TPPU Pre-Treatment Rescues Dendritic Spine Loss and Alleviates Depressive Behaviours during the Latent Period in the Lithium Chloride-Pilocarpine-Induced Status Epilepticus Rat Model
Abstract
:1. Introduction
2. Methods
2.1. Experimental Animals
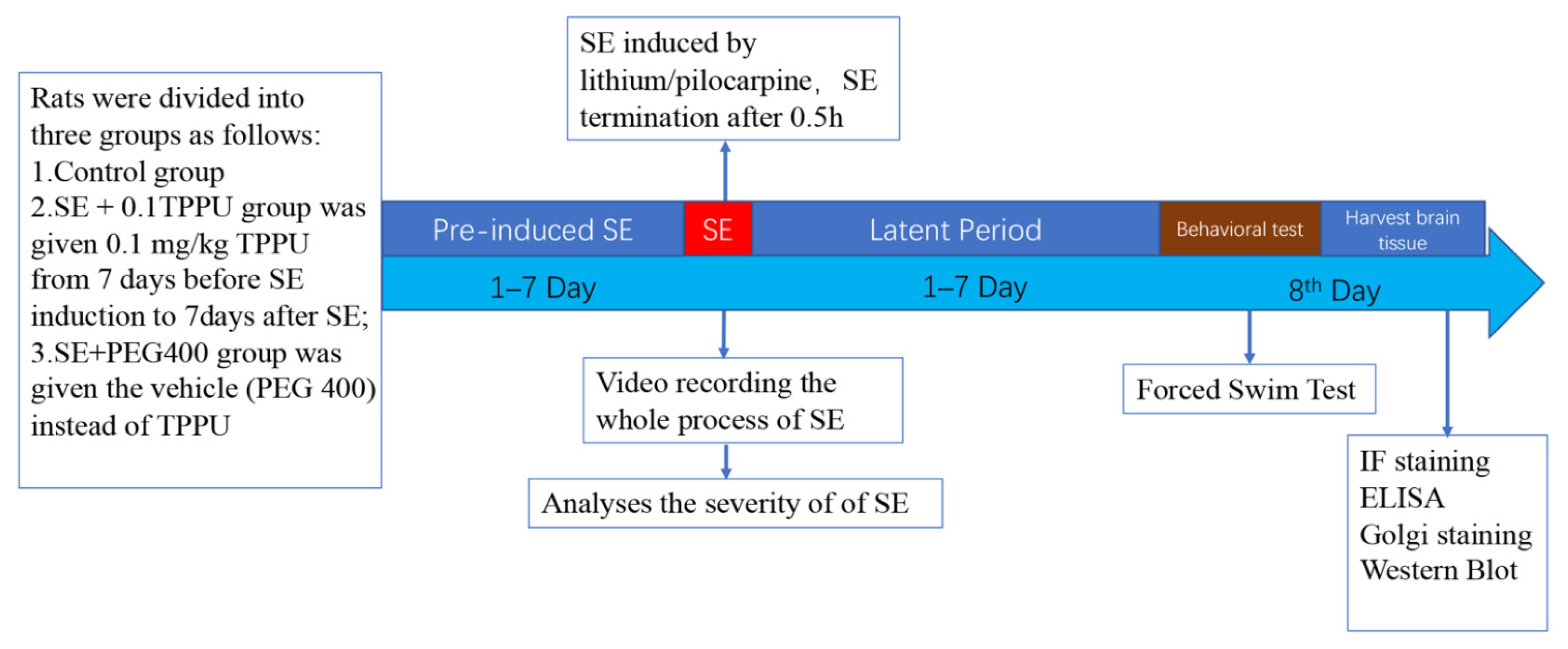
2.2. Establishment of the LiCl-Pilocarpine-Induced SE Rat Model and TPPU Treatment Paradigm
2.3. Forced Swim Test (FST)
2.4. Immunofluorescent Staining
2.5. Golgi Staining
2.6. Tissue Preparation and Protein Extraction
2.7. Enzyme-Linked Immunosorbent Assay (ELISA)
2.8. Western Blot Analysis
2.9. Statistical Analysis
3. Results
3.1. Pre-Treatment with TPPU Decreased the Seizure Severity during SE
3.2. Pre-Treatment with TPPU Attenuated the Depressive Behaviour of Despair 7 Days after SE Induction
3.3. Pre-Treatment with TPPU Alleviated Inflammations in the Hippocampus of LiCl-Pilocarpine-Induced SE Rat Model
3.4. Pre-Treatment with TPPU Selectively Rescued Neuronal and Dendritic Spine Loss in the Hippocampus of LiCl-Pilocarpine-Induced SE Rat Model
3.5. TPPU Pre-Treatment Activated the NR1/ERK/CREB Pathway in the Hippocampus of LiCl-Pilocarpine-Induced SE Rat Model
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Spencer, D.D.; Gerrard, J.; Zaveri, H.P. The roles of surgery and technology in understanding focal epilepsy and its comorbidities. Lancet Neurol. 2018, 17, 373–382. [Google Scholar] [CrossRef]
- Chen, Z.; Brodie, M.J.; Liew, D.; Kwan, P. Treatment Outcomes in Patients With Newly Diagnosed Epilepsy Treated With Established and New Antiepileptic Drugs: A 30-Year Longitudinal Cohort Study. JAMA Neurol. 2018, 75, 279–286. [Google Scholar] [CrossRef]
- Rayner, G. The Contribution of Cognitive Networks to Depression in Epilepsy. Epilepsy Curr. 2017, 17, 78–83. [Google Scholar] [CrossRef] [Green Version]
- Leistedt, S.J.; Linkowski, P. Brain, networks, depression, and more. Eur. Neuropsychopharmacol. 2013, 23, 55–62. [Google Scholar] [CrossRef] [PubMed]
- Chioza, B.; Osei-Lah, A.; Wilkie, H.; Nashef, L.; McCormick, D.; Asherson, P.; Makoff, A. Suggestive evidence for association of two potassium channel genes with different idiopathic generalised epilepsy syndromes. Epilepsy Res. 2002, 52, 107–116. [Google Scholar] [CrossRef]
- McNamara, J.O.; Huang, Y.Z.; Leonard, A.S. Molecular Signaling Mechanisms Underlying Epileptogenesis. Sci. STKE 2006, 2006, re12. [Google Scholar] [CrossRef]
- Vezzani, A.; Friedman, A.; Dingledine, R. The role of inflammation in epileptogenesis. Neuropharmacology 2013, 69, 16–24. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Barker-Haliski, M.L.; Löscher, W.; White, H.S.; Galanopoulou, A.S. Neuroinflammation in epileptogenesis: Insights and translational perspectives from new models of epilepsy. Epilepsia 2017, 58 (Suppl. S3), 39–47. [Google Scholar] [CrossRef] [Green Version]
- Tchekalarova, J.; Atanasova, D.; Kortenska, L.; Atanasova, M.; Lazarov, N. Chronic agomelatine treatment prevents comorbid depression in the post-status epilepticus model of acquired epilepsy through suppression of inflammatory signaling. Neurobiol. Dis. 2018, 115, 127–144. [Google Scholar] [CrossRef] [PubMed]
- Ren, Q.; Ma, M.; Ishima, T.; Morisseau, C.; Yang, J.; Wagner, K.M.; Zhang, J.-C.; Yang, C.; Yao, W.; Dong, C.; et al. Gene deficiency and pharmacological inhibition of soluble epoxide hydrolase confers resilience to repeated social defeat stress. Proc. Natl. Acad. Sci. USA 2016, 113, E1944–E1952. [Google Scholar] [CrossRef] [Green Version]
- Hammock, B.D.; McReynolds, C.B.; Wagner, K.; Buckpitt, A.; Cortes-Puch, I.; Croston, G.; Lee, K.S.S.; Yang, J.; Schmidt, W.K.; Hwang, S.H. Movement to the Clinic of Soluble Epoxide Hydrolase Inhibitor EC5026 as an Analgesic for Neuropathic Pain and for Use as a Nonaddictive Opioid Alternative. J. Med. Chem. 2021, 64, 1856–1872. [Google Scholar] [CrossRef]
- Iliff, J.J.; Jia, J.; Nelson, J.; Goyagi, T.; Klaus, J.; Alkayed, N.J. Epoxyeicosanoid signaling in CNS function and disease. Prostaglandins Other Lipid Mediat. 2010, 91, 68–84. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shen, Y.; Peng, W.; Chen, Q.; Hammock, B.D.; Liu, J.; Li, D.; Yang, J.; Ding, J.; Wang, X. Anti-inflammatory treatment with a soluble epoxide hydrolase inhibitor attenuates seizures and epilepsy-associated depression in the LiCl-pilocarpine post-status epilepticus rat model. Brain Behav. Immun. 2019, 81, 535–544. [Google Scholar] [CrossRef] [PubMed]
- Kurz, J.E.; Moore, B.J.; Henderson, S.C.; Campbell, J.N.; Churn, S.B. A cellular mechanism for dendritic spine loss in the pilocarpine model of status epilepticus. Epilepsia 2008, 49, 1696–1710. [Google Scholar] [CrossRef] [Green Version]
- Ma, Y.; Ramachandran, A.; Ford, N.; Parada, I.; Prince, D.A. Remodeling of dendrites and spines in the C1q knockout model of genetic epilepsy. Epilepsia 2013, 54, 1232–1239. [Google Scholar] [CrossRef] [Green Version]
- Bourne, J.N.; Harris, K.M. Balancing Structure and Function at Hippocampal Dendritic Spines. Annu. Rev. Neurosci. 2008, 31, 47–67. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hamilton, A.M.; Oh, W.C.; Vega-Ramirez, H.; Stein, I.S.; Hell, J.W.; Patrick, G.N.; Zito, K. Activity-Dependent Growth of New Dendritic Spines Is Regulated by the Proteasome. Neuron 2012, 74, 1023–1030. [Google Scholar] [CrossRef] [Green Version]
- Noguchi, J.; Matsuzaki, M.; Ellis-Davies, G.C.; Kasai, H. Spine-Neck Geometry Determines NMDA Receptor-Dependent Ca2+ Signaling in Dendrites. Neuron 2005, 46, 609–622. [Google Scholar] [CrossRef] [Green Version]
- Franchini, L.; Carrano, N.; Di Luca, M.; Gardoni, F. Synaptic GluN2A-Containing NMDA Receptors: From Physiology to Pathological Synaptic Plasticity. Int. J. Mol. Sci. 2020, 21, 1538. [Google Scholar] [CrossRef] [Green Version]
- Zhai, S.; Ark, E.D.; Parra-Bueno, P.; Yasuda, R. Long-Distance Integration of Nuclear ERK Signaling Triggered by Activation of a Few Dendritic Spines. Science 2013, 342, 1107–1111. [Google Scholar] [CrossRef] [Green Version]
- Hardingham, G.E.; Bading, H. Synaptic versus extrasynaptic NMDA receptor signalling: Implications for neurodegenerative disorders. Nat. Rev. Neurosci. 2010, 11, 682–696. [Google Scholar] [CrossRef] [Green Version]
- Moda-Sava, R.N.; Murdock, M.H.; Parekh, P.K.; Fetcho, R.N.; Huang, B.S.; Huynh, T.N.; Witztum, J.; Shaver, D.C.; Rosenthal, D.L.; Alway, E.J.; et al. Sustained rescue of prefrontal circuit dysfunction by antidepressant-induced spine formation. Science 2019, 364, 6436. [Google Scholar] [CrossRef]
- Peng, W.-F.; Ding, J.; Li, X.; Fan, F.; Zhang, Q.-Q.; Wang, X. N-methyl-d-aspartate receptor NR2B subunit involved in depression-like behaviours in lithium chloride-pilocarpine chronic rat epilepsy model. Epilepsy Res. 2016, 119, 77–85. [Google Scholar] [CrossRef]
- Racine, R.J. Modification of seizure activity by electrical stimulation: II. Motor seizure. Electroencephalogr. Clin. Neurophysiol. 1972, 32, 281–294. [Google Scholar] [CrossRef]
- Detke, M.J.; Rickels, M.; Lucki, I. Active behaviors in the rat forced swimming test differentially produced by serotonergic and noradrenergic antidepressants. Psychopharmacology 1995, 121, 66–72. [Google Scholar] [CrossRef] [PubMed]
- Asadi-Pooya, A.A.; Stewart, G.R.; Abrams, D.J.; Sharan, A. Prevalence and Incidence of Drug-Resistant Mesial Temporal Lobe Epilepsy in the United States. World Neurosurg. 2017, 99, 662–666. [Google Scholar] [CrossRef]
- Becker, A.J. Review: Animal models of acquired epilepsy: Insights into mechanisms of human epileptogenesis. Neuropathol. Appl. Neurobiol. 2018, 44, 112–129. [Google Scholar] [CrossRef] [PubMed]
- Goffin, K.; Nissinen, J.; Van Laere, K.; Pitkänen, A. Cyclicity of spontaneous recurrent seizures in pilocarpine model of temporal lobe epilepsy in rat. Exp. Neurol. 2007, 205, 501–505. [Google Scholar] [CrossRef] [PubMed]
- Jackson-Tarlton, C.S.; Whatley, B.P.; Kasheke, G.D.; Pohlmann-Eden, B.; Omisade, A. A prospective pilot study of cognitive impairment and mood in adults with first seizure, new-onset epilepsy, and newly diagnosed epilepsy at time of initial seizure presentation. Epilepsy Behav. 2020, 112, 107359. [Google Scholar] [CrossRef]
- Josephson, C.B.; Lowerison, M.; Vallerand, I.; Sajobi, T.T.; Patten, S.; Jette, N.; Wiebe, S. Association of Depression and Treated Depression with Epilepsy and Seizure Outcomes: A Multicohort Analysis. JAMA Neurol. 2017, 74, 533–539. [Google Scholar] [CrossRef]
- Gilliam, F.G.; Black, K.J.; Carter, J.; Freedland, K.E.; Sheline, Y.I.; Tsai, W.; Lustman, P.J. A Trial of Sertraline or Cognitive Behavior Therapy for Depression in Epilepsy. Ann. Neurol. 2019, 86, 552–560. [Google Scholar] [CrossRef] [PubMed]
- Tang, F.R.; Loke, W.K. Cyto-, axo- and dendro-architectonic changes of neurons in the limbic system in the mouse pilocarpine model of temporal lobe epilepsy. Epilepsy Res. 2010, 89, 43–51. [Google Scholar] [CrossRef]
- Isokawa, M. Remodeling Dendritic Spines of Dentate Granule Cells in Temporal Lobe Epilepsy Patients and the Rat Pilocarpine Model. Epilepsia 2000, 41, S14–S17. [Google Scholar] [CrossRef] [Green Version]
- Maysinger, D.; Gröger, D.; Lake, A.; Licha, K.; Weinhart, M.; Chang, P.K.-Y.; Mulvey, R.; Haag, R.; McKinney, R.A. Dendritic Polyglycerol Sulfate Inhibits Microglial Activation and Reduces Hippocampal CA1 Dendritic Spine Morphology Deficits. Biomacromolecules 2015, 16, 3073–3082. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Garré, J.M.; Silva, H.M.; Lafaille, J.J.; Yang, G. CX3CR1+ monocytes modulate learning and learning-dependent dendritic spine remodeling via TNF-α. Nat. Med. 2017, 23, 714–722. [Google Scholar] [CrossRef] [Green Version]
- Prada, I.; Gabrielli, M.; Turola, E.; Iorio, A.; D’Arrigo, G.; Parolisi, R.; De Luca, M.; Pacifici, M.; Bastoni, M.; Lombardi, M.; et al. Glia-to-neuron transfer of miRNAs via extracellular vesicles: A new mechanism underlying inflammation-induced synaptic alterations. Acta Neuropathol. 2018, 135, 529–550. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hu, J.; Dziumbla, S.; Lin, J.; Bibli, S.-I.; Zukunft, S.; De Mos, J.; Awwad, K.; Frömel, T.; Jungmann, A.; Devraj, K.; et al. Inhibition of soluble epoxide hydrolase prevents diabetic retinopathy. Nature 2017, 552, 248–252. [Google Scholar] [CrossRef]
- Swardfager, W.; Hennebelle, M.; Yu, D.; Hammock, B.; Levitt, A.; Hashimoto, K.; Taha, A. Metabolic/inflammatory/vascular comorbidity in psychiatric disorders; soluble epoxide hydrolase (sEH) as a possible new target. Neurosci. Biobehav. Rev. 2018, 87, 56–66. [Google Scholar] [CrossRef] [Green Version]
- Wang, L.; Luo, G.; Zhang, L.-F.; Geng, H.-X. Neuroprotective effects of epoxyeicosatrienoic acids. Prostaglandins Other Lipid Mediat. 2018, 138, 9–14. [Google Scholar] [CrossRef]
- Shen, H.C.; Hammock, B.D. Discovery of Inhibitors of Soluble Epoxide Hydrolase: A Target with Multiple Potential Therapeutic Indications. J. Med. Chem. 2011, 55, 1789–1808. [Google Scholar] [CrossRef] [Green Version]
- Inceoglu, B.; Zolkowska, D.; Yoo, H.J.; Wagner, K.M.; Yang, J.; Hackett, E.; Hwang, S.H.; Lee, K.S.S.; Rogawski, M.; Morisseau, C.; et al. Epoxy Fatty Acids and Inhibition of the Soluble Epoxide Hydrolase Selectively Modulate GABA Mediated Neurotransmission to Delay Onset of Seizures. PLoS ONE 2013, 8, e80922. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mule, N.; Leon, A.C.O.; Falck, J.R.; Arand, M.; Marowsky, A. 11,12-Epoxyeicosatrienoic acid (11,12 EET) reduces excitability and excitatory transmission in the hippocampus. Neuropharmacology 2017, 123, 310–321. [Google Scholar] [CrossRef] [PubMed]
- Noe, F.M.; Polascheck, N.; Frigerio, F.; Bankstahl, M.; Ravizza, T.; Marchini, S.; Beltrame, L.; Banderó, C.R.; Löscher, W.; Vezzani, A. Pharmacological blockade of IL-1beta/IL-1 receptor type 1 axis during epileptogenesis provides neuroprotection in two rat models of temporal lobe epilepsy. Neurobiol. Dis. 2013, 59, 183–193. [Google Scholar] [CrossRef] [PubMed]
- Paoletti, P.; Bellone, C.; Zhou, Q. NMDA receptor subunit diversity: Impact on receptor properties, synaptic plasticity and disease. Nat. Rev. Neurosci. 2013, 14, 383–400. [Google Scholar] [CrossRef] [PubMed]







Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Peng, W.; Shen, Y.; Wang, Q.; Ding, J.; Wang, X. TPPU Pre-Treatment Rescues Dendritic Spine Loss and Alleviates Depressive Behaviours during the Latent Period in the Lithium Chloride-Pilocarpine-Induced Status Epilepticus Rat Model. Brain Sci. 2021, 11, 1465. https://doi.org/10.3390/brainsci11111465
Peng W, Shen Y, Wang Q, Ding J, Wang X. TPPU Pre-Treatment Rescues Dendritic Spine Loss and Alleviates Depressive Behaviours during the Latent Period in the Lithium Chloride-Pilocarpine-Induced Status Epilepticus Rat Model. Brain Sciences. 2021; 11(11):1465. https://doi.org/10.3390/brainsci11111465
Chicago/Turabian StylePeng, Weifeng, Yijun Shen, Qiang Wang, Jing Ding, and Xin Wang. 2021. "TPPU Pre-Treatment Rescues Dendritic Spine Loss and Alleviates Depressive Behaviours during the Latent Period in the Lithium Chloride-Pilocarpine-Induced Status Epilepticus Rat Model" Brain Sciences 11, no. 11: 1465. https://doi.org/10.3390/brainsci11111465
APA StylePeng, W., Shen, Y., Wang, Q., Ding, J., & Wang, X. (2021). TPPU Pre-Treatment Rescues Dendritic Spine Loss and Alleviates Depressive Behaviours during the Latent Period in the Lithium Chloride-Pilocarpine-Induced Status Epilepticus Rat Model. Brain Sciences, 11(11), 1465. https://doi.org/10.3390/brainsci11111465




