Simultaneous Normalization and Compensatory Changes in Right Hemisphere Connectivity during Aphasia Therapy
Abstract
1. Introduction
- In the phonological task, we mainly expect left intra-hemispheric connectivity in the HC group, as phonological processing is more left lateralized in healthy adults [32,34]. Based on this assumption, we predict that in the phonological task, treatment-related changes among PWA in the left hemisphere will reflect normalization (i.e., will increase resemblance to HC), whereas changes in the RH will reflect compensation (i.e., will differ from HC connectivity);
- In HC, the semantic task is expected to involve the connections with bilateral ventral IFG, whereas the phonological task is expected to involve the connections with left dorsal IFG, since such functional subdivision of the IFG is evident in healthy individuals [41,42]. Accordingly, among PWA, treatment-related changes are expected to follow this pattern bilaterally, as contralateral right homologous regions were found to be involved in language processing following left hemisphere stroke [13,24,82,83].
2. Materials and Methods
2.1. Participants
2.2. Anomia Treatment
2.3. Behavioral Results
2.4. fMRI Task
2.5. fMRI Data Acquisition
2.6. Image Analysis
2.7. VOI Extraction
2.8. Effective Connectivity Analysis
2.9. DCM Specification and Estimation
2.10. Second- and Third-Level PEB of the RH MODEL
2.10.1. PWA Compared to Averaged Connectivity of HC
- To explore the average connectivity across the two time-points in each task in HC, as well as treatment-related connectivity changes in PWA, individual second-level PEBs were created, with one covariate for the mean across time-points and one for the difference between time-points. These second-level PEBs were taken to a third-level PEB-of-PEBs with a single covariate representing the mean across the group. The third-level analysis was done separately for PWA, where it was used to determine the change between S1 and S2, and for HC, where it was used to determine the average of S1 and S2.
- To explore connectivity in pre- and post-treatment separately in PWA, another individual second-level PEB was created, with a separate mean-centered covariate for each time-point (S1 and S2). These were taken to a third-level PEB-of-PEBs with a single covariate representing the mean across the group.
2.10.2. PWA Compared to Constant Connectivity of HC
3. Results
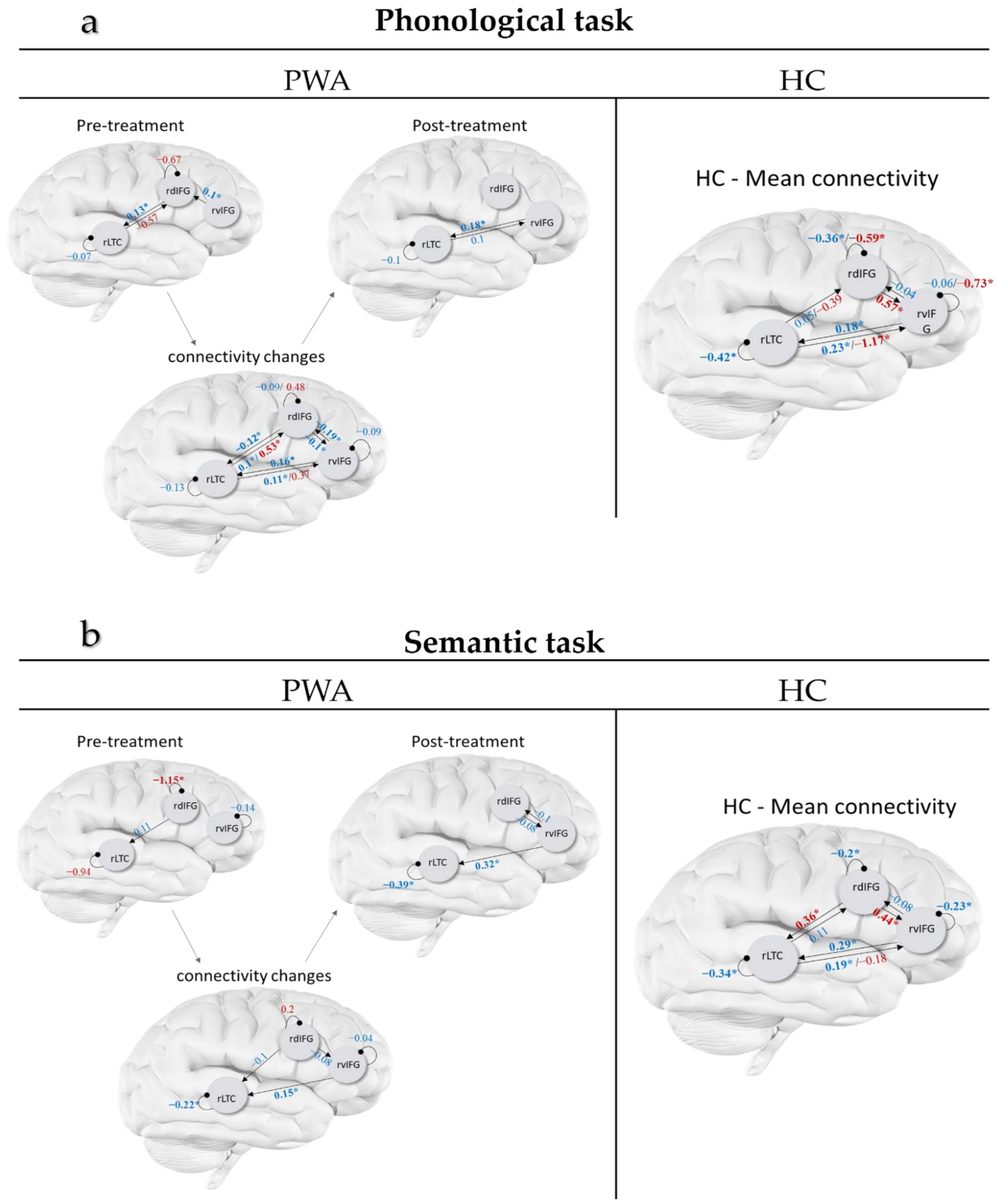
3.1. Connectivity of HC
3.2. Connectivity of PWA
4. Discussion
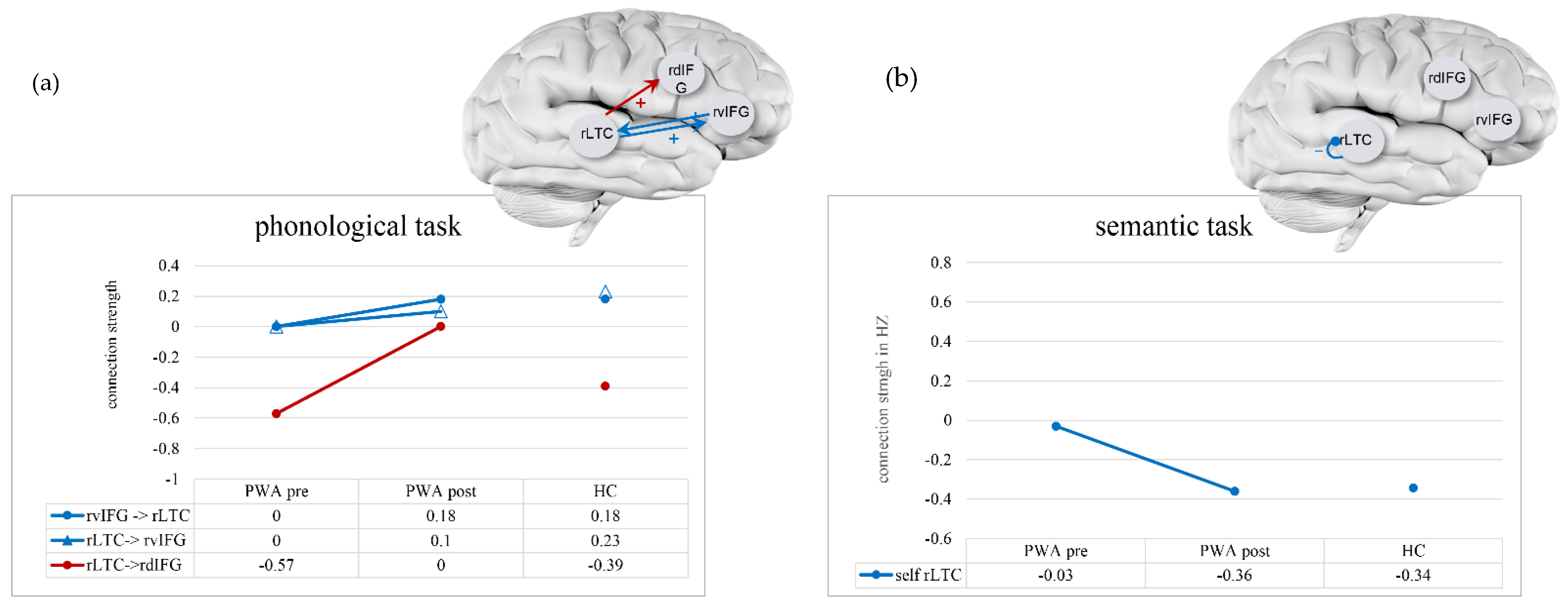
4.1. Connectivity of HC Participants
4.2. Connectivity of PWA
4.3. Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kertesz, A.; Harlock, W.; Coates, R. Computer tomographic localization, lesion size, and prognosis in aphasia and nonverbal impairment. Brain Lang. 1979, 8, 34–50. [Google Scholar] [CrossRef]
- Desai, R.H.; Riccardi, N. Cognitive Neuroscience of Language. In The Routledge Handbook of Cognitive Linguistics; Routledge: Oxfordshire, UK, 2021; pp. 615–642. [Google Scholar]
- Kiran, S.; Thompson, C.K. Neuroplasticity of Language Networks in Aphasia: Advances, Updates, and Future Challenges. Front. Neurol. 2019, 10, 295. [Google Scholar] [CrossRef] [PubMed]
- Wilson, S.M.; Eriksson, D.K.; Yen, M.; Demarco, A.T.; Schneck, S.; Lucanie, J.M. Language Mapping in Aphasia. J. Speech Lang. Hear. Res. 2019, 62, 3937–3946. [Google Scholar] [CrossRef] [PubMed]
- Fridriksson, J.; Richardson, J.D.; Fillmore, P.; Cai, B. Left hemisphere plasticity and aphasia recovery. NeuroImage 2012, 60, 854–863. [Google Scholar] [CrossRef] [PubMed]
- Hartwigsen, G.; Saur, D. Neuroimaging of stroke recovery from aphasia—Insights into plasticity of the human language network. NeuroImage 2019, 190, 14–31. [Google Scholar] [CrossRef]
- Hamilton, R.H.; Chrysikou, E.G.; Coslett, B. Mechanisms of aphasia recovery after stroke and the role of noninvasive brain stimulation. Brain Lang. 2011, 118, 40–50. [Google Scholar] [CrossRef]
- Turkeltaub, P.E.; Coslett, H.B.; Thomas, A.L.; Faseyitan, O.; Benson, J.; Norise, C.; Hamilton, R.H. The right hemisphere is not unitary in its role in aphasia recovery. Cortex 2012, 48, 1179–1186. [Google Scholar] [CrossRef]
- Fridriksson, J. Preservation and Modulation of Specific Left Hemisphere Regions is Vital for Treated Recovery from Anomia in Stroke. J. Neurosci. 2010, 30, 11558–11564. [Google Scholar] [CrossRef]
- Rochon, E.; Leonard, C.; Burianova, H.; Laird, L.; Sörös, P.; Graham, S.; Grady, C. Neural changes after phonological treatment for anomia: An fMRI study. Brain Lang. 2010, 114, 164–179. [Google Scholar] [CrossRef]
- Van Hees, S.; Mcmahon, K.; Angwin, A.; de Zubicaray, G.; Copland, D.A. Neural activity associated with semantic versus phonological anomia treatments in aphasia. Brain Lang. 2014, 129, 47–57. [Google Scholar] [CrossRef]
- Thiel, A.; Herholz, K.; Koyuncu, A.; Ghaemi, M.; Kracht, L.W.; Habedank, B.; Heiss, W.-D. Plasticity of language networks in patients with brain tumors: A positron emission tomography activation study. Ann. Neurol. 2001, 50, 620–629. [Google Scholar] [CrossRef] [PubMed]
- Warburton, E.; Price, C.; Swinburn, K.; Wise, R.J.S. Mechanisms of recovery from aphasia: Evidence from positron emission tomography studies. J. Neurol. Neurosurg. Psychiatry 1999, 66, 155–161. [Google Scholar] [CrossRef]
- Heiss, W.-D.; Thiel, A. A proposed regional hierarchy in recovery of post-stroke aphasia. Brain Lang. 2006, 98, 118–123. [Google Scholar] [CrossRef]
- Naeser, M.A.; Martin, P.I.; Nicholas, M.; Baker, E.H.; Seekins, H.; Kobayashi, M.; Theoret, H.; Fregni, F.; Maria-Tormos, J.; Kurland, J. Improved picture naming in chronic aphasia after TMS to part of right Broca’s area: An open-protocol study. Brain Lang. 2005, 93, 95–105. [Google Scholar] [CrossRef] [PubMed]
- Naeser, M.A.; Martin, P.I.; Nicholas, M.; Baker, E.H.; Seekins, H.; Helm-Estabrooks, N.; Cayer-Meade, C.; Kobayashi, M.; Theoret, H.; Fregni, F.; et al. Improved naming after TMS treatments in a chronic, global aphasia patient—Case report. Neurocase 2005, 11, 182–193. [Google Scholar] [CrossRef] [PubMed]
- Barwood, C.H.S.; Murdoch, B.E.; Whelan, B.-M.; Lloyd, D.; Riek, S.; Sullivan, J.D.O.; Coulthard, A.; Wong, A. Improved language performance subsequent to low-frequency rTMS in patients with chronic non-fluent aphasia post-stroke. Eur. J. Neurol. 2010, 18, 935–943. [Google Scholar] [CrossRef] [PubMed]
- Hamilton, R.H.; Sanders, L.; Benson, J.; Faseyitan, O.; Norise, C.; Naeser, M.; Martin, P.; Coslett, H.B. Stimulating conversation: Enhancement of elicited propositional speech in a patient with chronic non-fluent aphasia following transcranial magnetic stimulation. Brain Lang. 2010, 113, 45–50. [Google Scholar] [CrossRef] [PubMed]
- Abel, S.; Weiller, C.; Huber, W.; Willmes, K.; Specht, K. Therapy-induced brain reorganization patterns in aphasia. Brain 2015, 138, 1097–1112. [Google Scholar] [CrossRef]
- Meltzer, J.A.; Wagage, S.; Ryder, J.; Solomon, B.; Braun, A.R. Adaptive significance of right hemisphere activation in aphasic language comprehension. Neuropsychology 2013, 51, 1248–1259. [Google Scholar] [CrossRef]
- Bitan, T.; Simic, T.; Saverino, C.; Jones, C.; Glazer, J.; Collela, B.; Wiseman-Hakes, C.; Green, R.; Rochon, E. Changes in Resting-State Connectivity following Melody-Based Therapy in a Patient with Aphasia. Neural Plast. 2018, 2018, 1–13. [Google Scholar] [CrossRef]
- Abo, M.; Senoo, A.; Watanabe, S.; Miyano, S.; Doseki, K.; Sasaki, N.; Kobayashi, K.; Kikuchi, Y.; Yonemoto, K. Language-related brain function during word repetition in post-stroke aphasics. NeuroReport 2004, 15, 1891–1894. [Google Scholar] [CrossRef] [PubMed]
- Basso, A.; Gardelli, M.; Grassi, M.P.; Mariotti, M. The Role of the Right Hemisphere in Recovery from Aphasia. Two Case Studies. Cortex 1989, 25, 555–566. [Google Scholar] [CrossRef]
- Ohyama, M.; Senda, M.; Kitamura, S.; Ishii, K.; Mishina, M.; Terashi, A. Role of the Nondominant Hemisphere and Undamaged Area During Word Repetition in Poststroke Aphasics: A PET Activation Study. Stroke 1996, 27, 897–903. [Google Scholar] [CrossRef]
- Vitali, P.; Abutalebi, J.; Tettamanti, M.; Danna, M.; Ansaldo, A.-I.; Perani, D.; Joanette, Y.; Cappa, S. Training-Induced Brain Remapping in Chronic Aphasia: A Pilot Study. Neurorehabilit. Neural Repair 2007, 21, 152–160. [Google Scholar] [CrossRef]
- Kiran, S.; Meier, E.L.; Johnson, J.P. Neuroplasticity in Aphasia: A Proposed Framework of Language Recovery. J. Speech Lang. Hear. Res. 2019, 62, 3973–3985. [Google Scholar] [CrossRef]
- Saur, D.; Lange, R.; Baumgaertner, A.; Schraknepper, V.; Willmes, K.; Rijntjes, M.; Weiller, C. Dynamics of language reorganization after stroke. Brain 2006, 129, 1371–1384. [Google Scholar] [CrossRef]
- Heiss, W.-D.; Thiel, A.; Kessler, J.; Herholz, K. Disturbance and recovery of language function: Correlates in PET activation studies. NeuroImage 2003, 20, S42–S49. [Google Scholar] [CrossRef] [PubMed]
- Price, C.J.; Crinion, J. The latest on functional imaging studies of aphasic stroke. Curr. Opin. Neurol. 2005, 18, 429–434. [Google Scholar] [CrossRef] [PubMed]
- Regev, M.; Honey, C.; Simony, E.; Hasson, U. Selective and Invariant Neural Responses to Spoken and Written Narratives. J. Neurosci. 2013, 33, 15978–15988. [Google Scholar] [CrossRef] [PubMed]
- Deniz, F.; Nunez-Elizalde, A.O.; Huth, A.G.; Gallant, J.L. The Representation of Semantic Information Across Human Cerebral Cortex During Listening Versus Reading Is Invariant to Stimulus Modality. J. Neurosci. 2019, 39, 7722–7736. [Google Scholar] [CrossRef] [PubMed]
- Binder, J.R.; Desai, R.H.; Graves, W.W.; Conant, L.L. Where is the semantic system? A critical review and meta-analysis of 120 functional neuroimaging studies. Cereb. Cortex 2009, 19, 2767–2796. [Google Scholar] [CrossRef] [PubMed]
- Vigneau, M.; Beaucousin, V.; Hervé, P.-Y.; Jobard, G.; Petit, L.; Crivello, F.; Mellet, E.; Zago, L.; Mazoyer, B.; Tzourio-Mazoyer, N. What is right-hemisphere contribution to phonological, lexico-semantic, and sentence processing? Insights from a meta-analysis. NeuroImage 2011, 54, 577–593. [Google Scholar] [CrossRef] [PubMed]
- Spironelli, C.; Angrilli, A. Language lateralization in phonological, semantic and orthographic tasks: A slow evoked potential study. Behav. Brain Res. 2006, 175, 296–304. [Google Scholar] [CrossRef]
- Marslen-Wilson, W.D.; Tyler, L. Morphology, language and the brain: The decompositional substrate for language comprehension. Philos. Trans. R. Soc. B Biol. Sci. 2007, 362, 823–836. [Google Scholar] [CrossRef] [PubMed]
- Hickok, G.; Poeppel, D. Towards a functional neuroanatomy of speech perception. Trends Cogn. Sci. 2000, 4, 131–138. [Google Scholar] [CrossRef]
- Hickok, G.; Poeppel, D. Dorsal and ventral streams: A framework for understanding aspects of the functional anatomy of language. Cognition 2004, 92, 67–99. [Google Scholar] [CrossRef]
- Hickok, G.; Poeppel, D. The cortical organization of speech processing. Nat. Rev. Neurosci. 2007, 8, 393–402. [Google Scholar] [CrossRef]
- Hickok, G.; Poeppel, D. Neural Basis of Speech Perception. Neurobiol. Lang. 2016, 299–310. [Google Scholar] [CrossRef]
- Matchin, W.; Hickok, G. The Cortical Organization of Syntax. Cereb. Cortex 2020, 30, 1481–1498. [Google Scholar] [CrossRef]
- Gough, P.M.; Nobre, A.C.; Devlin, J. Dissociating Linguistic Processes in the Left Inferior Frontal Cortex with Transcranial Magnetic Stimulation. J. Neurosci. 2005, 25, 8010–8016. [Google Scholar] [CrossRef]
- Heim, S.; Eickhoff, S.B.; Ischebeck, A.; Friederici, A.D.; Stephan, K.E.; Amunts, K. Effective connectivity of the left BA 44, BA 45, and inferior temporal gyrus during lexical and phonological decisions identified with DCM. Hum. Brain Mapp. 2009, 30, 392–402. [Google Scholar] [CrossRef] [PubMed]
- Bassi, M.S.; Iezzi, E.; Gilio, L.; Centonze, D.; Buttari, F. Synaptic Plasticity Shapes Brain Connectivity: Implications for Network Topology. Int. J. Mol. Sci. 2019, 20, 6193. [Google Scholar] [CrossRef]
- Schevenels, K.; Price, C.J.; Zink, I.; De Smedt, B.; Vandermosten, M. A Review on Treatment-Related Brain Changes in Aphasia. Neurobiol. Lang. 2020, 1, 402–433. [Google Scholar] [CrossRef]
- Nair, V.A.; Young, B.M.; La, C.; Reiter, P.; Nadkarni, T.N.; Song, J.; Vergun, S.; Addepally, N.S.; Mylavarapu, K.; Swartz, J.L.; et al. Functional connectivity changes in the language network during stroke recovery. Ann. Clin. Transl. Neurol. 2015, 2, 185–195. [Google Scholar] [CrossRef]
- Klingbeil, J.; Wawrzyniak, M.; Stockert, A.; Saur, D. Resting-state functional connectivity: An emerging method for the study of language networks in post-stroke aphasia. Brain Cogn. 2019, 131, 22–33. [Google Scholar] [CrossRef]
- Yang, H.; Bai, L.; Zhou, Y.; Kang, S.; Liang, P.; Wang, L.; Zhu, Y. Increased inter-hemispheric resting-state functional connectivity in acute lacunar stroke patients with aphasia. Exp. Brain Res. 2017, 235, 941–948. [Google Scholar] [CrossRef] [PubMed]
- Sandberg, C.W. Hypoconnectivity of Resting-State Networks in Persons with Aphasia Compared with Healthy Age-Matched Adults. Front. Hum. Neurosci. 2017, 11, 91. [Google Scholar] [CrossRef] [PubMed]
- Van Hees, S.; McMahon, K.; Angwin, A.; de Zubicaray, G.; Read, S.; Copland, D.A. A functional MRI study of the relationship between naming treatment outcomes and resting state functional connectivity in post-stroke aphasia. Hum. Brain Mapp. 2014, 35, 3919–3931. [Google Scholar] [CrossRef] [PubMed]
- Duncan, E.S.; Small, S. Changes in dynamic resting state network connectivity following aphasia therapy. Brain Imaging Behav. 2018, 12, 1141–1149. [Google Scholar] [CrossRef]
- Bitan, T.; Booth, J.R.; Choy, J.; Burman, D.D.; Gitelman, D.R.; Mesulam, M.M. Behavioral/Systems/Cognitive Shifts of Effective Connectivity within a Language Network during Rhyming and Spelling. J. Neurosci. 2005, 25, 5397–5403. [Google Scholar] [CrossRef]
- Bitan, T.; Cheon, J.; Lu, D.; Burman, D.D.; Booth, J.R. Developmental Increase in Top–Down and Bottom–Up Processing in a Phonological Task: An Effective Connectivity, fMRI Study. J. Cogn. Neurosci. 2009, 21, 1135–1145. [Google Scholar] [CrossRef] [PubMed]
- Bitan, T.; Lifshitz, A.; Breznitz, Z.; Booth, J.R. Bidirectional Connectivity between Hemispheres Occurs at Multiple Levels in Language Processing but Depends on Sex. J. Neurosci. 2010, 30, 11576–11585. [Google Scholar] [CrossRef] [PubMed]
- Mechelli, A.; Price, C.J.; Noppeney, U.; Friston, K.J. A Dynamic Causal Modeling Study on Category Effects: Bottom–Up or Top–Down Mediation? J. Cogn. Neurosci. 2003, 15, 925–934. [Google Scholar] [CrossRef]
- Perrone-Bertolotti, M.; Kauffmann, L.; Pichat, C.; Vidal, J.R.; Baciu, M. Effective Connectivity between Ventral Occipito-Temporal and Ventral Inferior Frontal Cortex during Lexico-Semantic Processing. A Dynamic Causal Modeling Study. Front. Hum. Neurosci. 2017, 11, 325. [Google Scholar] [CrossRef]
- Seghier, M.L.; Price, C. Reading Aloud Boosts Connectivity through the Putamen. Cereb. Cortex 2009, 20, 570–582. [Google Scholar] [CrossRef]
- Seghier, M.L.; Josse, G.; Leff, A.P.; Price, C. Lateralization is Predicted by Reduced Coupling from the Left to Right Prefrontal Cortex during Semantic Decisions on Written Words. Cereb. Cortex 2010, 21, 1519–1531. [Google Scholar] [CrossRef] [PubMed]
- Sarasso, S.; Santhanam, P.; Määtta, S.; Poryazova, R.; Ferrarelli, F.; Tononi, G.; Small, S.; Poryiazova, R. Non-fluent aphasia and neural reorganization after speech therapy: Insights from human sleep electrophysiology and functional magnetic resonance imaging. Arch. Ital. Biol 2010, 148, 271–278. [Google Scholar]
- Iyer, K.K.; Angwin, A.J.; Van Hees, S.; McMahon, K.L.; Breakspear, M.; Copland, D.A. Alterations to dual stream connectivity predicts response to aphasia therapy following stroke. Cortex 2020, 125, 30–43. [Google Scholar] [CrossRef]
- Ekiran, S.; Meier, E.L.; Kapse, K.J.; Glynn, P.A. Changes in task-based effective connectivity in language networks following rehabilitation in post-stroke patients with aphasia. Front. Hum. Neurosci. 2015, 9, 316. [Google Scholar] [CrossRef]
- Meier, E.L.; Johnson, J.P.; Kiran, S. Behavioural Neurology Left frontotemporal effective connectivity during semantic feature judgments in patients with chronic aphasia and age-matched healthy controls. Cortex 2018, 108, 173–192. [Google Scholar] [CrossRef]
- Abutalebi, J.; Della Rosa, P.A.; Tettamanti, M.; Green, D.W.; Cappa, S. Bilingual aphasia and language control: A follow-up fMRI and intrinsic connectivity study. Brain Lang. 2009, 109, 141–156. [Google Scholar] [CrossRef]
- Chu, R.; Meltzer, J.A.; Bitan, T. Interhemispheric interactions during sentence comprehension in patients with aphasia. Cortex 2018, 109, 74–91. [Google Scholar] [CrossRef]
- Meier, E.L.; Kapse, K.J.; Ekiran, S. The Relationship between Frontotemporal Effective Connectivity during Picture Naming, Behavior, and Preserved Cortical Tissue in Chronic Aphasia. Front. Hum. Neurosci. 2016, 10, 109. [Google Scholar] [CrossRef] [PubMed]
- Leonard, C.; Rochon, E.; Laird, L. Treating naming impairments in aphasia: Findings from a phonological components analysis treatment. Aphasiology 2008, 22, 923–947. [Google Scholar] [CrossRef]
- Badre, D.; Poldrack, R.A.; Paré-Blagoev, E.J.; Insler, R.Z.; Wagner, A.D. Dissociable Controlled Retrieval and Generalized Selection Mechanisms in Ventrolateral Prefrontal Cortex. Neuron 2005, 47, 907–918. [Google Scholar] [CrossRef] [PubMed]
- Heim, S.; Opitz, B.; Müller, K.; Friederici, A. Phonological processing during language production: fMRI evidence for a shared production-comprehension network. Cogn. Brain Res. 2003, 16, 285–296. [Google Scholar] [CrossRef]
- Miceli, G.; Turriziani, P.; Caltagirone, C.; Capasso, R.; Tomaiuolo, F.; Caramazza, A. The Neural Correlates of Grammatical Gender: An fMRI Investigation. J. Cogn. Neurosci. 2002, 14, 618–628. [Google Scholar] [CrossRef]
- Zurowski, B.; Gostomzyk, J.; Grön, G.; Weller, R.; Schirrmeister, H.; Neumeier, B.; Spitzer, M.; Reske, S.N.; Walter, H. Dissociating a common working memory network from different neural substrates of phonological and spatial stimulus processing. Neuroimage 2002, 15, 45–57. [Google Scholar] [CrossRef] [PubMed]
- Doesborgh, S.J.; Van De Sandt-Koenderman, M.W.; Dippel, D.W.; Van Harskamp, F.; Koudstaal, P.J.; Visch-Brink, E.G. Ma Effects of Semantic Treatment on Verbal Communication and Linguistic Processing in Aphasia After Stroke. Stroke 2004, 35, 141–146. [Google Scholar] [CrossRef] [PubMed]
- Sadeghi, Z.; Baharloei, N.; Zadeh, A.M.; Ghasisin, L. Comparative Effectiveness of Semantic Feature Analysis (SFA) and Phonological Components Analysis (PCA) for Anomia Treatment in Persian Speaking Patients with Aphasia. Iran Rehabil. J. 2017, 15, 259–268. [Google Scholar] [CrossRef]
- Dell, G.S.; Schwartz, M.F.; Martin, N.; Saffran, E.M.; Gagnon, D.A. Lexical Access in Aphasic and Nonaphasic Speakers. Psychol. Rev. 1997, 104, 801–838. [Google Scholar] [CrossRef]
- Levelt, W.J.M.; Roelofs, A.; Meyer, A.S. A theory of lexical access in speech production. Behav. Brain Sci. 1999, 22, 1–38. [Google Scholar] [CrossRef]
- Howard, D. Cognitive neuropsychology and aphasia therapy: The case of word retrieval. In Acquired Neurogenic Communication Disorders A Clinical Perspective; Papathanasiou, I., Ed.; Whurr: London, UK, 2000; pp. 76–99. [Google Scholar]
- Hashimoto, N. The use of semantic- and phonological-based feature approaches to treat naming deficits in aphasia. Clin. Linguist. Phon. 2012, 26, 518–553. [Google Scholar] [CrossRef]
- Holland, R.; Johns, S.L.; Woollams, A.M. Neuropsychological Rehabilitation an International Journal The impact of phonological versus semantic repetition training on generalisation in chronic stroke aphasia reflects differences in dorsal pathway connectivity. Neuropsychol. Rehabil. 2016, 28, 548–567. [Google Scholar] [CrossRef]
- Howard, D.; Patterson, K.; Franklin, S.; Orchard-Lisle, V.; Morton, J. Treatment of word retrieval deficits in aphasia comparison of two therapy methods. Brain 1985, 108, 817–829. [Google Scholar]
- Kristinsson, S.; Basilakos, A.; Elm, J.; Spell, L.A.; Bonilha, L.; Rorden, C.; Ouden, D.B.D.; Cassarly, C.; Sen, S.; Hillis, A.; et al. Individualized response to semantic versus phonological aphasia therapies in stroke. Brain Commun. 2021, 3, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Lorenz, A.; Ziegler, W. Semantic vs. word-form specific techniques in anomia treatment: A multiple single-case study. J. Neurolinguistics 2009, 22, 515–537. [Google Scholar] [CrossRef]
- Wambaugh, J.; Cameron, R.; Kalinyak-Fliszar, M.; Nessler, C.; Wright, S. Retrieval of action names in aphasia: Effects of two cueing treatments. Aphasiology 2004, 18, 979–1004. [Google Scholar] [CrossRef]
- Leonard, C.; Laird, L.; Burianova, H.; Graham, S.; Grady, C.; Simic, T.; Rochon, E. Behavioural and neural changes after a “choice” therapy for naming deficits in aphasia: Preliminary findings. Aphasiology 2014, 29, 506–525. [Google Scholar] [CrossRef]
- Heiss, W.-D.; Kessler, J.; Thiel, A.; Ghaemi, M.; Karbe, H. Differential capacity of left and right hemispheric areas for compensation of poststroke aphasia. Ann. Neurol. 1999, 45, 430–438. [Google Scholar] [CrossRef]
- Weiller, C.; Isensee, C.; Rijntjes, M.; Huber, W.; Müller, S.; Bier, D.; Dutschka, K.; Woods, R.P.; Noth, J.; Diener, H.C. Recovery from wernicke’s aphasia: A positron emission tomographic study. Ann. Neurol. 1995, 37, 723–732. [Google Scholar] [CrossRef] [PubMed]
- Goodglass, H.; Kaplan, E.; Barresi, B. Boston Diagnostic Aphasia Examination (BDAE); Lippincott Williams & Wilkins: Baltimore, MD, USA, 2001; Available online: https://utdallas.primo.exlibrisgroup.com/discovery/fulldisplay/alma9927692213901421/01UT_DALLAS:UTDALMA (accessed on 5 April 2021).
- Busk, P.L.; Serlin, R.C. Meta-analysis for single-case research. In Single-Case Research Design and Analysis; Kratochwill, T.R., Levin, J.R., Eds.; Lawrence Erlbaum Associates Inc.: Hillsdale, NJ, USA, 1992; pp. 187–212. [Google Scholar]
- Howard, D.; Patterson, K. The Pyramids and Palm Trees Test: A Test of Semantic Access from Words and Pictures; Harcourt Assessment: San Antonio, TX, USA, 1992. [Google Scholar]
- Kay, J.; Coltheart, M.; Lesser, R. PALPA: Psycholinguistic Assessments of Language Processing in Aphasia. In Introduction, Auditory Processing, Reading & Spelling; Erlbaum: Mahwah, NJ, USA, 1992. [Google Scholar]
- Friston, K.J.; Harrison, L.; Penny, W. Dynamic causal modelling. Neuroimage 2003, 19, 1273–1302. [Google Scholar] [CrossRef]
- Zeidman, P.; Jafarian, A.; Corbin, N.; Seghier, M.L.; Razi, A.; Price, C.J.; Friston, K.J. A guide to group effective connectivity analysis, part 1: First level analysis with DCM for fMRI. NeuroImage 2019, 200, 174–190. [Google Scholar] [CrossRef] [PubMed]
- Zeidman, P.; Jafarian, A.; Seghier, M.L.; Litvak, V.; Cagnan, H.; Price, C.; Friston, K. A guide to group effective connectivity analysis, part 2: Second level analysis with PEB. NeuroImage 2019, 200, 12–25. [Google Scholar] [CrossRef]
- Sokolov, A.A.; Zeidman, P.; Erb, M.; Ryvlin, P.; Pavlova, M.A.; Friston, K.J. Linking structural and effective brain connectivity: Structurally informed Parametric Empirical Bayes (si-PEB). Brain Struct. Funct. 2019, 224, 205–217. [Google Scholar] [CrossRef]
- SPMcentral. spm12/spm_dcm_fmri_check.m at Master spm/spm12 GitHub. Available online: https://github.com/spm/spm12/blob/master/spm_dcm_fmri_check.m (accessed on 10 September 2021).
- Kan, I.P.; Thompson-Schill, S.L. Effect of name agreement on prefrontal activity during overt and covert picture naming. Cogn. Affect. Behav. Neurosci. 2004, 4, 43–57. [Google Scholar] [CrossRef] [PubMed]
- Levelt, W.J.M.; Schriefers, H.; Vorberg, D.; Havinga, J. The Time Course of Lexical Access in Speech Production: A Study of Picture Naming. Psychol. Rev. 1991, 98, 122–142. [Google Scholar] [CrossRef]
- Salmelin, R.; Hari, R.; Lounasmaa, O.V.; Sams, M. Dynamics of brain activation during picture naming. Nat. Cell Biol. 1994, 368, 463–465. [Google Scholar] [CrossRef]
- Kiyosawa, M.; Inoue, C.; Kawasaki, T.; Tokoro, T.; Ishii, K.; Ohyama, M.; Senda, M.; Soma, Y. Functional neuroanatomy of visual object naming: A PET study. Graefe’s Arch. Clin. Exp. Ophthalmol. 1996, 234, 110–115. [Google Scholar] [CrossRef]
- Price, C.J.; Devlin, J.T.; Moore, C.J.; Morton, C.; Laird, A. Meta-analyses of object naming: Effect of baseline. Hum. Brain Mapp. 2005, 25, 70–82. [Google Scholar] [CrossRef]
- Kang, W.; Azadikhah, A.; Mei, J. The Role of Right Hemisphere in Language Is Executive Rather than Linguistic. Neuropsychologia. 2020, 40, 1293–1299. [Google Scholar]
- Gertel, V.H.; Zhang, H.; Diaz, M.T. Stronger right hemisphere functional connectivity supports executive aspects of language in older adults. Brain Lang. 2020, 206, 104771. [Google Scholar] [CrossRef] [PubMed]
- Hartwigsen, G.; Neef, N.E.; Camilleri, J.; Margulies, D.; Eickhoff, S.B. Functional Segregation of the Right Inferior Frontal Gyrus: Evidence from Coactivation-Based Parcellation. Cereb. Cortex 2019, 29, 1532–1546. [Google Scholar] [CrossRef]
- Meier, E.L.; Johnson, J.P.; Pan, Y.; Kiran, S. A lesion and connectivity-based hierarchical model of chronic aphasia recovery dissociates patients and healthy controls. NeuroImage Clin. 2019, 23, 101919. [Google Scholar] [CrossRef]
- Wilmskoetter, J.; Fridriksson, J.; Basilakos, A.; Johnson, L.P.; Marebwa, B.; Rorden, C.; Warner, G.; Hickok, G.; Hillis, A.E.; Bonilha, L. Indirect White Matter Pathways Are Associated with Treated Naming Improvement in Aphasia. Neurorehabilit. Neural Repair 2021, 35, 346–355. [Google Scholar] [CrossRef] [PubMed]
| P2 a | P4 a | P5 (ATr1 a) | P6 (Atr2 a) | |
|---|---|---|---|---|
| Age (years) | 81 | 64 | 50 | 73 |
| Education (years) | 14 | 12 | 16 | 12 |
| Gender | M | M | F | M |
| Time post onset (years) | 1 | 1.75 | 3.5 | 4 |
| Lesion site | LMCA | Left frontotemporal | Left posterior frontal, temporal and parietal | Left frontotemporal |
| Aphasia type b | Broca’s | Broca’s | Broca’s | Mixed non-fluent |
| BNT [mean 94, normal range 78.3–100] c | 17 | 13 | 13 | 40 |
| Mean effect size across treated items | 3.94 | 1.53 | 3.00 | 3.47 |
| Phonological Task | |||||||||
| Participants with aphasia | Healthy Controls | ||||||||
| X | Y | Z | BA | X | Y | Z | BA | ||
| rvIFG | 36 | 30 | 2 | 45 | 50 | 20 | −8 | 47 | |
| lvIFG | −36 | 28 | 6 | 45 | −42 | 20 | −4 | 47 | |
| rdIFG | 50 | 18 | 32 | 44 | 38 | 22 | 30 | 9 | |
| ldIFG | −56 | 22 | 28 | 44 | −48 | 32 | 24 | 9 | |
| rLTC | 56 | −26 | 4 | 22 | 64 | −30 | −4 | 21 | |
| lLTC | −68 | −40 | 8 | 21 | −64 | −40 | 8 | 21 | |
| Semantic task | |||||||||
| Participants with aphasia | Healthy Controls | ||||||||
| X | Y | Z | BA | X | Y | Z | BA | ||
| rvIFG | 38 | 38 | −6 | 47 | 52 | 20 | 2 | 45 | 38 |
| lvIFG | −46 | 26 | −2; | 45 | −36 | 30 | −8 | 47 | −46 |
| rdIFG | 58 | 28 | 16 | 9 | 38 | 16 | 22 | 44 | 58 |
| ldIFG | −54 | 34 | 16 | 46 | −42 | 14 | 28 | 44 | −54 |
| rLTC | 52 | −8 | 0 | 41 | 64 | −46 | 6 | 21 | 52 |
| lLTC | −62 | −26 | −2 | 21 | −66 | −42 | 6 | 21 | −62 |
| Task | Group | RH Model | Bilateral Model |
|---|---|---|---|
| Phonological task | PWA | 14.49 (6.0–21.3) | 8.22 (6.2–11.7) |
| HC | 12.10 (5.73–20.1) | 16.98 (9.6–30.7) | |
| Semantic task | PWA | 4.49 (2.2–6.8) | 5.1 (2.8–7.8) |
| HC | 9.82 (5.3–21.7) | 16.66 (9.5–22.4) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Truzman, T.; Rochon, E.; Meltzer, J.; Leonard, C.; Bitan, T. Simultaneous Normalization and Compensatory Changes in Right Hemisphere Connectivity during Aphasia Therapy. Brain Sci. 2021, 11, 1330. https://doi.org/10.3390/brainsci11101330
Truzman T, Rochon E, Meltzer J, Leonard C, Bitan T. Simultaneous Normalization and Compensatory Changes in Right Hemisphere Connectivity during Aphasia Therapy. Brain Sciences. 2021; 11(10):1330. https://doi.org/10.3390/brainsci11101330
Chicago/Turabian StyleTruzman, Tammar, Elizabeth Rochon, Jed Meltzer, Carol Leonard, and Tali Bitan. 2021. "Simultaneous Normalization and Compensatory Changes in Right Hemisphere Connectivity during Aphasia Therapy" Brain Sciences 11, no. 10: 1330. https://doi.org/10.3390/brainsci11101330
APA StyleTruzman, T., Rochon, E., Meltzer, J., Leonard, C., & Bitan, T. (2021). Simultaneous Normalization and Compensatory Changes in Right Hemisphere Connectivity during Aphasia Therapy. Brain Sciences, 11(10), 1330. https://doi.org/10.3390/brainsci11101330







