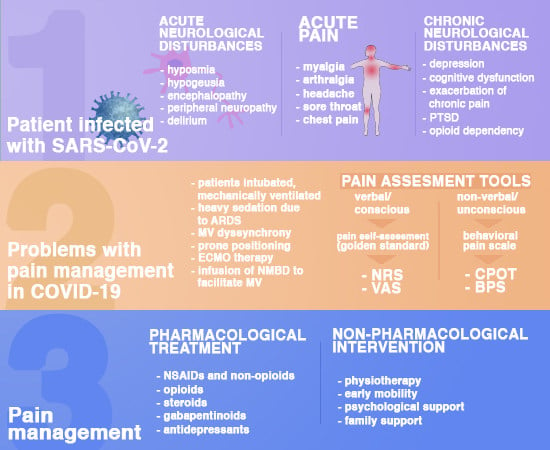
COVID-19: Pain Management in Patients with SARS-CoV-2 Infection—Molecular Mechanisms, Challenges, and Perspectives
Abstract
1. Introduction
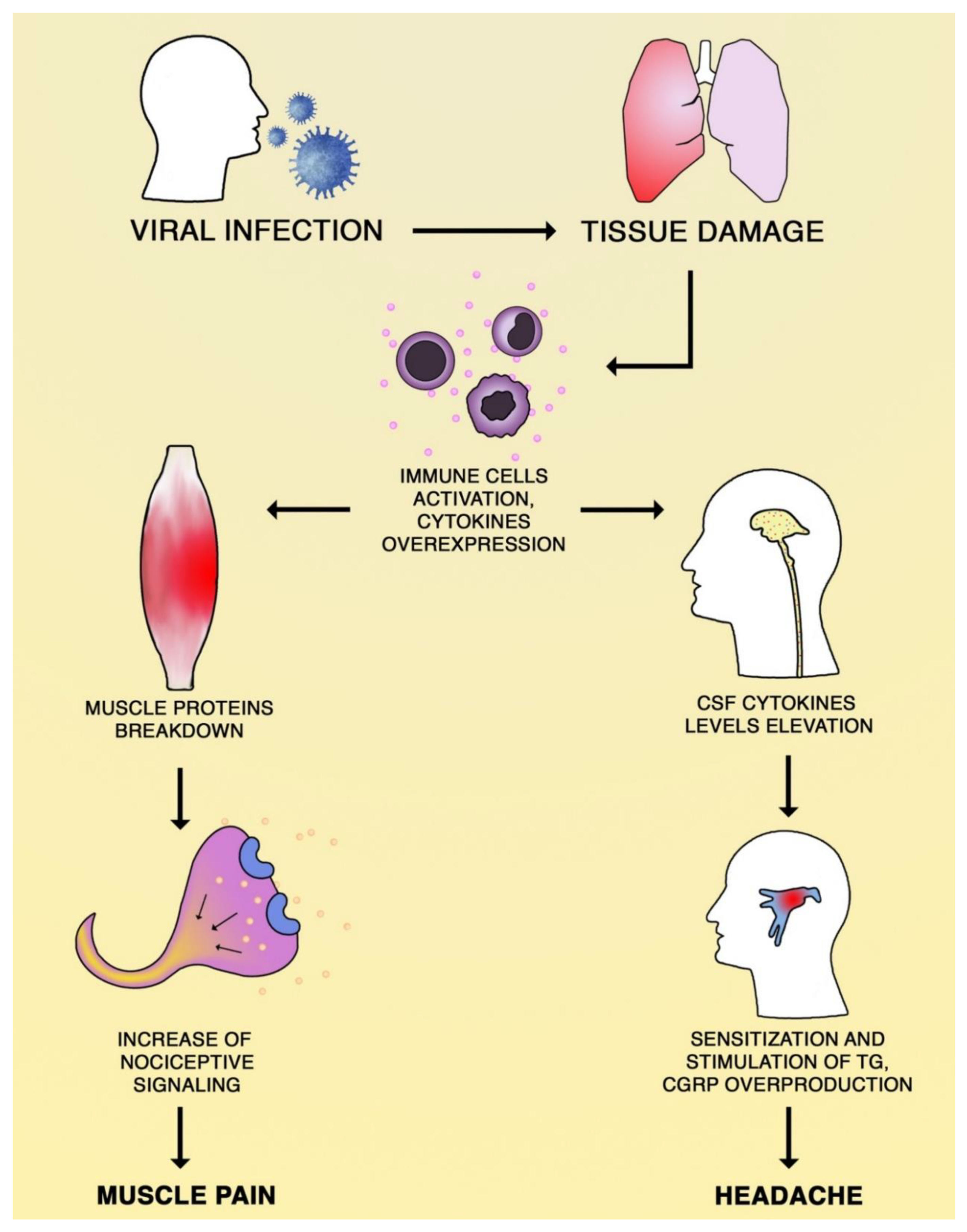
2. Pathogenesis of Pain during Coronavirus Infection
3. Difficulties Associated with Reliable Pain Assessment during COVID-19
4. Somatic Pain Treatment in SARS-CoV-2 Infections
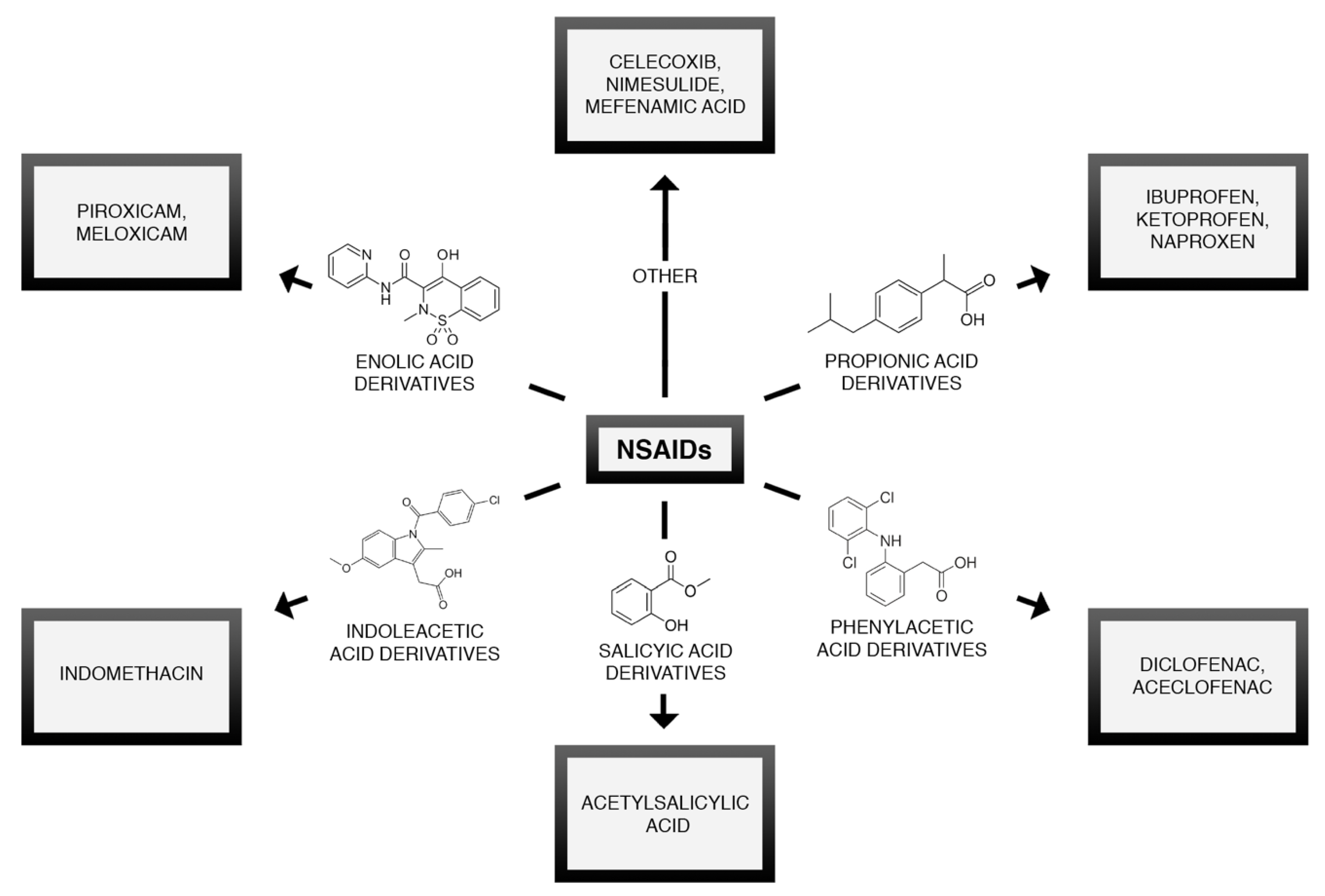
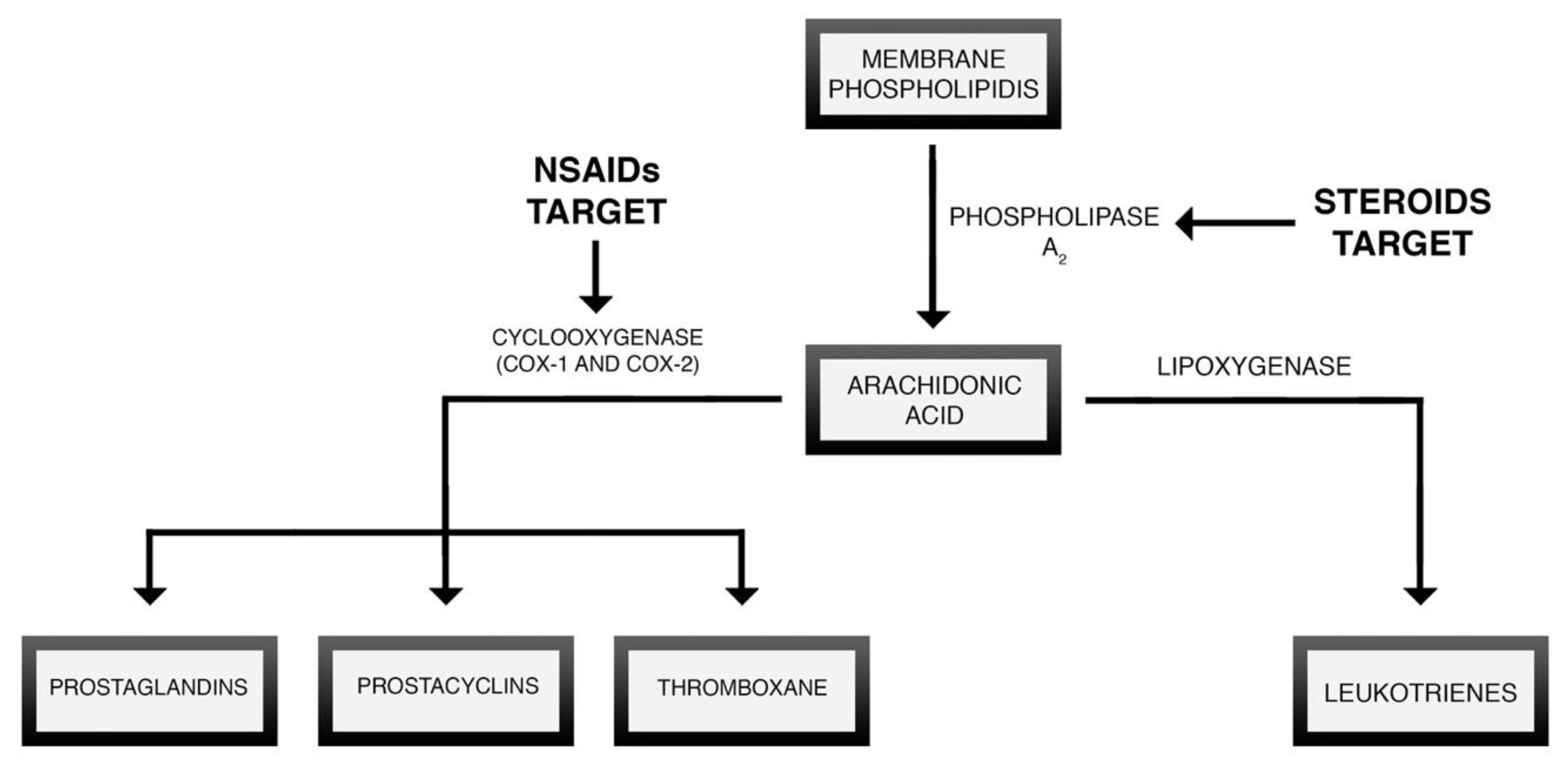
4.1. NSAIDs and Non-Opioid Analgesics
4.2. Opioids
4.3. Corticosteroids
5. Neuropathic Pain Treatment in SARS-CoV-2 Infection
Gabapentinoids
6. Summary
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| SARS-COV-2 | Severe acute respiratory syndrome coronavirus 2 |
| COVID-19 | Coronavirus Disease 2019 |
| ICU | Intensive Care Unit |
| ECMO | extracorporeal membrane oxygenation |
| NSAIDs | Nonsteroidal anti-inflammatory drugs |
| CPOT | Critical Care Pain Observation Tool |
| BPS | Behavioral Pain Scale |
| WHO | World Health Organization |
| RNA | ribonucleic acid |
| SARS | Severe Acute Respiratory Syndrome |
| MERS | Middle East Respiratory Syndrome |
| et al. | et alia |
| PNS | peripheral nervous system |
| CNS | Central nervous system |
| IL | Interleukin |
| ACE2 | Angiotensin-converting enzyme 2 |
| ANG | Angiotensin |
| TNF | tumor necrosis factor |
| PGE2 | Prostaglandin E2 |
| CSF | Cerebrospinal Fluid |
| TG | Trigeminal ganglia |
| CGRP | calcitonin gene-related peptide |
| VAS | visual analogue scale |
| NRS | numerical assessment scale |
| NMB | neuromuscular blocking medications |
| CoVs | Coronaviruses |
| COPD | chronic obstructive pulmonary disease |
| NK | Natural Killer cells |
| RR | Relative risk |
| COX | cyclooxygenase |
| LOX | lipoxygenase |
| EMA | European Medicines Agency |
| OCT | Over-the-counter drug |
| ASA | acetylsalicylic acid |
| NNT | numbers needed to treat |
References
- World Health Organization. General’s Opening Remarks at the Media Briefing on {COVID}-19. Available online: https://www.who.int/dg/speeches/detail/who-director-general-s-opening-remarks-at-the-media-briefing-on-covid-19---11-march-2020 (accessed on 15 June 2020).
- Cui, J.; Li, F.; Shi, Z.-L. Origin and evolution of pathogenic coronaviruses. Nat. Rev. Microbiol. 2019, 17, 181–192. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Zhang, Y. Animal coronaviruses: A brief introduction. Anim. Coronaviruses 2015, 1, 3–11. [Google Scholar] [CrossRef]
- Hui, D.S.C.; Zumla, A. Severe acute respiratory syndrome: Historical, epidemiologic, and clinical features. Infect. Dis. Clin. N. Am. 2019, 33, 869–889. [Google Scholar] [CrossRef] [PubMed]
- Nassar, M.S.; Bakhrebah, M.A.; Meo, S.A.; Alsuabeyl, M.S.; Zaher, W.A. Middle east respiratory syndrome coronavirus (MERS-CoV) infection: Epidemiology, pathogenesis and clinical characteristics. Eur. Rev. Med. Pharmacol. Sci. 2018, 22, 4956–4961. [Google Scholar] [CrossRef]
- Yu, P.; Zhu, J.; Zhang, Z.; Han, Y.; Huang, L. A familial cluster of infection associated with the 2019 novel coronavirus indicating potential person-to-person transmission during the incubation period. J. Infect. Dis. 2020, 11, 1757–1761. [Google Scholar] [CrossRef] [PubMed]
- Kotfis, K.; Skonieczna-Zydecka, K. COVID-19: Gastrointestinal symptoms and potential sources of 2019-NCoV transmission. Anaesthesiol. Intensive Ther. 2020, 52, 171–172. [Google Scholar] [CrossRef]
- Gu, J.; Han, B.; Wang, J. COVID-19: Gastrointestinal manifestations and potential fecal-oral transmission. Gastroenterology 2020, 158, 1518–1519. [Google Scholar] [CrossRef]
- Chan, J.F.-W.; Yuan, S.; Kok, K.-H.; To, K.K.-W.; Chu, H.; Yang, J.; Xing, F.; Liu, J.; Yip, C.C.-Y.; Poon, R.; et al. Familial cluster of pneumonia associated with the 2019 novel coronavirus indicating person-to-person transmission: A study of a family cluster. Lancet 2020, 395, 514–523. [Google Scholar] [CrossRef]
- Li, Q.; Guan, X.; Wu, P.; Wang, X.; Zhou, L.; Tong, Y.; Ren, R.; Leung, K.S.M.; Lau, E.H.Y.; Wong, J.Y.; et al. Early transmission dynamics in Wuhan, China, of novel coronavirus-infected pneumonia. N. Engl. J. Med. 2020, 382, 1199–1207. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.; Wang, Y.; Li, X.; Ren, L.; Zhao, J.; Hu, Y.; Zhang, L.; Fan, G.; Xu, J.; Gu, X.; et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet 2020, 395, 497–506. [Google Scholar] [CrossRef]
- WHO. Clinical Management of Severe Acute Respiratory Infection When Novel Coronavirus (NCoV) Infection Is Suspected; World Health Organization: Geneva, Switzerland, 2020. [Google Scholar]
- Kishor, K.; Marwah, R.; Anantharaj, A.; Kalra, S. Cardiovigilance in COVID 19. J. Pak. Med. Assoc. 2020, 70 (Suppl. S3), S77–S80. [Google Scholar] [CrossRef]
- Kałużna-Oleksy, M.; Gackowski, A.; Jankowska, E.A.; Kukulski, T.; Lelonek, M.; Nessler, J.; Pawlak, A.; Rozentryt, P.; Rubiś, P.; Straburzyńska-Migaj, E.; et al. The patient with heart failure in the face of the coronavirus disease 2019 pandemic: An Expert opinion of the heart failure working group of the polish cardiac society. Kardiol. Pol. 2020, 78, 618–631. [Google Scholar] [CrossRef] [PubMed]
- Hulot, J.-S. COVID-19 in patients with cardiovascular diseases. Arch. Cardiovasc. Dis. 2020, 113, 225–226. [Google Scholar] [CrossRef]
- Mercadante, S.; Adile, C.; Ferrera, P.; Giuliana, F.; Terruso, L.; Piccione, T. Palliative care in the time of COVID-19. J. Pain Symptom Manag. 2020, 60, E79–E80. [Google Scholar] [CrossRef] [PubMed]
- WHO. Report of the WHO-China Joint Mission on Coronavirus Disease 2019 (COVID-19); World Health Organization: Geneva, Switzerland, 2020. [Google Scholar]
- Kotfis, K.; Williams Roberson, S.; Wilson, J.E.; Dabrowski, W.; Pun, B.T.; Ely, E.W. COVID-19: ICU delirium management during SARS-CoV-2 pandemic. Crit. Care 2020, 24, 176. [Google Scholar] [CrossRef] [PubMed]
- Piraccini, E.; Byrne, H.; Taddei, S. Chronic pain management in COVID-19 Era. J. Clin. Anesth. 2020, 65, 109852. [Google Scholar] [CrossRef] [PubMed]
- Goesling, J.; Clauw, D.J.; Hassett, A.L. Pain and depression: An integrative review of neurobiological and psychological factors. Curr. Psychiatry Rep. 2013, 15, 421. [Google Scholar] [CrossRef] [PubMed]
- Eccleston, C.; Blyth, F.M.; Dear, B.F.; Fisher, E.A.; Keefe, F.J.; Lynch, M.E.; Palermo, T.M.; Reid, M.C.; de Williams, A.C.C. Managing patients with chronic pain during the COVID-19 outbreak: Considerations for the rapid introduction of remotely supported (EHealth) pain management services. Pain 2020, 161, 889–893. [Google Scholar] [CrossRef]
- Wang, H.; Zhang, L. Risk of COVID-19 for patients with cancer. The Lancet. Oncol. 2020, 21, e181. [Google Scholar] [CrossRef]
- Fausto, J.; Hirano, L.; Lam, D.; Mehta, A.; Mills, B.; Owens, D.; Perry, E.; Curtis, J.R. Creating a palliative care inpatient response plan for COVID-19-The UW medicine experience. J. Pain Symptom Manag. 2020, 60, e21–e26. [Google Scholar] [CrossRef]
- The British Pain Society. Pain Management during COVID-19 Viral Infection. Available online: https://www.britishpainsociety.org/static/uploads/resources/files/Pain_Management_during_COVID-19_viral_infection.pdf (accessed on 5 June 2020).
- Boziki, M.K.; Mentis, A.F.A.; Shumilina, M.; Makshakov, G.; Evdoshenko, E.; Grigoriadis, N. COVID-19 immunopathology and the central nervous system: Implication for multiple sclerosis and other autoimmune diseases with associated demyelination. Brain Sci. 2020, 10, 345. [Google Scholar] [CrossRef] [PubMed]
- Mao, L.; Jin, H.; Wang, M.; Hu, Y.; Chen, S.; He, Q.; Chang, J.; Hong, C.; Zhou, Y.; Wang, D.; et al. Neurologic manifestations of hospitalized patients with coronavirus disease 2019 in Wuhan, China. JAMA Neurol. 2020, 77, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Abdelnour, L.; Eltahir Abdalla, M.; Babiker, S. COVID 19 infection presenting as motor peripheral neuropathy. J. Formos. Med. Assoc. Taiwan Yi Zhi 2020, 119, 1119–1120. [Google Scholar] [CrossRef] [PubMed]
- Sheraton, M.; Deo, N.; Kashyap, R.; Surani, S. A review of neurological complications of COVID-19. Cureus 2020, 12, e8192. [Google Scholar] [CrossRef] [PubMed]
- Montalvan, V.; Lee, J.; Bueso, T.; de Toledo, J.; Rivas, K. Neurological manifestations of COVID-19 and other coronavirus infections: A systematic review. Clin. Neurol. Neurosurg. 2020, 194, 105921. [Google Scholar] [CrossRef] [PubMed]
- Helms, J.; Kremer, S.; Merdji, H.; Clere-Jehl, R.; Schenck, M.; Kummerlen, C.; Collange, O.; Boulay, C.; Fafi-Kremer, S.; Ohana, M.; et al. Neurologic features in severe SARS-CoV-2 infection. N. Engl. J. Med. 2020, 382, 2268–2270. [Google Scholar] [CrossRef]
- Su, S.; Cui, H.; Wang, T.; Shen, X.; Ma, C. Pain: A potential new label of COVID-19. Brain Behav. Immun. 2020, 87, 159–160. [Google Scholar] [CrossRef]
- Li, L.; Huang, T.; Wang, Y.; Wang, Z.; Liang, Y.; Huang, T.; Zhang, H.; Sun, W.; Wang, Y. COVID-19 Patients’ clinical characteristics, discharge rate, and fatality rate of meta-analysis. J. Med. Virol. 2020, 92, 577–583. [Google Scholar] [CrossRef]
- Manjavachi, M.N.; Motta, E.M.; Marotta, D.M.; Leite, D.F.P.; Calixto, J.B. Mechanisms involved in IL-6-induced muscular mechanical hyperalgesia in mice. Pain 2010, 151, 345–355. [Google Scholar] [CrossRef]
- Jiang, X.; Coffee, M.; Bari, A.; Wang, J.; Jiang, X.; Huang, J.; Shi, J.; Dai, J.; Cai, J.; Zhang, T.; et al. Towards an artificial intelligence framework for data-driven prediction of coronavirus clinical severity. Comput. Mater. Contin. 2020, 63, 537–551. [Google Scholar] [CrossRef]
- Chen, G.; Wu, D.; Guo, W.; Cao, Y.; Huang, D.; Wang, H.; Wang, T.; Zhang, X.; Chen, H.; Yu, H.; et al. Clinical and immunological features of severe and moderate coronavirus disease 2019. J. Clin. Investig. 2020, 130, 2620–2629. [Google Scholar] [CrossRef] [PubMed]
- Pedersen, S.F.; Ho, Y.-C. SARS-CoV-2: A storm is raging. J. Clin. Investig. 2020, 130, 2202–2205. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, S.; Beyer, C. A fatal alliance between microglia, inflammasomes, and central pain. Int. J. Mol. Sci. 2020, 21, 3764. [Google Scholar] [CrossRef] [PubMed]
- Fiebich, B.L.; Akter, S.; Akundi, R.S. The two-hit hypothesis for neuroinflammation: Role of exogenous atp in modulating inflammation in the brain. Front. Cell. Neurosci. 2014, 8, 260. [Google Scholar] [CrossRef] [PubMed]
- Martinon, F.; Burns, K.; Tschopp, J. The inflammasome: A molecular platform triggering activation of inflammatory caspases and processing of proil-beta. Mol. Cell 2002, 10, 417–426. [Google Scholar] [CrossRef]
- Trendelenburg, G. Acute neurodegeneration and the inflammasome: Central processor for danger signals and the inflammatory response? J. Cereb. Blood Flow Metab. 2008, 28, 867–881. [Google Scholar] [CrossRef] [PubMed]
- Song, L.; Pei, L.; Yao, S.; Wu, Y.; Shang, Y. NLRP3 inflammasome in neurological diseases, from functions to therapies. Front. Cell. Neurosci. 2017, 11, 63. [Google Scholar] [CrossRef]
- Schaefer, L. Complexity of danger: The diverse nature of damage-associated molecular patterns. J. Biol. Chem. 2014, 289, 35237–35245. [Google Scholar] [CrossRef]
- Evavold, C.L.; Kagan, J.C. How inflammasomes inform adaptive immunity. J. Mol. Biol. 2018, 430, 217–237. [Google Scholar] [CrossRef]
- Solomon, I.H.; Normandin, E.; Bhattacharyya, S.; Mukerji, S.S.; Keller, K.; Ali, A.S.; Adams, G.; Hornick, J.L.; Padera, R.F.; Sabeti, P. Neuropathological features of Covid-19. N. Engl. J. Med. 2020, NEJMc2019373. [Google Scholar] [CrossRef]
- Kigerl, K.A.; Gensel, J.C.; Ankeny, D.P.; Alexander, J.K.; Donnelly, D.J.; Popovich, P.G. Identification of two distinct macrophage subsets with divergent effects causing either neurotoxicity or regeneration in the injured mouse spinal cord. J. Neurosci. 2009, 29, 13435–13444. [Google Scholar] [CrossRef] [PubMed]
- Villa, A.; Della Torre, S.; Maggi, A. Sexual differentiation of microglia. Front. Neuroendocrinol. 2019, 52, 156–164. [Google Scholar] [CrossRef] [PubMed]
- Habib, P.; Beyer, C. Regulation of brain microglia by female gonadal steroids. J. Steroid Biochem. Mol. Biol. 2015, 146, 3–14. [Google Scholar] [CrossRef] [PubMed]
- Lim, H.; Kim, D.; Lee, S.J. Toll-Like receptor 2 mediates peripheral nerve injury-induced NADPH oxidase 2 expression in spinal cord microglia. J. Biol. Chem. 2013, 288, 7572–7579. [Google Scholar] [CrossRef] [PubMed]
- Rozen, T.; Swidan, S.Z. Elevation of CSF tumor necrosis factor α levels in new daily persistent headache and treatment refractory chronic migraine. Headache J. Head Face Pain 2007, 47, 1050–1055. [Google Scholar] [CrossRef] [PubMed]
- Eccles, R. Understanding the symptoms of the common cold and influenza. Lancet. Infect. Dis. 2005, 5, 718–725. [Google Scholar] [CrossRef]
- Acharya, A.; Kevadiya, B.D.; Gendelman, H.E.; Byrareddy, S.N. SARS-CoV-2 Infection leads to neurological dysfunction. J. Neuroimmune Pharmacol. 2020, 15, 167–173. [Google Scholar] [CrossRef]
- Pettus, K.; Cleary, J.F.; de Lima, L.; Ahmed, E.; Radbruch, L. Availability of internationally controlled essential medicines in the COVID-19 pandemic. J. Pain Symptom Manag. 2020, 60, e48–e51. [Google Scholar] [CrossRef]
- Kotfis, K.; Zegan-Barańska, M.; Szydłowski, Ł.; Żukowski, M.; Ely, E.W. Methods of pain assessment in adult intensive care unit patients—polish version of the CPOT (Critical Care Pain Observation Tool) and BPS (Behavioral Pain Scale). Anaesthesiol. Intensive Ther. 2017, 49, 66–72. [Google Scholar] [CrossRef]
- Gélinas, C.; Arbour, C. Behavioral and physiologic indicators during a nociceptive procedure in conscious and unconscious mechanically ventilated adults: Similar or different? J. Crit. Care 2009, 24, e7-17. [Google Scholar] [CrossRef]
- Gélinas, C.; Fillion, L.; Puntillo, K.A.; Viens, C.; Fortier, M. Validation of the critical-care pain observation tool in adult patients. Am. J. Crit. Care 2006, 15, 420–427. [Google Scholar] [CrossRef] [PubMed]
- Payen, J.F.; Bru, O.; Bosson, J.L.; Lagrasta, A.; Novel, E.; Deschaux, I.; Lavagne, P.; Jacquot, C. Assessing pain in critically Ill sedated patients by using a behavioral pain scale. Crit. Care Med. 2001, 29, 2258–2263. [Google Scholar] [CrossRef] [PubMed]
- Kotfis, K.; Williams Roberson, S.; Wilson, J.E.; Pun, B.T.; Ely, E.W. COVID-19: What do we need to know about ICU delirium during SARS-CoV-2 pandemic? Anaesthesiol. Intensive Ther. 2020, 52, 132–138. [Google Scholar] [CrossRef] [PubMed]
- Kotfis, K.; Zegan-Barańska, M.; Strzelbicka, M.; Safranow, K.; Żukowski, M.; Ely, E.W. Validation of the polish version of the critical care pain observation tool (CPOT) to assess pain intensity in adult, intubated intensive care unit patients: The POL-CPOT study. Arch. Med. Sci. 2018, 14, 880–889. [Google Scholar] [CrossRef] [PubMed]
- Kotfis, K.; Strzelbicka, M.; Zegan-Barańska, M.; Safranow, K.; Brykczyński, M.; Żukowski, M.; Ely, E.W. Validation of the behavioral pain scale to assess pain intensity in adult, intubated postcardiac surgery patients: A cohort observational study—POL-BPS. Medicine 2018, 97, e12443. [Google Scholar] [CrossRef]
- Madhok, J.; Mihm, F.G. Rethinking sedation during prolonged mechanical ventilation for COVID-19 respiratory failure. Anesth. Analg. 2020, 131, e123–e124. [Google Scholar] [CrossRef]
- Hanidziar, D.; Bittner, E. Sedation of mechanically ventilated COVID-19 patients: Challenges and special considerations. Anesth. Analg. 2020, 131, e40–e41. [Google Scholar] [CrossRef]
- Devlin, J.W.; O’Neal, H.R.J.; Thomas, C.; Barnes Daly, M.A.; Stollings, J.L.; Janz, D.R.; Ely, E.W.; Lin, J.C. Strategies to optimize ICU liberation (A to F) bundle performance in critically Ill adults with coronavirus disease 2019. Crit. Care Explor. 2020, 2, e0139. [Google Scholar] [CrossRef]
- Friedewald, V.E.; Bennett, J.S.; Christo, J.P.; Pool, J.L.; Scheiman, J.M.; Simon, L.S.; Strand, V.; White, W.B.; Williams, G.W.; Roberts, W.C. AJC editor’s consensus: Selective and nonselective nonsteroidal anti-inflammatory drugs and cardiovascular risk. Am. J. Cardiol. 2010, 106, 873–884. [Google Scholar] [CrossRef]
- Antman, E.M.; Bennett, J.S.; Daugherty, A.; Furberg, C.; Roberts, H.; Taubert, K.A. Use of nonsteroidal antiinflammatory drugs: An update for clinicians: A scientific statement from the american heart association. Circulation 2007, 115, 1634–1642. [Google Scholar] [CrossRef]
- Picot, D.; Loll, P.J.; Garavito, R.M. The X-ray crystal structure of the membrane protein prostaglandin H2 synthase-1. Nature 1994, 367, 243–249. [Google Scholar] [CrossRef] [PubMed]
- Khan, S.; Andrews, K.L.; Chin-dusting, J.P.F. Cyclo-Oxygenase (COX) inhibitors and cardiovascular risk: Are non-steroidal anti-inflammatory drugs really anti-inflammatory? Int. J. Mol. Sci. 2019, 20, 4262. [Google Scholar] [CrossRef] [PubMed]
- Russell, B.; Moss, C.; Rigg, A.; Van Hemelrijck, M. COVID-19 and treatment with NSAIDs and corticosteroids: Should we be limiting their use in the clinical setting? Ecancermedicalscience 2020, 14, 1023. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. The Use of Non-Steroidal Anti-Inflammatory Drugs (NSAIDs) in Patients with COVID-19. Available online: https://www.who.int/news-room/commentaries/detail/the-use-of-non-steroidal-anti-inflammatory-drugs-(nsaids)-in-patients-with-covid-19 (accessed on 5 June 2020).
- Day, M. Covid-19: European drugs agency to review safety of ibuprofen. BMJ 2020, 368, m1168. [Google Scholar] [CrossRef] [PubMed]
- Kotsiou, O.S.; Zarogiannis, S.G.; Gourgoulianis, K.I. Prehospital NSAIDs use prolong hospitalization in patients with pleuro-pulmonary infection. Respir. Med. 2017, 123, 28–33. [Google Scholar] [CrossRef]
- Micallef, J.; Soeiro, T.; Jonville-Béra, A.-P.; French Society of Pharmacology and Therapeutics. Non-Steroidal anti-inflammatory drugs, pharmacology, and COVID-19 infection. Therapie 2020, S0040-5957, 30092–30095. [Google Scholar] [CrossRef]
- Yousefifard, M.; Zali, A.; Zarghi, A.; Madani Neishaboori, A.; Hosseini, M.; Safari, S. Non-Steroidal anti-inflammatory drugs in management of COVID-19: A systematic review on current evidence. Int. J. Clin. Pract. 2020, e13557. [Google Scholar] [CrossRef]
- Rinott, E.; Kozer, E.; Shapira, Y.; Bar-Haim, A.; Youngster, I. Ibuprofen use and clinical outcomes in COVID-19 patients. Clin. Microbiol. Infect. 2020. [Google Scholar] [CrossRef]
- Bein, B.; Bachmann, M.; Huggett, S.; Wegermann, P. SARS-CoV-2/COVID-19: Evidence-Based recommendations on diagnosis and therapy. Geburtshilfe Frauenheilkd. 2020, 80, 491–498. [Google Scholar] [CrossRef]
- Morgan, E.T. Regulation of cytochromes P450 during inflammation and infection. Drug Metab. Rev. 1997, 29, 1129–1188. [Google Scholar] [CrossRef]
- Lampl, C.; Likar, R. Metamizole (dipyrone): Mode of action, drug-drug interactions, and risk of agranulocytosis. Schmerz 2014, 28, 584–590. [Google Scholar] [CrossRef] [PubMed]
- Aly, O. Molecular docking reveals the potential of aliskiren, dipyridamole, mopidamol, rosuvastatin, rolitetracycline and metamizole to inhibit COVID-19 virus main protease. ChemRxiv 2020. [Google Scholar] [CrossRef]
- Schönhöfer, P.; Offerhaus, L.; Herxheimer, A. Dipyrone and agranulocytosis: What is the risk? Lancet 2003, 361, 968–969. [Google Scholar] [CrossRef]
- Shanthanna, H.; Strand, N.H.; Provenzano, D.A.; Lobo, C.A.; Eldabe, S.; Bhatia, A.; Wegener, J.; Curtis, K.; Cohen, S.P.; Narouze, S. Caring for patients with pain during the COVID-19 pandemic: Consensus recommendations from an international expert panel. Anaesthesia 2020, 75, 935–944. [Google Scholar] [CrossRef] [PubMed]
- Ekström, M.; Nilsson, F.; Abernethy, A.A.; Currow, D.C. Effects of opioids on breathlessness and exercise capacity in chronic obstructive pulmonary disease: A systematic review. Ann. Am. Thorac. Soc. 2015, 12, 1079–1092. [Google Scholar] [CrossRef] [PubMed]
- Yamaguchi, T.; Matsunuma, R.; Suzuki, K.; Matsuda, Y.; Mori, M.; Watanabe, H. The current practice of opioid for cancer dyspnea: The result from the nationwide survey of japanese palliative care physicians. J. Pain Symptom Manag. 2019, 5, 672–677. [Google Scholar] [CrossRef] [PubMed]
- Vozoris, N.T. Opioid utility for dyspnea in chronic obstructive pulmonary disease: A Complicated and controversial story. Ann. Palliat. Med. 2020, 9, 571–578. [Google Scholar] [CrossRef] [PubMed]
- Woodcock, A.A.; Gross, E.R.; Gellert, A.; Shah, S.; Johnson, M.; Geddes, D.M. Effects of dihydrocodeine, alcohol, and caffeine on breathlessness and exercise tolerance in patients with chronic obstructive lung disease and normal blood gases. N. Engl. J. Med. 1981, 305, 1611–1616. [Google Scholar] [CrossRef]
- Light, R.W.; Muro, J.R.; Sato, R.I.; Stansbury, D.W.; Fischer, C.E.; Brown, S.E. Effects of oral morphine on breathlessness and exercise tolerance in patients with chronic obstructive pulmonary disease. Am. Rev. Respir. Dis. 1989, 139, 126–133. [Google Scholar] [CrossRef]
- Banzett, R.B.; Adams, L.; O’Donnell, C.R.; Gilman, S.A.; Lansing, R.W.; Schwartzstein, R.M. Using laboratory models to test treatment: Morphine reduces dyspnea and hypercapnic ventilatory response. Am. J. Respir. Crit. Care Med. 2011, 184, 920–927. [Google Scholar] [CrossRef]
- Vozoris, N.T.; Pequeno, P.; Li, P.; Austin, P.C.; O’Donnell, D.E.; Gershon, A.S. Predictors of opioid-related adverse pulmonary events among older adults with COPD [published online ahead of print, 2020 May 12]. Ann. Am. Thorac. Soc. 2020. [Google Scholar] [CrossRef]
- Vozoris, N.T.; Wang, X.; Fischer, H.D.; Bell, C.M.; Donnell, D.E.; Austin, P.C.; Stephenson, A.L.; Gill, S.S.; Rochon, P.A. Incident opioid drug use and adverse respiratory outcomes among older adults with COPD. Eur. Respir. J. 2016, 48, 683–693. [Google Scholar] [CrossRef] [PubMed]
- Cata, J.P.; Bhavsar, S.P. Pharmacology of opioids. Basic Sci. Anesth. 2017, 7, 123–137. [Google Scholar] [CrossRef]
- Yamamoto, Y.; Watanabe, H.; Sakurai, A.; Deguchi, Y.; Hirano, S.; Okumura, Y.; Kumon, A.; Watanabe, N.; Osada, T.; Yamamura, K.; et al. Effect of continuous intravenous oxycodone infusion in opioid-naïve cancer patients with dyspnea. Jpn. J. Clin. Oncol. 2018, 48, 748–752. [Google Scholar] [CrossRef] [PubMed]
- Kuo, A.; Wyse, B.D.; Meutermans, W.; Smith, M.T. In vivo profiling of seven common opioids for antinociception, constipation and respiratory depression: No two opioids have the same profile. Br. J. Pharmacol. 2015, 172, 532–548. [Google Scholar] [CrossRef] [PubMed]
- Flores, L.R.; Wahl, S.M.; Bayer, B.M. Mechanisms of morphine-induced immunosuppression: Effect of acute morphine administration on lymphocyte trafficking. J. Pharmacol. Exp. Ther. 1995, 272, 1246–1251. [Google Scholar]
- Hernandez, M.C.; Flores, L.R.; Bayer, B.M. Immunosuppression by Morphine is mediated by central pathways. J. Pharmacol. Exp. Ther. 1993, 267, 1336–1341. [Google Scholar]
- Shavit, Y.; Ben-Eliyahu, S.; Zeidel, A.; Beilin, B. Effects of fentanyl on natural killer cell activity and on resistance to tumor metastasis in rats. Dose and timing study. Neuroimmunomodulation 2004, 11, 255–260. [Google Scholar] [CrossRef]
- Beilin, B.; Shavit, Y.; Hart, J.; Mordashov, B.; Cohn, S.; Notti, I.; Bessler, H. Effects of anesthesia based on large versus small doses of fentanyl on natural killer cell cytotoxicity in the perioperative period. Anesth. Analg. 1996, 82, 492–497. [Google Scholar] [CrossRef]
- Forget, P.; Collet, V.; Lavand’homme, P.; De Kock, M. Does analgesia and condition influence immunity after surgery? Effects of fentanyl, ketamine and clonidine on natural killer activity at different ages. Eur. J. Anaesthesiol. 2010, 27, 233–240. [Google Scholar] [CrossRef]
- Narahara, H.; Kadoi, Y.; Hinohara, H.; Kunimoto, F.; Saito, S. Comparative effects of flurbiprofen and fentanyl on natural killer cell cytotoxicity, lymphocyte subsets and cytokine concentrations in post-surgical intensive care unit patients: Prospective, randomized study. J. Anesth. 2013, 27, 676–683. [Google Scholar] [CrossRef]
- Yardeni, I.Z.; Beilin, B.; Mayburd, E.; Alcalay, Y.; Bessler, H. Relationship between fentanyl dosage and immune function in the postoperative period. J. Opioid Manag. 2008, 4, 27–33. [Google Scholar] [CrossRef] [PubMed]
- Davis, M.P.; Behm, B. Reasons to avoid fentanyl. Ann. Palliat. Med. 2020, 9, 611–624. [Google Scholar] [CrossRef] [PubMed]
- Wiese, A.D.; Griffin, M.R.; Schaffner, W.; Stein, C.M.; Greevy, R.A.; Mitchel, E.F.; Grijalva, C.G. Long-Acting opioid use and the risk of serious infections: A retrospective cohort study. Clin. Infect. Dis. 2019, 68, 1862–1869. [Google Scholar] [CrossRef] [PubMed]
- Davis, M.P. Twelve reasons for considering buprenorphine as a frontline analgesic in the management of pain. J. Support. Oncol. 2012, 10, 209–219. [Google Scholar] [CrossRef]
- Davis, M.P.; Pasternak, G.; Behm, B. Treating chronic pain: An Overview of clinical studies centered on the buprenorphine option. Drugs 2018, 78, 1211–1228. [Google Scholar] [CrossRef]
- Wong, S.S.Y.; Yuen, K.-Y. The management of coronavirus infections with particular reference to SARS. J. Antimicrob. Chemother. 2008, 62, 437–441. [Google Scholar] [CrossRef]
- Hao, D.; He, L.; Qu, J.; Pan, J.; Hu, B.; Zhang, J.; Li, Z. A study of pulmonary inflammatory reaction induced by N-protein of SARS-CoV in rat models and effects of glucocorticoids on it. Zhonghua Nei Ke Za Zhi 2005, 44, 890–893. [Google Scholar] [PubMed]
- Zhang, X.; Alekseev, K.; Jung, K.; Vlasova, A.; Hadya, N.; Saif, L.J. Cytokine responses in porcine respiratory coronavirus-infected pigs treated with corticosteroids as a model for severe acute respiratory syndrome. J. Virol. 2008, 82, 4420–4428. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Stockman, L.J.; Bellamy, R.; Garner, P. SARS: Systematic review of treatment effects. PLoS Med. 2006, 3, e343. [Google Scholar] [CrossRef] [PubMed]
- Arabi, Y.M.; Mandourah, Y.; Al-Hameed, F.; Sindi, A.A.; Almekhlafi, G.A.; Hussein, M.A.; Jose, J.; Pinto, R.; Al-Omari, A.; Kharaba, A.; et al. Corticosteroid therapy for critically Ill patients with middle east respiratory syndrome. Am. J. Respir. Crit. Care Med. 2018, 197, 757–767. [Google Scholar] [CrossRef] [PubMed]
- Pleasure, S.J.; Green, A.J.; Josephson, S.A. The spectrum of neurologic disease in the severe acute respiratory syndrome coronavirus 2 pandemic infection: Neurologists move to the frontlines. JAMA Neurol. 2020, 77, 679–680. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.-C.; Bai, W.-Z.; Hashikawa, T. The neuroinvasive potential of SARS-CoV2 may play a role in the respiratory failure of COVID-19 patients. J. Med. Virol. 2020, 92, 552–555. [Google Scholar] [CrossRef] [PubMed]
- Egunsola, O.; Wylie, C.E.; Chitty, K.M.; Buckley, N.A. Systematic review of the efficacy and safety of gabapentin and pregabalin for pain in children and adolescents. Anesth. Analg. 2019, 128, 811–819. [Google Scholar] [CrossRef] [PubMed]
- Finnerup, N.B.; Attal, N.; Haroutounian, S.; McNicol, E.; Baron, R.; Dworkin, R.H.; Gilron, I.; Haanpää, M.; Hansson, P.; Jensen, T.S.; et al. Pharmacotherapy for neuropathic pain in adults: A systematic review and meta-analysis. Lancet. Neurol. 2015, 14, 162–173. [Google Scholar] [CrossRef]
- Brooks, K.G.; Kessler, T.L. Treatments for neuropathic pain. Clin. Pharm. 2017, 9, 1–14. [Google Scholar] [CrossRef]
- Pop-Busui, R.; Boulton, A.J.M.; Feldman, E.L.; Bril, V.; Freeman, R.; Malik, R.A.; Sosenko, J.M.; Ziegler, D. Diabetic neuropathy: A position statement by the american diabetes association. Diabetes Care 2017, 40, 136–154. [Google Scholar] [CrossRef] [PubMed]
- Zilliox, L.A. Neuropathic pain. Continuum (Minneap. Minn) 2017, 23, 512–532. [Google Scholar] [CrossRef] [PubMed]
- Accord Healthcare Limited. Pregabalin Accord SmPC. Available online: https://www.medicines.org.uk/emc/medicine/30815 (accessed on 18 June 2020).
- Gomes, T.; Juurlink, D.N.; Antoniou, T.; Mamdani, M.M.; Paterson, J.M.; van den Brink, W. Gabapentin, opioids, and the risk of opioid-related death: A Population-based nested case–control study. PLoS Med. 2017, 14, e1002396. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Drożdżal, S.; Rosik, J.; Lechowicz, K.; Machaj, F.; Szostak, B.; Majewski, P.; Rotter, I.; Kotfis, K. COVID-19: Pain Management in Patients with SARS-CoV-2 Infection—Molecular Mechanisms, Challenges, and Perspectives. Brain Sci. 2020, 10, 465. https://doi.org/10.3390/brainsci10070465
Drożdżal S, Rosik J, Lechowicz K, Machaj F, Szostak B, Majewski P, Rotter I, Kotfis K. COVID-19: Pain Management in Patients with SARS-CoV-2 Infection—Molecular Mechanisms, Challenges, and Perspectives. Brain Sciences. 2020; 10(7):465. https://doi.org/10.3390/brainsci10070465
Chicago/Turabian StyleDrożdżal, Sylwester, Jakub Rosik, Kacper Lechowicz, Filip Machaj, Bartosz Szostak, Paweł Majewski, Iwona Rotter, and Katarzyna Kotfis. 2020. "COVID-19: Pain Management in Patients with SARS-CoV-2 Infection—Molecular Mechanisms, Challenges, and Perspectives" Brain Sciences 10, no. 7: 465. https://doi.org/10.3390/brainsci10070465
APA StyleDrożdżal, S., Rosik, J., Lechowicz, K., Machaj, F., Szostak, B., Majewski, P., Rotter, I., & Kotfis, K. (2020). COVID-19: Pain Management in Patients with SARS-CoV-2 Infection—Molecular Mechanisms, Challenges, and Perspectives. Brain Sciences, 10(7), 465. https://doi.org/10.3390/brainsci10070465