Abstract
A considerable subset of mild traumatic brain injury (mTBI) patients fail to return to baseline functional status at or beyond 3 months postinjury. Identifying at-risk patients for poor outcome in the emergency department (ED) may improve surveillance strategies and referral to care. Subjects with mTBI (Glasgow Coma Scale 13–15) and negative ED initial head CT < 24 h of injury, completing 3- or 6-month functional outcome (Glasgow Outcome Scale-Extended; GOSE), were extracted from the prospective, multicenter Transforming Research and Clinical Knowledge in Traumatic Brain Injury (TRACK-TBI) Pilot study. Outcomes were dichotomized to full recovery (GOSE = 8) vs. functional deficits (GOSE < 8). Univariate predictors with p < 0.10 were considered for multivariable regression. Adjusted odds ratios (AOR) were reported for outcome predictors. Significance was assessed at p < 0.05. Subjects who completed GOSE at 3- and 6-month were 211 (GOSE < 8: 60%) and 185 (GOSE < 8: 65%). Risk factors for 6-month GOSE < 8 included less education (AOR = 0.85 per-year increase, 95% CI: (0.74–0.98)), prior psychiatric history (AOR = 3.75 (1.73–8.12)), Asian/minority race (American Indian/Alaskan/Hawaiian/Pacific Islander) (AOR = 23.99 (2.93–196.84)), and Hispanic ethnicity (AOR = 3.48 (1.29–9.37)). Risk factors for 3-month GOSE < 8 were similar with the addition of injury by assault predicting poorer outcome (AOR = 3.53 (1.17–10.63)). In mTBI patients seen in urban trauma center EDs with negative CT, education, injury by assault, Asian/minority race, and prior psychiatric history emerged as risk factors for prolonged disability.
1. Introduction
In 2014, at least 2.5 million people were treated for traumatic brain injury (TBI) in U.S. emergency departments (ED), of which 80%–90% were classified as “mild” (mTBI) with a Glasgow Coma Scale (GCS) score of 13–15 [1]. The frequency of mTBI-related emergency department visits increased from 569.4/100,000 persons in 2006 to 807.9/100,000 in 2012 and continues at an increase of 7% annually [2]. While all-cause mortality for mTBI is low (1.4%) [3], patients are at significant risk of psychiatric sequelae [4] and cognitive impairment postinjury [5]. It is estimated that 20%–70% of mTBI patients suffer from persistent functional, cognitive and/or neuropsychological symptoms at or beyond 3 months postinjury [6,7]. mTBI patients often suffer disruptions to functional and social wellbeing postinjury and require greater use of healthcare resources [8]—upwards of $17 billion annually in the U.S. [9]. However, indications for referral to these necessary resources remain poorly understood, masking the true burden of mTBI. In a recent large study of over 1.1 million adult patients who suffered TBI in the state of California from 2005–2014, 40.5% had a revisit during the first year, and, of those revisiting patients, 13.8% had five or more revisits within that first year [10]. A study of 2787 mTBI patients showed that 5% returned unexpectedly to the ED within 72 h, with post-concussion symptoms or otherwise needing repeat clinical evaluation [11]. Identifying the predictors of negative outcomes for patients with mTBI and using this information to inform ED management strategies could improve patient outcomes and decrease ED utilization.
The evidence-driven management of mTBI is limited compared to moderate and severe TBI, in which additional prognostic factors have been identified due to their association with the need for neurosurgical intervention [12]. The heterogeneous nature of mTBI and the need for guidelines in its evaluation were demonstrated in the Transforming Research and Clinical Knowledge in Traumatic Brain Injury Pilot (TRACK-TBI Pilot) study [13], which identified subgroups within the mTBI population with divergent functional outcomes, persistence of symptoms, and neuropsychiatric impairment at 90 and 180 days postinjury [14]. Current ED guidelines support the utilization of GCS and pupillary reactivity to assess and monitor severity in head injury, and computed tomography (CT) as first-line imaging; however, these assessments are often normal or negative in mTBI patients and may not directly correlate with post-traumatic complaints [15,16,17]. Data on additional clinical factors, such as loss of consciousness (LOC), post traumatic amnesia (PTA), hypoxia, hypotension, and the use of anticoagulants and antiplatelets, are often collected in the ED, though their clinical significance is not well defined [9,18]. Variables not yet described in guidelines due to minimal literature but found to be predictive of impaired outcomes following mTBI include education level and pre-injury psychiatric history, such as anxiety and depression [4,16,19,20].
With the increasing number of mTBI patients presenting to the ED and growing evidence of the presence of persistent post-traumatic symptomatology and disability, it is critical to identify risk factors for adverse outcomes, update ED assessment and treatment guidelines, and improve the efficiency of ED utilization. This is necessary to determine the appropriate level of care and services for this heterogeneous group of individuals. In the current ED mTBI management paradigm, patients who have a CT scan without evidence of intracranial abnormalities are routinely discharged from the ED with return precautions. However, it has been found that only 25% return to follow-up, of which one-third had not completely resumed pre-injury activities [21]. Thus, beyond improving outcome, identifying mTBI patients with negative CT who are at risk for poorer outcomes can improve ED evaluation and triage paradigms, as well as healthcare utilization. The current analysis of the TRACK-TBI Pilot dataset sought to identify a cohort of patients in the ED with mTBI who are at greater risk for adverse outcomes despite an initial negative head CT scan, and assess whether traditional clinical factors measured in the ED in this patient population are adequate for predicting outcome at 3 and 6 months postinjury.
2. Materials and Methods
2.1. Study Design of the TRACK-TBI Pilot Study
The TRACK-TBI Pilot study was conducted at three U.S. Level I trauma centers (Zuckerberg San Francisco General Hospital (San Francisco, CA, USA), University of Pittsburgh Medical Center (Pittsburgh, PA, USA), and University Medical Center Brackenridge (Austin, TX, USA)) using the National Institute of Neurological Disorders and Stroke (NINDS) TBI Common Data Elements (CDEs) [13,22,23,24,25]. The TRACK-TBI Pilot inclusion criteria were external force trauma to the head, presentation to one of the three trauma centers, and a clinically indicated head CT scan, as determined by the treating emergency physician, obtained within 24 h of injury. Exclusion criteria were pregnancy, ongoing life-threatening disease (e.g., end-stage malignancy), police custody, involuntary psychiatric hold, and inability to speak English due to limitations in participation with outcome assessments.
Eligible subjects were enrolled prospectively by trained clinical research coordinators via convenience sampling from years 2010 to 2012. Institutional Review Board approval was obtained at the participating sites. Informed consent was obtained prior to study enrollment. For subjects unable to provide consent due to their injury, consent was obtained from their legally authorized representatives. Subjects were then re-consented, if cognitively able, during the course of their clinical care and/or follow-up visits for continued participation in the study.
2.2. Participants Included in the Current Analysis
As the goal of the current analysis was to evaluate associations between demographic, clinical and injury factors routinely collected upon ED admission for mTBI and 3- and 6-month global functional outcome, subjects from the TRACK-TBI Pilot were retrospectively included if they were aged ≥18 years with an ED admission GCS score of 13–15, without evidence of intracranial injury on ED admission head CT as determined by a blinded, central board-certified neuroradiologist, and completed 3- or 6-month Glasgow Outcome Scale-Extended (GOSE) assessment. All study personnel conducting outcome assessments were trained in-person by a certified neuropsychology coordinator prior to study commencement. The GOSE was completed by patient interview in all cases with the exception of patients physically and/or cognitively debilitated to the extent where they were unable to reliably respond to the assessment (e.g., GOSE 3 or 4, requiring assistance with activities of daily living) in which case the designated caretaker was interviewed. Candidate predictors were included based on their utility as described in large prior studies in mTBI [26,27], as well as capacity to be routinely collected by ED personnel for patients with mTBI. Demographic factors, such as race, ethnicity and education level, and clinical factors, such as prior psychiatric history, were self reported.
2.3. Outcome Measure
The GOSE is a measure of functional disability covering cognition, independence, employability, and social/community participation. It is assessed via structured interview and has been widely used as a standard outcome measure for TBI [28,29]. Subjects are assigned one of eight categories: 1 = dead, 2 = vegetative state, 3 = lower severe disability, 4 = upper severe disability, 5 = lower moderate disability, 6 = upper moderate disability, 7 = lower good recovery, and 8 = upper good recovery (return to baseline function). The GOSE is anchored to baseline functional status; for instance, patients who returned to their baseline level of physical, mental health and/or socioeconomic status were scored as GOSE = 8. Patients with additional physical, cognitive, functional and/or social integration burden after TBI were scored as GOSE < 8, accordingly.
For the current analysis, 3- and 6-month GOSE outcomes were dichotomized to full return to baseline function (GOSE = 8) versus presence of functional deficits (GOSE < 8). By definition, a GOSE of 7 implies deficits in resumption of social activities, problems with social integration, and persistent postconcussive symptoms resulting in functional impairment compared to baseline. Recent studies have suggested that there are distinct neuropsychological differences between the GOSE 7 and GOSE 8 groups, more specifically the prominence of neurocognitive impairment in the GOSE 7 group, suggesting that “good recovery” for GOSE 7 patients may be misleading especially after mTBI [30].
2.4. Statistical Analysis
Descriptive statistics are presented as medians and interquartile range (IQR) for continuous variables and proportions for categorical variables. Group differences were assessed using the Wilcoxon Rank Sum test for continuous variables and Fisher’s exact test for categorical variables. Univariate predictors with p < 0.10 were included in multivariable regression models, with the exception of those with cell counts of 0 on univariate analyses due to limitations on model performance. AOR for multivariable logistic regressions to predict 3- and 6-month functional deficits, and their associated 95% confidence intervals (CI), were reported for each predictor in the regression analyses. Significance was assessed at p < 0.05. All analyses were performed using R [31] version 3.5.1.
3. Results
Overall, there were 271 eligible subjects with GCS 13–15 without evidence of intracranial injury on emergency department head CT. The majority of patients were male (69.4%) and white (73.4%). The median age was 39 years, IQR ranging from 26 to 52 years. The GCS distribution of these patients was: 15 (74.9%), 14 (22.1%), 13 (3.0%). The median years of education was 14 years (IQR: 12–16 years). Psychiatric history was noted as present for 37.3% of the patients. Nearly all patients (99.6%) had sustained a closed TBI. Motor vehicle accidents accounted for 35.8% of total injuries, falls accounted for 27.3%, bicycle accidents accounted for 20.7%, and assaults accounted for 18.1% of injuries. LOC was present in 51.3% of patients and PTA was present in 49.4% of patients. The prevalence of ED hypoxia and ED hypotension was 4.8% and 1.1%, respectively. Of the 271 eligible mTBIs, 211 completed 3-month outcomes and 185 completed 6-month outcomes. Subjects who completed the outcome assessments were similar in baseline characteristics to those who missed the assessments at 3 or 6 month postinjury. At 3-months, 60.2% of subjects (n = 127) and at 6-months, 65.4% (n = 121) had functional deficits (GOSE < 8). The flowchart of included subjects is presented in Figure 1.
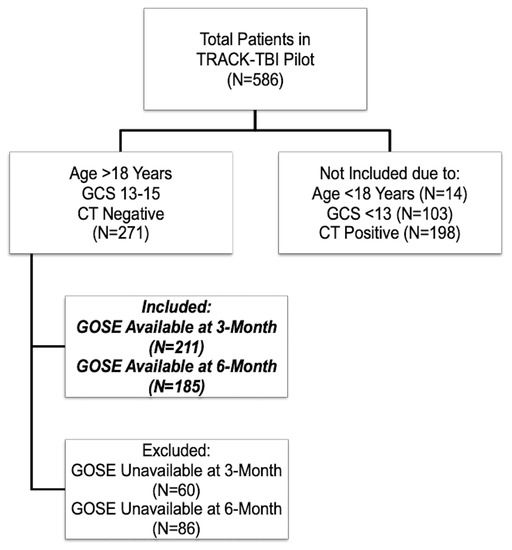
Figure 1.
Flowchart of includedsSubjects.
3.1. Univariate Predictors of 3- and 6-Month GOSE
Based on univariate analysis (Table 1), at the 3-month outcome, years of education were significantly lower in subjects with functional deficits (GOSE < 8) compared with those without functional deficits (GOSE = 8) (median 13 vs. 15, p < 0.01). A greater proportion of subjects with a baseline psychiatric history (e.g., anxiety, depression, post-traumatic stress, bipolar disorder, schizophrenia, sleep disorders, and others) had functional deficits compared with those without a psychiatric history (72% vs. 53%, p < 0.01). Individuals who suffered from assault as the mechanism of injury were more likely to have functional deficits than those with injuries from falls or accidents (81% vs. 56%, p = 0.01). A greater proportion of Asian (86%), American Indian or Alaskan (100%), and Hawaiian or Pacific Islander (100%) participants had functional deficits, compared to Caucasian participants (54%, p = 0.015). A nonsignificant statistical trend was observed for subjects with pre-hospital hypoxia more likely to have functional deficits compared with subjects without pre-hospital hypoxia (100% vs. 58%, p = 0.08). These results persist and remain significant at the 6-month GOSE measurement (Table 1) with the addition that Hispanic subjects had greater functional deficits, compared with non-Hispanic subjects (81% vs. 61%, p = 0.03).

Table 1.
Univariate associations between demographic and clinical characteristics and functional deficits at 3 and 6 months.
No statistical associations were observed of functional deficits on 3- or 6-month GOSE < 8 vs. GOSE = 8 with gender, GCS, anticoagulant use, LOC, PTA, pre-hospital hypotension, urine toxicology screen, blood alcohol level, ED hypotension, or ED hypoxia.
3.2. Multivariable Predictors of 3- and 6-Month GOSE
On multivariable analysis at 3 months (Table 2), significant risk factors associated with worse outcome included fewer years of education (AOR = 0.88 per year increase in education, 95% CI: 0.78–0.998, p = 0.046), the presense of psychiatric history (AOR 2.30, 95% CI: 1.18–4.49, p = 0.015), being Asian, American Indian or Alaskan, Hawaiian, or Pacific Islander (AOR = 12.40, 95% CI: 2.66–57.77, p = 0.006). Assault as the mechanism of injury was also associated with an increased odds of functional deficit (AOR = 3.53, 95% CI: 1.17–10.63, p = 0.025).

Table 2.
Multivariable analysis of Risk Factors for 3- and 6-month functional deficits (GOSE < 8).
Risk factors for functional deficits at 6 months postinjury were similar (Table 2). Lower education and the presense of psychatric history were associated with higher odds of worse outcome. Hispanic ethnicity was also a predictor for 6-month deficits (AOR = 3.48, 95% CI: 1.29–9.37, p = 0.01).
4. Discussion
This study is one of very few to examine the association of baseline clinical factors with postinjury functional outcomes in a cohort of mTBI patients presenting to EDs of Level 1 trauma centers with a negative head CT. Education level, pre-injury psychiatric history, assault as the mechanism of injury, Asian race and other minorities (American Indian or Alaskan, Hawaiian or Pacific Islander), and Hispanic ethnicity were predictive of heightened risk for functional deficits at three or six months postinjury [4,6,20,26,32,33,34,35,36,37,38]. Although there is a pervasive belief that all TBI will get better with time [39,40]. We found that the proportion of participants with functional deficits did not decrease from 3 to 6 months in patients who returned for follow-up. Therefore, modification of existing ED assessment and treatment guidelines to highlight the needs of these patients and provide appropriate referral to follow-up at earlier time points could prove beneficial.
Our finding that over 60% of patients with CT-negative, GCS 13–15 TBI have functional deficits at 3 and 6 months aligns with findings over the past decade that the proportion of deficits in mTBI patients is higher than traditionally appreciated [7,41]. Notably, commonly cited risk factors for poor outcome in severe TBI such as pre-hospital hypoxia and hypotension [42,43,44,45], were rarely seen in this cohort of patients who inherently have “milder” injuries, and factors used in ED guidelines such as LOC and PTA [46] were not associated with outcome. However, a negative CT with a GCS of 13–15 does not necessarily portend that the patient will recover to baseline after a short period of cognitive rest—in fact, a subset of these patients likely require increased surveillance and strict return to activity precautions. Our finding that GOSE deficits associated with a pre-injury history of psychiatric conditions were maintained at three and six months suggests that longer follow-up both in clinical settings and in research design may be critical in order to refer for treatment and obtain accurate outcome assessments, respectively.
Despite the evidence of persistent symptoms months to years postinjury [47,48,49,50] a large proportion of mTBI patients do not receive follow-up care. In a study of 831 mTBI patients, only 42% received TBI educational materials at ED discharge [51] despite evidence that patient educational information and targeted follow-up have been shown to reduce post-concussive symptoms [52,53,54,55]. In this same cohort, nearly 40% of CT-positive, mTBI patients had not seen a physician by three months following injury [51]. Even in those with persistent symptoms, only 41% were seen by a practitioner by two weeks and 44% by three months [51].
The ED physician is uniquely positioned to stratify patients that are in greater need of support and resources given that the majority of the triage, work-up, and injury education of mTBIs occur in the ED, and the vast majority of mTBI patients are discharged directly from the ED. Currently, patient education level and pre-injury psychiatric history information are not routinely collected in the ED. Mechanism of injury, race, and ethnicity are frequently collected in the ED; however, these factors rarely inform decision making after TBI. Including the assessment of these evidence-based clinical factors in the guidelines for ED management of mTBI would trigger appropriate referral for the patients most likely to develop persistent sequelae and demonstrate the justification for more accessible services.
Resources such as social work, follow-up referrals, and patient education regarding mTBI are sparse compared with those available for moderate and severe TBI [56,57]. One potential method to improve follow-up is to ensure ED social workers discuss follow-up with patients prior to discharge [58]. While treatment for mTBI remains variable, evidence has shown that cognitive rehabilitation, psychosocial support, and consistent follow-up with specialists can reduce symptomatology, decrease the time needed to return to work or baseline social integration, and improve the quality of life [59,60,61,62]. Utilizing existing infrastructure, such as patients’ primary care physicians, to improve follow-up when the brain is most receptive to rewiring [63,64], could help alleviate both post-concussive symptoms and observed long-term effects, such as neurocognitive decline [65,66].
Limitations
We recognize several limitations to our analysis. We analyzed mTBI patients from a convenience sample that may not be representative of the entire mTBI population. We also recruited from U.S. Level I trauma centers, which predominantly care for patients with more severe injuries and a greater likelihood of comorbidities. We focused on investigating variables routinely collected as part of ED admission that may be of value in overall functional recovery, and did not analyze for trajectories of recovery, which would be a prime topic for future investigations. The number of participants in non-White racial groups is small. Variables such as pre-hospital hypoxia also suffered from low cell counts on univariate analyses and were not included in multivariable models. Therefore, associated odds ratios should be interpreted cautiously, and will benefit from confirmation in future studies of ED patient populations with mTBI. As with all trauma studies, we were limited to patients who returned for follow-up, and attrition bias limits the generalizability of our study. Toxicology and blood alcohol level data were subject to institutional limitations and were not routinely drawn on all mTBI patients, which limits generalizability.
As the TRACK-TBI Pilot study variables were based on version 1 of the NINDS CDEs, insurance status, readmission, and granular post-discharge data were not available. Accordingly, post-acute interventions such as referrals and/or rehabilitation were not available. As we analyzed patients who were discharged from the ED, polytrauma was not categorized by the trauma registry, which is only available for admitted patients; however, this is likely a minor contribution, as patients’ peripheral injuries were deemed safe for discharge directly from the ED. Whether patients received TBI education materials is another data point informative for outcomes analysis [51] and was not tracked as part of the TRACK-TBI Pilot. These variables are being collected as part of the ongoing 18-center Transforming Research and Clinical Knowledge in Traumatic Brain Injury study [67] using version 2 of the NINDS TBI CDEs [68].
Regarding future directions, facilitating education and managing psychiatric issues may help reduce exposure and improve adherence to mTBI follow-up [51,52]. Comparison to military studies will also be of use, as the presence of psychiatric comorbidities, e.g., post-traumatic stress disorder (PTSD), consistently predicted poorer post-deployment outcome in deployed veterans compared to veteran controls, while demographics and injury mechanisms were less important [69,70,71]. This may be related to factors of intensity, and the frequency of trauma in military compared to civilian populations, as well as measures of resilience [72], which are ongoing topics of research. The implementation of qualified biomarkers sensitive to the presence of CT abnormalities can also improve diagnosis and the need to triage to follow-up for ED patients with mTBI [73,74].
5. Conclusions
In summary, it is our responsibility to implement compensatory resources and services for the over 65% of CT-negative mTBI patients who may predictably have functional deficits in the three to six months postinjury. The predictors of education, assault as the mechanism of injury, minority races, and the presence of baseline psychiatric disorders can be routinely collected at ED admission without significant burden to the provider or patient. These patients may benefit from education, follow-up resources, necessary referrals and/or early surveillance in clinic. Education, psychiatric history, race and ethnicity, along with mechanism of injury, should be considered for integration into current ED mTBI protocols.
Author Contributions
D.Y.M. was responsible for study concept and design, oversaw the acquisition of data and analysis and interpretation of the data, and drafted the manuscript. J.K.Y. assisted with study concept and design and drafted the manuscript. X.S. and S.J. were responsible for data analysis. C.G.S. and N.A.C. assisted with drafting the manuscript. G.T.M. contributed to the critical revision of the manuscript for important intellectual content. All authors have read and agreed to the published version of the manuscript.
Funding
This work was supported by the National Institute of Neurological Disorders and Stroke [grant numbers 1RC2NS069409-01, 3RC2NS069409-02S1, 5RC2NS069409-02, 1U01NS086090-01, 3U01NS086090-02S1, 3U01NS086090-02S2, 3U01NS086090-03S1, 5U01NS086090-02, and 5U01NS086090-03] and the US Department of Defense [grant numbers W81XWH-13-1-0441 and W81XWH-14-2-0176] for GTM. The funders had no role in the study design; data collection, analysis, and interpretation; writing of the report; and the decision to submit the article for publication.
Acknowledgments
Amy J. Markowitz, J.D. provided editorial support. The authors would like to thank the following contributors to the development of the TRACK-TBI database and repositories by organization and in alphabetical order by last name: One Mind for Research: General Peter Chiarelli, U.S. Army (Ret.), Garen Staglin, MBA; QuesGen Systems, Inc.: Vibeke Brinck, MS, Michael Jarrett, MBA; Thomson Reuters: Sirimon O’Charoen.
Conflicts of Interest
The authors declare no conflict of interest.
TRACK-TBI INVESTIGATORS (to be indexed under “Collaborators” in PubMed)
Shelly R. Cooper, PhD (Department of Psychology, Washington University in St. Louis, St. Louis, MO, USA); Kristen Dams-O’Connor (Department of Rehabilitation Medicine, Icahn School of Medicine at Mount Sinai, New York, NY, USA); Wayne A. Gordon, PhD (Department of Rehabilitation Medicine, Icahn School of Medicine at Mount Sinai, New York, NY, USA); Allison J. Hricik, MS (Department of Neurosurgery, University of Pittsburgh Medical Center, Pittsburgh, PA, USA); Hester F. Lingsma, PhD (Department of Public Health, Erasmus Medical Center, Rotterdam, The Netherlands); Andrew I. R. Maas, MD, PhD (Department of Neurosurgery, University Hospital Antwerp, Edegem, Belgium); David K. Menon, MD, PhD (Division of Anaesthesia, University of Cambridge, Cambridge, United Kingdom); Diane J. Morabito, RN, MPH (Department of Neurosurgery, University of California San Francisco, San Francisco, CA, USA); Pratik Mukherjee, MD, PhD (Department of Radiology and Biomedical Imaging, University of California San Francisco, San Francisco, CA, USA); David O. Okonkwo, MD, PhD (Department of Neurosurgery, University of Pittsburgh Medical Center, Pittsburgh, PA, USA); Ava M. Puccio, RN, PhD (Department of Neurosurgery, University of Pittsburgh Medical Center, Pittsburgh, PA, USA); David M. Schnyer, PhD (Department of Psychology, University of Texas, Austin, TX, USA); Sabrina R. Taylor, PhD (Department of Neurosurgery, University of California San Francisco, San Francisco, CA, USA); Alex B. Valadka, MD (Department of Neurosurgery, Virginia Commonwealth University, Richmond, VA, USA); Mary J. Vassar, RN, MS (Department of Neurosurgery, University of California San Francisco, San Francisco, CA, USA); Esther L. Yuh, MD, PhD (Department of Radiology and Biomedical Imaging, University of California San Francisco, San Francisco, CA, USA).
References
- Marincowitz, C.; Lecky, F.E.; Townend, W.; Borakati, A.; Fabbri, A.; Sheldon, T.A. The Risk of Deterioration in GCS13-15 Patients with Traumatic Brain Injury Identified by Computed Tomography Imaging: A Systematic Review and Meta-Analysis. J. Neurotrauma 2018, 35, 703–718. [Google Scholar] [CrossRef] [PubMed]
- Cancelliere, C.; Coronado, V.G.; Taylor, C.A.; Xu, L. Epidemiology of Isolated Versus Nonisolated Mild Traumatic Brain Injury Treated in Emergency Departments in the United States, 2006–2012: Sociodemographic Characteristics. J. Head Trauma Rehabil. 2017, 32, E37–E46. [Google Scholar] [CrossRef] [PubMed]
- Selassie, A.W.; Fakhry, S.M.; Ford, D.W. Population-based study of the risk of in-hospital death after traumatic brain injury: The role of sepsis. J. Trauma 2011, 71, 1226–1234. [Google Scholar] [CrossRef] [PubMed]
- Stein, M.B.; Jain, S.; Giacino, J.T.; Levin, H.; Dikmen, S.; Nelson, L.D.; Vassar, M.J.; Okonkwo, D.O.; Diaz-Arrastia, R.; Robertson, C.S.; et al. Risk of Posttraumatic Stress Disorder and Major Depression in Civilian Patients After Mild Traumatic Brain Injury: A TRACK-TBI Study. JAMA Psychiatry 2019, 76, 249–258. [Google Scholar] [CrossRef] [PubMed]
- Carroll, L.J.; Cassidy, J.D.; Cancelliere, C.; Cote, P.; Hincapie, C.A.; Kristman, V.L.; Holm, L.W.; Borg, J.; Nygren-de Boussard, C.; Hartvigsen, J.; et al. Systematic Review of the Prognosis after Mild Traumatic Brain Injury in Adults: Cognitive, Psychiatric, and Mortality Outcomes: Results of the International Collaboration on Mild Traumatic Brain Injury Prognosis. Arch. Phys. Med. Rehabil. 2014, 95, S152–S173. [Google Scholar] [CrossRef]
- Cnossen, M.C.; Winkler, E.A.; Yue, J.K.; Okonkwo, D.O.; Valadka, A.; Steyerberg, E.W.; Lingsma, H.; Manley, G.T.; the TRACK-TBI Investigators. Development of a Prediction Model for Post-Concussive Symptoms following Mild Traumatic Brain Injury: A TRACK-TBI Pilot Study. J. Neurotrauma 2017, 34, 2396–2409. [Google Scholar] [CrossRef]
- McMahon, P.; Hricik, A.; Yue, J.K.; Puccio, A.M.; Inoue, T.; Lingsma, H.F.; Beers, S.R.; Gordon, W.A.; Valadka, A.B.; Manley, G.T.; et al. Symptomatology and Functional Outcome in Mild Traumatic Brain Injury: Results from the Prospective TRACK-TBI study. J. Neurotrauma 2014, 31, 26–33. [Google Scholar] [CrossRef]
- Kraus, J.F.; Hsu, P.; Schafer, K.; Afifi, A.A. Sustained Outcomes Following Mild Traumatic Brain Injury: Results of a Five-Emergency Department Longitudinal Study. Brain Inj. 2014, 28, 1248–1256. [Google Scholar] [CrossRef]
- Ratcliff, J.J.; Adeoye, O.; Lindsell, C.J.; Hart, K.W.; Pancioli, A.; McMullan, J.T.; Yue, J.K.; Nishijima, D.K.; Gordon, W.A.; Valadka, A.B.; et al. ED Disposition of the Glasgow Coma Scale 13 to 15 Traumatic Brain Injury Patient: Analysis of the Transforming Research and Clinical Knowledge in TBI study. Am. J. Emerg. Med. 2014, 32, 844–850. [Google Scholar] [CrossRef]
- Hsia, R.Y.; Markowitz, A.J.; Lin, F.; Guo, J.; Madhok, D.Y.; Manley, G.T. Ten-Year Trends in Traumatic Brain Injury: A Retrospective Cohort Study of California Emergency Department and Hospital Revisits and Readmissions. BMJ Open 2018, 8, e022297. [Google Scholar] [CrossRef]
- Ganti, L.; Conroy, L.M.; Bodhit, A.; Daneshvar, Y.; Patel, P.S.; Ayala, S.; Kuchibhotla, S.; Hatchitt, K.; Pulvino, C.; Peters, K.R.; et al. Understanding Why Patients Return to the Emergency Department after Mild Traumatic Brain Injury within 72 Hours. West J. Emerg. Med. 2015, 16, 481–485. [Google Scholar] [CrossRef] [PubMed]
- Pandor, A.; Goodacre, S.; Harnan, S.; Holmes, M.; Pickering, A.; Fitzgerald, P.; Rees, A.; Stevenson, M. Diagnostic Management Strategies for Adults and Children with Minor Head Injury: A Systematic Review and an Economic Evaluation. Health Technol. Assess. 2011, 15, 1–202. [Google Scholar] [CrossRef] [PubMed]
- Yue, J.K.; Vassar, M.J.; Lingsma, H.F.; Cooper, S.R.; Okonkwo, D.O.; Valadka, A.B.; Gordon, W.A.; Maas, A.I.; Mukherjee, P.; Yuh, E.L.; et al. Transforming Research and Clinical Knowledge in Traumatic Brain Injury Pilot: Multicenter Implementation of the Common Data Elements for Traumatic Brain Injury. J. Neurotrauma 2013, 30, 1831–1844. [Google Scholar] [CrossRef] [PubMed]
- Si, B.; Dumkrieger, G.; Wu, T.; Zafonte, R.; Valadka, A.B.; Okonkwo, D.O.; Manley, G.T.; Wang, L.; Dodick, D.W.; Schwedt, T.J. Sub-classifying Patients with Mild Traumatic Brain Injury: A Clustering Approach Based on Baseline Clinical Characteristics and 90-day and 180-day Outcomes. PLoS ONE 2018, 13, e0198741. [Google Scholar] [CrossRef]
- Barbosa, R.R.; Jawa, R.; Watters, J.M.; Knight, J.C.; Kerwin, A.J.; Winston, E.S.; Barraco, R.D.; Tucker, B.; Bardes, J.M.; Rowell, S.E. Evaluation and Management of Mild Traumatic Brain Injury: An Eastern Association for the Surgery of Trauma Practice Management Guideline. J. Trauma Acute Care Surg. 2012, 73, S307–S314. [Google Scholar] [CrossRef]
- Silverberg, N.D.; Gardner, A.J.; Brubacher, J.R.; Panenka, W.J.; Li, J.J.; Iverson, G.L. Systematic Review of Multivariable Prognostic Models for Mild Traumatic Brain Injury. J. Neurotrauma 2015, 32, 517–526. [Google Scholar] [CrossRef]
- Tavender, E.J.; Bosch, M.; Green, S.; O’Connor, D.; Pitt, V.; Phillips, K.; Bragge, P.; Gruen, R.L. Quality and Consistency of Guidelines for the Management of Mild Traumatic Brain Injury in the Emergency Department. Acad. Emerg. Med. 2011, 18, 880–889. [Google Scholar] [CrossRef]
- Bossers, S.M.; Pol, K.M.; Ophuis, E.O.; Jacobs, B.; Visser, M.C.; Loer, S.A.; Boer, C.; van der Naalt, J.; Schober, P. Discrepancy between the Initial Assessment of Injury Severity and Post Hoc Determination of Injury Severity in Patients with Apparently Mild Traumatic Brain Injury: A Retrospective Multicenter Cohort Analysis. Eur. J. Trauma Emerg. Surg. 2018, 44, 889–896. [Google Scholar] [CrossRef]
- Theadom, A.; Starkey, N.; Barker-Collo, S.; Jones, K.; Ameratunga, S.; Feigin, V.; BIONIC4you Research Group. Population-Based Cohort Study of the Impacts of Mild Traumatic Brain Injury in Adults Four Years Post-Injury. PLoS ONE 2018, 13, e0191655. [Google Scholar] [CrossRef]
- van der Naalt, J.; Timmerman, M.E.; de Koning, M.E.; van der Horn, H.J.; Scheenen, M.E.; Jacobs, B.; Hageman, G.; Yilmaz, T.; Roks, G.; Spikman, J.M. Early Predictors of Outcome after Mild Traumatic Brain Injury (UPFRONT): An Observational Cohort Study. Lancet Neurol. 2017, 16, 532–540. [Google Scholar] [CrossRef]
- de Koning, M.E.; Scheenen, M.E.; van der Horn, H.J.; Hageman, G.; Roks, G.; Spikman, J.M.; van der Naalt, J. Non-Hospitalized Patients with Mild Traumatic Brain Injury: The Forgotten Minority. J. Neurotrauma 2017, 34, 257–261. [Google Scholar] [CrossRef] [PubMed]
- Duhaime, A.C.; Gean, A.D.; Haacke, E.M.; Hicks, R.; Wintermark, M.; Mukherjee, P.; Brody, D.; Latour, L.; Riedy, G.; Common Data Elements Neuroimaging Working Group Members; et al. Common Data Elements in Radiologic Imaging of Traumatic Brain Injury. Arch. Phys. Med. Rehabil. 2010, 91, 1661–1666. [Google Scholar] [CrossRef] [PubMed]
- Maas, A.I.; Harrison-Felix, C.L.; Menon, D.; Adelson, P.D.; Balkin, T.; Bullock, R.; Engel, D.C.; Gordon, W.; Orman, J.L.; Lew, H.L.; et al. Common Data Elements for Traumatic Brain Injury: Recommendations from the Interagency Working Group on Demographics and Clinical Assessment. Arch. Phys. Med. Rehabil. 2010, 91, 1641–1649. [Google Scholar] [CrossRef] [PubMed]
- Manley, G.T.; Diaz-Arrastia, R.; Brophy, M.; Engel, D.; Goodman, C.; Gwinn, K.; Veenstra, T.D.; Ling, G.; Ottens, A.K.; Tortella, F.; et al. Common Data Elements for Traumatic Brain Injury: Recommendations from the Biospecimens and Biomarkers Working Group. Arch. Phys. Med. Rehabil. 2010, 91, 1667–1672. [Google Scholar] [CrossRef]
- Wilde, E.A.; Whiteneck, G.G.; Bogner, J.; Bushnik, T.; Cifu, D.X.; Dikmen, S.; French, L.; Giacino, J.T.; Hart, T.; Malec, J.F.; et al. Recommendations for the Use of Common Outcome Measures in Traumatic Brain Injury Research. Arch. Phys. Med. Rehabil. 2010, 91, 1650–1660.e17. [Google Scholar] [CrossRef]
- Stulemeijer, M.; van der Werf, S.; Borm, G.F.; Vos, P.E. Early Prediction of Favourable Recovery 6 Months after Mild Traumatic Brain Injury. J. Neurol. Neurosurg. Psychiatry 2008, 79, 936–942. [Google Scholar] [CrossRef]
- Jacobs, B.; Beems, T.; Stulemeijer, M.; van Vugt, A.B.; van der Vliet, T.M.; Borm, G.F.; Vos, P.E. Outcome prediction in mild traumatic brain injury: Age and clinical variables are stronger predictors than CT abnormalities. J. Neurotrauma 2010, 27, 655–668. [Google Scholar] [CrossRef]
- Teasdale, G.M.; Pettigrew, L.E.; Wilson, J.T.; Murray, G.; Jennett, B. Analyzing Outcome of Treatment of Severe Head Injury: A Review and Update on Advancing the Use of the Glasgow Outcome Scale. J. Neurotrauma 1998, 15, 587–597. [Google Scholar] [CrossRef]
- Wilson, J.T.; Pettigrew, L.E.; Teasdale, G.M. Structured Interviews for the Glasgow Outcome Scale and the extended Glasgow Outcome Scale: Guidelines for Their Use. J. Neurotrauma 1998, 15, 573–585. [Google Scholar] [CrossRef]
- Nelson, L.D.; Ranson, J.; Ferguson, A.R.; Giacino, J.; Okonkwo, D.O.; Valadka, A.; Manley, G.; McCrea, M. Validating Multidimensional Outcome Assessment Using the TBI Common Data Elements: An Analysis of the TRACK-TBI Pilot Sample. J. Neurotrauma 2017, 34, 3158–3172. [Google Scholar] [CrossRef]
- The R Project for Statistical Computing. Available online: https://www.r-project.org (accessed on 20 March 2020).
- Chiang, C.C.; Guo, S.E.; Huang, K.C.; Lee, B.O.; Fan, J.Y. Trajectories And Associated Factors of Quality of Life, Global Outcome, and Post-Concussion Symptoms in the First Year Following Mild Traumatic Brain Injury. Qual. Life Res. 2016, 25, 2009–2019. [Google Scholar] [CrossRef] [PubMed]
- Voormolen, D.C.; Cnossen, M.C.; Polinder, S.; von Steinbuechel, N.; Vos, P.E.; Haagsma, J.A. Divergent Classification Methods of Post-Concussion Syndrome after Mild Traumatic Brain Injury: Prevalence Rates, Risk Factors, and Functional Outcome. J. Neurotrauma 2018, 35, 1233–1241. [Google Scholar] [CrossRef] [PubMed]
- McCauley, S.R.; Wilde, E.A.; Barnes, A.; Hanten, G.; Hunter, J.V.; Levin, H.S.; Smith, D.H. Patterns of Early Emotional and Neuropsychological Sequelae after Mild Traumatic Brain Injury. J. Neurotrauma 2014, 31, 914–925. [Google Scholar] [CrossRef] [PubMed]
- Arango-Lasprilla, J.C.; Rosenthal, M.; Deluca, J.; Komaroff, E.; Sherer, M.; Cifu, D.; Hanks, R. Traumatic Brain Injury and Functional Outcomes: Does Minority Status Matter? Brain Inj. 2007, 21, 701–708. [Google Scholar] [CrossRef] [PubMed]
- Arango-Lasprilla, J.C.; Rosenthal, M.; Deluca, J.; Cifu, D.X.; Hanks, R.; Komaroff, E. Functional Outcomes from Inpatient Rehabilitation after Traumatic Brain Injury: How do Hispanics Fare? Arch. Phys. Med. Rehabil. 2007, 88, 11–18. [Google Scholar] [CrossRef]
- Shafi, S.; Marquez de la Plata, C.; Diaz-Arrastia, R.; Shipman, K.; Carlile, M.; Frankel, H.; Parks, J.; Gentilello, L.M. Racial Disparities in Long-Term Functional Outcome After Traumatic Brain Injury. J. Trauma 2007, 63, 1263–1268. [Google Scholar] [CrossRef]
- Marquez de la Plata, C.; Hewlitt, M.; de Oliveira, A.; Hudak, A.; Harper, C.; Shafi, S.; Diaz-Arrastia, R. Ethnic Differences in Rehabilitation Placement and Outcome after TBI. J. Head Trauma Rehabil. 2007, 22, 113–121. [Google Scholar] [CrossRef]
- Maas, A.I.R.; Menon, D.K.; Adelson, P.D.; Andelic, N.; Bell, M.J.; Belli, A.; Bragge, P.; Brazinova, A.; Buki, A.; Chesnut, R.M.; et al. Traumatic Brain Injury: Integrated Approaches to Improve Prevention, Clinical Care, and Research. Lancet Neurol. 2017, 16, 987–1048. [Google Scholar] [CrossRef]
- Levin, H.S.; Diaz-Arrastia, R.R. Diagnosis, Prognosis, and Clinical Management of Mild Traumatic Brain Injury. Lancet Neurol. 2015, 14, 506–517. [Google Scholar] [CrossRef]
- Lingsma, H.F.; Yue, J.K.; Maas, A.I.; Steyerberg, E.W.; Manley, G.T.; the TRACK-TBI Investigators. Outcome Prediction after Mild and Complicated Mild Traumatic Brain Injury: External Validation of Existing Models and Identification of New Predictors Using The TRACK-TBI Pilot Study. J. Neurotrauma 2015, 32, 83–94. [Google Scholar] [CrossRef]
- Spaite, D.W.; Hu, C.; Bobrow, B.J.; Chikani, V.; Barnhart, B.; Gaither, J.B.; Denninghoff, K.R.; Adelson, P.D.; Keim, S.M.; Viscusi, C.; et al. The Effect of Combined Out-of-Hospital Hypotension and Hypoxia on Mortality in Major Traumatic Brain Injury. Ann. Emerg. Med. 2017, 69, 62–72. [Google Scholar] [CrossRef] [PubMed]
- Spaite, D.W.; Hu, C.; Bobrow, B.J.; Chikani, V.; Sherrill, D.; Barnhart, B.; Gaither, J.B.; Denninghoff, K.R.; Viscusi, C.; Mullins, T.; et al. Mortality and Prehospital Blood Pressure in Patients with Major Traumatic Brain Injury: Implications for the Hypotension Threshold. JAMA Surg. 2017, 152, 360–368. [Google Scholar] [CrossRef] [PubMed]
- Fearnside, M.R.; Cook, R.J.; McDougall, P.; McNeil, R.J. The Westmead Head Injury Project Outcome in Severe Head Injury. A Comparative Analysis of Pre-Hospital, Clinical and CT variables. Br. J. Neurosurg. 1993, 7, 267–279. [Google Scholar] [CrossRef] [PubMed]
- Hill, D.A.; Abraham, K.J.; West, R.H. Factors Affecting Outcome in the Resuscitation of Severely Injured Patients. Aust. N. Z. J. Surg. 1993, 63, 604–609. [Google Scholar] [CrossRef] [PubMed]
- Jagoda, A.S.; Bazarian, J.J.; Bruns, J.J.; Cantrill, S.V.; Gean, A.D.; Howard, P.K.; Ghajar, J.; Riggio, S.; Wright, D.W.; Wears, R.L.; et al. Clinical Policy: Neuroimaging and Decisionmaking in Adult Mild Traumatic Brain Injury in the Acute Setting. Ann. Emerg. Med. 2008, 52, 714–748. [Google Scholar] [CrossRef] [PubMed]
- Rabinowitz, A.R.; Li, X.; McCauley, S.R.; Wilde, E.A.; Barnes, A.; Hanten, G.; Mendez, D.; McCarthy, J.J.; Levin, H.S. Prevalence and Predictors of Poor Recovery from Mild Traumatic Brain Injury. J. Neurotrauma 2015, 32, 1488–1496. [Google Scholar] [CrossRef] [PubMed]
- Dikmen, S.; Machamer, J.; Temkin, N. Mild Traumatic Brain Injury: Longitudinal Study of Cognition, Functional Status, and Post-Traumatic Symptoms. J. Neurotrauma 2017, 34, 1524–1530. [Google Scholar] [CrossRef]
- McCrea, M.A.; Nelson, L.D.; Guskiewicz, K. Diagnosis and Management of Acute Concussion. Phys. Med. Rehabil. Clin. N. Am. 2017, 28, 271–286. [Google Scholar] [CrossRef]
- Hiploylee, C.; Dufort, P.A.; Davis, H.S.; Wennberg, R.A.; Tartaglia, M.C.; Mikulis, D.; Hazrati, L.N.; Tator, C.H. Longitudinal Study of Postconcussion Syndrome: Not Everyone Recovers. J. Neurotrauma 2017, 34, 1511–1523. [Google Scholar] [CrossRef]
- Seabury, S.A.; Gaudette, E.; Goldman, D.P.; Markowitz, A.J.; Brooks, J.; McCrea, M.A.; Okonkwo, D.O.; Manley, G.T.; the TRACK-TBI Investigators. Assessment of Follow-up Care after Emergency Department Presentation for Mild Traumatic Brain Injury and Concussion: Results from the TRACK-TBI Study. JAMA Netw. Open 2018, 1, e180210. [Google Scholar] [CrossRef]
- de Koning, M.E.; Scheenen, M.E.; van der Horn, H.J.; Hageman, G.; Roks, G.; Yilmaz, T.; Spikman, J.M.; van der Naalt, J. Outpatient Follow-Up After Mild Traumatic Brain Injury: Results of the UPFRONT-study. Brain Inj. 2017, 31, 1102–1108. [Google Scholar] [CrossRef] [PubMed]
- Janak, J.C.; Cooper, D.B.; Bowles, A.O.; Alamgir, A.H.; Cooper, S.P.; Gabriel, K.P.; Perez, A.; Orman, J.A. Completion of Multidisciplinary Treatment for Persistent Postconcussive Symptoms Is Associated With Reduced Symptom Burden. J. Head Trauma Rehabil. 2017, 32, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Nygren-de Boussard, C.; Holm, L.W.; Cancelliere, C.; Godbolt, A.K.; Boyle, E.; Stalnacke, B.M.; Hincapie, C.A.; Cassidy, J.D.; Borg, J. Nonsurgical Interventions after Mild Traumatic Brain Injury: A Systematic Review. Results of the International Collaboration on Mild Traumatic Brain Injury Prognosis. Arch. Phys. Med. Rehabil. 2014, 95, S257–S264. [Google Scholar] [CrossRef] [PubMed]
- Ponsford, J.; Willmott, C.; Rothwell, A.; Cameron, P.; Ayton, G.; Nelms, R.; Curran, C.; Ng, K. Impact of Early Intervention on Outcome after Mild Traumatic Brain Injury in Children. Pediatrics 2001, 108, 1297–1303. [Google Scholar] [CrossRef] [PubMed]
- Bazarian, J.J.; McClung, J.; Cheng, Y.T.; Flesher, W.; Schneider, S.M. Emergency Department Management of Mild Traumatic Brain Injury in the USA. Emerg. Med. J. 2005, 22, 473–477. [Google Scholar] [CrossRef] [PubMed]
- Stern, R.A.; Seichepine, D.; Tschoe, C.; Fritts, N.G.; Alosco, M.L.; Berkowitz, O.; Burke, P.; Howland, J.; Olshaker, J.; Cantu, R.C.; et al. Concussion Care Practices and Utilization of Evidence-Based Guidelines in the Evaluation and Management of Concussion: A Survey of New England Emergency Departments. J. Neurotrauma 2017, 34, 861–868. [Google Scholar] [CrossRef]
- Einstadter, D.; Cebul, R.D.; Franta, P.R. Effect of a Nurse Case Manager on Postdischarge Follow-Up. J. Gen. Intern. Med. 1996, 11, 684–688. [Google Scholar] [CrossRef]
- Cicerone, K.D.; Langenbahn, D.M.; Braden, C.; Malec, J.F.; Kalmar, K.; Fraas, M.; Felicetti, T.; Laatsch, L.; Harley, J.P.; Bergquist, T.; et al. Evidence-Based Cognitive Rehabilitation: Updated Review of the Literature from 2003 through 2008. Arch. Phys. Med. Rehabil. 2011, 92, 519–530. [Google Scholar] [CrossRef]
- Ponsford, J.L.; Downing, M.G.; Olver, J.; Ponsford, M.; Acher, R.; Carty, M.; Spitz, G. Longitudinal Follow-Up of Patients with Traumatic Brain Injury: Outcome at Two, Five, and Ten Years Post-Injury. J. Neurotrauma 2014, 31, 64–77. [Google Scholar] [CrossRef]
- Shames, J.; Treger, I.; Ring, H.; Giaquinto, S. Return to Work Following Traumatic Brain Injury: Trends and Challenges. Disabil. Rehabil. 2007, 29, 1387–1395. [Google Scholar] [CrossRef]
- Tiersky, L.A.; Anselmi, V.; Johnston, M.V.; Kurtyka, J.; Roosen, E.; Schwartz, T.; Deluca, J. A Trial of Neuropsychologic Rehabilitation in Mild-Spectrum Traumatic Brain Injury. Arch. Phys. Med. Rehabil. 2005, 86, 1565–1574. [Google Scholar] [CrossRef] [PubMed]
- Nahmani, M.; Turrigiano, G.G. Adult Cortical Plasticity Following Injury: Recapitulation of Critical Period Mechanisms? Neuroscience 2014, 283, 4–16. [Google Scholar] [CrossRef] [PubMed]
- Nudo, R.J. Recovery after Brain Injury: Mechanisms and Principles. Front. Hum. Neurosci. 2013, 7, 887. [Google Scholar] [CrossRef]
- Gardner, R.C.; Yaffe, K. Epidemiology of Mild Traumatic Brain Injury and Neurodegenerative Disease. Mol. Cell Neurosci. 2015, 66, 75–80. [Google Scholar] [CrossRef]
- Lee, Y.K.; Hou, S.W.; Lee, C.C.; Hsu, C.Y.; Huang, Y.S.; Su, Y.C. Increased Risk of Dementia in Patients with Mild Traumatic Brain Injury: A Nationwide Cohort Study. PLoS ONE 2013, 8, e62422. [Google Scholar] [CrossRef] [PubMed]
- Transforming Research and Clinical Knowledge in TBI. Available online: https://tracktbi.ucsf.edu/ (accessed on 20 March 2020).
- Hicks, R.; Giacino, J.; Harrison-Felix, C.; Manley, G.; Valadka, A.; Wilde, E.A. Progress in Developing Common Data Elements For Traumatic Brain Injury Research: Version Two—The End of the Beginning. J. Neurotrauma 2013, 30, 1852–1861. [Google Scholar] [CrossRef]
- Yurgil, K.A.; Barbauskas, D.A.; Vasterling, J.J.; Nievergelt, C.M.; Larson, G.E.; Schork, N.J.; Litz, B.T.; Nash, W.P.; Baker, D.G.; Marine Resiliency Study Team. Association between traumatic brain injury and risk of posttraumatic stress disorder in active-duty Marines. JAMA Psychiatry 2014, 71, 149–157. [Google Scholar] [CrossRef]
- Combs, H.L.; Berry, D.T.; Pape, T.; Babcock-Parziale, J.; Smith, B.; Schleenbaker, R.; Shandera-Ochsner, A.; Harp, J.P.; High, W.M., Jr. The Effects of Mild Traumatic Brain Injury, Post-Traumatic Stress Disorder, and Combined Mild Traumatic Brain Injury/Post-Traumatic Stress Disorder on Returning Veterans. J. Neurotrauma 2015, 32, 956–966. [Google Scholar] [CrossRef]
- Cooper, D.B.; Vanderploeg, R.D.; Armistead-Jehle, P.; Lewis, J.D.; Bowles, A.O. Factors associated with neurocognitive performance in OIF/OEF servicemembers with postconcussive complaints in postdeployment clinical settings. J. Rehabil. Res. Dev. 2014, 51, 1023–1034. [Google Scholar] [CrossRef]
- Elliott, T.R.; Hsiao, Y.Y.; Kimbrel, N.A.; Meyer, E.; DeBeer, B.B.; Gulliver, S.B.; Kwok, O.M.; Morissette, S.B. Resilience and Traumatic Brain Injury among Iraq/Afghanistan War Veterans: Differential Patterns of Adjustment and Quality of Life. J. Clin. Psychol. 2017, 73, 1160–1178. [Google Scholar] [CrossRef]
- Wang, K.K.; Yang, Z.; Zhu, T.; Shi, Y.; Rubenstein, R.; Tyndall, J.A.; Manley, G.T. An update on diagnostic and prognostic biomarkers for traumatic brain injury. Expert Rev. Mol. Diagn. 2018, 18, 165–180. [Google Scholar] [CrossRef] [PubMed]
- Yue, J.K.; Yuh, E.L.; Korley, F.K.; Sun, X.; Puffer, R.C.; Deng, H.; Choy, W.; Chandra, A.; Taylor, S.R.; Ferguson, A.R.; et al. Association between plasma GFAP concentrations and MRI abnormalities in patients with CT-negative traumatic brain injury in the TRACK-TBI cohort: A prospective multicentre study. Lancet Neurol. 2019, 18, 953–961. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).