The Modulation of Cognitive Performance with Transcranial Alternating Current Stimulation: A Systematic Review of Frequency-Specific Effects
Abstract
1. Introduction
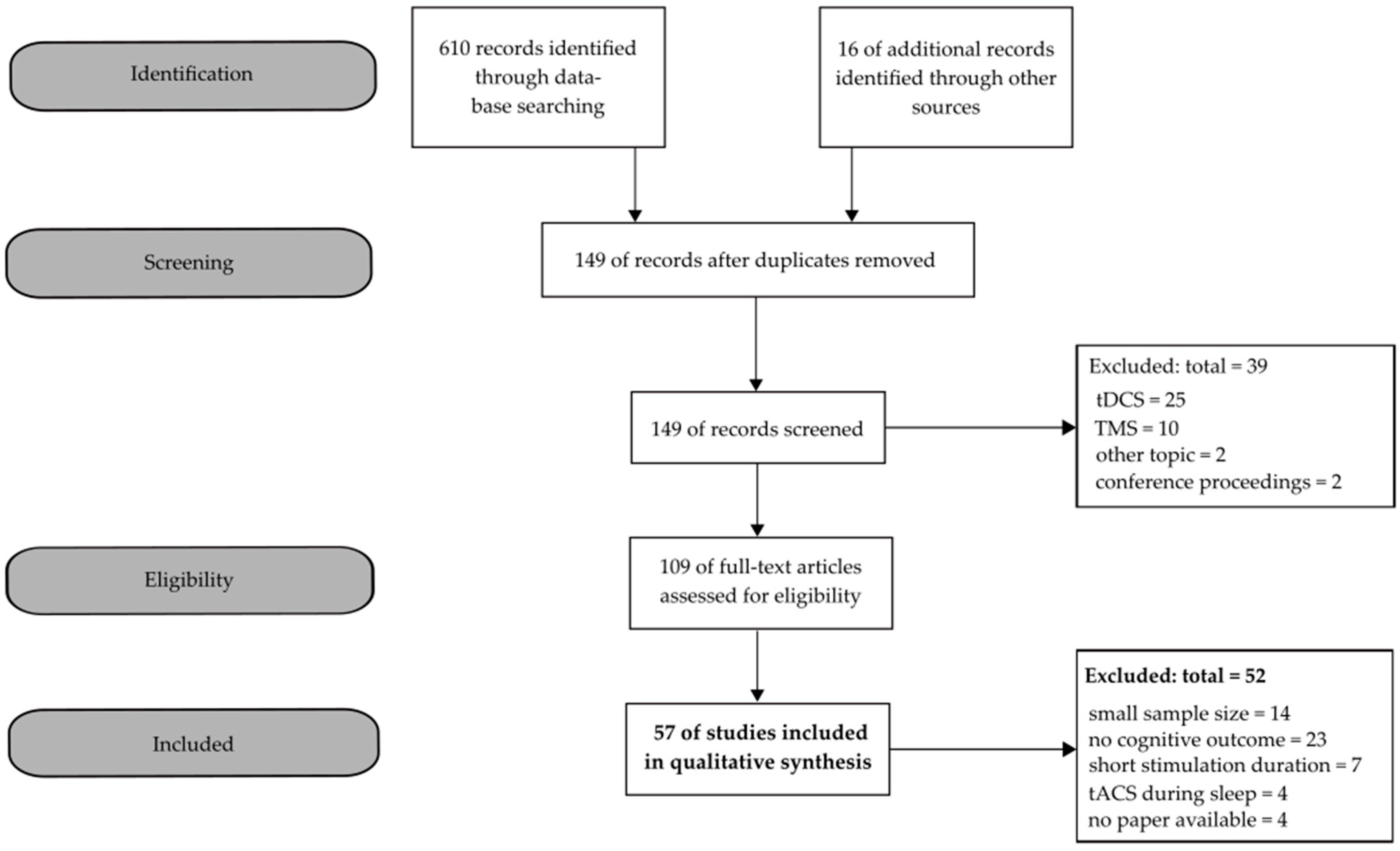
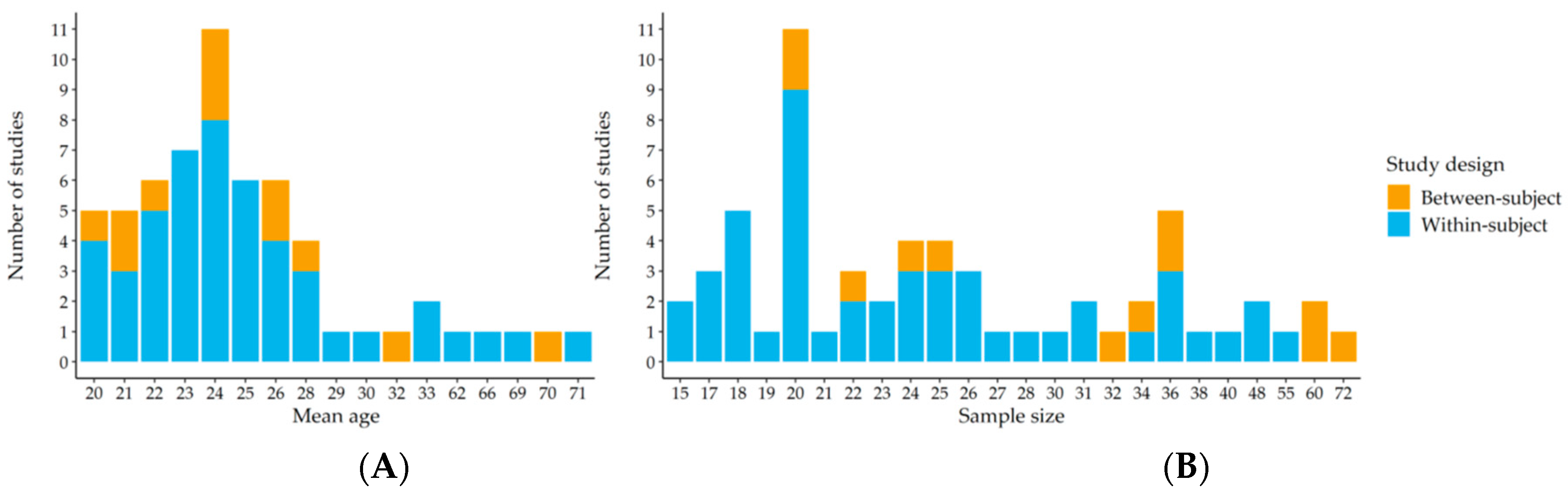
2. Materials and Methods
- “Transcranial AND alternating AND current AND stimulation AND *cognitive domain* AND healthy AND adults”
- “TACS AND *cognitive domain* AND healthy AND adults”.
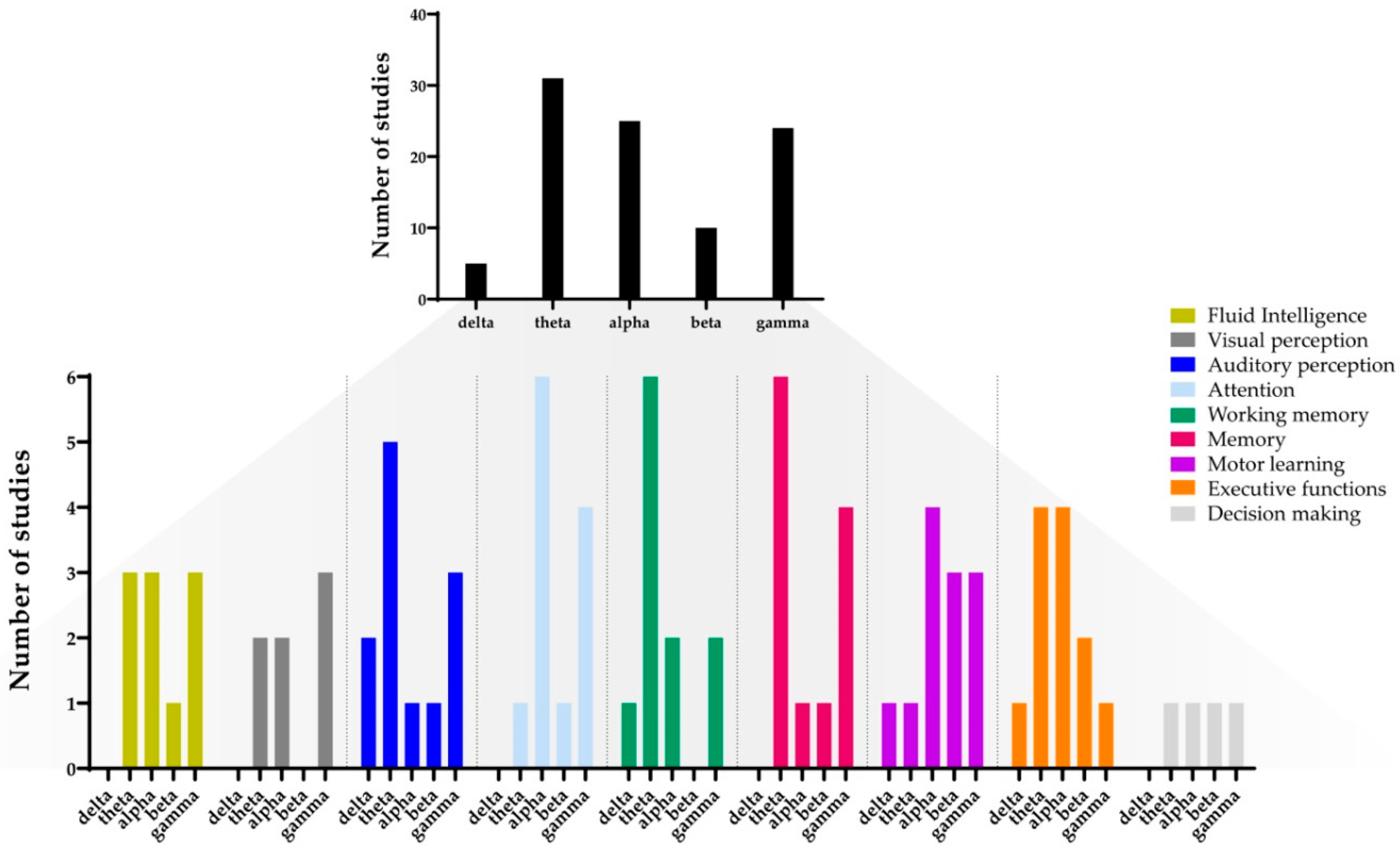
3. Results
3.1. Auditory Perception
3.2. Visual Perception
3.3. Attention
3.4. Executive Functions
3.5. Working Memory
3.6. Declarative Memory
3.7. Procedural Memory (Motor Learning)
3.8. Decision-Making
3.9. Intelligence
4. Discussion
Author Contributions
Funding
Conflicts of Interest
References
- Tavakoli, A.V.; Yun, K. Transcranial alternating current stimulation (tACS) mechanisms and protocols. Front. Cell. Neurosci. 2017, 11, 214. [Google Scholar] [CrossRef]
- Antal, A.; Herrmann, C.S. Transcranial alternating current and random noise stimulation: Possible mechanisms. Neural Plast. 2016, 2016, e3616807. [Google Scholar] [CrossRef]
- Johnson, L.; Alekseichuk, I.; Krieg, J.; Doyle, A.; Yu, Y.; Vitek, J.; Johnson, M.; Opitz, A. Dose-dependent effects of transcranial alternating current stimulation on spike timing in awake nonhuman primates. Sci. Adv. 2020, 6, eaaz2747. [Google Scholar] [CrossRef]
- Krause, M.R.; Vieira, P.G.; Csorba, B.A.; Pilly, P.K.; Pack, C.C. Transcranial alternating current stimulation entrains single-neuron activity in the primate brain. Proc. Natl. Acad. Sci. USA 2019, 116, 5747–5755. [Google Scholar] [CrossRef]
- Fröhlich, F.; McCormick, D.A. Endogenous electric fields may guide neocortical network activity. Neuron 2010, 67, 129–143. [Google Scholar] [CrossRef]
- Vossen, A.; Gross, J.; Thut, G. Alpha power increase after transcranial alternating current stimulation at alpha frequency (a-tACS) reflects plastic changes rather than entrainment. Brain Stimul. 2015, 8, 499–508. [Google Scholar] [CrossRef]
- Kasten, F.H.; Herrmann, C.S. Transcranial alternating current stimulation (tACS) enhances mental rotation performance during and after stimulation. Front. Hum. Neurosci. 2017, 11, 1–16. [Google Scholar] [CrossRef]
- Bland, N.S.; Sale, M.V. Current challenges: The ups and downs of tACS. Exp. Brain Res. 2019, 237, 3071–3088. [Google Scholar] [CrossRef]
- Sauseng, P.; Klimesch, W. What does phase information of oscillatory brain activity tell us about cognitive processes? Neurosci. Biobehav. Rev. 2008, 32, 1001–1013. [Google Scholar] [CrossRef]
- Morillon, B.; Arnal, L.H.; Schroeder, C.E.; Keitel, A. Prominence of delta oscillatory rhythms in the motor cortex and their relevance for auditory and speech perception. Neurosci. Biobehav. Rev. 2019, 107, 136–142. [Google Scholar] [CrossRef]
- Harmony, T. The functional significance of delta oscillations in cognitive processing. Front. Integr. Neurosci. 2013, 7, 83. [Google Scholar] [CrossRef]
- Sauseng, P.; Griesmayr, B.; Freunberger, R.; Klimesch, W. Control mechanisms in working memory: A possible function of EEG theta oscillations. Neurosci. Biobehav. Rev. 2010, 34, 1015–1022. [Google Scholar] [CrossRef]
- Hsieh, L.-T.; Ranganath, C. Frontal midline theta oscillations during working memory maintenance and episodic encoding and retrieval. Neuroimage 2014, 85 Pt 2, 721–729. [Google Scholar] [CrossRef]
- Herweg, N.A.; Solomon, E.A.; Kahana, M.J. Theta oscillations in human memory. Trends Cogn. Sci. 2020, 24, 208–227. [Google Scholar] [CrossRef]
- Kim, N.Y.; Wittenberg, E.; Nam, C.S. Behavioral and neural correlates of executive function: Interplay between inhibition and updating processes. Front. Neurosci. 2017, 11, 378. [Google Scholar] [CrossRef]
- Taylor, P.C.J.; Thut, G. Brain activity underlying visual perception and attention as inferred from TMS–EEG: A review. Brain Stimul. 2012, 5, 124–129. [Google Scholar] [CrossRef]
- Mierau, A.; Klimesch, W.; Lefebvre, J. State-dependent alpha peak frequency shifts: Experimental evidence, potential mechanisms and functional implications. Neuroscience 2017, 360, 146–154. [Google Scholar] [CrossRef]
- Engel, A.K.; Fries, P. Beta-band oscillations—Signalling the status quo? Curr. Opin. Neurobiol. 2010, 20, 156–165. [Google Scholar] [CrossRef]
- Schmidt, R.; Ruiz, M.H.; Kilavik, B.E.; Lundqvist, M.; Starr, P.A.; Aron, A.R. Beta oscillations in working memory, executive control of movement and thought, and sensorimotor function. J. Neurosci. 2019, 39, 8231. [Google Scholar] [CrossRef]
- Fries, P. Rhythms for cognition: Communication through coherence. Neuron 2015, 88, 220–235. [Google Scholar] [CrossRef]
- Pina, J.E.; Bodner, M.; Ermentrout, B. Oscillations in working memory and neural binding: A mechanism for multiple memories and their interactions. PLoS Comput. Biol. 2018, 14, e1006517. [Google Scholar] [CrossRef]
- Nyhus, E.; Curran, T. Functional role of gamma and theta oscillations in episodic memory. Neurosci. Biobehav. Rev. 2010, 34, 1023–1035. [Google Scholar] [CrossRef]
- Baltus, A.; Herrmann, C.S. The importance of individual frequencies of endogenous brain oscillations for auditory cognition—A short review. Brain Res. 2016, 1640, 243–250. [Google Scholar] [CrossRef]
- Schutter, D.J.L.G.; Wischnewski, M. A meta-analytic study of exogenous oscillatory electric potentials in neuroenhancement. Neuropsychologia 2016, 86, 110–118. [Google Scholar] [CrossRef]
- Naro, A.; Leo, A.; Russo, M.; Cannavò, A.; Milardi, D.; Bramanti, P.; Calabrò, R.S. Does transcranial alternating current stimulation induce cerebellum plasticity? Feasibility, safety and efficacy of a novel electrophysiological approach. Brain Stimul. 2016, 9, 388–395. [Google Scholar] [CrossRef]
- Dayan, E.; Censor, N.; Buch, E.R.; Sandrini, M.; Cohen, L.G. Noninvasive brain stimulation: From physiology to network dynamics and back. Nat. Neurosci. 2013, 16, 838–844. [Google Scholar] [CrossRef]
- Woods, A.J.; Antal, A.; Bikson, M.; Boggio, P.S.; Brunoni, A.R.; Celnik, P.; Cohen, L.G.; Fregni, F.; Herrmann, C.S.; Kappenman, E.S.; et al. A technical guide to tDCS, and related non-invasive brain stimulation tools. Clin. Neurophysiol. 2016, 127, 1031–1048. [Google Scholar] [CrossRef]
- Fröhlich, F.; Sellers, K.K.; Cordle, A.L. Targeting the neurophysiology of cognitive systems with transcranial alternating current stimulation. Expert Rev. Neurother. 2015, 15, 145–167. [Google Scholar] [CrossRef]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G.; Group, T.P. Preferred reporting items for systematic reviews and Meta-analyses: The PRISMA statement. PLoS Med. 2009, 6, e1000097. [Google Scholar] [CrossRef]
- Schuhmann, T.; Kemmerer, S.K.; Duecker, F.; de Graaf, T.A.; Ten Oever, S.; De Weerd, P.; Sack, A.T. Left parietal tACS at alpha frequency induces a shift of visuospatial attention. PLoS ONE 2019, 14, e0217729. [Google Scholar] [CrossRef]
- Deng, Y.; Reinhart, R.M.G.; Choi, I.; Shinn-Cunningham, B. Causal links between parietal alpha activity and spatial auditory attention. eLife 2019, 8, 1–23. [Google Scholar] [CrossRef]
- Otsuru, N.; Kamijo, K.; Otsuki, T.; Kojima, S.; Miyaguchi, S.; Saito, K.; Inukai, Y.; Onishi, H. 10 Hz transcranial alternating current stimulation over posterior parietal cortex facilitates tactile temporal order judgment. Behav. Brain Res. 2019, 368, 111899. [Google Scholar] [CrossRef]
- Yaple, Z.; Vakhrushev, R. Modulation of the frontal-parietal network by low intensity anti-phase 20 Hz transcranial electrical stimulation boosts performance in the attentional blink task. Int. J. Psychophysiol. 2018, 127, 11–16. [Google Scholar] [CrossRef]
- Clayton, M.S.; Yeung, N.; Cohen Kadosh, R. Electrical stimulation of alpha oscillations stabilizes performance on visual attention tasks. J. Exp. Psychol. Gen. 2019, 148, 203–220. [Google Scholar] [CrossRef]
- Hopfinger, J.B.; Parsons, J.; Fröhlich, F. Differential effects of 10-Hz and 40-Hz transcranial alternating current stimulation (tACS) on endogenous versus exogenous attention. Cogn. Neurosci. 2017, 8, 102–111. [Google Scholar] [CrossRef]
- Wöstmann, M.; Vosskuhl, J.; Obleser, J.; Herrmann, C.S. Opposite effects of lateralised transcranial alpha versus gamma stimulation on auditory spatial attention. Brain Stimul. 2018, 11, 752–758. [Google Scholar] [CrossRef]
- Meier, J.; Nolte, G.; Schneider, T.R.; Engel, A.K.; Leicht, G.; Mulert, C. Intrinsic 40Hz-phase asymmetries predict tACS effects during conscious auditory perception. PLoS ONE 2019, 14, e0213996. [Google Scholar] [CrossRef]
- Laczó, B.; Antal, A.; Niebergall, R.; Treue, S.; Paulus, W. Transcranial alternating stimulation in a high gamma frequency range applied over V1 improves contrast perception but does not modulate spatial attention. Brain Stimul. 2012, 5, 484–491. [Google Scholar] [CrossRef]
- Neubauer, A.C.; Wammerl, M.; Benedek, M.; Jauk, E.; Jaušovec, N. The influence of transcranial alternating current stimulation (tACS) on fluid intelligence: An fMRI study. Personal. Individ. Differ. 2017, 118, 50–55. [Google Scholar] [CrossRef]
- Pahor, A.; Jaušovec, N. Making brains run faster: Are they becoming smarter? Span. J. Psychol. 2016, 19, 1–27. [Google Scholar] [CrossRef]
- Grabner, R.H.; Krenn, J.; Fink, A.; Arendasy, M.; Benedek, M. Effects of alpha and gamma transcranial alternating current stimulation (tACS) on verbal creativity and intelligence test performance. Neuropsychologia 2018, 118, 91–98. [Google Scholar] [CrossRef]
- Santarnecchi, E.; Muller, T.; Rossi, S.; Sarkar, A.; Polizzotto, N.R.; Rossi, A.; Cohen Kadosh, R. Individual differences and specificity of prefrontal gamma frequency-tACS on fluid intelligence capabilities. Cortex 2016, 75, 33–43. [Google Scholar] [CrossRef]
- Santarnecchi, E.; Polizzotto, N.R.; Godone, M.; Giovannelli, F.; Feurra, M.; Matzen, L.; Rossi, A.; Rossi, S. Frequency-dependent enhancement of fluid intelligence induced by transcranial oscillatory potentials. Curr. Biol. 2013, 23, 1449–1453. [Google Scholar] [CrossRef]
- Brignani, D.; Ruzzoli, M.; Mauri, P.; Miniussi, C. Is transcranial alternating current stimulation effective in modulating brain oscillations? PLoS ONE 2013, 8, e56589. [Google Scholar] [CrossRef]
- Herring, J.D.; Esterer, S.; Marshall, T.R.; Jensen, O.; Bergmann, T.O. Low-frequency alternating current stimulation rhythmically suppresses gamma-band oscillations and impairs perceptual performance. NeuroImage 2019, 184, 440–449. [Google Scholar] [CrossRef]
- Gonzalez-Perez, M.; Wakui, E.; Thoma, V.; Nitsche, M.A.; Rivolta, D. Transcranial alternating current stimulation (tACS) at 40 Hz enhances face and object perception. Neuropsychologia 2019, 135, 107237. [Google Scholar] [CrossRef]
- Strüber, D.; Rach, S.; Trautmann-Lengsfeld, S.A.; Engel, A.K.; Herrmann, C.S. Antiphasic 40 Hz oscillatory current stimulation affects bistable motion perception. Brain Topogr. 2014, 27, 158–171. [Google Scholar] [CrossRef]
- Zoefel, B.; Archer-Boyd, A.; Davis, M.H. Phase entrainment of brain oscillations causally modulates neural responses to intelligible speech. Curr. Biol. 2018, 28, 401–408.e5. [Google Scholar] [CrossRef]
- Zoefel, B.; Allard, I.; Anil, M.; Davis, M.H. Perception of rhythmic speech is modulated by focal bilateral transcranial alternating current stimulation. J. Cogn. Neurosci. 2019, 32, 226–240. [Google Scholar] [CrossRef]
- Riecke, L.; Sack, A.T.; Schroeder, C.E. Endogenous delta/theta sound-brain phase entrainment accelerates the buildup of auditory streaming. Curr. Biol. 2015, 25, 3196–3201. [Google Scholar] [CrossRef]
- Riecke, L.; Formisano, E.; Sorger, B.; Başkent, D.; Gaudrain, E. Neural entrainment to speech modulates speech intelligibility. Curr. Biol. 2018, 28, 161–169.e5. [Google Scholar] [CrossRef]
- Moliadze, V.; Sierau, L.; Lyzhko, E.; Stenner, T.; Werchowski, M.; Siniatchkin, M.; Hartwigsen, G. After-effects of 10 Hz tACS over the prefrontal cortex on phonological word decisions. Brain Stimul. 2019, 12, 1464–1474. [Google Scholar] [CrossRef]
- Rufener, K.S.; Zaehle, T.; Oechslin, M.S.; Meyer, M. 40 Hz-Transcranial alternating current stimulation (tACS) selectively modulates speech perception. Int. J. Psychophysiol. 2016, 101, 18–24. [Google Scholar] [CrossRef]
- Rufener, K.S.; Oechslin, M.S.; Zaehle, T.; Meyer, M. Transcranial alternating current stimulation (tACS) differentially modulates speech perception in young and older adults. Brain Stimul. 2016, 9, 560–565. [Google Scholar] [CrossRef]
- Brauer, H.; Kadish, N.E.; Pedersen, A.; Siniatchkin, M.; Moliadze, V. No modulatory effects when stimulating the right inferior frontal gyrus with continuous 6Hz TACs and TRNs on response inhibition: A behavioral study. Neural Plast. 2018, 2018. [Google Scholar] [CrossRef]
- Reinhart, R.M.G. Disruption and rescue of interareal theta phase coupling and adaptive behavior. Proc. Natl. Acad. Sci. USA 2017, 114, 11542–11547. [Google Scholar] [CrossRef]
- Van Driel, J.; Sligte, I.G.; Linders, J.; Elport, D.; Cohen, M.X. Frequency band-specific electrical brain stimulation modulates cognitive control processes. PLoS ONE 2015, 10, e0138984. [Google Scholar] [CrossRef]
- Kasten, F.H.; Maess, B.; Herrmann, C.S. Facilitated event-related power modulations during transcranial alternating current stimulation (tACS) revealed by concurrent tACS-MEG. eNeuro 2018, 5. [Google Scholar] [CrossRef]
- Wiener, M.; Parikh, A.; Krakow, A.; Coslett, H.B. An intrinsic role of beta oscillations in memory for time estimation. Sci. Rep. 2018, 8, 7992. [Google Scholar] [CrossRef]
- Fusco, G.; Scandola, M.; Feurra, M.; Pavone, E.F.; Rossi, S.; Aglioti, S.M. Midfrontal theta transcranial alternating current stimulation modulates behavioural adjustment after error execution. Eur. J. Neurosci. 2018, 48, 3159–3170. [Google Scholar] [CrossRef]
- Yaple, Z.; Martinez-Saito, M.; Feurra, M.; Shestakova, A.; Klucharev, V. Transcranial alternating current stimulation modulates risky decision making in a frequency-controlled experiment. eNeuro 2017, 4. [Google Scholar] [CrossRef]
- Zavecz, Z.; Horváth, K.; Solymosi, P.; Janacsek, K.; Nemeth, D. Frontal-midline theta frequency and probabilistic learning: A transcranial alternating current stimulation study. Behav. Brain Res. 2020, 393, 112733. [Google Scholar] [CrossRef]
- Fresnoza, S.; Christova, M.; Bieler, L.; Körner, C.; Zimmer, U.; Gallasch, E.; Ischebeck, A. Age-dependent effect of transcranial alternating current stimulation on motor skill consolidation. Front. Aging Neurosci. 2020, 12, 1–15. [Google Scholar] [CrossRef]
- Brinkman, L.; Stolk, A.; Marshall, T.R.; Esterer, S.; Sharp, P.; Dijkerman, H.C.; de Lange, F.P.; Toni, I. Independent causal contributions of Alpha- and Beta-band oscillations during movement selection. J. Neurosci. 2016, 36, 8726–8733. [Google Scholar] [CrossRef]
- Nowak, M.; Hinson, E.; Van Ede, F.; Pogosyan, A.; Guerra, A.; Quinn, A.; Brown, P.; Stagg, C.J. Driving human motor cortical oscillations leads to behaviorally relevant changes in local GABAA inhibition: A tACS-TMS study. J. Neurosci. 2017, 37, 4481–4492. [Google Scholar] [CrossRef]
- Giustiniani, A.; Tarantino, V.; Bonaventura, R.E.; Smirni, D.; Turriziani, P.; Oliveri, M. Effects of low-gamma tACS on primary motor cortex in implicit motor learning. Behav. Brain Res. 2019, 376, 112170. [Google Scholar] [CrossRef]
- Sugata, H.; Yagi, K.; Yazawa, S.; Nagase, Y.; Tsuruta, K.; Ikeda, T.; Matsushita, K.; Hara, M.; Kawakami, K.; Kawakami, K. Modulation of motor learning capacity by transcranial alternating current stimulation. Neuroscience 2018, 391, 131–139. [Google Scholar] [CrossRef]
- Alekseichuk, I.; Pabel, S.C.; Antal, A.; Paulus, W. Intrahemispheric theta rhythm desynchronization impairs working memory. Restor. Neurol. Neurosci. 2017, 35, 147–158. [Google Scholar] [CrossRef]
- Bender, M.; Romei, V.; Sauseng, P. Slow theta tACS of the right parietal cortex enhances contralateral visual working memory capacity. Brain Topogr. 2019, 32, 477–481. [Google Scholar] [CrossRef]
- Jausovec, N.; Jausovec, K.; Pahor, A. The influence of theta transcranial alternating current stimulation (tACS) on working memory storage and processing functions. Acta Psychol. 2014, 146, 1–6. [Google Scholar] [CrossRef]
- Tseng, P.; Iu, K.C.; Juan, C.H. The critical role of phase difference in theta oscillation between bilateral parietal cortices for visuospatial working memory. Sci. Rep. 2018, 8, 1–9. [Google Scholar] [CrossRef]
- Polanía, R.; Nitsche, M.A.; Korman, C.; Batsikadze, G.; Paulus, W. The importance of timing in segregated theta phase-coupling for cognitive performance. Curr. Biol. 2012, 22, 1314–1318. [Google Scholar] [CrossRef]
- Violante, I.R.; Li, L.M.; Carmichael, D.W.; Lorenz, R.; Leech, R.; Hampshire, A.; Rothwell, J.C.; Sharp, D.J. Externally induced frontoparietal synchronization modulates network dynamics and enhances working memory performance. eLife 2017, 6, 1–22. [Google Scholar] [CrossRef]
- Wolinski, N.; Cooper, N.R.; Sauseng, P.; Romei, V. The speed of parietal theta frequency drives visuospatial working memory capacity. PLoS Biol. 2018, 16, 1–17. [Google Scholar] [CrossRef]
- Gutteling, T.P.; Schutter, D.J.L.G.; Medendorp, W.P. Alpha-band transcranial alternating current stimulation modulates precision, but not gain during whole-body spatial updating. Neuropsychologia 2017, 106, 52–59. [Google Scholar] [CrossRef]
- Hoy, K.E.; Bailey, N.; Arnold, S.; Windsor, K.; John, J.; Daskalakis, Z.J.; Fitzgerald, P.B. The effect of γ-tACS on working memory performance in healthy controls. Brain Cogn. 2015, 101, 51–56. [Google Scholar] [CrossRef]
- Tseng, P.; Chang, Y.T.; Chang, C.F.; Liang, W.K.; Juan, C.H. The critical role of phase difference in gamma oscillation within the temporoparietal network for binding visual working memory. Sci. Rep. 2016, 6, 1–15. [Google Scholar] [CrossRef]
- Borghini, G.; Candini, M.; Filannino, C.; Hussain, M.; Walsh, V.; Romei, V.; Zokaei, N.; Cappelletti, M. Alpha oscillations are causally linked to inhibitory abilities in ageing. J. Neurosci. 2018, 38, 4418–4429. [Google Scholar] [CrossRef]
- Antonenko, D.; Faxel, M.; Grittner, U.; Lavidor, M.; Flöel, A. Effects of transcranial alternating current stimulation on cognitive functions in healthy young and older adults. Neural Plast. 2016, 2016, 4274127. [Google Scholar] [CrossRef]
- De Lara, G.A.; Alekseichuk, I.; Turi, Z.; Lehr, A.; Antal, A.; Paulus, W. Perturbation of theta-gamma coupling at the temporal lobe hinders verbal declarative memory. Brain Stimul. 2018, 11, 509–517. [Google Scholar] [CrossRef]
- Klink, K.; Peter, J.; Wyss, P.; Klöppel, S. Transcranial electric current stimulation during associative memory encoding: Comparing tACS and tDCS effects in healthy aging. Front. Aging Neurosci. 2020, 12, 66. [Google Scholar] [CrossRef] [PubMed]
- Alekseichuk, I.; Turi, Z.; Veit, S.; Paulus, W. Model-driven neuromodulation of the right posterior region promotes encoding of long-term memories. Brain Stimul. 2019, 13, 474–483. [Google Scholar] [CrossRef]
- Lang, S.; Gan, L.S.; Alrazi, T.; Monchi, O. Theta band high definition transcranial alternating current stimulation, but not transcranial direct current stimulation, improves associative memory performance. Sci. Rep. 2019, 9. [Google Scholar] [CrossRef]
- Javadi, A.-H.; Glen, J.C.; Halkiopoulos, S.; Schulz, M.; Spiers, H.J. Oscillatory reinstatement enhances declarative memory. J. Neurosci. 2017, 37, 9939–9944. [Google Scholar] [CrossRef]
- Nomura, T.; Asao, A.; Kumasaka, A. Transcranial alternating current stimulation over the prefrontal cortex enhances episodic memory recognition. Exp. Brain Res. 2019, 237, 1709–1715. [Google Scholar] [CrossRef]
- Braun, V.; Sokoliuk, R.; Hanslmayr, S. On the effectiveness of event-related beta tACS on episodic memory formation and motor cortex excitability. Brain Stimul. 2017, 10, 910–918. [Google Scholar] [CrossRef]
- Henry, M.J.; Obleser, J. Frequency modulation entrains slow neural oscillations and optimizes human listening behavior. Proc. Natl. Acad. Sci. USA 2012, 109, 20095–20100. [Google Scholar] [CrossRef]
- Bridwell, D.A.; Henderson, S.; Sorge, M.; Plis, S.; Calhoun, V.D. Relationships between alpha oscillations during speech preparation and the listener N400 ERP to the produced speech. Sci. Rep. 2018, 8, 12838. [Google Scholar] [CrossRef]
- Klimesch, W. α-band oscillations, attention, and controlled access to stored information. Trends Cogn. Sci. (Regul. Ed.) 2012, 16, 606–617. [Google Scholar] [CrossRef]
- Clayton, M.S.; Yeung, N.; Cohen Kadosh, R. The many characters of visual alpha oscillations. Eur. J. Neurosci. 2018, 48, 2498–2508. [Google Scholar] [CrossRef]
- Proskovec, A.L.; Wiesman, A.I.; Wilson, T.W. The strength of alpha and gamma oscillations predicts behavioral switch costs. NeuroImage 2019, 188, 274–281. [Google Scholar] [CrossRef]
- Roux, F.; Uhlhaas, P.J. Working memory and neural oscillations: Alpha–gamma versus theta–gamma codes for distinct WM information? Trends Cogn. Sci. 2014, 18, 16–25. [Google Scholar] [CrossRef]
- Constantinidis, C.; Klingberg, T. The neuroscience of working memory capacity and training. Nat. Rev. Neurosci. 2016, 17, 438–449. [Google Scholar] [CrossRef]
- Meissner, S.N.; Krause, V.; Südmeyer, M.; Hartmann, C.J.; Pollok, B. The significance of brain oscillations in motor sequence learning: Insights from Parkinson’s disease. NeuroImage Clin. 2018, 20, 448–457. [Google Scholar] [CrossRef]
- Marco-Pallarés, J.; Münte, T.F.; Rodríguez-Fornells, A. The role of high-frequency oscillatory activity in reward processing and learning. Neurosci. Biobehav. Rev. 2015, 49, 1–7. [Google Scholar] [CrossRef]
- Bunzeck, N.; Guitart-Masip, M.; Dolan, R.J.; Düzel, E. Contextual novelty modulates the neural dynamics of reward anticipation. J. Neurosci. 2011, 31, 12816–12822. [Google Scholar] [CrossRef]
- Chuderski, A. Fluid intelligence and the cross-frequency coupling of neuronal oscillations. Span. J. Psychol. 2016, 19, E91. [Google Scholar] [CrossRef]
- Kral, A.; Tillein, J. Brain plasticity under cochlear implant stimulation. Cochlear Brainstem Implant. 2006, 64, 89–108. [Google Scholar]
- Heimrath, K.; Fiene, M.; Rufener, K.S.; Zaehle, T. Modulating human auditory processing by transcranial electrical stimulation. Front. Cell. Neurosci. 2016, 10, 53. [Google Scholar] [CrossRef]
- Smulders, T.V.; Black-Dominique, A.; Choudhury, T.S.; Constantinescu, S.E.; Foka, K.; Walker, T.J.; Dick, K.; Bradwel, S.; McAllister-Williams, R.H.; Gallagher, P. A real-world what-where-when memory test. JoVE 2017, 55646. [Google Scholar] [CrossRef]
- Jung, S.; Yoon, Y.; Han, S.W. Working memory-driven attention in real-world search. Perception 2018, 47, 966–975. [Google Scholar] [CrossRef]
- Çukur, T.; Nishimoto, S.; Huth, A.G.; Gallant, J.L. Attention during natural vision warps semantic representation across the human brain. Nat. Neurosci. 2013, 16, 763–770. [Google Scholar] [CrossRef]
- Brady, T.F.; Konkle, T.; Oliva, A.; Alvarez, G.A. Detecting changes in real-world objects: The relationship between visual long-term memory and change blindness. Commun Integr. Biol. 2009, 2, 1–3. [Google Scholar] [CrossRef]
- Haberman, J.; Whitney, D. Rapid extraction of mean emotion and gender from sets of faces. Curr. Biol. 2007, 17, R751–R753. [Google Scholar] [CrossRef]
- Debener, S.; Emkes, R.; De Vos, M.; Bleichner, M. Unobtrusive ambulatory EEG using a smartphone and flexible printed electrodes around the ear. Sci. Rep. 2015, 5, 16743. [Google Scholar] [CrossRef]
- Debener, S.; Minow, F.; Emkes, R.; Gandras, K.; de Vos, M. How about taking a low-cost, small, and wireless EEG for a walk? Psychophysiology 2012, 49, 1617–1621. [Google Scholar] [CrossRef]
- Bleichner, M.G.; Debener, S. Concealed, unobtrusive ear-centered EEG acquisition: cEEGrids for transparent EEG. Front. Hum. Neurosci. 2017, 11, 163. [Google Scholar] [CrossRef]
- Griffiths, B.; Mazaheri, A.; Debener, S.; Hanslmayr, S. Brain oscillations track the formation of episodic memories in the real world. NeuroImage 2016, 143, 256–266. [Google Scholar] [CrossRef]
- Seeber, M.; Scherer, R.; Wagner, J.; Solis-Escalante, T.; Müller-Putz, G.R. High and low gamma EEG oscillations in central sensorimotor areas are conversely modulated during the human gait cycle. Neuroimage 2015, 112, 318–326. [Google Scholar] [CrossRef]
- Liang, M.; Starrett, M.J.; Ekstrom, A.D. Dissociation of frontal-midline delta-theta and posterior alpha oscillations: A mobile EEG study. Psychophysiology 2018, 55, e13090. [Google Scholar] [CrossRef]
- Feurra, M.; Pasqualetti, P.; Bianco, G.; Santarnecchi, E.; Rossi, A.; Rossi, S. State-dependent effects of transcranial oscillatory currents on the motor system: What you think matters. J. Neurosci. 2013, 33, 17483–17489. [Google Scholar] [CrossRef]
- Ruhnau, P.; Neuling, T.; Fuscá, M.; Herrmann, C.S.; Demarchi, G.; Weisz, N. Eyes wide shut: Transcranial alternating current stimulation drives alpha rhythm in a state dependent manner. Sci. Rep. 2016, 6, 27138. [Google Scholar] [CrossRef]
- Stecher, H.I.; Pollok, T.M.; Strüber, D.; Sobotka, F.; Herrmann, C.S. Ten minutes of α-tACS and ambient illumination independently modulate EEG α-Power. Front. Hum. Neurosci. 2017, 11, 257. [Google Scholar] [CrossRef]
- Kasten, F.H.; Herrmann, C.S. Recovering brain dynamics during concurrent tACS-M/EEG: An overview of analysis approaches and their methodological and interpretational pitfalls. Brain Topogr. 2019, 32, 1013–1019. [Google Scholar] [CrossRef]
- Kasten, F.H.; Duecker, K.; Maack, M.C.; Meiser, A.; Herrmann, C.S. Integrating electric field modeling and neuroimaging to explain inter-individual variability of tACS effects. Nat. Commun. 2019, 10, 5427. [Google Scholar] [CrossRef]
- Huang, Y.; Thomas, C.; Datta, A.; Parra, L.C. Optimized tDCS for targeting multiple brain regions: An integrated implementation. Annu. Int. Conf. IEEE. Eng. Med. Biol. Soc. 2018, 2018, 3545–3548. [Google Scholar]
- Huang, Y.; Datta, A.; Bikson, M.; Parra, L.C. Realistic volumetric-approach to simulate transcranial electric stimulation—ROAST—a fully automated open-source pipeline. J. Neural. Eng. 2019, 16, 056006. [Google Scholar] [CrossRef]
- Saturnino, G.B.; Siebner, H.R.; Thielscher, A.; Madsen, K.H. Accessibility of cortical regions to focal TES: Dependence on spatial position, safety, and practical constraints. NeuroImage 2019, 203, 116183. [Google Scholar] [CrossRef]
- Wagner, S.; Burger, M.; Wolters, C.H. An optimization approach for well-targeted transcranial direct current stimulation. SIAM J. Appl. Math. 2016, 76, 2154–2174. [Google Scholar] [CrossRef]
- Button, K.S.; Ioannidis, J.P.A.; Mokrysz, C.; Nosek, B.A.; Flint, J.; Robinson, E.S.J.; Munafò, M.R. Power failure: Why small sample size undermines the reliability of neuroscience. Nat. Rev. Neurosci. 2013, 14, 365–376. [Google Scholar] [CrossRef]
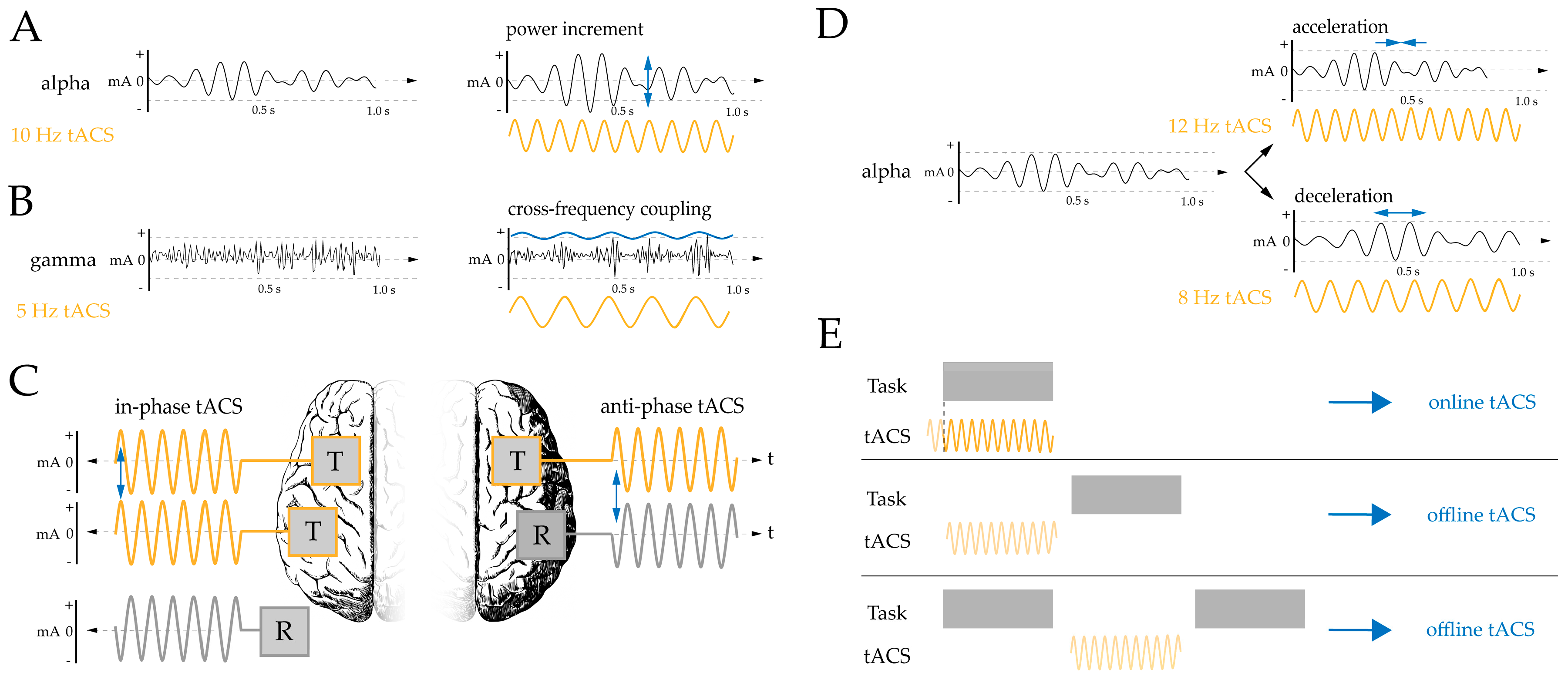
| Authors | Participants and Design | Task and Dependent Variables | Electrode Montage and Device | Stimulation Parameters | Outcome/Risk Bias |
|---|---|---|---|---|---|
| Attention | |||||
| Alpha | |||||
| [30] | N = 36 (mean age: 21.56 years, range 18–29 years) Crossover within-subject design (tACS vs. sham) | Endogenous and exogenous spatial attention task Reaction time bias score | Left parietal cortex (P3) + centered above target electrode Device: high-definition tES (NeuroConn, Ilmenau, Germany) | Frequency: 10 Hz/sham Intensity: 1 mA peak-to-peak Duration: 35–40 min Electrode size: target electrode (round-shaped, diameter: 2.1 cm), return electrode (round-shaped, outer diameter: 11 cm, inner diameter: 9 cm) Timing: online | ↗ (for endogenous task only) Low risk |
| [31] | Exp. 1 N = 20 (mean age: 26.6 ± 4.1 years) Exp. 2 N = 18 (mean age: 22.11 ± 2.4 years) All Exp. Crossover within-subject design (tACS vs. sham) | Selective auditory attention task Number of /ga/-syllables in spatial (left or right direction) or non-spatial trials (female/male speakers) | Parietal cortex (P2) + (CP2/P4/Pz/PO4) Soterix M × N 9 high definition-tES Stimulator (Soterix Medical, New York, NY) | Frequency: Exp. 1 10 Hz/sham; Exp. 2 6 Hz/sham Intensity: 1.5 mA peak-to-peak Duration: 20 min incl. 30 s ramp-in/-out Electrode Size: not provided Timing: online | ↘ (with alpha-tACS) − (with theta-tACS) Low risk |
| [32] | Exp. 1 N = 24 (mean age: 21) Exp. 2 N = 12 (mean age: 21.2 ± 0.2 years) All Exp. Crossover within-subject design (tACS vs. sham) | Temporal order judgement task Forced-choice verbal report of stimuli order | Exp. 1 Left somatosensory cortex (C3) or left posterior parietal cortex (P3) + ipsilateral lower cheek Exp. 2 Right posterior parietal cortex (P4) + ipsilateral lower cheek Device: Eldith (NeuroConn GmbH, Ilmenau, Germany) | Frequency: 10 Hz/sham Intensity: 1 mA peak-to-peak Duration: 270 s Electrode size: both 5 × 5 cm Timing: online | ↗ Low risk |
| Alpha and Beta | |||||
| [33] | Exp. 1 N = 18 (mean age: 20.66 ± 2.53 years) Crossover within-subject design (10 Hz versus 20 Hz versus sham) Exp. 2 N = 15 (mean age: 20.26 ± 2.35 years) Crossover within-subject design (sham vs. 10 Hz in-phase vs. 10 Hz anti-phase vs. 20 Hz in-phase vs. 20 Hz anti-phase) | Attentional blink task Detection of second target stimuli | Exp. 1 Right posterior parietal cortex (P4) + right deltoid Exp. 2 Right posterior parietal cortex (P4) and left frontal cortex (F3) + right deltoid Device: Exp. 1 BrainStim (EMS Medical, Bologna, Italy); Exp. 2 StarStim 8 (Neuroelectrics, Boston, Massachusetts) | Frequency: Exp. 1 10 Hz/20 Hz/sham; Exp. 2 10 Hz in-phase/10 Hz anti-phase/20 Hz in-phase/20 Hz anti-phase/sham Intensity: 0.35 mA Duration: Exp. 1 35 min; Exp. 2 50 min Electrode size: Exp. 1 both 7 × 5 cm; Exp. 2 both round-shaped electrodes (25 cm2) Timing: online | ↗ (with anti-phase beta-tACS) − (with alpha-tACS or in-phase beta-tACS) Low risk |
| Alpha and Gamma | |||||
| [34] | Exp. 1 N = 48 (mean age: 22.7 ± 2.9 years) Exp. 2 N = 37 (mean age: 22.7 ± 3.5 years) Exp. 3 N = 41 (mean age: 23.2 ± 2.7 years) Exp. 4 N = 43 (mean age: 23.1 ± 3.1 years) Crossover within-subject design (tACS vs. sham vs. control (50 Hz) tACS) | Visual or auditory sustained attention tasks Correct responses, reaction times | Occipital cortex (Oz) + vertex (Cz) Device: Starstim (Neuroelectrics, Barcelona) | Frequency: 10 Hz/50 Hz/sham Intensity: 2 mA peak-to-peak Duration: 11 min Electrode size: both 25 cm2 (round-shaped) Timing: online | ↗ (with alpha-tACS) − (with gamma-tACS) Low risk |
| [35] | N = 23 (age range: 18–27 years) Crossover within-subject design (alpha tACS vs. gamma tACS vs. sham) | Selective spatial attention task (endogenous or exogenous) Reaction times | Inferior parietal lobe (P6) + vertex (Cz) Device: NeuroConn DC Stimulator Plus (NeuroCare, Ilmenau, Germany) | Frequency: 10 Hz/40 Hz/sham Intensity: 2 mA peak-to-peak Duration: Endogenous blocks: ~33 min Exogenous blocks: ~21 min Electrode size: target electrode 5 × 5 cm, return electrode 5 × 7 cm Timing: online | ↗ (with alpha-tACS) − (with gamma-tACS) Low risk |
| [36] | N = 20 (age range: 19–31 years) Crossover within-subject design (tACS vs. control vs. sham) | Dichotic Listening task Correct responses | Fronto-central and temporo-parietal cortex (FC5) + TP7 Device: NeuroConn DC Stimulator Plus (NeuroCare, Ilmenau, Germany) | Frequency: 10 Hz/47.1 Hz/sham Intensity: 1 mA peak-to-peak Duration: 25 min Electrode Size: both 7 cm2 (round-shaped, 3 cm in diameter) Timing: online | alpha-tACS: ↗ (for left-attend condition) ↘ (for right-attend condition) gamma-tACS: ↘ (for left-attend condition) ↗ (for right-attend condition) Low risk |
| [37] | N = 26 (mean age: 28.5 ± 7.9 years) Crossover within-subject design (tACS vs. sham) | Dichotic Listening task Correct responses | Multifocal approach: (FT9 + FT10/F3 + F4/CP5 + CP6/P5 + P6) Device: NeuroConn DC Stimulator Plus (NeuroCare, Ilmenau, Germany) | Frequency: 40 Hz/sham Intensity: 1 mA peak-to-peak Duration: 20 min Electrode Size: all electrodes round-shaped, 12 mm in diameter Timing: online | ↗ (for left ear-perception only) Low risk |
| Gamma | |||||
| [38] | N = 20 (mean age: 25.8 ± 6.2) Crossover within-subject design (40 Hz vs. 60 Hz vs. 80 Hz vs. sham) | Visual spatial attention task Correct responses | Occipital area (Oz) + vertex (Cz) Device: DC-Stimulator Plus (NeuroConn, Ilmenau, Germany) | Frequency: 40 Hz/60 Hz/80 Hz/sham Intensity: 1.5 mA peak-to-peak Duration: Exp. 1: 45 ± 10 min, Exp. 2: 15 ± 5 min Electrode size: target electrode 4 × 4 cm, return electrode 7 × 4 cm Timing: online | ↗ (with 60 Hz gamma-tACS) − (with other tACS) Low risk |
| Intelligence | |||||
| Theta | |||||
| [39] | N = 20 (mean age: 24.9 ± 3.3 years) Crossover within-subject design (tACS vs. sham) | Raven’s progressive matrices Correct responses | left temporal cortex (T7) + C3 Device: DC-Stimulator Plus (NeuroConn, Ilmenau, Germany) | Frequency: 5 Hz/sham Intensity: 1.5 mA peak-to-peak Duration: 15 min Electrode size: both 5 × 7 cm Timing: offline | ↗ (for difficult items only) Low risk |
| Alpha | |||||
| [40] | N = 20 (mean age: 20.18 ± 0.40) Crossover within-subject design (tACS vs. sham) | Raven’s progressive matrices Correct responses | Frontal cortex (F3) + F4 Device: NeuroConn DC Stimulator Plus (NeuroCare, Ilmenau, Germany) | Frequency: 1 Hz above IAF/sham Intensity: Modus = 1.75 mA peak-to-peak (Range = 1 mA to 2 mA) Duration: 15 min Electrode Size: both 7 × 5 cm Timing: offline | − Low risk |
| Alpha and Gamma | |||||
| [41] | N = 22 (mean age: 23.00 ± 2.85 years) Crossover within-subject design (alpha tACS vs. gamma tACS vs. sham) | Verbal intelligence task Correct responses | Frontal cortex (F3) + F4 Device: Eldith (NeuroConn GmbH, Ilmenau, Germany) | Frequency: 10 Hz/40 Hz/sham Intensity: 1 mA peak-to-peak Duration: 30 min incl. 100 cycles ramp-in/-out Electrode size: both 5 × 7 cm Timing: online | − Low risk |
| Gamma and Theta | |||||
| [42] | Exp. 1 N = 24 (mean age: 23.8 ± 3.14 years) Exp. 2 N = 34 (mean age: 24.3 ± 2.76 years) Crossover within-subject design (tACS vs. sham vs. control) | Modified version of Raven’s progressive matrices Correct responses, response times | Frontal cortex (left MFG) + vertex (Cz) Device: Eldith DC-Stimulator, NeuroConn, Germany | Frequency: Exp. 1 40 Hz/ 5 Hz/sham; Exp. 2 40 Hz/sham Intensity: 0.75 mA peak-to-peak Duration: ~30 min Electrode Size: both 5 × 5 cm Timing: online | ↗ (with gamma-tACS, benefit for low performer only) − (with other tACS) Low risk |
| Multiple Frequencies | |||||
| [43] | N = 20 (mean age: 20.2 ± 12.3 years) Crossover within-subject design (tACS vs. sham vs. control) | Raven’s progressive matrices Correct responses, response times | Frontal cortex (left MFG) + vertex (Cz) Device: Eldith DC-Stimulator, NeuroConn, Germany | Frequency: 5 Hz/10 Hz/20 Hz/40 Hz/sham Intensity: 0.75 mA peak-to-peak Duration: through task (~48 min), per condition Electrode Size: both 5 × 7 cm Timing: online | ↗ (with gamma-tACS) − (with other tACS) Low risk |
| Visual Perception | |||||
| Alpha | |||||
| [44] | N = 96, divided into 4 groups with each N = 24 6 Hz (mean age: 21 ± 2.4 years) 10 Hz (mean age: 21 ± 3.2 years) 25 Hz (mean age: 22 ± 2.1 years) sham (mean age: 22 ± 2.3 years) Between-subject design | Visual detection and discrimination task Correct responses | Parieto-occipital cortex (PO7/PO8) + vertex (Cz) Device: NeuroConn DC Stimulator (NeuroCare, Ilmenau, Germany) | Frequency: 6 Hz/10 Hz/25 Hz/sham Intensity: 1 mA peak-to-peak Duration: 5 min Electrode size: target electrode 16 cm2, return electrode 35 cm2 Timing: online | ↘ (with theta- or alpha-tACS) Low risk |
| [45] | N = 15 (mean age: 24.3 ± 0.7 years) Crossover within-subject design | Visual rotation detection task Correct responses | Visual cortex stimulation montage (Oz) + Cz Retinal stimulation montage (Fpz) + Cz Device: NeuroConn DC Stimulator Plus (NeuroCare, Ilmenau, Germany) | Frequency: IAF/IAF+4 Hz/IAF–4 Hz Intensity: Oz–Cz: 0.96 ± 0.3; Fpz–Cz: 2.31 ± 1.1 mA peak-to-peak Duration: 27 min (~5.4 s per frequency-montage combination) Electrode size: both 5 × 5 cm Timing: online | ↘ Low risk |
| Gamma | |||||
| [46] | N = 60 (mean age: 32.22 ± 7.96 years) 40 Hz (N = 12) 40 Hz (N = 16) 5 Hz (N = 16) sham (N = 16) Between-subject design | Face perception task, object perception task, Correct responses, reaction times | Occipital cortex (PO8) + frontal cortex (FP1) Device: Neuroelectrics, Barcelona, Spain | Frequency: 40 Hz/5 Hz/sham Intensity: 1.5 mA peak-to-peak Duration: 20 min Electrode size: both 25 cm2 Timing: offline | ↗ (with gamma-tACS) − (with theta-tACS) Low risk |
| [47] | N = 45 (mean age: 24.9 ± 4.1) Exp. 1 N = 17 Exp. 2 N = 13 Exp. 3 N = 15 All Exp. Crossover within-subject design | Stroboscopic alternative motion task Motion dominance index (% duration of perceived motion direction) | Exp. 1 and Exp. 2: parieto-occipital cortex (P7-PO7) + (P8-PO8) Exp. 3: Central (C3/C4) + occipital areas (O1/O2) Device: NeuroConn, Ilmenau, Germany | Frequency: Exp. 1 anti-phase 40 Hz tACS/sham; Exp. 2 antiphase 6 Hz/sham; Exp. 3 in-phase 6 Hz/40 Hz/sham Intensity: 0.5–1.5 mA peak-to-peak Duration: 15 min Electrode size: Exp. 1 and Exp. 2: both 5 × 7 cm; Exp. 3: both 3.9 × 3.9 cm Timing: online | ↘ (with anti-phase gamma-tACS) − (with other tACS) Low risk |
| Auditory Perception | |||||
| Delta/Theta | |||||
| [48] | N = 17 (mean age: 33 ± 8 years) Crossover within-subject design | Word detection task Correct responses | Left temporal cortex (T7) + C3 Device: DC-Stimulator MR (NeuroConn, Ilmenau, Germany) | Frequency: 3.125 Hz/sham Intensity: 1.7 mA peak-to-peak Duration: ~30 min Electrode size: target electrode 3 × 3 cm, return electrode 5 × 7 cm Timing: online | − High risk (small sample sizes) |
| [49] | Exp. 1 N = 27 (mean age: 31 ± 7 years) Exp. 2 N = 19 (mean age: 21 ± 2 years) Crossover within-subject design | Speech perception task Correct responses | Exp. 1 Unilateral montage: left temporal cortex (T7) + C3 Exp. 2 Bilateral montage: temporal cortex (T7/T8) + 2 large ring electrodes (around target electrode) Device: NeuroConn DC Stimulator MR (NeuroCare, Ilmenau, Germany) | Frequency: 3.125 Hz/sham Intensity: Exp. 1 1.2 mA peak-to-peak; Exp. 2 1.7 mA peak-to-peak Duration: ~30 min Electrode Size: both 5 × 7 cm Timing: online | Bilateral stimulation: ↗ (when stimulation and speech were aligned) ↘ (when stimulation and speech were not aligned) − (unilateral stimulation) Low risk for Exp. 1 High risk for Exp. 2 (small sample size) |
| Theta | |||||
| [50] | N = 20 (age range: 18–38 years) Crossover within-subject design | Stream perception task Correct responses | Temporal cortex (T7/T8) + L/R side of midline (Cz) Device: NeuroConn DC Stimulator (NeuroCare, Ilmenau, Germany) | Frequency: 4 Hz/sham Intensity: 0.8 ± 0.1 mA peak-to-peak Duration: 40 min Electrode Size: both target electrodes 5 × 5, both return electrodes 5 × 7 cm Timing: online | ↗ (when streams were synchronous with entrained oscillatory activity) Low risk |
| [51] | N = 22 (age range: 18–28 years) Crossover within-subject design | Speech perception task Correct responses | Temporal cortex (T7/T8) + Cz Device: DC-Stimulator MR (NeuroConn, Ilmenau, Germany) | Frequency: 4 Hz/sham Intensity: Exp. 1: 0.9 ± 0.1 mA peak-to-peak, Exp. 2: 1 ± 0.1 mA peak-to-peak Duration: 36 min Electrode size: both target electrodes 5 × 5 cm, return electrode 10 × 7 cm Timing: online | ↗ Low risk |
| Alpha and Beta | |||||
| [52] | N = 26 (mean age: 21.4 ± 4.7 years) Crossover within-subject design | Phonological decision task Correct responses, reaction times | Fronto-temporal cortex (Crossing point of T3-Fz × F7-Cz) + (T4-Fz × F8-Cz) Device: NeuroConn DC Stimulator (NeuroCare, Ilmenau, Germany) | Frequency: 10 Hz/16.18 Hz/sham Intensity: 1 mA peak-to-peak Duration: 20 min Electrode Size: both 9 cm2 (round-shaped) Timing: offline | With alpha-tACS: ↗ (reaction time) ↘ (error rates) − (with other tACS) Low risk |
| Gamma and Theta | |||||
| [53] | Exp. 1 N = 21 (mean age: 24.3 ± 2.0 years) Exp. 2 (control) N = 17 (mean age: 27.5 ± 3.3 years) All Exp. Crossover within-subject design | Phonetic categorization task Correct responses, perceptual learning | Exp. 1 only temporal areas (T7) + T8 Device: NeuroConn DC Stimulator (NeuroCare, Ilmenau, Germany) | Frequency: Exp. 1 40 Hz/6 Hz; Exp. 2 No stimulation Intensity: 1 mA peak-to-peak Duration: 18 min in total, in 6 min blocks Electrode Size: both 5 × 7 cm Timing: online | ↗ (with gamma-tACS in older adults) ↘ (with gamma-tACS in younger adults) − (with theta-tACS) Low risk |
| [54] | N = 25 (mean age: 24.1 ± 2.24 years) N = 20 (mean age: 69.8 ± 4.4 years) Between-subject/Crossover design | Phonetic categorization task Correct responses, perceptual learning | Temporal cortex (T7) + T8 Device: NeuroConn DC Stimulator (NeuroCare, Ilmenau, Germany) | Frequency: 40 Hz/6 Hz Intensity: 1 mA peak-to-peak Duration: 10 min Electrode Size: both 5 × 7 cm Timing: online | ↘ (with gamma-tACS) ↗ (with theta-tACS) Low risk |
| Executive functions | |||||
| Theta | |||||
| [55] | N = 23 (mean age: 22.91 years, range 18–30) Crossover within-subject design | Go/NoGo task Correct responses, reaction times | Fronto-temporal area (T4-Fz × F8-Cz) + supraorbital Device: NeuroConn DC Stimulator (NeuroCare, Ilmenau, Germany) | Frequency: 6 Hz/sham Intensity: 1 mA peak-to-peak Duration: 20 min Electrode Size: both 5 × 5 cm Timing: online/offline | − Low risk |
| [56] | N = 90 (N = 30 for each experiment) Exp. 1 mean age: 26 years Exp. 2 mean age: 27 years Exp. 3 mean age: 26 years Crossover within-subject design | Time estimation task Correct responses | Exp. 1/3 medial frontal and lateral prefrontal cortex (right: MFC/IPFC) + 4 surrounding return electrodes Exp. 2 similar to Exp. 1, but left hemisphere Device: M x N 9 high-definition Stimulator (Model 9002A, Soterix Medical, New York, NY) | Frequency: 6 Hz in-phase/anti-phase/35 Hz/sham Intensity: 1 mA peak-to-peak Duration: Exp. 1/2: 20 min; Exp. 3: 20 min Electrode size: high definition-tACS Timing: online | ↗ (tACS on right hemisphere) − (tACS on left hemisphere or gamma-tACS) Low risk |
| Theta and Alpha | |||||
| [57] | N = 31 (age range: 19-31 years) Crossover within-subject design | Simon task Correct responses, response times | left frontal cortex (between FCz and Cz) + both cheeks Device: DC-Stimulator MR (NeuroConn, Ilmenau, Germany) | Frequency: ITF, IAF Intensity: 2 mA peak-to-peak Duration: ~20 min Electrode size: target electrode 9 cm2, both return electrodes 35 cm2 Timing: online | ↗ (with theta-tACS) − (with alpha-tACS) Low risk |
| Alpha | |||||
| [58] | N = 20 (mean age: 26 ± 3 years) tACS (N = 10) sham (N = 10) Between-subject design | Mental rotation task Correct responses, reaction times | Occipital cortex (Oz) + vertex (Cz) Device: NeuroConn (NeuroCare, Ilmenau, Germany) | Frequency: 10.5 ± 0.9 Hz/sham Intensity: 0.7 ± 0.3 mA peak-to-peak Duration: 20 min Electrode size: target electrode 4 × 4 cm, return electrode 5 × 7 cm Timing: online | ↗ (accuracy) Low risk |
| Alpha and Beta | |||||
| [59] | N = 19 (mean age: 24 ± 4 years) Crossover within-subject design | Temporal bisection task Choice (long or short) Reaction times | SMA (FC1) + FC2 Device: NeuroConn MagStim (NeuroCare, Ilmenau, Germany) | Frequency: 10 Hz/20 Hz/sham Intensity: 1.5 mA peak-to-peak Duration: 20 min Electrode size: both 5 × 5 cm Timing: online | ↗ Low risk |
| Multiple Frequencies | |||||
| [60] | N = 36 (mean age: 24.4 ± 3.5 years) Crossover within-subject design | Flanker task Speed-accuracy-trade off and reaction times | Medial-frontal Cortex (FCz) + Pz Device: Neuroelectrics, Barcelona, Spain | Frequency: 2 Hz/6 Hz/11 Hz/21 Hz/60 Hz/sham Intensity: 1.5 mA peak-to-peak Duration: 20 min Electrode size: both 25 cm2 Timing: online | ↗ (with theta-tACS) − (with other tACS) Low risk |
| Decision-making | |||||
| Multiple Frequencies | |||||
| [61] | N = 34 in total left frontal montage: N = 17 (mean age: 20.52 ± 2.52 years) right frontal montage: N = 17 (mean age: 21.17 ± 2.78 years) Between-subject design | Decision making task Gains and losses and response times | Left frontal cortex (F3) + ipsilateral shoulder Right frontal cortex (F4) + ipsilateral shoulder Device: BrainStim, EMS Medical, Bologna, Italy | Frequency: 5Hz/10 Hz/20 Hz/40 HZ/sham Intensity: 1 mA peak-to-peak Duration: 40 min Electrode Size: both 5 × 7 cm Timing: online | ↘ (with beta-tACS to left hemisphere = riskier decisions) ↗ (with tACS to the right hemisphere) Low risk |
| Procedural Memory (motor learning) | |||||
| Theta | |||||
| [62] | N = 26 (mean age: 21.38 ± 1.52 years) Crossover within-subject design | Implicit motor learning task Correct responses, reaction times | Frontal area (FPz) + Pz Device: NeuroConn DC Stimulator Plus (NeuroCare, Ilmenau, Germany) | Frequency: 6 Hz/sham Intensity: 1 mA peak-to-peak Duration: 20 min Electrode Size: both 5 × 5 cm Timing: online | − Low risk |
| Alpha | |||||
| [63] | N = 20 (mean age: 23.8 ± 3.90 years) N = 15 (mean age: 61.66 ± 3.71 years) Between-subject design (older vs. younger participants)/Crossover within-subject design | Implicit motor learning task Correct responses, reaction times | Central area (C3) + supraorbital region Device: Eldith DC Stimulator, NeuroConn ((NeuroCare, Ilmenau, Germany) | Frequency: IAF/IAF + 2 Hz/sham Intensity: 1.5 mA peak-to-peak Duration: 10 min Electrode Size: target electrode 5 × 7 cm, return electrode 10 × 10 cm Timing: offline | − (accuracy) Reaction times: ↗ (in older adults with IAF and IAF + 2 Hz) ↗ (in younger adults with IAF + 2 Hz only) Low risk |
| Alpha and Beta | |||||
| [64] | N = 38 (mean age: 23 ± 3 years) Crossover within-subject design | Movement selection task Duration between stimulus onset and verbal responses | Central area (C3 or C4) + Pz Device: NeuroConn DC Stimulator Plus (NeuroCare, Ilmenau, Germany) | Frequency: 10 Hz/20 Hz/sham Intensity: 1.1 ± 0.3 mA peak-to-peak (range: 0.4–1.9 mA) Duration: ~30 min Electrode Size: both 5 × 5 cm Timing: online | ↗ (with alpha-tACS ipsilateral to requested hand) − (with beta-tACS) Low risk |
| Beta and Gamma | |||||
| [65] | N = 20 (mean age: 24.9 years) Crossover within-subject design | Cued reaction time task Reaction times | Central area (left M1) + supraorbital region Device: NeuroConn DC Stimulator (NeuroCare, Ilmenau, Germany); BiStim (MagStim) | Frequency: IBF (or fixed 20.1 Hz)/75 Hz/sham Intensity: 0.69 ± 0.11 mA peak-to-peak; 1.3 ± 0.36 mA peak-to-peak Duration: 20 min Electrode Size: both 5 × 7 cm Timing: online | ↗ (with gamma-tACS) − (with beta-tACS) Low risk |
| Gamma | |||||
| [66] | N = 17 (mean age: 24.5 ± 3.5 years) Crossover within-subject design | Implicit motor learning task Reaction times | Central area (left M1) + right supraorbital region Device: NeuroConn DC Stimulator (E.M.S., Bologna, Italy) | Frequency: 1 Hz/40 Hz/sham Intensity: 2 mA peak-to-peak Duration: 5.08 ± 1.13 min Electrode Size: both 5 × 5 cm Timing: online | ↘ (with gamma tACS) − (with other tACS) Low risk |
| Multiple Frequencies | |||||
| [67] | N = 55 (mean age: 32.7 ± 6.8 years) Crossover within-subject design | Implicit motor learning task Correct responses, Reaction times | Central area (C3) + supraorbital Device: NeuroConn DC Stimulator Plus (NeuroCare, Ilmenau, Germany) | Frequency: 10 Hz/20 Hz/70 Hz/sham Intensity: 1 mA peak-to-peak Duration: 10 min Electrode Size: both 5 × 7 cm Timing: offline | ↗ (with gamma-tACS) − (with other tACS) Low risk |
| Working memory | |||||
| Theta | |||||
| [68] | N = 25 (mean age: 23.5 ± 2.9 years) Crossover within-subject design | N-back task Correct responses, reaction times | Frontal/parietal cortex Sync: AF3/P3 in-phase + AF4/P4 in-phase Desync: AF3/AF4 in-phase + P3/P4 in-phase Device: Starstim (Neuroelectrics, Barcelona) | Frequency: 6 Hz/sham Intensity: 2 mA peak-to-peak Duration: 17–19 min Electrode Size: 3.14 cm2 (round-shaped) Timing: online | ↘ (with anti-phase tACS) − (with in-phase tACS) Low risk |
| [69] | Exp. 1 N = 18 (mean age: 29.8 ± 8.3 years) Exp. 2 N = 14 (mean age: 21.9 ± 5.9 years) Crossover within-subject design | Delayed match-to-sample task Working memory capacity | Parietal cortex (P4) + (Cz/Oz/T8) Device: Starstim (Neuroelectrics, Barcelona) | Frequency: 4 Hz/7 Hz/sham Intensity: Exp. 1 1 mA peak-to-peak; Exp. 2 2 mA peak-to-peak Duration: at least 6 min per condition Electrode Size: target electrode 19.6 cm², each return electrode 4.9 cm² Timing: online | ↗ (with low theta-tACS, for items presented contralateral to stimulation site only) − (with high theta-tACS) Low risk for Exp. 1 High risk for Exp. 2 (small sample size) |
| [70] | N = 36 (mean age: 20 years ± 4.25 months) Between-subject design | Corsi block-tapping task, Digit-span task, N-back task Working memory capacity, correct responses (n-back only) | Left parietal cortex (P3) + supraorbital region Frontal cortex (F3) + supraorbital region Right parietal cortex (P4) + supraorbital region Device: NeuroConn DC Stimulator (NeuroCare, Ilmenau, Germany) | Frequency: ITF/sham Intensity: 1–2.25 mA peak-to-peak Duration: 15 min Electrode Size: both 5 × 7 cm Timing: offline | ↗ Low risk (but imbalanced gender distribution: 27 females) |
| [71] | N = 48 (mean age: 23 years) Crossover within-subject design | Visuo-spatial working memory task Working memory capacity | Parietal cortex (P3/P4) + left cheek Exp. 1 in-phase between P3 and P4 Exp. 2 anti-phase between P3 and P4 Device: NeuroConn DC Stimulator MC (NeuroCare, Ilmenau, Germany) | Frequency: 6 Hz/sham Intensity: 1.6 mA peak-to-peak Duration: 20–24 min Electrode Size: target electrode 4 × 4 cm, return electrode 5 × 7 cm Timing: online | ↗ (with in-phase for low performer) − (with anti-phase or in-phase for high performer) Low risk |
| [72] | N = 46 (age range: 22–30 years) Exp. 1: N = 10 Exp. 2: N = 18 Exp. 3 (control): N = 18 Crossover within-subject design | Letter discrimination task Reaction times | Exp. 1: no stimulation Exp. 2/3: Frontal/parietal cortex (F3/P3) + Vertex (Cz) Device: NeuroConn (NeuroCare, Ilmenau, Germany) | Frequency: Exp. 1: no stimulation; Exp. 2: 6 Hz in-phase or anti-phase/sham; Exp. 3: 35 Hz in-phase or anti-phase/sham Intensity: Exp. 2/3 1 mA peak-to-peak Duration: Exp. 2/3 14 ± 1.5 min Electrode size: Exp. 2/3 all 5 × 5 cm Timing: Exp. 2/3 online | ↗ (with in-phase theta-tACS) − (with anti-phase theta- or gamma-tACS) Low risk |
| [73] | Exp. 1 N = 10 (mean age: 28.6 ± 5.0 years) Exp. 2 N = 21 (mean age: 27.38 ± 4.56 years) Crossover within-subject design | Working memory tasks Exp. 1 2-back/1-back vs. choice reaction task Exp. 2 2-back vs. choice reaction task + MRT Correct responses, reaction times | Exp. 1 and Exp. 2 frontal/parietal areas (F4/P4) + T8 All Exp. either in-phase or anti-phase between F4 and P4 Device: NeuroConn DC Stimulator MR (NeuroCare, Ilmenau, Germany) | Frequency: 6 Hz/sham Intensity: 1 mA peak-to-peak Duration: Exp. 1: 26.5 min; Exp. 2: 20 min in short runs of 20 s or 30 s Electrode Size: all 5 cm diameter (‘donut’-shaped) Timing: online | ↗ (for choice reaction task with in-phase tACS) − (accuracy) High risk in Exp. 1 (small sample size) Low risk in Exp. 2 |
| [74] | N = 2 × 16 (mean age: 28.3 years (±7.6)/22.8 years (±5.2) Between-subject design (experimental vs. control montage)/Crossover within-subject design | Visuo-spatial working memory task Correct responses, K-Value (Load x (Hits – False)) | Target stimulation: Parietal cortex (P4) + supraorbital region Active control: Parietal cortex (P4) + Vertex (Cz) Device: Magstim DC-Stimulator Plus | Frequency: 4 Hz/7 Hz/control (5.5 Hz) Intensity: 1.24 ± 0.3 mA peak-to-peak Duration: ~12 min Electrode Size: both 5 × 5 cm Timing: online | for arrays pointing to the left: ↗ (with 4 Hz-tACS) ↘ (with 7 Hz-tACS) − (for arrays pointing to the right) Low risk |
| Alpha | |||||
| [75] | N = 22 (mean age: 24.1 years) Between-subject design | Spatial updating paradigm Updating bias | Left parieto-occipital cortex (P3-O1) + vertex (Cz) Right parieto-occipital cortex (P4-O2) + vertex (Cz) Device: NeuroConn (NeuroCare, Ilmenau, Germany) | Frequency: 10 Hz Intensity: 1 mA peak-to-peak Duration: 25 min Electrode size: target electrode 4 × 3 cm, return electrode 9 × 5 cm Timing: online | ↗ Low risk |
| Gamma | |||||
| [76] | N = 18 (mean age: 29.3 ± 7.65 years) Crossover within-subject design | N-back tasks Correct responses, reaction times | Frontal cortex (F3) + subraorbital region Device: NeuroConn DC Stimulator Plus (NeuroCare, Ilmenau, Germany) | Frequency: 40 Hz/tDCS/sham Intensity: 1.5 mA peak-to-peak Duration: 20 min Electrode Size: both 5 × 7 cm Timing: online | ↗ (for 3-back condition when applied offline) − (with online-tACS, as well as for the 1- and 2-back condition) High risk (small sample size) |
| Gamma and Theta | |||||
| [77] | Exp. 1 N = 20 (mean age: 21 years) Exp. 2 N = 20 (mean age: 23 years) Control 1 N = 20 (mean age: 23 years) Control 2 N = 20 (mean age: 23 years) Crossover within-subject design | Visuo-spatial working memory task d′ | Centro-to-temporal areas (CP1/T5) + right cheek Exp. 1 anti-phase between CP1 and T5 Exp. 2 in-phase between CP1 and T5 Control 1 in-phase between CP1 and T5 Control 2 anti-phase between CP1 and T5 Device: NeuroConn DC Stimulator MC (NeuroCare, Ilmenau, Germany) | Frequency: Exp. 1/2: 40 Hz/sham; Control 1/2: 6 Hz/sham Intensity: 1.5 mA peak-to-peak Duration: 20 min Electrode Size: both target electrodes 5 × 5 cm; return electrode 5 × 7 cm Timing: online | ↗ (with anti-phase gamma-tACS) − (with in-phase gamma-tACS or theta-tACS) Low risk |
| Multiple Frequencies | |||||
| [78] | N = 25 (mean age: 69.1 ± 4.5 years) Exp. 1 no stimulation Exp. 2/3 Crossover within-subject design | Retro-cue working memory paradigm Correct responses | Parietal cortex (P3) + P4 | Frequency: Exp. 1: no stimulation; Exp. 2: 4 Hz/10 Hz/40 Hz; Exp. 3: 10 Hz/sham Intensity: 1.5 mA peak-to-peak Duration: 20 min Electrode Size: both 5 × 7 cm Timing: online | ↗ (with alpha-tACS but only in Exp. 2, no replication in Exp. 3) − (with other tACS) Low risk |
| Declarative memory | |||||
| Theta | |||||
| [79] | N = 12 young (YA, mean age: 22.3 ± 1.5 years) N = 12 older adults (OA, mean age: 66.3 ± 3.9 years) Crossover within-subjects design | Language-learning paradigm Correct responses | Centro-parietal area (CP5) + supraorbital region Device: NeuroConn (NeuroCare, Ilmenau, Germany) | Frequency: 6 Hz/sham Intensity: 1 mA peak-to-peak Duration: 20 min Electrode size: target electrode 5 × 7 cm, return electrode 10 × 10 cm Timing: online | ↗ (in older adults) − (in younger adults) High risk (small sample size) |
| [80] | N = 72, (N = 24 for each experiment) Exp. 1 (mean age: 23.5 ± 3.1 years) Exp. 2 (mean age: 24.3 ± 2.9 years) Exp. 3 (mean age: 23.2 ± 2.2 years) Between-subject design | Paired-associative learning task Correct responses | Left temporal cortex (T7) + (FPz/T8) Device: NeuroConn (NeuroCare, Ilmenau, Germany) | Frequency: 5 Hz modulated bursts of 80 Hz gamma/sham Intensity: 1 mA peak-to-baseline Duration: 10 min Electrode size: all 3 cm² Timing: online | ↘ (with gamma-bursts coupled with troughs of theta-waves) − (with every other coupling) Low |
| [81] | N = 28 (mean age: 71.2 ± 6.4 years) Crossover within-subject design | Face-occupation task (Paired-associative learning) Correct responses | Ventrolateral PFC (intersection of T3-F3 and F7-C3 and midpoint F7-F3) + supraorbital region Device: NeuroConn (NeuroCare, Ilmenau, Germany) | Frequency: 5 Hz/sham Intensity: 1 mA peak-to-peak Duration: 20 min Electrode size: target electrode 5 × 7 cm, return electrode 10 × 10 cm Timing: online | − Low risk |
| [82] | N = 25 (age range: 18–28 years) Crossover within-subjects design | Short-term recognition task Correct responses Long-term memory recognition task Strength of recollection and familiarity | Target stimulation: Right parietal cortex (P4) + (T8/C2/CP1/Oz) Active control: Left parietal cortex (P3) + (T7/C1/CP2/Oz) Device: Multi-channel stimulator (StarStim, Neuroelectrics, Barcelona, Spain) | Frequency: 4 Hz/sham Intensity: 3 mA peak-to-peak Duration: 20 min Electrode size: 5 round-shaped electrodes with 1 cm radius Timing: online | ↗ (with theta-tACS; for familiarity) Low risk |
| [83] | N = 60 (age range: 18–45 years) N = 19 (mean age: 28.4 ± 6.9 years) N = 21 (mean age: 25.3 ± 5.6 years) N = 19 (mean age: 24.9 + 4.7 years) Between-subject design | Visual associative memory task Correct responses | Right fusiform cortex (P10) + (FP1/P2/P3/PO7) Device: Soterix MxN TES device (Soterix Medical Inc., New York, USA) | Frequency: 6 Hz/sham Intensity: 2 mA peak-to-baseline Duration: 10 min Electrode size: HD-tES electrodes Timing: online | ↗ (accuracy and forgetting) Low risk |
| Gamma | |||||
| [84] | N = 70 (mean age: 22.12 + 2.16 years) Congruent group 60-60 (N = 17) Congruent group 90-90 (N = 18) Incongruent group 60-90 (N = 17) Incongruent group 90-60 (N = 18) Between-subject design/Crossover within-subjects design | Word-list learning Correct responses; d’; Reaction times | Dorsolateral prefrontal cortex (F3) + left wrist Device: NeuroConn DC Brain Stimulator Plus (NeuroCare, Ilmenau, Germany) | Frequency: 60 Hz/90 Hz/sham Intensity: 1.5 mA peak-to-peak Duration: 15 min during encoding, 15 min during retrieval Electrode size: both 5 × 7 cm Timing: online | ↗ (with the same gamma-tACS during encoding and retrieval) − (with different gamma-tACS during encoding and retrieval) Low risk |
| [85] | N = 36 (mean age: 21.3 ± 0.5 years) N = 18 (mean age: 21.2 ± 0.4 years) N = 18 (mean age: 21.3 ± 0.5 years) Between-subject design, mixed with repeated-measures within-subject design | Episodic memory task (Word learning and recognition) Correct responses, d’, Reaction times | PFC (F3) + left wrist Device: Eldith Brain Stimulator (NeuroCare, Ilmenau, Germany) | Frequency: 60 Hz/sham Intensity: 0.75 mA peak-to-peak Duration: 15 min during encoding, 15 min during retrieval Electrode size: both 5 × 7 cm Timing: online | ↗ (accuracy) − (reaction times) Low risk |
| [86] | Exp. 1 N = 36 (mean age: 20.03 ± 2.38) Exp. 2 N = 36 (mean age: 20.97 ± 2.22) Crossover within-subject design | Episodic memory task (Word and face learning and recognition) Correct responses | Left IFG (FP1) + C5 Right IFG (FP2) + C6 Device: 4-channel DC Stimulator MC (NeuroCare, Ilmenau, Germany) | Frequency: 18.5 Hz/control frequencies: 6.8 Hz, 10.7 Hz, 30 Hz, 48 Hz/sham Intensity: Exp. 1: 2 mA peak-to-peak Exp. 2: 1.6 mA peak-to-peak Duration: event-related stimulation for 2 s during each stimulus presentation (2 s × 360 = 720 s total stimulation duration) Electrode size: Exp. 1: 4 donut-shaped electrodes, each 14 cm2; Exp. 2: 4 round electrodes, each 10.75 cm2 Timing: online | − Low risk |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Klink, K.; Paßmann, S.; Kasten, F.H.; Peter, J. The Modulation of Cognitive Performance with Transcranial Alternating Current Stimulation: A Systematic Review of Frequency-Specific Effects. Brain Sci. 2020, 10, 932. https://doi.org/10.3390/brainsci10120932
Klink K, Paßmann S, Kasten FH, Peter J. The Modulation of Cognitive Performance with Transcranial Alternating Current Stimulation: A Systematic Review of Frequency-Specific Effects. Brain Sciences. 2020; 10(12):932. https://doi.org/10.3390/brainsci10120932
Chicago/Turabian StyleKlink, Katharina, Sven Paßmann, Florian H. Kasten, and Jessica Peter. 2020. "The Modulation of Cognitive Performance with Transcranial Alternating Current Stimulation: A Systematic Review of Frequency-Specific Effects" Brain Sciences 10, no. 12: 932. https://doi.org/10.3390/brainsci10120932
APA StyleKlink, K., Paßmann, S., Kasten, F. H., & Peter, J. (2020). The Modulation of Cognitive Performance with Transcranial Alternating Current Stimulation: A Systematic Review of Frequency-Specific Effects. Brain Sciences, 10(12), 932. https://doi.org/10.3390/brainsci10120932






