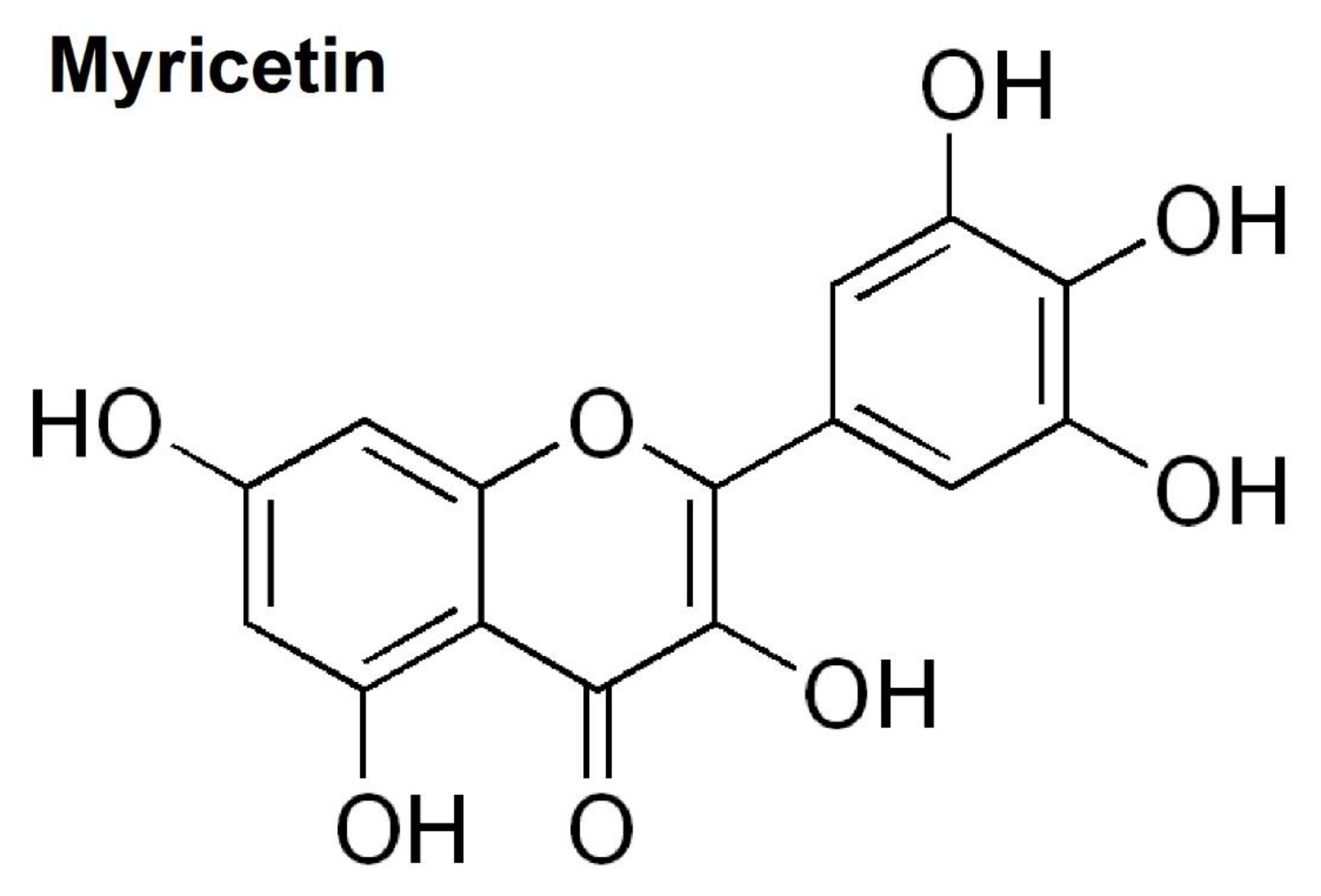
Inhibitory Effects of Myricetin on Lipopolysaccharide-Induced Neuroinflammation
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals and Reagents
2.2. Cell Culture and Cytotoxicity Measurements
2.3. Measurement of Nitric Oxide (NO), MAPK Signaling, TNF-α, IL-1β, PGE2, iNOS, and COX-2 Levels
2.4. Animals
2.5. Drug Administration
2.6. Tissue Preparation and Immunohistochemistry
2.7. Statistical Analysis
3. Results
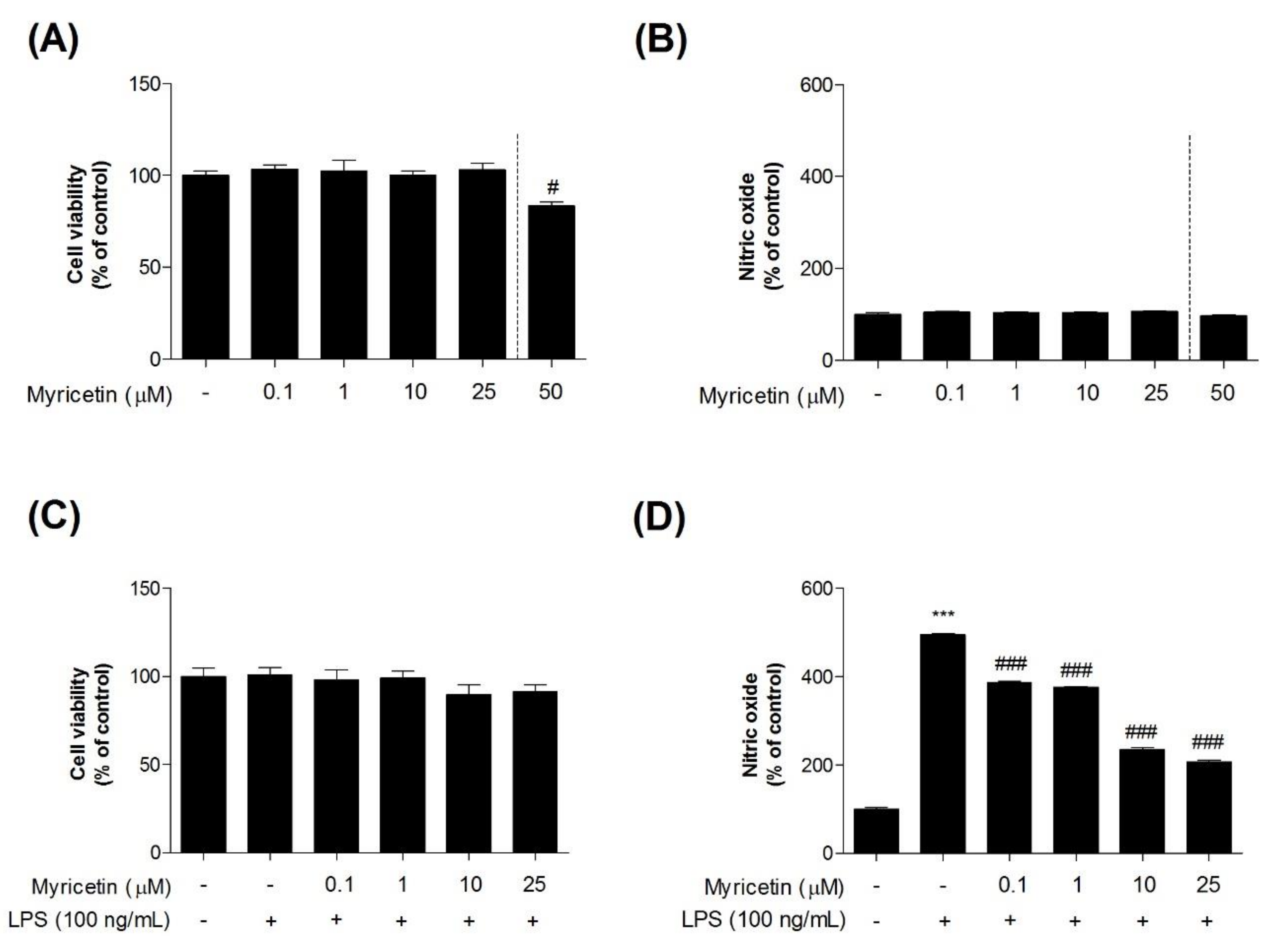
3.1. Effects of Myricetin on LPS-Induced Cytotoxicity and NO Generation in Microglia BV2 Cells
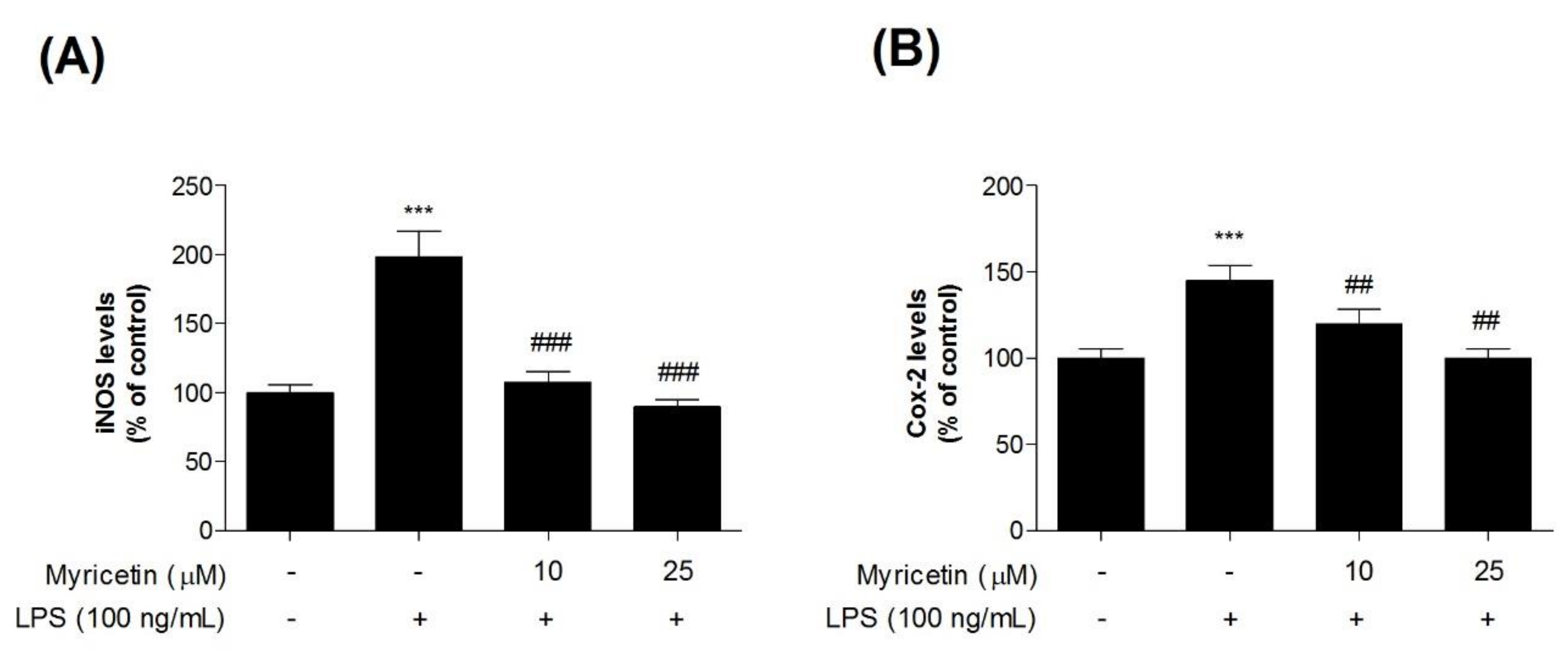
3.2. Inhibitory Effects of Myricetin on LPS-Induced iNOS and COX-2 Levels
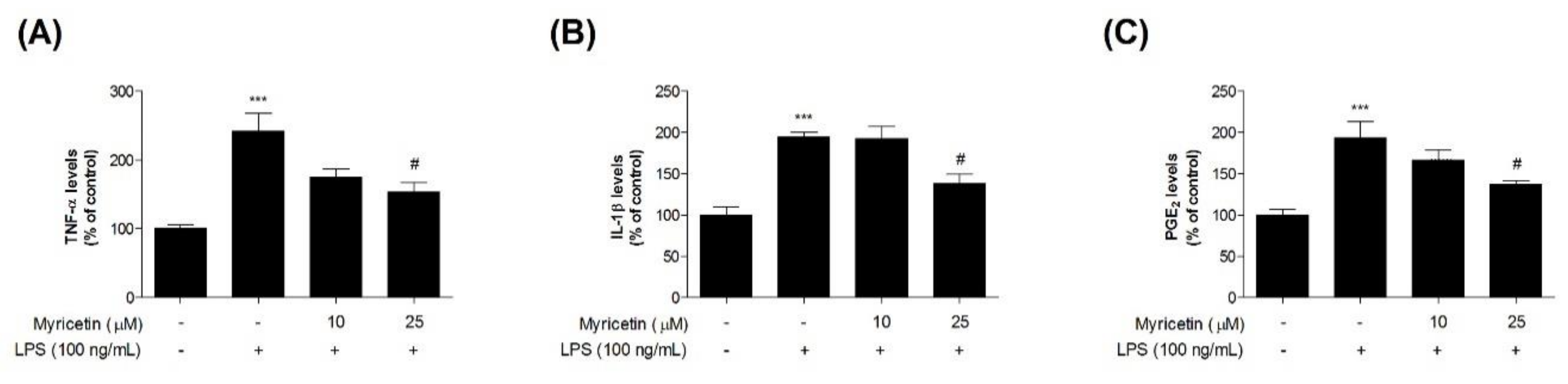
3.3. Inhibitory Effects of Myricetin on LPS-Induced TNF-α, IL-1β, and PGE2 Levels
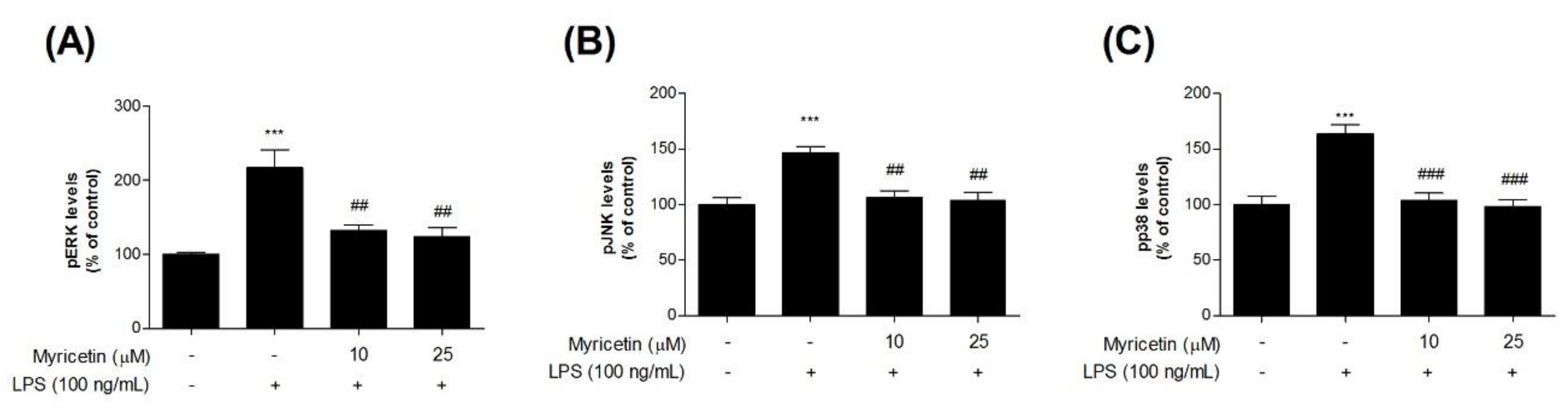
3.4. Inhibitory Effects of Myricetin on LPS-Induced Phosphor-MAPKs Signaling ERK, JNK, and p38 Levels
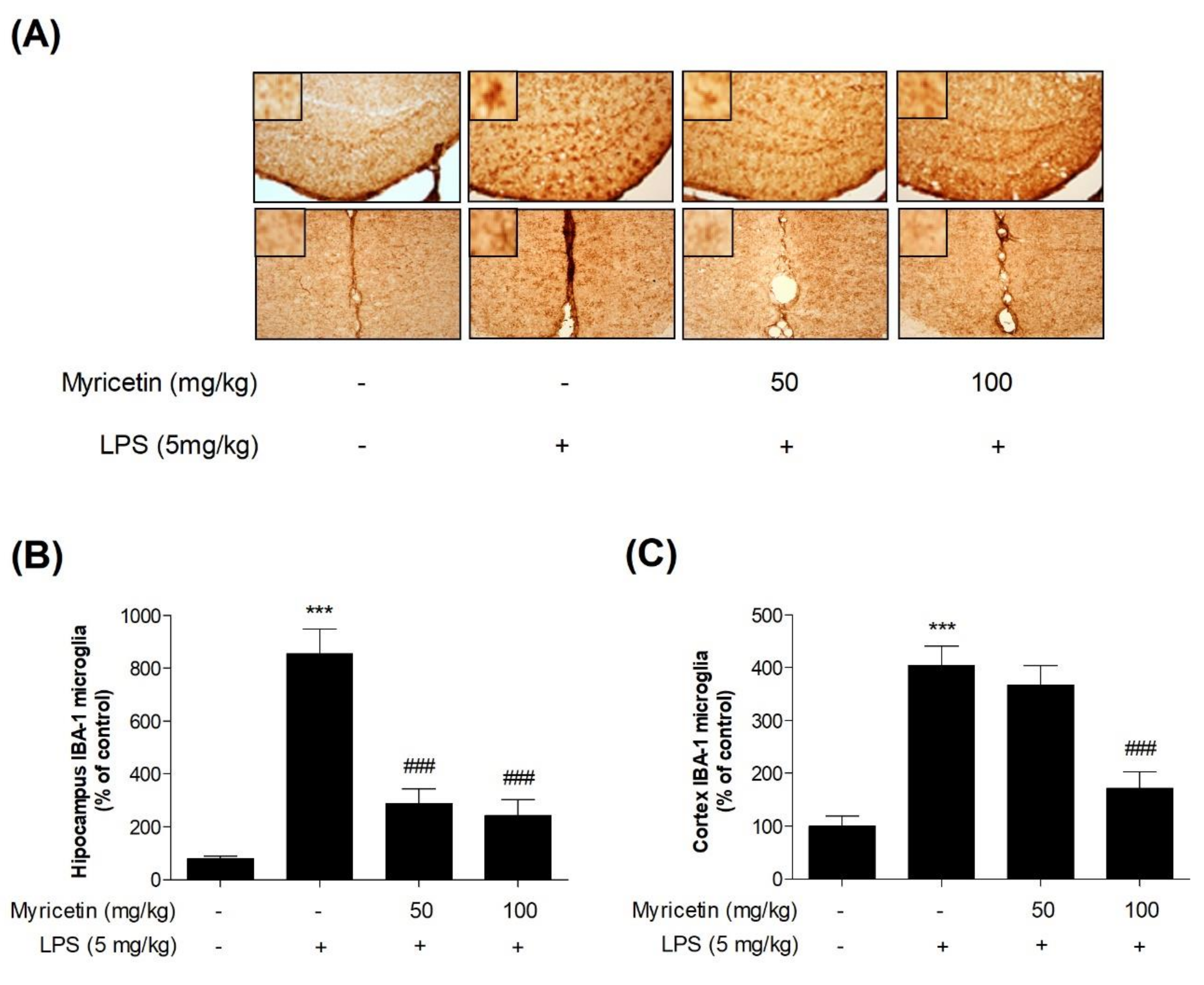
3.5. Inhibitory Effects of Myricetin on LPS-Induced Activation of Microglia in Hippocampus and Cortex in Mice
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Block, M.L.; Hong, J.S. Microglia and inflammation-mediated neurodegeneration: Multiple triggers with a common mechanism. Prog. Neurobiol. 2005, 76, 77–98. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.C.; Quang, T.H.; Oh, H.; Kim, Y.C. Steppogenin Isolated from Cudrania tricuspidata Shows Antineuroinflammatory Effects via NF-kappaB and MAPK Pathways in LPS-Stimulated BV2 and Primary Rat Microglial Cells. Molecules 2017, 22, 2130. [Google Scholar] [CrossRef] [PubMed]
- Stansley, B.; Post, J.; Hensley, K. A comparative review of cell culture systems for the study of microglial biology in Alzheimer’s disease. J. Neuroinflamm. 2012, 9, 115. [Google Scholar] [CrossRef] [PubMed]
- Muhammad, T.; Ikram, M.; Ullah, R.; Rehman, S.U.; Kim, M.O. Hesperetin, a Citrus Flavonoid, Attenuates LPS-Induced Neuroinflammation, Apoptosis and Memory Impairments by Modulating TLR4/NF-kappaB Signaling. Nutrients 2019, 11, 648. [Google Scholar] [CrossRef]
- Liu, S.Y.; Xu, P.; Luo, X.L.; Hu, J.F.; Liu, X.H. (7R,8S)-Dehydrodiconiferyl Alcohol Suppresses Lipopolysaccharide-Induced Inflammatory Responses in BV2 Microglia by Inhibiting MAPK Signaling. Neurochem. Res. 2016, 41, 1570–1577. [Google Scholar] [CrossRef]
- Block, M.L.; Zecca, L.; Hong, J.S. Microglia-mediated neurotoxicity: Uncovering the molecular mechanisms. Nat. Rev. Neurosci. 2017, 8, 57–69. [Google Scholar] [CrossRef]
- Park, J.; Min, J.S.; Kim, B.; Chae, U.B.; Yun, J.W.; Choi, M.S.; Kong, I.K.; Chang, K.T.; Lee, D.S. Mitochondrial ROS govern the LPS-induced pro-inflammatory response in microglia cells by regulating MAPK and NF-kappaB pathways. Neurosci. Lett. 2015, 584, 191–196. [Google Scholar] [CrossRef]
- Colonna, M.; Butovsky, O. Microglia Function in the Central Nervous System During Health and Neurodegeneration. Annu. Rev. Immunol. 2017, 35, 441–468. [Google Scholar] [CrossRef]
- Gertig, U.; Hanisch, U.K. Microglial diversity by responses and responders. Front. Cell. Neurosci. 2014, 8, 101. [Google Scholar] [CrossRef]
- Majid, M.; Khan, M.R.; Shah, N.A.; Haq, I.U.; Farooq, M.A.; Ullah, S.; Sharif, A.; Zahra, Z.; Younis, T.; Sajid, M. Studies on phytochemical, antioxidant, anti-inflammatory and analgesic activities of Euphorbia dracunculoides. BMC Complement. Altern. Med. 2015, 15, 349. [Google Scholar] [CrossRef]
- Semwal, D.K.; Semwal, R.B.; Combrinck, S.; Viljoen, A. Myricetin: A Dietary Molecule with Diverse Biological Activities. Nutrients 2016, 8, 90. [Google Scholar] [CrossRef] [PubMed]
- Sun, L.; Xu, P.; Fu, T.; Huang, X.; Song, J.; Chen, M.; Tian, X.; Yin, H.; Han, J. Myricetin against ischemic cerebral injury in rat middle cerebral artery occlusion model. Mol. Med. Rep. 2018, 17, 3274–3280. [Google Scholar] [CrossRef] [PubMed]
- Ma, Z.; Wang, G.; Cui, L.; Wang, Q. Myricetin Attenuates Depressant-Like Behavior in Mice Subjected to Repeated Restraint Stress. Int. J. Mol. Sci. 2015, 16, 28377–28385. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.M.; Wang, G.L.; Ma, Z.G. Protective effects of myricetin on chronic stress-induced cognitive deficits. Neuroreport 2016, 27, 652–658. [Google Scholar] [CrossRef]
- Zhang, K.; Ma, Z.; Wang, J.; Xie, A.; Xie, J. Myricetin attenuated MPP(+)-induced cytotoxicity by anti-oxidation and inhibition of MKK4 and JNK activation in MES23.5 cells. Neuropharmacology 2011, 61, 329–335. [Google Scholar] [CrossRef]
- Ma, Z.G.; Wang, J.; Jiang, H.; Liu, T.W.; Xie, J.X. Myricetin reduces 6-hydroxydopamine-induced dopamine neuron degeneration in rats. Neuroreport 2007, 18, 1181–1185. [Google Scholar] [CrossRef]
- Dhanraj, V.; Karuppaiah, J.; Balakrishnan, R.; Elangovan, N. Myricetin attenuates neurodegeneration and cognitive impairment in Parkinsonism. Front. Biosci. 2018, 10, 481–494. [Google Scholar]
- Lim, H.S.; Kim, J.S.; Moon, B.C.; Choi, G.; Ryu, S.M.; Lee, J.; Ang, M.J.; Jeon, M.; Moon, C.; Park, G. Cicadidae Periostracum, the Cast-Off Skin of Cicada, Protects Dopaminergic Neurons in a Model of Parkinson’s Disease. Oxid. Med. Cell. Longev. 2019, 2019, 5797512. [Google Scholar] [CrossRef]
- Nam, K.N.; Choi, Y.S.; Jung, H.J.; Park, G.H.; Park, J.M.; Moon, S.K.; Cho, K.H.; Kang, C.; Kang, I.; Oh, M.S.; et al. Genipin inhibits the inflammatory response of rat brain microglial cells. Int. Immunopharmacol. 2010, 10, 493–499. [Google Scholar] [CrossRef]
- Sonar, S.A.; Lal, G. The iNOS Activity During an Immune Response Controls the CNS Pathology in Experimental Autoimmune Encephalomyelitis. Front. Immunol. 2019, 10, 710. [Google Scholar] [CrossRef]
- Pautz, A.; Art, J.; Hahn, S.; Nowag, S.; Voss, C.; Kleinert, N. Regulation of the expression of inducible nitric oxide synthase. Nitric Oxide 2010, 23, 75–93. [Google Scholar] [CrossRef] [PubMed]
- Minghetti, L. Role of COX-2 in inflammatory and degenerative brain diseases. Sub-Cell. Biochem. 2007, 42, 127–141. [Google Scholar]
- Murphy, S. Production of nitric oxide by glial cells: Regulation and potential roles in the CNS. Glia 2000, 29, 1–13. [Google Scholar] [CrossRef]
- Minghetti, L. Cyclooxygenase-2 (COX-2) in inflammatory and degenerative brain diseases. J. Neuropathol. Exp. Neurol. 2004, 63, 901–910. [Google Scholar] [CrossRef] [PubMed]
- Shi, S.; Liang, D.; Chen, Y.; Xie, Y.; Wang, Y.; Wang, L.; Wang, Z.; Qiao, Z. Gx-50 reduces beta-amyloid-induced TNF-alpha, IL-1beta, NO, and PGE2 expression and inhibits NF-kappaB signaling in a mouse model of Alzheimer’s disease. Eur. J. Immunol. 2016, 46, 665–676. [Google Scholar] [CrossRef]
- Kim, E.K.; Choi, E.J. Compromised MAPK signaling in human diseases: An update. Arch. Toxicol. 2015, 89, 867–882. [Google Scholar] [CrossRef]
- Peti, W.; Page, R. Molecular basis of MAP kinase regulation. Protein Sci. 2013, 22, 1698–1710. [Google Scholar] [CrossRef]
- Arthur, J.S.; Ley, S.C. Mitogen-activated protein kinases in innate immunity. Nat. Rev. Immunol. 2013, 13, 679–692. [Google Scholar] [CrossRef]
- Sabio, G.; Davis, R.J. TNF and MAP kinase signalling pathways. Semin. Immunol. 2014, 26, 237–245. [Google Scholar] [CrossRef]
- Hou, W.; Hu, S.; Su, Z.; Wang, Q.; Meng, G.; Guo, T.; Zhang, J.; Gao, P. Myricetin attenuates LPS-induced inflammation in RAW 264.7 macrophages and mouse models. Future Med. Chem. 2018, 10, 2253–2264. [Google Scholar] [CrossRef]
- Kang, K.A.; Wang, Z.H.; Zhang, R.; Piao, M.J.; Kim, K.C.; Kang, S.S.; Kim, Y.W.; Lee, J.; Park, D.; Hyun, J.W. Myricetin protects cells against oxidative stress-induced apoptosis via regulation of PI3K/Akt and MAPK signaling pathways. Int. J. Mol. Sci. 2010, 11, 4348–4360. [Google Scholar] [CrossRef] [PubMed]
- Kempuraj, D.; Thangavel, R.; Natteru, P.A.; Selvakumar, G.P.; Saeed, D.; Zahoor, H.; Zaheer, S.; Iyer, S.S.; Zaheer, A. Neuroinflammation Induces Neurodegeneration. J. Neurol. Neurosurg. Spine 2016, 1, 1003. [Google Scholar] [PubMed]
- Stolmeier, D.; Thangavel, R.; Anantharam, P.; Khan, M.M.; Kempuraj, D.; Zaheer, A. Glia maturation factor expression in hippocampus of human Alzheimer’s disease. Neurochem. Res. 2013, 38, 1580–1589. [Google Scholar] [CrossRef] [PubMed]
- Ramezani, M.; Darbandi, N.; Khodagholi, F.; Hashemi, A. Myricetin protects hippocampal CA3 pyramidal neurons and improves learning and memory impairments in rats with Alzheimer’s disease. Neural Regen. Res. 2016, 11, 1976–1980. [Google Scholar] [CrossRef]
- Chang, Y.; Chang, C.Y.; Wang, S.J.; Huang, S.K. Myricetin inhibits the release of glutamate in rat cerebrocortical nerve terminals. J. Med. Food 2015, 18, 516–523. [Google Scholar] [CrossRef]
- Yao, Y.; Lin, G.; Xie, Y.; Ma, P.; Li, G.; Meng, Q.; Wu, T. Preformulation studies of myricetin: A natural antioxidant flavonoid. Die Pharm. 2014, 69, 19–26. [Google Scholar]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jang, J.-H.; Lee, S.H.; Jung, K.; Yoo, H.; Park, G. Inhibitory Effects of Myricetin on Lipopolysaccharide-Induced Neuroinflammation. Brain Sci. 2020, 10, 32. https://doi.org/10.3390/brainsci10010032
Jang J-H, Lee SH, Jung K, Yoo H, Park G. Inhibitory Effects of Myricetin on Lipopolysaccharide-Induced Neuroinflammation. Brain Sciences. 2020; 10(1):32. https://doi.org/10.3390/brainsci10010032
Chicago/Turabian StyleJang, Jung-Hee, Seung Hoon Lee, Kyungsook Jung, Horyong Yoo, and Gunhyuk Park. 2020. "Inhibitory Effects of Myricetin on Lipopolysaccharide-Induced Neuroinflammation" Brain Sciences 10, no. 1: 32. https://doi.org/10.3390/brainsci10010032
APA StyleJang, J.-H., Lee, S. H., Jung, K., Yoo, H., & Park, G. (2020). Inhibitory Effects of Myricetin on Lipopolysaccharide-Induced Neuroinflammation. Brain Sciences, 10(1), 32. https://doi.org/10.3390/brainsci10010032





