Generation of High Quality Biogenic Silica by Combustion of Rice Husk and Rice Straw Combined with Pre- and Post-Treatment Strategies—A Review
Abstract
:1. Introduction
2. Fuel Properties of Rice Husk and Rice Straw
3. Production of Biogenic Silica
3.1. Combustion of Rice Husk and Rice Straw
3.2. Combination of Fuel Pre-Treatment with Combustion
3.3. Combination of Fuel Pre-Treatment, Ash Post-Treatment and Combustion of Rice Husk and Rice Straw
4. Summary and Conclusions
- (1)
- By studying the ash melting tendency of original and treated RH and RS, it was shown that there is a gap in knowledge, and further investigation is required in this field.
- (2)
- Fuel pre-treatment has a significant impact on controlling ash melting issue in RH and RS, and it seems that at least water washing prior to combustion is essential in RS to avoid slag formation.
- (3)
- Increasing combustion temperature (CT) and residence time (RT) improves the purity of the biogenic silica with respect to the carbon content. At the same time, specific surface area (SSA) and pore volume of the biogenic silica decrease while the risk of crystallization is increased. The alternative strategy to improve all quality characteristics of rice husk ash (RHA) and rice straw ash (RSA) is using fuel pre-treatment prior to the combustion. Crystallization tendency of RHA possibly correlates with the alkali metal content, which facilitates formation of cristobalite. Crystallization temperature is around 600–900 °C in untreated RH and RS depending on the elemental composition, whereas pre-treated RH and RS remain amorphous up to higher temperatures of about 1000 °C.
- (4)
- In RHA from untreated RH, the maximum silica purity, SSA and pore volume are around 97.2 wt %, 220 m2/g and 0.26 cm3/g, respectively. However, these values can be improved to around 99.8 wt %, 353 m2/g and 0.52 cm3/g using fuel pre-treatment strategies prior to the combustion. Further improvement of silica purity is possible by a combination of fuel pre-treatment, combustion and ash post-treatment strategies.
- (5)
- Although high quality biogenic silica can be produced from RH and RS, most studies were performed in lab-scale muffle furnaces and investigations at a bench scale are scarce.
- (6)
- Economically feasible production of biogenic silica from RH was demonstrated at a bench scale though with silica purity limited to 98 wt %. For higher silica purities, a harsh pre- and post-treatment environment may be required, which could increase the technical efforts and thus the overall process costs.
- (7)
- Finally, it was shown that a preliminary practical study has been done to scale the production procedure up to a bench scale with the aim of developing an environmentally friendly and economically feasible process with coupled energetic and material utilization of RH. However, further investigations are required to understand the behavior of RH and RS in combustion. Moreover, there is no report in literature to predict the quality of biogenic silica as well as the ash melting tendency and gaseous emissions using thermodynamic calculations. By using thermodynamic calculations, the effect of chemical composition of input material can also be evaluated in order to transfer to other biomasses since there are limited experimental data available in literature. No information is available in literature on computational fluid dynamic (CFD) simulation of the combustion process of biomass to simulate their conversion mechanism to biogenic silica inside the boiler. If a CFD simulation is used, thermal efficiency of the combustion unit can also be evaluated and optimized. In addition, such a simulation will be valuable to calculate the exact CT and RT of each singular biomass particle during the real operational condition of the combustion unit. In the literature, fundamental lab-scale studies are usually carried out in static airflow. However, the effect of dynamic airflow and air to fuel ratio on the quality of resulting ash have not being studied in the literature. Although many people have measured the biogenic silica purity and pore structure of the ash obtained from RH and RS, there is no information about the accuracy and repeatability of the measurements in the literature, and it seems this gap should be considered in future investigations. Sustainability, economic, and energetic analysis are essential once a commercial plant becomes available to produce biogenic silica from RH and RH in a process including the combination of pre- and post-treatment strategies with combustion.
Author Contributions
Acknowledgments
Conflicts of Interest
Abbreviations
| DT | initial deformation ash fusion temperature |
| db | dry basis |
| LOI | loss of ignition |
| LRH | acid-leached rice husk |
| LRHA | acid-leached rice husk ash |
| LRS | acid-leached rice straw |
| LRSA | acid-leached rice straw ash |
| PRH | pyrolized rice husk |
| RH | as received rice husk |
| RHA | rice husk ash |
| RS | as received rice straw |
| RSA | rice straw ash |
| WRH | water-washed rice husk |
| WRHA | water-washed rice husk ash |
| WRS | water-washed rice straw |
| WRSA | water-washed rice straw ash |
| λ | excess air ratio |
References
- International Energy Agency. World Energy Outlook 2018; ISBN 978-92-64-06452-2. Available online: https://webstore.iea.org/world-energy-outlook-2018 (accessed on 4 March 2019).
- McKendry, P. Energy Production from Biomass (Part 1): Overview of Biomass. Bioresour. Technol. 2002, 83, 37–46. [Google Scholar] [CrossRef]
- Food and Agriculture Organization of the United Nations. Countries by Commodity (Rice, Paddy) [Statistical Database], 2008. Available online: http://faostat.fao.org/site/339/default.aspx (accessed on 4 March 2019).
- Food and Agriculture Organization of the United Nations (FAO). Rice Market Monitor: April 2018. Available online: http://www.fao.org/3/I9243EN/i9243en.pdf (accessed on 13 November 2018).
- Food and Agriculture Organization of the United Nations (FAO). Rice Market Monitor: December 2017. Available online: http://www.fao.org/3/I8317EN/I8317EN.pdf (accessed on 13 November 2018).
- Food and Agriculture Organization of the United Nations (FAO). Rice Market Monitor: December 2006. Available online: http://www.fao.org/tempref/docrep/fao/009/ah614e/ah614e00.pdf (accessed on 13 November 2018).
- Food and Agriculture Organization of the United Nations (FAO). Rice Market Monitor: December 2007. Available online: http://www.fao.org/tempref/docrep/fao/010/ai458e/ai458e00.pdf (accessed on 13 November 2018).
- Food and Agriculture Organization of the United Nations (FAO). Rice market Monitor: July 2008. Available online: http://www.fao.org/tempref/docrep/fao/011/ak001e/ak001e00.pdf (accessed on 13 November 2018).
- Food and Agriculture Organization of the United Nations (FAO). Rice Market Monitor: December 2009. Available online: http://www.fao.org/docrep/012/al004e/al004e00.pdf (accessed on 13 November 2018).
- Food and Agriculture Organization of the United Nations (FAO). Rice Market Monitor: November 2010. Available online: http://www.fao.org/docrep/013/am016e/am016e00.pdf (accessed on 13 November 2018).
- Food and Agriculture Organization of the United Nations (FAO). Rice Market Monitor: November 2011. Available online: http://www.fao.org/docrep/014/am945e/am945e00.pdf (accessed on 13 November 2018).
- Food and Agriculture Organization of the United Nations (FAO). Rice Market Monitor: November 2012. Available online: http://www.fao.org/docrep/017/ap772e/ap772e.pdf (accessed on 13 November 2018).
- Food and Agriculture Organization of the United Nations (FAO). Rice Market Monitor: November 2013. Available online: http://www.fao.org/docrep/019/as201e/as201e.pdf (accessed on 13 November 2018).
- Food and Agriculture Organization of the United Nations (FAO). Rice Market Monitor: December 2014. Available online: http://www.fao.org/3/a-i4294e.pdf (accessed on 13 November 2018).
- Food and Agriculture Organization of the United Nations (FAO). Rice Market Monitor: December 2015. Available online: http://www.fao.org/fileadmin/templates/est/COMM_MARKETS_MONITORING/Rice/Images/RMM/RMM_DEC15_H.pdf (accessed on 13 November 2018).
- Food and Agriculture Organization of the United Nations (FAO). Rice Market Monitor: December 2016. Available online: http://www.fao.org/fileadmin/templates/est/COMM_MARKETS_MONITORING/Rice/Images/RMM/RMM-Dec16_H.pdf (accessed on 13 November 2018).
- Chen, H.; Wang, W.; Martin, J.C.; Oliphant, A.J.; Doerr, P.A.; Xu, J.F.; DeBorn, K.M.; Chen, C.; Sun, L. Extraction of Lignocellulose and Synthesis of Porous Silica Nanoparticles from Rice Husks: A Comprehensive Utilization of Rice Husk Biomass. ACS Sustain. Chem. Eng. 2013, 1, 254–259. [Google Scholar] [CrossRef]
- Kadam, K.L.; Forrest, L.H.; Jacobson, W.A. Rice straw as a lignocellulosic resource: Collection, processing, transportation, and environmental aspects. Biomass Bioenergy 2000, 18, 369–389. [Google Scholar] [CrossRef]
- Blissett, R.; Sommerville, R.; Rowson, N.; Jones, J.; Laughlin, B. Valorisation of rice husks using a TORBED® combustion process. Fuel Process. Technol. 2017, 159, 247–255. [Google Scholar] [CrossRef]
- Chen, X.-G.; Lv, S.-S.; Zhang, P.-P.; Zhang, L.; Ye, Y. Thermal destruction of rice hull in air and nitrogen. J. Therm. Anal. Calorim. 2011, 104, 1055–1062. [Google Scholar] [CrossRef]
- Food and Agriculture Organization of the United Nations (FAO). Sustainable Rice Production for Food Security 2002 [Statistical Database]. Available online: http://www.fao.org/docrep/006/Y4751E/y4751e00.htm (accessed on 13 November 2018).
- Jain, A.; Rajeswara Rao, T.; Sambi, S.S.; Grover, P.D. Energy and chemicals from rice husk. Biomass Bioenergy 1994, 7, 285–289. [Google Scholar] [CrossRef]
- Fang, M.; Yang, L.; Chen, G.; Shi, Z.; Luo, Z.; Cen, K. Experimental study on rice husk combustion in a circulating fluidized bed. Fuel Process. Technol. 2004, 85, 1273–1282. [Google Scholar] [CrossRef]
- Lim, J.S.; Abdul Manan, Z.; Wan Alwi, S.R.; Hashim, H. A review on utilisation of biomass from rice industry as a source of renewable energy. Renew. Sustain. Energy Rev. 2012, 16, 3084–3094. [Google Scholar] [CrossRef]
- Van, V.-T.-A.; Rößler, C.; Bui, D.-D.; Ludwig, H.-M. Pozzolanic reactivity of mesoporous amorphous rice husk ash in portlandite solution. Constr. Build. Mater. 2014, 59, 111–119. [Google Scholar] [CrossRef]
- Zain, M.F.M.; Islam, M.N.; Mahmud, F.; Jamil, M. Production of rice husk ash for use in concrete as a supplementary cementitious material. Constr. Build. Mater. 2011, 25, 798–805. [Google Scholar] [CrossRef]
- Rêgo, J.H.S.; Nepomuceno, A.A.; Figueiredo, E.P.; Hasparyk, N.P. Microstructure of cement pastes with residual rice husk ash of low amorphous silica content. Constr. Build. Mater. 2015, 80, 56–68. [Google Scholar]
- Foo, K.Y.; Hameed, B.H. Utilization of rice husk ash as novel adsorbent: A judicious recycling of the colloidal agricultural waste. Adv. Colloid Interface Sci. 2009, 152, 39–47. [Google Scholar] [CrossRef] [PubMed]
- Srivastava, V.C.; Mall, I.D.; Mishra, I.M. Competitive adsorption of cadmium(II) and nickel(II) metal ions from aqueous solution onto rice husk ash. Chem. Eng. Process. Process Intensif. 2009, 48, 370–379. [Google Scholar] [CrossRef]
- Arabahmadi, V.; Ghorbani, M. Pb (II) removal from water using surface-modified polythiophene-coated rice husk ash nanocomposite. Inorg. Nano-Met. Chem. 2017, 47, 1614–1624. [Google Scholar] [CrossRef]
- Ghorbani, M.; Eisazadeh, H. Fixed bed column study for Zn, Cu, Fe and Mn removal from wastewater using nanometer size polypyrrole coated on rice husk ash. Synth. Met. 2012, 162, 1429–1433. [Google Scholar] [CrossRef]
- Ghorbani, M.; Eisazadeh, H. Removal of COD, color, anions and heavy metals from cotton textile wastewater by using polyaniline and polypyrrole nanocomposites coated on rice husk ash. Compos. Part B Eng. 2013, 45, 1–7. [Google Scholar] [CrossRef]
- Ghorbani, M.; Lashkenari, M.S.; Eisazadeh, H. Application of polyaniline nanocomposite coated on rice husk ash for removal of Hg(II) from aqueous media. Synth. Met. 2011, 161, 1430–1433. [Google Scholar] [CrossRef]
- Liu, D.; Seeburg, D.; Kreft, S.; Bindig, R.; Hartmann, I.; Schneider, D.; Enke, D.; Wohlrab, S. Rice Husk Derived Porous Silica as Support for Pd and CeO2 for Low Temperature Catalytic Methane Combustion. Catalysts 2019, 9, 26. [Google Scholar] [CrossRef]
- Feng, Q.; Lin, Q.; Gong, F.; Sugita, S.; Shoya, M. Adsorption of lead and mercury by rice husk ash. J. Colloid Interface Sci. 2004, 278, 1–8. [Google Scholar] [CrossRef]
- Naiya, T.K.; Bhattacharya, A.K.; Mandal, S.; Das, S.K. The sorption of lead(II) ions on rice husk ash. J. Hazard. Mater. 2009, 163, 1254–1264. [Google Scholar] [CrossRef] [PubMed]
- Tiwari, D.P.; Singh, D.K.; Saksena, D.N. Hg(II) Adsorption from Aqueous Solutions Using Rice-Husk Ash. J. Environ. Eng. 1995, 121, 479–481. [Google Scholar] [CrossRef]
- Srivastava, V.C.; Mall, I.D.; Mishra, I.M. Removal of cadmium(II) and zinc(II) metal ions from binary aqueous solution by rice husk ash. Colloids Surf. A Physicochem. Eng. Asp. 2008, 312, 172–184. [Google Scholar] [CrossRef]
- Chareonpanich, M.; Namto, T.; Kongkachuichay, P.; Limtrakul, J. Synthesis of ZSM-5 zeolite from lignite fly ash and rice husk ash. Fuel Process. Technol. 2004, 85, 1623–1634. [Google Scholar] [CrossRef]
- Vempati, R.K.; Borade, R.; Hegde, R.S.; Komarneni, S. Template free ZSM-5 from siliceous rice hull ash with varying C contents. Microporous Mesoporous Mater. 2006, 93, 134–140. [Google Scholar] [CrossRef]
- Jang, H.T.; Park, Y.; Ko, Y.S.; Lee, J.Y.; Margandan, B. Highly siliceous MCM-48 from rice husk ash for CO2 adsorption. Int. J. Greenh. Gas Control 2009, 3, 545–549. [Google Scholar] [CrossRef]
- Ahmad-Alyosef, H.; Gert Kloess, T.M.; Einicke, W.-D.; Gläser, R.; Enke, D. Biogenic Silica from Rice Husk Ash—Sustainable Sources for the Synthesis of Value Added Silica; A Publication of AIDIC The Italian Association of Chemical Engineering; The Italian Association of Chemical Engineering: Milan, Italy, 2014. [Google Scholar]
- Alyosef, H.A.; Roggendorf, H.; Schneider, D.; Inayat, A.; Welscher, J.; Schwieger, W.; Münster, T.; Kloess, G.; Ibrahim, S.; Enke, D. Comparative Study between Direct and Pseudomorphic Transformation of Rice Husk Ash into MFI-Type Zeolite. Molecules 2017, 23. [Google Scholar] [CrossRef]
- Ng, E.-P.; Awala, H.; Tan, K.-H.; Adam, F.; Retoux, R.; Mintova, S. EMT-type zeolite nanocrystals synthesized from rice husk. Microporous Mesoporous Mater. 2015, 204, 204–209. [Google Scholar] [CrossRef]
- Mohamed, R.M.; Mkhalid, I.A.; Barakat, M.A. Rice husk ash as a renewable source for the production of zeolite NaY and its characterization. Arab. J. Chem. 2015, 8, 48–53. [Google Scholar] [CrossRef]
- Jullaphan, O.; Witoon, T.; Chareonpanich, M. Synthesis of mixed-phase uniformly infiltrated SBA-3-like in SBA-15 bimodal mesoporous silica from rice husk ash. Mater. Lett. 2009, 63, 1303–1306. [Google Scholar] [CrossRef]
- He, Q.; Shi, J. Mesoporous silica nanoparticle based nano drug delivery systems: Synthesis, controlled drug release and delivery, pharmacokinetics and biocompatibility. J. Mater. Chem. 2011, 21, 5845. [Google Scholar] [CrossRef]
- Kwon, S.; Singh, R.K.; Perez, R.A.; Abou Neel, E.A.; Kim, H.-W.; Chrzanowski, W. Silica-based mesoporous nanoparticles for controlled drug delivery. J. Tissue Eng. 2013, 4. [Google Scholar] [CrossRef]
- Pode, R. Potential applications of rice husk ash waste from rice husk biomass power plant. Renew. Sustain. Energy Rev. 2016, 53, 1468–1485. [Google Scholar] [CrossRef]
- Soltani, N.; Bahrami, A.; Pech-Canul, M.I.; González, L.A. Review on the physicochemical treatments of rice husk for production of advanced materials. Chem. Eng. J. 2015, 264, 899–935. [Google Scholar] [CrossRef]
- Shen, Y. Rice husk silica derived nanomaterials for sustainable applications. Renew. Sustain. Energy Rev. 2017, 80, 453–466. [Google Scholar] [CrossRef]
- Yao, X.; Xu, K.; Liang, Y. Comparing the Thermo-Physical Properties of Rice Husk and Rice Straw as Feedstock for Thermochemical Conversion and Characterization of their Waste Ashes from Combustion. BioResources 2016, 11. [Google Scholar] [CrossRef]
- Chandrasekhar, S.; Pramada, P.N.; Praveen, L. Effect of organic acid treatment on the properties of rice husk silica. J. Mater. Sci. 2005, 40, 6535–6544. [Google Scholar] [CrossRef]
- Chandrasekhar, S.; Pramada, P.N.; Majeed, J. Effect of calcination temperature and heating rate on the optical properties and reactivity of rice husk ash. J. Mater. Sci. 2006, 41, 7926–7933. [Google Scholar] [CrossRef]
- Shen, Y.; Zhao, P.; Shao, Q. Porous silica and carbon derived materials from rice husk pyrolysis char. Microporous Mesoporous Mater. 2014, 188, 46–76. [Google Scholar] [CrossRef]
- Tchakouté, H.K.; Rüscher, C.H.; Kong, S.; Ranjbar, N. Synthesis of sodium waterglass from white rice husk ash as an activator to produce metakaolin-based geopolymer cements. J. Build. Eng. 2016, 6, 252–261. [Google Scholar] [CrossRef]
- Pfeiffer, T.; Enke, D.; Roth, R.; Roggendorf, H. Hydrothermal Dissolution of Opal in Sodium Hydroxide Lyes for the Synthesis of Water Glass. ACES 2017, 07, 76–90. [Google Scholar] [CrossRef]
- Wang, W.; Martin, J.C.; Zhang, N.; Ma, C.; Han, A.; Sun, L. Harvesting silica nanoparticles from rice husks. J. Nanopart. Res. 2011, 13, 6981–6990. [Google Scholar] [CrossRef]
- Yang, H.; Liu, B.; Chen, Y.; Li, B.; Chen, H. Influence of Inherent Silicon and Metals in Rice Husk on the Char Properties and Associated Silica Structure. Energy Fuels 2015, 29, 7327–7334. [Google Scholar] [CrossRef]
- Rice, F.; Park, R.; Stayner, L.; Smith, R.; Gilbert, S.; Checkoway, H. Crystalline silica exposure and lung cancer mortality in diatomaceous earth industry workers: A quantitative risk assessment. Occupat. environ. medicine 2001, 58, 38–45. [Google Scholar] [CrossRef]
- DHHS, Department of Health and Human Services, Centers for Disease Control and Prevention. Health Effects of Occupational Exposure to Respirable Crystalline Silica, DHHS (NIOSH) Publication No. 2002-129, 2002.
- Bhattacharya, S.C.; Shah, N.; Alikhani, Z. Some aspects of fluidized bed combustion of paddy husk. Appl. Energy 1984, 16, 307–316. [Google Scholar] [CrossRef]
- Bhattacharya, S.C.; Wu, W. Fluidized Bed Combustion of Rice Husk for Disposal and Energy Recovery. Energy Biomass Wastes XII 1989, 12, 591–601. [Google Scholar]
- Della, V.P.; Kühn, I.; Hotza, D. Rice husk ash as an alternate source for active silica production. Mater. Lett. 2002, 57, 818–821. [Google Scholar] [CrossRef]
- Nehdi, M.; Duquette, J.; El Damatty, A. Performance of rice husk ash produced using a new technology as a mineral admixture in concrete. Cem. Concr. Res. 2003, 33, 1203–1210. [Google Scholar] [CrossRef]
- Rozainee, M.; Ngo, S.P.; Salema, A.A.; Tan, K.G. Fluidized bed combustion of rice husk to produce amorphous siliceous ash. Energy Sustain. Dev. 2008, 12, 33–42. [Google Scholar] [CrossRef]
- Umeda, J.; Kondoh, K. High-purity amorphous silica originated in rice husks via carboxylic acid leaching process. J. Mater. Sci 2008, 43, 7084–7090. [Google Scholar] [CrossRef]
- Martínez, J.D.; Pineda, T.; López, J.P.; Betancur, M. Assessment of the rice husk lean-combustion in a bubbling fluidized bed for the production of amorphous silica-rich ash. Energy 2011, 36, 3846–3854. [Google Scholar] [CrossRef]
- Bie, R.-S.; Song, X.-F.; Liu, Q.-Q.; Ji, X.-Y.; Chen, P. Studies on effects of burning conditions and rice husk ash (RHA) blending amount on the mechanical behavior of cement. Cem. Concr. Compos. 2015, 55, 162–168. [Google Scholar] [CrossRef]
- Faé Gomes, G.M.; Philipssen, C.; Bard, E.K.; Zen, L.D.; Souza, G.d. Rice husk bubbling fluidized bed combustion for amorphous silica synthesis. J. Environ. Chem. Eng. 2016, 4, 2278–2290. [Google Scholar] [CrossRef]
- Chen, P.; Gu, W.; Fang, W.; Ji, X.; Bie, R. Removal of metal impurities in rice husk and characterization of rice husk ash under simplified acid pretreatment process. Environ. Prog. Sustain. Energy 2017, 36, 830–837. [Google Scholar] [CrossRef]
- Schneider, D.; Wassersleben, S.; Weiß, M.; Denecke, R.; Stark, A.; Enke, D. A Generalized Procedure for the Production of High-Grade, Porous Biogenic Silica. Waste Biomass Valor 2018, 37, 667. [Google Scholar] [CrossRef]
- Schliermann, T.; Hartmann, I.; Beidaghy Dizaji, H.; Zeng, T.; Schneider, D.; Wassersleben, S.; Enke, D.; Jobst, T.; Lange, A.; Roelofs, F.; et al. High quality biogenic silica from combined energetic and material utilization of agricultural residues. In Proceedings of the 7th International Symposium of Energy from Biomass and Waste, Venice, Italy, 15–18 October 2018; ISBN 978-8-86-265013-7. [Google Scholar]
- Tutsek, A.; Bartha, P. Method of Producing Low-Carbon White Husk Ash. US Patent No. US4049464, 20 September 1977. [Google Scholar]
- Chandrasekhar, S.; Satyanarayana, K.G.; Pramada, P.N.; Raghavan, P.; Gupta, T.N. Review Processing, properties and applications of reactive silica from rice husk—An overview. J. Mater. Sci. 2003, 38, 3159–3168. [Google Scholar] [CrossRef]
- Chuah, T.G.; Jumasiah, A.; Azni, I.; Katayon, S.; Thomas Choong, S.Y. Rice husk as a potentially low-cost biosorbent for heavy metal and dye removal: An overview. Desalination 2005, 175, 305–316. [Google Scholar] [CrossRef]
- Adam, F.; Appaturi, J.N.; Iqbal, A. The utilization of rice husk silica as a catalyst: Review and recent progress. Catal. Today 2012, 190, 2–14. [Google Scholar] [CrossRef]
- Malhotra, V.M. Fly Ash, Slag, Silica Fume, and Rice Husk Ash in Concrete: A Review. Concr. Int. 1993, 15, 23–28. [Google Scholar]
- Quispe, I.; Navia, R.; Kahhat, R. Energy potential from rice husk through direct combustion and fast pyrolysis: A review. Waste Manag. 2017, 59, 200–210. [Google Scholar] [CrossRef]
- Karim, M.R.; Zain, M.F.M.; Jamil, M. Strength of mortar and concrete as influenced by rice husk ash: A review. World Appl. Sci. J. 2012, 19, 1501–1513. [Google Scholar]
- Chen, Y.; Zhu, Y.; Wang, Z.; Li, Y.; Wang, L.; Ding, L.; Gao, X.; Ma, Y.; Guo, Y. Application studies of activated carbon derived from rice husks produced by chemical-thermal process—A review. Adv. Colloid Interface Sci. 2011, 163, 39–52. [Google Scholar] [CrossRef] [PubMed]
- Natarajan, E.; Nordin, A.; Rao, A.N. Overview of combustion and gasification of rice husk in fluidized bed reactors. Biomass Bioenergy 1998, 14, 533–546. [Google Scholar] [CrossRef]
- Muthadhi, A.; Anitha, R.; Kothandaraman, S. Rice husk ash—Properties and its uses: A review. J. Inst. Eng. Civ. Eng. Divis. 2007, 88, 50–56. [Google Scholar]
- Prasad, R.; Pandey, M. Rice Husk Ash as a Renewable Source for the Production of Value Added Silica Gel and its Application: An Overview. Bull. Chem. React. Eng. Catal. 2012, 7. [Google Scholar] [CrossRef]
- Prasara-A, J.; Gheewala, S.H. Sustainable utilization of rice husk ash from power plants: A review. J. Clean. Prod. 2017, 167, 1020–1028. [Google Scholar] [CrossRef]
- Sandhu, R.K.; Siddique, R. Influence of rice husk ash (RHA) on the properties of self-compacting concrete: A review. Constr. Build. Mater. 2017, 153, 751–764. [Google Scholar] [CrossRef]
- Khan, M.N.N.; Jamil, M.; Karim, M.R.; Zain, M.F.M.; Kaish, A.B.M.A. Utilization of Rice Husk Ash for Sustainable Construction: A Review. RJASET 2015, 9, 1119–1127. [Google Scholar] [CrossRef]
- Ahmaruzzaman, M.; Gupta, V.K. Rice Husk and Its Ash as Low-Cost Adsorbents in Water and Wastewater Treatment. Ind. Eng. Chem. Res. 2011, 50, 13589–13613. [Google Scholar] [CrossRef]
- Sun, L.; Gong, K. Silicon-Based Materials from Rice Husks and Their Applications. Ind. Eng. Chem. Res. 2001, 40, 5861–5877. [Google Scholar] [CrossRef]
- Shen, Y. Rice Husk Silica-Derived Nanomaterials for Battery Applications: A Literature Review. J. Agric. Food Chem. 2017, 65, 995–1004. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.H.; Kwon, J.H.; Lee, J.-W.; Lee, H.-S.; Chang, J.H.; Sang, B.-I. Preparation of high purity silica originated from rice husks by chemically removing metallic impurities. J. Ind. Eng. Chem. 2017, 50, 79–85. [Google Scholar] [CrossRef]
- Pollex, A.; Zeng, T.; Khalsa, J.; Erler, U.; Schmersahl, R.; Schön, C.; Kuptz, D.; Lenz, V.; Nelles, M. Content of potassium and other aerosol forming elements in commercially available wood pellet batches. Fuel 2018, 232, 384–394. [Google Scholar] [CrossRef]
- Vassilev, S.V.; Baxter, D.; Andersen, L.K.; Vassileva, C.G. An overview of the composition and application of biomass ash. Part 1. Phase–mineral and chemical composition and classification. Fuel 2013, 105, 40–76. [Google Scholar] [CrossRef]
- Zevenhoven-Onderwater, M.; Blomquist, J.-P.; Skrifvars, B.-J.; Backman, R.; Hupa, M. The prediction of behaviour of ashes from five different solid fuels in fluidised bed combustion. Fuel 2000, 79, 1353–1361. [Google Scholar] [CrossRef]
- Lindström, E.; Öhman, M.; Backman, R.; Boström, D. Influence of Sand Contamination on Slag Formation during Combustion of Wood Derived Fuels. Energy Fuels 2008, 22, 2216–2220. [Google Scholar] [CrossRef]
- Lindström, E.; Larsson, S.H.; Boström, D.; Öhman, M. Slagging Characteristics during Combustion of Woody Biomass Pellets Made from a Range of Different Forestry Assortments. Energy Fuels 2010, 24, 3456–3461. [Google Scholar] [CrossRef]
- Öhman, M.; Boman, C.; Hedman, H.; Nordin, A.; Boström, D. Slagging tendencies of wood pellet ash during combustion in residential pellet burners. Biomass Bioenergy 2004, 27, 585–596. [Google Scholar] [CrossRef]
- Alyosef, H.A.; Eilert, A.; Welscher, J.; Ibrahim, S.S.; Denecke, R.; Schwieger, W.; Enke, D. Characterization of Biogenic Silica Generated by Thermo Chemical Treatment of Rice Husk. Part. Sci. Technol. 2013, 31, 524–532. [Google Scholar] [CrossRef]
- Santana Costa, J.A.; Paranhos, C.M. Systematic evaluation of amorphous silica production from rice husk ashes. J. Clean. Prod. 2018, 192, 688–697. [Google Scholar] [CrossRef]
- Alyosef, H.A. Rice Husk ash (RHA) as a Renewable Source for Value Added Silica: Products the Way to Standardized Educts, Comprehensive Characterization, Modification and Phase Transformation. Ph.D. Thesis, Universität Leipzig, Leipzig, Germany, 2014. [Google Scholar]
- Zareihassangheshlaghi, A.; Beidaghy Dizaji, H.; Zeng, T.; Hartmann, I.; Lenz, V.; Enke, D.; Bidabadi, M. Impact of Varying Heating Regimes on Biogenic Silica Obtained from Rice Husk. In Proceedings of the Deutsches Doktorandenkolloquium Bioenergie, Leipzig, Germany, 20–21 September 2018. [Google Scholar]
- Gu, S.; Zhou, J.; Luo, Z.; Wang, Q.; Ni, M. A detailed study of the effects of pyrolysis temperature and feedstock particle size on the preparation of nanosilica from rice husk. Ind. Crops Prod. 2013, 50, 540–549. [Google Scholar] [CrossRef]
- Rivas, A.L.; Vera, G.; Palacios, V.; Cornejo, M.; Rigail, A.; Solórzano, G. Phase Transformation of Amorphous Rice Husk Silica. In Frontiers in Materials Processing, Applications, Research and Technology; Muruganant, M., Chirazi, A., Raj, B., Eds.; Springer: Singapore, 2018; ISBN 978-981-10-4818-0. [Google Scholar]
- Mochidzuki, K.; Sakoda, A.; Suzuki, M.; Izumi, J.; Tomonaga, N. Structural Behavior of Rice Husk Silica in Pressurized Hot-Water Treatment Processes. Ind. Eng. Chem. Res. 2001, 40, 5705–5709. [Google Scholar] [CrossRef]
- Bakker, R.R.; Jenkins, B.M.; Williams, R.B. Fluidized Bed Combustion of Leached Rice Straw. Energy Fuels 2002, 16, 356–365. [Google Scholar] [CrossRef]
- Knudsen, J.N.; Jensen, P.A.; Dam-Johansen, K. Transformation and Release to the Gas Phase of Cl, K, and S during Combustion of Annual Biomass. Energy Fuels 2004, 18, 1385–1399. [Google Scholar] [CrossRef]
- Allendorf, M.D.; Spear, K.E. Thermodynamic Analysis of Silica Refractory Corrosion in Glass-Melting Furnaces. J. Electrochem. Soc. 2001, 148, B59. [Google Scholar] [CrossRef]
- Besmann, T.M.; Spear, K.E. Thermochemical Modeling of Oxide Glasses. J. Am. Ceram. Soc. 2002, 85, 2887–2894. [Google Scholar] [CrossRef]
- Jenkins, B.M.; Bakker, R.R.; Wei, J.B. On the properties of washed straw. Biomass Bioenergy 1996, 10, 177–200. [Google Scholar] [CrossRef]
- Vassilev, S.V.; Baxter, D.; Andersen, L.K.; Vassileva, C.G.; Morgan, T.J. An overview of the organic and inorganic phase composition of biomass. Fuel 2012, 94, 1–33. [Google Scholar] [CrossRef]
- Haykiri-Acma, H.; Yaman, S.; Kucukbayrak, S. Effect of biomass on temperatures of sintering and initial deformation of lignite ash. Fuel 2010, 89, 3063–3068. [Google Scholar] [CrossRef]
- Jenkins, B.M.; Baxter, L.L.; Miles, T.R. Combustion properties of biomass. Fuel Process. Technol. 1998, 54, 17–46. [Google Scholar] [CrossRef]
- Sun, S.; Zhao, Y.; Su, F.; Ling, F. Gasification of rice husk in a cyclone gasifier. Korean J. Chem. Eng. 2009, 26, 528–533. [Google Scholar] [CrossRef]
- Madhiyanon, T.; Sathitruangsak, P.; Soponronnarit, S. Co-combustion of rice husk with coal in a cyclonic fluidized-bed combustor (ψ-FBC). Fuel 2009, 88, 132–138. [Google Scholar] [CrossRef]
- Williams, P.T.; Nugranad, N. Comparison of products from the pyrolysis and catalytic pyrolysis of rice husks. Energy 2000, 25, 493–513. [Google Scholar] [CrossRef]
- Rahman, I.A.; Ismail, J.; Osman, H. Effect of nitric acid digestion on organic materials and silica in rice husk. J. Mater. Chem. 1997, 7, 1505–1509. [Google Scholar] [CrossRef]
- Vallios, I.; Tsoutsos, T.; Papadakis, G. Design of biomass district heating systems. Biomass Bioenergy 2009, 33, 659–678. [Google Scholar] [CrossRef]
- Hartiniati, S.A.; Youvial, M. (Eds.) Performance of a pilot scale flu-idized bed gasifier fueled by rice husks, pyrolysis and gasifi-cation. International Conference on Pyrolysis and Gasification, Luxembourg City, Luxembourg, 15–18 October 2018. [Google Scholar]
- Perera, K.K.C.K.; Rathnasiri, P.G.; Senarath, S.A.S.; Sugathapala, A.G.T.; Bhattacharya, S.C.; Abdul Salam, P. Assessment of sustainable energy potential of non-plantation biomass resources in Sri Lanka. Biomass Bioenergy 2005, 29, 199–213. [Google Scholar] [CrossRef]
- Gaur, S.; Reed, T.B. An Atlas of Thermal Data for Biomass and Other Fuels; National Renewable Energy Lab.: Golden, CO, USA, 1995. [CrossRef]
- Lv, D.; Xu, M.; Liu, X.; Zhan, Z.; Li, Z.; Yao, H. Effect of cellulose, lignin, alkali and alkaline earth metallic species on biomass pyrolysis and gasification. Fuel Process. Technol. 2010, 91, 903–909. [Google Scholar] [CrossRef]
- Singh, R.I.; Mohapatra, S.K.; Gangacharyulu, D. Studies in an atmospheric bubbling fluidized-bed combustor of 10MW power plant based on rice husk. Energy Convers. Manag. 2008, 49, 3086–3103. [Google Scholar] [CrossRef]
- Mansaray, K.G.; Ghaly, A.E. Determination of kinetic parameters of rice husks in oxygen using thermogravimetric analysis. Biomass Bioenergy 1999, 17, 19–31. [Google Scholar] [CrossRef]
- Sathitruangsak, P.; Madhiyanon, T.; Soponronnarit, S. Rice husk co-firing with coal in a short-combustion-chamber fluidized-bed combustor (SFBC). Fuel 2009, 88, 1394–1402. [Google Scholar] [CrossRef]
- Channiwala, S.A.; Parikh, P.P. A unified correlation for estimating HHV of solid, liquid and gaseous fuels. Fuel 2002, 81, 1051–1063. [Google Scholar] [CrossRef]
- Gungor, A. Two-dimensional biomass combustion modeling of CFB. Fuel 2008, 87, 1453–1468. [Google Scholar] [CrossRef]
- Ebeling, J.M.; Jenkins, B.M. Physical and chemical properties of biomass fuels. Trans. Am. Soc. Agric. Eng. 1985, 28. [Google Scholar] [CrossRef]
- Buragohain, B.; Mahanta, P.; Moholkar, V.S. Biomass gasification for decentralized power generation: The Indian perspective. Renew. Sustain. Energy Rev. 2010, 14, 73–92. [Google Scholar] [CrossRef]
- Zhang, Y.; Ashizawa, M.; Kajitani, S.; Hara, S. A new approach to catalytic coal gasification: The recovery and reuse of calcium using biomass derived crude vinegars. Fuel 2010, 89, 417–422. [Google Scholar] [CrossRef]
- Couhert, C.; Commandré, J.-M.; Salvador, S. Failure of the component additivity rule to predict gas yields of biomass in flash pyrolysis at 950 °C. Biomass Bioenergy 2009, 33, 316–326. [Google Scholar] [CrossRef]
- Chen, W.-H.; Wu, J.-S. An evaluation on rice husks and pulverized coal blends using a drop tube furnace and a thermogravimetric analyzer for application to a blast furnace. Energy 2009, 34, 1458–1466. [Google Scholar] [CrossRef]
- Hossain, M.S.; Islam, M.R.; Rahman, M.S.; Kader, M.A.; Haniu, H. Biofuel from Co-pyrolysis of Solid Tire Waste and Rice Husk. Energy Procedia 2017, 110, 453–458. [Google Scholar] [CrossRef]
- Zhang, S.; Dong, Q.; Zhang, L.; Xiong, Y. Effects of water washing and torrefaction on the pyrolysis behavior and kinetics of rice husk through TGA and Py-GC/MS. Bioresour. Technol. 2016, 199, 352–361. [Google Scholar] [CrossRef]
- Kumar, A. An approach to the estimation of the value of agricultural residues used as biofuels. Biomass Bioenergy 2002, 22, 195–203. [Google Scholar] [CrossRef]
- Sun, L.; Gu, Z.Z.; Guo, D.Y.; Xu, M. Demonstration Systems of Cooking Gas Produced by Crop Straw Gasifier for Villages; Bridgwater, A.V., Boocock, D.G.B., Eds.; Developments in Thermochemical Biomass Conversion; Springer: Dordrecht, The Netherlands, 1997; Volume 1. [Google Scholar] [CrossRef]
- Raveendran, K.; Ganesh, A.; Khilar, K.C. Influence of mineral matter on biomass pyrolysis characteristics. Fuel 1995, 74, 1812–1822. [Google Scholar] [CrossRef]
- Chao, C.Y.H.; Kwong, P.C.W.; Wang, J.H.; Cheung, C.W.; Kendall, G. Co-firing coal with rice husk and bamboo and the impact on particulate matters and associated polycyclic aromatic hydrocarbon emissions. Bioresour. Technol. 2008, 99, 83–93. [Google Scholar] [CrossRef] [PubMed]
- Huang, H.-J.; Yuan, X.-Z.; Zeng, G.-M.; Liu, Y.; Li, H.; Yin, J.; Wang, X.-L. Thermochemical liquefaction of rice husk for bio-oil production with sub- and supercritical ethanol as solvent. J. Anal. Appl. Pyrolysis 2013, 102, 60–67. [Google Scholar] [CrossRef]
- Armesto, L.; Bahillo, A.; Veijonen, K.; Cabanillas, A.; Otero, J. Combustion behaviour of rice husk in a bubbling fluidised bed. Biomass Bioenergy 2002, 23, 171–179. [Google Scholar] [CrossRef]
- Permchart, W.; Kouprianov, V.I. Emission performance and combustion efficiency of a conical fluidized-bed combustor firing various biomass fuels. Bioresour. Technol. 2004, 92, 83–91. [Google Scholar] [CrossRef]
- Sun, S.; Tian, H.; Zhao, Y.; Sun, R.; Zhou, H. Experimental and numerical study of biomass flash pyrolysis in an entrained flow reactor. Bioresour. Technol. 2010, 101, 3678–3684. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.-C.; Ku, Y.; Tsai, H.-H.; Kuo, Y.-L.; Tseng, Y.-H. Rice husk as solid fuel for chemical looping combustion in an annular dual-tube moving bed reactor. Chem. Eng. J. 2015, 280, 82–89. [Google Scholar] [CrossRef]
- Kwong, C.W.; Chao, C.Y.H. Fly-ash products from biomass co-combustion for VOC control. Bioresour. Technol. 2010, 101, 1075–1081. [Google Scholar] [CrossRef]
- Couhert, C.; Commandre, J.-M.; Salvador, S. Is it possible to predict gas yields of any biomass after rapid pyrolysis at high temperature from its composition in cellulose, hemicellulose and lignin? Fuel 2009, 88, 408–417. [Google Scholar] [CrossRef]
- Tao, G.; Lestander, T.A.; Geladi, P.; Xiong, S. Biomass properties in association with plant species and assortments I: A synthesis based on literature data of energy properties. Renew. Sustain. Energy Rev. 2012, 16, 3481–3506. [Google Scholar] [CrossRef]
- Wang, X.; Lv, W.; Guo, L.; Zhai, M.; Dong, P.; Qi, G. Energy and exergy analysis of rice husk high-temperature pyrolysis. Int. J. Hydrogen Energy 2016, 41, 21121–21130. [Google Scholar] [CrossRef]
- Janvijitsakul, K.; Kuprianov, V.I. Major gaseous and PAH emissions from a fluidized-bed combustor firing rice husk with high combustion efficiency. Fuel Process. Technol. 2008, 89, 777–787. [Google Scholar] [CrossRef]
- Zhang, S.; Su, Y.; Zhu, S.; Zhang, H.; Zhang, Q. Effects of pretreatment and FeCl3 preload of rice husk on synthesis of magnetic carbon composites by pyrolysis for supercapacitor application. J. Anal. Appl. Pyrolysis 2018, 135, 22–31. [Google Scholar] [CrossRef]
- Zhang, S.; Su, Y.; Ding, K.; Zhu, S.; Zhang, H.; Liu, X.; Xiong, Y. Effect of inorganic species on torrefaction process and product properties of rice husk. Bioresour. Technol. 2018, 265, 450–455. [Google Scholar] [CrossRef]
- Zhang, S.; Dong, Q.; Zhang, L.; Xiong, Y.; Liu, X.; Zhu, S. Effects of water washing and torrefaction pretreatments on rice husk pyrolysis by microwave heating. Bioresour. Technol. 2015, 193, 442–448. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Dong, Q.; Chen, T.; Xiong, Y. Combination of Light Bio-oil Washing and Torrefaction Pretreatment of Rice Husk: Its Effects on Physicochemical Characteristics and Fast Pyrolysis Behavior. Energy Fuels 2016, 30, 3030–3037. [Google Scholar] [CrossRef]
- Zhang, S.; Zhu, S.; Zhang, H.; Chen, T.; Xiong, Y. Catalytic fast pyrolysis of rice husk: Effect of coupling leaching with torrefaction pretreatment. J. Anal. Appl. Pyrolysis 2018, 133, 91–96. [Google Scholar] [CrossRef]
- Zhang, S.; Su, Y.; Xu, D.; Zhu, S.; Zhang, H.; Liu, X. Effects of torrefaction and organic-acid leaching pretreatment on the pyrolysis behavior of rice husk. Energy 2018, 149, 804–813. [Google Scholar] [CrossRef]
- Zhang, S.; Chen, T.; Xiong, Y.; Dong, Q. Effects of wet torrefaction on the physicochemical properties and pyrolysis product properties of rice husk. Energy Convers. Manag. 2017, 141, 403–409. [Google Scholar] [CrossRef]
- Zhang, S.; Su, Y.; Xiong, Y. Influence of coupling demineralization with the torrefaction pretreatment process on the pyrolysis characteristics and kinetics of rice husk. Energy Sour. Part A Recov. Utiliz. Environ. Eff. 2017, 39, 726–732. [Google Scholar] [CrossRef]
- Bakker, R.R.; Jenkins, B.M.; Williams, R.B.; Pfaff, D. Fluidized bed combustion of leached rice straw. Proceedings of the 4th biomass conference of the Americas. Available online: https://phyllis.nl/ (accessed on 13 November 2018).
- Jung, S.-H.; Kang, B.-S.; Kim, J.-S. Production of bio-oil from rice straw and bamboo sawdust under various reaction conditions in a fast pyrolysis plant equipped with a fluidized bed and a char separation system. J. Anal. Appl. Pyrolysis 2008, 82, 240–247. [Google Scholar] [CrossRef]
- LU, Y.; GUO, L.; JI, C.; ZHANG, X.; HAO, X.; YAN, Q. Hydrogen production by biomass gasification in supercritical water: A parametric study. Int. J. Hydrogen Energy 2006, 31, 822–831. [Google Scholar] [CrossRef]
- Liu, H.; Feng, Y.; Wu, S.; Liu, D. The role of ash particles in the bed agglomeration during the fluidized bed combustion of rice straw. Bioresour. Technol. 2009, 100, 6505–6513. [Google Scholar] [CrossRef] [PubMed]
- Miles, T.R.; Baxter, L.L.; Bryers, R.W.; Jenkins, B.M.; Oden, L.L. Boiler deposits from firing biomass fuels. Biomass Bioenergy 1996, 10, 125–138. [Google Scholar] [CrossRef]
- Thy, P.; Jenkins, B.M.; Williams, R.B.; Lesher, C.E.; Bakker, R.R. Bed agglomeration in fluidized combustor fueled by wood and rice straw blends. Fuel Process. Technol. 2010, 91, 1464–1485. [Google Scholar] [CrossRef]
- Grover, P.D.; Iyer, P.V.R.; Rao, T.R. Biomass-Thermochemical Characterization, 3rd ed.; IIT-Delhi and MNES: Delhi, Indian, 2002. [Google Scholar]
- Thy, P.; Jenkins, B.; Grundvig, S.; Shiraki, R.; Lesher, C. High temperature elemental losses and mineralogical changes in common biomass ashes. Fuel 2006, 85, 783–795. [Google Scholar] [CrossRef]
- Yang, T.; Wang, J.; Li, B.; Kai, X.; Li, R. Effect of residence time on two-step liquefaction of rice straw in a CO2 atmosphere: Differences between subcritical water and supercritical ethanol. Bioresour. Technol. 2017, 229, 143–151. [Google Scholar] [CrossRef] [PubMed]
- Thy, P.; Jenkins, B.M.; Lesher, C.E.; Grundvig, S. Compositional constraints on slag formation and potassium volatilization from rice straw blended wood fuel. Fuel Process. Technol. 2006, 87, 383–408. [Google Scholar] [CrossRef]
- Ding, L.; Gong, Y.; Wang, Y.; Wang, F.; Yu, G. Characterisation of the morphological changes and interactions in char, slag and ash during CO2 gasification of rice straw and lignite. Appl. Energy 2017, 195, 713–724. [Google Scholar] [CrossRef]
- Wu, Y.; Wu, S.; Li, Y.; Gao, J. Physico-Chemical Characteristics and Mineral Transformation Behavior of Ashes from Crop Straw. Energy Fuels 2009, 23, 5144–5150. [Google Scholar] [CrossRef]
- Chen, C.; Yu, C.; Zhang, H.; Zhai, X.; Luo, Z. Investigation on K and Cl release and migration in micro-spatial distribution during rice straw pyrolysis. Fuel 2016, 167, 180–187. [Google Scholar] [CrossRef]
- Asadullah, M.; Miyazawa, T.; Ito, S.-I.; Kunimori, K.; Yamada, M.; Tomishige, K. Gasification of different biomasses in a dual-bed gasifier system combined with novel catalysts with high energy efficiency. Appl. Catal. A Gen. 2004, 267, 95–102. [Google Scholar] [CrossRef]
- Jenkins, B.M.; Mannapperuma, J.D.; Bakker, R.R. Biomass leachate treatment by reverse osmosis. Fuel Process. Technol. 2003, 81, 223–246. [Google Scholar] [CrossRef]
- Li, K.; Zhang, R.; Bi, J. Experimental study on syngas production by co-gasification of coal and biomass in a fluidized bed. Int. J. Hydrogen Energy 2010, 35, 2722–2726. [Google Scholar] [CrossRef]
- Chiang, K.-Y.; Liao, C.-K.; Lu, C.-H. The effects of prepared iron-based catalyst on the energy yield in gasification of rice straw. Int. J. Hydrogen Energy 2016, 41, 21747–21754. [Google Scholar] [CrossRef]
- Dayton, D.C.; Jenkins, B.M.; Turn, S.Q.; Bakker, R.R.; Williams, R.B.; Belle-Oudry, D.; Hill, L.M. Release of Inorganic Constituents from Leached Biomass during Thermal Conversion. Energy Fuels 1999, 13, 860–870. [Google Scholar] [CrossRef]
- Chen, H.; Chen, X.; Qin, Y.; Wei, J.; Liu, H. Effect of torrefaction on the properties of rice straw high temperature pyrolysis char: Pore structure, aromaticity and gasification activity. Bioresour. Technol. 2017, 228, 241–249. [Google Scholar] [CrossRef] [PubMed]
- Dong, Q.; Zhang, S.; Ding, K.; Zhu, S.; Zhang, H.; Liu, X. Pyrolysis behavior of raw/torrefied rice straw after different demineralization processes. Biomass Bioenergy 2018, 119, 229–236. [Google Scholar] [CrossRef]
- Miccio, F.; Moersch, O.; Spliethoff, H.; Hein, K.R.G. Generation and conversion of carbonaceous fine particles during bubbling fluidised bed gasification of a biomass fuel. Fuel 1999, 78, 1473–1481. [Google Scholar] [CrossRef]
- Storm, C.; Spliethoff, H.; Hein, K.R.G. Generation of a gaseous fuel by gasification or pyrolysis of biomass for use as reburn gas in coal-fired boilers. In Proceedings of the 5th European Conference on Industrial Furnaces and Boilers, Porto, Portugal, 11–14 April 2000; Volume 1. [Google Scholar]
- WEI, X.; SCHNELL, U.; HEIN, K. Behaviour of gaseous chlorine and alkali metals during biomass thermal utilisation. Fuel 2005, 84, 841–848. [Google Scholar] [CrossRef]
- Fernandes, I.J.; Calheiro, D.; Kieling, A.G.; Moraes, C.A.M.; Rocha, T.L.A.C.; Brehm, F.A.; Modolo, R.C.E. Characterization of rice husk ash produced using different biomass combustion techniques for energy. Fuel 2016, 165, 351–359. [Google Scholar] [CrossRef]
- Souza, M.F.d.; Magalhães, W.L.E.; Persegil, M.C. Silica Derived from Burned Rice Hulls. Mater. Res. 2002, 5, 467–474. [Google Scholar] [CrossRef]
- Yalçin, N.; Sevinç, V. Studies on silica obtained from rice husk. Ceram. Int. 2001, 27, 219–224. [Google Scholar] [CrossRef]
- Vassilev, S.V.; Baxter, D.; Vassileva, C.G. An overview of the behaviour of biomass during combustion: Part I. Phase-mineral transformations of organic and inorganic matter. Fuel 2013, 112, 391–449. [Google Scholar] [CrossRef]
- Vassilev, S.V.; Baxter, D.; Andersen, L.K.; Vassileva, C.G. An overview of the chemical composition of biomass. Fuel 2010, 89, 913–933. [Google Scholar] [CrossRef]
- Vassilev, S.V.; Baxter, D.; Vassileva, C.G. An overview of the behaviour of biomass during combustion: Part II. Ash fusion and ash formation mechanisms of biomass types. Fuel 2014, 117, 152–183. [Google Scholar] [CrossRef]
- Pollex, A.; Zeng, T.; Khalsa, J.H.A.; Erler, U.; Schmersahl, R.; Schön, C.; Kuptz, D.; Lenz, V.; Nelles, M. Data Set of Potassium and Other Aerosol Forming Elements in High Quality Wood Pellets, 2017. [CrossRef]
- Alvarez, J.; Lopez, G.; Amutio, M.; Bilbao, J.; Olazar, M. Bio-oil production from rice husk fast pyrolysis in a conical spouted bed reactor. Fuel 2014, 128, 162–169. [Google Scholar] [CrossRef]
- Umeda, J.; Kondoh, K. High-purification of amorphous silica originated from rice husks by combination of polysaccharide hydrolysis and metallic impurities removal. Ind. Crops Prod. 2010, 32, 539–544. [Google Scholar] [CrossRef]
- Krishnarao, R.V.; Subrahmanyam, J.; Jagadish Kumar, T. Studies on the formation of black particles in rice husk silica ash. J. Eur. Ceram. Soc. 2001, 21, 99–104. [Google Scholar] [CrossRef]
- Shen, J.; Liu, X.; Zhu, S.; Zhang, H.; Tan, J. Effects of calcination parameters on the silica phase of original and leached rice husk ash. Mater. Lett. 2011, 65, 1179–1183. [Google Scholar] [CrossRef]
- Chen, G.; Du, G.; Ma, W.; Yan, B.; Wang, Z.; Gao, W. Production of amorphous rice husk ash in a 500 kW fluidized bed combustor. Fuel 2015, 144, 214–221. [Google Scholar] [CrossRef]
- Liou, T.-H. Preparation and characterization of nano-structured silica from rice husk. Mater. Sci. Eng. A 2004, 364, 313–323. [Google Scholar] [CrossRef]
- Liou, T.-H. Evolution of chemistry and morphology during the carbonization and combustion of rice husk. Carbon 2004, 42, 785–794. [Google Scholar] [CrossRef]
- Lu, P.; Hsieh, Y.-L. Highly pure amorphous silica nano-disks from rice straw. Powder Technol. 2012, 225, 149–155. [Google Scholar] [CrossRef]
- Ahmad Alyosef, H.; Schneider, D.; Wassersleben, S.; Roggendorf, H.; Weiß, M.; Eilert, A.; Denecke, R.; Hartmann, I.; Enke, D. Meso/Macroporous Silica from Miscanthus, Cereal Remnant Pellets, and Wheat Straw. ACS Sustain. Chem. Eng. 2015, 3, 2012–2021. [Google Scholar] [CrossRef]
- Gu, S.; Zhou, J.; Yu, C.; Luo, Z.; Wang, Q.; Shi, Z. A novel two-staged thermal synthesis method of generating nanosilica from rice husk via pre-pyrolysis combined with calcination. Ind. Crops Prod. 2015, 65, 1–6. [Google Scholar] [CrossRef]
- Liaw, S.B.; Wu, H. Leaching Characteristics of Organic and Inorganic Matter from Biomass by Water: Differences between Batch and Semi-continuous Operations. Ind. Eng. Chem. Res. 2013, 52, 4280–4289. [Google Scholar] [CrossRef]
- Werkelin, J.; Skrifvars, B.-J.; Zevenhoven, M.; Holmbom, B.; Hupa, M. Chemical forms of ash-forming elements in woody biomass fuels. Fuel 2010, 89, 481–493. [Google Scholar] [CrossRef]
- Said, N.; Bishara, T.; García-Maraver, A.; Zamorano, M. Effect of water washing on the thermal behavior of rice straw. Waste Manag. 2013, 33, 2250–2256. [Google Scholar] [CrossRef] [PubMed]
- Bakar, R.A.; Yahya, R.; Gan, S.N. Production of High Purity Amorphous Silica from Rice Husk. Procedia Chem. 2016, 19, 189–195. [Google Scholar] [CrossRef]
- Umeda, J.; Kondoh, K.; Michiura, Y. Process Parameters Optimization in Preparing High-Purity Amorphous Silica Originated from Rice Husks. Mater. Trans. 2007, 48, 3095–3100. [Google Scholar] [CrossRef]
- Chakraverty, A.; Mishra, P.; Banerjee, H.D. Investigation of combustion of raw and acid-leached rice husk for production of pure amorphous white silica. J. Mater. Sci. 1988, 23, 21–24. [Google Scholar] [CrossRef]
- Schliermann, T.; Hartmann, I.; Schneider, D.; Wassersleben, S.; Enke, D.; Jobst, T.; Lange, A.; Roelofs, F.; Fellner, A.; Schneider, P. High-quality biogenic silica from agricultural residues. In Tagungsband Waste-to-Resources, Proceedings of the 7th International Symposium MBT, MRF & Recycling, Hannover, Germany, 16–18 May 2017; Cuvillier Verlag: Göttingen, Germany, 2017; pp. 676–687. [Google Scholar]
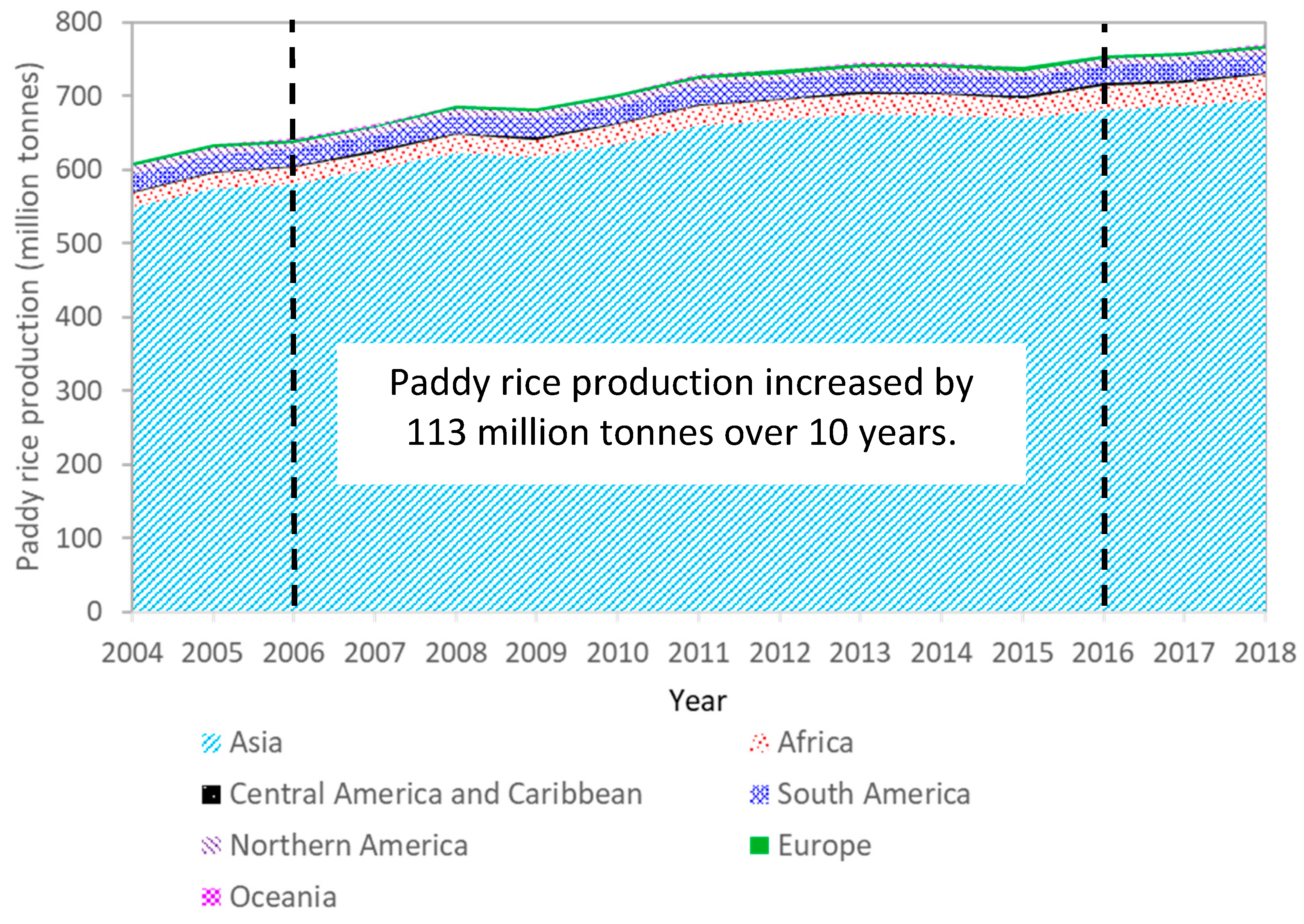
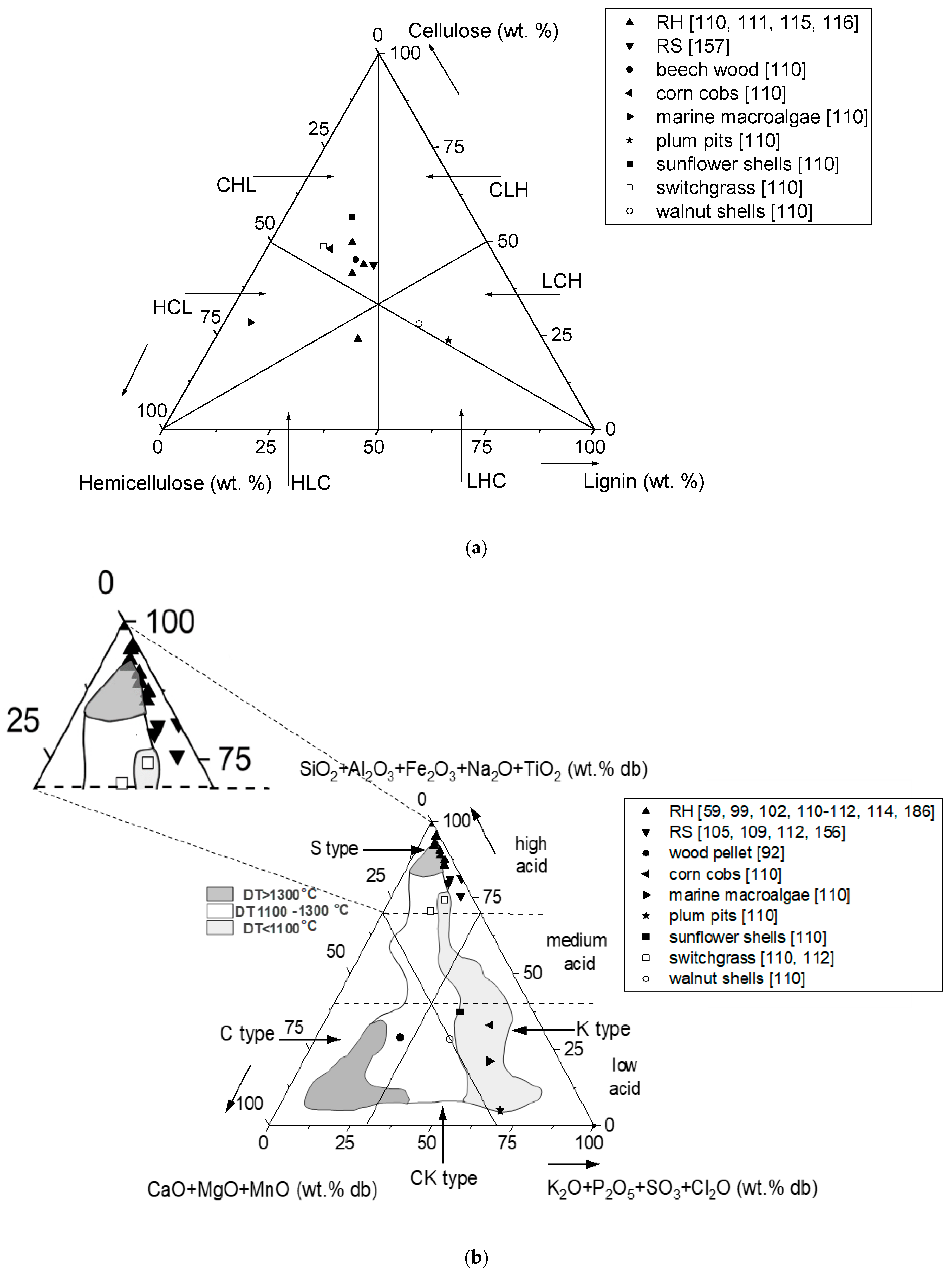

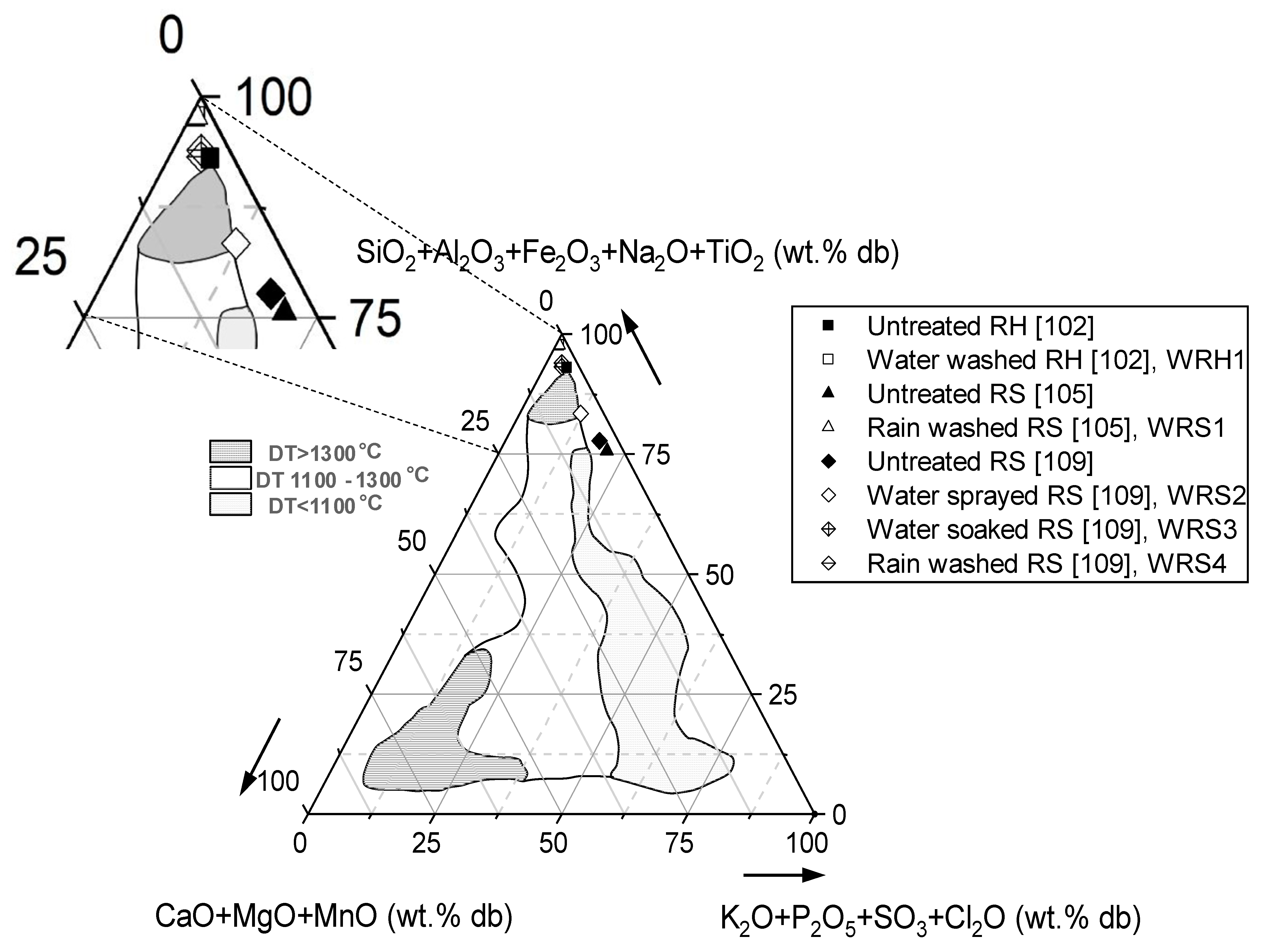
| Characteristic | Rice Husk (RH) | Rice Straw (RS) | Clean Wood Pellet 1 (W) | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Organic Type | CHL and HLC | CHL | CHL | |||||||||
| Inorganic Type | S Type | S Type | C Type | |||||||||
| Inorganic Sub-Type | HA | HA | LA | |||||||||
| Mean | Minimum | Maximum | n | Mean | Minimum | Maximum | n | Mean | Minimum | Maximum | n | |
| Moisture content (MC), wt % | 8.79 | 4.64 | 12.08 | 21 | 8.74 | 5.58 | 13.06 | 12 | 6.90 | 3.30 | 10.80 | 249 |
| Ash content (AC), wt % db | 16.30 | 7.68 | 24.60 | 48 | 16.52 | 9.22 | 22.60 | 26 | 0.40 | 0.22 | 0.68 | 249 |
| Volatile matter (VM), wt % db | 65.77 | 51.98 | 81.60 | 43 | 67.82 | 49.00 | 80.20 | 18 | 83.75 | 82.00 | 84.90 | 4 |
| Fixed carbon (FC), wt % db | 15.92 | 12.40 | 25.10 | 41 | 15.18 | 6.62 | 28.40 | 17 | 15.59 | 14.07 | 17.20 | 4 |
| C, daf 2 | 41.49 | 36.42 | 50.70 | 48 | 40.06 | 35.61 | 49.40 | 27 | 46.39 | 42.18 | 49.8 | 3 |
| O, daf | 38.51 | 29.33 | 53.70 | 48 | 38.01 | 29.23 | 53.64 | 26 | 46.95 | 43.7 | 50.54 | 3 |
| H, daf | 5.31 | 4.30 | 7.50 | 48 | 5.06 | 3.56 | 6.88 | 27 | 6.14 | 6.03 | 6.30 | 3 |
| N, daf | 0.84 | 0.09 | 4.26 | 47 | 0.76 | 0.17 | 1.41 | 27 | 0.11 | 0.05 | 0.51 | 174 |
| S, daf | 0.08 | 0.00 | 0.34 | 5 | 0.20 | 0.06 | 0.66 | 23 | 0.008 | <0.005 | 0.037 | 249 |
| Cl, daf | 0.11 | 0.01 | 0.20 | 7 | 0.57 | 0.08 | 1.01 | 11 | 0.009 | <0.005 | 0.042 | 249 |
| Low heating value (LHV) (MJ/kg) | 14.14 | 12.30 | 15.70 | 12 | 14.94 | 12.73 | 17.25 | 3 | 17.7 | 16.6 | 19.2 | 220 |
| Cellulose, wt % | 32.87 | 25.20 | 43.80 | 9 | 37.40 | 37.00 | 37.80 | 2 | 45.20 | 45.20 | 45.20 | 1 |
| Hemicellulose, wt % | 26.09 | 18.10 | 44.90 | 9 | 24.00 | 22.70 | 25.30 | 2 | 32.70 | 32.70 | 32.70 | 1 |
| Lignin, wt % | 21.73 | 13.60 | 34.80 | 9 | 18.45 | 13.60 | 23.30 | 2 | 22.10 | 22.10 | 22.10 | 1 |
| 3 | 0.34 | 0.17 | 0.78 | 7 | 0.59 | 0.07 | 1.94 | 14 | 3.63 | 4.43 | 2.65 | 81 |
| 1.23 | 0.24 | 3.21 | 8 | 2.99 | 1.60 | 10.12 | 13 | 37.94 | <5.97 | 19.94 | 249 | |
| 0.34 | 0.08 | 0.73 | 3 | 3.76 | 0.00 | 12.55 | 6 | 0.16 | 0.16 | 0.16 | 1 | |
| 0.23 | 0.10 | 0.40 | 8 | 0.46 | 0.10 | 0.98 | 13 | 1.72 | 1.09 | 2.67 | 3 | |
| 4.31 | 2.29 | 8.30 | 8 | 13.85 | 11.30 | 20.92 | 14 | 19.84 | <4.37 | 10.53 | 249 | |
| 0.72 | 0.19 | 2.13 | 8 | 2.13 | 1.49 | 5.02 | 13 | 8.24 | 32.53 | 5.00 | 249 | |
| 0.13 | 0.02 | 0.24 | 2 | 0.28 | 0.27 | 0.29 | 3 | 2.51 | 0.40 | 4.61 | 2 | |
| 0.19 | 0.03 | 0.37 | 7 | 1.31 | 0.14 | 2.71 | 14 | 1.20 | <1.15 | 8.64 | 249 | |
| 1.05 | 0.43 | 3.70 | 7 | 1.65 | 0.61 | 2.65 | 12 | 4.68 | 3.76 | 6.39 | 208 | |
| 89.92 | 86.92 | 94.38 | 8 | 71.33 | 55.08 | 82.13 | 14 | 23.72 | <45.61 | 42.90 | 249 | |
| 0.68 | 0.34 | 0.96 | 5 | 1.56 | 0.84 | 4.95 | 10 | 3.38 | 0.80 | 5.30 | 3 | |
| 0.02 | 0.01 | 0.02 | 5 | 0.03 | 0.01 | 0.09 | 9 | 0.89 | 0.1 | 1.6 | 3 | |
| Location | SiO2 | Na2O | K2O | CaO | MgO | Al2O3 | Fe2O3 | P2O5 | SO3 | Cl | Others 1 | Method | Ref. |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Egypt | 57.90 | N.R. | 14.60 | 2.30 | 3.40 | 1.00 | 1.00 | 13.80 | 3.20 | 2.30 | 0.50 | XRF 2 | [98] |
| Cambodia | 80.18 | N.R. | 3.89 | 1.55 | 0.81 | 0.83 | 4.29 | 3.80 | 2.05 | N.R. | 2.60 | XRF 2 | [72] |
| Italy | 83.20 | N.R. | 2.73 | 1.60 | 1.00 | 0.33 | 3.57 | 4.12 | 1.52 | N.R. | 1.92 | XRF 2 | [72] |
| Input Material, Source, Silica Content in Fuel Ash | Combustion Process | Exp. Technique | Scale | Main Findings | Reference |
|---|---|---|---|---|---|
| Material: RH Source: Riceland Foods, Arkansas in the US 1. Silica content: N.R. | RH was combusted at different temperatures (700, 750, 800, 850, 900, and 950 °C). Atmosphere: air | TORBED reactor | The reactor used was 400 mm in diameter, and average fuel feed rate was 26.5 kg/h. | Resulting RHA had silica purity between 95.2 and 96.7 wt %. With low reactor RT 2 of <10 min, RHA crystallization started at 950 °C. Loss of ignition (LOI) decreased from 1.8 to 1.0 wt % by increasing CT 3 from 700 to 900 °C. Furthermore, by raising CT from 700 to 900 °C, SSA 4, pore volume and average pore radius decreased from 37 to 6 m2/g, 0.11 to 0.02 cm3/g, and 1.9 to 1.2 nm, respectively. | Blissett et al., 2017 [19] |
| Material: RH Source: Heilongjiang province, China. Silica content: N.R. | RH was put in the furnace at 600, 700, 800, or 900 °C for 30 min. Atmosphere: air | Muffle furnace | Lab-scale | Silica purity in RHA obtained at 600 °C was 92.1 wt %. Both pore volume and SSA of the RHA drastically declined by increasing CT from 600 to 700 °C, and then slowly decreased by raising CT from 700 to 900 °C. SSA of the RHA was 145 and 25 m2/g at 600 and 700 °C, respectively. | Chen et al., 2017 [71] |
| Material: RH Source: state of Rio Grande do Sul, Brazil. Silica content: N.R. | Combustion at around 700 °C Atmosphere: air | RH was combusted in three different combustion technologies: moving grate reactor, suspension/entrained combustion chamber, and fluidized bed. | The output electricity power capacity of the moving grate reactor, suspension/entrained combustion chamber, and fluidized bed were 3.8, 12.5, and 5 MWh, respectively. | Quality of the RHA (silica purity and carbon content, and the structure of the ash) was affected by combustion technologies. The highest silica purity (96.7 wt %) and the lowest LOI (2.96%) were obtained using fluidized bed technology. Consequently, SSA was 11, 27 and 39 m2/g in fluidized bed, suspension/entrained combustion chamber, and moving grate reactor, respectively. XRD results indicated that the ashes from suspension/entrained combustion chamber are completely amorphous, while products from moving grate reactor and fluidized bed were completely or partially crystalline, respectively. | Fernandes et al., 2016 [179] |
| Material: RH Source: Harbin, Heilongjiang province, China. Silica content: N.R. | Heating RH at 5 K/min to 600 °C for 1 or 2 h, or to 700 °C for 1 h. Atmosphere: air | Muffle furnace with half-opened door for airflow control. | Lab-scale | Silica purity was 92.09, 93.00, and 93.42 wt %, LOI was 1.52, 1.48 and 3.24, and SSA was 86, 90 and 27 m2/g for combustion at 600 °C for 1 and 2 h, and combustion at 700 °C for 1 h, respectively. Results indicated that RHA obtained at 700 °C was darker than RHA obtained at 600 °C. It was interpreted that when conversion temperature is higher than decomposition temperature of potassium oxide in the fuel sample K2O melts and entraps the unburned carbon content. Therefore, remaining carbon in the ash is high for RHA obtained at 700 °C compared to the RHA produced at 600 °C. | Bie et al., 2015 [69] |
| Material: RH Source: Quzhou, Zhejiang, China. Silica content: N.R. | RH was combusted at 5 K/min from room temperature to 300 to 750 °C with RT of 2 h. Atmosphere: air | Muffle furnace | Lab-Scale | Silica purity of RHA increased from 29.7 wt % at 300 °C to 94.1 and 96.3 wt % at 600 and 750 °C, respectively. Consequently, the carbon content decreased from 48.8 wt % at 300 °C to 5.4 and 3.2 wt % at 600 and 750 °C, respectively. The structure of RHA remained amorphous up to 600 °C, and then a crystalline crystobalite peak appeared in XRD patterns. Results showed that SSA, total pore volume, micropore and mesopore volumes decrease by raising the CT. For instance, SSA decreased from 60 to 6 m2/g by increasing the CT from 300 to 750 °C. | Chen et al., 2011 [20] |
| Material: RH Source: N.R. Silica content: N.R. | Combustion of RH was investigated with slow and fast heating rates. In slow heating, the furnace was heated from room temperature to 700 °C at 5 K/min with RT of 4 h. In fast heating, RH was transferred to the furnace at 700 °C for 3 h. Atmosphere: N.R. | Muffle furnace with steel trays with size of 15 × 10 × 2.5 cm. | Lab-scale | The purity of biogenic silica and the carbon content were around the same in both RHAs obtained from slow and fast heating. The value of the silica purity for slow and fast heating rates was 90.20 and 89.80 wt %, respectively; whereas, the carbon content was around 4.2 wt % for both heating rates. However, brightness of the RHA obtained in slow heating was higher than the brightness for the fast heating rate. | Krishnarao et al., 2001 [188] |
| Symbol | Fuel Pre-Treatment Strategy |
|---|---|
| WRH 1 | RH was soaked in distilled water. No details have been reported for water washing. |
| WRS 1 | RS was collected after having received a total of 451 mm/m2 of rain. |
| WRS 2 | Laboratory washed, 100 g whole straw, hand sprayed for 1 min with tap water. |
| WRS 3 | Laboratory washed, 100 g whole straw, submerged in 7 liters of distilled water for 24 h. |
| WRS 4 | RS was collected after having received a total of 65 mm of rain (after first rain). |
| Input Material, Source, Silica Content in Fuel Ash | Fuel Pre-Treatment | Combustion Process | Scale | Main Findings | Reference |
|---|---|---|---|---|---|
| Material: RH and RS Source: Cambodia, Italy and Vietnam Silica content of the fuel ash: 50.7–83.2 wt %. |
| Multi-step sequential combustion at a heating rate of 10 K/min: 310 °C for 30 min, 450 °C for 60 min, 510 °C for 210 min and finally, 600 °C for 30 min. Atmosphere: air | Muffle furnace | Both RHA and RSA showed silica purity higher than 99 wt %. Italian RHA had the highest purity which was around 99.7 wt %. SSA1 was higher than 260 m2/g for all RHAs, and the highest value was for Cambodian RHA (300 m2/g). As the maximum processing temperature was lower than normal crystallization temperature, all RHAs and RSA were completely amorphous. It was concluded that water washing, which is swelling of the cell walls and pre-hydrolysis the carbohydrates, allow citric acid to reach and contact with the inner parts and as a result, remove the inner inorganic matter effectively. Regardless of the silicon purity in the starting material, the process had a potential to produce biogenic silica with almost uniform quality. | Schneider et al., 2018 [72] |
| Material: RH Source: N.R. Silica content of the fuel ash: N.R. |
| Combustion at 800 °C. Although it was called pyrolysis in the paper, but there is no indication for it. Furthermore, the results were compared with combustion studies. Atmosphere: N.R. | Muffle furnace | XRD patterns showed that all RHAs were amorphous. Silica purity of RHA increased using fuel pre-treatment. The value for untreated samples and those treated with sulfuric acid, hydrogen chloride, oxalic acid, and ionic liquid was 94.7, 99.6, 98.0 and 99.5 wt %, respectively, whereas the carbon content for these ashes was around the same (between 0.02 and 0.08 wt %. Results indicated that ashes obtained from treated RH had no potassium content while untreated sample had around 1.7 wt % of K2O. N2 sorption data showed that SSA and pore volume of untreated samples and treated with sulfuric acid, hydrogen chloride, oxalic acid, and ionic liquid were 99, 85, 66, 71 and 185 m2/g and 0.17, 0.22, 0.18, 0.21 and 0.41 cm3/g, respectively. | Lee et al., 2017 [91] |
| Material: RH Source: Heilongjiang province, China. Silica content of the fuel ash: N.R. |
| As-received RH and acid-leached RH (LRH) were burned at 600, 700, 800, and 900 °C for 30 min while air flow was continuously fed in during the combustion process. Atmosphere: air | Muffle furnace | Result of inductively coupled plasma–optical emission spectrometry (ICP-OES) showed that silica purity in acid leached RHA (LRHA) samples, which were obtained at 600 °C, was in the range of 96.5–98.6 wt %. This range is much higher than the silica purity of untreated RHA 92.1 wt %. Furthermore, K2O content was around 0.2–0.6 wt % in LRHAs; while, it was 4.0 wt % in untreated RHA. Both pore volume and SSA of the LRHA were higher than these values of original RHA. The maximum value of the SSA and pore volume obtained from sulfuric acid leached ash at 60 °C, which were 237 m2/g and 0.084 cm3/g, whereas these parameters were 145 m2/g and 0.051 cm3/g in untreated RHA, respectively. It is worth mentioning that the reported pore volumes are not reliable, as the authors did not measured the complete isotherm for their calculation. | Chen et al., 2017 [71] |
| Material: RH Source: Selangor, Malaysia. Silica content of the fuel ash: N.R. |
| Combustion at 500, 600. 700, 800 and 900 °C for 2 h. Atmosphere: N.R. | Muffle furnace | Silica purity measured by XRF analysis, Brunauer–Emmett–Teller specific surface area (BET SSA) and total pore volume improved from 95.8 wt %, 116 m2/g and 0.23 cm3/g in non-leached RHA to 99.1 wt %, 208 m2/g and 0.31 cm3/g in sulfuric acid leached RHA and to 99.6 wt %, 218 m2/g and 0.32 cm3/g in hydrochloric acid leached RHA, respectively. Moreover, crystallization temperature was decreased and impurities were drastically removed from the ash samples by using acid leaching strategy before the combustion. | Bakar el al., 2016 [199] |
| Material: RH Source: Jiangsu, China. Silica content of the fuel ash: N.R. |
| A sequential burning process was applied as follows: (1) 5 g of LRH was pyrolyzed at 300–800 °C under nitrogen or carbon dioxide atmosphere with flow rate of 1 l/min at heating rate of 20 K/min for 30 min. (2) combustion of pyrolyzed RH (PRH) was carried out at 610 °C for 2–3 h under oxygen flow with the same flow rate at heating rate of 10 K/min. Atmosphere: Oxygen | Tubular furnace | Silica purity, SSA and total pore volume of synthesized amorphous ashes were in the range of 95.8–99.6 wt %, 204–353 m2/g and 0.35–0.52 cm3/g, respectively. | Gu et al., 2015 [195] |
| Material: RH Source: Kafr El-Daowar, Egypt. Silica content of the fuel ash: 57.9 wt % |
| Multi-step sequential combustion in air at a heating rate of 10 K/min: (1) 310 °C for 60 min, 400 °C for 120 min, 510 °C for 300 min and finally, 600 °C for 30 min. Atmosphere: air | Muffle furnace | XRF results indicated that silica purity increased by simply washing the RH from 57.9 wt % in the unwashed RH fuel ash to 85.6 wt % in washed RH fuel ash. In addition, selected multi-step temperature program improved silica purity to 97.7 wt % in LRHA. Nitrogen sorption analysis showed that both SSA and pore volume were increased from 220 m2/g and 0.26 cm3/g in non-leached RHA to 313 m2/g and 0.38 cm3/g in LRHA, respectively. Furthermore, internal structure analysis revealed that both RHA and LRHA had amorphous structure, since the maximum processing temperature (600 °C) was lower than the crystallization temperature. | Ahmad Alyosef et al., 2013 [98] |
| Material: RS Source: different regions in Spain and Egypt. Silica content of the fuel ash: N.R. |
| RS and WRS was heated up from 30 to 1000 °C at a heating rate of 10 K/min. Air flow rate was 100 mL/min. Atmosphere: air | TG-DSC | Simply washing of RS with tap water reduced Na, K, Mg, P, S and Cl elements in the ashes with the removal percentages of 35.6–60.0, 26.1–49.5, 0.0–38.8, 0.0–34.8, 25.5–59.4 and 59.0–87.0%, respectively. As a result, water washing controlled slag formation during the combustion of RS from different regions. | Said et al., 2013 [198] |
| Material: RH Source: Wuyunjing, China. Silica content of the fuel ash: N.R. | RH was leached with deionized water at 25 ± 1 °C for 4 h. | RH and WRH was calcined and held at a maximum temperature (600, 700 and 800 °C) for 0.5, 1.5, 2.5 and 3.5 h. Atmosphere: air | N.R. | The effect of calcination parameters (CT2 and RT3) on RHA content was studied in RH and WRH. It was shown that the ash content decreases by increasing CT in both RH and WRH samples. That is because some of the metallic elements show higher volatility at higher calcination temperatures. Results indicated that in the original RH, ash content was dramatically decreased by raising RT, which is because of the slow oxidation of carbon residues in original RH. On the other hand, WRH had no change in ash content when the RT is longer than 1.5 h, which implies no carbon residues in RHA, and it was interpreted as an indication for pure silica production from WRH. It was shown that increasing the CT and RT decreases the SSA of the WRHA, which is because of the agglomeration effect and diminishing porosity of the ash. | Shen et al., 2011 [189] |
| Material: RH Source: Niigata, Japan. Silica content of the fuel ash: N.R. |
| LRH was burned at 800 °C for 30 min under airflow rate of 0.42 mL/s using a small air-compressor. Atmosphere: air flow. | Muffle furnace | XRF results indicated that leaching RH with citric acid solution and a concentration over 1 wt % produces RHA with silica purity higher than 99.5 wt %, whereas silica purity of untreated RHA was 97.2 wt %. Potassium oxide content was 1.39 wt % in untreated RHA. However, acid leaching process reduced its level into 0.01–0.03 wt % in LRHA samples. Furthermore, it was shown that silica purity is not sensitive to concentration of acid solution and temperature of acid leaching process, and only 1 wt % of citric acid leaching at room temperature was enough to remove potassium and carbon contents to produce pure biogenic silica. Maximum silica purity was obtained from RH leached with 5 wt % of acid solution at 80 °C, which was 99.77 wt % (compared to 99.47 wt % upon leaching with 1 wt % of citric acid at room temperature). XRD patterns showed that acid leaching improves LRHA resistance to crystallization. | Umeda and Kondoh 2010 [187] |
| Material: RH Source: Niigata, Japan. Silica content of the fuel ash: N.R. |
| As-received RH and LRH were burned in air (150 mL/min) at 600–1150 °C. Atmosphere: air | TG-DSC | Results of XRF analysis showed that silica purity was improved from 94.6 in untreated RHA to higher than 99 wt % in LRHA. Furthermore, LRHA with lower metallic impurities remained amorphous until 1050 °C, which is the highest value published in literature. | Umeda and Kondoh, 2008 [67] |
| Material: RH Source: Niigata, Japan. Silica content of the fuel ash: N.R. |
| As-received RH and LRH were burned in air at 600 and 1000 °C. Atmosphere: air | TG-DTA | Leaching RH with only 1 wt % of sulfuric acid solution reduced calcium, potassium and sodium oxide impurities from chemical composition of LRHA drastically. Consequently, not only the silica purity improved, but also ash remained amorphous up to 1000 °C, whereas untreated original raw RH transformed into crystalline species at temperatures above 854 °C. | Umeda et al., 2007 [200] |
| Material: RH Source: Andhra Pradesh State, India. Silica content of the fuel ash: N.R. |
| LRH was heated up from 300 to 1000 °C at an interval of 100 °C at different heating rates of 1, 2, 3, 5, 7 or 10 K/min and RTs of 2, 4 or 6 h. Atmosphere: N.R. | Muffle furnace | Acid leaching followed by combustion at 700 °C for 2 h increased silica purity from 89.5 wt % in ash from untreated RH to 97.8 wt % in the ash from LRH sample. Loss of ignition (LOI) test showed that in RHA obtained from untreated RH, in spite of the presence of some black particles compare to the ash from LRH, the value of LOI is almost the same with LRHA. It was concluded that the black particles contributes to fix carbon in RHA. It was shown that ash obtained from LRH switch from amorphous to crystalline form at higher temperatures compared to the RHA produced from untreated RH. Increasing the heating rate improved surface area and pore volume and decreased the brightness of the ash samples. It is because of higher carbon content in RHAs obtained at higher heating rates. | Chandrasekhar et al., 2006 [54] |
| Material: RH Source: Kerala state (KRH) and Andhra Pradesh state (APRH), India. Silica content of the fuel ash: N.R. |
| LRH, WRH, as received RH samples were burned out at 5 K/min to 700 °C with RT of 2 h. Atmosphere: N.R. | Muffle furnace | Irrespective of CT and RT, black particles appeared in the ash obtained from APRH. In spite of visible black particles in ashes obtained from APRH source, LOI had around the same value all resulting ashes from two different sources, which was in the range of 1.8–4.6 wt %. Therefore, it indicates that there is no direct connection between black particles in the ash and the carbon content obtained by LOI. Further pre-treatment using different acids improved the silica purity, and the maximum reported value was 97.8 wt %. Nitrogen sorption analysis showed that RHA produced from RH with lower potassium content (KRH) had much higher SSA (approx. 150 m2/g), while in the sample with higher potassium content (APRH), this value was less than 10 m2/g for the same combustion conditions. Pore volume behaves in the same manner, and it was higher in the ash of KRH compared to the ash of APRH. Furthermore, acid leaching improved both SSA and pore volume in both samples. | Chandrasekhar et al., 2005 [53] |
| Material: RH Source: N.R. Silica content of the fuel ash: N.R. |
| LRH was heated up from room temperature to 727 °C at heating rates of 5, 10, 15, and 20 K/min. Atmosphere: air | Thermogravimetric analysis (TG-DTG) in a form of tube reactor | The maximum silica purity, SSA and total pore volume in LRHA reached to 99.7 wt %, 235 m2/g and 0.32 cm3/g for combustion at lower heating rate. All LRHA obtained from different heating rates were completely amorphous. | Liou, 2004 [191] |
| Material: RH Source: Trakya Region, Turkey. Silica content of the fuel ash: N.R. |
| 20 g of RH, WRH and LRH was incinerated at 600 °C in four different ways: (1) combustion for 4 h in static air in muffle furnace; (2) incineration in tubular reactor under argon (1.5 L/min, for 3 h) and then supplying oxygen (1.0 L/min for 1 h); (3) combustion in tubular reactor under air flow (3 L/min, for 3 h) and; (4) combustion in tubular reactor under oxygen flow (1.0 L/min, for 2 h). Atmosphere: air and oxygen. | Muffle furnace & a tubular reactor placed horizontal inside the muffle oven | Incineration of untreated RH under oxygen atmosphere produced amorphous RHA with silica purity of 98.3 wt %. However, silica purity in the static air condition was 91.5 wt %. Result of RH, WRH, and LRH combustion under static air showed that silica purity increased from 91.5 wt % in the untreated sample to 95.5 wt % and 99.2 wt % in distilled water washed and HCl-leached samples, respectively. Furthermore, maximum purity obtained from sulfuric acid pre-treatment (99.6 wt %). | Yalçin and Secinç, 2001 [181] |
| Input Material, Source, Silica Content in Fuel Ash | Pre-Treatment Process | Combustion Process | Scale | Post-Treatment Process | Main Findings | Reference |
|---|---|---|---|---|---|---|
| Material: RH Source: Cambodia and Italy. Silica content of the fuel ash: N.R. |
| Combustion was carried out using ÖKOTHERM® boilers, and RH was continuously supplied into the boilers. | Muffle oven for lab scale studies and post-treatment and ÖKOTHERM® boilers with power up to 120 kW for bench-scale studies. |
| This research is the first known attempt to consider both energy and material applications of agricultural residues in a real combustion unit. Some optimization for huge ash loading was done by modifying the conveying screw and using additional mechanical equipment to mobilize and transport the voluminous ash inside the boiler. As a result, the operation time was prolonged with no limit in fuel handling and ash discharged. Considering the principles of green chemistry by using less chemical resources, the cost of the process as well as the quality of the obtained biogenic silica, water washing and 2 wt % citric acid leaching at 50 °C for 2 h were selected, respectively, as the pre-treatment and post-treatment. The final product had an amorphous internal structure with BET SSA 1, specific mesoporous volume and silica purity of 185 m2/g, 0.25 cm3/g, and >98 wt %, respectively. | Schliermann et al., 2018 [73] |
| Material: RHA was supplied by a local industry (Fumacense, Morro da Fumaça, SC, Brazil). Silica content: 72.1 wt % | - | N.R. | Electric oven with crucible of 24.5 cm diameter. | RHA was heated from room temperature to 400, 500, 600, or 700 °C for 3–6 h with heating rate of 10 K/min. | Simply heating the RHA to 700 °C for 6 h improved silica purity of the ash from 72.1 to 95 wt %, and RHA remained in amorphous form. However, SSA was changed from 177 m2/g in the as-received RHA to 54 m2/g in reheated RHA sample. | Della et al., 2002 [64] |
| Material: RH Source: Trakya Region, Turkey. Silica content of the fuel ash: N.R. |
| 20 g of RH, WRH, LRH was incinerated at 600 °C. Atmosphere: air | Muffle furnace | RHA produced from untreated and acid leached RH was leached with 3% (v/v) HCl at a ratio of 50 g WRH/l for 2 h. | The order of the silica purity was acid pre- and post-treated sample (99.7 wt %) > acid pre-treated sample (99.2 wt %) > water pre-treated sample (95.5 wt %) > acid post-treated sample (95.1 wt %) > untreated sample (91.5 wt %). SSA results revealed that the order of the SSA was untreated sample (63 m2/g), water washed sample (194 m2/g), acid pre- and post-treated sample (244 m2/g), and acid pre-treated sample (321 m2/g), respectively. | Yalçin and Secinç, 2001 [181] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Beidaghy Dizaji, H.; Zeng, T.; Hartmann, I.; Enke, D.; Schliermann, T.; Lenz, V.; Bidabadi, M. Generation of High Quality Biogenic Silica by Combustion of Rice Husk and Rice Straw Combined with Pre- and Post-Treatment Strategies—A Review. Appl. Sci. 2019, 9, 1083. https://doi.org/10.3390/app9061083
Beidaghy Dizaji H, Zeng T, Hartmann I, Enke D, Schliermann T, Lenz V, Bidabadi M. Generation of High Quality Biogenic Silica by Combustion of Rice Husk and Rice Straw Combined with Pre- and Post-Treatment Strategies—A Review. Applied Sciences. 2019; 9(6):1083. https://doi.org/10.3390/app9061083
Chicago/Turabian StyleBeidaghy Dizaji, Hossein, Thomas Zeng, Ingo Hartmann, Dirk Enke, Thomas Schliermann, Volker Lenz, and Mehdi Bidabadi. 2019. "Generation of High Quality Biogenic Silica by Combustion of Rice Husk and Rice Straw Combined with Pre- and Post-Treatment Strategies—A Review" Applied Sciences 9, no. 6: 1083. https://doi.org/10.3390/app9061083
APA StyleBeidaghy Dizaji, H., Zeng, T., Hartmann, I., Enke, D., Schliermann, T., Lenz, V., & Bidabadi, M. (2019). Generation of High Quality Biogenic Silica by Combustion of Rice Husk and Rice Straw Combined with Pre- and Post-Treatment Strategies—A Review. Applied Sciences, 9(6), 1083. https://doi.org/10.3390/app9061083







