Potential Applications of Clay-Based Therapy for the Reduction of Pesticide Exposures in Humans and Animals
Abstract
Featured Application
Abstract
1. Introduction
2. Materials and Methods
2.1. Reagents
2.2. Synthesis of Sorbents
2.3. Coefficient of Linear Expansibility in Water
2.4. Surface Area Determination
2.5. In Vitro Isothermal Adsorption
2.6. Data Calculations and Curve Fitting
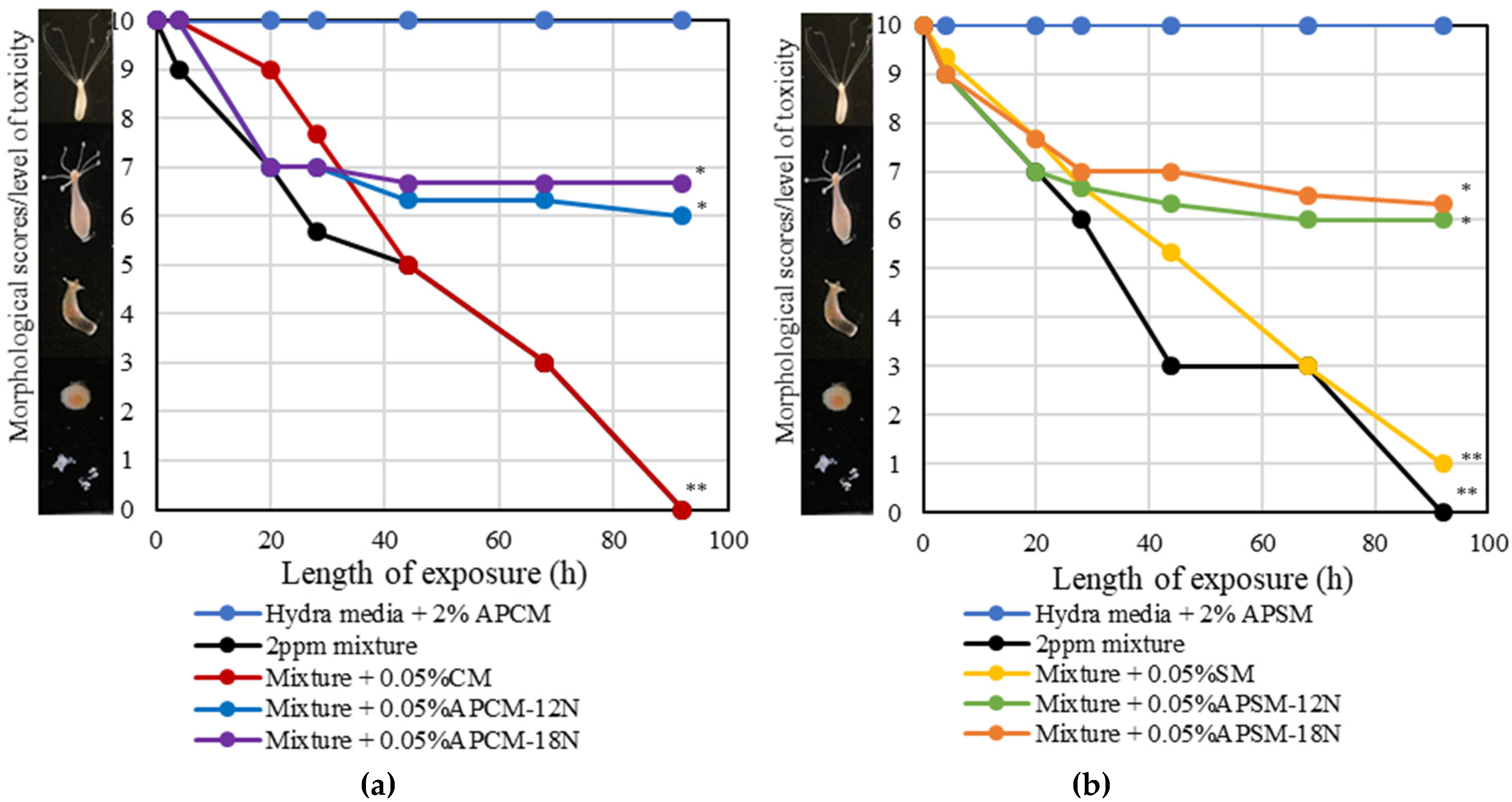
2.7. Hydra Assay
2.8. Statistical Analysis
3. Results
3.1. Coefficient of Linear Expansibility in Water
3.2. Surface Area
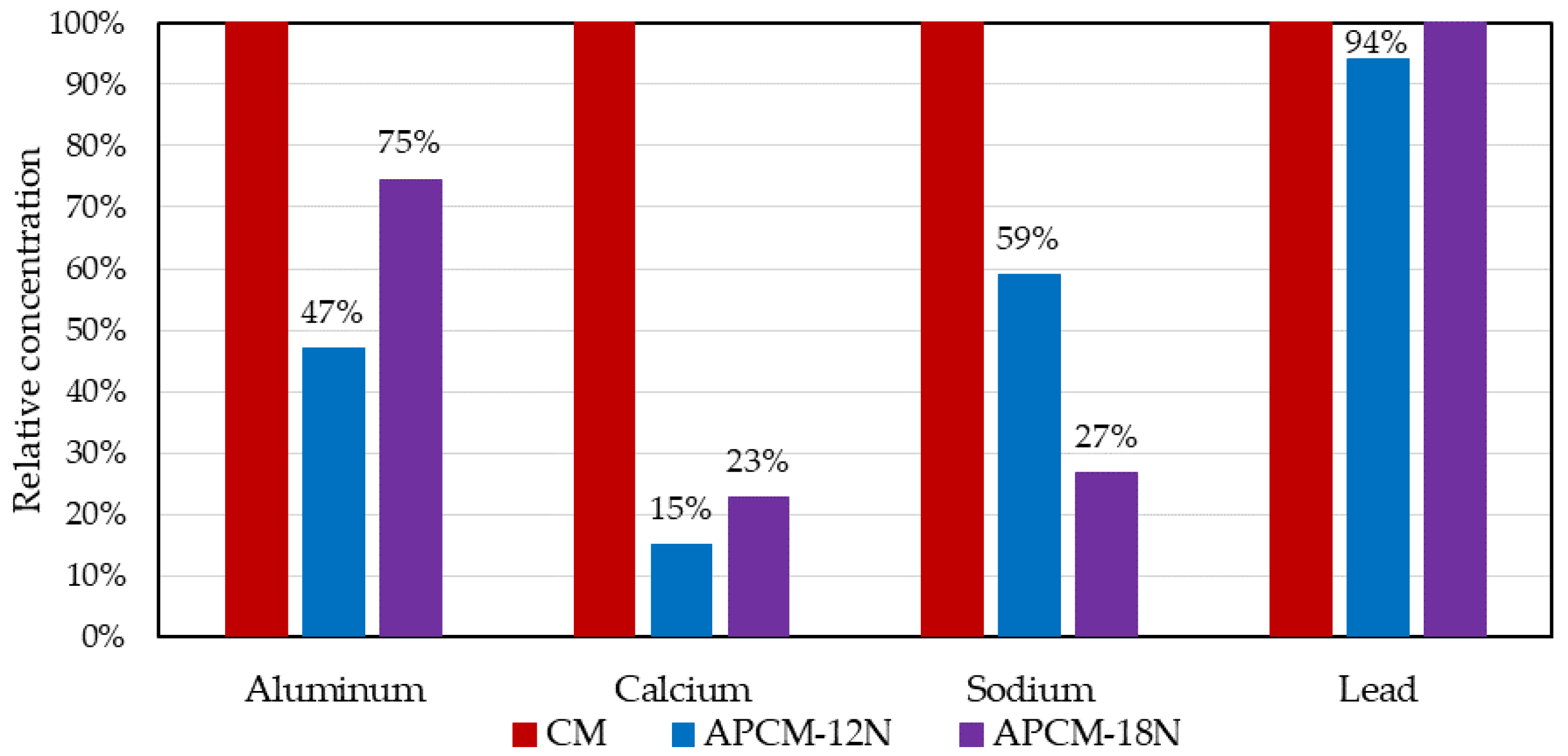
3.3. Trace Metals
3.4. Adsorption Analyses
4. Discussion
Author Contributions
Funding
Conflicts of Interest
References
- Alavanja, M.C.R. Pesticides use and exposure extensive worldwide. Rev. Environ. Health 2009, 24, 303–309. [Google Scholar] [CrossRef] [PubMed]
- Gavrilescu, M. Fate of pesticides in the environment and its bioremediation. Eng. Life Sci. 2005, 5, 497–526. [Google Scholar] [CrossRef]
- Yavari, S.; Malakahmad, A.; Sapari, N.B. Biochar efficiency in pesticides sorption as a function of production variables—A review. Environ. Sci. Pollut. Res. 2015, 22, 13824–13841. [Google Scholar] [CrossRef] [PubMed]
- Mughari, A.R.; Vázquez, P.P.; Galera, M.M. Analysis of phenylurea and propanil herbicides by solid-phase microextraction and liquid chromatography combined with post-column photochemically induced fluorimetry derivatization and fluorescence detection. Anal. Chim. Acta 2007, 593, 157–163. [Google Scholar] [CrossRef]
- FAO (Food and Agriculture Organization of the United Nations); WHO (World Health Organization). International Code of Conduct on Pesticides Management. Rome, 2014. Available online: http://www.fao.org/fileadmin/templates/agphome/documents/Pests_Pesticides/Code/CODE_2014Sep_ENG.pdf (accessed on 5 December 2019).
- UTZ. List of Banned Pesticides and Pesticides Watchlist. The Netherlands, 2015. Available online: https://utz.org/wp-content/uploads/2015/12/EN_UTZ_List-of-Banned-PesticidesWatchlist_v1.0_2015.pdf (accessed on 5 December 2019).
- Patnaik, P. Pesticides and herbicides: Classification, structure, and analysis. In A Comprehensive Guide to the Hazardous Properties of Chemical Substances, 3rd ed.; Esposito, B., Ed.; John Wiley & Sons, Inc: Hoboken, NJ, USA, 2007; pp. 741–742. [Google Scholar]
- Dai, M.; Copley, S.D. Genome shuffling improves degradation of the anthropogenic Pesticide Pentachlorophenol by Sphingobium chlorophenolicum ATCC 39723. Appl. Environ. Microbiol. 2004, 70, 2391–2397. [Google Scholar] [CrossRef] [PubMed]
- Zheng, W.; Yu, H.; Wang, X.; Qu, W. Systematic review of pentachlorophenol occurrence in the environment and in humans in China: Not a negligible health risk due to the re-emergence of schistosomiasis. Environ. Int. 2012, 42, 105–116. [Google Scholar] [CrossRef]
- Hameed, B.H. Equilibrium and kinetics studies of 2,4,6-trichlorophenol adsorption onto activated clay. Colloid Surf. A Physicochem. Eng. Asp. 2007, 307, 45–52. [Google Scholar] [CrossRef]
- Gao, J.; Liu, L.; Liu, X.; Zhou, H.; Huang, S.; Wang, Z. Levels and spatial distribution of chlorophenols-2,4-Dichlorophenol, 2,4,6-trichlorophenol, and pentachlorophenol in surface water of China. Chemosphere 2008, 71, 1181–1187. [Google Scholar] [CrossRef]
- Pool-Zobel, B.L.; Guigas, C.; Klein, R.; Neudecker, C.; Renner, H.W.; Schmezer, P. Assessment of genotoxic effects by Lindane. Food Chem. Toxicol. 1993, 31, 271–283. [Google Scholar] [CrossRef]
- Kalender, S.; Ogutcu, A.; Uzunhisarcikli, M.; Acikgoz, F.; Durak, D.; Ulusoy, Y.; Kalender, Y. Diazinon-induced hepatotoxicity and protective effect of vitamin E on some biochemical indices and ultrastructural changes. Toxicology 2005, 211, 197–206. [Google Scholar] [CrossRef]
- Dejonghe, W.; Berteloot, E.; Goris, J.; Boon, N.; Crul, K.; Maertens, S.; Hofte, M.; Vos, P.D.; Verstraete, W.; Top, E.M. Synergistic degradation of linuron by a bacterial consortium and isolation of a single linuron-degrading variovorax strain. Appl. Environ. Microbiol. 2002, 69, 1532–1541. [Google Scholar] [CrossRef] [PubMed]
- Lambright, C.; Ostby, J.; Bobseine, K.; Wilson, V.; Hotchkiss, A.K.; Mann, P.C.; Gray, L.E. Cellular and molecular mechanisms of action of linuron: An antiandrogenic herbicide that produces reproductive malformations in male rats. Toxicol. Sci. 2000, 56, 389–399. [Google Scholar] [CrossRef] [PubMed]
- Probst, G.W.; Golab, T.; Herberg, R.J.; Holzer, F.J.; Parka, S.J. Fate of trifluralin in soils and plants. J. Agric. Food Chem. 1976, 15, 592–599. [Google Scholar] [CrossRef]
- Wallace, D.R. Trifluralin. In Encyclopedia of Toxicology, 3rd ed.; Wexler, P., Ed.; Elsevier: Amsterdam, The Netherlands, 2014; pp. 846–848. [Google Scholar]
- Zen, J.; Jeng, S.; Chen, H. Determination of paraquat by square-wave voltammetry at a perfluorosulfonated ionomer/clay-modified electrode. Anal. Chem. 1996, 68, 498–502. [Google Scholar] [CrossRef] [PubMed]
- Bertrand, D.B. Photodegradation of the carbamate pesticides aldicarb, carbaryl and carbofuran in water. Anal. Chim. Acta 1991, 254, 235–244. [Google Scholar] [CrossRef]
- Tiemann, U. In vivo and in vitro effects of the organochlorine pesticides DDT, TCPM, methoxychlor, and lindane on the female reproductive tract of mammals: A review. Reprod. Toxicol. 2008, 25, 316–326. [Google Scholar] [CrossRef] [PubMed]
- Yarsan, E.; Tanyuksel, M.; Celik, S.; Aydin, A. Effects of aldicarb and malathion on lipid peroxidation. Bull. Environ. Contam. Toxicol. 1999, 63, 575–581. [Google Scholar] [CrossRef]
- Phillips, T.D.; Wang, M.; Elmore, S.E.; Hearon, S.; Wang, J.S. NovaSil clay for the protection of humans and animals from aflatoxins and other contaminants. Clays Clay Miner. 2019, 67, 99–110. [Google Scholar] [CrossRef]
- Phillips, T.D. Dietary clay in the chemoprevention of aflatoxin-induced disease. Toxicol. Sci. 1999, 52 (Suppl. S2), 118–126. [Google Scholar] [CrossRef]
- Wang, M.; Hearon, S.E.; Phillips, T.D. Development of enterosorbents that can be added to food and water to reduce toxin exposures during disasters. J. Environ. Sci. Health B 2019, 54, 514–524. [Google Scholar] [CrossRef]
- Wang, M.; Safe, S.; Hearon, S.E.; Phillips, T.D. Strong adsorption of polychlorinated biphenyls by processed montmorillonite clays: Potential applications as toxin enterosorbents during disasters and floods. Environ. Pollut. 2019, 255, 113210. [Google Scholar] [CrossRef] [PubMed]
- Tkac, I.; Konadel, P.; Muller, D. Acid-treated montmorillonites-a study by Si and Al MAS NMR. Clay Miner. 1994, 29, 11–19. [Google Scholar] [CrossRef]
- Tyagi, B.; Chudasama, C.D.; Jasra, R.V. Determination of structural modification in acid activated montmorillonite clay by FT-IR spectroscopy. Spectrochim. Acta A Mol. Biomol. Spectrosc. 2006, 64, 273–278. [Google Scholar] [CrossRef] [PubMed]
- Grant, P.G.; Phillips, T.D. Isothermal adsorption of aflatoxin B (1) on HSCAS clay. J. Agric. Food Chem. 1998, 46, 599–605. [Google Scholar] [CrossRef]
- Wang, M.; Hearon, S.E.; Johnson, N.M.; Phillips, T.D. Development of broad-acting clays for the tight adsorption of benzo [a] pyrene and aldicarb. Appl. Clay Sci. 2019, 168, 196–202. [Google Scholar] [CrossRef]
- Wang, M.; Maki, C.R.; Phillips, T.D. Development of high capacity enterosorbents for aflatoxin B1 and other hazardous chemicals. Chem. Res. Toxicol. 2017, 30, 1694–1701. [Google Scholar] [CrossRef]
- Carter, D.L.; Mortland, M.M.; Kemper, W.D. Specific surface. In Methods of Soil Analysis, Part 1, Physical and Mineralogical Methods, 2nd ed.; Klute, A., Ed.; Soil Science Society of America: Madison, WI, USA; American Society of Agronomy: Madison, WI, USA, 1986; pp. 413–422. [Google Scholar]
- Diaz, P.E.; Leyva, R.; Guerrero, R.M.; Mendoza, J. Adsorption of pentachlorophenol from aqueous solution onto activated carbon fiber. Ind. Eng. Chem. Res. 2006, 45, 330–336. [Google Scholar] [CrossRef]
- Gonen, Y.; Rytwo, G. Using a Matlab implemented algorithm for UV-vis spectral resolution for pKa determination and multicomponent analysis. Anal. Chem. Insights 2009, 4, 21–27. [Google Scholar] [CrossRef]
- Yi, A.; Feng, Y.; Du, Z.; Li, H. Mechanism of 2, 4, 6-Trichlorophenol degradation in microbial fuel cells system with microbe isolated from submarine sediment. Int. J. Electrochem. Sci. 2015, 10, 1459–1468. [Google Scholar]
- Challa, R.; Naidu, N.V.S. Development and validation of HPLC method for estimation of lindane in its formulation. Sch. Res. Libr. 2016, 8, 23–26. [Google Scholar]
- Sanchez-Martin, M.J.; Delgado-Pascual, R.; Iglesias-Jimenez, E.; Sanchez-Camazano, M. Determination of linuron in aqueous soil extracts by high-performance liquid chromatography. J. Chromatogr. A 1996, 754, 295–299. [Google Scholar] [CrossRef]
- Sigma-Aldrich Co. Application Note 134. Available online: https://www.sigmaaldrich.com/content/dam/sigma-aldrich/docs/Supelco/Application_Notes/1/46741.pdf (accessed on 5 December 2019).
- Wittrig, B. Comprehensive Pesticide Residue Analysis by LC-MS/MS Using an Ultra Aqueous C18 Column. 2019. Available online: https://www.restek.com/adv004 (accessed on 5 December 2019).
- Zou, T.; He, P.; Cao, J.; Li, Z. Determination of paraquat in vegetables using HPLC-MS-MS. J. Chromatogr. Sci. 2014, 53, 204–209. [Google Scholar] [CrossRef] [PubMed]
- Abdellaoui, Y.; Olguinm, M.T.; Abatal, M.; Ali, B.; Mendez, S.E.D.; Santiago, A.A. Comparison of the divalent heavy metals (Pb, Cu and Cd) adsorption behavior by montmorillonite-KSF and their calcium- and sodium-forms. Superlattice Microst. 2019, 127, 165–175. [Google Scholar] [CrossRef]
- Brown, K.A.; Mays, T.; Romoser, A.; Marroquin-Cardona, A.; Mitchell, N.J.; Elmore, S.E.; Phillips, T.D. Modified hydra bioassay to evaluate the toxicity of multiple mycotoxins and predict the detoxification efficacy of a clay-based sorbent. J. Appl. Toxicol. 2014, 34, 40–48. [Google Scholar] [CrossRef]
- Dash, B.; Phillips, T.D. Molecular Characterization of a Catalase from Hydra vulgaris. Gene 2012, 501, 144–152. [Google Scholar] [CrossRef]
- Caglar, B.; Afsin, B.; Koksal, E.; Tabak, A.; Eren, E. Characterization of Unye bentonite after treatment with sulfuric acid. Quim. Nova 2013, 36, 955–959. [Google Scholar] [CrossRef]
- Kooli, F.; Hian, P.C.; Werong, Q.; Alshahateet, S.F.; Chen, F. Effect of the acid-activated clays on the properties of porous clay heterostructures. J. Porous Mater. 2006, 13, 319–324. [Google Scholar] [CrossRef]
- Krupskaya, V.V.; Zakusin, S.V.; Tyupina, E.A.; Dorzhieva, O.V.; Zhukhlistov, A.P.; Belousov, P.E.; Timofeeva, M.N. Experimental study of montmorillonite structure and transformation of its properties under treatment with inorganic acid solutions. Minerals 2017, 7, 49. [Google Scholar] [CrossRef]
- Amari, A.; Gannouni, H.; Khan, M.I.; Almesfer, M.K.; Elkhaleefa, A.M.; Gannouni, A. Effect of structure and chemical activation on the adsorption properties of green clay minerals for the removal of cationic dye. Appl. Sci. 2018, 8, 2302. [Google Scholar] [CrossRef]
- Mitchell, N.J.; Xue, K.S.; Lin, S.; Marroquin-Cardona, A.; Brown, K.A.; Elmore, S.E.; Tang, L.; Romoser, A.; Gelderblom, W.C.; Wang, J.S.; et al. Calcium montmorillonite clay reduces AFB1 and FB1 biomarkers in rats exposed to single and co-exposures of aflatoxin and fumonisin. J. Appl. Toxicol. 2014, 34, 795–804. [Google Scholar] [CrossRef]
- Wang, M.; Orr, A.A.; He, S.; Dalaijamts, C.; Chiu, W.A.; Tamamis, P.; Phillips, T.D. Montmorillonites Can Tightly Bind Glyphosate and Paraquat Reducing Toxin Exposures and Toxicity. ACS Omega 2019, 4, 17702–17713. [Google Scholar] [CrossRef] [PubMed]
- Maki, C.R.; Haney, S.; Wang, M.; Ward, S.H.; Rude, B.J.; Bailey, R.H.; Harvey, R.B.; Phillips, T.D. Calcium montmorillonite clay for the reduction of aflatoxin residues in milk and dairy products. JDVS 2017, 2, 1–8. [Google Scholar]
- Phillips, T.D.; Afriyie-Gyawu, E.; Williams, J.; Huebner, H.; Ankrah, N.A.; Ofori-Adjei, D.; Jolly, P.; Johnson, N.; Taylor, J.; Marroquin-Cardona, A.; et al. Reducing human exposure to aflatoxin through the use of clay: A review. Food Addit. Contam. A 2008, 25, 134–145. [Google Scholar] [CrossRef] [PubMed]



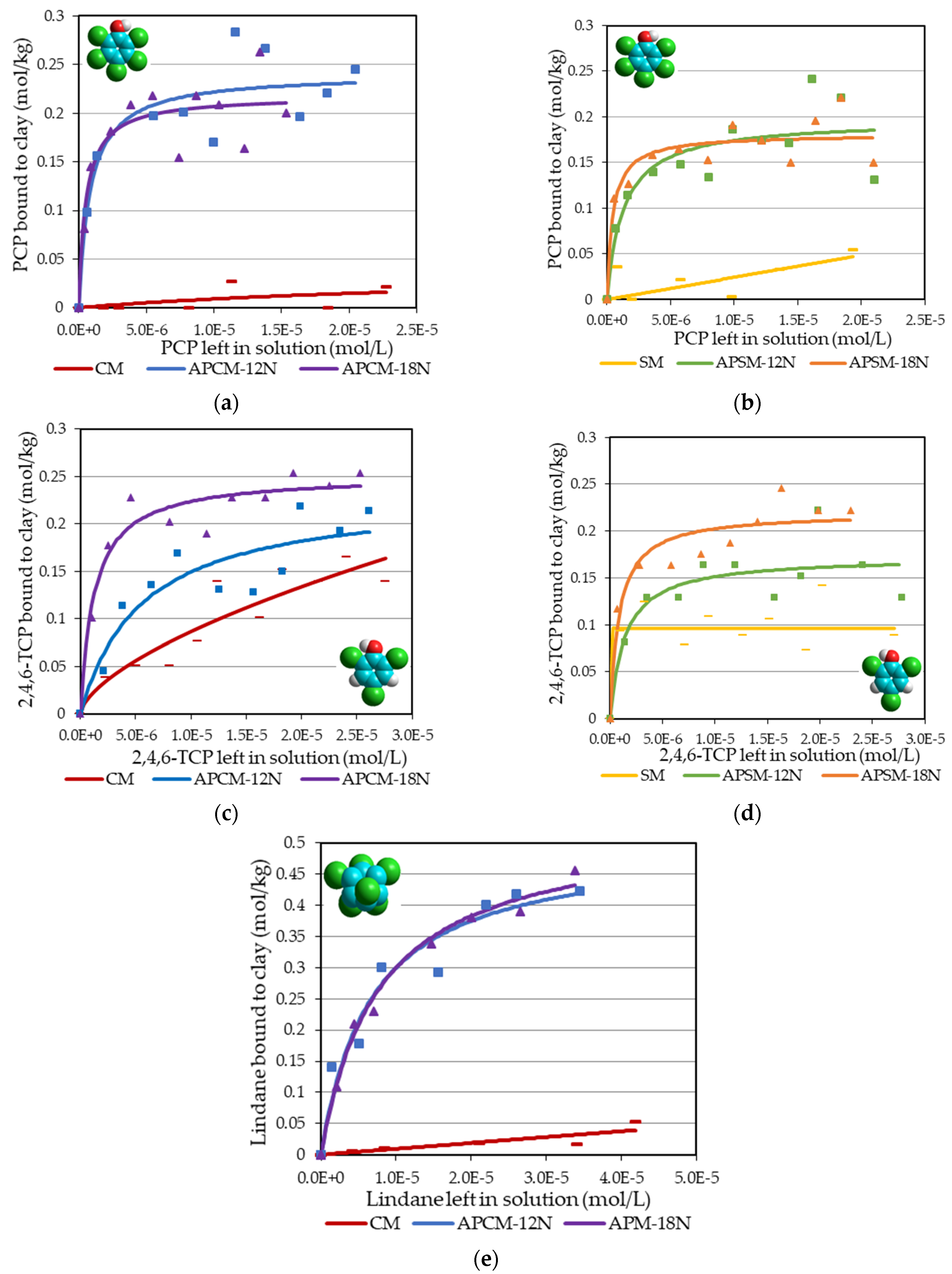
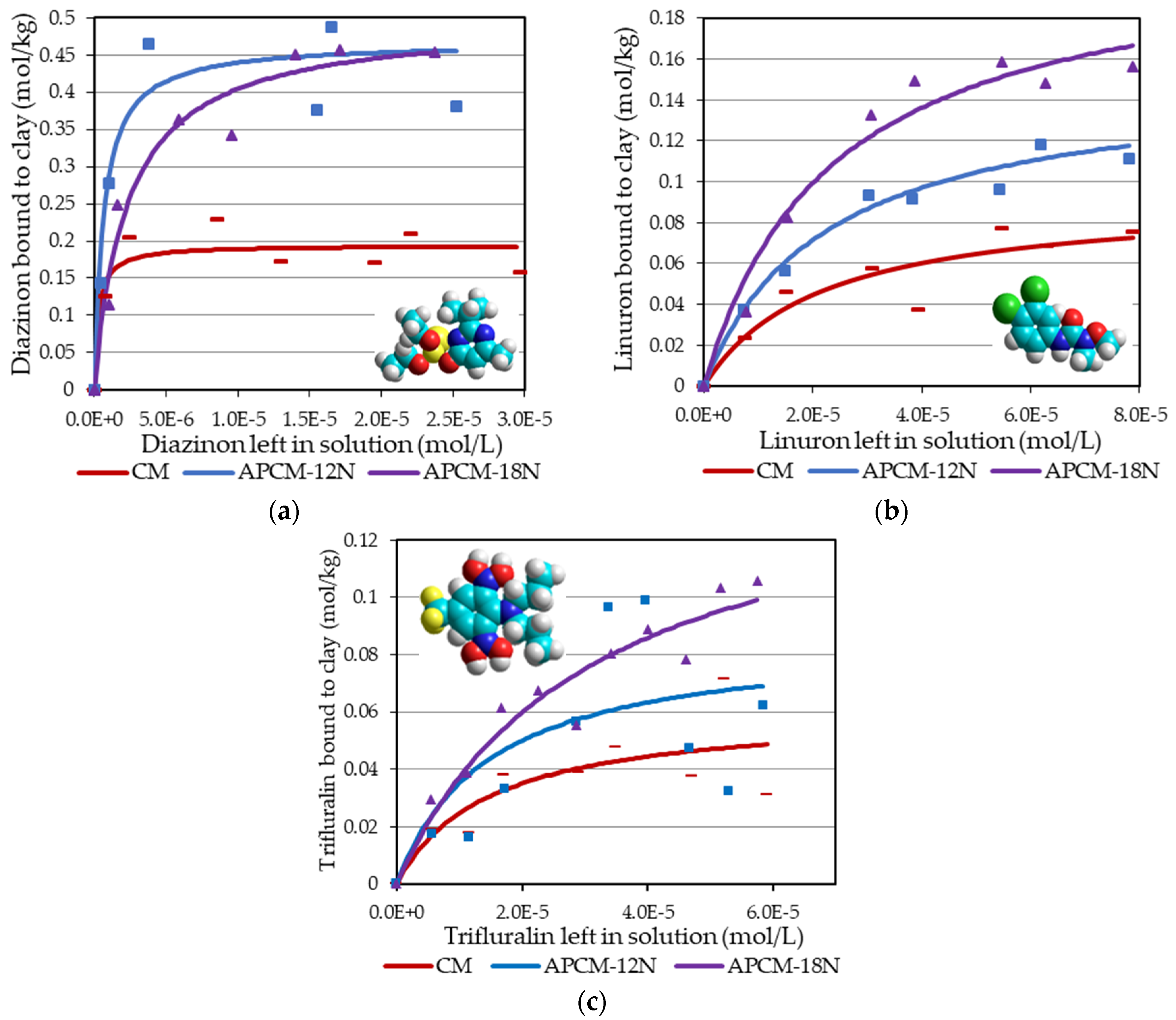

| Sorbents | PCP | 2,4,6-TCP | Lindane | Diazinon | Linuron | Trifluralin | Paraquat |
|---|---|---|---|---|---|---|---|
| CM | Kd = 2.7 × 104 | Kd = 4 × 102 | Kd = 2.8 × 102 | Qmax = 0.19 Kd = 3.6 × 106 | Qmax = 0.09 Kd = 4.7 × 104 | Qmax = 0.06 Kd = 6.9 × 104 | Qmax = 0.29 Kd = 2.8 × 106 |
| SM | Kd = 5.1 × 102 | Kd = 5 × 1020 | - | - | - | Qmax = 0.44 Kd = 3.4 × 106 | |
| APCM-12N | Qmax = 0.24 Kd = 1.2 × 106 | Qmax = 0.23 Kd = 1.8 × 105 | Qmax = 0.5 Kd = 1.5 × 105 | Qmax = 0.47 ** Kd = 1.6 × 106 | Qmax = 0.15 * Kd = 4.5 × 104 | Qmax = 0.09 Kd = 6.9 × 104 | Qmax = 0.21 Kd = 2.4 × 106 |
| APSM-12N | Qmax = 0.2 Kd = 7.8 × 105 | Qmax = 0.17 Kd = 7.1 × 105 | - | - | - | Qmax = 0.3 * Kd = 4.1 × 106 | |
| APCM-18N | Qmax = 0.22 Kd = 1.9 × 106 | Qmax = 0.25 Kd = 8.1 × 105 | Qmax = 0.53 Kd = 1.3 × 105 | Qmax = 0.5 ** Kd = 4.3 × 105 | Qmax = 0.22 ** Kd = 4.2 × 104 | Qmax = 0.15 * Kd = 3.2 × 104 | Qmax = 0.24 Kd = 5.1 × 106 |
| APSM-18N | Qmax = 0.18 Kd = 2.2 × 106 | Qmax = 0.22 Kd = 1.2 × 106 | - | - | - | Qmax = 0.37 Kd = 3.9 × 105 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, M.; Phillips, T.D. Potential Applications of Clay-Based Therapy for the Reduction of Pesticide Exposures in Humans and Animals. Appl. Sci. 2019, 9, 5325. https://doi.org/10.3390/app9245325
Wang M, Phillips TD. Potential Applications of Clay-Based Therapy for the Reduction of Pesticide Exposures in Humans and Animals. Applied Sciences. 2019; 9(24):5325. https://doi.org/10.3390/app9245325
Chicago/Turabian StyleWang, Meichen, and Timothy D. Phillips. 2019. "Potential Applications of Clay-Based Therapy for the Reduction of Pesticide Exposures in Humans and Animals" Applied Sciences 9, no. 24: 5325. https://doi.org/10.3390/app9245325
APA StyleWang, M., & Phillips, T. D. (2019). Potential Applications of Clay-Based Therapy for the Reduction of Pesticide Exposures in Humans and Animals. Applied Sciences, 9(24), 5325. https://doi.org/10.3390/app9245325





