Effect of Tinospora cordifolia-Derived Phytocomponents on Cancer: A Systematic Review
Abstract
1. Introduction
2. Methods
2.1. Inclusion Criteria
2.2. Exclusion Criteria
2.3. Information Sources and Search Strategies
2.4. Study Selection
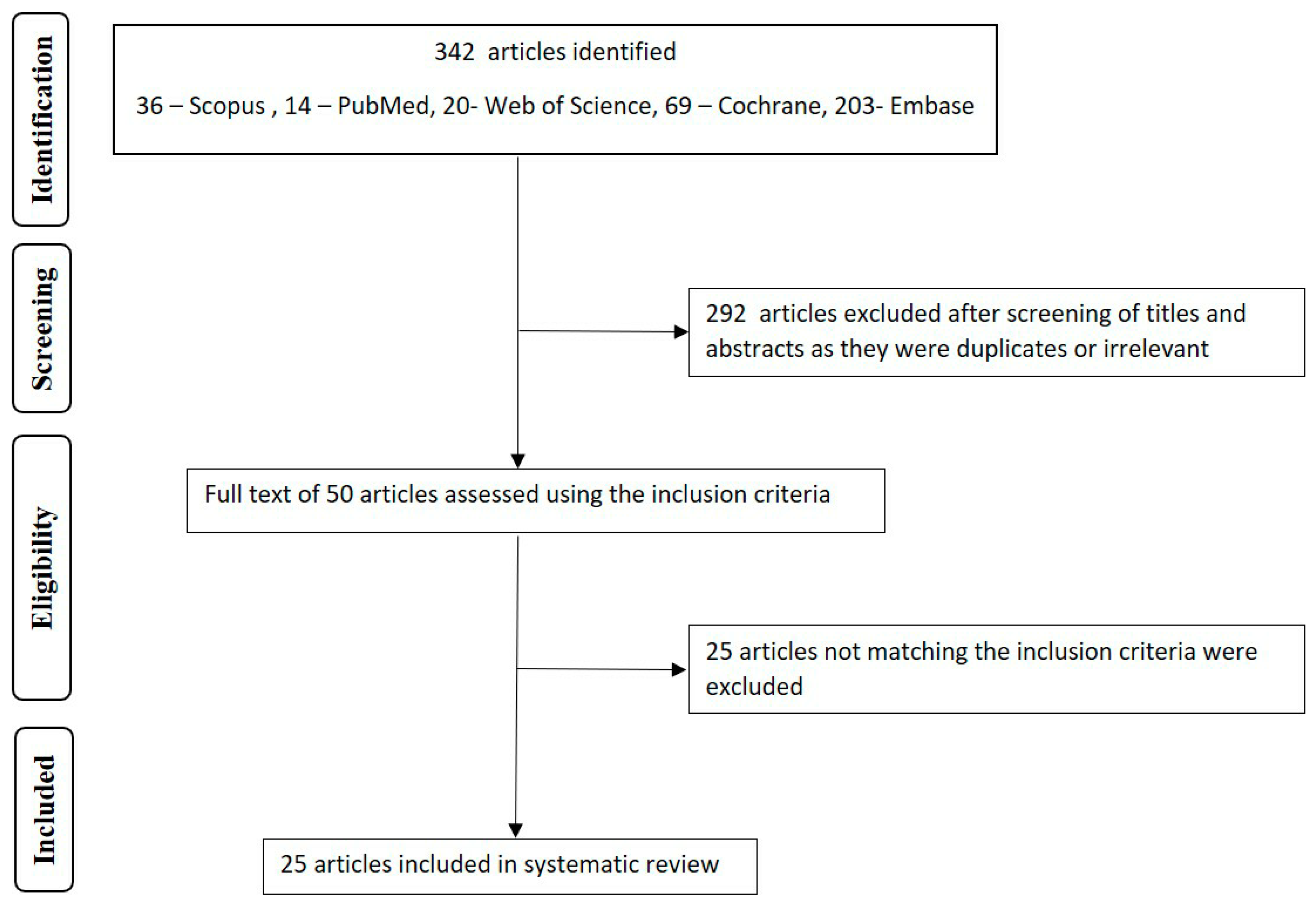
3. Results
4. Discussion
4.1. Parts of the Tc with Anti-Carcinogenic Effect
4.2. Tc-Based Extracts
4.3. Dose Effect of Phytocomponents
4.4. Phytocomponents and Its Mechanism of Action against Cancer Cells
4.5. Anti-Carcinogenic Effect of Tc Phytocomponents
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, L.A.; Jemal, A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2018, 68, 394–424. [Google Scholar] [CrossRef] [PubMed]
- Anastyuk, S.D.; Shevchenko, N.M.; Ermakova, S.P.; Vishchuk, O.S.; Nazarenko, E.L.; Dmitrenok, P.S.; Zvyagintseva, T.N. Anticancer activity in vitro of a fucoidan from the brown alga Fucusevanescens and its low-molecular fragments, structurally characterized by tandem mass-spectrometry. Carbohydr. Polym. 2012, 87, 186–194. [Google Scholar] [CrossRef]
- Saha, S.; Ghosh, S. Tinosporacordifolia: One plant, many roles. Anc. Sci. Life 2012, 31, 151–159. [Google Scholar]
- Tiwari, P.; Nayak, P.; Prusty, S.K.; Sahu, P. Phytochemistry and pharmacology of Tinosporacordifolia: A Review. Syst. Rev. Pharm. 2018, 9, 70–78. [Google Scholar] [CrossRef]
- Palmieri, A.; Iapichino, A.; Cura, F.; Scapoli, L.; Carinci, F.; Mandrone, M.; Martinelli, M. Pre-treatment with berberine enhances effect of 5-fluorouracil and cisplatin in HEP2 laryngeal cancer cell line. J. Biol. Regul. Homeost. Agents 2018, 32, 167–177. [Google Scholar]
- Sharma, N.; Kumar, A.; Sharma, P.R.; Qayum, A.; Singh, S.K.; Dutt, P.; Paul, S.; Gupta, V.; Verma, M.K.; Satti, N.K.; et al. A new clerodane furano diterpene glycoside from Tinosporacordifolia triggers autophagy and apoptosis in HCT-116 colon cancer cells. J. Ethnopharmacol. 2018, 211, 295–310. [Google Scholar] [CrossRef]
- Priya, M.S.; Venkateswaran, K.V.; Vijayanand, T. Determination of apoptosis by flow cytometric analysis in MCF-7 cells treated with TinosporaCordifolia. Ind. Vet. J. 2017, 94, 73–75. [Google Scholar]
- Singh, B.; Sharma, P.; Kumar, A.; Chadha, P.; Kaur, R.; Kaur, A. Antioxidant and in vivo genoprotective effects of phenolic compounds identified from an endophytic Cladosporium velox and their relationship with its host plant Tinosporacordifolia. J. Ethnopharmacol. 2016, 194, 450–456. [Google Scholar] [CrossRef]
- Bala, M.; Pratap, K.; Verma, P.K.; Singh, B.; Padwad, Y. Validation of ethnomedicinal potential of Tinosporacordifolia for anticancer and immunomodulatory activities and quantification of bioactive molecules by HPTLC. J. Ethnopharmacol. 2015, 175, 131–137. [Google Scholar] [CrossRef]
- Shaikh, R.; Pund, M.; Dawane, A.; Iliyas, S. Evaluation of anticancer, antioxidant, and possible anti-inflammatory properties of selected medicinal plants used in indian traditional medication. J. Tradit. Complement. Med. 2014, 4, 253–257. [Google Scholar] [CrossRef]
- Maliyakkal, N.; Udupa, N.; Pai, K.S.R.; Rangarajan, A. Cytotoxic and apoptotic activities of extracts of withaniasomnifera and tinosporacordifolia in human breast cancer cells. Int. J. Appl. Res. Nat. Prod. 2013, 6, 1–10. [Google Scholar]
- Mishra, A.; Kumar, S.; Pandey, A.K. Scientific validation of the medicinal efficacy of tinosporacordifolia. Sci. World J. 2013, 2013, 1–8. [Google Scholar]
- Rashmi, K.C.; Raj, M.H.; Paul, M.; Girish, K.S.; Salimath, B.P.; Aparna, H.S. A new pyrrole based small molecule from Tinosporacordifolia induces apoptosis in MDA-MB-231 breast cancer cells via ROS mediated mitochondrial damage and restoration of p53 activity. Chem. Biol. Interact. 2019, 299, 120–130. [Google Scholar] [CrossRef] [PubMed]
- Mishra, R.; Kaur, G. Tinosporacordifolia Induces Differentiation and Senescence Pathways in Neuroblastoma cells. Mol. Neurobiol. 2015, 52, 719–733. [Google Scholar] [CrossRef] [PubMed]
- Ansari, J.A.; Rastogi, N.; Ahmad, M.K.; Mahdi, A.A.; Khan, A.R.; Thakur, R.; Srivastava, V.K.; Mishra, D.P.; Fatima, N.; Khan, H.J.; et al. ROS mediated pro-apoptotic effects of Tinosporacordifolia on breast cancer cells. Front. Biosci. 2017, 9, 89–100. [Google Scholar]
- Mishra, R.; Kaur, G. Aqueous ethanolic extract of Tinosporacordifolia as a potential candidate for differentiation based therapy of glioblastomas. PLoS ONE 2013, 8, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Bansal, P.; Malik, M.A.; Das, S.N.; Kaur, J. TinosporaCordifolia Induces Cell Cycle Arrest in Human Oral Squamous Cell Carcinoma Cells. Gulf J. Oncol. 2017, 1, 10–14. [Google Scholar]
- Mohan, V.; Koul, A. Anticancer potential of Tinosporacordifolia and arabinogalactan against benzo(a) pyrene induced pulmonary tumorigenesis: A study in relevance to various biomarkers. J. Herbmed. Pharmacol. 2018, 7, 225–235. [Google Scholar] [CrossRef]
- Bakrania, A.K.; Nakka, S.; Variya, B.C.; Shah, P.V.; Patel, S.S. Antitumor potential of herbomineral formulation against breast cancer: Involvement of inflammation and oxidative stress. Indian J. Exp. Biol. 2017, 55, 680–687. [Google Scholar]
- Ali, H.; Dixit, S. Extraction optimization of Tinosporacordifolia and assessment of the anticancer activity of its alkaloid palmatine. Sci. World J. 2013, 2013, 1–10. [Google Scholar] [CrossRef]
- Jagetia, G.C.; Rao, S.K. Evaluation of the antineoplastic activity of guduchi (Tinosporacordifolia) in Ehrlich ascites carcinoma bearing mice. Biol. Pharm. Bull. 2006, 29, 460–466. [Google Scholar] [CrossRef] [PubMed]
- Jagetia, G.C.; Reddy, TK.; Malagi, K.J.; Nayak, B.S.; Naidu, M.B.; Ravikiran, P.B.; Kamath, S.U.; Shetty, P.C.; Reddy, D.S. Antarth, a polyherbal preparation protects against the doxorubicin-induced toxicity without compromising its Antineoplastic activity. Phytother. Res. 2005, 19, 772–778. [Google Scholar]
- Mathew, S.; Kuttan, G. Immunomodulatory and antitumour activities of Tinosporacordifolia. Fitoterapia 1999, 70, 35–43. [Google Scholar] [CrossRef]
- Thippeswamy, G.; Salimath, B.P. Induction of caspase-3 activated DNase mediated apoptosis by hexane fraction of Tinosporacordifolia in EAT cells. Environ. Toxicol. Pharmacol. 2007, 23, 212–220. [Google Scholar] [CrossRef]
- Leyon, P.V.; Kuttan, G. Inhibitory effect of a polysaccharide from Tinosporacordifolia on experimental metastasis. J. Ethnopharmacol. 2004, 90, 233–237. [Google Scholar] [CrossRef]
- Dhanasekaran, M.; Baskar, A.A.; Ignacimuthu, S.; Agastian, P.; Duraipandiyan, V. Chemopreventive potential of Epoxy clerodane diterpene from Tinosporacordifolia against diethylnitrosamine-induced hepatocellular carcinoma. Investig. New Drugs 2009, 27, 347–355. [Google Scholar] [CrossRef]
- Adhvaryu, M.R.; Reddy, N.; Parabia, M.H. Anti-tumor activity of four Ayurvedic herbs in Dalton lymphoma ascites bearing mice and their short-term in vitro cytotoxicity on DLA-cell-line. Afr. J. Tradit. Complement. Altern. Med. 2008, 5, 409–418. [Google Scholar] [CrossRef]
- Singh, N.; Singh, S.M.; Shrivastava, P. Effect of Tinosporacordifolia on the antitumor activity of tumor-associated macrophages-derived dendritic cells. Immunopharmacol. Immunotoxicol. 2005, 27, 1–14. [Google Scholar]
- Ahamad, R.; Shrivastava, A.N.; Khan, M.A. Evaluation of in vitro anticancer activity of stem of Tinosporacordiflia against human breast cancer Vero cell lines. J. Med. Plants Stud. 2015, 3, 33–37. [Google Scholar]
| S. No. | Phytocomponents | Part in Use | Cancer Cell Lines | Anticancer Drugs Used with Tc | Effect on the Cancer Cells |
|---|---|---|---|---|---|
| 1 | Berberine and isoquinolone [5] | Information not provided | HEP2 human laryngeal cancer cell line (in vitro) | 5-fluorouracil and cisplatin | Decreased gene expression of cell cycle, differentiation, and epithelial–mesenchymal transition |
| 2 | A new clerodane furano diterpene glycoside [6] | Fresh stems aqueous alcoholic extract | (in vitro) Human lung carcinoma cell line (A549), Prostate (PC-3), SF-269 (CNS), MDA-MB-435 (Melanoma), HCT-116 (Colon) and Breast (MCF-7) | Paclitaxel | Induction of mitochondria-mediated apoptosis and autophagy in HCT-116 cells |
| 5 | Information was not provided [7] | stem part aqueous and hydroalcohol extracts | Human breast carcinoma cell line MCF-7 | Doxorubicin | Induction of apoptosis |
| 6 | Phenol (ellagic acid and kaempferol) [8] | Leaf and stem Phenolic extract | CHO (Chinese Hamster Ovary) cell line | Doxorubicin | Mild cytotoxic effect noted at a high concentration of the extract |
| 7 | N-formylannonain, magnoflorine, jatrorrhizine palmatine, 11-hydroxymustakone, cordifolioside A, tinocordiside and yangambin [9] | Stem Ethanol extract | Human cancer cell lines, KB (human oral squamous carcinoma), CHOK-1 (hamster ovary), HT-29 (human colon cancer) and SiHa (human cervical cancer) and murine primary cells | Doxorubicin | Cytotoxicity of cells |
| 8 | Ethanol phytofraction [10] | Powdered plant samples hexane, ethanol, and water extract | Human cancer cell lines HeLa-B75, HL-60, HEP-3B, PN-15, and normal liver cell lines | Suramin | Cytotoxicity of cells and induction of apoptosis |
| 9 | Phytochemicals of Ethanolic extract [11] | Whole plant and stem ethanolic extracts aqueous extracts | on human breast cancer cells (MCF7 and MDA MB 231) | Doxorubicin | Cytotoxicity of cells, induction of apoptosis, cell cycle arrest in the G2/M phase |
| 11 | Anthraquinones, terpenoids, and saponins and phenol [12] | Stem solvents like petroleum, ether, chloroform, ethyl acetate, acetone, and water extract | prostrate (DU-145), ovary (IGROV- 1), and breast (MCF-7) cell lines | mitomycin-C (DU-145), paclitaxel against breast (MCF-7), and adriamycin (against ovary (IGR-OV-1) | Cell growth inhibition |
| 15 | Pyrrole-based small molecule [13] | Leaves ethyl acetate and aqueous extract | MDA-MB-231 breast cancer cells | Doxorubicin | Induction of apoptosis |
| 17 | Information was not provided [14] | Stem alcohol extract | human IMR-32 cell line | None | Upregulation of senescence and apoptosis |
| 18 | Phenolics contents quercetin and rutin [15] | Stems methanol extract | human breast cancer MDA-MB-231 cells | None | Anti-proliferative activity |
| 19 | Information was not provided [16] | ethanolic extract | Rat C6 glioma, U87MG human glioma, PC3 prostate cancer cell line, and HeLa cell line | None | Anti-proliferative, anti-migratory/anti-metastatic potential activity, and induction of apoptosis. |
| 20 | Ready product from standard ayurvedic pharmacy [17] | Information was not provided | KB cancer cell lines | Methotrexate | Cell cycle arrest at G0/G1 phase |
| S. No. | Phytocomponents | Part in Use | Animals | Outcome |
|---|---|---|---|---|
| 3 | Arabinogalactan a polysaccharide [18] | Stem Aqueous extract | Male BALB/c mice (25–30 g) benzo(a) pyrene-induced pulmonary tumor | Reduced tumor incidence and multiplicity, induction of apoptosis |
| 4 | Phenolic component [19] | Plant semiautomated capsule | DABA-induced mammary carcinogenesis in female Sprague-Dawley rats (breast cancer) | Tumor inhibition |
| 6 | Phenol (ellagic acid and kaempferol) [8] | Stem phenolic extract | Freshwater air-breathing fish Channa punctatus with DNA damage induction by nonionic surfactant nonylphenol | Non-cytotoxic, non-mutagenic, significant antioxidant activity, genoprotective effect |
| 10 | Alkaloid palmatine [20] | Stems methanol and aqueous extracts | Swiss albino mice injected with DMBA | Tumor inhibition |
| 12 | alkaloids including berberine [21] | stems dichloromethane alcoholic Extract | Swiss albino mice injected with Ehrlich ascites carcinoma (EAC) | Cytotoxicity of the cells |
| 13 | Antarth [22] | Plants aqueous extract | male Swiss albino mice injected with Ehrlich ascites carcinoma (EAC). | Reduces the cardiotoxicity associated with doxorubicin, but independently has no anti-carcinogenic effect |
| 14 | Triterpenoids and alkaloids [23] | stem methanolic extract | BALB RC and Swiss albino mice injected with Ehrlich ascites tumor cells | Tumor inhibition |
| 15 | Pyrrole-based small molecule [13] | Leaves methanolic extract | female Swiss albino mice injected with Ehrlich ascites tumor cells | Reduced tumor burden and two-fold increase in survival |
| 16 | Hexane fraction [24] | Stems solvents like hexane, benzene and chloroform | Swiss albino female mice injected intraperitoneally with Ehrlich ascites tumor (EAT) cells | Cell growth inhibition and induction of apoptosis |
| 20 | Polysaccharide [25] | Stem methanolic extract | C57BL/6 MICE injected by B16F-10 melanoma cell lines | Tumor inhibition |
| 21 | Clerodane-derived diterpenoids [26] | Stems alcoholic extraction | Male Wistar albino strain rats with diethylnitrosamine-induced hepatocellular carcinoma | Inhibiting tumor growth by blocking carcinogen metabolic activation and enhancing carcinogen detoxification. |
| 22 | Crude powder [27] | hydroethanolic (1:1) extract | Dalton lymphoma ascites (DLA) tumor model in Swiss albino mice | Reduced clonogenicity |
| 23 | Information was not provided [28] | Plant alcoholic extract | Inbred BALB/c mice tumor-associated macrophage (TAM)-derived dendritic cell to Dalton’s lymphoma-bearing mice | Enhances the differentiation of dendritic cells |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Deepa, B.; Babaji, H.V.; Hosmani, J.V.; Alamir, A.W.H.; Mushtaq, S.; Raj, A.T.; Patil, S. Effect of Tinospora cordifolia-Derived Phytocomponents on Cancer: A Systematic Review. Appl. Sci. 2019, 9, 5147. https://doi.org/10.3390/app9235147
Deepa B, Babaji HV, Hosmani JV, Alamir AWH, Mushtaq S, Raj AT, Patil S. Effect of Tinospora cordifolia-Derived Phytocomponents on Cancer: A Systematic Review. Applied Sciences. 2019; 9(23):5147. https://doi.org/10.3390/app9235147
Chicago/Turabian StyleDeepa, Babji, Harsha V. Babaji, Jagadish V. Hosmani, Abdul Wahab H. Alamir, Shazia Mushtaq, A. Thirumal Raj, and Shankargouda Patil. 2019. "Effect of Tinospora cordifolia-Derived Phytocomponents on Cancer: A Systematic Review" Applied Sciences 9, no. 23: 5147. https://doi.org/10.3390/app9235147
APA StyleDeepa, B., Babaji, H. V., Hosmani, J. V., Alamir, A. W. H., Mushtaq, S., Raj, A. T., & Patil, S. (2019). Effect of Tinospora cordifolia-Derived Phytocomponents on Cancer: A Systematic Review. Applied Sciences, 9(23), 5147. https://doi.org/10.3390/app9235147