Ni3Se4@MoSe2 Composites for Hydrogen Evolution Reaction
Abstract
Featured Application
Abstract
1. Introduction
2. Experimental Section
2.1. Materials
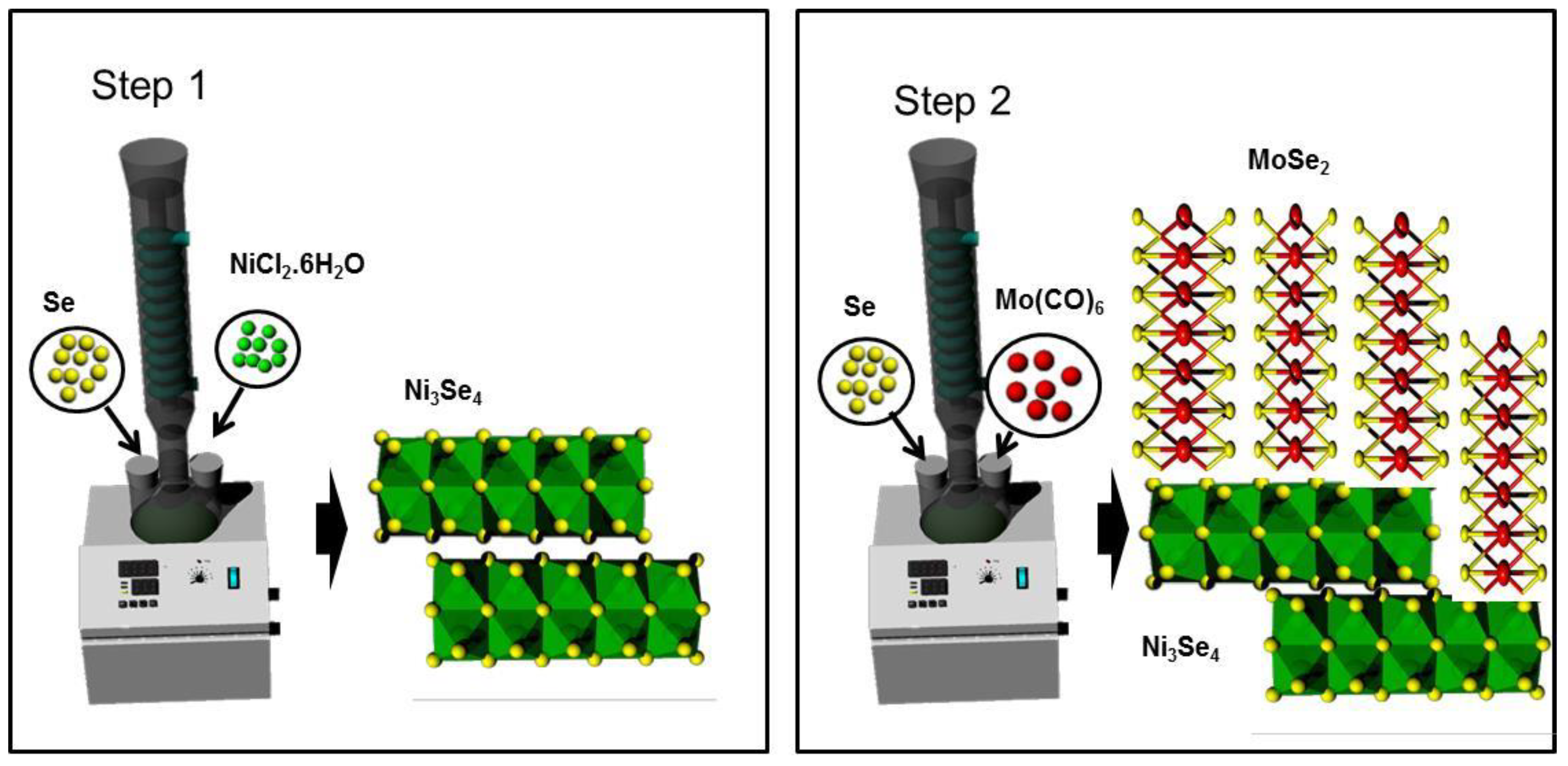
2.2. Synthesis of Ni3Se4 Nanosheets
2.3. Synthesis of Ni3Se4–MoSe2 Composites
2.4. Characterization
2.5. Electrochemical
3. Results and Discussion
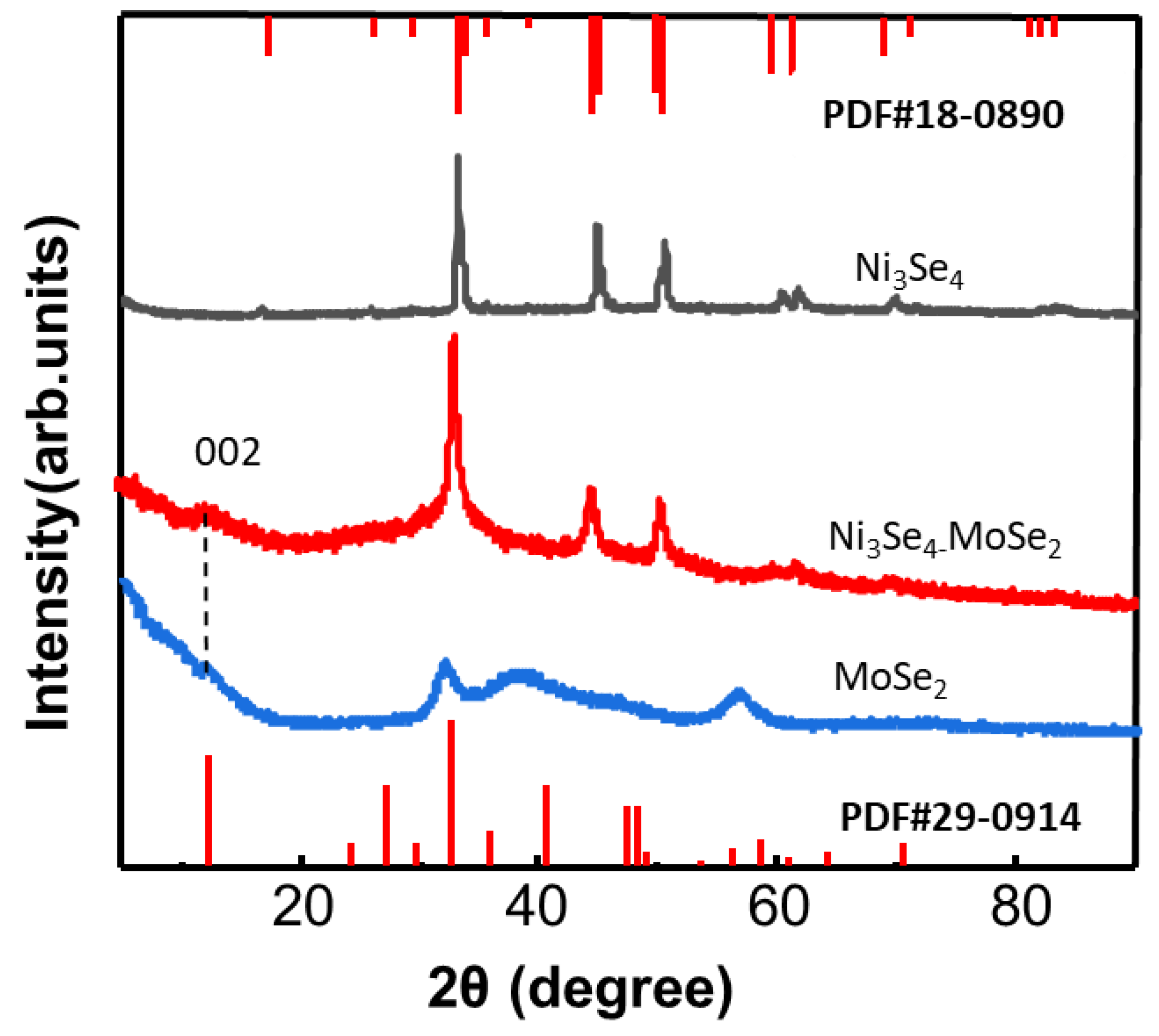
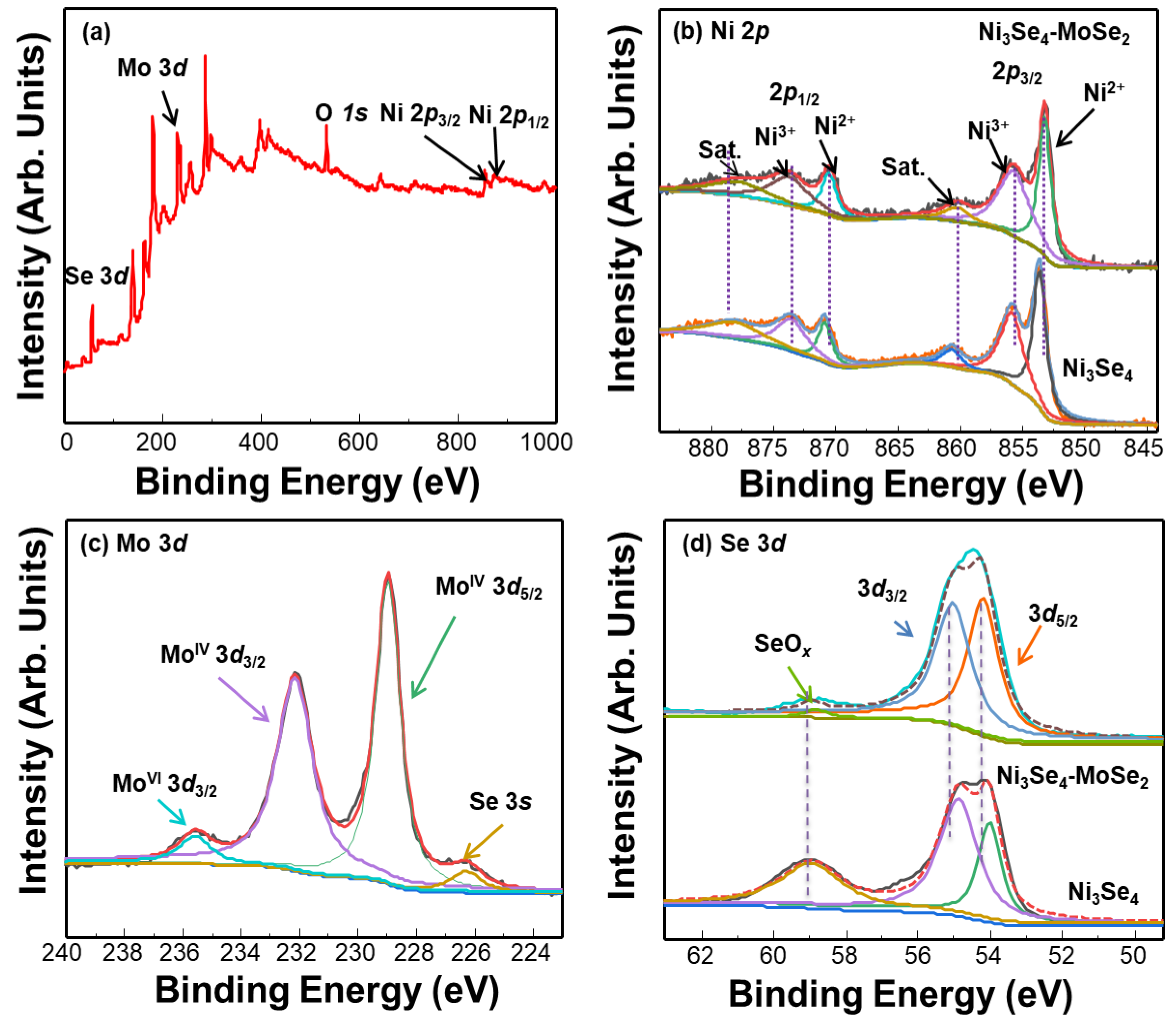
3.1. Synthesis and Structural Characterization
3.2. Electrocatalytic Properties
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Dunn, S. Hydrogen futures: Toward a sustainable energy system. Int. J. Hydrog. Energy 2002, 27, 235–264. [Google Scholar] [CrossRef]
- Momirlan, M.; Veziroglu, T. Current status of hydrogen energy. Renew. Sustain. Energy Rev. 2002, 6, 141–179. [Google Scholar] [CrossRef]
- Dresselhaus, M.; Thomas, I. Alternative energy technologies. Nature 2001, 414, 332. [Google Scholar] [CrossRef] [PubMed]
- Walter, M.G.; Warren, E.L.; McKone, J.R.; Boettcher, S.W.; Mi, Q.; Santori, E.A.; Lewis, N.S. Solar water splitting cells. Chem. Rev. 2010, 110, 6446–6473. [Google Scholar] [CrossRef]
- Xie, J.; Zhang, H.; Li, S.; Wang, R.; Sun, X.; Zhou, M.; Zhou, J.; Lou, X.W.; Xie, Y. Defect-rich MoS2 ultrathin nanosheets with additional active edge sites for enhanced electrocatalytic hydrogen evolution. Adv. Mater. 2013, 25, 5807–5813. [Google Scholar] [CrossRef] [PubMed]
- Cortright, R.D.; Davda, R.; Dumesic, J.A. Hydrogen from catalytic reforming of biomass-derived hydrocarbons in liquid water. In Materials For Sustainable Energy: A Collection of Peer-Reviewed Research and Review Articles from Nature Publishing Group; World Scientific: Singapore, 2011; pp. 289–292. [Google Scholar]
- Ball, M.; Wietschel, M. The future of hydrogen–opportunities and challenges. Int. J. Hydrog. Energy 2009, 34, 615–627. [Google Scholar] [CrossRef]
- Deng, J.; Ren, P.; Deng, D.; Yu, L.; Yang, F.; Bao, X. Highly active and durable non-precious-metal catalysts encapsulated in carbon nanotubes for hydrogen evolution reaction. Energy Environ. Sci. 2014, 7, 1919–1923. [Google Scholar] [CrossRef]
- Bard, A.J.; Fox, M.A. Artificial photosynthesis: Solar splitting of water to hydrogen and oxygen. Chem. Res. 1995, 28, 141–145. [Google Scholar] [CrossRef]
- Turner, J.A. Sustainable hydrogen production. Science 2004, 305, 972–974. [Google Scholar] [CrossRef]
- Li, Y.; Wang, H.; Xie, L.; Liang, Y.; Hong, G.; Dai, H. MoS2 nanoparticles grown on graphene: An advanced catalyst for the hydrogen evolution reaction. J. Am. Chem. Soc. 2011, 133, 7296–7299. [Google Scholar] [CrossRef]
- Conway, B.; Tilak, B. Interfacial processes involving electrocatalytic evolution and oxidation of H2, and the role of chemisorbed H. Electrochim. Acta 2002, 47, 3571–3594. [Google Scholar] [CrossRef]
- Kemppainen, E.; Bodin, A.; Sebok, B.; Pedersen, T.; Seger, B.; Mei, B.; Bae, D.; Vesborg, P.C.K.; Halme, J.; Hansen, O. Scalability and feasibility of photoelectrochemical H2 evolution: The ultimate limit of Pt nanoparticle as an HER catalyst. Energy Environ. Sci. 2015, 8, 2991–2999. [Google Scholar] [CrossRef]
- Elezović, N.R.; Gajić-Krstajić, L.; Radmilović, V.; Vračar, L.; Krstajić, N.V. Effect of chemisorbed carbon monoxide on Pt/C electrode on the mechanism of the hydrogen oxidation reaction. Electrochim. Acta 2009, 54, 1375–1382. [Google Scholar] [CrossRef]
- Chen, C.; Kang, Y.; Huo, Z.; Zhu, Z.; Huang, W.; Xin, H.L.; Snyder, J.D.; Li, D.; Herron, J.A.; Mavrikakis, M. Highly crystalline multimetallic nanoframes with three-dimensional electrocatalytic surfaces. Science 2014, 343, 1339–1343. [Google Scholar] [CrossRef] [PubMed]
- Ring, L.; Pollet, B.G.; Chatenet, M.; Abbou, S.; Küpper, K.; Schmidt, M.; Huck, M.; Gries, A.; Steinhart, M.; Schäfer, H. From bad electrochemical practices to an environmental and waste reducing approach for the generation of active hydrogen evolving electrodes. Angew. Chem. 2019, 131, 2–12. [Google Scholar] [CrossRef]
- Zou, X.; Zhang, Y. Noble metal-free hydrogen evolution catalysts for water splitting. Chem. Soc. Rev. 2015, 44, 5148–5180. [Google Scholar] [CrossRef]
- Liu, J.; Liu, Y.; Liu, N.; Han, Y.; Zhang, X.; Huang, H.; Lifshitz, Y.; Lee, S.-T.; Zhong, J.; Kang, Z. Metal-free efficient photocatalyst for stable visible water splitting via a two-electron pathway. Science 2015, 347, 970–974. [Google Scholar] [CrossRef]
- Guo, S.; Zhang, S.; Wu, L.; Sun, S. Co/CoO nanoparticles assembled on graphene for electrochemical reduction of oxygen. Angew. Chem. 2012, 124, 11940–11943. [Google Scholar] [CrossRef]
- Cao, R.; Lee, J.S.; Liu, M.; Cho, J. Recent progress in non-precious catalysts for metal-air batteries. Adv. Energy Mater. 2012, 2, 816–829. [Google Scholar] [CrossRef]
- Wang, H.; Lu, Z.; Xu, S.; Kong, D.; Cha, J.J.; Zheng, G.; Hsu, P.-C.; Yan, K.; Bradshaw, D.; Prinz, F.B. Electrochemical tuning of vertically aligned MoS2 nanofilms and its application in improving hydrogen evolution reaction. Proc. Natl. Acad. Sci. USA 2013, 110, 19701–19706. [Google Scholar] [CrossRef]
- Tan, C.; Zhang, H. Two-dimensional transition metal dichalcogenide nanosheet-based composites. Chem. Soc. Rev. 2015, 44, 2713–2731. [Google Scholar] [CrossRef] [PubMed]
- Chhowalla, M.; Shin, H.S.; Eda, G.; Li, L.-J.; Loh, K.P.; Zhang, H. The chemistry of two-dimensional layered transition metal dichalcogenide nanosheets. Nat. Chem. 2013, 5, 263. [Google Scholar] [CrossRef] [PubMed]
- Nicolosi, V.; Chhowalla, M.; Kanatzidis, M.G.; Strano, M.S.; Coleman, J.N. Liquid exfoliation of layered materials. Science 2013, 340, 1226419. [Google Scholar] [CrossRef]
- Kibsgaard, J.; Chen, Z.; Reinecke, B.N.; Jaramillo, T.F. Engineering the surface structure of MoS2 to preferentially expose active edge sites for electrocatalysis. Nat. Mater. 2012, 11, 963. [Google Scholar] [CrossRef]
- Choi, W.I.; Wood, B.C.; Schwegler, E.; Ogitsu, T. Site-dependent free energy barrier for proton reduction on MoS2 edges. J. Phys. Chem. C 2013, 117, 21772–21777. [Google Scholar] [CrossRef]
- Jaramillo, T.F.; Jørgensen, K.P.; Bonde, J.; Nielsen, J.H.; Horch, S.; Chorkendorff, I. Identification of active edge sites for electrochemical H2 evolution from MoS2 nanocatalysts. Science 2007, 317, 100–102. [Google Scholar] [CrossRef]
- Lauritsen, J.; Nyberg, M.; Nørskov, J.K.; Clausen, B.; Topsøe, H.; Lægsgaard, E.; Besenbacher, F. Hydrodesulfurization reaction pathways on MoS2 nanoclusters revealed by scanning tunneling microscopy. J. Catal. 2004, 224, 94–106. [Google Scholar] [CrossRef]
- Guo, W.; Le, Q.; Hasani, A.; Lee, T.; Jang, H.; Luo, Z.; Kim, S. MoSe2-GO/rGO Composite Catalyst for Hydrogen Evolution Reaction. Polymers 2018, 10, 1309. [Google Scholar] [CrossRef]
- Tang, H.; Dou, K.; Kaun, C.-C.; Kuang, Q.; Yang, S. MoSe2 nanosheets and their graphene hybrids: Synthesis, characterization and hydrogen evolution reaction studies. J. Mater. Chem. 2014, 2, 360–364. [Google Scholar] [CrossRef]
- Qu, B.; Li, C.; Zhu, C.; Wang, S.; Zhang, X.; Chen, Y. Growth of MoSe2 nanosheets with small size and expanded spaces of (002) plane on the surfaces of porous N-doped carbon nanotubes for hydrogen production. Nanoscale 2016, 8, 16886–16893. [Google Scholar] [CrossRef]
- Gao, M.-R.; Lin, Z.-Y.; Zhuang, T.-T.; Jiang, J.; Xu, Y.-F.; Zheng, Y.-R.; Yu, S.-H. Mixed-solution synthesis of sea urchin-like NiSe nanofiber assemblies as economical Pt-free catalysts for electrochemical H2 production. J. Mater. Chem. 2012, 22, 13662–13668. [Google Scholar] [CrossRef]
- Tang, C.; Cheng, N.; Pu, Z.; Xing, W.; Sun, X. NiSe nanowire film supported on nickel foam: An efficient and stable 3D bifunctional electrode for full water splitting. Angew. Chem. Int. Ed. 2015, 54, 9351–9355. [Google Scholar] [CrossRef] [PubMed]
- Ji, L.; Lv, C.; Chen, Z.; Huang, Z.; Zhang, C. Nickel-Based (Photo) Electrocatalysts for Hydrogen Production. Adv. Mater. 2018, 30, 1705653. [Google Scholar] [CrossRef] [PubMed]
- Zhang, T.; Liu, X.; Cui, X.; Chen, M.; Liu, S.; Geng, B. Colloidal Synthesis of Mo–Ni Alloy Nanoparticles as Bifunctional Electrocatalysts for Efficient Overall Water Splitting. Adv. Mater. Interfaces 2018, 5, 1800359. [Google Scholar] [CrossRef]
- Tian, J.; Cheng, N.; Liu, Q.; Sun, X.; He, Y.; Asiri, A.M. Self-supported NiMo hollow nanorod array: An efficient 3D bifunctional catalytic electrode for overall water splitting. J. Mater. Chem. A 2015, 3, 20056–20059. [Google Scholar] [CrossRef]
- Zhou, X.; Liu, Y.; Ju, H.; Pan, B.; Zhu, J.; Ding, T.; Wang, C.; Yang, Q. Design and epitaxial growth of MoSe2–NiSe vertical heteronanostructures with electronic modulation for enhanced hydrogen evolution reaction. Chem. Mater. 2016, 28, 1838–1846. [Google Scholar] [CrossRef]
- Merki, D.; Vrubel, H.; Rovelli, L.; Fierro, S.; Hu, X. Fe, Co, and Ni ions promote the catalytic activity of amorphous molybdenum sulfide films for hydrogen evolution. Chem. Sci. 2012, 3, 2515–2525. [Google Scholar] [CrossRef]
- Kuraganti, V.; Jain, A.; Bar-Ziv, R.; Ramasubramaniam, A.; Bar-Sadan, M. Manganese doping of MoSe2 promotes active defect sites for hydrogen evolution. ACS Appl. Mater. Interfaces 2019, 11, 25155–25162. [Google Scholar] [CrossRef]
- Peng, H.; Zhou, J.; Sun, K.; Ma, G.; Zhang, Z.; Feng, E.; Lei, Z. High-performance asymmetric supercapacitor designed with a novel NiSe@ MoSe2 nanosheet array and nitrogen-doped carbon nanosheet. ACS Sustain. Chem. Eng. 2017, 5, 5951–5963. [Google Scholar] [CrossRef]
- Chen, H.; Wu, L.; Zhang, K.; Qin, A.; Chen, S. New Synthesis of Polycrystalline NiSe Nanoarrays on Ni Foam as the Electrode for High Performance Supercapacitors. Int. J. Electrochem. Sci. 2018, 13, 12437–12449. [Google Scholar] [CrossRef]
- Lee, C.-T.; Peng, J.-D.; Li, C.-T.; Tsai, Y.-L.; Vittal, R.; Ho, K.-C. Ni3Se4 hollow architectures as catalytic materials for the counter electrodes of dye-sensitized solar cells. Nano Energy 2014, 10, 201–211. [Google Scholar] [CrossRef]
- Zhang, J.-Y.; Tian, X.; He, T.; Zaman, S.; Miao, M.; Yan, Y.; Qi, K.; Dong, Z.; Liu, H.; Xia, B.Y. In situ formation of Ni3Se4 nanorod arrays as versatile electrocatalysts for electrochemical oxidation reactions in hybrid water electrolysis. J. Mater. Chem. A 2018, 6, 15653–15658. [Google Scholar] [CrossRef]
- Anantharaj, S.; Kennedy, J.; Kundu, S. Microwave-initiated facile formation of Ni3Se4 nanoassemblies for enhanced and stable water splitting in neutral and alkaline media. ACS Appl. Mater. Interfaces 2017, 9, 8714–8728. [Google Scholar] [CrossRef] [PubMed]
- Bhat, K.S.; Nagaraja, H. Nickel selenide nanostructures as an electrocatalyst for hydrogen evolution reaction. Int. J. Hydrog. Energy 2018, 43, 19851–19863. [Google Scholar] [CrossRef]
- Anantharaj, S.; Karthik, K.; Amarnath, T.S.; Chatterjee, S.; Subhashini, E.; Swaathini, K.C.; Karthick, P.E.; Kundu, S. Membrane free water electrolysis under 1.23 V with Ni3Se4/Ni anode in alkali and Pt cathode in acid. Appl. Surf. Sci. 2019, 478, 784–792. [Google Scholar] [CrossRef]
- Peng, H.; Wei, C.; Wang, K.; Meng, T.; Ma, G.; Lei, Z.; Gong, X. Ni0.85Se@ MoSe2 nanosheet arrays as the electrode for high-performance supercapacitors. ACS Appl. Mater. Interfaces 2017, 9, 17067–17075. [Google Scholar] [CrossRef]
- Du, J.; Zou, Z.; Liu, C.; Xu, C. Hierarchical Fe-doped Ni3Se4 ultrathin nanosheets as an efficient electrocatalyst for oxygen evolution reaction. Nanoscale 2018, 10, 5163–5170. [Google Scholar] [CrossRef]
- Zhang, L.; Sun, L.; Huang, Y.; Sun, Y.; Hu, T.; Xu, K.; Ma, F. Hydrothermal synthesis of N-doped RGO/MoSe2 composites and enhanced electro-catalytic hydrogen evolution. J. Mater. Sci. 2017, 52, 13561–13571. [Google Scholar] [CrossRef]
- Xu, C.; Peng, S.; Tan, C.; Ang, H.; Tan, H.; Zhang, H.; Yan, Q. Ultrathin S-doped MoSe2 nanosheets for efficient hydrogen evolution. J. Mater. Chem. A 2014, 2, 5597–5601. [Google Scholar] [CrossRef]
- Voiry, D.; Yang, J.; Chhowalla, M. Recent strategies for improving the catalytic activity of 2D TMD nanosheets toward the hydrogen evolution reaction. Adv. Mater. 2016, 28, 6197–6206. [Google Scholar] [CrossRef]

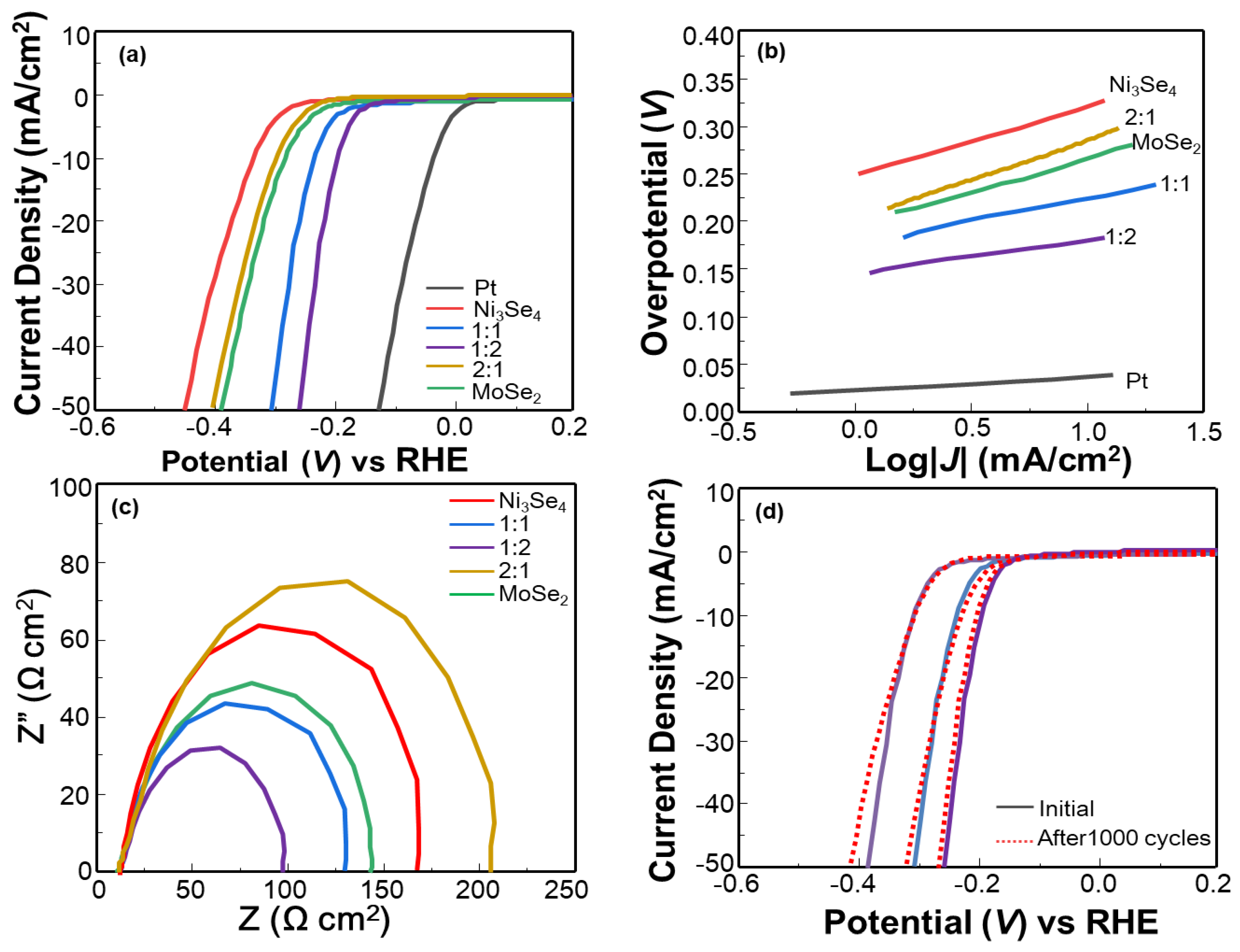
| η1 (mV) | η10 (mV) | Tafel Slope (mV/dec) | Rct (Ω cm2) | |
|---|---|---|---|---|
| Ni3Se4 | 240 | 330 | 109 | 108 |
| Ni3Se4–MoSe2 (1:2) | 145 | 206 | 65 | 68 |
| Ni3Se4–MoSe2 (1:1) | 160 | 242 | 76 | 98 |
| Ni3Se4–MoSe2 (2:1) | 214 | 310 | 96 | 156 |
| MoSe2 | 210 | 295 | 80 | 134 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Guo, W.; Le, Q.V.; Do, H.H.; Hasani, A.; Tekalgne, M.; Bae, S.-R.; Lee, T.H.; Jang, H.W.; Ahn, S.H.; Kim, S.Y. Ni3Se4@MoSe2 Composites for Hydrogen Evolution Reaction. Appl. Sci. 2019, 9, 5035. https://doi.org/10.3390/app9235035
Guo W, Le QV, Do HH, Hasani A, Tekalgne M, Bae S-R, Lee TH, Jang HW, Ahn SH, Kim SY. Ni3Se4@MoSe2 Composites for Hydrogen Evolution Reaction. Applied Sciences. 2019; 9(23):5035. https://doi.org/10.3390/app9235035
Chicago/Turabian StyleGuo, Wenwu, Quyet Van Le, Ha Huu Do, Amirhossein Hasani, Mahider Tekalgne, Sa-Rang Bae, Tae Hyung Lee, Ho Won Jang, Sang Hyun Ahn, and Soo Young Kim. 2019. "Ni3Se4@MoSe2 Composites for Hydrogen Evolution Reaction" Applied Sciences 9, no. 23: 5035. https://doi.org/10.3390/app9235035
APA StyleGuo, W., Le, Q. V., Do, H. H., Hasani, A., Tekalgne, M., Bae, S.-R., Lee, T. H., Jang, H. W., Ahn, S. H., & Kim, S. Y. (2019). Ni3Se4@MoSe2 Composites for Hydrogen Evolution Reaction. Applied Sciences, 9(23), 5035. https://doi.org/10.3390/app9235035








