Elemental Composition of Biochar Obtained from Agricultural Waste for Soil Amendment and Carbon Sequestration
Abstract
1. Introduction
2. Materials and Methods
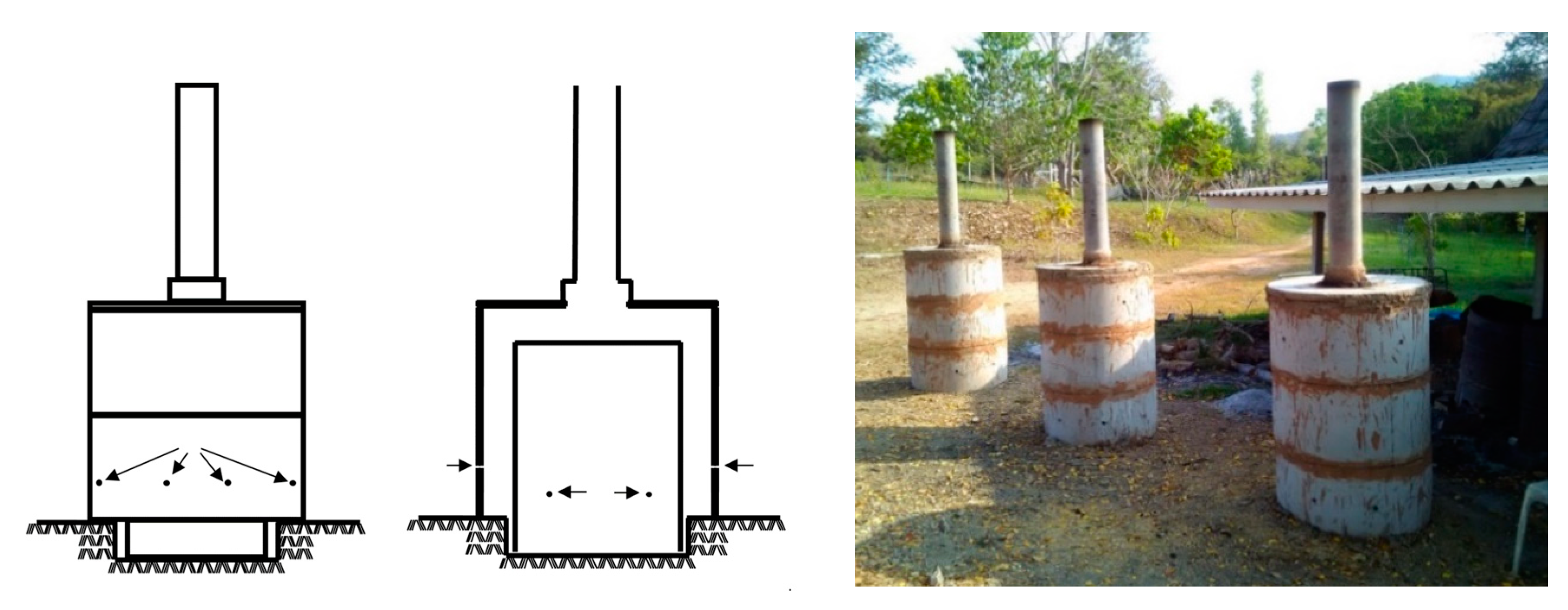
2.1. Biochar Production
2.2. Analysis Methodology
2.3. Statistical Analysis
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Hingsamer, M.; Jungmeier, G. Chapter 5 Biorefinery. In The Role of Bioenergy in the Emerging Bioeconomy: Resources, Technology, Sustainability and Policy, 1st ed.; Lago, C., Caldés, N., Lechón, Y., Eds.; Academic Press: London, UK, 2019; pp. 179–222. [Google Scholar]
- Cherubini, F.; Jungmeier, G. LCA of a biorefinery concept producing bioethanol, bioenergy, and chemicals from switchgrass. Int. J. Life Cycle Assess. 2010, 15, 53–66. [Google Scholar] [CrossRef]
- Aguilar-Reynosa, A.; Romaní, A.; Rodríguez-Jasso, R.M.; Aguilar, C.N.; Garrote, G.; Ruiz, H.A. Microwave heating processing as alternative of pretreatment in secondgeneration biorefinery: An overview. Energy Convers. Manag. 2017, 136, 50–65. [Google Scholar] [CrossRef]
- Lara-Flores, A.A.; Araújo, R.G.; Rodríguez-Jasso, R.M.; Aguedo, M.; Aguilar, C.N.; Trajano, H.L.; Ruiz, H.A. Chapter 8 Bioeconomy and Biorefinery: Valorization of Hemicellulose from Lignocellulosic Biomass and Potential Use of Avocado Residues as a Promising Resource of Bioproducts. In Waste to Wealth, Energy, Environment, and Sustainabilit, 1st ed.; Singhania, R.R., Agarwal, R.A., Kumar, R.P., Sukumaran, R.K., Eds.; Springer: Singapore, Singapore, 2018; pp. 141–170. [Google Scholar]
- Diep, N.Q.; Sakanishi, K.; Nakagoshi, N.; Fujimoto, S.; Minowa, T.; Tran, X.D. Biorefinery: Concepts, current status, and development trends. Int. J. Biomass Renew. 2012, 1, 1–8. [Google Scholar]
- Ferreira, A.F. Chapter 1 Biorefinery Concept. In Biorefineries: Targeting Energy, High Value Products and Waste Valorisation, 1st ed.; Rabaçal, M., Ferreira, A.F., Silva, C.A.M., Costa, M., Eds.; Springer: Basel, Switzerland, 2017; pp. 1–20. [Google Scholar]
- Food and Agriculture Organization of the United Nations. FAO Statistical Pocketbook 2015; Food and Agriculture Organization of the United Nations: Rome, Italy, 2015. [Google Scholar]
- de Jong, E.; Jungmeier, G. Biorefinery Concepts in Comparison to Petrochemical Refineries. In Industrial Biorefineries & White Biotechnology, 1st ed.; Pandey, A., Höfer, R., Taherzadeh, M., Nampoothiri, K.M., Larroche, C., Eds.; Elsevier B.V.: Amsterdam, The Netherlands, 2015; pp. 1–31. [Google Scholar]
- Sadhukhan, J.; Martinez-Hernandez, E.; Murphy, R.J.; Ng, D.K.S.; Hassim, M.H.; Ng, K.S.; Kin, W.K.; Jaye, I.F.M.; Hang, M.Y.L.H.; Andiappan, V. Role of bioenergy, biorefinery and bioeconomy in sustainable development: Strategic pathways for Malaysia. Renew. Sustain. Energy Rev. 2018, 81, 1966–1987. [Google Scholar] [CrossRef]
- Amoah, J.; Kahar, P.; Ogino, C.; Kondo, A. Bioenergy and Biorefinery: Feedstock, Biotechnological Conversion, and Products. Biotechnol. J. 2019, 1800494. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Li, S.; Ho, S.H.; Wang, C.; Lin, Y.C.; Nagarajan, D.; Chang, J.S.; Ren, N. Integration of sludge digestion and microalgae cultivation for enhancing bioenergy and biorefinery. Renew. Sustain. Energy Rev. 2018, 96, 76–90. [Google Scholar] [CrossRef]
- Taylor, G. Biofuels and the biorefinery concept. Energy Policy 2008, 36, 4406–4409. [Google Scholar] [CrossRef]
- Valdivia, M.; Galan, J.L.; Laffarga, J.; Ramos, J.L. Biofuels 2020: Biorefineries based on lignocellulosic materials. Microb. Biotechnol. 2016, 9, 585–594. [Google Scholar] [CrossRef]
- Almeida, J.R.M.; Fávaro, L.C.L.; Quirino, B.F. Biodiesel biorefinery: Opportunities and challenges for microbial production of fuels and chemicals from glycerol waste. Biotechnol. Biofuels 2012, 5, 48. [Google Scholar] [CrossRef]
- Sarma, S.J.; Ayadi, M.; Brar, S.K. Chapter 2 Biorefinery: General Overview. In Platform Chemical Biorefinery: Future Green Inustry, 1st ed.; Brar, S.K., Sarma, S.J., Pakshirajan, K., Eds.; Elsevier: Amsterdam, The Netherlands, 2016; pp. 21–32. [Google Scholar]
- IEA Bioenergy. Biofuel-driven Biorefineries. 2013. Available online: https://www.nachhaltigwirtschaften.at/resources/iea_pdf/iea_task_42_biofuel_driven_biorefineries_lr.pdf (accessed on 10 June 2019).
- Cherubini, F. The biorefinery concept: Using biomass instead of oil for producing energy and chemicals. Energy Convers. Manag. 2010, 51, 1412–1421. [Google Scholar] [CrossRef]
- Liu, Z.; Liao, W.; Liu, Y. A sustainable biorefinery to convert agricultural residues into value-Added chemicals. Biotechnol. Biofuels 2016, 9, 197. [Google Scholar] [CrossRef] [PubMed]
- Ghysela, S.; Léon, A.E.E.; Pala, M.; Schoder, K.A.; Ackerc, J.V.; Ronsse, F. Fast pyrolysis of mannan-Rich ivory nut (Phytelephas aequatorialis) to valuable biorefinery products. Chem. Eng. J. 2019, 373, 446–457. [Google Scholar] [CrossRef]
- Dahiya, S.; Kumar, A.N.; Sravan, J.S.; Chatterjee, S.; Sarkar, O.; Mohan, S.V. Food waste biorefinery: Sustainable strategy for circular bioeconomy. Bioresour. Technol. 2018, 248, 2–12. [Google Scholar] [CrossRef] [PubMed]
- Vaidya, A.A.; Collet, C.; Gaugler, M.; Lloyd-Jones, G. Integrating softwood biorefinery lignin into polyhydroxybutyrate composites and application in 3D printing. Mater. Today Commun. 2019, 19, 286–296. [Google Scholar] [CrossRef]
- Sriburi, T.; Wijitkosum, S. Biochar Amendment Experiments in Thailand: Practical Examples. In Biochar a Regional Supply Chain Approach in View of Climate Change Mitigation, 1st ed.; Bruckman, V.J., Varol, E.A., Uzun, B.B., Liu, J., Eds.; Cambridge University Press: Cambridge, UK, 2016; pp. 351–367. [Google Scholar]
- Wijitkosum, S.; Jiwnok, P. Effect of biochar on Chinese kale and carbon storage in an agricultural area on a high rise building. AIMS Agric. Food 2019, 4, 177–193. [Google Scholar] [CrossRef]
- Daful, A.G.; Chandraratne, M. Biochar Production from Biomass Waste-Derived Material. In Encyclopedia of Renewable and Sustainable Materials, 1st ed.; Choudhury, I., Hashmi, S., Eds.; Elsevier Science Publishing Co. Inc.: New York, NY, USA, 2018; pp. 1–9. (in press)
- Masek, O. Chapter 21 Biochar in thermal and thermochemical biorefineries-production of biochar as a coproduct. In Handbook of Biofuels Production: Process and Technologies, 2nd ed.; Luque, R., Lin, C.S.K., Wilson, K., Clark, J., Eds.; Woodhead Publishing: Duxford, UK, 2016; pp. 655–671. [Google Scholar]
- Wijitkosum, S.; Sriburi, T. Increasing the Amount of Biomass in Field Crops for Carbon Sequestration and Plant Biomass Enhancement Using Biochar. In Biochar-An Imperative Amendment for Soil and the Environment; IntechOpen: London, UK, 2018; Available online: http://www.intechopen.com/online-first/increasing-the-amount-of-biomass-in-field-crops-for-carbon-sequestration-and-plant-biomass-enhanceme (accessed on 10 June 2019).
- Li, Z.; Delvaux, B. Phytolith-Rich biochar: A potential Si fertilizer in desilicated soils. Gcb Bioenergy 2019. [Google Scholar] [CrossRef]
- Department of Alternative Energy Development and Efficiency. Biomass Energy, 1st ed.; Energy Research Centre, Department of Alternative Energy Development and Efficiency: Bangkok, Thailand, 2015. [Google Scholar]
- Lehmann, J. Bioenergy in the black. Front. Ecol. Environ. 2007, 5, 381–387. [Google Scholar] [CrossRef]
- Sirijanusorn, S.; Sriprateep, K.; Pattiya, A. Pyrolysis of cassava rhizome in a counter-Rotating twin screw reactor unit. Bioresour. Technol. 2013, 139, 343–348. [Google Scholar] [CrossRef]
- Kambo, H.S.; Dutta, A. A comparative review of biochar and hydrochar in terms of production physico-Chemical properties and applications. Renew. Sustain. Energy. Rev. 2015, 45, 359–378. [Google Scholar] [CrossRef]
- Afeng, Z.; Genxing, P.; Jinwei, Z.; Jufeng, Z.; Lianqing, L.; Liqiang, C.; Qaiser, H.; Rongjun, B.; Xiaojun, H.; Xinyan, Y.; et al. Effects of biochar amendment on soil quality, crop yield and greenhouse gas emission in a Chinese rice paddy: A field study of 2 consecutive rice growing cycles. Field Crop. Res. 2012, 127, 153–160. [Google Scholar]
- de la Rosa, J.M.; Paneque, M.; Miller, A.Z.; Knicker, H. Relating physical and chemical properties of four different biochars and their application rate to biomass production of Lolium perenne on a Calcic Cambisol during a pot experiment of 79 days. Sci. Total Environ. 2014, 499, 175–184. [Google Scholar] [CrossRef] [PubMed]
- Wijitkosum, S.; Kallayasiri, W. The Use of Biochar to Increase Productivity of Indigenous Upland Rice (Oryza sativa L.) and Improve Soil Properties. Res. J. Pharm. Biol. Chem. Sci. 2015, 6, 1326–1336. [Google Scholar]
- Gaskin, J.; Steiner, C.; Harris, K.; Das, K.; Bibens, B. Effect of low temperature pyrolysis conditions on biochar for agricultural use. Trans. ASABE 2008, 51, 2061–2069. [Google Scholar] [CrossRef]
- Kim, K.; Kim, T.; Lee, S.; Choi, D.; Yeo, H. Comparison of physicochemical features of biooils and biochars produced from various woody biomasses by fast pyrolysis. Renew. Energy 2013, 50, 188–195. [Google Scholar] [CrossRef]
- Kim, K.H.; Kim, J.; Cho, T.; Choi, J.W. Influence of pyrolysis temperature on physicochemical properties of biochar obtained from the fast pyrolysis of pitch pine (Pinus rigida). Bioresour. Technol. 2012, 118, 158–162. [Google Scholar] [CrossRef]
- Lehmann, J.; Rillig, M.C.; Thies, J.; Masiello, C.A.; Hockaday, W.C.; Crowley, D. Biochar effects on soil biota—A review. Soil Biol. Biochem. 2011, 43, 1812–1836. [Google Scholar] [CrossRef]
- Lehmann, J.; Gaunt, J.; Rondon, M. Bio-Char sequestration in terrestrial ecosystems—A review. Mitig. Adapt. Strateg. Glob. Chang. 2006, 11, 403–427. [Google Scholar] [CrossRef]
- Stewart, C.E.; Zheng, J.; Botte, J.; Cotrufo, F. Co-Generated fast pyrolysis biochar mitigates greenhouse gas emission and increases carbon sequestration in temperature soil. Glob. Change Biol. Bioenergy 2013, 5, 153–164. [Google Scholar] [CrossRef]
- Sriburi, T. Biochar researches for soil amendment at Pa-Deng Biochar Research Center (PdBRG (CC294I)), Thailand. In Proceedings of the International Symposium on Biochar for Climate Change Mitigation and Soil and Environmental Management, Kangwon, Korea, 8–9 December 2011. [Google Scholar]
- Sriburi, T.; Wijitkosum, S. Using Biochar for soil Stabilization and Increasing of Yield for Food Security and Sustainable Agriculture Initiatives; National Research Council of Thailand (NRCT): Bangkok, Thailand, 2015. [Google Scholar]
- Wijitkosum, S. The use of biochar for food security in maize production. Unisearch J. 2017, 4, 9–14. [Google Scholar]
- Intagun, W.; Khamdaeng, T.; Prom-ngarm, P.; Panyoyai, N. Effect of core puncture diameter on Bio-Char Kiln efficiency. World Acad. Sci. Eng. Technol. Int. J. Biotechnol. Bioeng. 2018, 12, 11. Available online: https://waset.org/publications/10009773/effect-of-core-puncture-diameter-on-bio-char-kiln-efficiency (accessed on 9 August 2019).
- Cornelissen, G.; Pandit, N.R.; Taylor, P.; Pandit, B.H.; Sparrevik, M.; Schmidt, H.P. Emissions and char quality of flame-Curtain "Kon Tiki" kilns for farmer-scale charcoal/biochar production. PLoS ONE 2016, 11, e0154617. [Google Scholar] [CrossRef]
- Pandit, N.R.; Mulder, J.; Hale, S.E.; Schmidt, H.P.; Cornelissen, G. Biochar from “Kon Tiki” flame curtain and other kilns: Effects of nutrient enrichment and kiln type on crop yield and soil chemistry. PLoS ONE 2017, 12, e0176378. [Google Scholar] [CrossRef]
- O’Toole, A.; Knoth de Zarruk, K.; Steffens, M.; Rasse, D.P. Characterization, stability, and plant effects of Kiln-Produced wheat straw biochar. J. Environ. Qual. 2013, 42, 429–436. [Google Scholar] [CrossRef]
- Beis, S.H.; Onay, O.; Atabay, E.; Kockar, O.M. Pyrolysis of walnut shell in a fixed-Bed reactor. In World Renewable Energy Congress VI, 1st ed.; Sayigh, A.A.M., Ed.; Pergamon Press: Oxford, UK, 2000; pp. 1360–1363. [Google Scholar]
- Yooyen, J.; Wijitkosum, S.; Sriburi, T. The use of biochar in soybean fields for carbon sequestration. In Proceedings of the International Conference on Solid Wastes 2015: Knowledge Transfer for Sustainable Resource Management (ICSWHK2015), Hong Kong, China, 19–23 May 2015. [Google Scholar]
- Ronsse, F.; Van Hecke, S.; Nachenius, R.; Prins, W. Production and characterisation of slow pyrolysis biochar. In Proceedings of the 19th European Biomass Conference and Exhibition, Berlin, Germany, 10–14 June 2011. [Google Scholar]
- Wang, T.; Stewart, C.E.; Suna, C.; Wang, Y.; Zheng, J. Effects of biochar addition on evaporation in the five typical Loess Plateau Soils. Catena 2018, 162, 29–39. [Google Scholar] [CrossRef]
- Liu, W.J.; Jiang, H.; Yu, H.Q. Development of Biochar-Based Functional Materials: Toward a Sustainable Platform Carbon Material. Chem. Rev. 2015, 115, 12251–12285. [Google Scholar] [CrossRef]
- Lee, Y.; Park, J.; Ryu, C.; Gang, K.S.; Yang, W.; Park, Y.; Jung, J.; Hyun, S. Comparison of biochar properties from biomass residues produced by slow pyrolysis at 500 °C. Bioresour. Technol. 2013, 148, 196–201. [Google Scholar] [CrossRef]
- IBI. Standardized Product Definition and Product Testing Guidelines for Biochar That is Use in Soil. Version 2.1. Available online: https://biochar-international.org/wp-content/uploads/2019/01/IBI_Biochar_Standards_V2.1_ Final1.pdf (accessed on 1 May 2017).
- McKendry, P. Energy production from biomass (part 1): Overview of biomass. Bioresour. Technol. 2002, 83, 37–46. [Google Scholar] [CrossRef]
- Yang, H.; Yan, R.; Chen, H.; Lee, D.H.; Zheng, C. Characteristics of hemicellulose, cellulose and lignin pyrolysis. Fuel 2007, 86, 1781–1788. [Google Scholar] [CrossRef]
- Chen, Y.; Duan, J.; Luo, Y. Investigation of agricultural residues pyrolysis behavior under inert and oxidative conditions. J. Anal. Appl. Pyrolysis 2008, 83, 165–174. [Google Scholar] [CrossRef]
- Novak, J.M.; Lima, I.; Xing, B.; Gaskin, J.W.; Steiner, C.; Das, K.C.; Ahmedna, M.; Rehrah, D.; Watts, D.W.; Busscher, W.J.; et al. Characterization of designer biochar produced at different temperatures and their effects on a loamy sand. Ann. Environ. Sci. 2009, 3, 195–206. [Google Scholar]
- Antal, M.J., Jr.; Gronli, M. The Art, Science, and Technology of Charcoal Production. Ind. Eng. Chem. Res. 2003, 42, 1619–1640. [Google Scholar] [CrossRef]
- Sun, Y.; Gao, B.; Yao, Y.; Fang, J.; Zhang, M.; Zhou, Y.; Chen, H.; Yang, L. Effects of feedstock type, production method, and pyrolysis temperature on biochar and hydrochar properties. Chem. Eng. J. 2014, 240, 574–578. [Google Scholar] [CrossRef]
- Brownsort, P.A. Biomass Pyrolysis Processes: Performance Parameters and Their Influence on Biochar System Benefits. Master’s Thesis, The University of Edinburg, Edinburgh, UK, 2009. [Google Scholar]
- Fang, Q.; Chen, B.; Lin, Y.; Guan, Y. Aromatic and Hydrophobic surfaces of wood-derived biochar enhance perchlorate adsorption via hydrogen bonding to oxygen oxygen-containing organic Groups. Environ. Sci. Technol. 2013, 48, 279–288. [Google Scholar] [CrossRef]
- Doumer, M.E.; Arízaga, G.G.C.; Silva, D.A.; Yamamoto, C.I.; Novotny, E.H.; Santos, J.M.; dos Santos, L.O.; Wisniewski, A., Jr.; de Andrade, J.B.; Mangrich, A.S. Slow pyrolysis of different Brazilian waste biomasses as sources of soil conditioners and energy, and for environmental protection. J. Anal. Appl. Pyrolysis 2015, 113, 434–443. [Google Scholar] [CrossRef]
- Lee, Y.; Eum, P.; Ryu, C.; Park, Y.; Jung, J. Characteristics of biochar produced from slow pyrolysis of Geodae-Uksae 1. Bioresour. Technol. 2013, 130, 345–350. [Google Scholar] [CrossRef]
- Al-Wabel, M.I.; Al-Omran, A.; El-Naggar, A.H.; Nadeem, M.; Usman, A.R.A. Pyrolysis temperature induced changes in characteristics and chemical composition of biochar produced from conocarpus wastes. Bioresour. Technol. 2013, 131, 374–379. [Google Scholar] [CrossRef]
- Gul, S.; Whalen, J.K.; Thomas, B.W.; Sachdeva, V.; Deng, H. Physico-chemical properties and microbial responses in biochar-Amended soil mechanism and future directions. Agric. Ecosyst. Environ. 2015, 206, 46–59. [Google Scholar] [CrossRef]
- Ennis, C.J.; Evans, A.G.; Islam, M.; Komang, K.; Senior, R. Biochar carbon sequestration, land remediation, and impact on soil microbiology. Crit. Rev. Environ. Sci. Technol. 2012, 42, 2311–2364. [Google Scholar] [CrossRef]
- Lai, W.Y.; Lai, C.M.; Ke, G.R.; Chung, R.S.; Chen, C.T.; Cheng, C.H.; Pai, C.W.; Chen, S.Y.; Chen, C.C. The effects of woodchip biochar application on crop yield, carbon sequestration and greenhouse gas emission from soils planted with rice or leaf beet. J. Taiwan Int. Chem. Eng. 2013, 44, 1039–1044. [Google Scholar] [CrossRef]
- Chia, C.H.; Downie, A.; Munroe, P. Characteristics of Biochar: Physical and Structural Properties. In Biochar for Environmental Management Science, Technology and Implementation, 2nd ed.; Lehmann, J., Joseph, S., Eds.; Routledge: London, UK, 2015; pp. 89–110. [Google Scholar]
- Jin, W.; Singh, K.; Zondlo, J. Pyrolysis Kinetics of Physical Components of Wood and Wood-Polymers Using Isoconversion Method. Agriculture 2013, 3, 12–32. [Google Scholar] [CrossRef]
- Wei, S.; Zhu, M.; Fan, X.; Song, J.; Peng, P.; Li, K.; Jia, W.; Song, H. Influence of pyrolysis temperature and feedstock on carbon fractions of biochar produced from pyrolysis of rice straw, pine wood, pig manure and sewage sludge. Chemosphere 2018, 218, 624–631. [Google Scholar] [CrossRef]
- Laird, D.A.; Brown, R.C.; Amonette, J.E.; Lehmann, J. Review of the pyrolysis platform for coproducing bio-oil and biochar. Biofuel Bioprod. Biorein. 2009, 3, 547–562. [Google Scholar] [CrossRef]
- Leng, L.; Yuan, X.; Zeng, G.; Shao, J.; Chen, X.; Wu, Z.; Wang, H.; Peng, X. Surface characterization of rice husk bio-Char produced by liquefaction and application for cationic dye (malachite green) adsorption. Fuel 2015, 155, 77–85. [Google Scholar] [CrossRef]
- Ayhan, D. Effects of Temperature and Particle Size on Bio-Char Yield from Pyrolysis of Agricultural Residues. J. Anal. Appl. Pyrolysis 2004, 72, 243–248. [Google Scholar]
- Downie, A.; Crosky, A.; Munroe, P. Physical properties of biochar. In Biochar for Environmental Management: Science and Technology, 1st ed.; Lehmann, J., Joseph, S., Eds.; Earthscan: London, UK, 2009; pp. 13–32. [Google Scholar]
- Lehmann, J.; Joseph, S. Biochar for Environmental Management: An Introduction. 2009. Available online: http://www.biochar-international.org/images/Biochar_book_Chapter_1.pdf (accessed on 5 January 2016).
- Sohi, S.P.; Lopez-Capel, E.; Krull, E.; Bol, R. Biochar, climate change and soil: A review to guide future research. CSIRO Land Water Sci. Rep. 2009, 5, 17–31. [Google Scholar]
- Batista, E.M.C.C.; Shultz, J.; Matos, T.T.S.; Fornari, M.R.; Ferreira, T.M.; Szpoganicz, B.; de Freitas, R.A.; Mangrich, A.S. Effect of surface and porosity of biochar on water holding capacity aiming indirectly at preservation of the Amazon biome. Sci. Rep. 2018, 8, 10677. [Google Scholar] [CrossRef]
- Pituello, C.; Francioso, O.; Simonetti, G.; Pisi, A.; Torreggiani, A.; Berti, A.; Morari, F. Characterization of chemical—Physical, structural and morphological properties of biochars from biowastes produced at different temperatures. J. Soils Sediments 2015, 15, 792–804. [Google Scholar] [CrossRef]
- Hassler, J.W. Purification with Activated Carbon: Industrial, Commercial, Environmental; Chemical Publishing Company: New York, NY, USA, 1974. [Google Scholar]
- Wilon, K. How biochar works in soil, the Biochar Journal 2014, Arbaz, Switzerland. Available online: www.biochar-journal.org/en/ct/32 (accessed on 14 June 2019).
- Yuan, J.; Xu, R.; Zhang, H. The forms of alkalis in the biochar produced from crop residues at different temperatures. Bioresour. Technol. 2011, 102, 3488–3497. [Google Scholar] [CrossRef]
- Hmid, A.; Mondelli, D.; Fiore, S.; Fanizzi, F.P.; Al Chami, Z.; Dumontet, S. Production and characterization of biochar from three-Phase olive mill waste through slow pyrolysis. Biomass Bioenergy 2014, 71, 330–339. [Google Scholar] [CrossRef]
- McHenry, M.P. Agricultural bio-Char production, renewable energy generation and farm carbon sequestration in Western Australia: Certainty, uncertainty and risk. Agric. Ecosyst. Environ. 2008, 129, 1–7. [Google Scholar] [CrossRef]
- Manya, J.J. Pyrolysis for biochar purposes: A review to establish current knowledge gaps and research needs. Environ. Sci. Technol. 2012, 46, 7939–7954. [Google Scholar] [CrossRef]
- Budai, A.; Wang, L.; Gronli, M.; Strand, L.T.; Antal, M.J.; Abiven, S.; Dieguez-Alonso, A.; Anca-Couce, A.; Rasse, D.P. Surface properties and chemical composition of corncob and miscanthus biochars: Effects of production temperature and method. J. Agric. Food Chem. 2014, 62, 3791–3799. [Google Scholar] [CrossRef]
- Liu, X.; Ye, Y.; Liu, Y.; Zhang, A.; Zhang, X.; Li, L.; Pan, G.; Kibue, G.W.; Zheng, J.; Zheng, J. Sustainable biochar effects for low carbon crop production: A 5-crop season field experiment on a low fertility soil from Central China. Agric. Syst. 2014, 129, 22–29. [Google Scholar] [CrossRef]
- Masulili, A.; Utomo, W.H. Rice husk biochar for rice based cropping system in acid soil 1. The Characteristics of rice husk biochar and Its Influence on the Properties of Acid Sulfate Soils and Rice Growth in West Kalimantan, Indonesia. J. Agric. Sci. 2010, 2, 39–47. [Google Scholar] [CrossRef]
- Liang, B.; Lehmann, J.; Solomon, D.; Kinyangi, J.; Grossman, J.; O’Neill, B.; Skjemstad, J.O.; Thies, J.; Luizao, F.J.; Petersen, J.; et al. Black Carbon increases cation exchange capacity in soils. Soil Sci. Soc. Am. J. 2006, 70, 1719–1730. [Google Scholar] [CrossRef]
- Ronsse, F.; Hecke, S.V.; Dickinson, D.; Prins, W. Production and characterization of slow pyrolysis biochar: Influence of feedstock type and pyrolysis conditions. Glob. Chang. Biol. Bioenergy 2013, 5, 104–115. [Google Scholar] [CrossRef]
- Zhao, S.X.; Ta, N.; Wang, X.D. Effect of temperature on the structural and physicochemical properties of biochar with apple tree branches as feedstock material. Energies 2017, 10, 1293. [Google Scholar] [CrossRef]
- Zhang, J.; Liu, J.; Liu, R. Effects of pyrolysis temperature and heating time on biochar obtained from the pyrolysis of straw and lignosulfonate. Bioresour. Technol. 2015, 176, 288–291. [Google Scholar] [CrossRef]
- Babu, B.V. Biomass pyrolysis: A state-of-the-Art review. Biofuels Bioprod. Biorefin. 2008, 2, 393–414. [Google Scholar] [CrossRef]
- Chen, Y.; Zhang, X.; Chen, W.; Yang, H.; Chen, H. The structure evolution of biochar from biomass pyrolysis and its correlation with gas pollutant adsorption performance. Bioresour. Technol. 2017, 246, 101–109. [Google Scholar] [CrossRef]
- Chen, Y.; Yang, H.; Wang, X.; Zhang, S.; Chen, H. Biomass-Based pyrolytic polygeneration system on cotton stalk pyrolysis: Influence of temperature. Bioresour. Technol. 2012, 107, 411–418. [Google Scholar] [CrossRef]
- Xiao, R.R.; Yang, W. Influence of temperature on organic structure of biomass pyrolysis products. Renew. Energy 2013, 50, 136–141. [Google Scholar] [CrossRef]
- Zhao, Y.J.; Feng, D.D.; Zhang, Y.; Huang, Y.D.; Sun, S.Z. Effect of pyrolysis temperature on char structure and chemical speciation of alkali and alkaline earth metallic species in biochar. Fuel Process. Technol. 2016, 141, 54–60. [Google Scholar] [CrossRef]
- Oh, T.K.; Choi, B.S.; Shinogi, Y.; Chikushi, J. Characterization of biochar derived from three types of biomass. J. Fac. Agric. Kyushu. Univ. 2012, 57, 61–66. [Google Scholar]
- Gul, S.; Whalen, J.K. Biochemical cycling of nitrogen and phosphorus in biochar-amended soils. Soil Biol. Biochem. 2016, 103, 1–15. [Google Scholar] [CrossRef]
- Liang, B.; Lehmann, J.; Sohi, S.P.; Thies, J.E.; O’Neill, B.; Trujillo, L.; Gaunt, J.; Solomon, D.; Grossman, J.; Neves, E.G.; et al. Black carbon affects the cycling of non-black carbon in soil. Org. Geochem. 2010, 41, 206–213. [Google Scholar] [CrossRef]
- Amonette, J.; Joseph, S. Characteristics of Biochar: Micro Chemical Properties. In Biochar for Environmental Management: Science and Technology; Lehmann, J., Joseph, S., Eds.; Earthscan: London, UK, 2009; pp. 33–52. [Google Scholar]
- Glaser, B.; Lehmann, J.; Zech, W. Ameliorating physical and chemical properties of highly weathered soils in the tropics with charcoal—A review. Biol. Fertil. Soils 2002, 35, 219–230. [Google Scholar] [CrossRef]
- Lehmann, J.; da Silva, J.; Steiner, C.; Nehls, T.; Zech, W.; Glaser, B. Nutrient availability and leaching in an archaeological Anthrosol and a Ferralsol of the Central Amazon basin: Fertilizer, manure and charcoal amendments. Plant Soil 2003, 249, 343–357. [Google Scholar] [CrossRef]
- Laird, D.A.; Fleming, P.; Wang, B.Q.; Horton, R.; Karlen, D. Biochar impact on nutrient leaching from a Midwestern agricultural soil. Geoderma 2010, 158, 436–442. [Google Scholar] [CrossRef]
- Steiner, C.; Teixeira, W.G.; Lehmann, J.; Nehls, T.; de Macêdo, J.L.; Blum, W.E.; Zech, W. Long term effects of manure, charcoal, and mineral fertilization on crop production and fertility on a highly weathered central Amazonian upland soil. Plant Soil 2007, 291, 275–290. [Google Scholar] [CrossRef]
- Li, Z.; Unzué-Belmonte, D.; Cornelis, J.T.; Linden, C.V.; Struyf, E.; Ronsse, F.; Delvaux, B. Effects of phytolithic rice-Straw biochar, soil buffering capacity and pH on silicon bioavailability. Plant Soil 2019, 438, 187–203. [Google Scholar] [CrossRef]
- Nwajiaku, I.M.; Sato, K.; Tokunari, T.; Kitano, S.; Masunaga, T. Improvement of rice husk residue silicon availability for replenishing available silicon in paddy soil. Int. J. Plant Soil Sci. 2018, 24, 1–11. [Google Scholar] [CrossRef]
- Zhang, A.; Liu, Y.; Pan, G.; Hussain, Q.; Li, L.Q.; Zheng, J.; Zhang, X. Effect of biochar amendment on maize yield and greenhouse gas emissions from a soil organic carbon poor calcareous loamy soil from Central China Plain. Plant Soil 2012, 351, 263–272. [Google Scholar] [CrossRef]
- Major, J.; Rondon, M.; Molina, D.; Riha, S.J.; Lehmann, J. Maize yield and nutrition during 4 years after biochar application to a Colombian savanna oxisol. Plant Soil 2010, 333, 117–128. [Google Scholar] [CrossRef]
- Zheng, W.; Sharma, B.K.; Rajagopalan, N. Using Biochar as a Soil Amendment for Sustainable Agriculture; Illinois Department of Agriculture: Champaign, IL, USA, 2010; pp. 1–36. [Google Scholar]
- Yamato, M.; Okimori, Y.; Wibowo, I.F.; Anshori, S.; Ogawa, M. Effects of the application of charred bark of Acacia mangium on the yield of maize, cowpea and peanut, and soil chemical properties in South Sumatra, Indonesia. J. Soil Sci. Plant Nutr. 2006, 52, 489–495. [Google Scholar] [CrossRef]
- Asai, H.; Saito, K.; Samson, B.; Songyikhangsuthor, K.; Homma, K.; Shiraiwa, T.; Kiyono, Y.; Inoue, Y.; Horie, T. Yield response of indica and tropical japonica genotypes to soil fertility conditions under rainfed uplands in northern Laos. Field Crops Res. 2009, 112, 141–148. [Google Scholar] [CrossRef]
- Knowles, O.A.; Robinson, B.H.; Contangelo, A.; Clucas, L. Biochar for the mitigation of nitrate leaching from soil amended with biosolids. Sci. Total Environ. 2011, 409, 3206–3210. [Google Scholar] [CrossRef]
- Doan, T.T.; Henry-des-Tureaux, T.; Rumpel, C.; Janeau, J.; Jouquet, P. Impact of compost, vermicompost and biochar on soil fertility, maize yield and soil erosion in Northern Vietnam: A three year mesocosm experiment. Sci. Total Environ. 2015, 514, 147–154. [Google Scholar] [CrossRef]
- Carter, S.; Shackley, S.; Sohi, S.; Suy, T.B.; Haefele, S. The Impact of biochar application on soil properties and plant growth of pot grown Lettuce (Lactuca sativa) and Cabbage (Brassica chinensis). Agronomy 2013, 3, 404–418. [Google Scholar] [CrossRef]
- Wijitkosum, S.; Mattayom, B.; Sriburi, T. The effects of biochar on increasing the yield of seeda tomatoes. In Proceedings of the International Conference on Solid Wastes 2015: Knowledge Transfer for Sustainable Resource Management (ICSWHK2015), Hong Kong, China, 19–23 May 2015. [Google Scholar]
- Liang, F.; Li, G.T.; Lin, Q.M.; Zhao, X.R. Crop yield and soil properties in the first 3 years after biochar application to a calcareous Soil. J. Integr. Agric. 2014, 13, 525–532. [Google Scholar] [CrossRef]
- Sohi, S.P.; Krull, E.; Lopez-Capel, E.; Bol, R. A review of biochar and its use and function in soil. Adv. Agron. 2010, 105, 47–82. [Google Scholar]
- Spokas, K.A.; Reicosky, D.C. Impacts of sixteen different biochars on soil greenhouse gas production. Ann. Environ. Sci. 2009, 3, 179–193. [Google Scholar]
- Windeatt, J.H.; Ross, A.B.; Williams, P.T.; Forster, P.M.; Nahil, M.A.; Singh, S. Characteristics of biochars from crop residues: Potential for carbon sequestration and soil amendment. J. Environ. Manag. 2014, 146, 189–197. [Google Scholar] [CrossRef]
- Thomazini, A.; Spokas, K.; Hall, K.; Ippolito, J.; Lentz, R.; Novak, J. GHG impacts of biochar: Predictability for the same biochar. Agric. Ecosyst. Environ. 2015, 207, 183–191. [Google Scholar] [CrossRef]
| Corncob | Cassava Rhizome | Cassava Stem | |
|---|---|---|---|
| C (wt%) | 41.66 a ± 1.12 | 37.60 a ± 2.22 | 41.55 a ± 0.92 |
| H (wt%) | 6.84 a ± 0.36 | 6.15 a ± 0.58 | 6.04 a ± 0.13 |
| N (wt%) | 0.74 b ± 0.02 | 0.88 b ± 0.04 | 1.27 a ± 0.14 |
| O (wt%) | 50.76 a ± 0.89 | 55.37 a ± 1.64 | 51.14 a ± 1.18 |
| H/C ratio | 1.97 a ± 0.17 | 1.96 a ± 0.27 | 1.74 a ± 0.06 |
| O/C ratio | 0.91 a ± 0.05 | 1.10 a ± 0.09 | 0.92 a ± 0.05 |
| C/N ratio | 65.63 a ± 1.89 | 50.16 b ± 3.86 | 39.30 b ± 3.53 |
| Molecular formula | CH1.97O0.91 | CH1.96O1.10 | CH1.74O0.92 |
| Surface area (m2/g) | 2.54 b ± 0.41 | 2.78 b ± 0.23 | 2.51 a ± 0.03 |
| Total pore volume (cm3/g) | 0.0034 b ± 0.0007 | 0.0043 b ± 0.0004 | 0.0058 a ± 0.0006 |
| Average pore diameter (Å) | 31.05 c ± 0.75 | 69.57 b± 5.58 | 83.34 a ± 1.79 |
| Corncob | Cassava Rhizome | Cassava Stem | |
|---|---|---|---|
| C (wt%) | 81.35 a ± 1.42 | 64.25 b ± 1.53 | 62.95 b ± 3.71 |
| H (wt%) | 2.42 a ± 0.17 | 2.73 a ± 0.17 | 2.24 a ± 0.12 |
| N (wt%) | 1.22 b ± 0.14 | 1.65 a ± 0.08 | 1.37 ab ± 0.06 |
| O (wt%) | 15.23 b ± 1.27 | 31.80 a ± 1.27 | 33.44 a ± 3.81 |
| H/C ratio | 0.36 a ± 0.02 | 0.43 a ± 0.12 | 0.42 a ± 0.01 |
| O/C ratio | 0.14 b ± 0.015 | 0.37 a ± 0.02 | 0.39 a ± 0.08 |
| C/N ratio | 80.60 a ± 8.41 | 45.72 b ± 2.34 | 54.08 b ± 4.51 |
| Molecular formula | CH0.36O0.14 | CH0.51O0.37 | CH0.43O0.40 |
| Surface area (m2/g) | 56.35 b ± 0.79 | 18.38 b ± 2.21 | 200.46 a ± 26.18 |
| Total pore volume (cm3/g) | 0.0405 b ± 0.0041 | 0.0284 b ± 0.0029 | 0.1219 a ± 0.01 |
| Average pore diameter (Å) | 28.72 b ± 2.29 | 61.69 a ± 1.64 | 24.35 b ± 1.82 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wijitkosum, S.; Jiwnok, P. Elemental Composition of Biochar Obtained from Agricultural Waste for Soil Amendment and Carbon Sequestration. Appl. Sci. 2019, 9, 3980. https://doi.org/10.3390/app9193980
Wijitkosum S, Jiwnok P. Elemental Composition of Biochar Obtained from Agricultural Waste for Soil Amendment and Carbon Sequestration. Applied Sciences. 2019; 9(19):3980. https://doi.org/10.3390/app9193980
Chicago/Turabian StyleWijitkosum, Saowanee, and Preamsuda Jiwnok. 2019. "Elemental Composition of Biochar Obtained from Agricultural Waste for Soil Amendment and Carbon Sequestration" Applied Sciences 9, no. 19: 3980. https://doi.org/10.3390/app9193980
APA StyleWijitkosum, S., & Jiwnok, P. (2019). Elemental Composition of Biochar Obtained from Agricultural Waste for Soil Amendment and Carbon Sequestration. Applied Sciences, 9(19), 3980. https://doi.org/10.3390/app9193980




