The Antifungal Potential of Carvacrol against Penicillium Digitatum through 1H-NMR Based Metabolomics Approach
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Preparation of P. Digitatum Spores
2.3. Antifungal Effects of Carvacrol against P. Digitatum
2.4. The Effect of Carvacrol on Mycelial Weights and Water-Retention Rate
2.5. Sample Preparation for 1H NMR Spectroscopy
2.6. Spectral Pre-Processing and Data Analysis
2.7. Statistical Analysis
3. Results
3.1. Effects of Carvacrol on P. Digitatum Mycelial Growth on PDA
3.2. Effects of Carvacrol on Mycelial Weights in PDB
3.3. Effect of Carvacrol on Water-Retention Rate of P. Digitatum
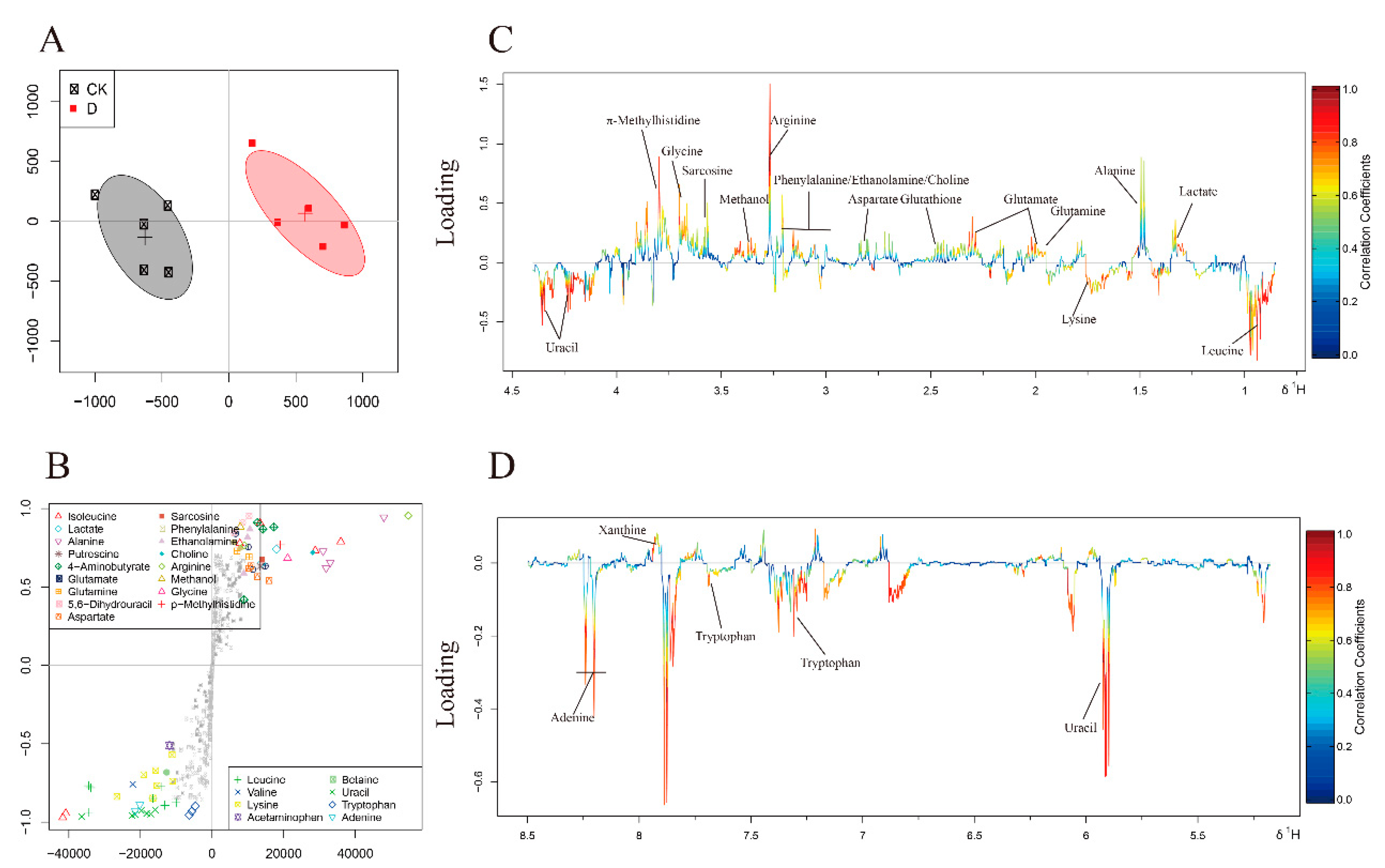
3.4. Metabolites Identified in 1H-NMR Spectra
3.5. Multivariate Analysis of 1H-NMR Spectral Data
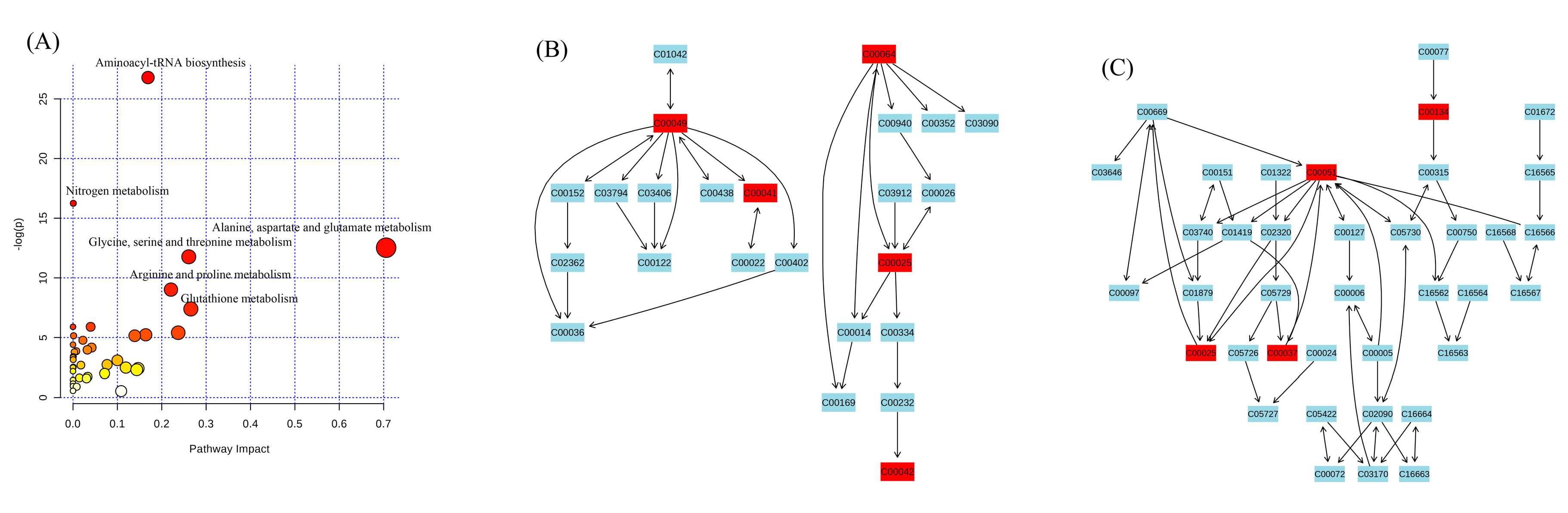
4. Discussion
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Nicosia, M.G.; Pangallo, S.; Raphael, G.; Romeo, F.V.; Strano, M.C.; Rapisarda, P.; Droby, S.; Schena, L. Control of postharvest fungal rots on citrus fruit and sweet cherries using a pomegranate peel extract. Postharvest Biol. Technol. 2016, 114, 54–61. [Google Scholar] [CrossRef]
- Chen, J.; Shen, Y.; Chen, C.; Wan, C. Inhibition of key citrus postharvest fungal strains by plant extracts in vitro and in vivo: A review. Plants 2019, 8, 26. [Google Scholar] [CrossRef]
- Chen, C.; Cai, N.; Chen, J.; Peng, X.; Wan, C. Chitosan-Based Coating Enriched with Hairy Fig (Ficus hirta Vahl.) Fruit Extract for “Newhall” Navel Orange Preservation. Coatings 2018, 8, 445. [Google Scholar] [CrossRef]
- Zafar, I.; Zora, S.; Ravjit, K.; Saeed, A. Management of citrus blue and green moulds through application of organic elicitors. Australas. Plant Pathol. 2012, 41, 69–77. [Google Scholar]
- Hao, W.; Li, H.; Hu, M.Y.; Liu, Y.; Rizwan-ul-Haq, M. Integrated control of citrus green and blue mold and sour rot by Bacillus amyloliquefaciens incombination with tea saponin. Postharvest Biol. Technol. 2011, 59, 316–323. [Google Scholar] [CrossRef]
- Palou, L.; Ali, L.; Fallik, E.; Romanazzi, G. GRAS, plant- and animal-derived compounds as alternatives to conventional fungicides for the control of postharvest dieseases of fresh horticultural produce. Postharvest Biol. Technol. 2016, 122, 41–52. [Google Scholar] [CrossRef]
- Gong, T.; Li, C.; Bian, B.; Wu, Y.; Dawuda, M.M.; Liao, W. Advances in application of small molecule compounds for extending the shelf life of perishable horticultural products: A review. Sci. Hortic. 2018, 230, 25–34. [Google Scholar] [CrossRef]
- Zhang, M.; Xu, L.; Zhang, L.; Guo, Y.; Qi, X.; He, L. Effects of quercetin on postharvest blue mold control in kiwifruit. Sci. Hortic. 2018, 228, 18–25. [Google Scholar] [CrossRef]
- Ncama, K.; Magwaza, L.S.; Mditshwa, A.; Tesfay, S.Z. Plant-based edible coatings for managing postharvest quality of fresh horticultural produce: A review. Food Packag. Shelf Life 2018, 16, 157–167. [Google Scholar] [CrossRef]
- Zhang, X.F.; Guo, Y.J.; Guo, L.Y.; Jiang, H.; Ji, Q.H. In Vitro Evaluation of Antioxidant and Antimicrobial Activities of Melaleuca alternifolia Essential Oil. BioMed Res. Int. 2018, 2018, 2396109. [Google Scholar] [CrossRef]
- Wan, C.P.; Pei, L.; Chen, C.Y.; Peng, X.; Li, M.X.; Chen, M.; Wang, J.S.; Chen, J.Y. Antifungal Activity of Ramulus cinnamomi Explored by 1H-NMR Based Metabolomics Approach. Molecules 2017, 22, 2237. [Google Scholar] [CrossRef]
- Trabelsi, D.; Hamdane, A.M.; Said, M.B.; Abdrrsbba, M. Chemical composition and antifungal activity of essential oils from flowers, leaves and peels of Tunisian Citrus aurantium against Penicillium digitatum and Penicillium italicum. J. Essent. Oil Bear. Plants 2016, 19, 1660–1674. [Google Scholar] [CrossRef]
- Can Baser, K.H. Biological and pharmacological activities of carvacrol and carvacrol bearing essential oils. Curr. Pharm. Des. 2008, 14, 3106–3119. [Google Scholar] [CrossRef]
- Zotti, M.; Colaianna, M.; Morgese, M.; Tucci, P.; Schiavone, S.; Avato, P.; Trabace, L. Carvacrol: From ancient flavoring to neuromodulatory agent. Molecules 2013, 18, 6161–6172. [Google Scholar] [CrossRef]
- Pérez-Alfonso, C.O.; Martínez-Romero, D.; Zapata, P.J.; Serrano, M.; Valero, D.; Castillo, S. The effects of essential oils carvacrol and thymol on growth of Penicillium digitatum and P. italicum involved in lemon decay. Int. J. Food Microbiol. 2012, 158, 101–106. [Google Scholar]
- Boubaker, H.; Karim, H.; Hamdaoui, A.E.; Msanda, F.; Leach, D.; Bombarda, I.; Vanloot, P.; Abbad, A.; Boudyach, E.H.; Aoumar, A.B. Chemical characterization and antifungal activities of four Thymus species essential oils against postharvest fungal pathogens of citrus. Ind. Crop. Prod. 2016, 86, 95–101. [Google Scholar] [CrossRef]
- Sun, X.; Narciso, J.; Wang, Z.; Ference, C.; Bai, J.; Zhou, K. Effects of chitosan-essential oil coatings on safety and quality of fresh blueberries. J. Food Sci. 2014, 79, M955–M960. [Google Scholar] [CrossRef]
- Keun, H.C.; Ebbels, T.M.; Antti, H.; Bollard, M.E.; Beckonert, O.; Schlotterbeck, G.; Senn, H.; Niederhauser, U.; Holmes, E.; Lindon, J.C.; et al. Analytical Reproducibility in 1H NMR-Based Metabonomic Urinalysis. Chem. Res. Toxicol. 2002, 15, 1380–1386. [Google Scholar] [CrossRef]
- Liu, X.; Zhang, L.; You, L.; Cong, M.; Zhao, J.; Wu, H.; Li, C.; Liu, D.; Yu, J. Toxicological responses to acute mercury exposure for three species of Manila clam Ruditapes philippinarum by NMR-based metabolomics. Environ. Toxicol. Pharmacol. 2011, 31, 323–332. [Google Scholar] [CrossRef]
- Liu, Q.; Wu, J.E.; Lim, Z.Y.; Aggarwal, A.; Yang, H.; Wang, S. Evaluation of the metabolic response of Escherichia coli to electrolysed water by 1H NMR spectroscopy. LWT Food Sci. Technol. 2017, 79, 428–436. [Google Scholar] [CrossRef]
- Picone, G.; Laghi, L.; Gardini, F.; Lanciotti, R.; Siroli, L.; Capozzi, F. Evaluation of the effect of carvacrol on the Escherichia coli 555 metabolome by using 1H-NMR spectroscopy. Food Chem. 2013, 141, 4367–4374. [Google Scholar] [CrossRef]
- Chen, C.; Qi, W.; Peng, X.; Chen, J.; Wan, C. Inhibitory Effect of 7-Demethoxytylophorine on Penicillium italicum and its Possible Mechanism. Microorganisms 2019, 7, 36. [Google Scholar] [CrossRef]
- Tian, J.; Wang, Y.; Zeng, H.; Li, Z.; Zhang, P.; Tessema, A.; Peng, X. Efficacy and possible mechanisms of perillaldehyde in control of Aspergillus niger causing grape decay. Int. J. Food Microbiol. 2015, 202, 27–34. [Google Scholar] [CrossRef]
- Chen, C.; Fu, Y.H.; Li, M.H.; Li, M.H.; Ruan, L.Y.; Xu, H.; Chen, J.F.; Wang, J.S. Nuclear magnetic resonance-based metabolomics approach to evaluate preventive and therapeutic effects of Gastrodia elata Blume on chronic atrophic gastritis. J. Pharm. Biomed. Anal. 2019, 164, 231–240. [Google Scholar] [CrossRef]
- Ballatori, N.; Jacob, R.; Boyer, J.L. Intrabiliary glutathione hydrolysis. A source of glutamate in bile. J. Biol. Chem. 1986, 261, 7860–7865. [Google Scholar]
- Apel, K.; Hirt, H. Reactive oxygen Species: Metabolism, Oxidative Stress, and Signal Transduction. Annu. Rev. Plant Biol. 2004, 55, 373–399. [Google Scholar] [CrossRef]
- Fu, Y.; Si, Z.; Li, P.; Li, M.; Zhao, H.; Jiang, L.; Xing, Y.; Hong, W.; Ruan, L.; Wang, J.S. Acute psychoactive and toxic effects of D. metel on mice explained by 1H NMR based metabolomics approach. Metab. Brain Dis. 2017, 32, 1295–1309. [Google Scholar] [CrossRef]
- Boncompagni, E.; Østerås, M.; Poggi, M.C.; le Rudulier, D. Occurrence of Choline and Glycine Betaine Uptake and Metabolism in the Family Rhizobiaceae and Their Roles in Osmoprotection. Appl. Environ. Microbiol. 1999, 65, 2072–2077. [Google Scholar]
- Armstrong, C.W.; McGregor, N.R.; Sheedy, J.R.; Buttfield, I.; Butt, H.L.; Gooley, P.R. NMR metabolic profiling of serum identifies amino acid disturbances in chronic fatigue syndrome. Clin. Chim. Acta 2012, 413, 1525–1531. [Google Scholar] [CrossRef]
- Sakurai, K.; Shinkai, S. Molecular Recognition of Adenine, Cytosine, and Uracil in a Single-Stranded RNA by a Natural Polysaccharide: Schizophyllan. J. Am. Chem. Soc. 2000, 122, 1–8. [Google Scholar] [CrossRef]
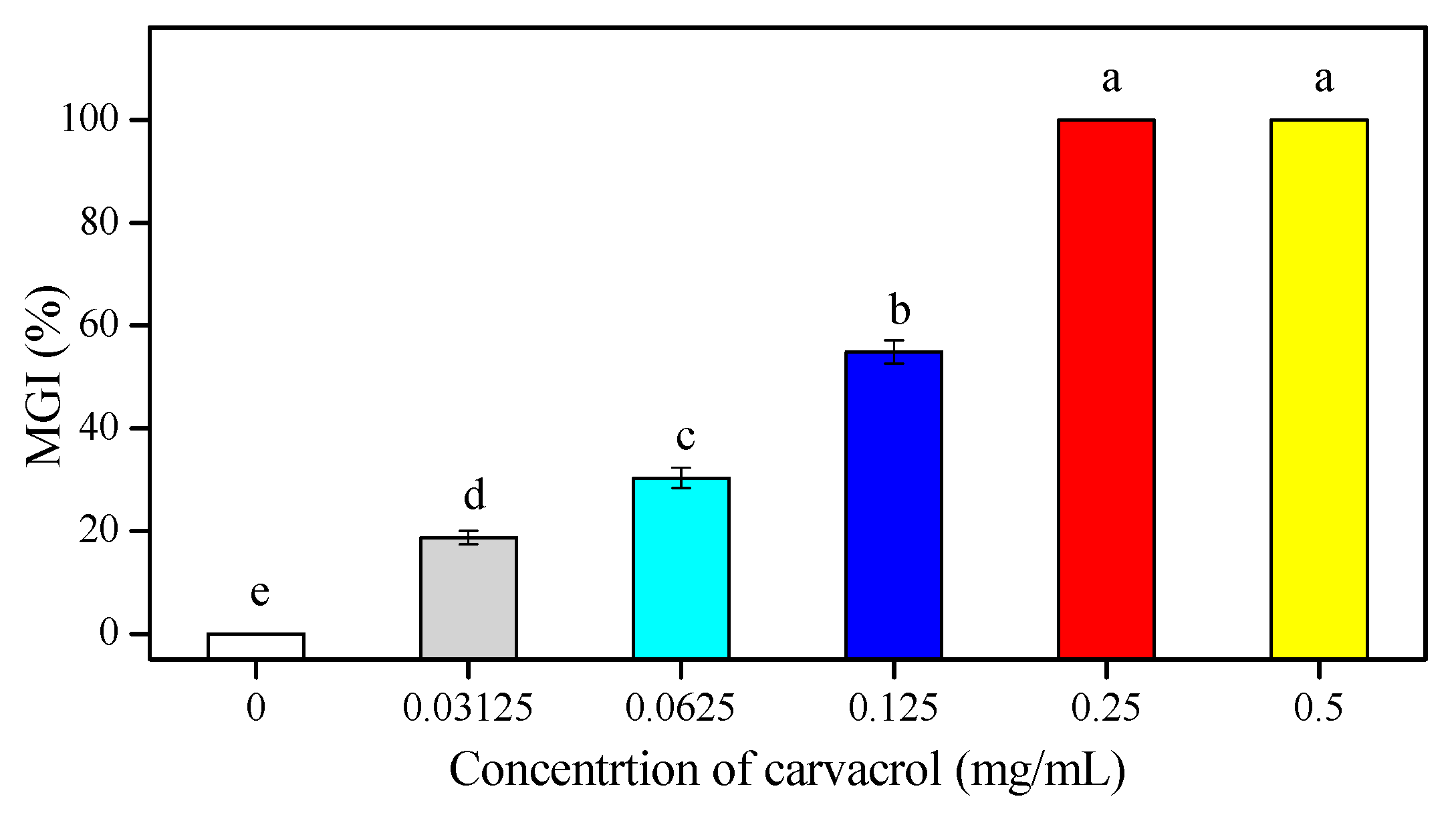
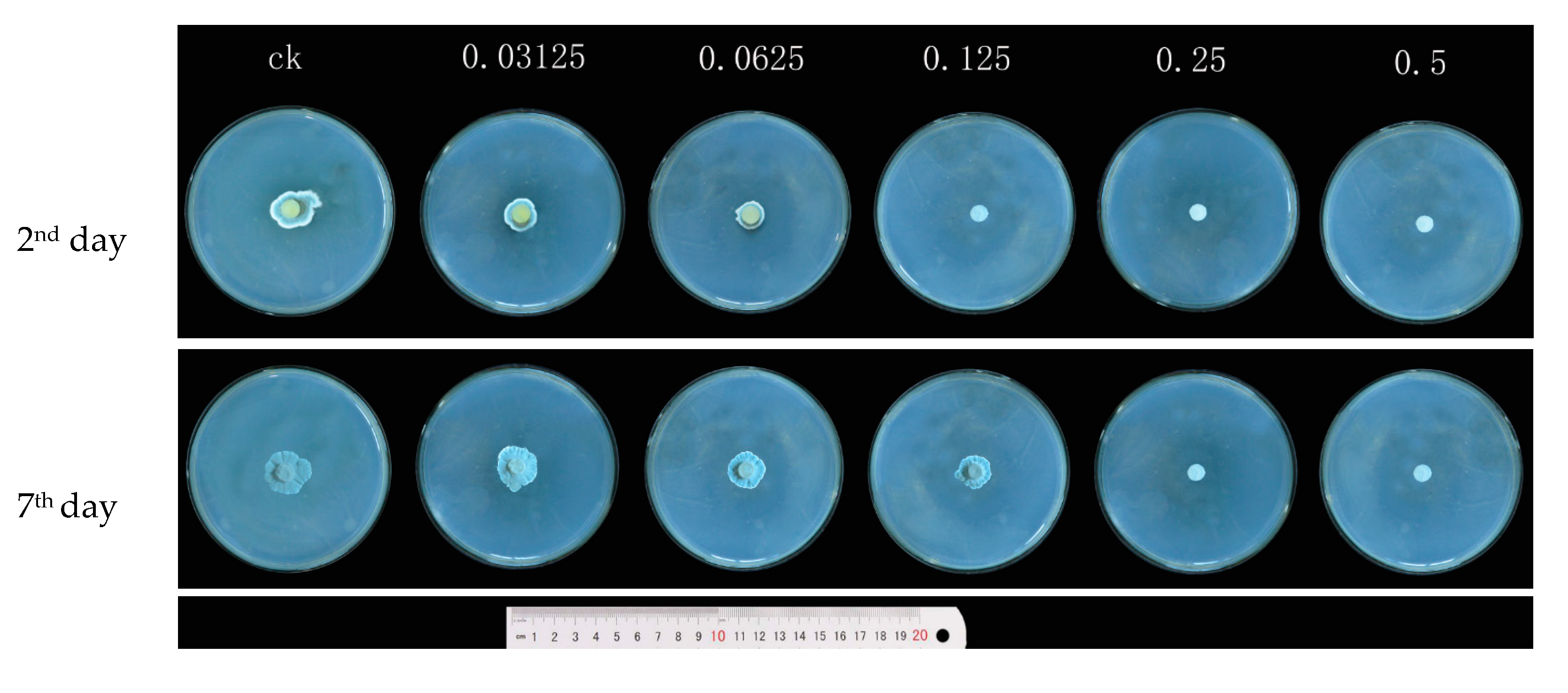
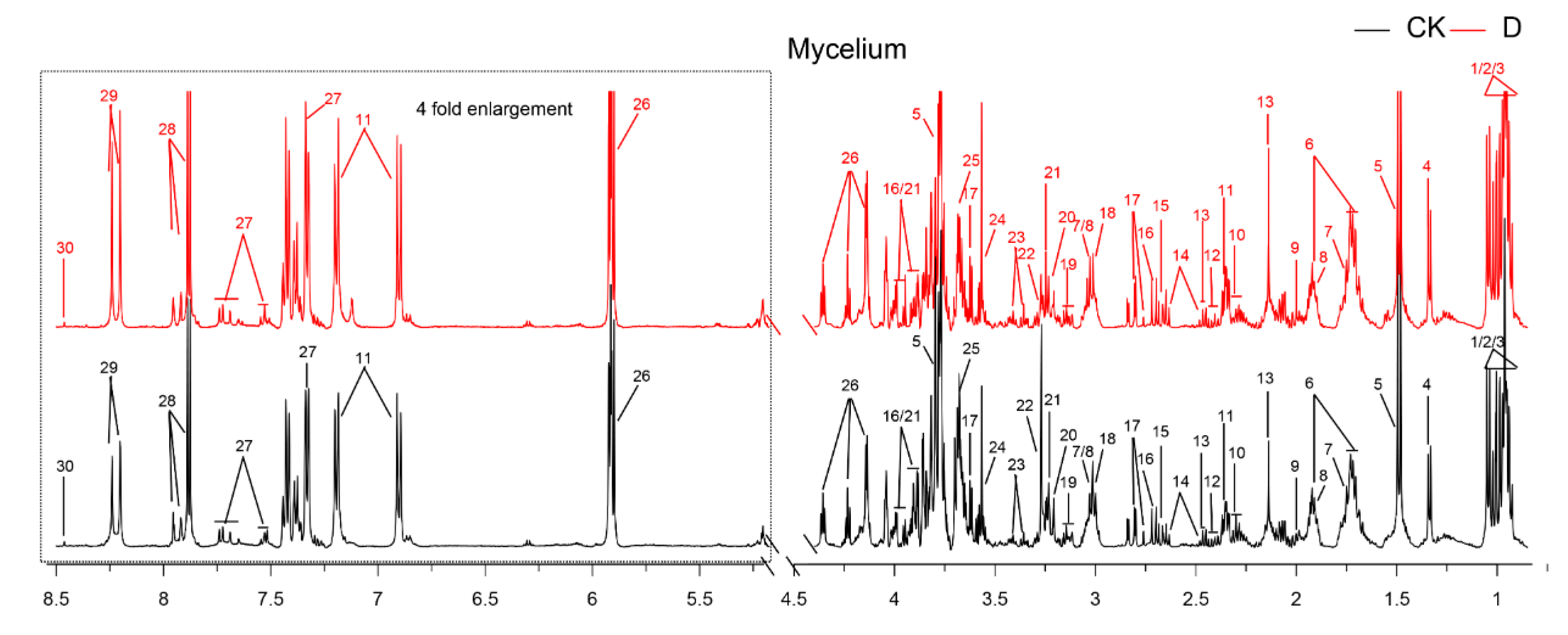
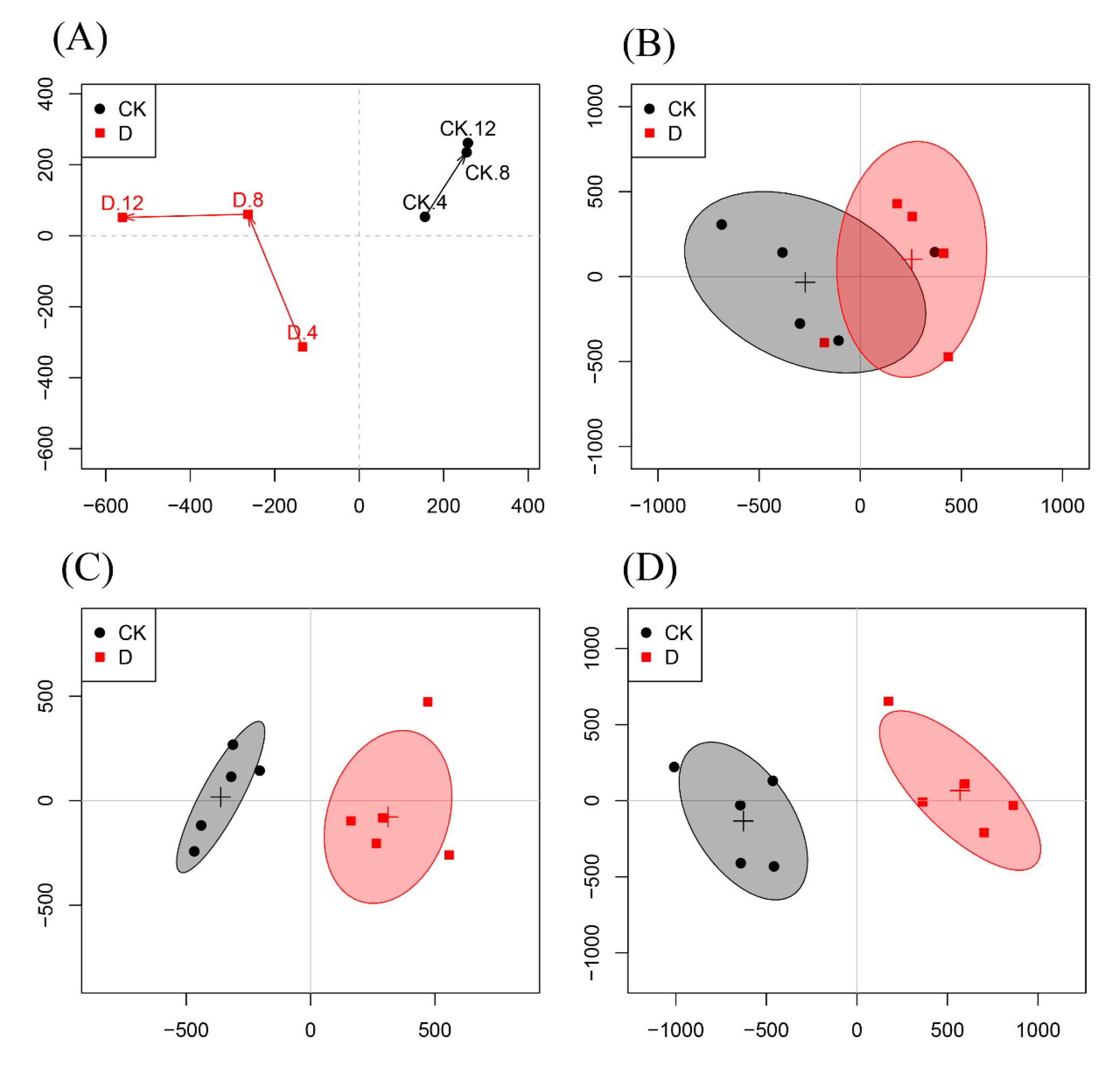
| Concentrations (mg/mL) | Mycelial Weight (g/100 mL) | Water-retention Rate (%) | |
|---|---|---|---|
| Wet Weight | Dry Weight | ||
| 0 | 4.868 ± 0.288 a | 0.419 ± 0.011 a | 91.38 ± 0.29 a |
| 0.03125 | 4.487 ± 0.185 a | 0.407 ± 0.008 a | 90.89 ± 0.33 a |
| 0.0625 | 3.736 ± 0.113 b | 0.393 ± 0.015 ab | 89.47 ± 0.48 b |
| 0.125 | 3.065 ± 0.267 c | 0.368 ± 0.016 bc | 87.69 ± 0.77 c |
| 0.25 | 2.895 ± 0.170 c | 0.357 ± 0.012 c | 87.57 ± 0.39 c |
| 0.5 | 2.671 ± 0.232 c | 0.345 ± 0.020 c | 86.96 ± 0.65 c |
| No | Metabolites | D/CK | |
|---|---|---|---|
| a FC | b P | ||
| 1 | Isoleucine | 0.04 | |
| 2 | Leucine | 0.5 | * |
| 3 | Valine | −0.11 | |
| 4 | Lactate | −0.52 | * |
| 5 | Alanine | −0.82 | ** |
| 6 | Lysine | 0.25 | |
| 7 | Putrescine | −0.18 | |
| 8 | 4-Aminobutyrate | −0.27 | |
| 9 | Acetate | 0.1 | |
| 10 | Glutamate | −0.41 | * |
| 11 | Acetaminophen | 0.04 | |
| 12 | Succinate | −0.26 | |
| 13 | Glutamine | −1.17 | *** |
| 14 | Glutathione | −0.65 | * |
| 15 | 5-6-Dihydrouracil | −0.3 | |
| 16 | Aspartate | −0.71 | ** |
| 17 | Sarcosine | −1.02 | ** |
| 18 | Phenylalanine | −1.65 | *** |
| 19 | Ethanolamine | −0.51 | * |
| 20 | Choline | −1.51 | *** |
| 21 | Betaine | 0.06 | |
| 22 | Arginine | −1.86 | *** |
| 23 | Methanol | −0.75 | ** |
| 24 | Glycine | −1.09 | ** |
| 25 | π-Methylhistidine | −1 | * |
| 26 | Uracil | 1.13 | ** |
| 27 | Tryptophan | 0.37 | * |
| 28 | Xanthine | −1.42 | *** |
| 29 | Adenine | 1.47 | *** |
| 30 | Formate | −0.07 | |
 . b P values corrected by Benjamini-Hochberg methods were calculated based on a parametric Student’s t-test or a nonparametric Mann-Whitney test (dependent on the conformity to the normal distribution). * p < 0.05, ** p < 0.01, *** p < 0.001.
. b P values corrected by Benjamini-Hochberg methods were calculated based on a parametric Student’s t-test or a nonparametric Mann-Whitney test (dependent on the conformity to the normal distribution). * p < 0.05, ** p < 0.01, *** p < 0.001.© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wan, C.; Shen, Y.; Nisar, M.F.; Qi, W.; Chen, C.; Chen, J. The Antifungal Potential of Carvacrol against Penicillium Digitatum through 1H-NMR Based Metabolomics Approach. Appl. Sci. 2019, 9, 2240. https://doi.org/10.3390/app9112240
Wan C, Shen Y, Nisar MF, Qi W, Chen C, Chen J. The Antifungal Potential of Carvacrol against Penicillium Digitatum through 1H-NMR Based Metabolomics Approach. Applied Sciences. 2019; 9(11):2240. https://doi.org/10.3390/app9112240
Chicago/Turabian StyleWan, Chunpeng, Yuting Shen, Muhammad Farrukh Nisar, Wenwen Qi, Chuying Chen, and Jinyin Chen. 2019. "The Antifungal Potential of Carvacrol against Penicillium Digitatum through 1H-NMR Based Metabolomics Approach" Applied Sciences 9, no. 11: 2240. https://doi.org/10.3390/app9112240
APA StyleWan, C., Shen, Y., Nisar, M. F., Qi, W., Chen, C., & Chen, J. (2019). The Antifungal Potential of Carvacrol against Penicillium Digitatum through 1H-NMR Based Metabolomics Approach. Applied Sciences, 9(11), 2240. https://doi.org/10.3390/app9112240






