Low-Field NMR and MRI to Analyze the Effect of Edible Coating Incorporated with MAP on Qualities of Half-Smooth Tongue Sole (Cynoglossus Semilaevis Günther) Fillets during Refrigerated Storage
Abstract
:Featured Application
Abstract
1. Introduction
2. Material and Methods
2.1. Preparation of Edible Coating Solution
2.2. Immersion Treatment of Fillets
2.3. MAP Treatment of Fillets
2.4. LF-NMR Analysis
2.5. MRI Analysis
2.6. Water Content
2.7. Fat Content
2.8. TVB-N Value
2.9. K Value
2.10. Ca2+-ATPase Activity
2.11. Malondialdehyde (MDA) Value
2.12. Sensory Scores
2.13. Statistical Analysis
3. Results and Discussion
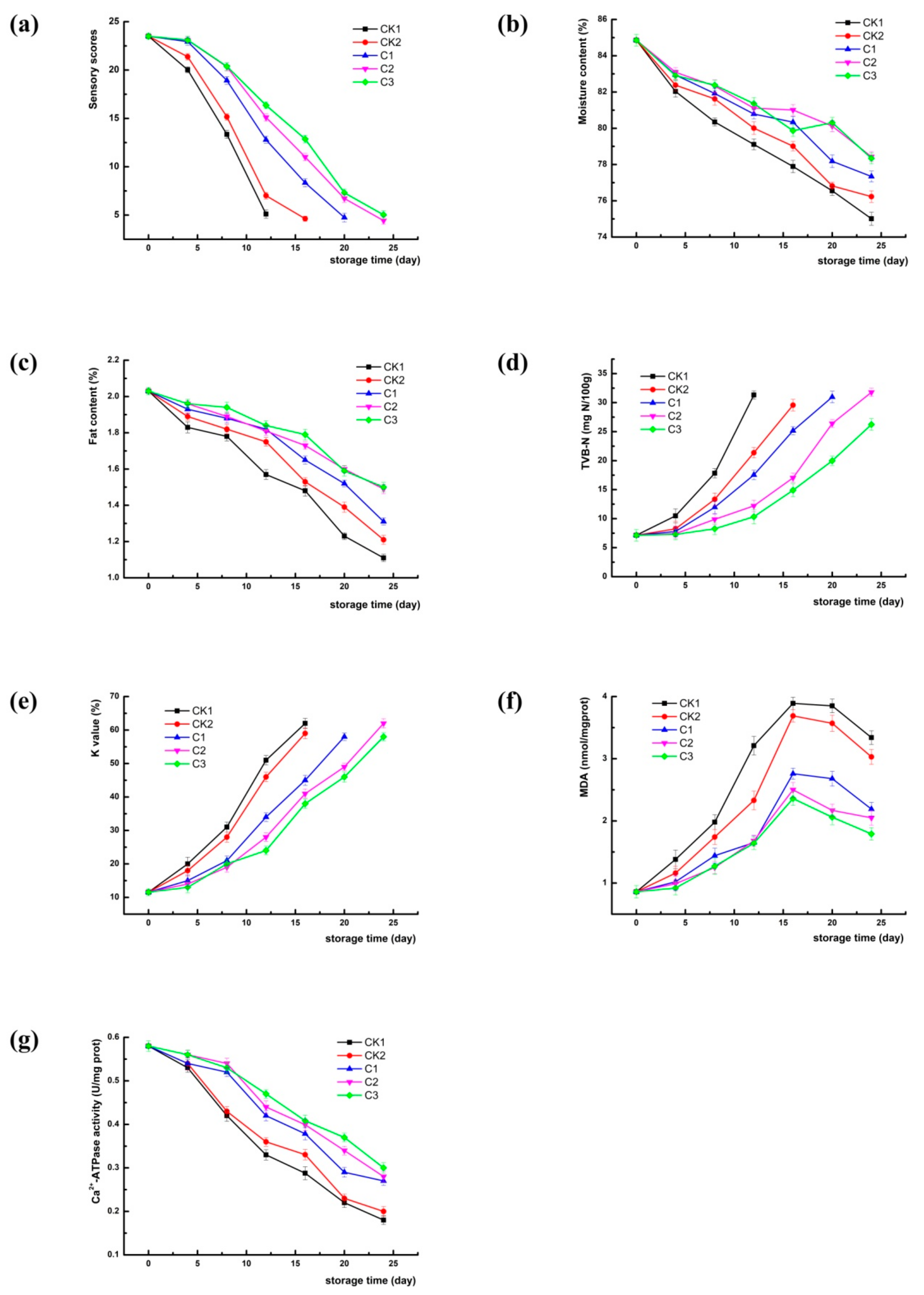
3.1. Quality Attributes
3.2. Measurements of LF-NMR
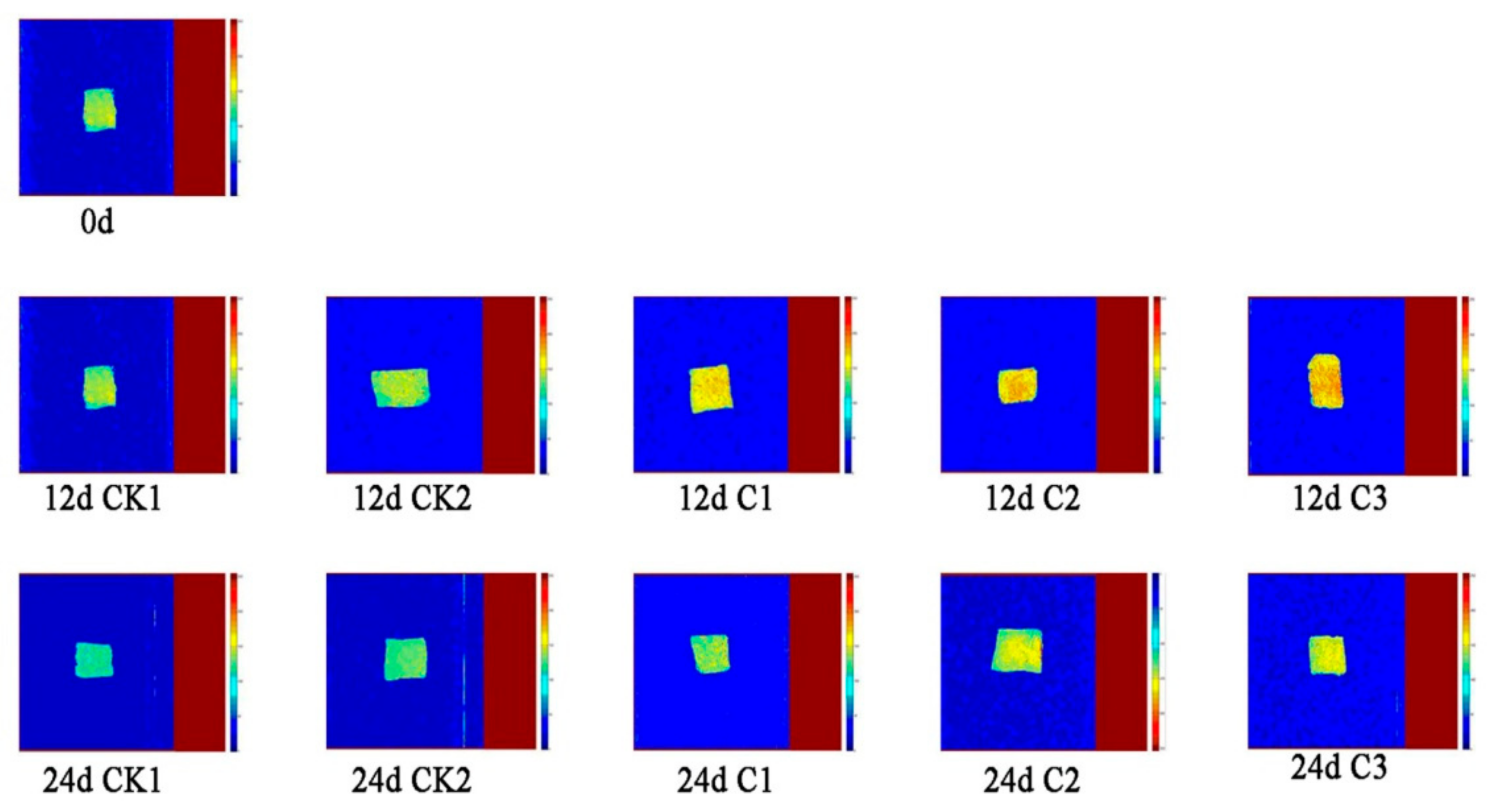
3.3. Analysis of MRI
3.4. Linear Regression Analysis
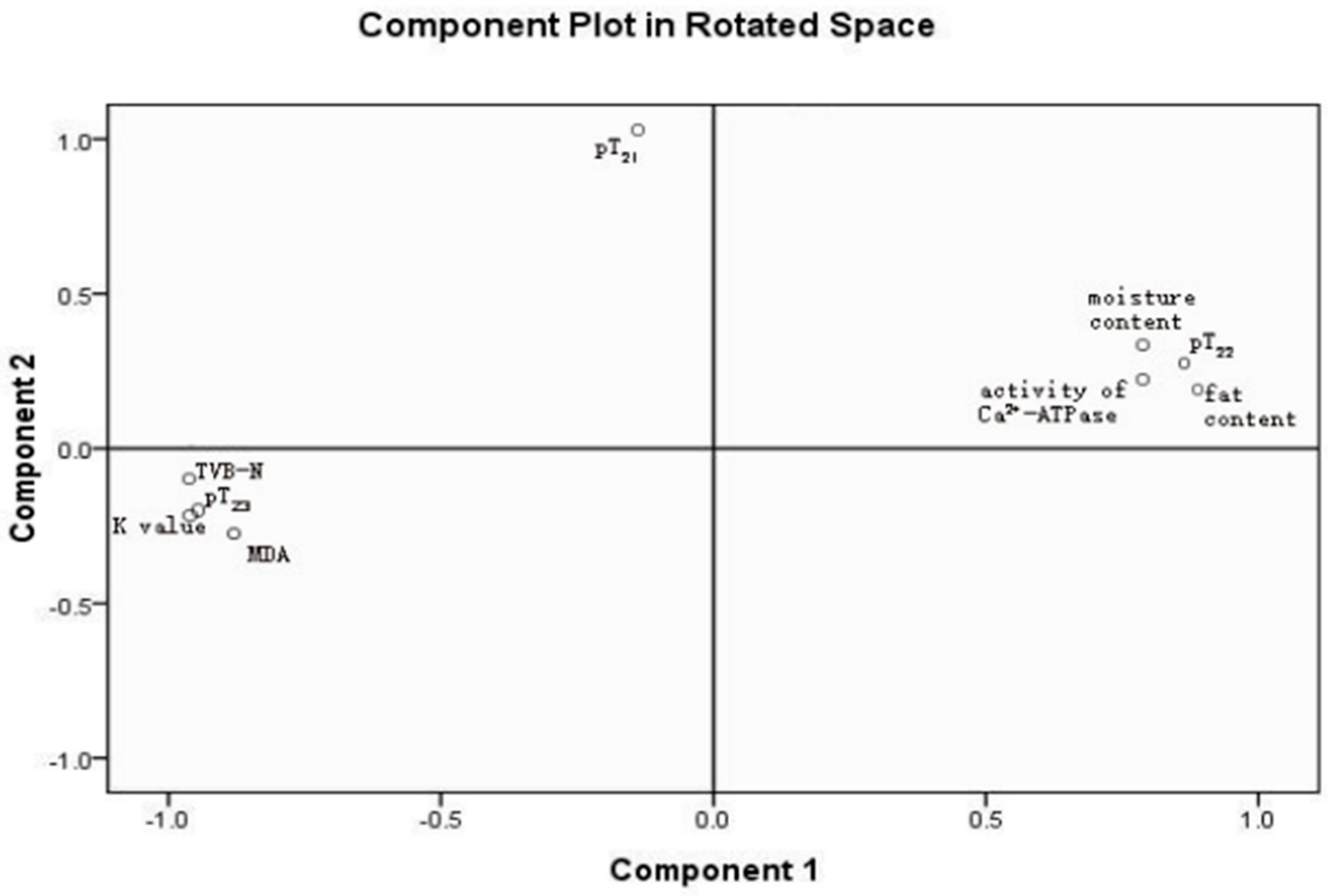
3.5. Analysis of PCA
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Yu, D.; Xu, Y.; Regenstein, J.M.; Xia, W.; Yang, F.; Jiang, Q.; Wang, B. The effects of edible chitosan-based coatings on flavor quality of raw grass carp (Ctenopharyngodon idellus) fillets during refrigerated storage. Food Chem. 2018, 242, 412–420. [Google Scholar] [CrossRef] [PubMed]
- Benbettaïeb, N.; Tanner, C.; Cayot, P.; Karbowiak, T.; Debeaufort, F. Impact of functional properties and release kinetics on antioxidant activity of biopolymer active films and coatings. Food Chem. 2017, 242, 369–377. [Google Scholar] [CrossRef] [PubMed]
- Dehghani, P.; Hosseini, S.M.H.; Golmakani, M.T.; Majdinasab, M.; Esteghlal, S. Shelf-life extension of refrigerated rainbow trout fillets using total farsi gum-based coatings containing clove and thyme essential oils emulsions. Food Hydrocoll. 2018, 77, 677–688. [Google Scholar] [CrossRef]
- Martínez, O.; Salmeron, J.; Epelde, L.; Vicente, M.S.; de Vega, C. Quality enhancement of smoked sea bass (Dicentrarchus labrax) fillets by adding resveratrol and coating with chitosan and alginate edible films. Food Control 2018, 85, 168–176. [Google Scholar] [CrossRef]
- Mizielińska, M.; Kowalska, U.; Jarosz, M.; Sumińska, P. A comparison of the effects of packaging containing nano ZnO or polylysine on the microbial purity and texture of cod (Gadus morhua) fillets. Nanomaterials 2018, 8, 158. [Google Scholar] [CrossRef] [PubMed]
- Hernández, E.J.G.P.; de Carvalho, R.N., Jr.; Joele, M.R.S.P.; Araújo, C.D.S.; Lourenço, L.F.H. Effects of modified atmosphere packing over the shelf life of sous vide, from captive pirarucu (Arapaima gigas). Innov. Food Sci. Emerg. Technol. 2017, 39, 94–100. [Google Scholar] [CrossRef]
- Kuuliala, L.; Al Hage, Y.; Ioannidis, A.G.; Sader, M.; Kerckhof, F.M.; Vanderroost, M.; Boon, N.; Baets, B.D.; Meulenaer, B.D.; Ragaert, P.; et al. Microbiological, chemical and sensory spoilage analysis of raw Atlantic cod (Gadus morhua) stored under modified atmospheres. Food Microbiol. 2018, 70, 232–244. [Google Scholar] [CrossRef] [PubMed]
- Odeyemi, O.A.; Burke, C.M.; Bolch, C.J.; Stanley, R. Evaluation of spoilage potential and volatile metabolites production by Shewanella baltica isolated from modified atmosphere packaged live mussels. Food Res. Int. 2018, 103, 415–425. [Google Scholar] [CrossRef] [PubMed]
- Zang, X.; Lin, Z.; Zhang, T.; Wang, H.; Cong, S.; Song, Y.; Li, Y.; Cheng, S. Nondestructive measurement of water and fat contents, water dynamics during drying and adulteration detection of intact small yellow croaker by low field NMR. J. Food Meas. Charact. 2017, 11, 1550–1558. [Google Scholar] [CrossRef]
- Sánchez-Alonso, I.; Martinez, I.; Sánchez-Valencia, J.; Careche, M. Estimation of freezing storage time and quality changes in hake (Merluccius merluccius L.) by low field NMR. Food Chem. 2012, 135, 1626–1634. [Google Scholar] [CrossRef] [PubMed]
- Huang, L.; Song, Y.; Kamal, T.; Li, Y.; Xia, K.; Lin, Z.; Qi, L.; Cheng, S.; Zhu, B.; Tan, M. A non-invasive method based on low-field NMR to analyze the quality changes in caviar from hybrid sturgeon (Huso dauricus, Acipenser schrenckiid). J. Food Process. Pres. 2017, 41, e13256. [Google Scholar] [CrossRef]
- Patel, K.K.; Khan, M.A.; Kar, A. Recent developments in applications of MRI techniques for foods and agricultural produce—An overview. J. Food Sci. Technol. MYS 2015, 52, 1–26. [Google Scholar] [CrossRef]
- Albertos, I.; Martin-Diana, A.B.; Cullen, P.J.; Tiwari, B.K.; Ojha, S.K.; Bourke, P.; Rico, D. Shelf-life extension of herring (Clupea harengus) using in-package atmospheric plasma technology. Innov. Food Sci. Emerg. Technol. 2017. [Google Scholar] [CrossRef]
- Sun, X.H.; Xiao, L.; Lan, W.Q.; Liu, S.C.; Wang, Q.; Yang, X.H.; Zhang, W.J.; Xie, J. Effects of temperature fluctuation on quality changes of large yellow croaker (Pseudosciaena crocea) with ice storage during logistics process. J. Food Process. Pres. 2018, 42, e13505. [Google Scholar] [CrossRef]
- Su, H.; Wei, C.; Fu, S.; Chu, W.; Kai, L.; Huang, Z.; Li, J. Antimicrobial effect of bayberry leaf extract forthe preservation of large yellow croaker (Pseudosciaena crocea). J. Sci. Food Agric. 2013, 94, 935–942. [Google Scholar] [CrossRef] [PubMed]
- Soares, N.; Silva, P.; Barbosa, C.; Pinheiro, R.; Vicente, A.A. Comparing the effects of glazing and chitosan-based coating applied on frozen salmon on its organoleptic and physicochemical characteristics over six-months storage. J. Food Eng. 2017, 194, 79–86. [Google Scholar] [CrossRef]
- Wang, S.; Xiang, W.; Fan, H.; Xie, J.; Qian, Y.F. Study on the mobility of water and its correlation with the spoilage process of salmon (Salmo solar) stored at 0 and 4 °C by low-field nuclear magnetic resonance (LF NMR 1H). J. Food Sci. Technol. MYS 2018, 55, 173–182. [Google Scholar] [CrossRef] [PubMed]
- Gonçalves, A.A.; Santos, T.C.L. The effects of vacuum and modified atmosphere packaging on quality changes in seasoned cobia (Rachycentron canadum) sticks stored under refrigeration. Braz. J. Food Technol. 2018, 21, e2017029. [Google Scholar] [CrossRef]
- Rahman, A.; Kondo, N.; Ogawa, Y.; Suzuki, T.; Shirataki, Y.; Wakita, Y. Prediction of K value for fish flesh based on ultraviolet–visible spectroscopy of fish eye fluid using partial least squares regression. Comput. Electron. Agric. 2015, 117, 149–153. [Google Scholar] [CrossRef]
- Li, X.P.; Zhou, M.Y.; Liu, J.F.; Xu, Y.X.; Mi, H.B.; Yi, S.M.; Li, J.R.; Lin, H. Shelf-life extension of chilled olive flounder (Paralichthys olivaceus) using chitosan coatings containing clove oil. J. Food Process. Pres. 2017, 41, e13204. [Google Scholar] [CrossRef]
- Li, Q.; Zhang, L.; Lu, H.; Song, S.; Luo, Y. Comparison of postmortem changes in ATP-related compounds, protein degradation and endogenous enzyme activity of white muscle and dark muscle from common carp (Cyprinus carpio) stored at 4 °C. LWT Food Sci. Technol. 2017, 78, 317–324. [Google Scholar] [CrossRef]
- Ozogul, Y.; Balikci, E. Effect of various processing methods on quality of mackerel (Scomber scombrus). Food Bioprocess Technol. 2013, 6, 1091–1098. [Google Scholar] [CrossRef]
- Shariat, M.; Raftari, M.; Bakar, F.A. Evaluation of sensory and biochemical changes in fresh water catfish stored under vacuum and different modified atmospheres. Anal. Methods 2013, 5, 231–238. [Google Scholar] [CrossRef]
- Masniyom, P.; Benjakul, S.; Visessanguan, W. ATPase activity, surface hydrophobicity, sulfhydryl content and protein degradation in refrigerated seabass muscle in modified atmosphere packaging. J. Food Biochem. 2004, 28, 43–60. [Google Scholar] [CrossRef]
- Reza, M.S.; Bapary, M.A.; Ahasan, C.T.; Islam, M.N.; Kamal, M. Shelf life of several marine fish species of Bangladesh during ice storage. Int. J. Food Sci. Technol. 2009, 44, 1485–1494. [Google Scholar] [CrossRef]
- Wang, S.; Lin, Z.; Xia, K.; Li, Y.; Tan, M. Dynamics of water mobility and distribution in Sur clam (Mactra chinensis) during dehydration and rehydration processes assessed by low-field NMR and MRI. J. Food Meas. Charact. 2017, 11, 1342–1354. [Google Scholar] [CrossRef]
- Qin, N.; Zhang, L.; Zhang, J.; Song, S.; Wang, Z.; Regenstein, J.M.; Luo, Y. Influence of lightly salting and sugaring on the quality and water distribution of grass carp (Ctenopharyngodon idellus) during super-chilled storage. J. Food Eng. 2017, 215, 104–112. [Google Scholar] [CrossRef]
- Li, H.; Lin, B.; Hong, Y.; Liu, T.; Huang, Z.; Wang, R.; Wang, Z. Assessing the moisture migration during microwave drying of coal using low-field nuclear magnetic resonance. Drying Technol. 2018, 36, 567–577. [Google Scholar] [CrossRef]
- Brown, R.J.; Capozzi, F.; Cavani, C.; Cremonini, M.A.; Petracci, M.; Placucci, G. Relationships between (1)H NMR relaxation data and some technological parameters of meat: A chemometric approach. J. Magn. Reson. 2000, 147, 89–94. [Google Scholar] [CrossRef] [PubMed]
- Li, T.; Tu, C.; Rui, X.; Gao, Y.; Li, W.; Wang, K.; Xiao, Y.; Dong, M. Study of water dynamics in the soaking, steaming and solid-state fermentation of glutinous rice by LF-NMR: A novel monitoring approach. J. Agric. Food Chem. 2015, 63, 3261–3270. [Google Scholar] [CrossRef] [PubMed]
- Huang, L.; Cheng, S.; Song, Y.; Xia, K.; Xu, X.; Zhu, B.W.; Tan, M. Nondestructive analysis of caviar compositions using low-field nuclear magnetic resonance technique. J. Food Meas. Charact. 2016, 11, 621–628. [Google Scholar] [CrossRef]
- Xia, K.; Xu, W.; Huang, L.; Song, Y.; Zhu, B.W.; Tan, M. Water dynamics of turbot flesh during frying, boiling, and stewing processes and its relationship with color and texture properties: Low-field NMR and MRI studies. J. Food Process. Pres. 2018, 42, e13338. [Google Scholar] [CrossRef]
- Li, J.; Xia, K.; Li, Y.; Tan, M. Influence of freezing-thawing cycle on water dynamics of turbot flesh assessed by low-field nuclear magnetic resonance and magnetic resonance imaging. Int. J. Food Eng. 2018, 14, 14. [Google Scholar] [CrossRef]
- Li, M.; Li, B.; Zhang, W. Rapid and non-invasive detection and imaging of the hydrocolloid-injected prawns with low-field NMR and MRI. Food Chem. 2018, 242, 16–21. [Google Scholar] [CrossRef] [PubMed]
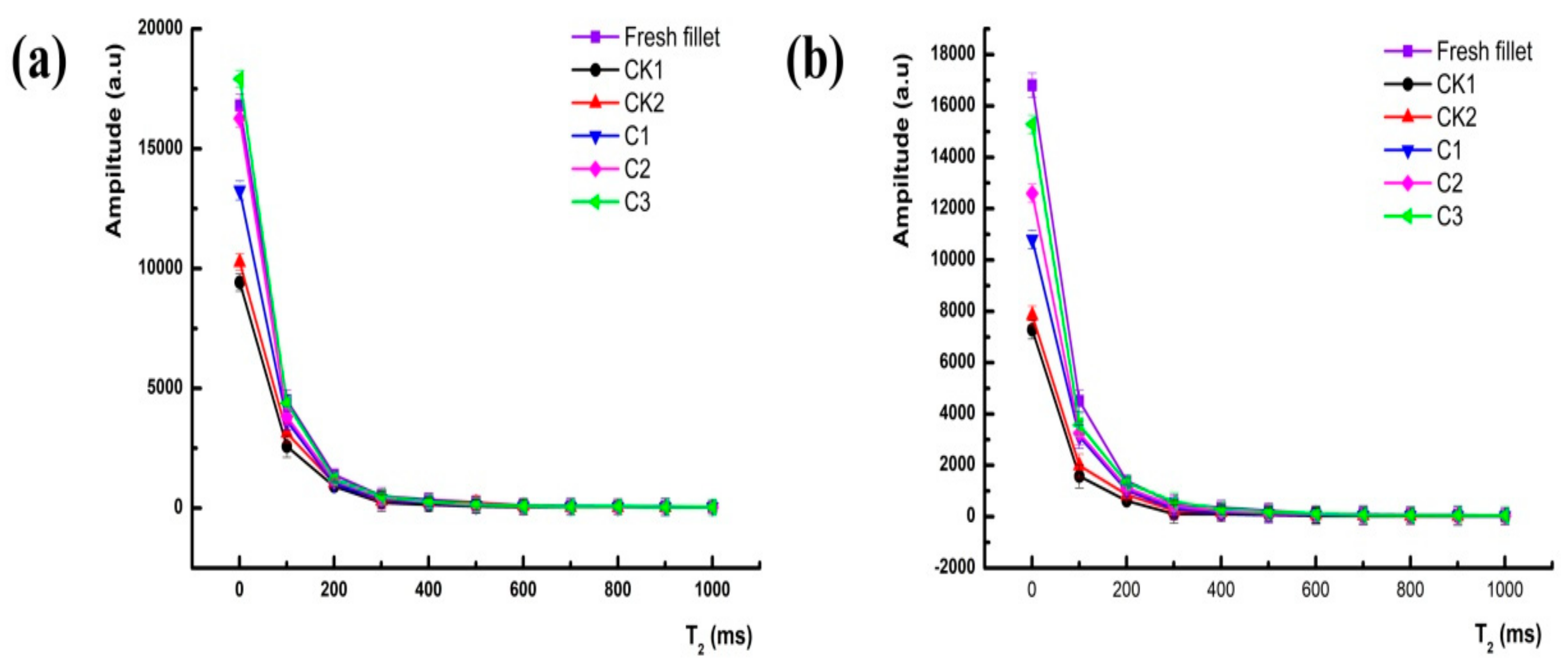
| Storage Time (Day) | T21 (ms) | T22 (ms) | T23 (ms) | pT21 | pT22 | pT23 |
|---|---|---|---|---|---|---|
| 0d | 0.079 ± 0.002 | 75.65 ± 2.79 | 1232.84 ± 5.4 | 1.96 ± 0.13 | 87.63 ± 3.26 | 10.41 ± 0.56 |
| 12d | ||||||
| CK1 | 0.15 ± 0.02 | 79.38 ± 1.84 | 932.60 ± 3.82 | 1.78 ± 0.21 | 77.83 ± 2.18 | 20.39 ± 0.93 |
| CK2 | 0.064 ± 0.003 | 65.79 ± 3.27 | 717.13 ± 4.92 | 1.74 ± 0.09 | 79.19 ± 2.84 | 19.12 ± 1.13 |
| C1 | 0.166 ± 0.046 | 75.83 ± 2.64 | 705.48 ± 4.04 | 1.84 ± 0.34 | 81.23 ± 3.16 | 16.93 ± 0.92 |
| C2 | 0.194 ± 0.032 | 69.79 ± 2.18 | 533.67 ± 1.84 | 1.83 ± 0.22 | 84.98 ± 2.21 | 13.19 ± 1.26 |
| C3 | 0.141 ± 0.027 | 73.93 ± 2.35 | 642.84 ± 3.15 | 1.74 ± 0.13 | 85.13 ± 2.19 | 13.11 ± 1.17 |
| 24d | ||||||
| CK1 | 0.15 ± 0.026 | 65.79 ± 4.19 | 1072.27 ± 4.2 | 1.84 ± 0.08 | 69.29 ± 3.17 | 28.87 ± 1.04 |
| CK2 | 0.132 ± 0.025 | 73.19 ± 2.83 | 1232.85 ± 6.8 | 1.86 ± 0.13 | 70.54 ± 2.56 | 27.6 ± 1.22 |
| C1 | 0.116 ± 0.011 | 86.97 ± 3.62 | 1417.48 ± 3.2 | 1.79 ± 0.36 | 73.19 ± 3.9 | 25.02 ± 1.47 |
| C2 | 0.102 ± 0.027 | 68.59 ± 2.91 | 1069.57 ± 5.6 | 1.85 ± 0.32 | 79.92 ± 2.89 | 18.23 ± 1.02 |
| C3 | 0.107 ± 0.034 | 74.27 ± 3.15 | 1243.38 ± 3.9 | 1.92 ± 0.24 | 80.18 ± 3.04 | 17.9 ± 0.93 |
| Groups | T2 | TVB-N | K Value | Moisture Content | Fat Content | Activity of Ca2+-ATPase | MDA | |
|---|---|---|---|---|---|---|---|---|
| CK1 | pT21 | R | −0.116 | −0.252 | 0.376 | 0.257 | 0.334 | −0.356 |
| R2 | 0.01346 | 0.06351 | 0.14138 | 0.02146 | 0.111556 | 0.12674 | ||
| p | ns | ns | ns | ns | ns | ns | ||
| pT22 | R | 0.95119 | 0.29514 | 0.92732 | 0.98812 | 0.98214 | 0.99312 | |
| R2 | 0.90476 | 0.08711 | 0.85992 | 0.97638 | 0.9646 | 0.98629 | ||
| p | 0.05 | ns | 0.01 | 0.01 | 0.01 | 0.05 | ||
| pT23 | R | 0.86271 | 0.94352 | 0.84978 | 0.22136 | 0.94675 | 0.94978 | |
| R2 | 0.74427 | 0.89023 | 0.72213 | 0.0449 | 0.89634 | 0.90208 | ||
| p | 0.01 | 0.01 | 0.05 | ns | 0.05 | 0.01 | ||
| CK2 | pT21 | R | 0.56437 | 0.34126 | 0.39643 | 0.23168 | 0.41154 | 0.28746 |
| R2 | 0.31851 | 0.11646 | 0.15716 | 0.05367 | 0.16937 | 0.08263 | ||
| p | ns | ns | ns | ns | ns | ns | ||
| pT22 | R | 0.38324 | 0.29435 | 0.93293 | 0.97317 | 0.97224 | 0.89146 | |
| R2 | 0.3705 | 0.08644 | 0.87036 | 0.94706 | 0.94525 | 0.7947 | ||
| p | ns | ns | 0.01 | 0.01 | 0.01 | 0.05 | ||
| pT23 | R | 0.90213 | 0.97568 | 0.82514 | 0.28246 | 0.87412 | 0.93562 | |
| R2 | 0.81384 | 0.95195 | 0.68086 | 0.07978 | 0.76173 | 0.87539 | ||
| p | 0.01 | 0.01 | 0.05 | ns | 0.05 | 0.01 | ||
| C1 | pT21 | R | 0.54873 | 0.28534 | 0.42674 | 0.32143 | 0.37892 | 0.29836 |
| R2 | 0.30111 | 0.08142 | 0.18211 | 0.10332 | 0.14358 | 0.08902 | ||
| p | ns | ns | ns | ns | ns | ns | ||
| pT22 | R | 0.88412 | 0.38423 | 0.96612 | 0.97412 | 0.97733 | 0.88911 | |
| R2 | 0.78167 | 0.14763 | 0.93339 | 0.94628 | 0.94731 | 0.79052 | ||
| p | 0.05 | ns | 0.01 | 0.01 | 0.01 | 0.05 | ||
| pT23 | R | 0.93215 | 0.97346 | 0.79354 | 0.58421 | 0.74278 | 0.96542 | |
| R2 | 0.8689 | 0.94762 | 0.62971 | 0.3413 | 0.55172 | 0.93204 | ||
| p | 0.01 | 0.01 | 0.05 | ns | 0.05 | 0.01 | ||
| C2 | pT21 | R | 0.32145 | 0.31238 | 0.50156 | 0.16973 | 0.45672 | 0.37829 |
| R2 | 0.10333 | 0.0969 | 0.25156 | 0.02881 | 0.20859 | 0.1431 | ||
| p | ns | ns | ns | ns | ns | ns | ||
| pT22 | R | 0.82356 | 0.49324 | 0.94582 | 0.96374 | 0.91214 | 0.62569 | |
| R2 | 0.67825 | 0.24329 | 0.89458 | 0.9288 | 0.832 | 0.39149 | ||
| p | 0.05 | ns | 0.01 | 0.01 | 0.01 | ns | ||
| pT23 | R | 0.96739 | 0.95678 | 0.94718 | 0.50047 | 0.81248 | 0.45768 | |
| R2 | 0.93584 | 0.91543 | 0.89715 | 0.25047 | 0.66012 | 0.20947 | ||
| p | 0.01 | 0.01 | 0.05 | ns | 0.05 | ns | ||
| C3 | pT21 | R | 0.45362 | 0.37288 | 0.51118 | 0.19463 | 0.47284 | 0.32145 |
| R2 | 0.20577 | 0.13904 | 0.26131 | 0.03789 | 0.22358 | 0.10333 | ||
| p | ns | ns | ns | ns | ns | ns | ||
| pT22 | R | 0.73246 | 0.30123 | 0.94378 | 0.94178 | 0.89236 | 0.64113 | |
| R2 | 0.53649 | 0.09074 | 0.89072 | 0.88695 | 0.79631 | 0.41105 | ||
| p | 0.05 | ns | 0.01 | 0.01 | 0.01 | 0.05 | ||
| pT23 | R | 0.98275 | 0.95467 | 0.79932 | 0.5101 | 0.80213 | 0.95467 | |
| R2 | 0.9658 | 0.9114 | 0.79389 | 0.2602 | 0.64341 | 0.9114 | ||
| p | 0.01 | 0.01 | 0.05 | ns | 0.05 | 0.01 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, N.; Shen, Y.; Liu, W.; Mei, J.; Xie, J. Low-Field NMR and MRI to Analyze the Effect of Edible Coating Incorporated with MAP on Qualities of Half-Smooth Tongue Sole (Cynoglossus Semilaevis Günther) Fillets during Refrigerated Storage. Appl. Sci. 2018, 8, 1391. https://doi.org/10.3390/app8081391
Li N, Shen Y, Liu W, Mei J, Xie J. Low-Field NMR and MRI to Analyze the Effect of Edible Coating Incorporated with MAP on Qualities of Half-Smooth Tongue Sole (Cynoglossus Semilaevis Günther) Fillets during Refrigerated Storage. Applied Sciences. 2018; 8(8):1391. https://doi.org/10.3390/app8081391
Chicago/Turabian StyleLi, Na, Yong Shen, Wenru Liu, Jun Mei, and Jing Xie. 2018. "Low-Field NMR and MRI to Analyze the Effect of Edible Coating Incorporated with MAP on Qualities of Half-Smooth Tongue Sole (Cynoglossus Semilaevis Günther) Fillets during Refrigerated Storage" Applied Sciences 8, no. 8: 1391. https://doi.org/10.3390/app8081391







