A Review of the Developed New Model Biodiesels and Their Effects on Engine Combustion and Emissions
Abstract
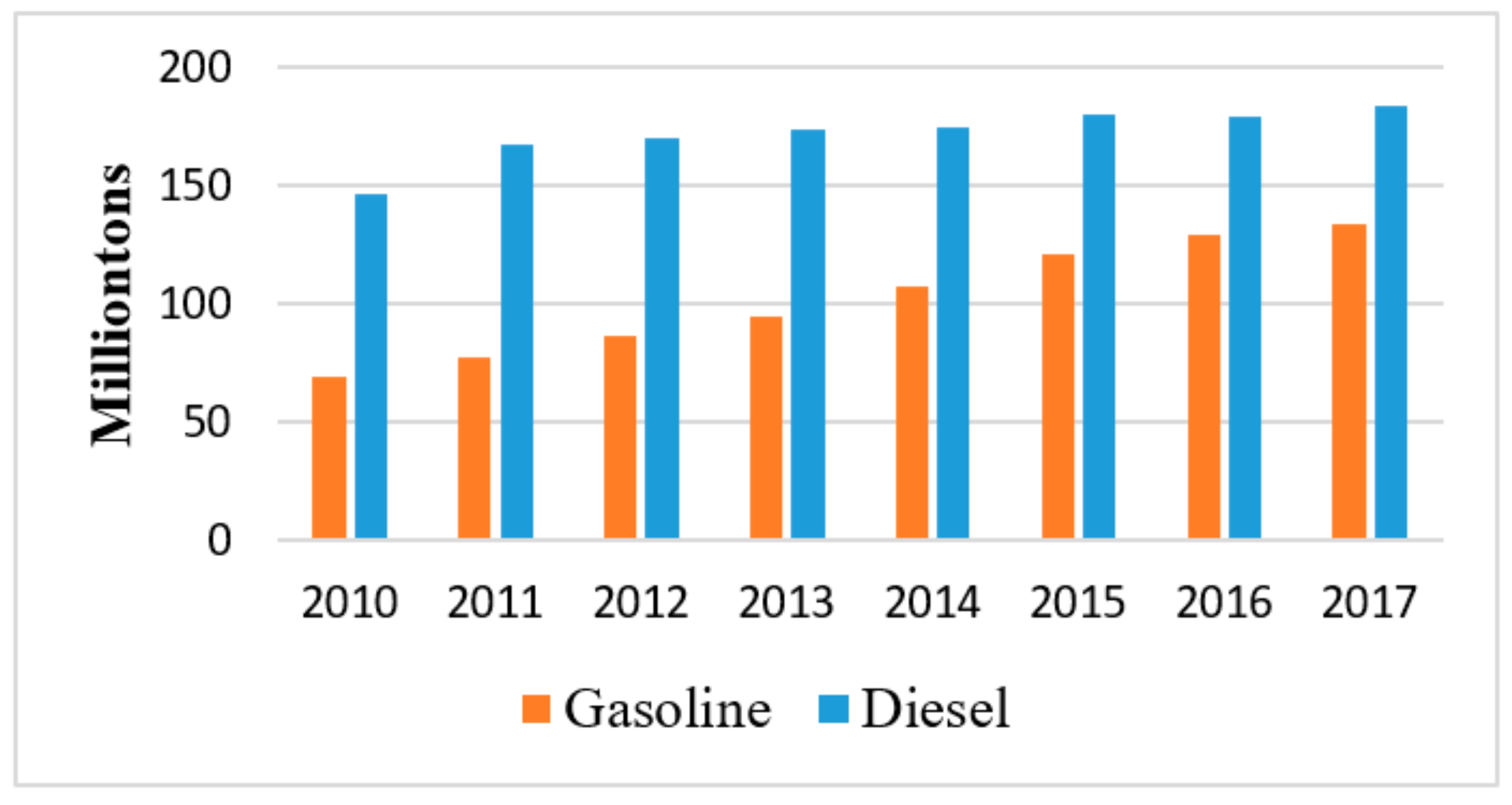
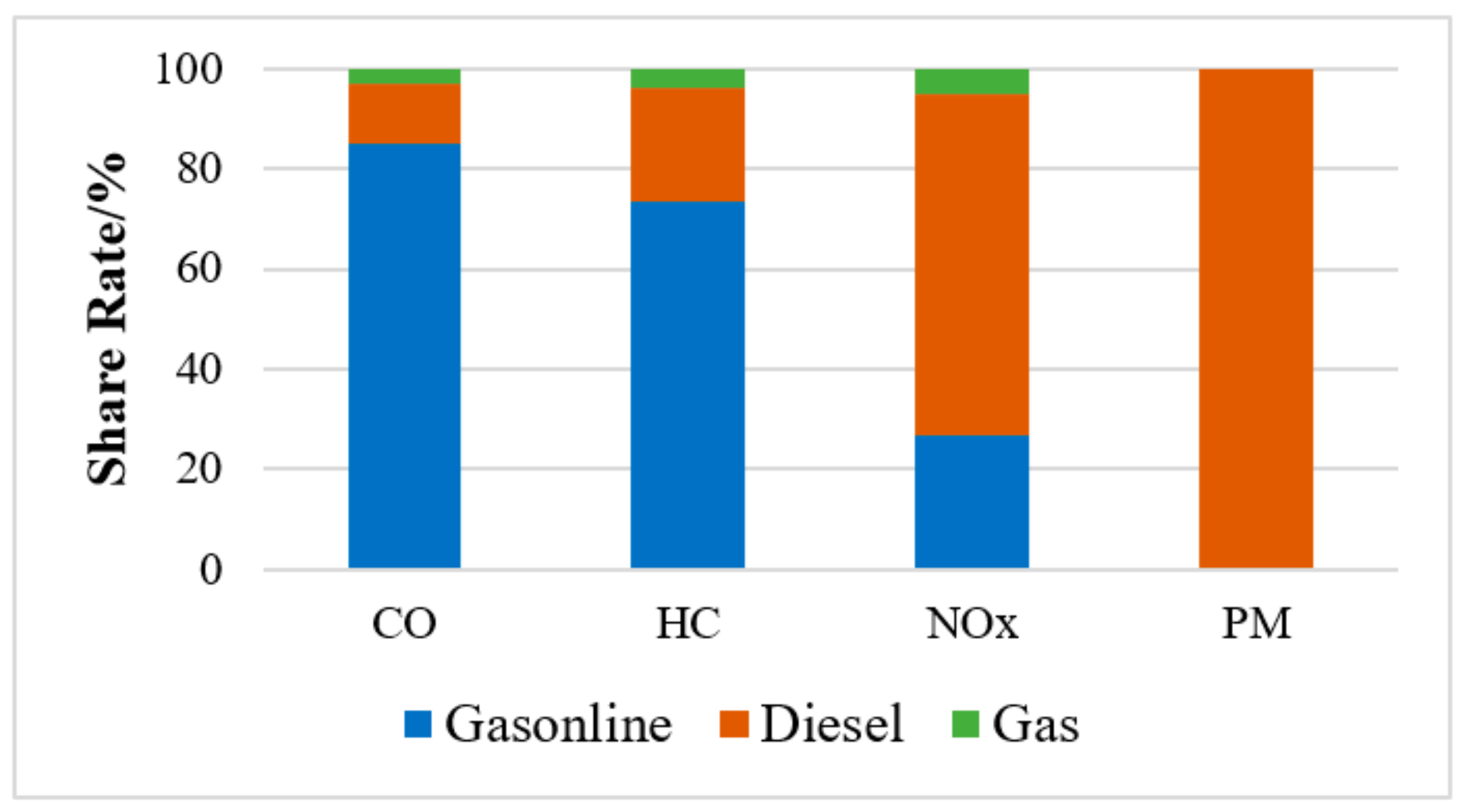
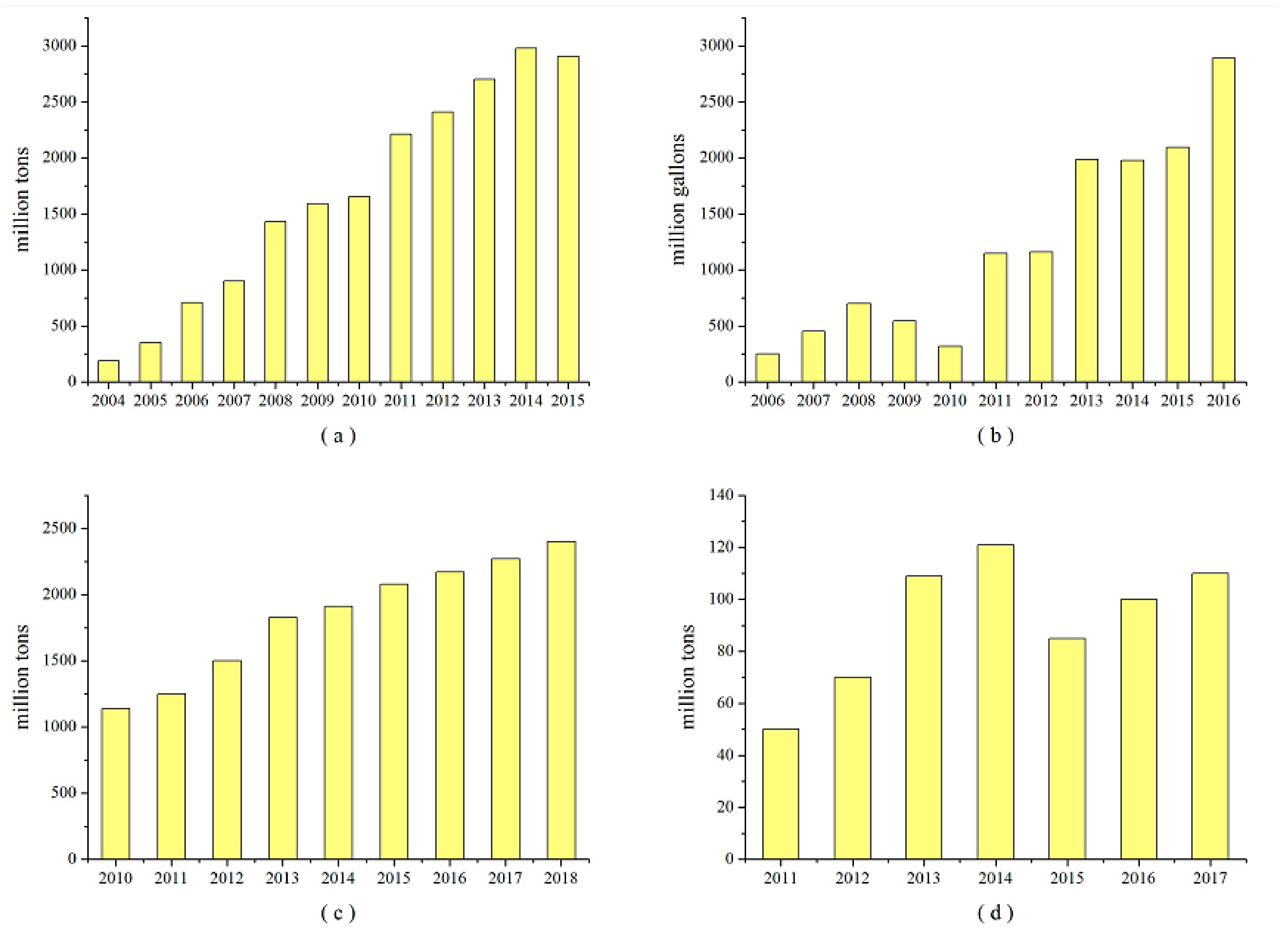
1. Introduction
- It is renewable, safe and biodegradable. Biodiesel can be made from vegetable oils, animal fats, or recycled restaurant greases (waste cooking oils). Both the original materials and the product of biodiesel are safe and can be degraded naturally.
- It can reduce greenhouse gas emissions. A life cycle analysis of biodiesel showed that overall CO2 emissions were reduced by 78% compared with petrodiesel fuel, which will contribute to domestic and international targets of greenhouse gas reductions.
- It has a similar cetane number as compared to petrodiesel, so it can be used alone, or blended with petrodiesel in any proportions.
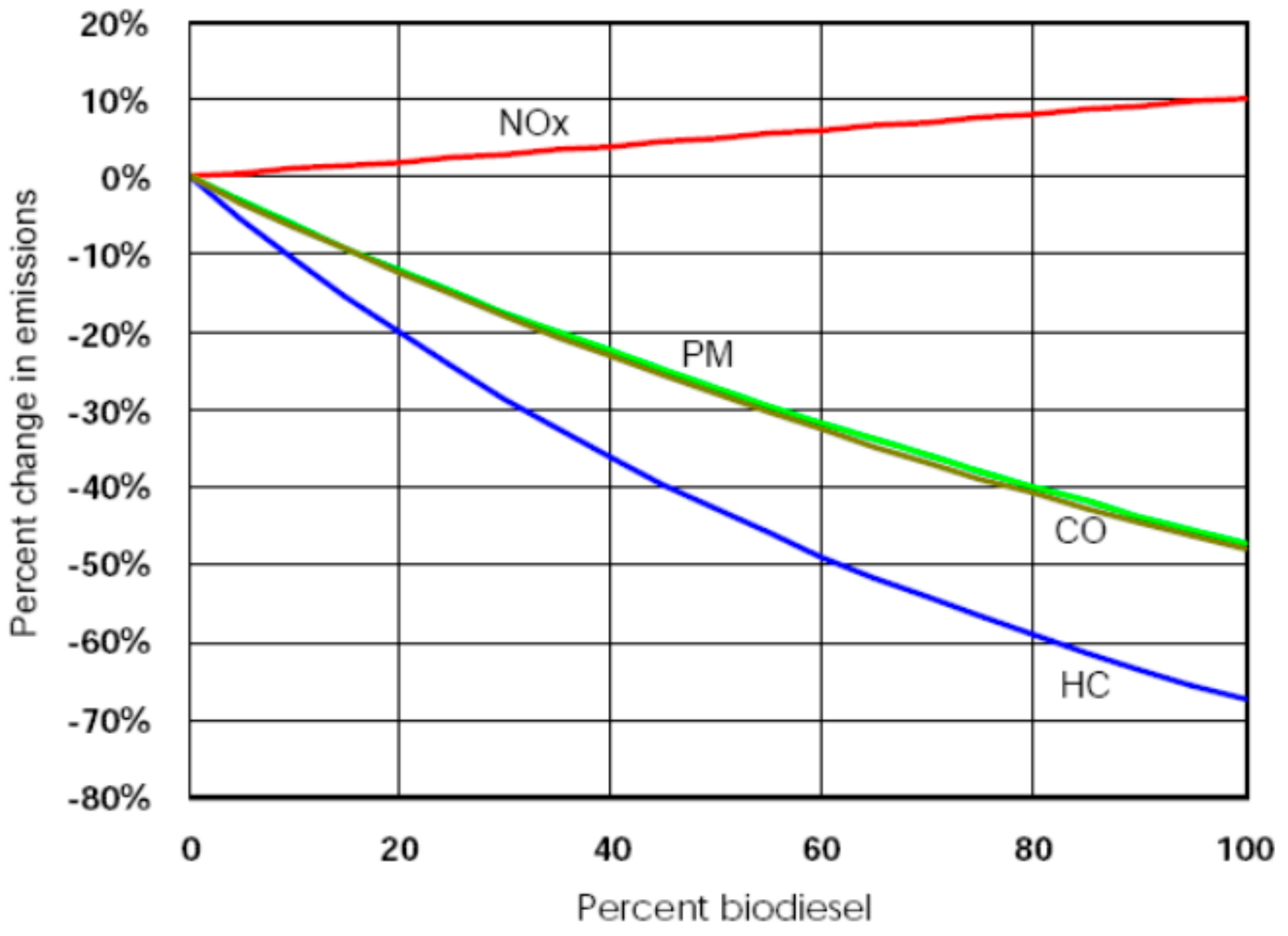
- It is an oxygenated fuel without sulfur content, which can promote engine combustion and reduce emissions of HC, CO and PM, which helps to reduce the environmental pollution.
- It has better lubricity, and unsaturated esters possess a slight advantage. So, there is no need to reconsider the lubricity of the engine fuel injection system when biodiesel is applied.
- It is a mix of mono-alkyl esters of long chain fatty acids. It has a higher cloud and pour point (CP and PP) temperatures, density, and kinematic viscosity as well as the acid value compared to diesel, affecting the utility of the fuel, especially in cold conditions.
- The calorific value of biodiesel is about 37.27 MJ/kg. There is 9% lower than regular petrodiesel. Fuel injection systems measure fuel by volume, and thus, engine output power may be affected under high ratio or pure application conditions.
- It comprises of saturated and unsaturated esters. Saturated fatty esters are very stable, while unsaturated esters are likely to react with oxygen, therefore reduce oxidation stability.
- And finally, because of its renewable feature, the use of biodiesel can reduce its dependence on foreign fossil fuels. Sustainable energy supply is necessary for the economic development of a country like China, whose petroleum depends much on import.
2. New Model Biodiesels and Their Fuel Properties
2.1. New Model Biodiesels
2.2. Biodiesel Fuel Properties
3. Test Engine and Parameter Measurement
4. Engine Test Results
4.1. FAME
4.2. Ethylene Glycol Monomethyl Ether Based Biodiesel
- 1.
- Effect on Combustion
- Effects on emissions
- 2.
- Effects on energy consumption
- Effects on emissions
- 3.
- Ethylene Glycol Methyl Ether Peanut Oil Monoester [32].
- Effect on Combustion
- Effects on emissions
- 4.
- 5.
- Ethylene Glycol Methyl Ether Cottonseed Oil Monoester [42].
4.3. Other Models of New Biodiesels
- 1.
- Ethylene glycol ethyl ether Soyate oil monoester
- 2.
- Ethylene glycol ethyl ether palm oil monoester
- 3.
- Ethylene Glycol n-Propyl Ether Palm Oil Monoester [45].
- 4.
- Propylene glycol methyl ether palm oil monoester [44].
- 5.
- DGMECOM [46].
- 6.
- TGMECOM [47].
5. Conclusions and Discussions
5.1. Ether Group Has Significant Effects on Fuel Properties
5.2. Ether Group Has Significant Effects on Engine Combustion and Emissions
5.3. One Methyl Ether Group Is Important
5.4. Biodiesel Combustion Chemical Kinetics Should Be Studied
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| AT | Autoignition Temperature/°C |
| BT | Boiling Temperature/°C |
| CA | Crank angle/(o) |
| CO | Carbon monoxide |
| CO2 | Carbon dioxide |
| CN | Cetane number |
| CP | Cold Filter Plugging Point/°C |
| OS | Oxidative Stability |
| DOE | Department of Energy, US |
| DU | Degree of unsaturation |
| EGME | Ethylene Glycol Monoethyl Ether |
| FAME | Fatty acid methyl ester |
| EGMEROM | Ethylene Glycol Methyl Ether Rapeseed Oil Monoester |
| EGMESOM | Ethylene Glycol Methyl Ether Soyate Oil Monoester |
| EGMEPOM (Peanut) | Ethylene Glycol Methyl Ether Peanut Oil Monoester |
| EGMEPOM (Palm) | Ethylene Glycol Methyl Ether Palm Oil Monoester |
| EGMECOM | Ethylene Glycol Methyl Ether Cottonseed Oil Monoester |
| EGEESOM | Ethylene glycol ethyl ether Soyate oil monoester |
| EGEEPOM | Ethylene glycol ethyl ether palm oil monoester |
| EGnPEPOM | Ethylene Glycol n-Propyl Ether Palm Oil Monoester |
| PGMEPOM | Propylene glycol methyl ether palm oil monoester |
| DGMECOM | Di-ethylene Glycol Methyl Ether Cottonseed Oil Monoester |
| TGMECOM | Tri-ethylene Glycol Methyl Ether Cottonseed Oil Monoester |
| FTIR | Fourier Transform Infrared Spectroscopy |
| HC | Hydrocarbons |
| HCI | Hydrochloric acid |
| LHV | Lower Heating Value/(KJ/Kg) |
| K | Kalium; potassium |
| Na | Sodium |
| NOx | Nitrogen oxides |
| OC | Oxygen Content/% |
| PM | Particulate matters |
Appendix A
| Fatty Acids wt.% | Rapseed | Soybean | Vernicia Fordii | Cornus Wilsoniana | Idesia Polycarpa | Zanthoxylum Bungeanum | Xanthoceras Sorbifolia | Armeniaca Sibirica | Jatropha Curcas | Pistacia Chinensis | Elaeis Guineensis |
|---|---|---|---|---|---|---|---|---|---|---|---|
| C14:0 | 0.07 | 0.05 | 0.03 | 1.00 | |||||||
| C16:0 | 3.49 | 10.58 | 3.56 | 16.53 | 15.50 | 20.14 | 5.27 | 3.79 | 19.75 | 23.14 | 44.80 |
| C16:1 | 0.00 | 0.00 | 0.97 | 6.65 | 0.30 | 0.67 | 0.99 | 0.30 | |||
| C18:0 | 0.85 | 4.76 | 2.62 | 1.77 | 1.39 | 0.15 | 1.92 | 1.01 | 4.63 | 1.18 | 3.80 |
| C18:1 | 64.40 | 22.52 | 10.57 | 30.50 | 9.53 | 29.72 | 31.17 | 65.23 | 46.83 | 44.35 | 39.90 |
| C18:2 | 22.30 | 52.34 | 14.64 | 48.50 | 64.81 | 29.62 | 44.47 | 28.92 | 28.50 | 28.51 | 9.28 |
| C18:3 | 8.23 | 8.19 | 59.20 | 1.60 | 2.08 | 17.91 | 6.46 | 0.14 | 0.01 | 0.84 | 0.22 |
| C20:0 | 0.10 | 0.20 | 0.09 | 0.17 | 0.10 | 0.35 | |||||
| C20:1 | 0.90 | 0.04 | 0.48 | 0.12 | |||||||
| C20:3 | 7.27 | ||||||||||
| Others | 0.73 | 1.61 | 8.51 | 0.05 | 1.53 | 3.24 | 0.89 | 0.03 | |||
| CP/°C | −4 | −0.5 | −11.0 | 0.0 | −5.0 | −6.0 | −8.0 | −14.0 | 0 | −3.0 | 12.0 |
| OS/h@110 °C | 2.0 | 1.3 | 0.4 | 0.6 | 0.7 | 1.2 | 1.7 | 2.7 | 3.3 | 4.2 | 7.7 |
| CN | 52.9 | 50.9 | 37 | 49 | 45 | 46 | 47.6 | 48.8 | 51 | 51.3 | 62.0 |
References
- 2017 China Petroleum Market Analysis. Available online: https://wenku.baidu.com/view/3946d7dc64ce0508763231126edb6f1aff0071fa.html (accessed on 26 January 2018).
- Cárdenas, M.D.; Armas, O.; Mata, C.; Soto, F. Performance and pollutant emissions from transient operation of a common rail diesel engine fueled with different biodiesel fuels. Fuel 2016, 185, 743–762. [Google Scholar] [CrossRef]
- Liu, S. Internal Combustion Engine Fundamentals, 4th ed.; China Machine Press: Beijing, China, 2017; ISBN 978-7-111-56286-3. [Google Scholar]
- Ministry of Ecology and Environment of the People’s Republic of China. China Vehicle Environmental Management Annual Report 2018. Available online: http://dqhj.mep.gov.cn/jdchjgl/zhgldt/201806/P020180604354753261746.pdf (accessed on 1 June 2018).
- Production Statistics. Available online: http://biodiesel.org/ (accessed on 6 June 2018).
- EU to Adjust Biodiesel Policy. Available online: http://www.china-nengyuan.com/news/126188.html (accessed on 1 July 2018).
- Analysis of China’s Biodiesel Market in 2018. Available online: http://www.chyxx.com/industry/201805/636866.html (accessed on 18 July 2018).
- The State Council Circular on the Issuance of the 12th Five-year Plan for Energy Development. Available online: http://www.nea.gov.cn/2013-01/28/c_132132808.htm (accessed on 7 July 2018).
- The National Development and Reform Commission Circular on the Issuance of the 13th Five-year Plan for Energy Development. Available online: http://www.ndrc.gov.cn/zcfb/zcfbtz/201612/t20161216_830264.html (accessed on 10 July 2018).
- The National Energy Administration Circular on the Issuance of Biodiesel Industry Development Policy. Available online: http://zfxxgk.nea.gov.cn/auto83/201501/t20150123_1882.htm (accessed on 10 July 2018).
- Lapuerta, M.; Armas, O.; Rodriguezfernandez, J. Effect of biodiesel fuels on diesel engine emissions. Prog. Energy Combust Sci. 2008, 34, 198–223. [Google Scholar] [CrossRef]
- Tanzer, E.; Murat, K.Y. Influence of blending ratio on the physicochemical properties of safflower oil methyl ester-safflower oil, safflower oil methyl ester-diesel and safflower oil-diesel. Renew. Energy 2016, 95, 233–247. [Google Scholar] [CrossRef]
- Gerhard, K. “Designer” Biodiesel: Optimizing Fatty Ester Composition to Improve Fuel Properties. Energy Fuel 2008, 22, 1358–1364. [Google Scholar] [CrossRef]
- Anantharaman, G.; Krishnamurthy, S.; Ramalingam, V. A review on combustion, performance, and emission characteristics of fuels derived from oil seed crops (biodiesels). Aust. J. Crop Sci. 2013, 7, 1350–1354. [Google Scholar]
- Pankin, K.E.; Ivanova, Y.V.; Kuz’Mina, R.I.; Shtykov, S.N. Comparison of the physicochemical characteristics of biofuels and petroleum fuels. Chem. Technol. Fuels Oils 2011, 47, 7–11. [Google Scholar] [CrossRef]
- Han, B.; Zhang, W.; Yin, F.; Liu, S.; Zhao, X. Advantages, Problems and Countermeasure on the Development of Biodiesel Industry in Yunnan Province. Farm Prod. Process. 2016, 2016, 48–52. [Google Scholar] [CrossRef]
- Tyson, S.; McCormick, R. Biodiesel Handling and Use Guidelines, 3rd ed.; US Department of Energy. Available online: http://www.biodiesel.org/reports/20060909_gen-317.pdf (accessed on 21 June 2018).
- McCormick, R.L.; Williams, A.; Ireland, J.; Brimhall, M.; Hayes, R.R. Effects of Biodiesel Blends on Vehicle Emissions. National Renewable Energy Laboratory, U.S. Department of Energy. Available online: https://www.nrel.gov/docs/fy07osti/40554.pdf (accessed on 1 June 2018).
- European Parliament Directive 2009/30/EC. Available online: https://www.mendeley.com/research-papers/european-parliament-directive-200930ec/ (accessed on 1 July 2018).
- Xin, Y. Developing Status of Biodiesel in Germany and Europe. China Oils Fats 2008, 2008, 1–6. [Google Scholar]
- Wang, L.; Yu, H.; He, X. Influence of fatty acid composition of woody biodiesel plants on the fuel properties. J. Fuel Chem. Technol. 2012, 40, 397–404. [Google Scholar] [CrossRef]
- Canakci, M.; Sanli, H. Biodiesel Production from Various Feedstocks and their effects on the fuel properties. J. Ind. Microbiol. Biotechnol. 2008, 35, 431–441. [Google Scholar] [CrossRef] [PubMed]
- Serrano, M.; Bouaid, A.; Martínez, M.; Aracil, J. Oxidation stability of biodiesel from different feedstocks: Influence of commercial additives and purification step. Fuel 2013, 113, 50–58. [Google Scholar] [CrossRef]
- Karmakar, A.; Karmakar, S.; Mukherjee, S. Properties of various plants and animals feedstocks for biodiesel production. Bioresource Technol. 2010, 101, 7201–7210. [Google Scholar] [CrossRef] [PubMed]
- Islam, M.A.; Magnusson, M.; Brown, R.J.; Ayoko, G.; Nabi, M.; Heimann, K. Microalgal Species Selection for Biodiesel Production Based on Fuel Properties Derived from Fatty Acid Profiles. Energies 2013, 6, 5676–5702. [Google Scholar] [CrossRef]
- Ramos, M.J.; Fernández, C.M.; Casas, A.; Rodríguez, L.; Pérez, Á. Influence of fatty acid composition of raw materials on biodiesel properties. Bioresour. Technol. 2008, 100, 261–268. [Google Scholar] [CrossRef] [PubMed]
- Knothe, G.; Matheaus, A.C.; Ryan, T.W. Cetane numbers of branched and straight chain fatty esters determined in an ignition tester. Fuel 2003, 82, 971–975. [Google Scholar] [CrossRef]
- Zhu, Z.; Guo, H.; Zhou, A.; Li, D.; Liu, S. One Way to reduce the NOx emission of biodiesel: The increase of cetane number. Int. J. Green Energy 2016, 13, 957–962. [Google Scholar] [CrossRef]
- Chen, L.; Wang, Z.; Li, M. Effects of CN Improver DTBP on biodiesel combustion and emissions. In Proceeding of the China Society of ICE, Shanghai, China, 8–11 August 2011. [Google Scholar]
- Rakopoulos, D.C.; Rakopoulos, C.D.; Giakoumis, E.G.; Dimaratos, A.M.; Kyritsis, D.C. Effects of butanol-diesel fuel blends on the performance and emissions of a high-speed DI diesel engine. Energy Convers. Manag. 2010, 51, 1989–1997. [Google Scholar] [CrossRef]
- Sebastian, J.; Nagarajan, G. Experimental Study on Influence of Fuel Oxygen Content on Combustion and Emission Characteristics of a Direct Injection CI Engine; SAE 2010-01-1969; SAE: Warrendale, PA, USA, 2010. [Google Scholar]
- Guo, H.; Liu, S.; Zhou, L. Study on Ethylene Glycol Monomethyl Ether Peanut Oil Monoester as a Novel Biodiesel; SAE 2008-01-1575; SAE: Warrendale, PA, USA, 2008. [Google Scholar]
- Jiang, D.Y.; Guo, H.J.; Liu, S.H. Research on a new rapeseed oil monoester as a clean fuel for diesel engine. Fuel 2006, 51, 859–861. [Google Scholar]
- Jiang, D.; Wang, X.; Liu, S.; Guo, H. Rapeseed Oil Monoester of Ethylene Glycol Monomethyl Ether as a New Biodiesel. Biomed. Res. Int. 2011, 2011, 293161. [Google Scholar] [CrossRef]
- Mani, N.; Edwin, A.F.; David, W.N. Oxygenates for Advanced Petroleum Based Diesel Fuels: Part 1. Screening and Selection Methodology for the Oxygenates; SAE 2001-01-3631; SAE: Warrendale, PA, USA, 2001. [Google Scholar]
- Guo, H.; Liu, S.; He, J. Performances and Emissions of New Glycol Ether Blends in Diesel Fuel used as Oxygenated Fuel for Diesel Engines. Am. Soc. Civ. Eng. 2015, 142. [Google Scholar] [CrossRef]
- Rakopoulos, C.D.; Giakoumis, E.G.; Dimaratos, A.M. Investigation of the combustion of neat cottonseed oil or its neat bio-diesel in a HSDI diesel engine by experimental heat release and statistical analyses. Fuel 2010, 89, 3814–3826. [Google Scholar] [CrossRef]
- Guo, H.; Xun, Q.; Liu, S. Investigation of Ethylene Glycol Monomethyl Ether Soyate as a Biofuel; SAE 2015-01-0955; SAE: Warrendale, PA, USA, 2015. [Google Scholar]
- Zhang, H.; Li, Q.; Li, H.; Ma, Z. Study of the ethylene glycol monoether of soyate for biodiesel production. Renew. Energy Resour. 2010, 28, 43–47. [Google Scholar] [CrossRef]
- Chen, X.; Guo, H.; Zhang, L. Research on Ethylene Glycol Monoethyl Ether Palm Oil Monoester as a Novel Biodiesel-Like Fuel and Its Effecton Diesel Engine Performance; SAE 2014-01-1469; SAE: Warrendale, PA, USA, 2014. [Google Scholar]
- Jiang, D.; Bai, Y.; Guo, H. Exhaust emissions and combustion performances of ethylene glycol monomethyl ether palm oil monoester as a novel biodiesel. Afr. J. Biotechnol. 2011, 10, 16300–16313. [Google Scholar] [CrossRef]
- Guo, H.; Wang, R.; Su, J. Study of Ether Group Effects on Physicochemical Properties of Cottonseed Oil Ether Monoesters as Novel Biodiesels; SAE 2013-01-2601; SAE: Warrendale, PA, USA, 2013. [Google Scholar]
- Li, Y.; Guo, H.; Zhu, Z.; Feng, Y.; Liu, S. Ethylene glycol monomethyl ether cottonseed oil monoester: Properties evaluation as biofuel. Int. J. Green Energy 2012, 9, 376–387. [Google Scholar] [CrossRef]
- Ma, J.; Wang, X.; Guo, H. Research on the new kinds of oxygenated diesel fuels. Energy Eng. 2010, 2010, 28–31. [Google Scholar] [CrossRef]
- Gao, G.; Feng, Y.; Guo, H.; Liu, S. Synthesis, Structure Characterization, and Engine Performance Test of Ethylene Glycol n-Propyl Ether Palm Oil Monoester as Biodiesel. Energy Fuel 2011, 25, 4686–4692. [Google Scholar] [CrossRef]
- Wang, W.; Wang, X.; Guo, H.; Jiang, D.; Zhang, J. Research on the synthesis and performances of a new biodiesel, propylene glycol methyl ether palm oil monoester. In Proceedings of the 2011 International Conference on Electric Information and Control Engineering, Wuhan, China, 15–17 April 2011. [Google Scholar]
- Li, Z.; Guo, H.; Su, J.; Wang, R. Synthesis of Diethylene Glycol Monomethyl Ether Cottonseed Oil as a Novel Biodiesel. China Contemp. Chem. Ind. 2012, 41, 116–119. [Google Scholar] [CrossRef]
- Zhou, A.; Guo, H.; Chen, X.; Wang, H.; Liu, S. Preparation, Structure Analysis, and Engine Performance Test of Triethylene Glycol Monomethyl Ether Cottonseed Oil Monoester as Biodiesel; SAE 2013-6-0096; SAE: Warrendale, PA, USA, 2013. [Google Scholar]
- Lou, S.; Jia, L.; Guo, X.; Wu, P.; Gao, L.; Wang, J. Preparation of diethylene glycol monomethyl ether monolaurate catalyzed by active carbon supported KF/CaO. Springerplus 2015, 4, 686. [Google Scholar] [CrossRef] [PubMed]
- Guo, H.; Liu, S. Study of Ether Species Effects on Physicochemical Properties of Palm Oil Ether Monoesters as Novel Biodiesels. Int. J. Chem. Mol. Nucl. Mater. Metall. Eng. 2013, 7, 11. [Google Scholar] [CrossRef]
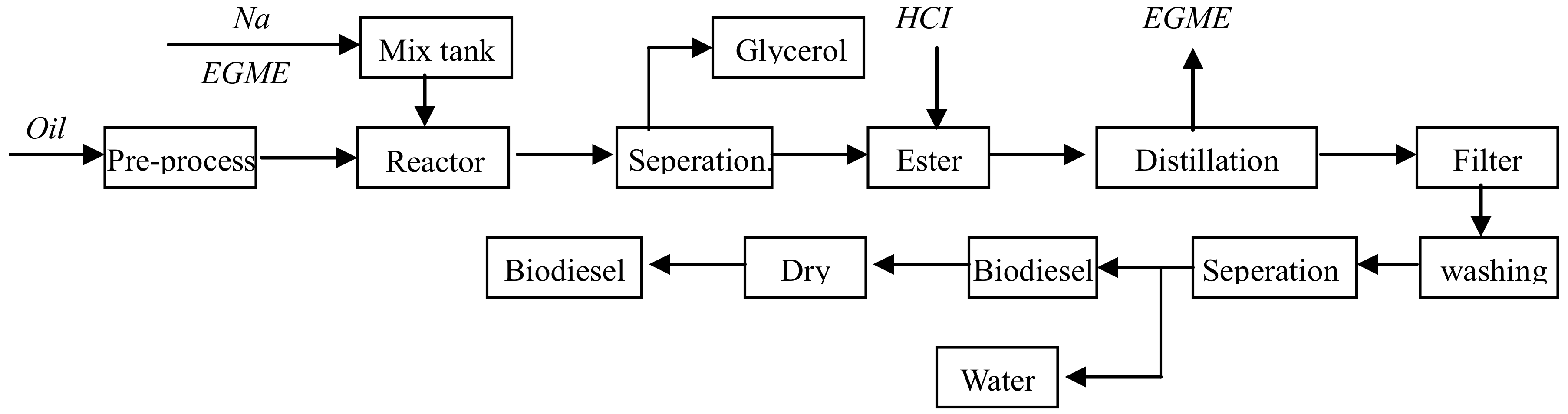
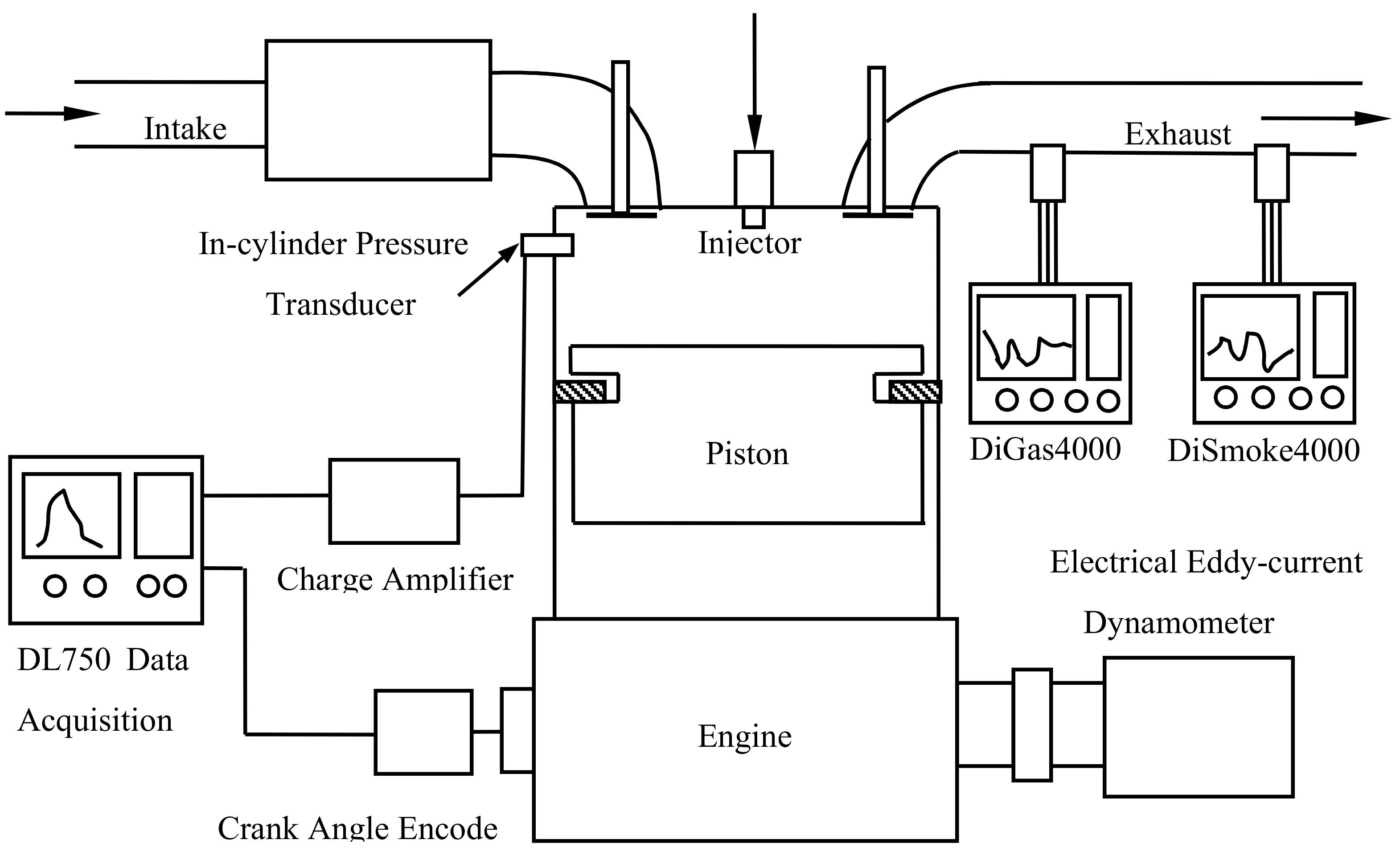
| Oil | Alcohol | Product | Density | LHV | OC | CN |
|---|---|---|---|---|---|---|
| 0# diesel | / | 0# diesel | 860 | 42.5 | 0 | 50 |
| Rapeseed | methanol | RCOOCH3 | 884 | / | 10.9 | 51 |
| Cottonseed | 886 | 37.5 | 10 | 52 | ||
| Rapeseed | ethylene glycol methyl ether | RCOOCH2CH2OCH3 | 910 | 38.7 | / | / |
| Soybean | 907.2 | 38.5 | / | 66 | ||
| Peanut | 939.1 | 37.0 | / | / | ||
| Cottonseed | 901.5 | 38.5 | 14.3 | 70.1 | ||
| Palm | ethylene glycol ethyl ether | RCOOCH2CH2OC2H5 | 893.4 | 36.2 | 80.4 | |
| Soybean | 902.7 | 38.6 | / | / | ||
| Palm | ethylene glycol n-propyl ether | RCOOCH2CH2OC3H7 | / | 37.3 | 16.35 | 72.3 |
| Palm | propylene glycol methyl ether | RCOOCH(CH3)CH2OCH3 | 889.2 | / | / | / |
| Cottonseed | diethylene glycol methyl ether | RCO(OCH2CH2)2OCH3 | 912.0 | 37.0 | 16.88 | 76.9 |
| triethylene glycol methyl ether | RCO(OCH2CH2)3OCH3 | 935.5 | 35.8 | 18.90 | 80.9 | |
| Standard | ASTM | ASTM | ASTM | ASTM |
| Engine Type | 2102 QB | TY1100 |
|---|---|---|
| Bore × Stroke (mm) | 102 × 115 | 100 × 115 |
| Displacement (cm3) | 1880 | 903 |
| Chamber shape | ω | ω |
| Compression ratio | 17.5:1 | 18:1 |
| Rated power (kW) @ speed (r·min−1) | 23.5/2300 | 11/2300 |
| Injector type | ZCK154S432 | ZCK154S432 |
| Nozzle hole number—diameter (mm) | 4×Φ0.3 | 4×Φ0.3 |
| Delivery timing (deg. BTDC) | 25 | 25 |
| Injection pressure (MPa) | 19.0 | 19.0 |
| Fuel Type | Engine | Operation Condition | Smoke | HC | CO | NOx |
|---|---|---|---|---|---|---|
| FAME | 2102QB | 1400 r·min−1 | 47.4% | 36.4% | 62.5% | 16.2% |
| 2200 r·min−1 | 45.5% | 20.0% | 50.0% | 24.2% | ||
| EGMEROM | TY110 | 2200 r·min−1 | 78.2% | 61.5% | 50.0% | Increase slightly |
| EGMESOM | TY110 | 2200 r·min−1 | 74.6% | 61.6% | 79.1% | changeless |
| EGMEPOM (Peanut) | TY110 | 1400 r·min−1 | 65.2% | 53.8% | 50.0% | changeless |
| 2000 r·min−1 | 75.0% | 69.2% | 50.0% | changeless | ||
| EGMEPOM (Palm) | 2102QB | 1400 r·min−1 | 58.3% | 66.7% | 60.0% | changeless |
| 2000 r·min−1 | 55.6% | 66.7% | 54.5% | 32.2% | ||
| EGMECOM | 2102QB | 1400 r·min−1 | 50.0% | 55.6% | A little | 39.3% |
| 2000 r·min−1 | 45.5% | 55.6% | A little | 39.3% |
| Fuel Type | Smoke | HC | CO | NOx |
|---|---|---|---|---|
| FAME | 47.4% | 36.4% | 62.5% | 24.2% |
| EGEESOM | 83.3% | 76.7% | 64.7% | Increase slightly |
| EGEEPOM | 60.0% | 66% | 55.6% | 32.2% |
| EGnPEPOM | 50% | 66.6% | 27.1% | 23.7% |
| PGMEPOM | 75% | 76.2% | 67.7% | changeless |
| DGMECOM | 80% | 33.3% | 33.3% | 33.3% |
| TGMECOM | 54.6% | Increase 20% | 20% | 40% |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, S.; Chen, W.; Zhu, Z.; Jiang, S.; Ren, T.; Guo, H. A Review of the Developed New Model Biodiesels and Their Effects on Engine Combustion and Emissions. Appl. Sci. 2018, 8, 2303. https://doi.org/10.3390/app8112303
Liu S, Chen W, Zhu Z, Jiang S, Ren T, Guo H. A Review of the Developed New Model Biodiesels and Their Effects on Engine Combustion and Emissions. Applied Sciences. 2018; 8(11):2303. https://doi.org/10.3390/app8112303
Chicago/Turabian StyleLiu, Shenghua, Wei Chen, Zengqiang Zhu, Sa Jiang, Tongtong Ren, and Hejun Guo. 2018. "A Review of the Developed New Model Biodiesels and Their Effects on Engine Combustion and Emissions" Applied Sciences 8, no. 11: 2303. https://doi.org/10.3390/app8112303
APA StyleLiu, S., Chen, W., Zhu, Z., Jiang, S., Ren, T., & Guo, H. (2018). A Review of the Developed New Model Biodiesels and Their Effects on Engine Combustion and Emissions. Applied Sciences, 8(11), 2303. https://doi.org/10.3390/app8112303






