Supercritical Regeneration of an Activated Carbon Fiber Exhausted with Phenol
Abstract
:Featured Application
Abstract
1. Introduction
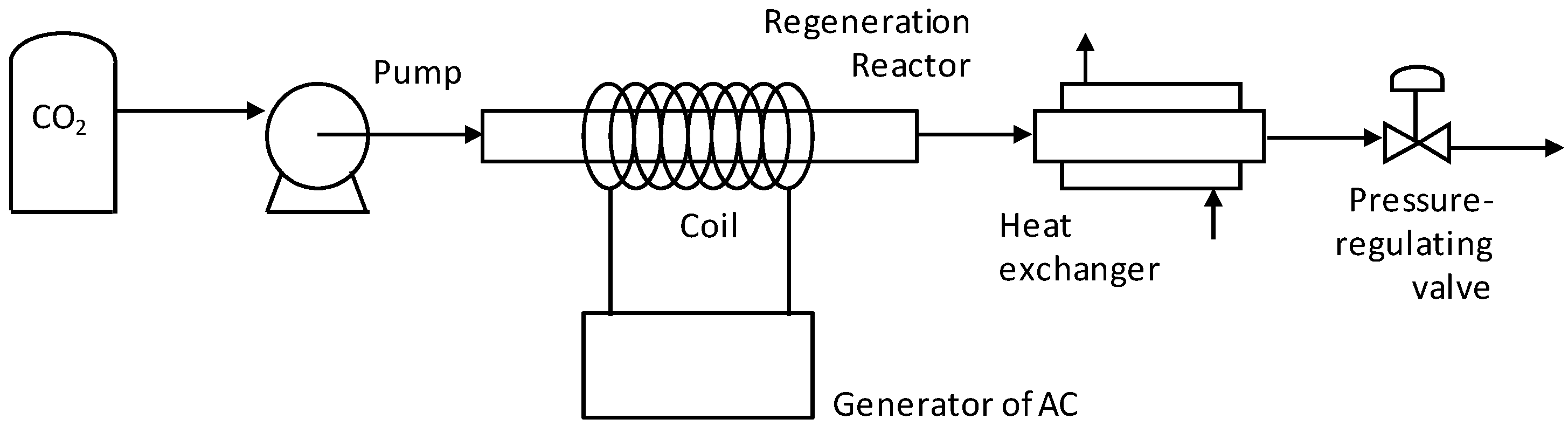
2. Materials and Methods
2.1. Materials
2.2. Adsorption–Regeneration Cycles
2.3. Textural Characterization
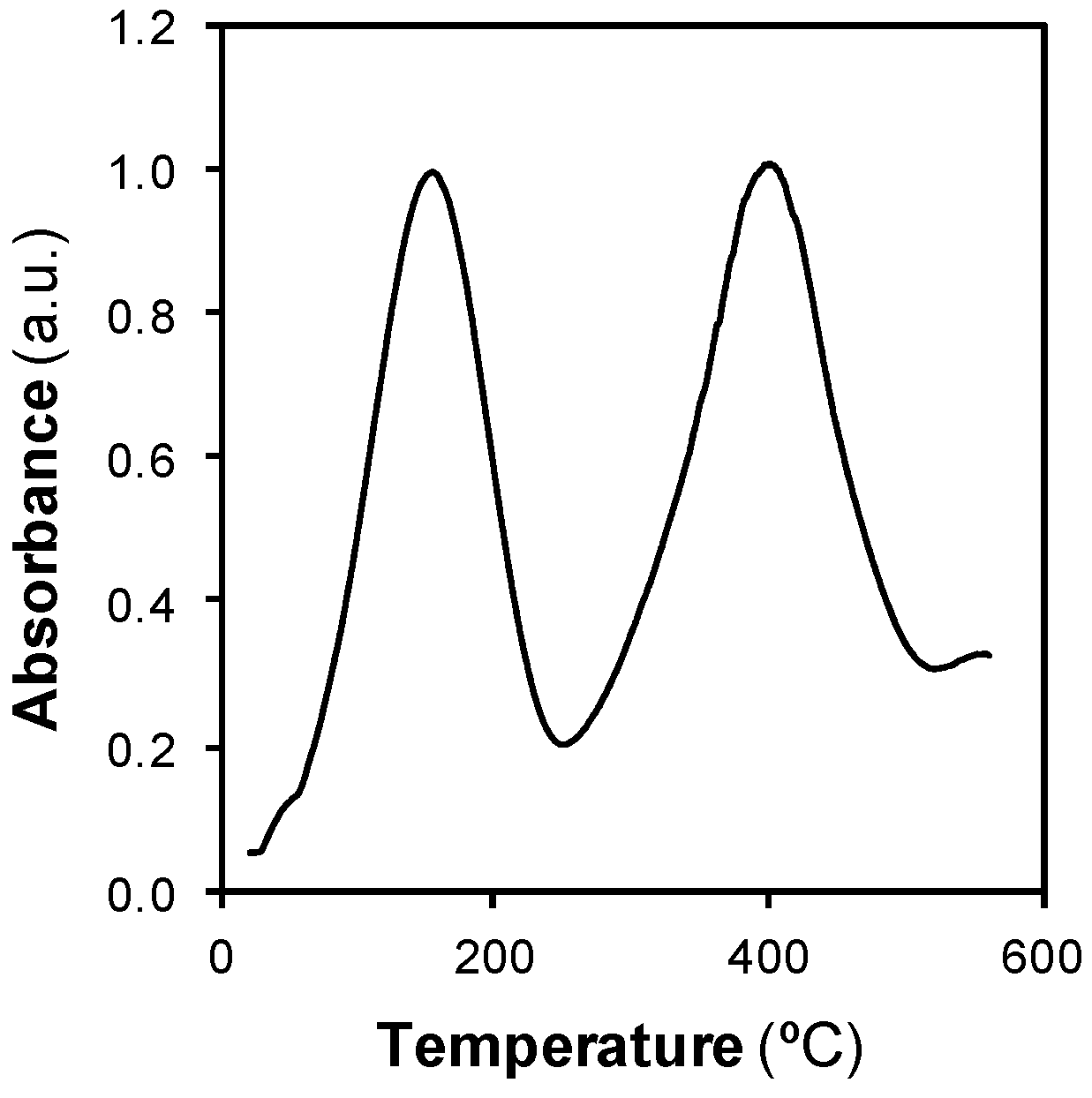
2.4. Temperature Programmed Desorption (TPD) with Pressurized Water
3. Results and Discussion
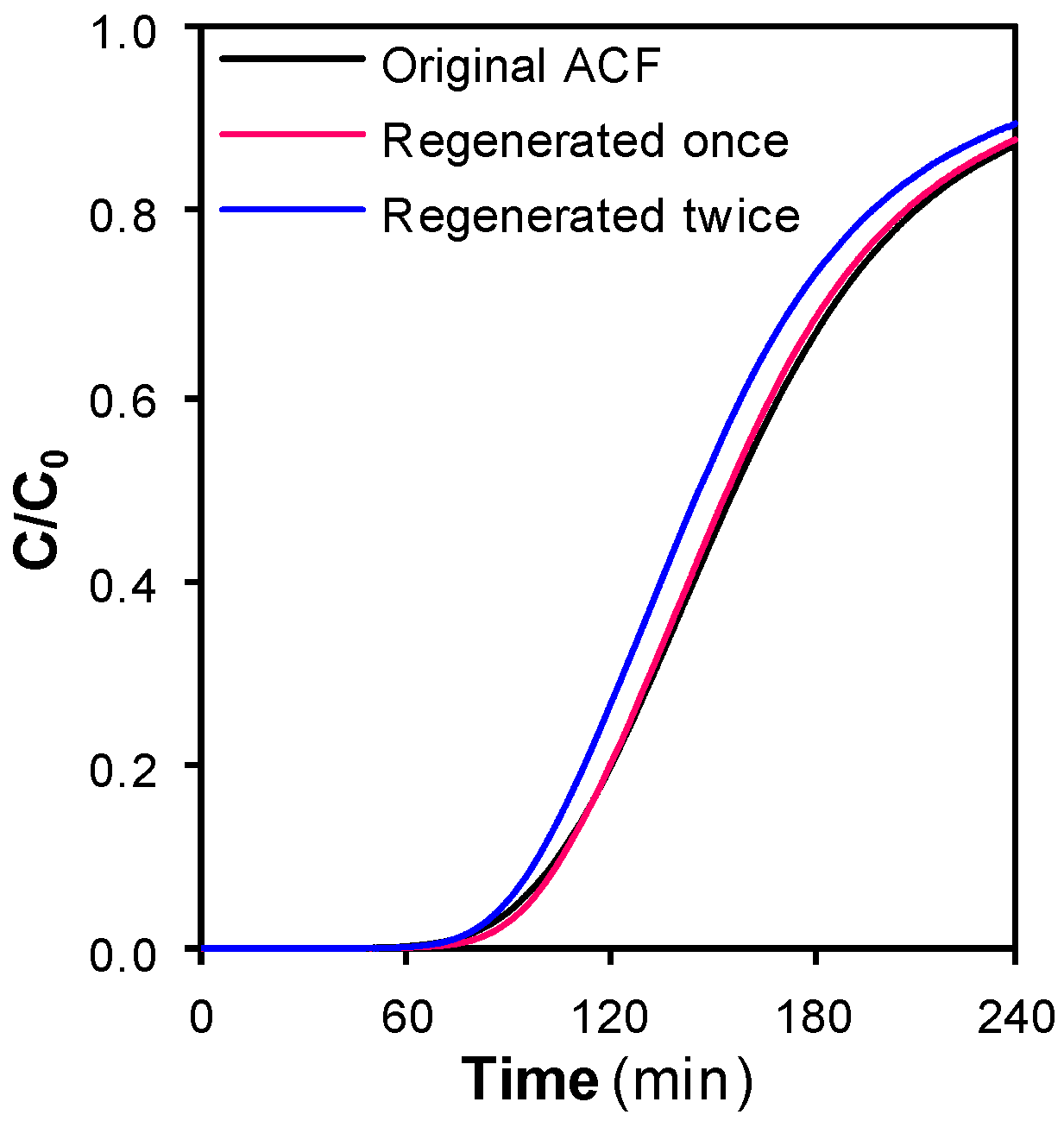
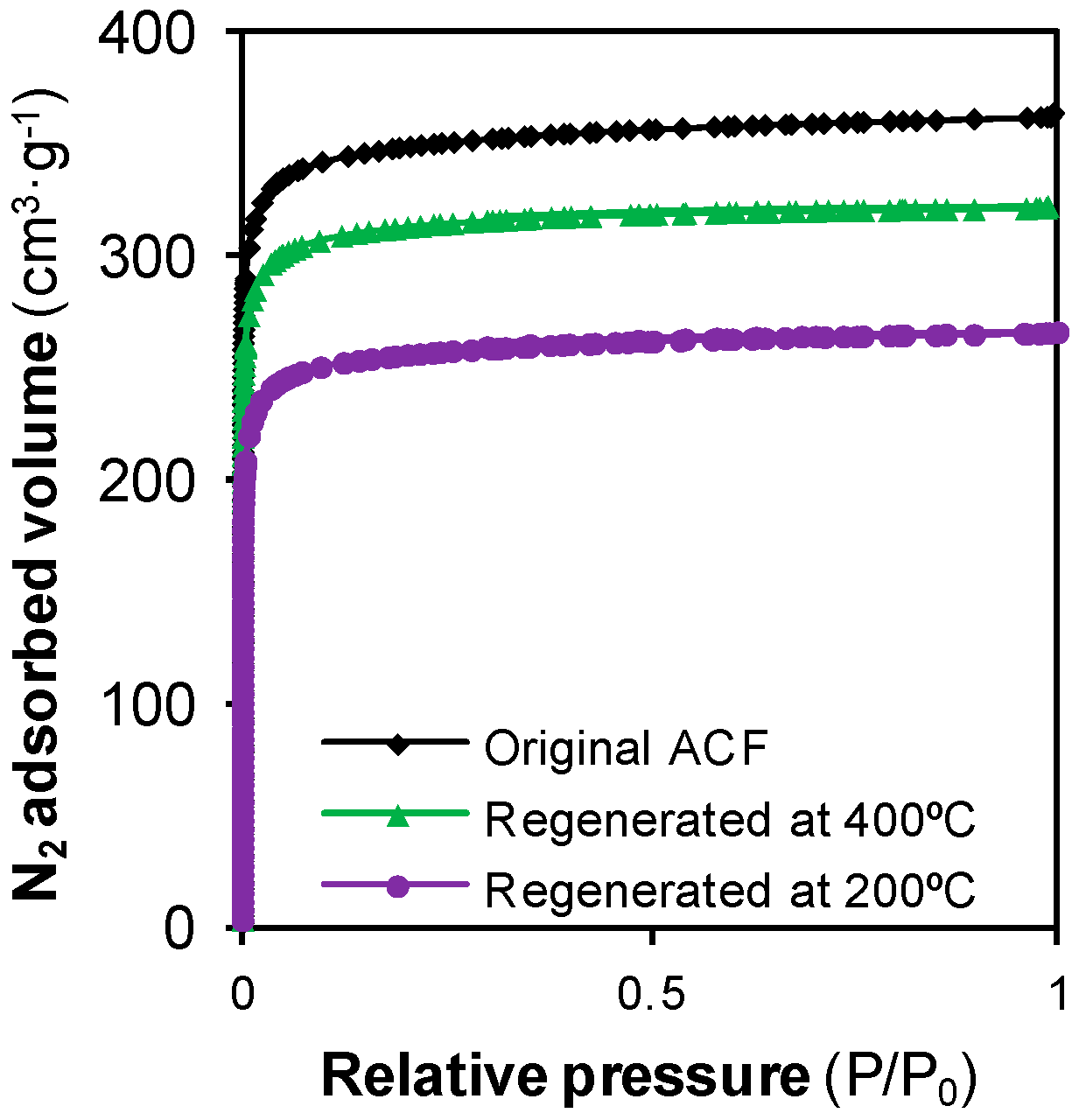
3.1. Preliminary Studies: Effect of Supercritical Treatments on the Textural Properties of the ACF
3.2. SCCO2 Regeneration
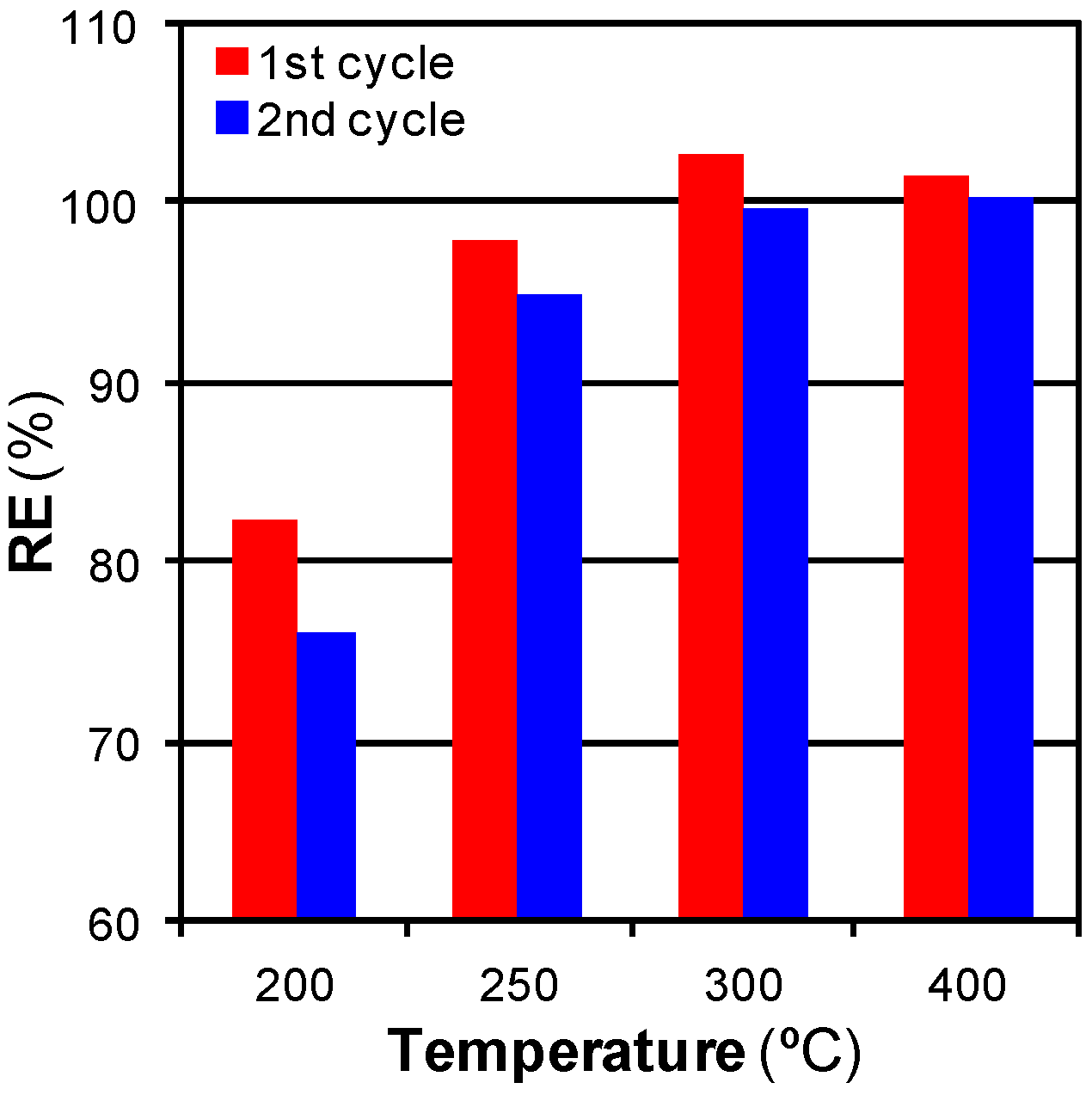
3.2.1. Effect of Temperature
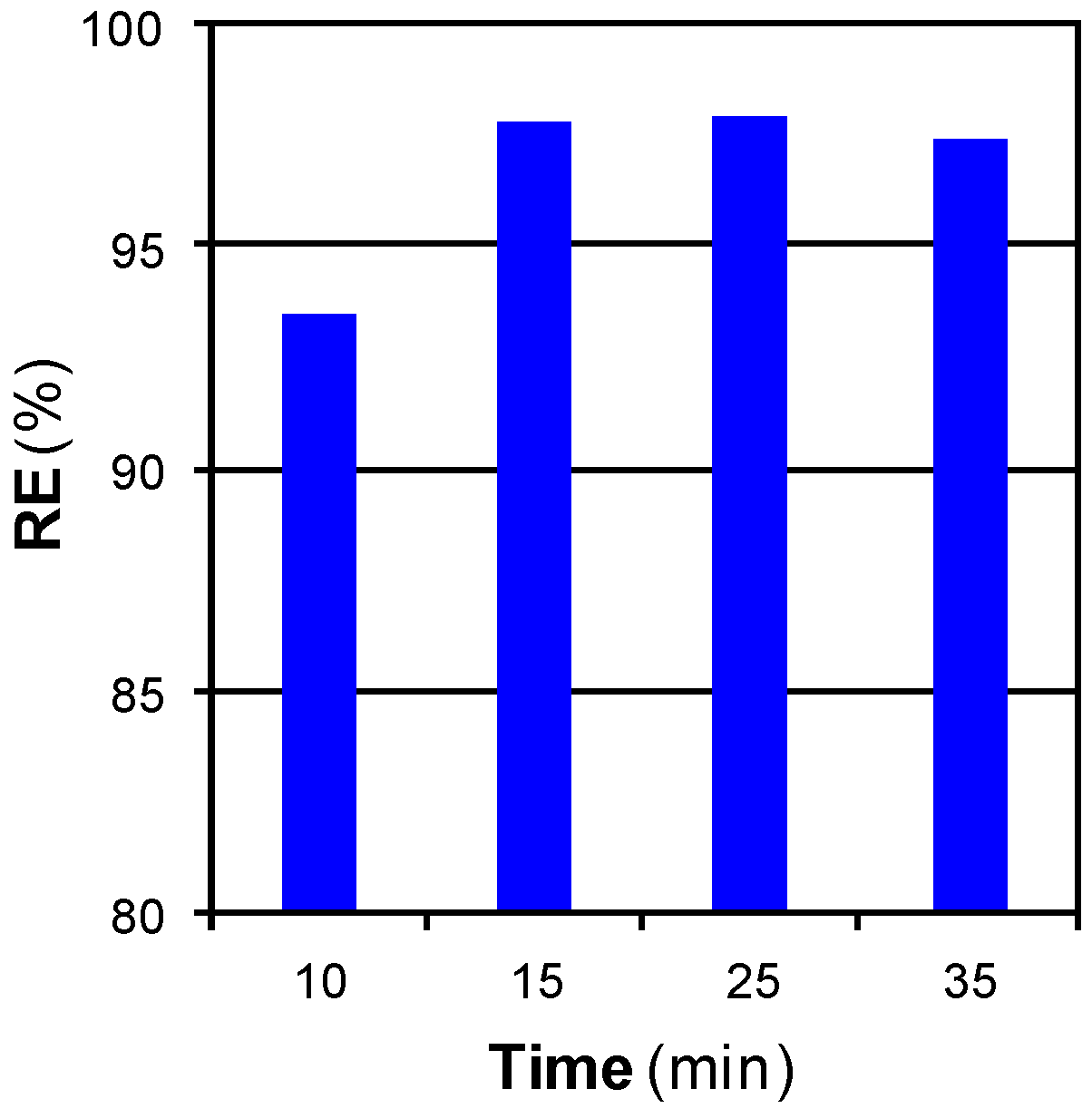
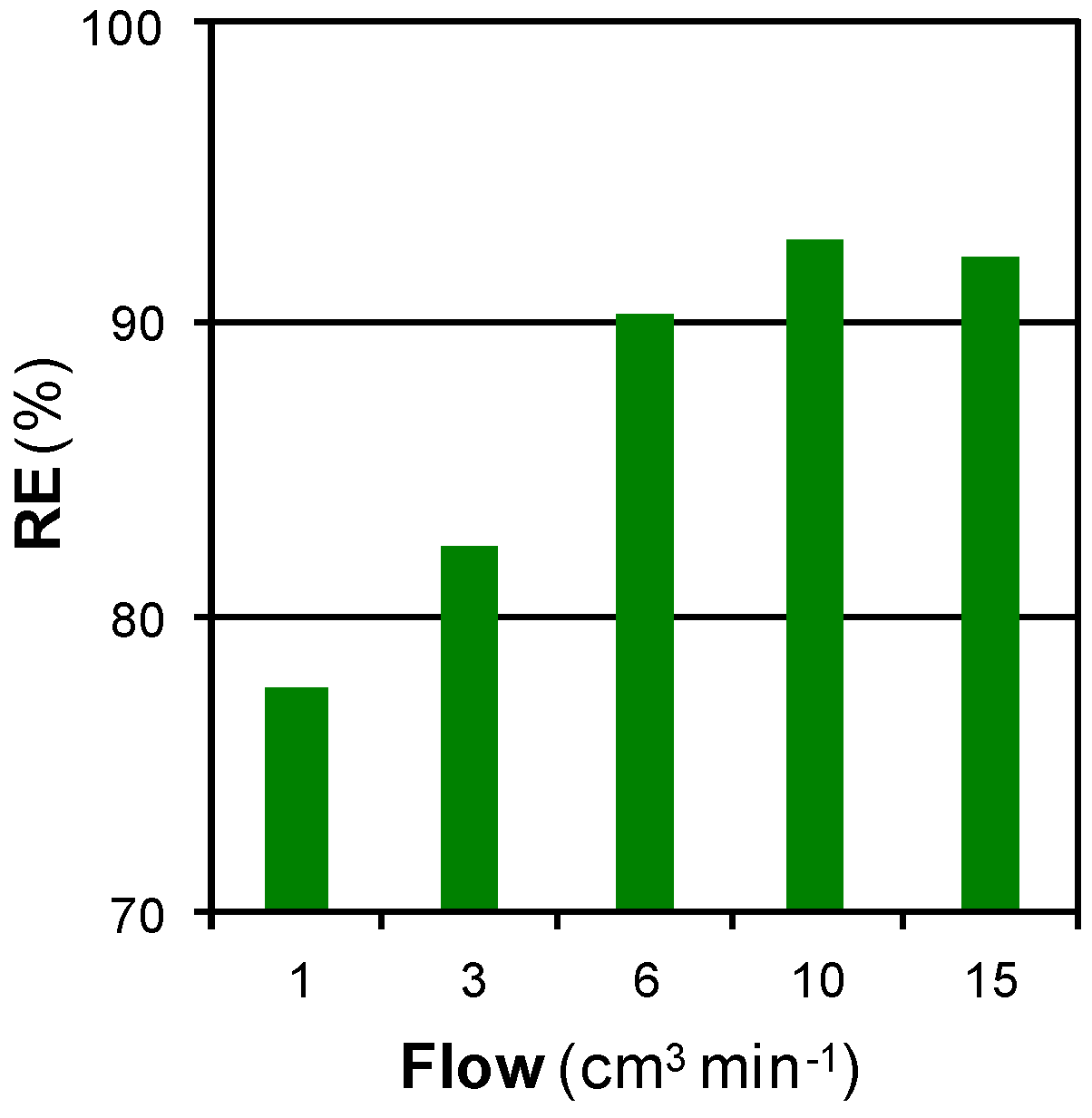
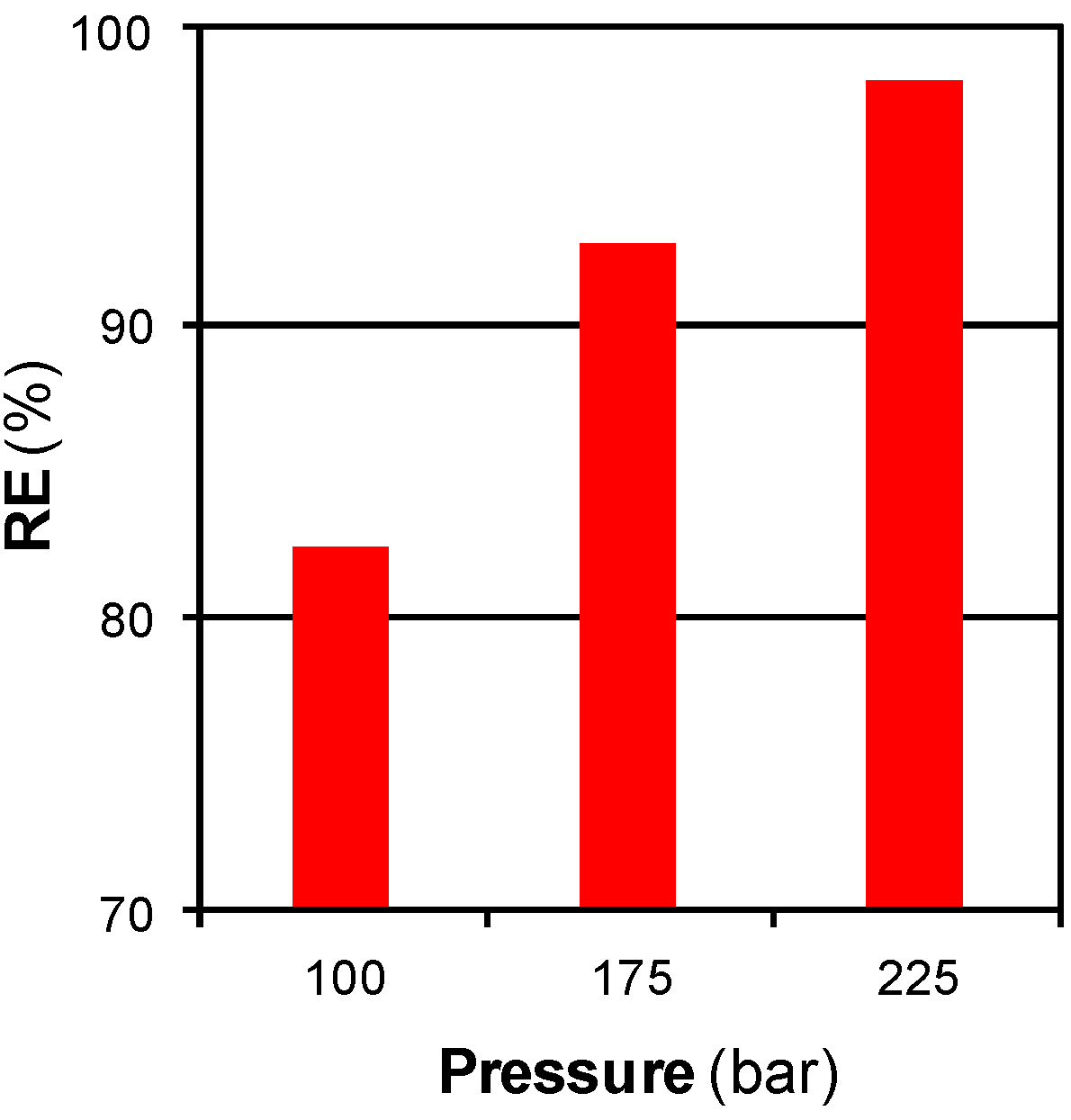
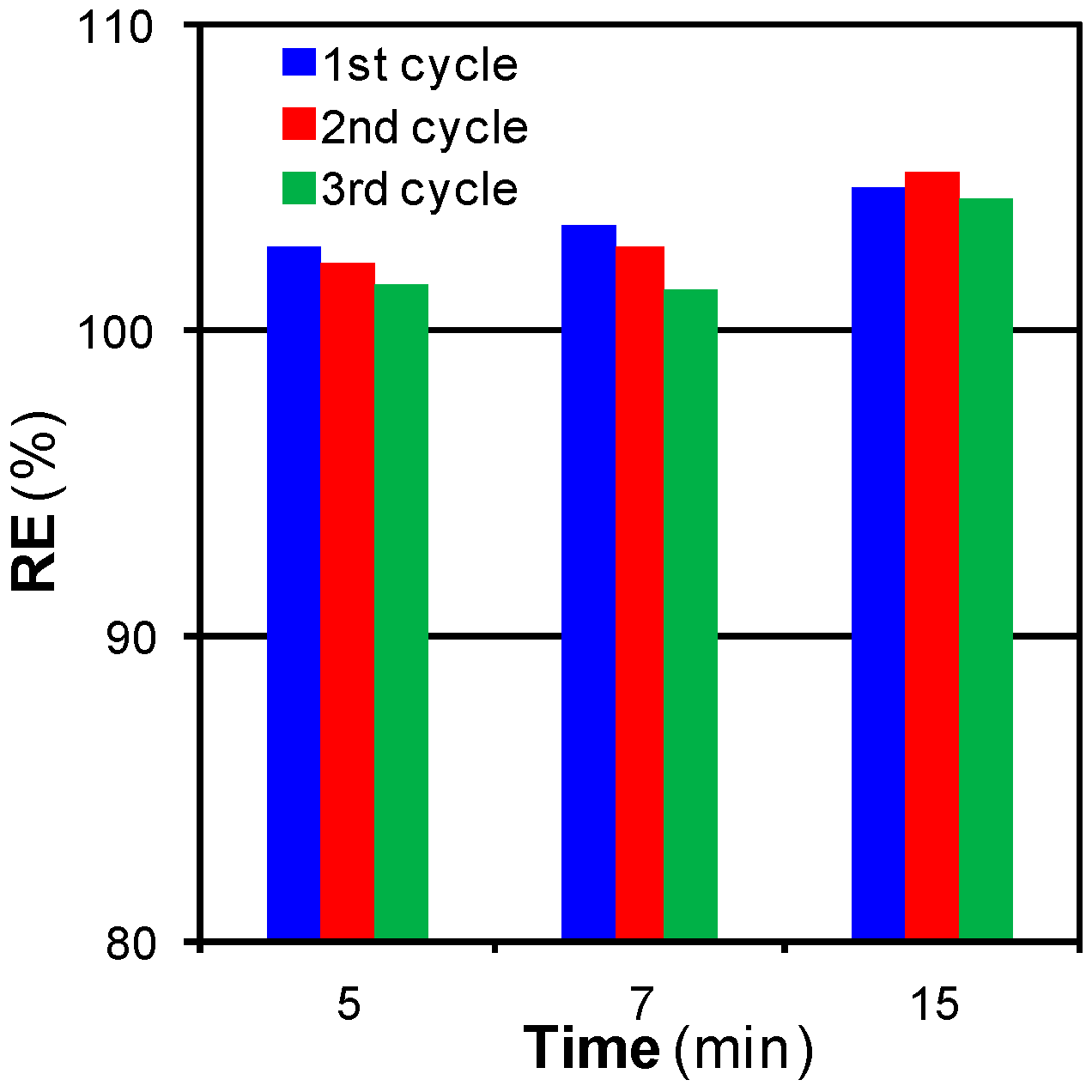
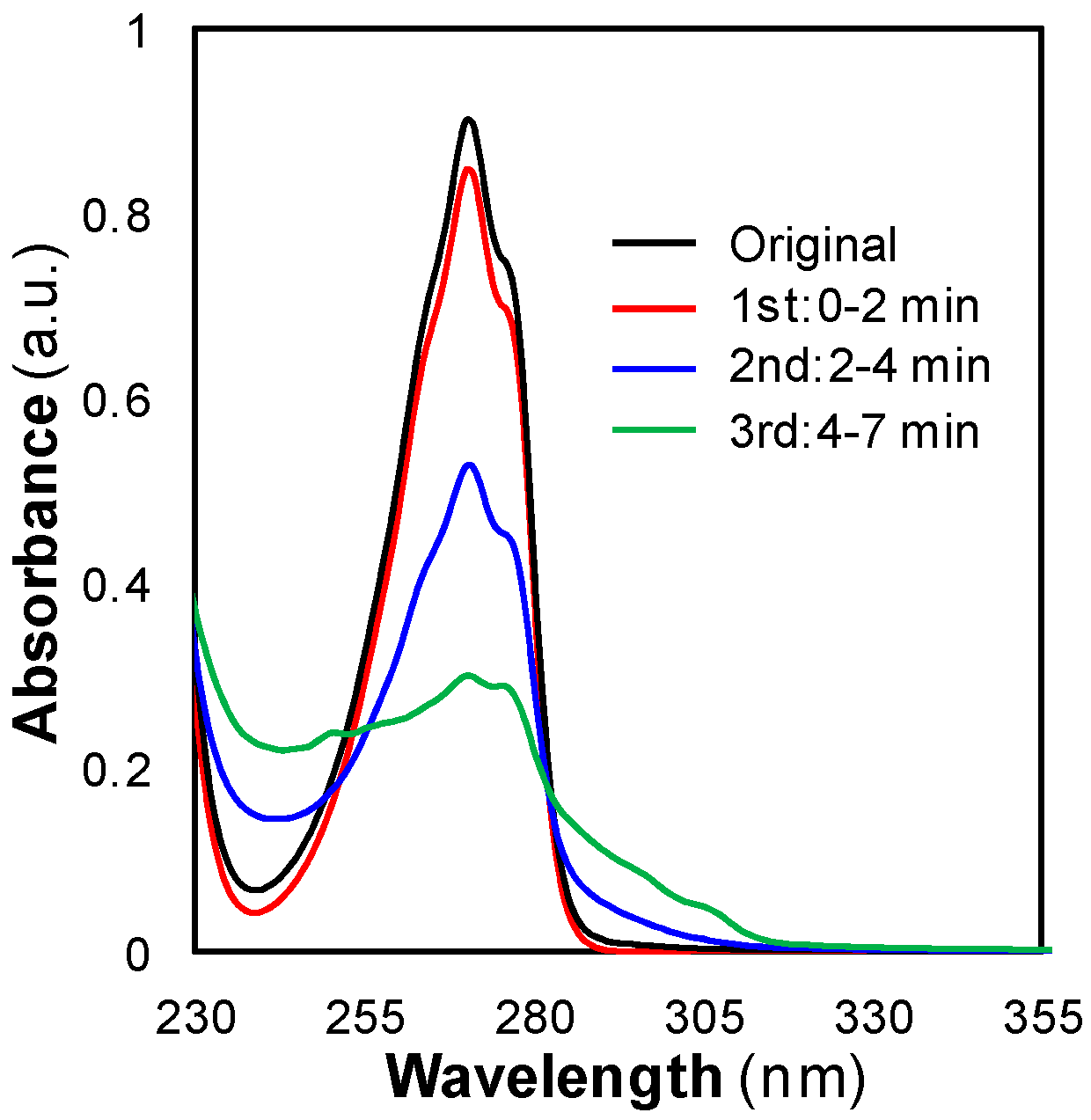
3.2.2. Mass Transfer Limitations: Effect of Time, Flow, and Pressure
3.3. Comparison between SCCO2 Regeneration and SCW Regeneration
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Souza-Machado, B.A.; Gambini-Pereira, C.; Baptista-Nunes, S.; Ferreira-Padilha, F.; Umsza-Guez, M.A. Supercritical fluid extraction using CO2: Main applications and future perspectives. Sep. Sci. Technol. 2013, 48, 2741–2760. [Google Scholar] [CrossRef]
- Ahmed, M.J. Preparation of activated carbons from date (Phoenix dactylifera L.) palm stones and application for wastewater treatments: Review. Process Saf. Environ. Prot. 2016, 102, 168–182. [Google Scholar] [CrossRef]
- Salvador, F.; Martin-Sanchez, N.; Sanchez-Hernandez, R.; Sanchez-Montero, M.J.; Izquierdo, C. Regeneration of carbonaceous adsorbents. Part I: Thermal regeneration. Microporous Mesoporous Mater. 2015, 202, 259–276. [Google Scholar] [CrossRef]
- Salvador, F.; Martin-Sanchez, N.; Sanchez-Hernandez, R.; Sanchez-Montero, M.J.; Izquierdo, C. Regeneration of carbonaceous adsorbents. Part II: Chemical, Microbiological and Vacuum Regeneration. Microporous Mesoporous Mater. 2015, 202, 277–296. [Google Scholar] [CrossRef]
- Wang, J.H.; Smith, J.M. Thermal regeneration of the phenol-carbon system. AIChE J. 1985, 31, 496–498. [Google Scholar] [CrossRef]
- Moreno-Castilla, C.; Rivera-Utrilla, J.; Joly, J.P.; López-Ramón, M.V.; Ferro-García, M.A.; Carrasco-Marín, F. Thermal regeneration of an activated carbon exhausted with different substituted phenols. Carbon 1995, 33, 1417–1423. [Google Scholar] [CrossRef]
- Tan, C.S.; Liou, D.C. Supercritical regeneration of activated carbon loaded with benzene and toluene. Ind. Eng. Chem. Res. 1989, 28, 1222–1226. [Google Scholar] [CrossRef]
- Tan, C.S.; Liou, D.C. Regeneration of activated carbon loaded with toluene by supercritical carbon dioxide. Sep. Sci. Technol. 1989, 24, 111–127. [Google Scholar] [CrossRef]
- Ryu, Y.K.; Kim, K.L.; Lee, C.H. Adsorption and desorption of n-hexane, methyl ethyl ketone, and toluene on an activated carbon fiber from supercritical carbon dioxide. Ind. Eng. Chem. Res. 2000, 39, 2510–2518. [Google Scholar] [CrossRef]
- Takahashi, N.; Ushiki, I.; Hamabe, Y.; Sato, Y.; Inomata, H. Measurement and prediction of desorption behavior of five volatile organic compunds (acetone, n-hexane, methanol, toluene, and n-decane) from activated carbon for supercritical carbon dioxide regeneration. J. Supercrit. Fluids 2016, 107, 226–233. [Google Scholar] [CrossRef]
- Ushiki, I.; Kikuchi, K.; Takahashi, N.; Sato, Y.; Ito, Y.; Inomata, H. Desorption behavior of various volatile organic compounds from activated carbon in supercritical carbon dioxide: Measurement and kinetic modeling. J. Supercrit. Fluids 2017, 121, 41–51. [Google Scholar] [CrossRef]
- Benkhedda, J.; Jaubert, J.N.; Barth, D.; Zetzl, C.; Brunner, G. Adsorption and desorption of m-xylene from supercritical carbon dioxide on activated carbon. Sep. Sci. Technol. 2001, 36, 2197–2211. [Google Scholar] [CrossRef]
- Heidari, A.; Lotfollahi, M.N.; Baseri, H. Regeneration of activated carbon loaded with cyclohexane using supercritical carbon dioxide: Experimental results and modeling. Chem. Eng. Technol. 2013, 36, 315–322. [Google Scholar] [CrossRef]
- Srinivasan, M.P.; Smith, J.M.; McCoy, B.J. Supercritical fluid desorption from activated carbon. Chem. Eng. Sci. 1990, 45, 1885–1895. [Google Scholar] [CrossRef]
- Tan, C.S.; Liou, D.C. Desorption of ethyl acetate from activated carbon by supercritical carbon dioxide. Ind. Eng. Chem. Res. 1988, 27, 988–991. [Google Scholar] [CrossRef]
- Liang, M.T.; Liang, R.C.; Lin, C.H. Regeneration of cobalt-contaminated activated carbon by supercritiacl carbon dioxide extraction. Adsorption 2012, 18, 461–467. [Google Scholar] [CrossRef]
- Chihara, K.; Oomori, K.; Oono, T.; Mochizuki, Y. Supercritical CO2 regeneration of activated carbon loaded with organic adsorbates. Water Sci. Technol. 1997, 35, 261–268. [Google Scholar]
- Humayun, R.; Karakas, G.; Dahlstrom, P.R.; Ozkan, U.S.; Tomasko, D.L. Supercritical fluid extraction and temperature-programmed desorption of phenol and Its oxidative coupling products from activated carbon. Ind. Eng. Chem. Res. 1998, 37, 3089–3097. [Google Scholar] [CrossRef]
- Carmona, M.; Garcia, M.T.; Carnicer, A.; Madrid, M.; Rodriguez, J.F. Adsorption of phenol and chlorophenols onto granular activated carbon and their desorption by supercritical CO2. J. Chem. Technol. Biotechnol. 2014, 89, 1660–1667. [Google Scholar] [CrossRef]
- Madras, G.; Erkey, C.; Akgerman, A. Supercritical fluid regeneration of activated carbon loaded with heavy molecular weight organics. Ind. Eng. Chem. Res. 1993, 32, 1163–1168. [Google Scholar] [CrossRef]
- Taylor, P.N.; Lester, J.N. Rapid extraction of isoproturon and diuron from granular activated carbon. Chemosphere 1996, 33, 1227–1240. [Google Scholar] [CrossRef]
- Macnaughton, S.J.; Foster, N.R. Supercritical adsorption and desorption behavior of DDT on activated carbon using carbon dioxide. Ind. Eng. Chem. Res. 1995, 34, 275–282. [Google Scholar] [CrossRef]
- Salvador, F.; Martin-Sanchez, N.; Sanchez-Montero, M.J.; Montero, J.; Izquierdo, C. Regeneration of activated carbons contaminated by phenol using supercritical water. J. Supercrit. Fluids 2013, 74, 1–7. [Google Scholar] [CrossRef]
- Rivera-Utrilla, J.; Ferro-García, M.A.; Bautista-Toledo, I.; Sánchez-Jiménez, C.; Salvador, F.; Merchán, M.D. Regeneration of ortho-chlorophenol-exhausted activated carbons with liquid water at high pressure and temperature. Water Res. 2003, 37, 1905–1911. [Google Scholar] [CrossRef]
- Das, D.; Gaur, V.; Verma, N. Removal of volatile organic compound by activated carbon fiber. Carbon 2004, 42, 2949–2962. [Google Scholar] [CrossRef]
- Liu, Q.S.; Zheng, T.; Wang, P.; Jiang, J.P.; Li, N. Adsorption isotherm, kinetic and mechanism studies of some substituted phenols on activated carbon fibers. Chem. Eng. J. 2010, 157, 348–356. [Google Scholar] [CrossRef]
- Salvador, F.; Merchán, M.D. A simple apparatus for studies of thermoprogrammed desorption in solution. Langmuir 1992, 8, 1226–1229. [Google Scholar] [CrossRef]
- Sanchez-Montero, M.J.; Salvador, F.; Izquierdo, C. Reactivity and Porosity of a Carbon Fiber Activated with Supercritical CO2. J. Phys. Chem. C 2008, 112, 4991–4999. [Google Scholar] [CrossRef]
- Salvador, F.; Sánchez Montero, M.J.; Montero, J.; Izquierdo, C. Activated carbon fibers prepared from a phenolic fiber by supercritical water and steam activation. J. Phys. Chem. C 2008, 112, 20057–20064. [Google Scholar] [CrossRef]
- Montero, J.; de la Casa-Lillo, M.A.; Sanchez-Montero, M.J.; Martin-Sanchez, N.; Izquierdo, C.; Salvador, F. Effect of supercritical fluid activation on the diameter of carbon fibres. J. Supercrit. Fluids 2015, 101, 131–139. [Google Scholar] [CrossRef]
- Suzuki, M.; Misic, D.M.; Koyama, O.; Kawazoe, K. Study of thermal regeneration of spent activated carbons: Thermogravimetric measurement of various single component organics loaded on activated carbons. Chem. Eng. Sci. 1978, 33, 271–279. [Google Scholar] [CrossRef]
- Franz, M.; Arafat, H.A.; Pinto, N.G. Effect of chemical surface heterogeneity on the adsorption mechanism of dissolved aromatics on activated carbon. Carbon 2000, 38, 1807–1817. [Google Scholar] [CrossRef]
- Sabio, E.; González, E.; González, J.F.; González-García, C.M.; Ramiro, A.; Gañan, J. Thermal regeneration of activated carbon saturated with p-nitrophenol. Carbon 2004, 42, 2285–2293. [Google Scholar] [CrossRef]
- Martin-Sanchez, N.; Soares, O.S.G.P.; Pereira, M.F.R.; Sanchez-Montero, M.J.; Figueiredo, J.L.; Salvador, F. Oxidative dehydrogenation of isobutane catalyzed by an activated carbon fiber cloth exposed to supercritical fluids. Appl. Catal. A Gen. 2015, 502, 71–77. [Google Scholar] [CrossRef]
- Cooney, D.O.; Nagerl, A.; Hines, A.L. Solvent regeneration of activated carbon. Water Res. 1983, 17, 403–410. [Google Scholar] [CrossRef]
- Sutikno, T.; Himmelstein, K.J. Desorption of phenol from activated carbon by solvent regeneration. Ind. Eng. Chem. Fundam. 1983, 22, 420–425. [Google Scholar] [CrossRef]
- Tamon, H.; Saito, T.; Kishimura, M.; Okazaki, M.; Toei, R. Solvent regeneration of spent activated carbon in wastewater treatment. J. Chem. Eng. Jpn. 1990, 23, 426–432. [Google Scholar] [CrossRef]
- Martin, R.J.; Ng, W.J. The repeated exhaustion and chemical regeneration of activated carbon. Water Res. 1987, 21, 961–965. [Google Scholar] [CrossRef]
- Martin-Sanchez, N.; Sanchez-Montero, M.J.; Izquierdo, C.; Salvador, F. Conversion of a resistant pollutant, phenol, into green fuels by gasification using supercritical water compressed up to 1000 bar. React. Chem. Eng. 2017, 2, 799–810. [Google Scholar] [CrossRef]
| Sample | SBET (m2·g−1) | V0(N2) (cm3·g−1) | Vmeso (cm3·g−1) | V0(CO2) (cm3·g−1) | Mass Losses (%) |
|---|---|---|---|---|---|
| Original | 1030 | 0.511 | 0.008 | 0.397 | - |
| SCCO2.250 | 1112 | 0.524 | 0.002 | 0.393 | Negligible |
| SCCO2.700 | 1300 | 0.540 | 0.001 | 0.384 | 8 |
| SCW.650 | 1438 | 0.590 | 0.001 | 0.341 | 15 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sanchez-Montero, M.J.; Pelaz, J.; Martin-Sanchez, N.; Izquierdo, C.; Salvador, F. Supercritical Regeneration of an Activated Carbon Fiber Exhausted with Phenol. Appl. Sci. 2018, 8, 81. https://doi.org/10.3390/app8010081
Sanchez-Montero MJ, Pelaz J, Martin-Sanchez N, Izquierdo C, Salvador F. Supercritical Regeneration of an Activated Carbon Fiber Exhausted with Phenol. Applied Sciences. 2018; 8(1):81. https://doi.org/10.3390/app8010081
Chicago/Turabian StyleSanchez-Montero, M. Jesus, Jennifer Pelaz, Nicolas Martin-Sanchez, Carmen Izquierdo, and Francisco Salvador. 2018. "Supercritical Regeneration of an Activated Carbon Fiber Exhausted with Phenol" Applied Sciences 8, no. 1: 81. https://doi.org/10.3390/app8010081
APA StyleSanchez-Montero, M. J., Pelaz, J., Martin-Sanchez, N., Izquierdo, C., & Salvador, F. (2018). Supercritical Regeneration of an Activated Carbon Fiber Exhausted with Phenol. Applied Sciences, 8(1), 81. https://doi.org/10.3390/app8010081





