Abstract
In the last few decades, the search for bioactive compounds or “target molecules” from natural sources or their by-products has become the most important application of the supercritical fluid extraction (SFE) process. In this context, the present research had two main objectives: (i) to verify the effectiveness of a two-step SFE process (namely, a preliminary Sc-CO2 extraction of carotenoids followed by the recovery of polyphenols by ethanol coupled with Sc-CO2) in order to obtain bioactive extracts from two widespread different matrices (chili pepper and tomato by-products), and (ii) to test the validity of the mathematical model proposed to describe the kinetics of SFE of carotenoids from different matrices, the knowledge of which is required also for the definition of the role played in the extraction process by the characteristics of the sample matrix. On the basis of the results obtained, it was possible to introduce a simplified kinetic model that was able to describe the time evolution of the extraction of bioactive compounds (mainly carotenoids and phenols) from different substrates. In particular, while both chili pepper and tomato were confirmed to be good sources of bioactive antioxidant compounds, the extraction process from chili pepper was faster than from tomato under identical operating conditions.
1. Introduction
In the last two decades, compounds with antioxidant capacities have attracted increasing interest [1], in particular polyphenols, carotenoids and vitamins (mainly E and C). This specific attention derives from the ability of these compounds to scavenge free radicals and reactive oxygen species which are known to be involved in the development of cardiovascular diseases and several cancers [2]. Since one of the main sources of antioxidants are fruits and vegetables, these foodstuffs have gained great interest and widespread usage in the nutritional strategies applied to prevent these pathologies [2].
In this context, tomato (Lycopersicon esculentum L.) and pepper (Capsicum spp.), both belonging to the Solanaceae family, are considered to be important sources of natural carotenoids and phenols [3,4].
Among vegetables, tomato, which is consumed either as raw fruit or as a processed product, is the second most important vegetable crop in the world and one of the main components of the Mediterranean diet [5]. Furthermore, the industrial processing of tomato leads to by-products, namely tomato seeds and peels, representing 10–40% of total processed tomatoes [6]. The management of tomato by-products is considered an important problem faced by tomato processing companies, as they cannot be discharged into the environment due to their high polluting potential [4]. The bioactive compounds present in industrial tomatoes and their processing by-products include tocopherols, polyphenols, carotenoids (mainly lycopene), some terpenes, and sterols [7]. Thus, tomato wastes are a cheap resource to be recovered and recycled within the food chain, and a sustainable strategy able to address the current challenges of the industrialized world is required [8].
Besides tomato, red pepper (Capsicum spp.) is also an important vegetable consumed worldwide. Due to their circulatory stimulant functions, chili peppers (Capsicum annuum L.) are of ethnopharmacological importance and are also widely used as fresh fruits and savoury food additives due to their colour, pungency, and aroma [9,10,11]. Furthermore, the presence of many bioactive components such as vitamin C, phenolics and carotenoids [11,12,13,14,15,16] makes peppers extremely attractive for the phytochemical manufacturing industry as well. In particular, among carotenoids, which increase in concentration greatly during pepper maturation [14], the most representative are α and β-carotene, β-cryptoxanthin, capsanthin, lutein, and zeaxanthin [16]. The phenolic fraction includes mainly phenolic acids (cinnamic acid derivatives and hydroxy-substituted benzoic acids) and flavonoids (e.g., quercetin and luteolin) [10,15].
In this context, the recovery of bioactive compounds (mainly carotenoids and polyphenols) from tomato wastes and peppers requires the use of mild extraction technologies, able to preserve the nutritional and pharmacological properties of these molecules, but also their antioxidant power [17,18]. Conventional Solvent Extraction (CSE), such as organic solvent extraction, has been widely used to extract carotenoids and/or phenols from plant material. Traditionally, CSE used n-hexane, propanol, methanol, tetrahydrofuran or ethyl acetate to extract carotenoids. This method usually requires long extraction times, large amounts of organic solvents and high temperatures, which can lead to extensive degradation of thermo-sensible molecules, as well as leave trace amounts of potentially toxic solvents in the extract [17]. Moreover, the sustainability of the extraction process and the purification of the bioactive compounds is of the utmost importance [19].
Nowadays, supercritical fluid extraction (SFE) employing Sc-CO2 is an established industrial process for the production of high added-value products. In 2014 there were more than 150 SFE industrial plants with a total extraction volume of more than 500 L in the world [19]. Many of these production plants are generally devoted to the SFE process involving a preliminary Sc-CO2 extraction of natural products, leading to the recovery of high-value products which provide interesting options for their use in the nutraceutical and functional food industry [20].
In the last decades, the search for bioactive compounds or “target molecules” from natural sources or their by-products has become the most important application of SFE technology [21,22,23,24].
As reported in the literature [21], the bioactivities from natural compounds extracted by SFE from 2010 to 2015 were mainly the antioxidant (41%), antitumor (18%), and antibacterial (10%) ones. As widely reported [25,26] SFE is a green technology that shows immediate advantages over traditional extraction techniques: (i) it is a flexible process due to the possibility of continuous modulation of the solvent power/selectivity of the supercritical fluid (SF); (ii) it allows the removal of polluting organic solvents as well as that of the expensive extract post-processing used for solvent elimination, thus ensuring a safe separation process both for human health and the environment.
Based on the work of Melo et al. [26] and references within, it is possible to affirm that the last decades have seen great advances; among them full characterization and quantification of supercritical extracts, assessment of kinetic and equilibrium aspects, and phenomenological modelling and optimization of operating conditions. In particular, many authors have studied the dependence of the solubility of different carotenoids in supercritical CO2 with temperature and pressure [26,27]. Most of the solubility data of these works were correlated using the semi-empirical Chrastil’s model [28], which provides a proportional relationship between the solubility and density of CO2 [27]. Furthermore, the majority of the SFE studies for the recovery of carotenoids have focused on tomato products and industrial tomato by-products, as they constitute a good source of lycopene, and, to a lower extent, of β-carotene [4].
Nonetheless, according to Melo et al. [26], solute-matrix interactions can be better understood and correctly taken into account by reliable predictive models. In such a context, to better analyze the experimental results and to optimize the working parameters (temperature, pressure, etc.), this extractive technology could greatly benefit from mathematical models that are not only suitable and reliable, but also easy to use.
Different phenomena as phase equilibrium, mass transfer, and flow of Sc-CO2 through packed beds, are differently involved in the kinetic models reported in the literature [29]. In many cases, the extraction of the first fraction of extracts is essentially limited by its solubility, whereas the extraction rate of the remaining fraction is limited by internal diffusion through the matrix [30].
As reported in previous papers, a simplified mathematical model was introduced to describe the time evolution of SFE of the lipid fraction from oilseeds [31,32,33] and microalgae [34,35,36]. Using Chrastil’s equation [28], it was also possible to correlate the maximum extraction rate with both working pressures and temperatures.
On this basis, a simplified method to estimate the time evolution of carotenoid extraction by Sc-CO2 from two different matrices (chili peppers and tomato by-products) can be developed in order to highlight the “matrix effect” on the SFE of carotenoids. This new method could potentially simplify the identification of the best working conditions to promote SFE of carotenoids from these two matrices as a function of temperature, pressure, flow-rate of Sc-CO2, and amount of matrix, and to reduce the load of the related experimental activity [29,37].
Furthermore, in a previous paper [31] we reported on the use of a pilot scale SFE apparatus aimed at studying a two-sequential step procedure to intensify the extraction of oil and phenolic compounds from sunflower seeds.
In this context, this research had two main objectives: (i) to verify the effectiveness of a two-step SFE process (a preliminary Sc-CO2 extraction of carotenoids followed by the recovery of polyphenols with ethanol coupled with Sc-CO2) to obtain bioactive extracts from two different matrices (chili pepper and tomato by-products); (ii) to test the validity of the mathematical model proposed to describe the kinetics of SFE of carotenoids, the knowledge of which is required to establish the role played in the extraction process by the characteristics of the matrix.
2. Materials and Methods
As reported in a previous paper [36], SFE were performed using a commercial pilot plant apparatus (Sitec, Maur, Switzerland) which allows the recovery and the subsequent recycling of the solvent, with a minimal loss of CO2. A supplementary pump provides the addition of a co-solvent to the CO2 stream, when desired.
In order to obtain tomato peels as by-products, fresh fruits of L. esculentum L. were washed, cut, and parenchyma, seeds and percolation juice were removed. Then, both tomato peels and fresh fruits of C. annuum L. were lyophilized and ground to a particle size of 0.37 mm. All the samples were then stored under inert atmosphere (N2) and protected from light until use.
SFE of the carotenoidic fraction was performed using 280 g of lyophilized material for each run, with working pressures (P) of 40 and 70 MPa and temperatures (T) of 40 °C and 60 °C. The extraction time was 180 min, while the flow rate of Sc-CO2 was 10 kg·h−1. Extraction yields were determined gravimetrically, while the carotenoid concentration in extracts and lyophilized fruits was determined spectrophotometrically (Cary 300 UV-Vis, Agilent Technologies, Santa Clara, CA, USA) [38]. In particular, samples were first solubilized in hexane:acetone:ethanol 2:1:1 (by volume), shaken for 30 min, then distilled water was added and the samples were left to separate as a function of their polarity; the content of carotenoids was then obtained by measuring the specific absorbance and expressed as β-carotene (λ = 479 nm) for pepper and as lycopene (λ = 472 nm) for tomato. Extraction of carotenoids by percolation with n-hexane for 180 min was also performed using a Soxhlet apparatus (SER 148-3, Velp Scientifica, Usmate, Italy).
The recovery of the phenolic fraction was carried out using the samples resulting from carotenoid SFE and left in the extractor.
As reported in the literature [16], pure Sc-CO2 is a poor solvent for these polar compounds and water is not suitable as co-solvent because, in the operating conditions adopted, its very reduced presence in the homogeneous phase is unable to significantly modify the polarity of the final Sc-CO2/water mixture; thus, ethanol (EtOH) was used, both coupled to Sc-CO2 (50% w/w) and in pure form. Extractions were carried out using a pressure (P) of 30 MPa and 50 MPa and a temperature (T) of 50 °C and 80 °C. Because of the different EtOH/CO2 ratios used, variable solvent flow rates were employed, whereas the extraction time was fixed to a maximum of 180 min. Extraction yields were determined gravimetrically, while the polyphenolic concentration in both extracts and lyophilized fruits was determined spectrophotometrically according to the Folin-Ciocalteau method [39], and expressed as chlorogenic acid (λ = 765 nm). Extraction of polyphenols by percolation with pure EtOH for 180 min was also performed using a Soxhlet apparatus.
All the reagents utilized were provided by Sigma Aldrich s.r.l. Milano, Italy, while the CO2 was supplied by SOL s.p.a, Monza, Italy.
3. Results
3.1. Kinetic Evaluation of Carotenoid Extraction
Applying the Fick’s law to the diffusion between two heterogeneous phases (solid matrix and Sc-CO2), the carotenoid accumulation rate (d[Ce]t=t/dt) in the mobile phase (Sc-CO2) was assumed to be described by the following equation:
where ke = mass transfer constant (s−1·m−2); A = area of contact between the two phases: solid matrix and Sc-CO2 (m2); [Ce]t=t = concentration of carotenoids already extracted at time t=t (mg/g); [Ce*]t=t = H∙[Cs]t=t = concentration of carotenoids already extracted if, at that time t=t, the equilibrium between the two phases involved has been reached (mg/g); H = the equilibrium constant related to the partition of carotenoids between the two phases (H = [Ce*]t=t/[Cs]t=t); [Cs]t=t = concentration of unextracted carotenoids at time t=t (mg/g).
The differential Equation (1) can be integrated by adopting some simplifying hypothesis [37] and utilizing the equation related to the mass balance of carotenoids between the two phases:
where [Cs]t=0 = carotenoid concentration initially present in one gram of starting material (mg/g).
Then Equations (3) and (4) could be obtained:
where H* = an adimensional constant ranging from zero to one, related to the equilibrium constant H (H* = H/(H + 1)); k = (H + 1) × ke × A = kinetic constant (s−1).
The extraction rate (R) calculated as first derivative of the exponential equation:
reaches its maximum value (Rmax) at the beginning of the extraction, when t is close to zero:
As in Yu et al. [40], the value of Rmax (s−1) was assumed as an index to evaluate the efficiency of the SFE system versus the carotenoid fraction of the matrix. In particular, while the constant k provides information on the kinetics of the SFE, the product H* × [Cs], which represents the asymptotic value of the extraction curve when t → ∞, measures the maximum amount of extractable carotenoids under the working conditions adopted.
As reported in a previous paper [41], the identification of the best values to be assigned to the equation parameters H* × [Cs] and k was carried out by a commercial statistical program (BURENL©, CMIC, Politecnico Milano, Italy, 1996).
Table 1 reports the values assumed by Rmax and by the functional parameters H* × [Cs] and k for the two matrices (tomato peels and chili pepper).

Table 1.
Extraction of carotenoids from pepper and tomato. Values were assumed by Rmax and the equation parameters k and H* × [Cs] as a function of the working conditions. T = temperature; P = pressure; r2 = square of correlation coefficient (p = 0.05).
Figure 1 reports the time evolution of carotenoid extraction from pepper (Figure 1a) and tomato peels (Figure 1b) as a function of the working conditions, together with the data calculated on the basis of the functional parameters reported in Table 1.
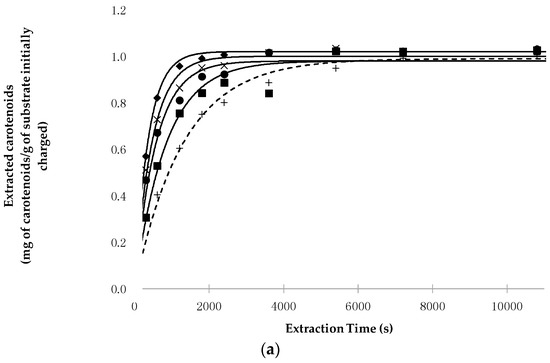
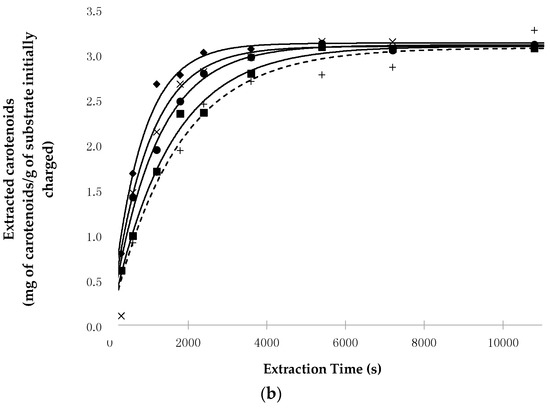
Figure 1.
(a) Evolution of the experimental and calculated points related to carotenoid extraction from lyophilized fruits of C. annuum L. as a function of extraction time and working conditions (■ 40 °C, 40 MPa; × 40 °C, 70 MPa; ● 60 °C, 40 MPa; ◆ 60 °C, 70 MPa; + Soxhlet extraction n-hexane); (b) Evolution of the experimental and calculated points related to carotenoid extraction from lyophilized peels of L. esculentum L. as a function of extraction time and working conditions (■ 40 °C, 40 MPa; × 40 °C, 70 MPa; ● 60 °C, 40 MPa; ◆ 60 °C, 70 MPa; + Soxhlet extraction n-hexane).
On the basis of the reported data, regardless of the raw material used, the following remarks can be made:
- (a)
- The increase in pressure and temperature does not significantly influence the total amount of carotenoids extractable at equilibrium (extraction time = ∞), as shown by the values assumed by the H* × [Cs] parameter. Indeed, this product—which represents the yield in milligrams of carotenoids extracted from one gram of lyophilized material when the equilibrium is reached—assumes that for all the SFE runs, the values used were close to the concentration of carotenoids in the starting material (1016 mg/kg for pepper and 3125 mg/kg for tomato peels) and also to the concentrations obtained with percolation with n-hexane;
- (b)
- P highly affects the extraction kinetics, as confirmed by the values of the k constant when working at the same T;
- (c)
- Secondarily, T also affects the kinetics of the SFE, as shown by the value that k assumes when working at the same P;
- (d)
- The binomial pressure-temperature combination is more crucial than T and P alone in determining the kinetics of the SFE process. In particular, transitioning from the mildest (40 °C and 40 MPa) to the most intense (60 °C and 70 MPa) working conditions, the value of k increases 2.3- and 1.9-fold for chili peppers and tomato peels, respectively.
- (e)
- In all conditions, extraction with Sc-CO2 was completed faster than percolation with n-hexane, as shown by the values assumed by k and/or Rmax. For example, the ratio between the value of k obtained at the most intense SFE conditions and that of the Soxhlet extraction was about 3.5 and 2.2 for chili pepper and tomato peels, respectively.
These considerations, together with the high values of the square of the correlation coefficient, demonstrate the suitability of the hypotheses introduced and give a measure of validity for the mathematical model proposed.
As described by Zinnai et al. [37], in order to relate Rmax to the Sc-CO2 density, which is influenced by both pressure and temperature, the following equation introduced by Chrastil was used:
where R*max = maximum value assumed by the extraction rate expressed in grams of extracted carotenoids per liter of Sc-CO2 flowed through the substrate bed (g/L); Rmax = the maximum value of the extraction rate (s−1); m = amount of substrate (g); ρ = density of Sc-CO2; (g/L); Φ = flow rate of Sc-CO2 (g/s); a, b, c = equation parameters; T = temperature (K).
Table 2 reports the value of the parameters a, b and c of both matrices calculated by the statistical program BURENL© using the values assumed by Rmax as a function of the working conditions adopted (Table 1).

Table 2.
Values of parameters involved in Chrastil’s equation, used to correlate the solvent power of Sc-CO2 to its density and working temperatures. c.i. = confidence interval (p = 0.05); r2 = square of correlation coefficient.
The high values assumed by the square of the correlation coefficients give a measure of the validity of Chrastil’s equation in order to describe the evolution of the maximum extraction rate as a function of both temperature and pressure. Knowing the values assumed by the three functional parameters (a, b, c) and the working temperature and pressure, it is possible to calculate the corresponding R*max value and then that of the kinetic constant k.
In fact, combining Equation (6) with (7), the following expression can be obtained:
The value of the density assumed by Sc-CO2 (ρSc-CO2, T; P) as a function of temperature and pressure used can be evaluated by examining the state diagram of carbon dioxide.
Thus, for the two substrates it was possible to calculate the theoretical evolution of the kinetic constant k related to Sc-CO2 extraction of carotenoids, as a function of temperature and pressure (see Table 3).

Table 3.
Values calculated for the kinetic constant (k) by Chrastil’s equation and related to carotenoid extraction from pepper and tomato peels as a function of pressure of Sc-CO2 (P = 40; 70 MPa) and temperature (T = 40; 60 °C).
The proposed mathematical model allowed us to analyze the role played by the matrix in the SFE of carotenoids. In particular, regardless of the experimental conditions, the values attributed to the kinetic constant k for carotenoids doubled when chili pepper was utilized as starting material, in comparison with the calculated value for tomato peels at the same P and T.
3.2. Kinetic Evaluation of Polyphenol Extraction
The same approach based on Fick’s law and used to describe the kinetics of carotenoid SFE, was used to optimize the working parameters related to the extraction of polyphenols from pepper and tomato by-products with pure EtOH or Sc-CO2 coupled with EtOH (1:1, w/w). In particular, the following equation was used to determine the maximum extraction rate (R′max):
where: k′ = kinetic constant (s−1); H′* = an adimensional constant, ranging from zero to one, and related to the equilibrium constant H′; [Ps] = polyphenolic concentration in the starting lyophilized material (adimensional).
The values assumed by R′max and the functional parameters k′ and H′* × [Ps] are reported in Table 4.

Table 4.
Extraction of polyphenols from pepper and tomato by-products. Values assumed by R′max and the equation parameters k′ and H′* × [Ps] as a function of the working conditions. T = temperature; P = pressure; r = correlation coefficient.
Figure 2 shows the time evolution of polyphenols extracted from pepper (Figure 2a) and from tomato peels (Figure 2b) as a function of the working conditions, together with the data calculated on the basis of the kinetic parameters reported in Table 4.
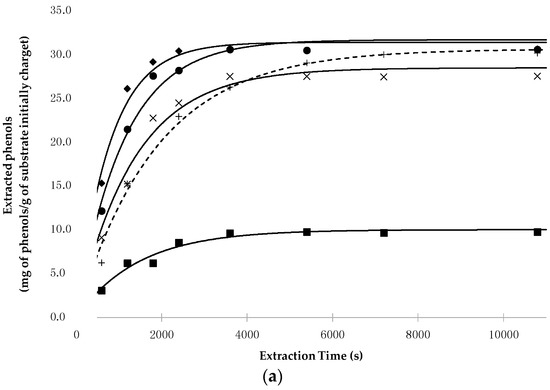
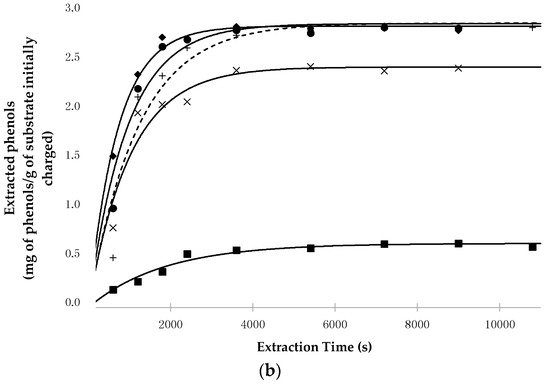
Figure 2.
(a) Evolution of the experimental and calculated points related to the phenol extraction from lyophilized fruits of C. annuum L. as a function of extraction time and working conditions (■ 50 °C, 30 MPa, Sc-CO2/EtOH (1:1, w/w); × 80 °C, 30 MPa, Sc-CO2/EtOH (1:1, w/w); ● 80 °C, 30 MPa, EtOH 100%; ◆ 80 °C, 50 MPa, EtOH 100%; + Soxhlet extraction, EtOH 100%); (b) Evolution of the experimental and calculated points related to phenol extraction from lyophilized peels of L. esculentum L. as a function of extraction time and working conditions (■ 50 °C, 30 MPa, Sc-CO2/EtOH (1:1, w/w); × 80 °C, 30 MPa, Sc-CO2/EtOH (1:1, w/w); ● 80 °C, 30 MPa, EtOH 100%; ◆ 80 °C, 50 MPa, EtOH 100%; + Soxhlet extraction, EtOH 100%).
On the basis of the data reported in Table 4, regardless of the raw material utilized, the following remarks can be made:
- (a)
- As previously observed with potato by-products [42], pure Sc-CO2 was confirmed to be a poor solvent for polyphenols, even when high density values were used;
- (b)
- EtOH is a suitable co-solvent used to pilot the polarity of the solvent phase, provided that high percentages are utilized (≥50%);
- (c)
- At an equal EtOH/Sc-CO2 ratio (1:1) and P (30 MPa), T greatly affects the extraction process, with particular reference to the total amount of extractable polyphenols. Indeed, while the kinetic constant k′ does not change markedly when T increases from 50 °C to 80 °C, the equation parameter H′* × [Ps] increases almost three-fold for chili peppers and four-fold for tomato by-products, from 50 °C to 80 °C. This means that such temperature increases determine the solubilization of phenolic compounds that are otherwise not easily collectable;
- (d)
- To obtain the extraction of the whole phenolic fraction, pure EtOH at 80 °C, and 30 MPa or 50 MPa conditions were needed. In such conditions, the extraction process was faster than when it was carried out using the Soxhlet apparatus, as shown by the values of the kinetic constant k′. For example, working at 80 °C and 30 MPa, the ratio between the value of Rmax obtained in SFE conditions and of the Soxhlet extraction, was about 2.0 and 1.3 for chili pepper and tomato peels, respectively;
- (e)
- At 80 °C and 30 MPa, if EtOH decreased from 100% to 50%, only a small decrease in the extraction yield and kinetics was observed.
4. Conclusions
As widely reported in the literature [26,27,28,29], SFE is based on the solvating properties of SF. In particular, the extraction by SF depends on a tuneable nature of SF like temperature, pressure and some extrinsic features like the characteristics of the sample matrix, interactions with targeted analysts, and many environmental factors [43,44].
In this context, on the basis of the results obtained, it was possible to introduce a simplified kinetic model that was able to describe the time evolution of extraction of bioactive compounds (mainly carotenoids and phenols) from different substrates. Moreover, the utilization of this simplified kinetic model together with the Chrastil’s equation allowed the prediction of the time evolution of SFE as a function of the main working conditions adopted.
The high values assumed by the square of the correlation coefficient seemed to confirm the suitability of the hypotheses introduced and gave a measure of the validity of the kinetic model proposed.
In particular, while both C. annuum L. and L. esculentum L. were confirmed as good sources of bioactive antioxidant compounds, when the same operating conditions (T, P, pre-treatments carried out on the substrate) were used, the extraction process of both carotenoids and phenols from C. annuum L. was significantly faster. The obtained results allowed us to tentatively describe the role played by the matrix in SFE of bioactive compounds.
Acknowledgments
We thank our colleague Mike F. Quartacci for the language check of the manuscript.
Author Contributions
F.V. and A.Z. conceived and designed the experiments; C.S. and I.T. performed the experiments; C.S. and G.A. analyzed the data; F.V. wrote the paper.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Prior, R.L.; Wu, X.; Schaich, K. Standardized methods for the determination of antioxidant capacity and phenolics in foods and dietary supplements. J. Agric. Food Chem. 2005, 53, 4290–4302. [Google Scholar] [CrossRef] [PubMed]
- Le Grandois, J.; Guffond, D.; Hamon, E.; Marchioni, E.; Werner, D. Combined microplate-ABTS and HPLC-ABTS analysis of tomato and pepper extracts reveals synergetic and antagonist effect of their lipophilic antioxidative components. Food Chem. 2017, 223, 62–71. [Google Scholar] [CrossRef] [PubMed]
- Arimboor, R.; Natarajan, R.B.; Menon, K.R.; Chandrasekhar, L.P.; Moorkoth, V. Red pepper (Capsicum annuum) carotenoids as a source of natural food colors: Analysis and stability—A review. J. Food Sci. Technol. 2015, 52, 1258–1271. [Google Scholar] [CrossRef] [PubMed]
- Strati, I.F.; Oreopoulou, V. Recovery of carotenoids from tomato processing by-products—A review. Food Res. Int. 2014, 65, 311–321. [Google Scholar] [CrossRef]
- FAOSTAT. 2005. Available online: http://faostat.fao.org (accessed on 25 March 2006).
- Al-Wandawi, H.; Abdul-Rahman, M.; Al-Shaikhly, K. Tomato processing wastes as essential raw materials source. J. Agric. Food Chem. 1985, 33, 804–807. [Google Scholar] [CrossRef]
- Kalogeropoulos, N.; Chiou, A.; Pyriochou, V.; Peristeraki, A.; Karathanos, V.T. Bioactive phytochemicals in industrial tomatoes and their processing byproducts. LWT Food Sci. Technol. 2012, 49, 213–216. [Google Scholar] [CrossRef]
- Pinela, J.; Prieto, M.A.; Barreiro, M.F.; Carvalho, A.M.; Oliveira, M.B.P.P.; Curran, T.P.; Ferreira, I.C.F.R. Valorisation of tomato wastes for development of nutrient-rich antioxidant ingredients: A sustainable approach towards the needs of the today’s society. Innov. Food Sci. Emerg. Technol. 2017. [Google Scholar] [CrossRef]
- Walsh, B.M.; Hoot, S.B. Phylogenetic relashionships of Capsicum (Solanaceae) using DNA sequence from two noncoding regions: The chloroplast atpB-rbcL spacer regions and nuclear waxy introns. Int. J. Plant Sci. 2001, 162, 1409–1418. [Google Scholar] [CrossRef]
- Khan, F.A.; Mahmood, T.; Ali, M.; Saeed, A.; Maalik, A. Pharmacological importance of an ethnobotanical plant: Capsicum annuum L. Nat. Prod. Res. 2014, 28, 1267–1274. [Google Scholar] [CrossRef] [PubMed]
- De Aguiar, A.C.; Sales, L.P.; Coutinho, J.P.; Barbero, J.F.; Godoy, H.B.; Martínez, J. Supercritical carbon dioxide extraction of Capsicum peppers: Global yield and capsaicinoid content. J. Supercrit. Fluids 2013, 81, 210–216. [Google Scholar] [CrossRef]
- Materska, M.; Perucka, I. Antioxidant activity of the main phenolic compounds isolated from hot pepper fruit (Capsicum annuum L.). J. Agric. Food Chem. 2005, 53, 1750–1756. [Google Scholar] [CrossRef] [PubMed]
- Topuz, A.; Ozdemir, F. Assessment of carotenoids, capsaicinoids and ascorbic acid composition of some selected pepper cultivars (Capsicum annuum L.) grown in Turkey. J. Food Compos. Anal. 2007, 20, 596–602. [Google Scholar] [CrossRef]
- Deepa, N.; Kaur, C.; George, B.; Singh, B.; Kapoor, H.C. Antioxidant constituents in some sweet pepper (Capsicum annuum L.) genotypes during maturity. LWT Food Sci. Technol. 2007, 40, 121–129. [Google Scholar] [CrossRef]
- Jeong, W.Y.; Jin, J.S.; Cho, Y.A.; Lee, J.H.; Park, S.; Jeong, S.W.; Kim, Y.; Lim, C.; Abd El-Aty, A.M.; Kim, G.; et al. Determination of polyphenols in three Capsicum annuum L. (bell pepper) varieties using high-performance liquid chromatography-tandem mass spectrometry: Their contribution to overall antioxidant and anticancer activity. J. Sep. Sci. 2011, 34, 2967–2974. [Google Scholar] [PubMed]
- Meireles, M.A.A. Extracting Bioactive Compounds for Food Products: Theory and Applications, 1st ed.; CRC Press: Boca Raton, FL, USA, 2008; pp. 288–326. [Google Scholar]
- Durante, M.; Lenucci, M.S.; Mita, G. Supercritical Carbon Dioxide Extraction of Carotenoids from Pumpkin (Cucurbita spp.): A Review. Int. J. Mol. Sci. 2014, 15, 6725–6740. [Google Scholar] [CrossRef] [PubMed]
- Maadane, A.; Merghoub, N.; Ainane, T.; ElArroussi, H.; Benhima, R.; Amzazi, S.; Bakri, Y.; Wahby, I. Antioxidant activity of some Moroccan marine microalgae: Pufa profiles, carotenoids and phenolic content. J. Biotechnol. 2015, 215, 13–19. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Camargo, A.P.; García-Cañas, V.; Herrero, M.; Cifuentes, A.; Ibáñez, E. Comparative study of green sub- and supercritical processes to obtain carnosic acid and carnosol-enriched rosemary extracts with in vitro anti-proliferative activity on colon cancer cells. Int. J. Mol. Sci. 2016, 17, 2046. [Google Scholar] [CrossRef] [PubMed]
- Campardelli, R.; Baldino, L.; Reverchon, E. Supercritical fluids applications in nanomedicins. J. Supercrit. Fluids 2015, 101, 193–214. [Google Scholar] [CrossRef]
- Da Silva, R.P.F.F.; Rocha-Santos, T.A.P.; Duarte, A.C. Supercritical fluid extraction of bioactive compounds. TrAC Trends Anal. Chem. 2016, 76, 40–51. [Google Scholar] [CrossRef]
- Machado, B.A.S.; Pereira, C.G.; Nunes, S.B.; Padilha, F.F.; Umsza-Guez, M.A. Supercritical fluid extraction using CO2. Main applications and future perspectives. Sep. Sci. Technol. 2013, 48, 2741–2760. [Google Scholar] [CrossRef]
- Herrero, M.; Castro-Puyana, M.; Mendiola, J.A.; Ibañez, E. Compressed fluids for the extraction of bioactive compounds. TrAC Trends Anal. Chem. 2013, 43, 67–83. [Google Scholar] [CrossRef]
- Herrero, M.; Sánchez-Camargo, A.D.P.; Cifuentes, A.; Ibáñez, E. Plants, seaweeds, microalgae and food by-products as natural sources of functional ingredients obtained using pressurized liquid extraction and supercritical fluid extraction. TrAC Trends Anal. Chem. 2015, 71, 26–38. [Google Scholar] [CrossRef]
- Reverchon, E.; de Marco, I. Supercritical fluid extraction and fractionation of natural matter. J. Supercrit. Fluids 2006, 38, 146–166. [Google Scholar] [CrossRef]
- De Melo, M.M.R.; Silvestre, A.J.D.; Silva, C.M. Supercritical fluid extraction of vegetable matrices: Applications, trends and future perspectives of a convincing green technology. J. Supercrit. Fluids 2014, 92, 115–176. [Google Scholar] [CrossRef]
- Mattea, F.; Martín, Á.; Cocero, M.J. Carotenoid processing with supercritical fluids. J. Food Eng. 2009, 93, 255–265. [Google Scholar] [CrossRef]
- Chrastil, J. Solubility of Solids and Liquids in Supercritical Gases. J. Phys. Chem. 1982, 86, 3016–3021. [Google Scholar] [CrossRef]
- Sharif, K.M.; Rahman, M.M.; Azmir, J.; Mohamed, A.; Jahurul, M.H.A.; Sahena, F.; Zaidul, I.S.M. Experimental design of supercritical fluid extraction. J. Food Eng. 2014, 124, 105–116. [Google Scholar] [CrossRef]
- Sovová, H. Steps of Supercritical Fluid Extraction of Natural Products and Their Characteristic Times. J. Supercrit. Fluids 2012, 66, 73–79. [Google Scholar] [CrossRef]
- Andrich, G.; Balzini, S.; Zinnai, A.; de Vitis, V.; Silvestri, S.; Venturi, F.; Fiorentini, R. Supercritical fluid extraction in sunflower seed technology. Eur. J. Lipid Sci. Technol. 2001, 103, 151–157. [Google Scholar] [CrossRef]
- Andrich, G.; Zinnai, A.; Nottoli, S.; Venturi, F.; Fiorentini, R. A Mathematical Model Describing the Supercritical Fluid Extraction (SFE) of Oil from Soybean Seeds. Chem. Eng. Trans. 2002, 2, 397–401. [Google Scholar]
- Andrich, G.; Zinnai, A.; Venturi, F.; Fiorentini, R. A Mathematical Model Describing the Supercritical Fluid Extraction (SFE) of Rapeseed (Brassica Napus) Oil. Chem. Eng. Trans. 2003, 3, 1605–1610. [Google Scholar]
- Andrich, G.; Nesti, U.; Venturi, F.; Zinnai, A.; Fiorentini, R. Supercritical Fluid Extraction of Bioactive Lipids from Microalga Nannochloropsis sp. Eur. J. Lipid Sci. Technol. 2005, 107, 381–386. [Google Scholar] [CrossRef]
- Andrich, G.; Zinnai, A.; Nesti, U.; Venturi, F.; Fiorentini, R. Supercritical Fluid Extraction of Oil from Microalga (Arthrospira platensis). Acta Aliment. 2006, 35, 195–203. [Google Scholar] [CrossRef]
- Zinnai, A.; Sanmartin, C.; Taglieri, I.; Andrich, G.; Venturi, F. Supercritical fluid extraction from microalgae with high content of LC-PUFAs. A case of study: Sc-CO2 oil extraction from Schizochytrium spp. J. Supercrit. Fluids 2016, 116, 126–131. [Google Scholar] [CrossRef]
- Zinnai, A.; Venturi, F.; Sanmartin, C.; Andrich, G. A Simplified Method to Estimate the Time Evolution of Oil Extraction from Different Substrates by Supercritical CO2. Am. J. Anal. Chem. 2012. [Google Scholar] [CrossRef]
- Martinez-Valverde, I.; Periago, M.J.; Provan, G.; Chesson, A. Phenolic compounds, lycopene and antioxidant activity in commercial varieties of tomato (Lycopersicon esculentum). J. Sci. Food Agric. 2002, 82, 323–330. [Google Scholar] [CrossRef]
- Slinkard, K.; Singleton, V.L. Total phenol analysis: Automation and comparison with manual methods. Am. J. Enol. Vitic. 1997, 28, 49–55. [Google Scholar]
- Yu, Z.; Sing, B.; Rizvi, S.H.; Zollweg, J.A. Solubilities of fatty acids, fatty acid esters, triglycerides, and fats and oils in supercritical carbon dioxide. J Supercrit. Fluids. 1994, 7, 51–59. [Google Scholar] [CrossRef]
- Venturi, F.; Sanmartin, C.; Taglieri, I.; Xiaoguo, Y.; Andrich, G.; Zinnai, A. The influence of packaging on the sensorial evolution of white wine as a function of the operating conditions adopted during storage. Agrochimica 2016, 60, 150–160. [Google Scholar]
- Andrich, G.; Stevanin, E.; Zinnai, A.; Venturi, F.; Fiorentini, R. Extraction kinetics of natural antioxidants from potato industry by-products. In Proceedings of the VI Symposium on Supercritical Fluids, Versailles, France, 28–30 April 2003; Volume 1, pp. 159–163. [Google Scholar]
- Pereira, C.; Meireles, M.A. Supercritical fluid extraction of bioactive compounds: Fundamentals, applications and economic perspectives. Food Bioprocess Technol. 2010, 3, 340–372. [Google Scholar] [CrossRef]
- Cavalcanti, R.N.; Meireles, M.A.A. Fundamentals of supercritical fluid extraction. In Comprehensive Sampling and Sample Preparation; Janusz, P., Ed.; Academic Press: Oxford, UK, 2012; pp. 117–133. [Google Scholar]
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).