Photoacoustic Imaging in Oxygen Detection
Abstract
1. Introduction
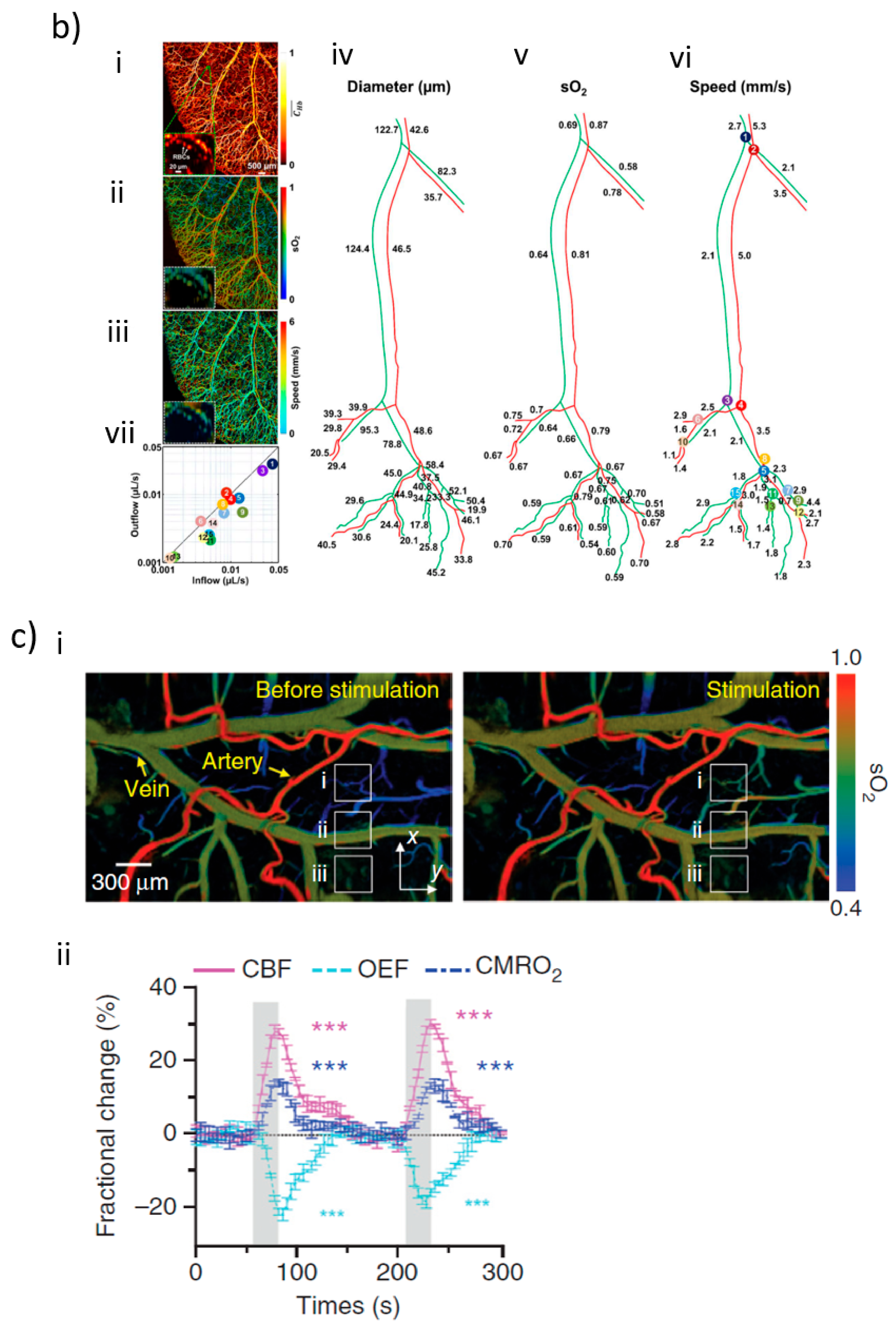
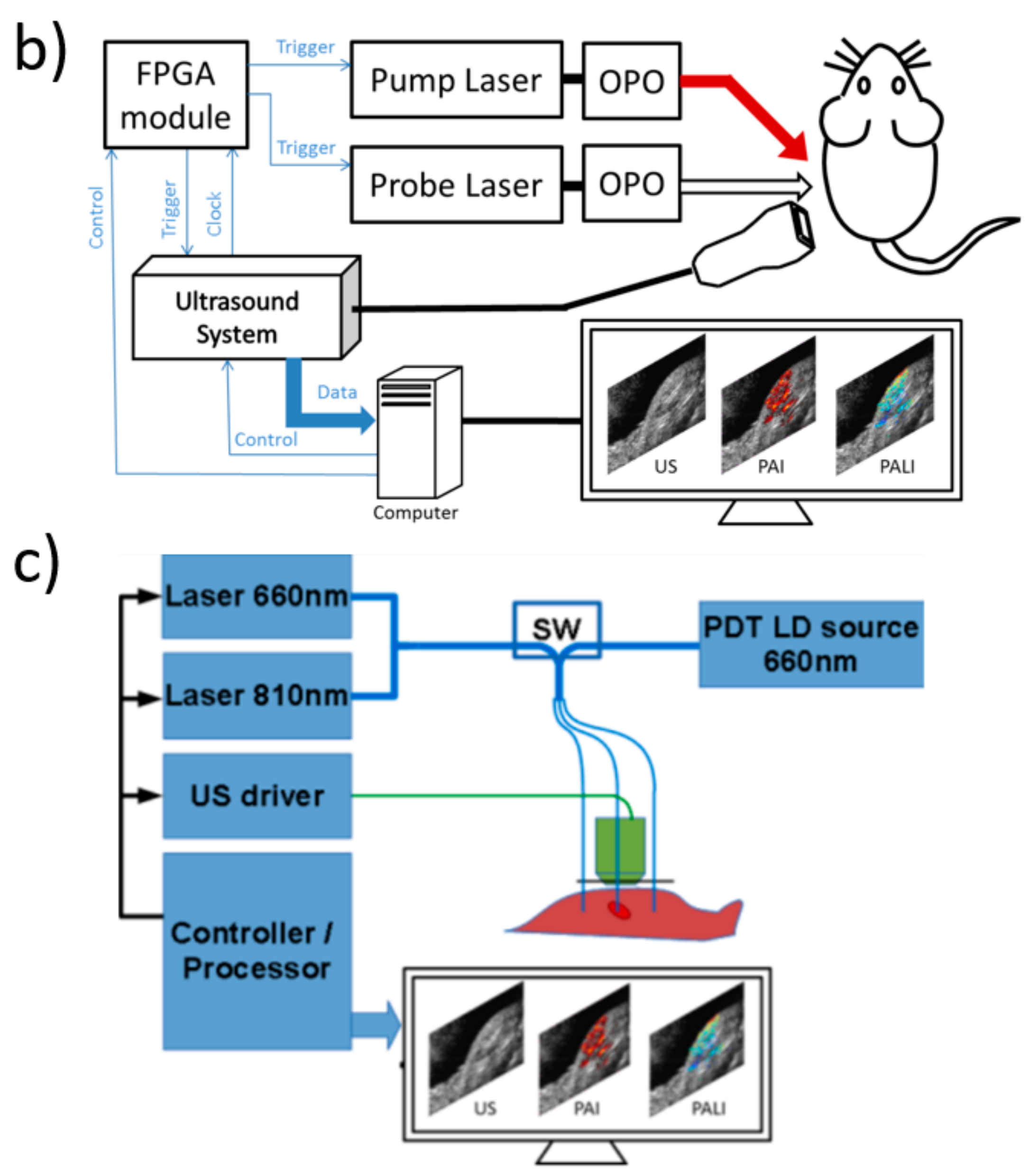
2. Photoacoustic Imaging in sO2 Measurement
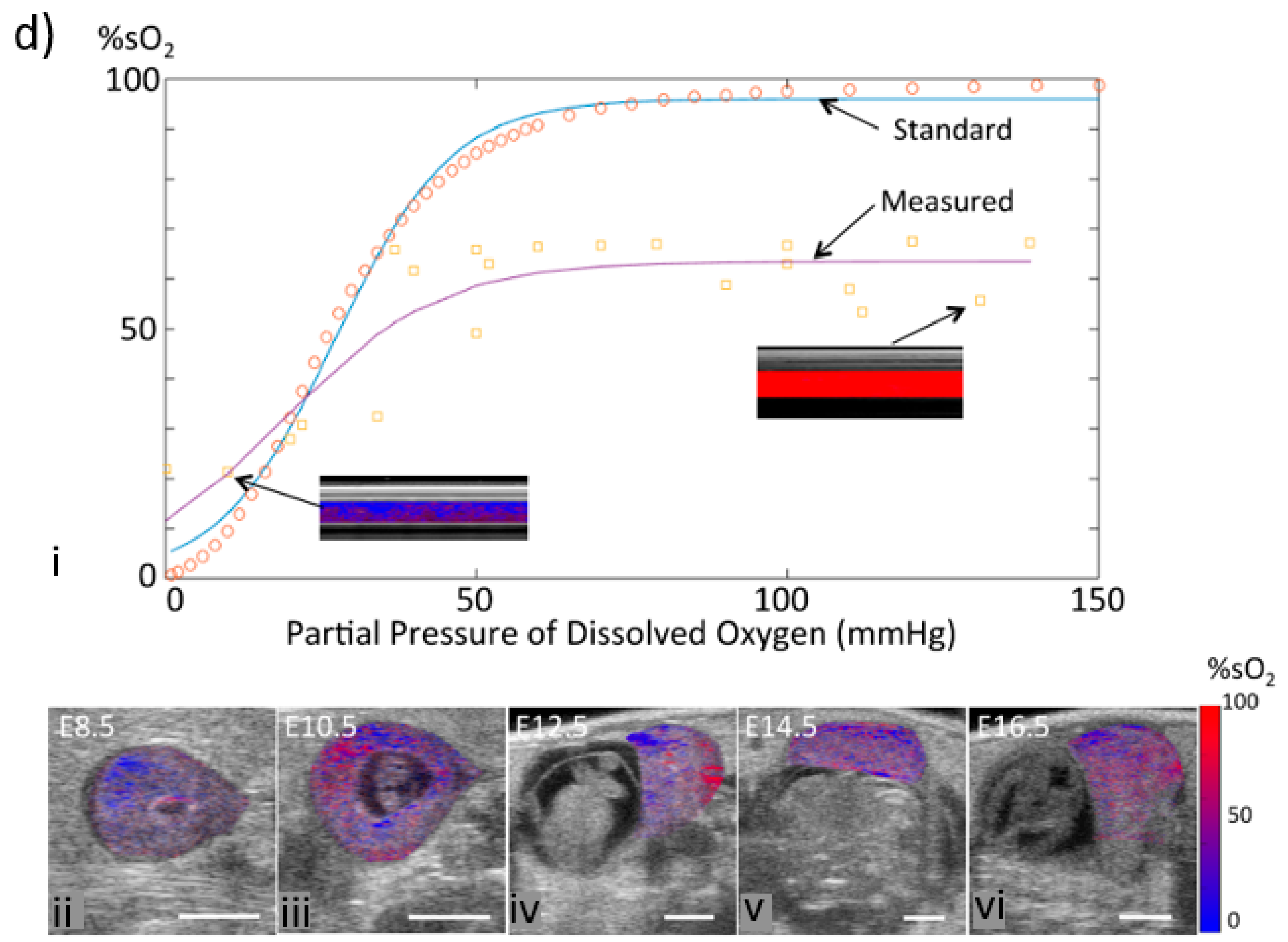
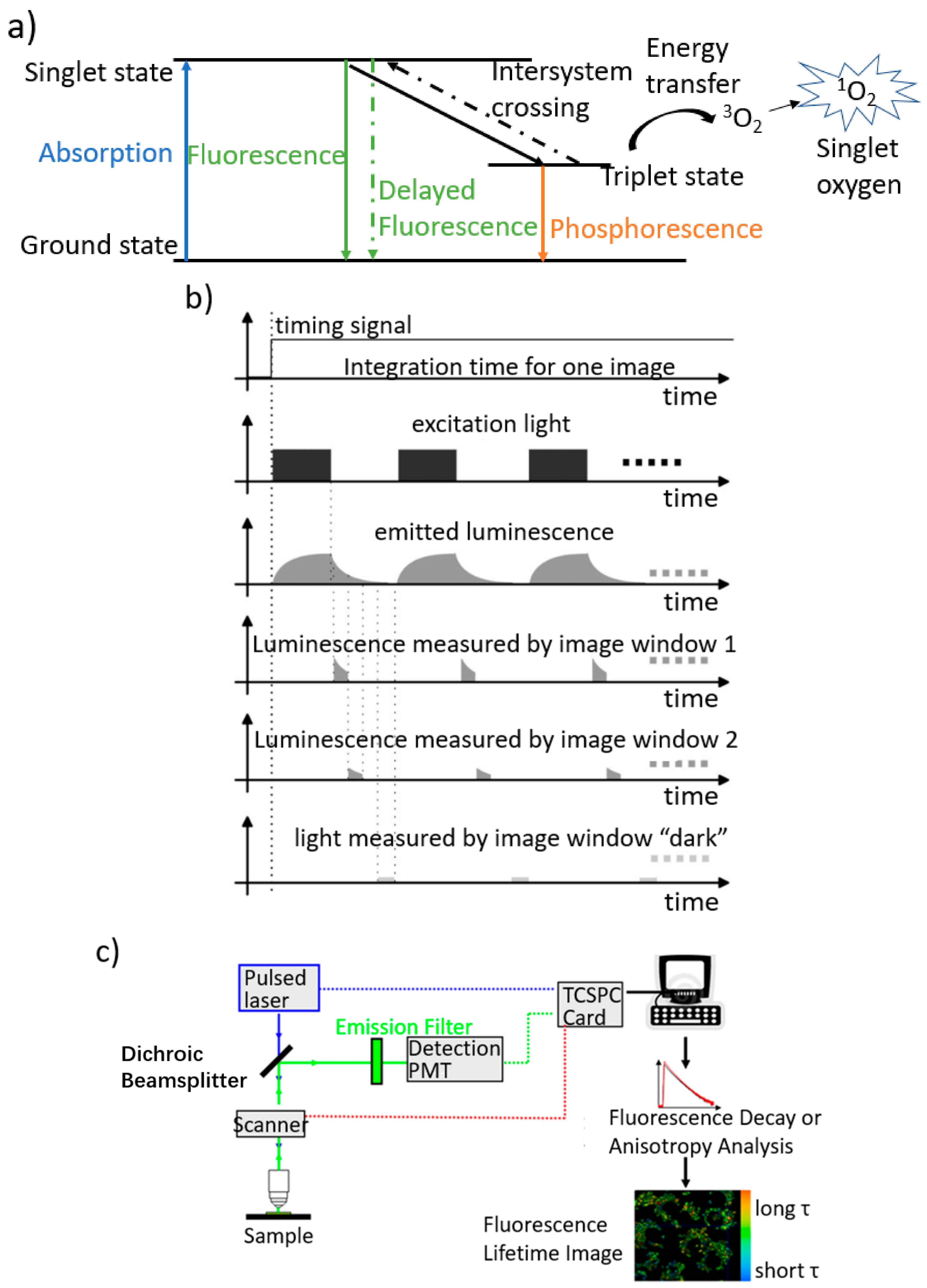
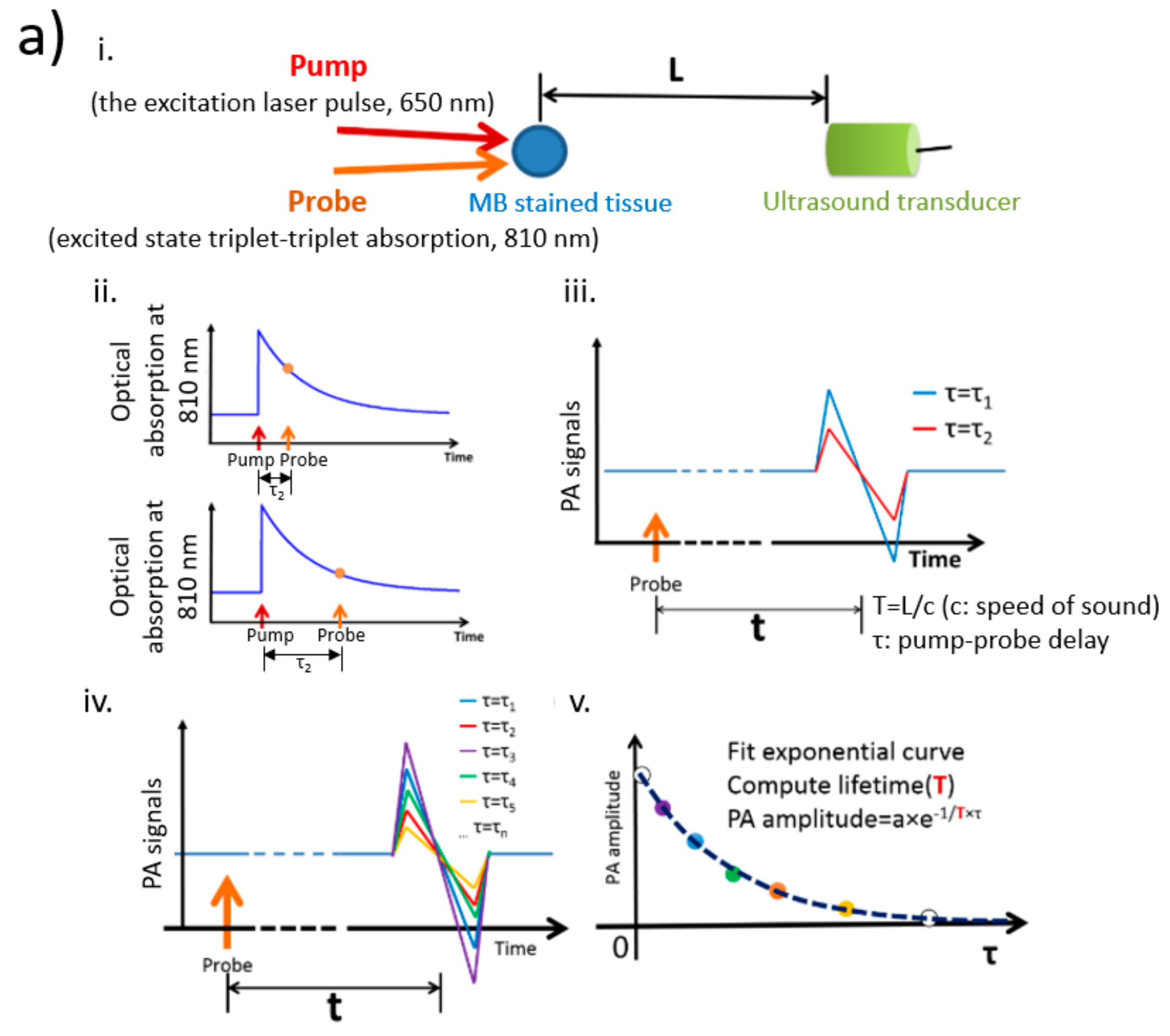
3. Photoacoustic Imaging in pO2 Measurement
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Giaccia, A.J.; Simon, M.C.; Johnson, R. The biology of hypoxia: The role of oxygen sensing in development, normal function, and disease. Genes Dev. 2004, 18, 2183–2194. [Google Scholar] [CrossRef] [PubMed]
- Fidler, I.J. Angiogenesis and cancer metastasis. Cancer J. 2000, 6, S134–S141. [Google Scholar] [PubMed]
- Hockel, M.; Vaupel, P. Tumor Hypoxia: Definitions and Current Clinical, Biologic, and Molecular Aspects. J. Natl. Cancer Inst. 2001, 93, 266–276. [Google Scholar] [CrossRef] [PubMed]
- Moen, I.; Stuhr, L.E.B. Hyperbaric oxygen therapy and cancer—A review. Target. Oncol. 2012, 7, 233–242. [Google Scholar] [CrossRef] [PubMed]
- Song, G.; Ji, C.; Liang, C.; Song, X.; Yi, X.; Domg, Z.; Yang, K.; Liu, Z. TaOx decorated perfluorocarbon nanodroplets as oxygen reservoirs to overcome tumor hypoxia and enhance cancer radiotherapy. Biomaterials 2017, 112, 257–263. [Google Scholar] [CrossRef] [PubMed]
- Littlejohns, L.R.; Bader, M.K.; March, K. Brain Tissue Oxygen Monitoring in Severe Brain Injury, I: Research and Usefullness in Critical Care. Crit. Care Nurse 2003, 23, 17–25. [Google Scholar] [PubMed]
- Rosenthal, G.; Hemphill, J.C., 3rd; Sonani, M.; Martin, C.; Morabito, D.; Obrist, W.D.; Manley, G.T. Brain tissue oxygen tension is more indicative of oxygen diffusion than oxygen delivery and metabolism in patients with traumatic brain injury. Crit. Care Med. 2008, 36, 1917–1924. [Google Scholar] [CrossRef] [PubMed]
- Sheffield, P.J. Measuring tissue oxygen tension: A review. Undersea Hyperb. Med. 1998, 25, 179–188. [Google Scholar] [PubMed]
- Diaz-Tena, E.; Barona, A.; Gallastegui, G.; López de Lacalle, L.N.; Elías, A. Biomachining: Metal etching via microorganisms. Crit. Rev. Biotechnol. 2017, 37, 323–332. [Google Scholar] [CrossRef] [PubMed]
- Díaz-Tena, E.; Gallastegui, G.; Hipperdinger, M.; Donati, E.R.; Ramírez, M.; Rodríguez, A.; López de Lacalle, L.N.; Elías, A. New advances in copper biomachining by iron-oxidizing bacteria. Corros. Sci. 2016, 112, 385–392. [Google Scholar] [CrossRef]
- Hummler, H.D.; Engelmann, A.; Pohlandt, F.; Högel, J.; Franz, A.R. Accuracy of pulse oximetry readings in an animal model of low perfusion caused by emerging pneumonia and sepsis. Intensive Care Med. 2004, 30, 709–713. [Google Scholar] [CrossRef] [PubMed]
- De Santis, V.; Singer, M. Tissue oxygen tension monitoring of organ perfusion: Rationale, methodologies, and literature review. Br. J. Anaesth. 2015, 115, 357–365. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.V.; Yao, J. A practical guide to photoacoustic tomography in the life sciences. Nat. Methods 2016, 13, 627–638. [Google Scholar] [CrossRef] [PubMed]
- Xu, M.; Wang, L.V. Photoacoustic imaging in biomedicine. Rev. Sci. Instrum. 2006, 77, 041101. [Google Scholar] [CrossRef]
- Beard, P. Biomedical photoacoustic imaging. Interface Focus 2011, 1, 602–631. [Google Scholar] [CrossRef] [PubMed]
- Yao, J.; Wang, L.V. Sensitivity of photoacoustic microscopy. Photoacoustics 2014, 2, 87–101. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Xie, X.; Ku, G.; Wang, L.V.; Stoica, G. Noninvasive imaging of hemoglobin concentration and oxygenation in the rat brain using high-resolution photoacoustic tomography. J. Biomed. Opt. 2006, 11, 024015. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.F.; Maslov, K.; Sivaramakrishnan, M.; Wang, L.V. Imaging of hemoglobin oxygen saturation variations in single vessels in vivo using photoacoustic microscopy. Appl. Phys. Lett. 2007, 90, 053901. [Google Scholar] [CrossRef]
- Li, W.; Lin, L.; Li, G. Wavelength selection method based on test analysis of variance: Application to oximetry. Anal. Methods 2014, 6, 1082–1089. [Google Scholar] [CrossRef]
- Lopez Silva, S.M.; Dotor Castilla, M.L.; Silveira Martin, J.P. Near-infrared transmittance pulse oximetry with laser diodes. J. Biomed. Opt. 2003, 8, 525–533. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.F.; Masloc, K.; Stoica, G.; Wang, L.V. Imaging acute thermal burns by photoacoustic microscopy. J. Biomed. Opt. 2006, 11, 054033. [Google Scholar] [CrossRef] [PubMed]
- Yao, J.; Maslov, K.I.; Zhang, Y.; Xia, Y.; Wang, L.V. Label-free oxygen-metabolic photoacoustic microscopy in vivo. J. Biomed. Opt. 2011, 16, 076003. [Google Scholar] [CrossRef] [PubMed]
- Ning, B.; Kennedy, M.J.; Dixon, A.J.; Sun, N.; Cao, R.; Soetikno, B.T.; Chen, R.; Zhou, Q.; Kirk Shuang, K.; Hossack, J.A.; et al. Simultaneous photoacoustic microscopy of microvascular anatomy, oxygen saturation, and blood flow. Opt. Lett. 2015, 40, 910–913. [Google Scholar] [CrossRef] [PubMed]
- Yao, J.; Wang, L.; Yang, J.M.; Maslov, K.I.; Wong, T.T.; Li, L.; Huang, C.H.; Zou, J.; Wang, L.V. High-speed label-free functional photoacoustic microscopy of mouse brain in action. Nat. Methods 2015, 12, 407–410. [Google Scholar] [CrossRef] [PubMed]
- Bayer, C.L.; Wlodarczyk, B.J.; Finnell, R.H.; Emelianov, S.Y. Ultrasound-guided spectral photoacoustic imaging of hemoglobin oxygenation during development. Biomed. Opt. Express 2017, 8, 757–763. [Google Scholar] [CrossRef] [PubMed]
- Rich, L.J.; Seshadri, M. Photoacoustic monitoring of tumor and normal tissue response to radiation. Sci. Rep. 2016, 6, 21237. [Google Scholar] [CrossRef] [PubMed]
- Dasu, A.; Toma-Dasu, I. Vascular oxygen content and the tissue oxygenation—A theoretical analysis. Med. Phys. 2008, 35, 539–545. [Google Scholar] [CrossRef] [PubMed]
- Hockel, M.; Knoop, C.; Schlenger, K.; Vorndran, B.; Baulmann, E.; Mitze, M.; Knapstein, P.G.; Vaupel, P. Intratumoral pO2 predicts survival in advanced cancer of the uterine cervix. Radiother. Oncol. 1993, 26, 45–50. [Google Scholar] [CrossRef]
- Cramer, T.; Schmitt, C.A. Metabolism in Cancer; Springer International Publishing: Cham, Switzerland, 2016. [Google Scholar]
- Lu, F.M.; Yuan, Z. PET/SPECT molecular imaging in clinical neuroscience: Recent advances in the investigation of CNS diseases. Quant. Imaging Med. Surg. 2015, 5, 433–447. [Google Scholar] [PubMed]
- Krohn, K.A.; Link, J.M.; Mason, R.P. Molecular imaging of hypoxia. J. Nucl. Med. 2008, 49, 129S–148S. [Google Scholar] [CrossRef] [PubMed]
- Haris, M.; Yadav, S.K.; Rizwan, A.; Singh, A.; Wang, E.; Hariharan, H.; Reddy, R.; Marincola, F.M. Molecular magnetic resonance imaging in cancer. J. Trans. Med. 2015, 13, 313–328. [Google Scholar] [CrossRef] [PubMed]
- Lövblad, K.O.; Anzalone, N.; Dörfler, A.; Essig, M.; Hurwitz, B.; Kappos, L.; Lee, S.K.; Filippi, M. MR Imaging in Multiple Sclerosis: Review and Recommendations for Current Practice. Am. J. Neuroradiol. 2010, 31, 983–989. [Google Scholar] [CrossRef] [PubMed]
- Tatum, J.L.; Kelloff, G.J.; Gillies, R.J. Hypoxia: Importance in tumor biology, noninvasive measurement by imaging, and value of its measurement in the management of cancer therapy. Int. J. Radiat. Biol. 2006, 82, 699–757. [Google Scholar] [CrossRef] [PubMed]
- Ntziachristos, V. Going deeper than microscopy: The optical imaging frontier in biology. Nat. Methods 2010, 7, 603–614. [Google Scholar] [CrossRef] [PubMed]
- Kautsky, H.; Hirsch, A. Original observation of effectdyes absorbed on silica gel. Z. Anorg. Allg. Chem. 1935, 222, 126–134. [Google Scholar]
- Vanderkooi, J.M.; Maniara, G.; Green, T.J.; Wilson, D.F. An Optical Method for Measurement of Dioxygen Concentration Based upon Quenching of Phosphorescence. J. Biol. Chem. 1987, 262, 5476–5482. [Google Scholar] [PubMed]
- Holst, G.; Grunwald, B. Luminescence lifetime imaging with transparent oxygen optodes. Sens. Actuators B Chem. 2001, 74, 78–90. [Google Scholar] [CrossRef]
- Becker, W. Advanced Time Correlated Single Photon Counting Applications; Springer International Publishing: Cham, Switzerland, 2015. [Google Scholar]
- Piffaretti, F.; Novello, A.M.; Kumar, R.S.; Forte, E.; Paulou, C.; Nowak-Sliwinska, P.; van den Bergh, H.; Wagnières, G. Real-time, in vivo measurement of tissular pO2 through the delayed fluorescence of endogenous protoporphyrin IX during photodynamic therapy. J. Biomed. Opt. 2012, 17, 115007. [Google Scholar] [CrossRef] [PubMed]
- Suhling, K.; Hirvonen, L.M.; Levitt, J.A.; Chung, P.H.; Tregidgo, C.; Le Marois, A.; Rusakov, D.A.; Zheng, K.; Ameer-Beg, S.; Poland, S.; et al. Fluorescence lifetime imaging (FLIM): Basic concepts and some recent developments. Med. Photonics 2015, 27, 3–40. [Google Scholar] [CrossRef]
- Shao, Q. Photoacoustic Lifetime Imaging and Its Biomedical Applications. Ph.D Thesis, University of Minnesota, Minneapolis, MN, USA, January 2016; p. 8. [Google Scholar]
- Ashkenazi, S.; Huang, S.W.; Horvath, T.; Koo, Y.E.; Kopelman, R. Photoacoustic probing of fluorophore excited state lifetime with application to oxygen sensing. J. Biomed. Opt. 2008, 13, 034023. [Google Scholar] [CrossRef] [PubMed]
- Ashkenazi, S. Photoacoustic lifetime imaging of dissolved oxygen using methylene blue. J. Biomed. Opt. 2010, 15, 040501. [Google Scholar] [CrossRef] [PubMed]
- Shao, Q.; Morgounova, E.; Jiang, C.; Choi, J.; Bischof, J.; Ashkenazi, S. In vivo photoacoustic lifetime imaging of tumor hypoxia in small animals. J. Biomed. Opt. 2013, 18, 076019. [Google Scholar] [CrossRef] [PubMed]
- Shao, Q.; Ashkenazi, S. Photoacoustic lifetime imaging for direct in vivo tissue oxygen monitoring. J. Biomed. Opt. 2015, 20, 036004. [Google Scholar] [CrossRef] [PubMed]
- Shao, Q.; Biel, M.A.; Ashkenazi, S. Noninvasive tumor oxygen imaging by photoacoustic lifetime imaging integrated with photodynamic therapy. Proc. SPIE 2014, 8931, 89310H. [Google Scholar]
- Shao, Q.; Morgounova, E.; Ashkenazi, S. Tissue oxygen monitoring by photoacoustic lifetime imaging (PALI) and its application to image-guided photodynamic therapy (PDT). Proc. SPIE 2015, 9323, 932320. [Google Scholar]
- Ray, A.; Rajian, J.R.; Lee, Y.E.; Wang, X.; Kopelman, R. Lifetime-based photoacoustic oxygen sensing in vivo. J. Biomed. Opt. 2012, 17, 057004. [Google Scholar] [CrossRef] [PubMed]
- Raya, A.; Rajian, J.R.; Lee, Y.-E.K.; Wang, X.; Kopelman, R. In vivo oxygen sensing using lifetime based photoacoustic measurements. Proc. SPIE 2013, 8581, 85811Q. [Google Scholar]
- Jo, J.; Lee, C.H.; Kopeman, R.; Wang, X. Lifetime-resolved photoacoustic (LPA) spectroscopy for monitoring oxygen change and photodynamic therapy (PDT). Proc. SPIE 2016, 9708, 97081L. [Google Scholar]
- Lai, P.; Wang, L.; Tay, J.W.; Wang, L.V. Photoacoustically guided wavefront shaping for enhanced optical focusing in scattering media. Nat. Photonics 2015, 9, 126–132. [Google Scholar] [CrossRef] [PubMed]


© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cao, F.; Qiu, Z.; Li, H.; Lai, P. Photoacoustic Imaging in Oxygen Detection. Appl. Sci. 2017, 7, 1262. https://doi.org/10.3390/app7121262
Cao F, Qiu Z, Li H, Lai P. Photoacoustic Imaging in Oxygen Detection. Applied Sciences. 2017; 7(12):1262. https://doi.org/10.3390/app7121262
Chicago/Turabian StyleCao, Fei, Zhihai Qiu, Huanhao Li, and Puxiang Lai. 2017. "Photoacoustic Imaging in Oxygen Detection" Applied Sciences 7, no. 12: 1262. https://doi.org/10.3390/app7121262
APA StyleCao, F., Qiu, Z., Li, H., & Lai, P. (2017). Photoacoustic Imaging in Oxygen Detection. Applied Sciences, 7(12), 1262. https://doi.org/10.3390/app7121262





