Qualitative and Quantitative Changes of Carbonate Rocks Exposed to SC CO2 (Basque-Cantabrian Basin, Northern Spain)
Abstract
1. Introduction and Objectives
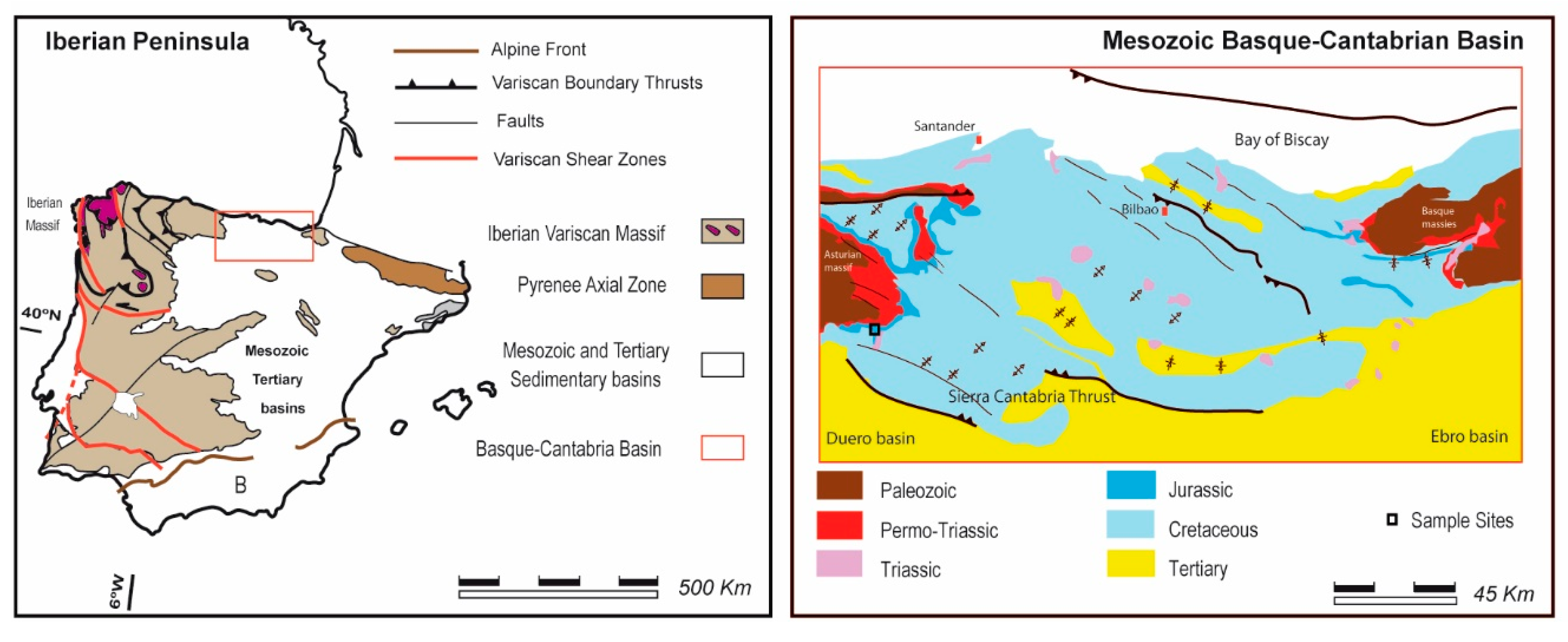
2. Geological Setting
3. Methodology
- Selection of a study area with real potential for CO2 geological storage (Figure 1).
- Identification of reservoir rocks in this area and their sampling for laboratory experimentation (Figure 1).
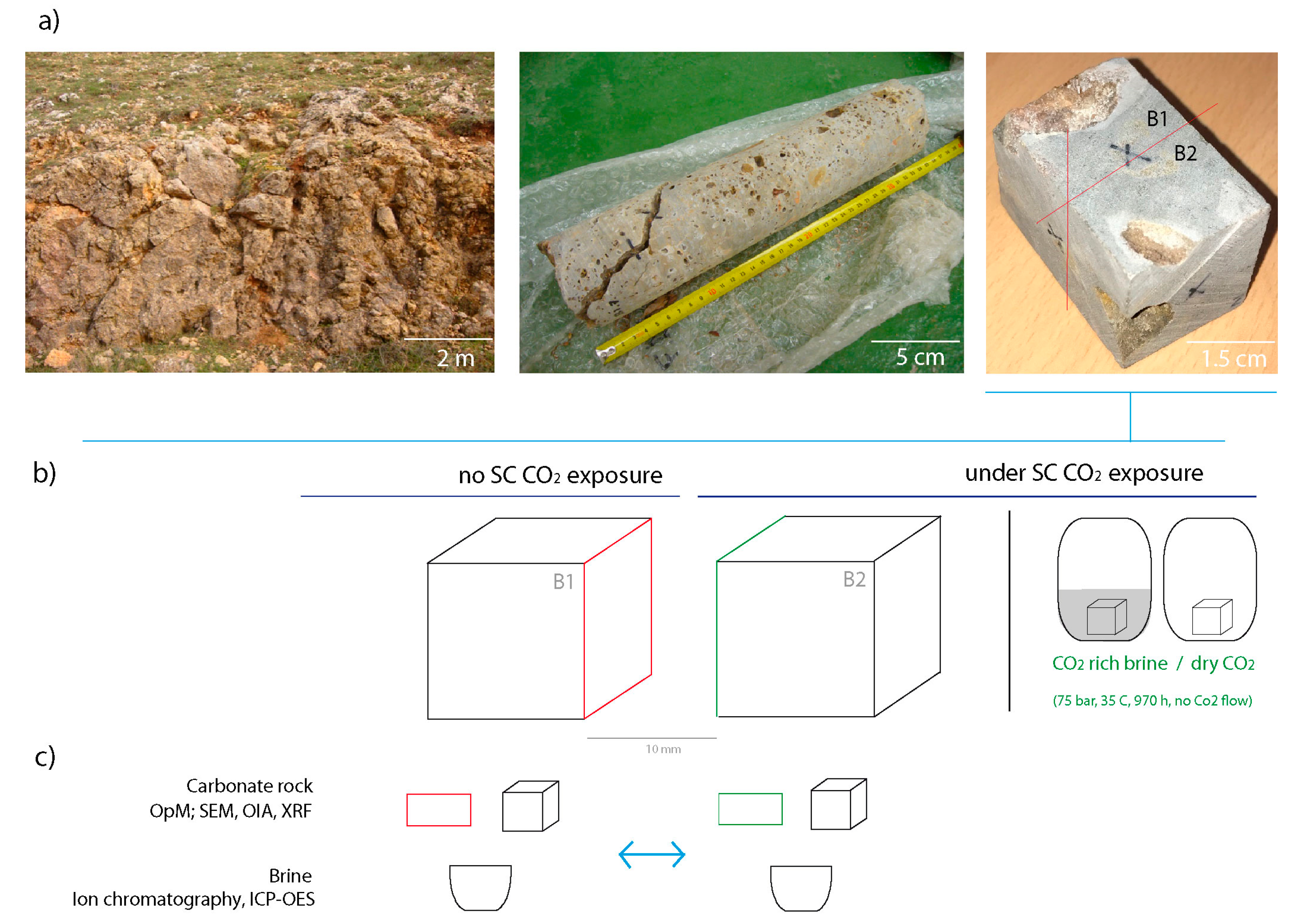
- Preparation of rock samples for laboratory experiments providing sample pairs in order to conduct comparative studies before and after SC CO2 exposure. The sample pairs are prepared to have one matching surface as close to each other in the original block as possible (Figure 2).
- Mineralogical and chemical characterization of rock samples and definition of brine composition before the experiment.
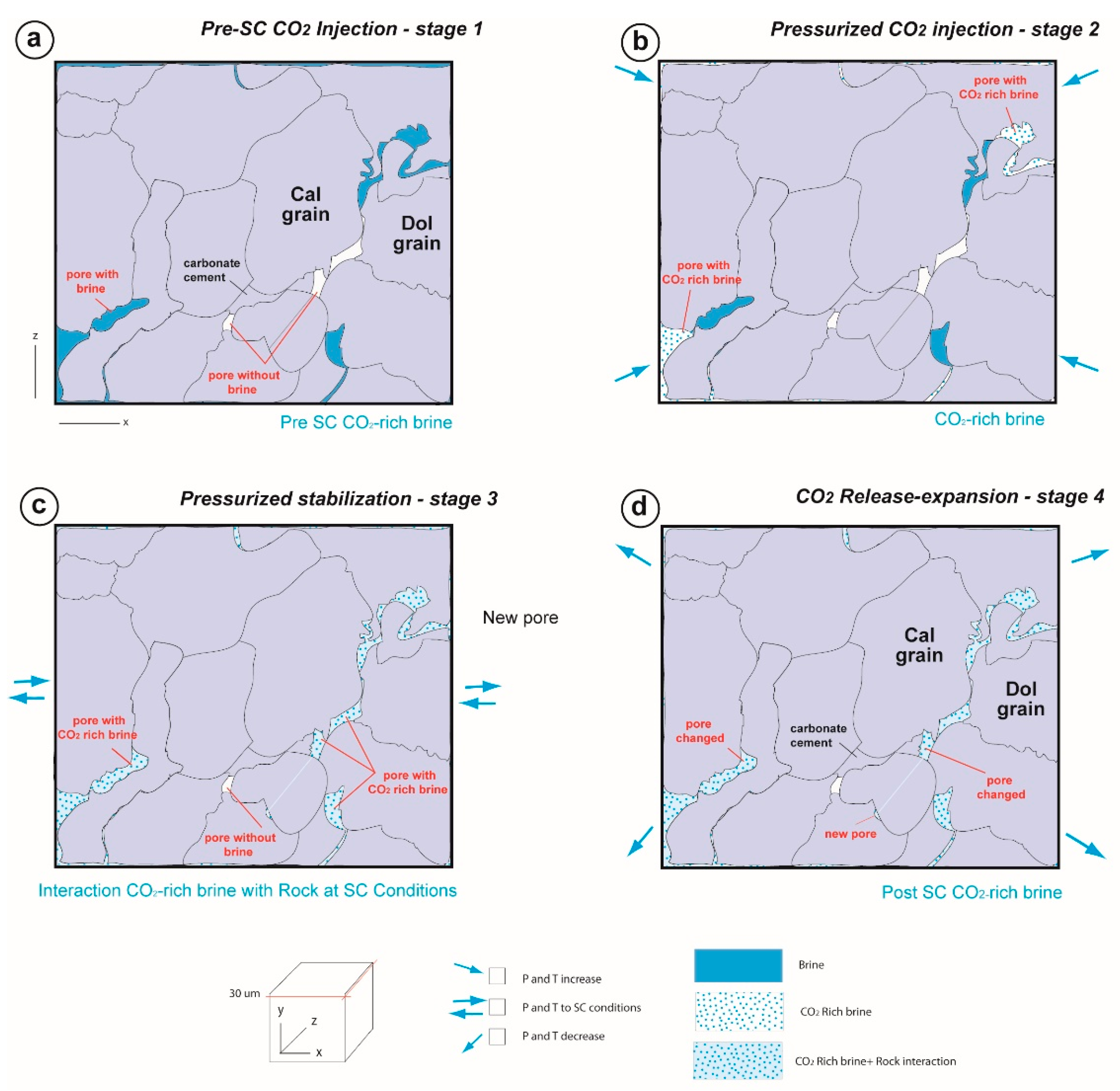
- Rock sample exposure to SC CO2 in a hyperbaric chamber simulating conditions of CO2 injection in a geological reservoir (P ≈ 75 bar, T ≈ 35 °C, 970 h exposure time and no CO2 flow) (Figure 2).
- Mineralogical and chemical characterization of rock samples and analysis of brine composition after the experiment.
- Comparison of analytical results obtained in the two stages of the experimentation (before and after SC CO2 exposure).Mass balance calculation and numerical modelling for validation of obtained results.
3.1. Experimental Setup (Sample Preparation and SC CO2 Injection)
3.2. Applied Techniques and Methods
4. Results
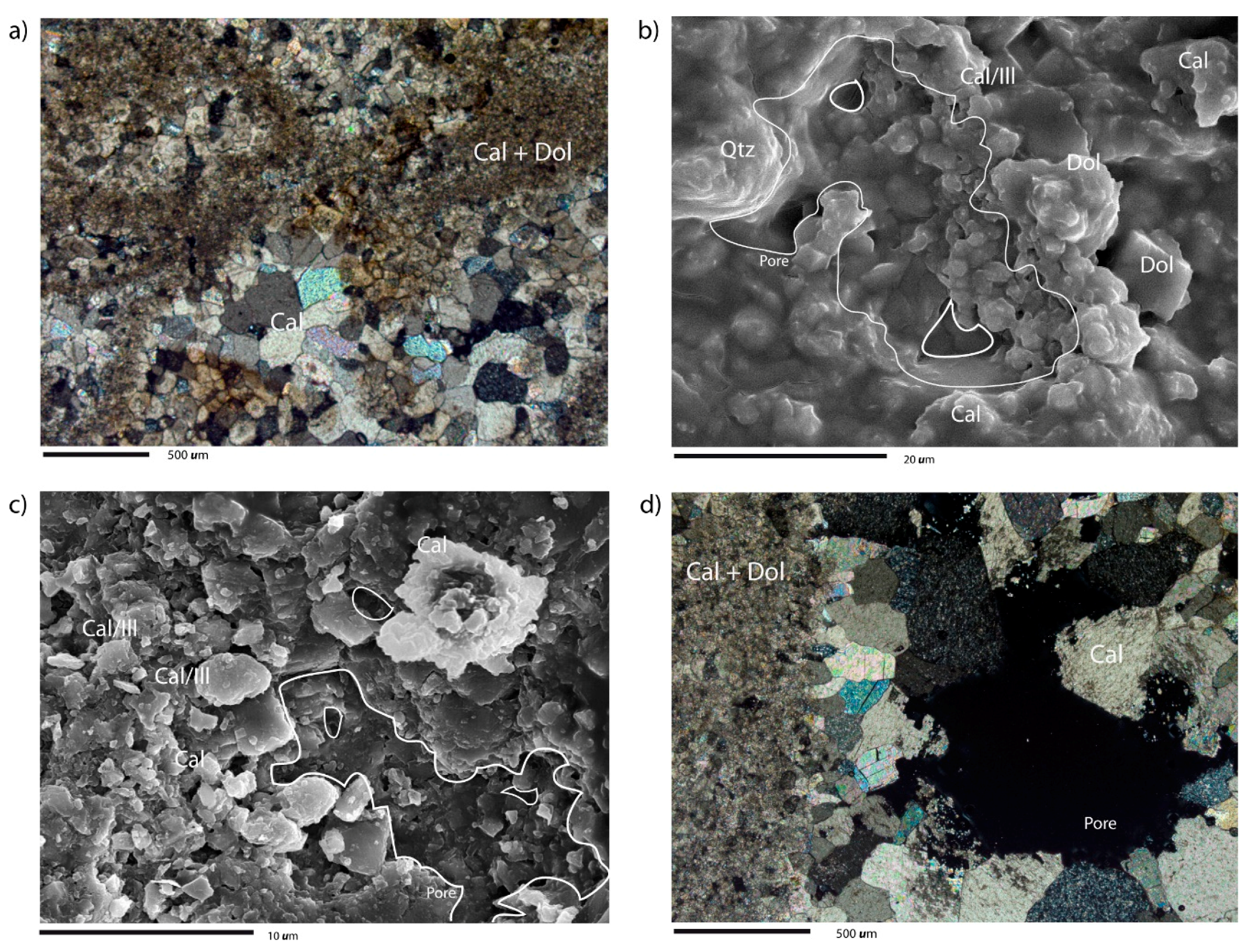
4.1. Petrographic and Chemical Characterization of Carbonate Rocks before SC CO2 Injection
4.2. Petrographic and Chemical Characterization of Carbonate Rocks after SC CO2 Injection
4.2.1. Dry SC CO2
4.2.2. SC CO2 Rich-Brine
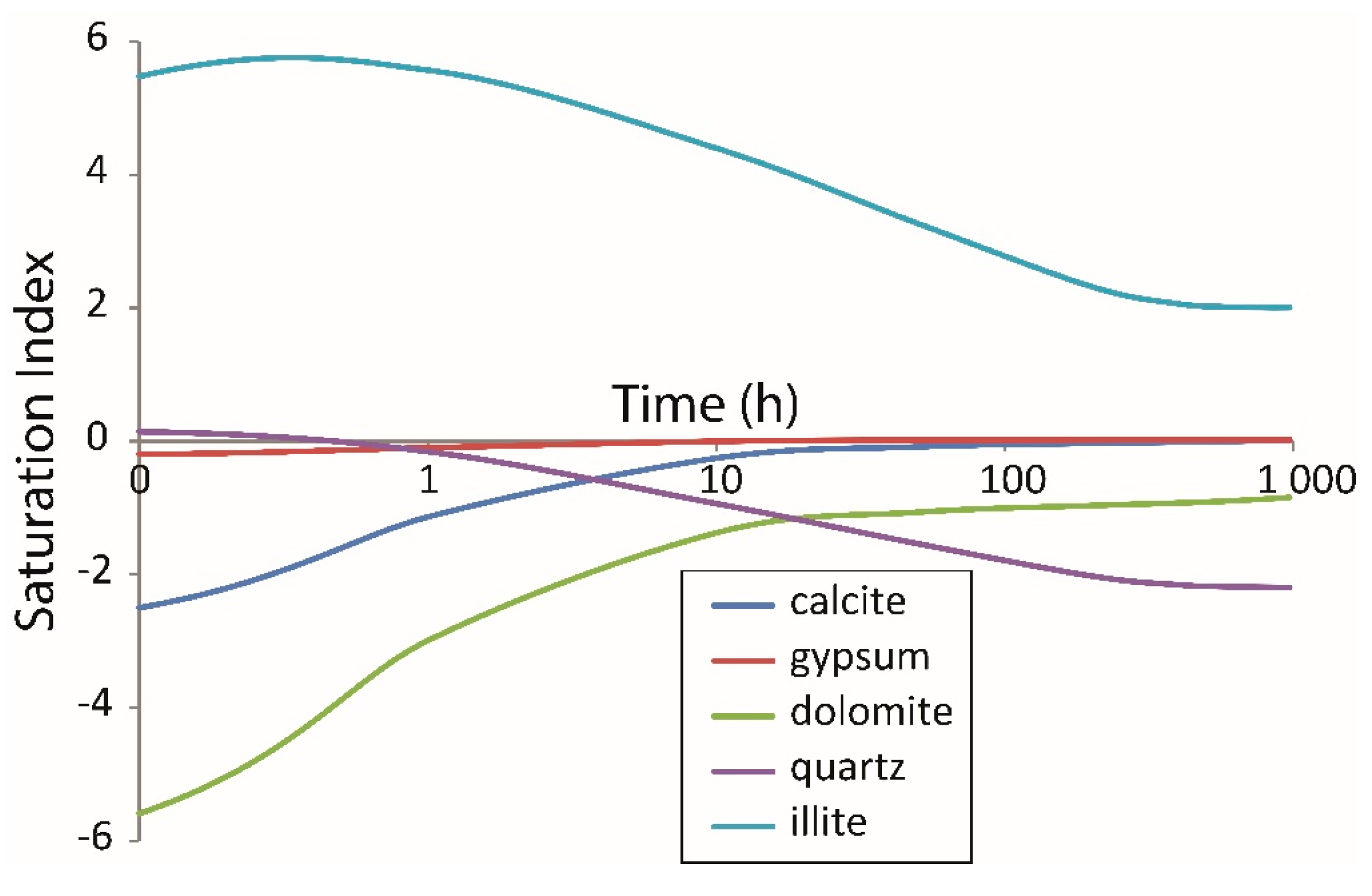
4.3. Mass Balance Calculation and Numerical Modelling
5. Discussion
6. Conclusions
- (i)
- The dissolution of CO2 in the brine, which created an acidified environment (HCO3− increase in brine: 1400%; pH decrease).
- (ii)
- Calcite dissolution in the low pH brine (Ca2+ increase in brine: 290%), producing dissolution phenomena observable by microscopic techniques (OpM and SEM).
- (iii)
- Increased porosity (absolute porosity variation: 0.32%) and modified pore morphology due to the calcite dissolution (Ro decrease: 2.40%; As increase: 2.26%; and increase of pore number: 12.26%).
- (iv)
- Dolomite crystals also present in the rock were found to be less reactive than the calcite.
- (v)
- Illite and gypsum precipitation was evaluated by both mass balance calculation from the water chemistry and numerical modelling.
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| SC | Supercritical Conditions |
| OpM | Optical Microscopy |
| SEM | Scanning Electron Microscopy |
| OIA | Optical Image Analysis |
| XRF | X-ray fluorescence |
| IGME | Geological Survey of Spain |
| UNIOVI | University of Oviedo |
| ICP-EOS | Inductively coupled plasma optical emission spectrometry |
| Cal | Calcite |
| Dol | Dolomite |
References
- Bachu, S. Sequestration of CO2 in geological media: Criteria and approach for site selection in response to climate change. Energy Convers. Manag. 2000, 41, 953–970. [Google Scholar] [CrossRef]
- Benson, S.M.; Cole, D.R. CO2 sequestration in deep sedimentary formations. Elements 2008, 4, 325–331. [Google Scholar] [CrossRef]
- Gaus, I. Role and impact of CO2-rock interactions during CO2 storage in sedimentary rocks. Int. J. Greenh. Gas Control 2010, 4, 73–89. [Google Scholar] [CrossRef]
- Izgec, O.; Demiral, B.; Bertin, H.J.; Akin, S. CO2 injection into saline carbonate aquifer formations I: Laboratory investigation. Transp. Porous Media 2008, 72, 1–24. [Google Scholar] [CrossRef]
- Bacci, G.; Korr, A.; Durucan, S. Experimental investigation into salt precipitation during CO2 injection in saline aquifers. Energy Procedia 2011, 4, 4450–4456. [Google Scholar] [CrossRef]
- Intergovernmental Panel on Climate Change (IPCC). Carbon Dioxide Capture and Storage. In IPCC 2005; Metz, B., Davidson, O., de Coninck, H.C., Loos, M., Meyer, L.A., Eds.; Cambridge University Press: Cambridge, UK; New York, NY, USA, 2005; p. 442. [Google Scholar]
- André, L.; Audigane, P.; Azaroual, M.; Menjoz, A. Numerical modelling of fluid-rock chemical interactions at the supercritical CO2-liquid interface during CO2 injection into a carbonate reservoir, the Dogger aquifer (Paris Basin, France). Energy Convers. Manag. 2007, 48, 1782–1797. [Google Scholar] [CrossRef]
- Ross, G.D.; Todd, A.C.; Tweedie, J.A.; Will, A.G.S. The Dissolution effects of CO2-brine systems on the permeability of UK and North Sea calcareous sandstones. Presented at the SPE/DOE Symposium on Enhanced Oil Recovery, Tulsa, OK, USA, 4–7 April 1982. [Google Scholar]
- Saeedi, A.; Rezaee, R.; Evans, B.; Clennell, B. Multiphase flow behaviour during CO2 geo-sequestration: Emphasis on the effect of cyclic CO2-brine flooding. J. Petrol. Sci. Eng. 2011, 79, 65–85. [Google Scholar] [CrossRef]
- Sayegh, S.G.; Krause, F.F.; Girard, M.; DeBree, C. Rock/fluid interactions of carbonated brines in a sandstone reservoir: Pembina Cardium, Alberta, Canada. SPE Form. Eval. 1990, 5, 399–405. [Google Scholar] [CrossRef]
- Garcia-Rios, M.; Luquot, L.; Soler, J.M.; Cama, J. Influence of the flow rate on dissolution and precipitation features during percolation of CO2 rich sulphate solutions through fractured limestone samples. Chem. Geol. 2015, 414, 95–108. [Google Scholar] [CrossRef]
- Egermann, P.; Bazin, B.; Vizika, O. An experimental investigation of reaction-transport phenomena during CO2 injection. In Proceedings of the SPE Middle East Oil and Gas Show and Conference, Manama, Bahrain, 12–15 March 2005. [Google Scholar]
- Gunter, W.D.; Bachu, S.; Benson, S. The Role of Hydrogeological and Geochemical Trapping in Sedimentary Basins for Secure Geological Storage of Carbon Dioxide. In Geological Storage of Carbon Dioxide; Special Publication; Baines, S.J., Worden, R.H., Eds.; Geological Society: London, UK, 2004; Volume 233, pp. 129–145. [Google Scholar]
- Izgec, Ö.; Demiral, B.; Bertin, H.; Akın, S. Experimental and numerical investigation of carbon sequestration in deep saline aquifers. In Proceedings of the SPE/EPA/DOE Exploration and Production Environmental Conference, Galveston, TX, USA, 7–9 March 2005. [Google Scholar]
- Luquot, L.; Gouze, P. Experimental determination of porosity and permeability changes induced by injection of CO2 into carbonate Rocks. Chem. Geol. 2009, 265, 148–159. [Google Scholar] [CrossRef]
- Cailly, B.; Le Thiez, P.; Egermann, P.; Audibert, A.; Vidal-Gilbert, S.; Longaygue, X. Geological storage of CO2: A state of the art of injection processes and technologies. Oil Gas Sci. Technol. 2005, 60, 517–525. [Google Scholar] [CrossRef]
- Kaszuba, J.P.; Janecky, D.R.; Snow, M.G. Carbon dioxide reaction processes in a model brine aquifer at 200 °C and 200 bars: Implications for geologic sequestration of carbon. Appl. Geochem. 2003, 18, 1065–1080. [Google Scholar] [CrossRef]
- Kaszuba, J.P.; Janecky, D.R.; Snow, M.G. Experimental evaluation of mixed fluid reactions between supercritical carbon dioxide and NaCl brine: Relevance to the integrity of a geologic carbon repository. Chem. Geol. 2005, 217, 277–293. [Google Scholar] [CrossRef]
- Mito, S.; Xue, Z.; Ohsumi Mito, S.; Xue, Z.; Ohsumi, T. Case study of geochemical reactions at the Nagaoka CO2 injection site, Japan. Int. J. Greenh. Gas Control 2008, 2, 309–318. [Google Scholar] [CrossRef]
- Sterpenich, J.; Sausse, J.; Pironon, J.; Géhin, A.; Hubert, G.; Perfetti, E.; Grgic, D. Experimental ageing of oolitic limestones under CO2 storage conditions. Petrographical and chemical evidence. Chem. Geol. 2009, 265, 99–112. [Google Scholar] [CrossRef]
- Miri, R.; Hellevang, H. Salt precipitation during CO2 storage—A review. Int. J. Greenh. Gas Control 2016, 51, 136–147. [Google Scholar] [CrossRef]
- Burton, M.; Kumar, K.; Bryant, S.L. Time-dependent injectivity during CO2 storage in aquifers. In Proceedings of the SPE Symposium on Improved Oil Recovery, Tulsa, OK, USA, 20–23 April 2008. [Google Scholar]
- Vickerd, M.A.; Thring, R.W.; Arocena, J.M.; Li, J.B. Changes in porosity due to acid gas injection as determined by X-ray computed tomography. J. Can. Pet. Technol. 2006, 45, 17–22. [Google Scholar] [CrossRef]
- Gaus, I.; Audigane, P.; Andre, L.; Lions, J.; Jacquement, N.; Durst, P.; Czernichowski, I.; Azaroual, M. Geochemical and solute transport modelling for CO2 storage, what to expect from it? Int. J. Greenh. Gas Control 2008, 2, 605–625. [Google Scholar] [CrossRef]
- Ketzer, J.M.; Iglesias, R.; Einloft, S.; Dullius, J.; Ligabue, R.; De Lima, V. Water-rock-CO2 interactions in saline aquifers aimed for carbon dioxide storage: Experimental and numerical modelling studies of the Rio Bonito Formation (Permian), Southern Brazil. Appl. Geochem. 2009, 24, 760–767. [Google Scholar] [CrossRef]
- Berrezueta, E.; González-Menéndez, L.; Breitner, D.; Luquot, L. Pore system changes during experimental CO2 injection into detritic rocks: Studies of potential storage rocks from some sedimentary basins of Spain. Int. J. Greenh. Gas Control 2013, 17, 411–422. [Google Scholar] [CrossRef]
- Fischer, S.; Liebscher, A.; De Lucia, M.; Hecht, L. Reactivity of sandstone and siltstone samples from the Ketzin pilot CO2 storage site-Laboratory experiments and reactive geochemical modelling. Environ. Earth Sci. 2013, 70, 3687–3708. [Google Scholar] [CrossRef]
- Tarkowski, R.; Wdowin, M. Petrophysical and mineralogical research on the influence of CO2 injection on Mesozoic reservoir and caprocks from the polish lowlands. Oil Gas Sci. Technol. 2011, 66, 137–150. [Google Scholar] [CrossRef]
- García-Lobón, J.L.; Reguera-García, M.I.; Martín-León, J.; Rey-Moral, C.; Berrezueta, E. Plan de Selección y Caracterización de Áreas y Estructuras Favorables Para el Almacenamiento Geológico de CO2 en España. Resumen Ejecutivo; Instituto Geológico y Minero de España (IGME): Madrid, Spain, 2010. [Google Scholar]
- Quesada, S.; Robles, S.; Pujalte, V. El «Jurásico Marino» del margen suroccidental de la Cuenca Vascocantábrica y su relación con la exploración de hidrocarburos. Geogaceta 1993, 13, 92–96. [Google Scholar]
- Álvarez, C. Hydrocarbons in Spain—Exploration and production. First Break 1994, 12, 43–46. [Google Scholar] [CrossRef]
- Merten, R. Petroleum exploration and production in Spain. Z. Dtsch. Ges. Geowiss. 2006, 157, 717–732. [Google Scholar] [CrossRef]
- Ambrose, T.; Carballeira, J.; López-Rico, J.; Wagner, R.H. Mapa Geológico de España E, 1:50.000, Hoja Núm. 107 (Baruelo de Santullan), Segunda Serie, Primera Edición; Instituto Geológico y Minero de España (IGME): Madrid, Spain, 1978. [Google Scholar]
- Aurell, M.; Meléndez, G.; Oloriz, F.; Bádenas, B.; Caracuel, J.E.; García-Ramos, J.C.; Gay, A.; Linares, A.; Quesada, S.; Robles, S.; et al. Jurassic. In The Geology of Spain, 1st ed.; Gibbons, W., Moreno, T., Eds.; Geological Society: London, UK, 2002; pp. 213–253. [Google Scholar]
- Quesada, S.; Robles, S.; Pujalte, V. Depositional architecture and transgressive regressive cycles within Liassic backstepping carbonate ramps in the Basque-Cantabrian basin, Northern Spain. J. Geol. Soc. Lond. 2005, 162, 531–548. [Google Scholar] [CrossRef]
- Robles, S.; Aramburu, A.; Apraiz, A. The Basque-Cantabrian basin: Birth and tectonosedimentary evolution. Enseñanza de las Ciencias de la Tierra 2014, 22.2, 99–114. [Google Scholar]
- Alcalde, L.; Martí, D.; Juhlin, C.; Malehmir, A.; Sopher Saura, E.; Marzán, I.; Ayarza, P.; Calahorrano, A.; Perez-Estaún, A.; Carbonell, R. 3-D reflection seismic imaging of the Hontomín structure in the Basque–Cantabrian Basin (Spain). Solid Earth 2013, 4, 481–496. [Google Scholar] [CrossRef]
- Ogaya, X.; Ledo, J.; Queralt, P.; Marcuello, A.; Quintà, A. First geoelectrical image of the subsurface of the Hontomín site (Spain) for CO2 geological storage: A magnetotelluric 2D characterization. Int. J. Greenh. Gas Control 2013, 13, 168–179. [Google Scholar] [CrossRef]
- Steefel, C.I.; Appelo, C.A.J.; Arora, B.; Jacques, D.; Kalbacher, T.; Kolditz, O.; Lagneau, V.; Lichner, P.C.; Mayer, K.U.; Meeussen, J.C.L.; et al. Reactive transport codes for subsurface environmental simulation. Comput. Geosci. 2015, 19, 445–478. [Google Scholar] [CrossRef]
- Davila, G.; Cama, J.; Luquoy, L.; Soler, J.; Ayora, C. Experimental and modelling study of the interaction between a crushed marl caprock and CO2-rich solutions under different pressure and temperature conditions. Chem. Geol. 2016. [Google Scholar] [CrossRef]
- Deng, H.; Molins, S.; Steefel, C.; Depaolo, D.; Voltolini, M.; Yang, L.; Ajo-Franklin, J. A 2.5D reactive transport model for fracture alteration simulation. Environ. Sci. Technol. 2016, 50, 7564. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Rios, M.; Luquot, L.; Soler, J.M.; Cama, J. The role of mineral heterogeneity on the hydrogeochemical response of two fractured reservoir rocks in contact with dissolved CO2. Appl. Geochem. 2017, 84, 202–217. [Google Scholar] [CrossRef]
- Luquot, L.; Gouze, P.; Niemi, A.; Bensabat, J.; Carrera, J. CO2-rich brine percolation experiments through Heletz reservoir rock samples (Israel): Role of the flow rate and brine composition. Int. J. Greenh. Gas Control 2016, 48, 44–58. [Google Scholar] [CrossRef]
- Bibi, I.; Singh, B.; Silvester, E. Dissolution of illite in saline acidic solutions at 25 °C. Geochim. Cosmochim. Acta 2011, 75, 3237–3249. [Google Scholar] [CrossRef]
- Domènech, C.; Ayora, C.; De Pablo, J. Oxidative dissolution of pyritic sludge from the Aznalcóllar mine (SW Spain). Chem. Geol. 2002, 190, 339–353. [Google Scholar] [CrossRef]
- Nourtier-Mazauric, E.; Guy, B.; Fritz, B.; Brosse, E.; Garcia, D.; Clément, A. Modelling the Dissolution/Precipitation of Ideal Solid Solutions. Oil Gas Sci. Technol. Rev. IFP 2005, 60, 401–415. [Google Scholar] [CrossRef]
- Palandri, J.L.; Kharaha, Y.K. A Compilation of Rate Parameters of Water-Mineral Interaction Kinetics for Application to Geochemical Modelling; Open File Report 2004-1068; U.S. Geological Survey: Denver, CO, USA, 2004.
- Wolery, T.J.; Jackson, K.J.; Bourcier, W.L.; Bruton, C.J.; Viani, B.E.; Knauss, K.G.; Delany, J.M. Current status of the EQ3/6 software package for geochemical modelling. ACS Symp. Ser. 1990, 416, 104–116. [Google Scholar]
- Luquot, L.; Andreani, M.; Gouze, P.; Camps, P. CO2 percolation experiment through chlorite/zeolite-rich sandstone (Pretty Hill Formation—Otway Basin—Australia). Chem. Geol. 2012, 294–295, 75–88. [Google Scholar] [CrossRef]
- Tutolo, B.; Luhmann, A.; Kong, X.Z.; Saar, M.; Seyfried, W.E., Jr.; William, E. CO2 sequestration in feldspar-rich sandstone: Coupled evolution of fluid chemistry, mineral reaction rates, and hydrogeochemical properties. Geochim. Cosmochim. Acta 2015, 160, 132–154. [Google Scholar] [CrossRef]
- Carroll, S.; McNab, W.; Dai, Z.; Torres, S. Reactivity of Mt. Simon sandstone and the Eau Claire shale under CO2 storage conditions. Environ. Sci. Technol. 2013, 47, 252–261. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Soong, Y.; Dilmore, R.; Lopano, C. Numerical simulation of porosity and permeability evolution of Mount Simon sandstone under geological carbon sequestration conditions. Chem. Geol. 2015, 403, 1–12. [Google Scholar] [CrossRef]
- Gledhill, D.K.; Morse, J.W. Calcite dissolution kinetics in Na-Ca-Mg-Cl brines. Geochim. Cosmochim. Acta 2006, 70, 5802–5813. [Google Scholar] [CrossRef]
- Plummer, L.N.; Wigley, T.M.L.; Parkhurst, D.L. The kinetics of calcite dissolution in CO2-water systems at 5 degrees to 60 degrees C and 0.0 to 1.0 atm CO2. Am. J. Sci. 1978, 278, 179–216. [Google Scholar] [CrossRef]
- Pokrovsky, O.S.; Golubev, S.V.; Schott, J.; Castillo, A. Calcite, dolomite and magnesite dissolution kinetics in aqueous solutions at acid to circumneutral pH, 25 to 150 °C and 1 to 55 atm pCO2: New constraints on CO2 sequestration in sedimentary basins. Chem. Geol. 2009, 265, 20–32. [Google Scholar] [CrossRef]
- Guo, H.; Wang, Y.B.; Ni, X.M.; Tao, C.Q.; Han, W.L. Chemical kinetics of Ca/Mg elements from calcite/dolomite in carbonic acid solution. J. China Coal Soc. 2016, 41, 1806–1812. [Google Scholar]
- Gunter, W.; Wiwehar, B.; Perkins, E. Aquifer disposal of CO2-rich greenhouse gases: Extension of the time scale of experiment for CO2-sequestering reactions by geochemical modelling. Mineral. Petrol. 1997, 59, 121–140. [Google Scholar] [CrossRef]
- Hangs, S.; van der Linden, A.; Marcelis, F.; Bauer, A. The effect of CO2 on the mechanical properties of the Captain Sandstone: Geological storage of CO2 at the Goldeneye field (UK). Int. J. Greenh. Gas Control 2013, 19, 609–619. [Google Scholar] [CrossRef]
- Lyu, Q.; Gamage-Ranjith, P.; Long, X.; Ji, B. Experimental Investigation of Mechanical Properties of Black Shales after CO2-Water-Rock Interaction. Materials 2016, 9, 663. [Google Scholar] [CrossRef] [PubMed]
- Al-Shalabi, E.W.; Sepehmoori, K. A comprehensive review of low salinity/engineered water injections and their applications in sandstone and carbonate rocks. J. Pet. Sci. Eng. 2016, 139, 137–161. [Google Scholar] [CrossRef]
- Lebedev, M.; Zhang, Y.; Sarmadivaleh, M.; Barifcani, A.; Al-Khdheeawi, E.; Iglauer, S. Carbon geosequestration in limestone: Pore-scale dissolution and geomechanical weakening. Int. J. Greenh. Gas Control 2017, 66, 106–119. [Google Scholar] [CrossRef]
- Luquot, L.; Roetting, T.S.; Carrera, J. Characterization of flow parameters and evidence of pore clogging during limestone dissolution experiments. Water Resour. Res. 2014, 50, 6305–6321. [Google Scholar] [CrossRef]
- Qajar, J.; Francois, N.; Arns, C.H. Micro-Tomographic Characterization of Dissolution-Induced Local Porosity Changes Including Fines Migration in Carbonate Rock, Presented at the 2012 SPE EOR Conference at Oil and Gas West Asia(OGWA), Muscat, Oman, 16–18 April 2012; EOR: Building Towards Sustainable Growth; Society of Petroleum Engineers: London, UK, 2012; Volume 1, pp. 117–134. [Google Scholar]
- Wdowin, M.; Tarkowski, R.; Franus, W. Determination of changes in the reservoir and cap rocks of the Chabowo Anticline caused by CO2-brine-rock interactions. Int. J. Coal Geol. 2014, 130, 79–88. [Google Scholar] [CrossRef]
- Yu, Z.; Liu, L.; Yang, S.; Li, S.; Yang, Y. An experimental study of CO2–brine–rock interaction at in situ pressure—Temperature reservoir conditions. Chem. Geol. 2012, 326–327, 88–101. [Google Scholar] [CrossRef]

| Component | Rock before CO2 (%) (1) | Rock after Dry CO2 (%) (2) | Interpretation (1) to (2) | Rock after CO2 and Brine (%) (3) | Interpretation (1) to (3) |
|---|---|---|---|---|---|
| SiO2 | 2.080 ± 0.170 | 2.150 ± 0.176 | 2.510 ± 0.206 | ||
| Al2O3 | 0.620 ± 0.072 | 0.630 ± 0.073 | 0.720 ± 0.083 | ||
| Fe2O3 | 0.250 ± 0.024 | 0.240 ± 0.023 | 0.400 ± 0.38 | ||
| MnO | 0.200 ± 0.001 | 0.150 ± 0.014 | 0.050 ± 0.005 | ||
| MgO | 0.980 ± 0.078 | 0.860 ± 0.068 | 1.13 ± 0.900 | ||
| CaO | 52.810 ± 3.590 | 54.150 ± 3.682 | No relevant | 52.200 ± 3.545 | No relevant |
| Na2O | 0.070 ± 0.005 | 0.080 ± 0.007 | changes | 0.065 ± 0.005 | changes |
| K2O | 0.320 ± 0.064 | 0.410 ± 0.082 | 0.340 ± 0.068 | ||
| TiO2 | 0.040 ± 0.003 | 0.040 ± 0.003 | 0.100 ± 0.010 | ||
| P2O5 | 0.010 ± 0.001 | 0.010 ± 0.001 | 0.045 ± 0-005 | ||
| LOI | 42.580 ± 5.111 | 40.720 ± 4.886 | 41.5 ± 4.980 | ||
| Total | 99.810 | 99.830 | 99.60 |
| Component | Pure Brine (mg/L) | Brine Post Test (mg/L) | Interpretation |
|---|---|---|---|
| Na+ | 982 ± 98.2 | 1000 ± 100 | No change |
| K+ | 94 ± 9.4 | 92 ± 9.2 | No change |
| Mg2+ | 84 ± 8.4 | 106 ± 10.6 | Increase (26%). Dissolution of Dolomite |
| Ca2+ | 266 ± 26.6 | 1040 ± 104 | Increase (290%). Dissolution of Calcite |
| SO42− | 223 ± 22.3 | 240 ± 24 | No change |
| Cl− | 2180 ± 218 | 2180 ± 218 | No change |
| HCO−3 | 17 ± 1.7 | 2400 ± 240 | Increase (1400%) |
| CO3 | 0 | 0 | No change |
| NO−3 | 1 ± 0.1 | 7 ± 0.7 | Increase (600%) |
| SiO2 | 13 ± 1.3 | 17 ± 1.7 | Increase (31%) |
| pH | 7.4 (pH) | 6.4 (pH) | Decrease (13%) |
| Conductivity | 6450 (mS/cm) | 13650 (mS/cm) | Increase (112%) |
| Number of Pores | Δ Number of Pores | Porosity (%) | Δ Porosity | Ro | ΔRo | As | ΔAs | |
|---|---|---|---|---|---|---|---|---|
| Before | 11.17 × 104 | 12.26 | 4.12 ± 0.15 ** | 7.77 (0.32) * | 0.625 | −2.40 | 2.207 | 2.26 |
| After | 12.54 × 104 | 4.44 ± 0.17 ** | 0.610 | 2.257 |
| Mineral and Chemical Composition | Initial (mg) | Final (Mass Balance, mg) | Final (Numerical Model, mg) | Reactive Surface Area (m2/m3) | km,25 (mol m−2 s−1) | n H+ | n O2 | Ea (Kcal mol−1) | References |
|---|---|---|---|---|---|---|---|---|---|
| Calcite CaCO3 | 259.051 | 258.492 | 257.803 | 100 | 5.01 × 10−3 | 0.5 | 11 | [46] | |
| Dolomite CaMg(CO3)2 | 12.536 | 12.484 | 12.472 | 0.1 | 6.46 × 10−4 | 0.5 | 8.63 | [47] | |
| Quartz SiO2 | 3.719 | 3.717 | 3.706 | 10 | 3.98 × 10−14 | −0.3 | 21.72 | [47] | |
| Illite (K, H3O)Al2Si3AlO10(OH)2 | 3.387 | 3.389 | 3.390 | 10,000 | 6.31 × 10−14 | 13 | [44] | ||
| Pyrite FeS2 | 0.465 | 0.233 | 0.185 | 1000 | 6.46 × 10−9 | −0.11 | 0.5 | 13.61 | [45] |
| Gypsum CaSO4 2H2O | 0.002 | 0.265 | 0.033 | 0.1 | 1.62 × 10−3 | 0 | [47] |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Berrezueta, E.; Kovacs, T.; Luquot, L. Qualitative and Quantitative Changes of Carbonate Rocks Exposed to SC CO2 (Basque-Cantabrian Basin, Northern Spain). Appl. Sci. 2017, 7, 1124. https://doi.org/10.3390/app7111124
Berrezueta E, Kovacs T, Luquot L. Qualitative and Quantitative Changes of Carbonate Rocks Exposed to SC CO2 (Basque-Cantabrian Basin, Northern Spain). Applied Sciences. 2017; 7(11):1124. https://doi.org/10.3390/app7111124
Chicago/Turabian StyleBerrezueta, Edgar, Timea Kovacs, and Linda Luquot. 2017. "Qualitative and Quantitative Changes of Carbonate Rocks Exposed to SC CO2 (Basque-Cantabrian Basin, Northern Spain)" Applied Sciences 7, no. 11: 1124. https://doi.org/10.3390/app7111124
APA StyleBerrezueta, E., Kovacs, T., & Luquot, L. (2017). Qualitative and Quantitative Changes of Carbonate Rocks Exposed to SC CO2 (Basque-Cantabrian Basin, Northern Spain). Applied Sciences, 7(11), 1124. https://doi.org/10.3390/app7111124






