Kinetic Adsorption Study of Silver Nanoparticles on Natural Zeolite: Experimental and Theoretical Models
Abstract
:1. Introduction
2. Experimental Section
3. Results and Discussion
3.1. Chemical Characterization of Natural Zeolite


| Element | wt. % Zeolite | Oxides | wt. % Zeolite |
|---|---|---|---|
| Si | 81.7 | SiO2 | 77.04 |
| Al | 8.01 | Al2O3 | 13.2 |
| K | 6.04 | K2O | 6.42 |
| Fe | 1.145 | FeO | 0.66 |
| Ca | 1.2 | CaO | 0.75 |
| Mg | 0.725 | MgO | 0.53 |
| Na | 1.18 | Na2O | 1.4 |
3.2. Experimental Study of the Ag Adsorption by Atomic Absorption Spectrometry (AAS)
3.2.1. Calibration Curves for the Ag Ions and Ag Nanoparticles
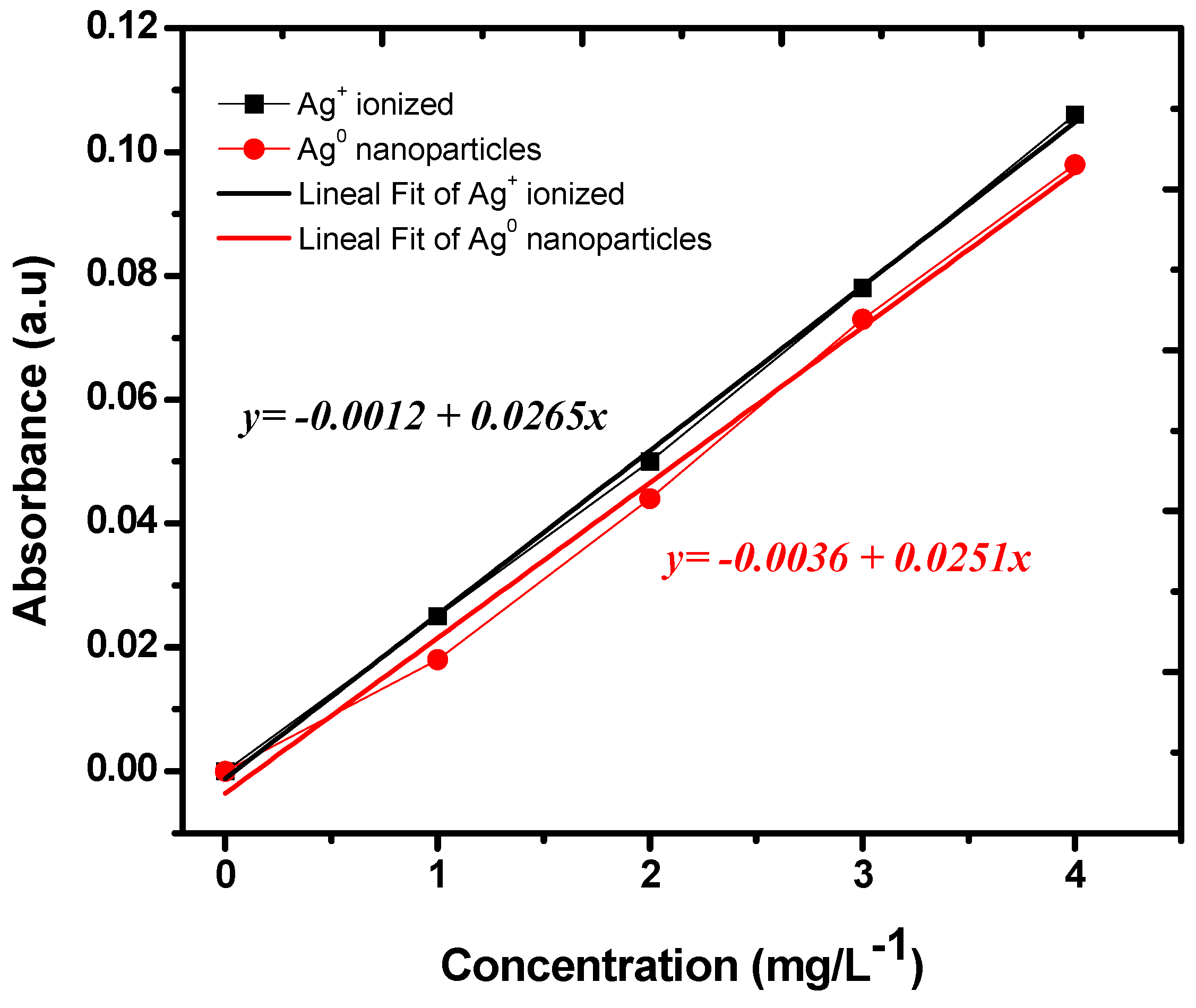
3.2.2. Adsorption Efficiency of Ag Ions and AgNPs on the Zeolite

3.3. Theoretical Models
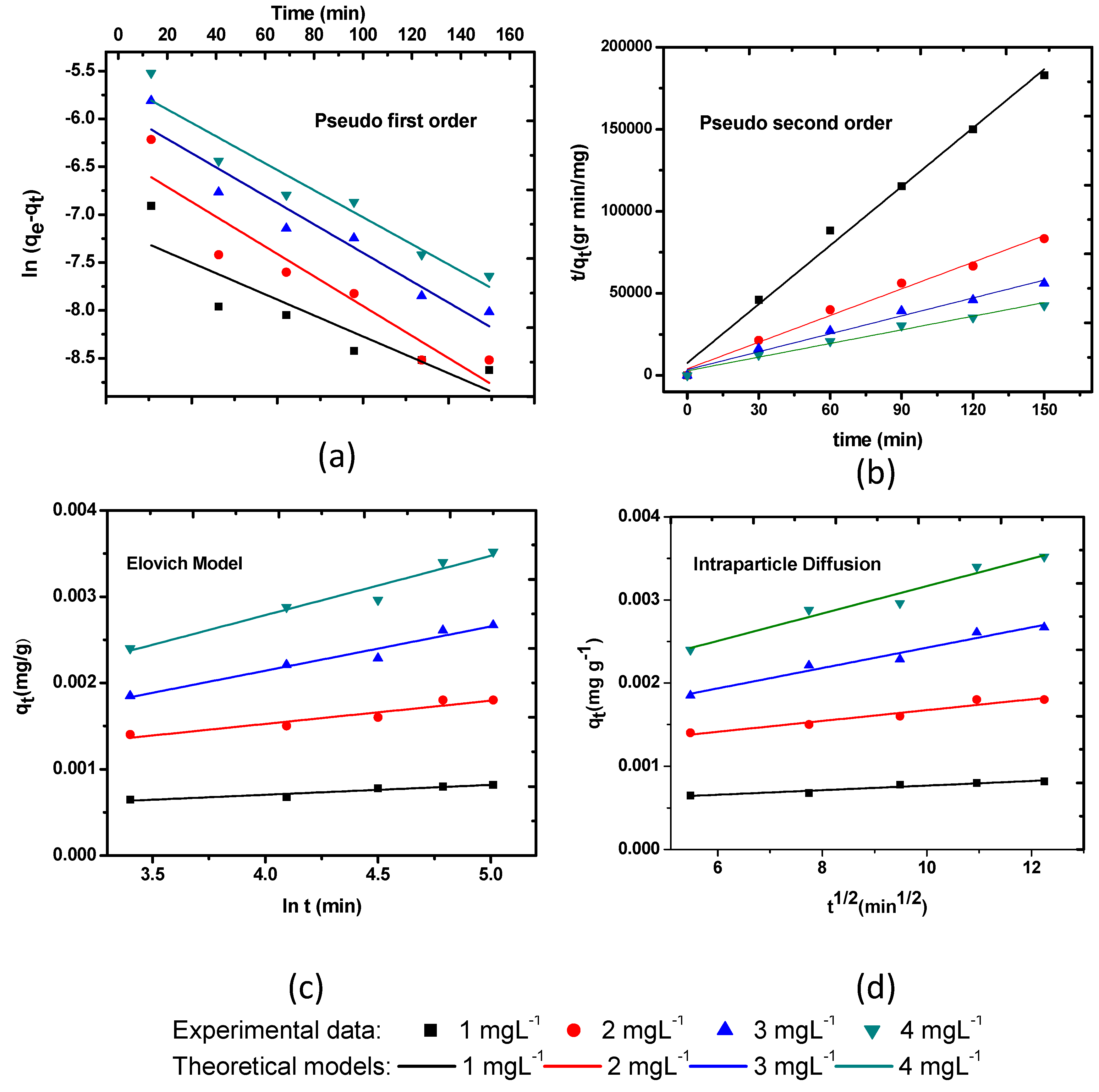
3.3.1. Lagergren Pseudo-First-Order Model
3.3.2. Pseudo-Second-Order Model
3.3.3. Elovich Model
3.3.4. Intraparticle Diffusion Model
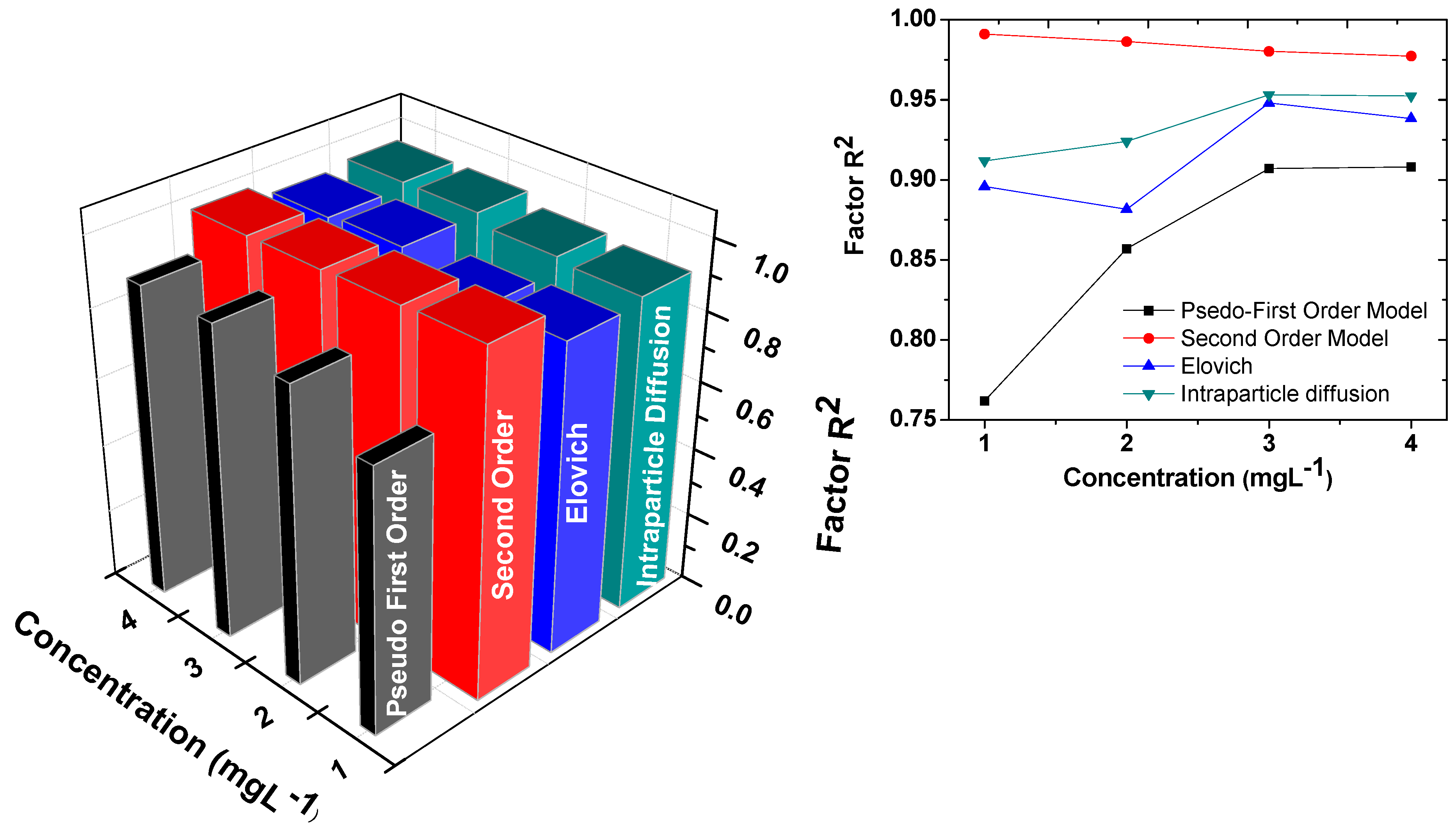
| Kinetic Model | Lagergren First-Order (K1) min−1 | Pseudo-Second-Order K2 (g·mg−1·min−1) qe (mg·g−1) | Elovich α (mg·g−1·min) β (g·mg−1) | Intraparticle Duffusion Ki (mg·g−1·min−0.5) | ||||
|---|---|---|---|---|---|---|---|---|
| 1 mg·L−1 | K1 | 0.023 | K2 | 1.88 × 102 | α | 73,690.88 | Ci | 4.93 × 10−4 |
| qe | 8.38 × 10−4 | β | 0.000115 | Ki | 2.76 × 10−5 | |||
| R2 | 0.762 | R2 | 0.99108 | R2 | 0.89589 | R2 | 0.91193 | |
| 2 mg·L−1 | K1 | 0.032 | K2 | 73.4 | α | 1.98 × 104 | Ci | 0.00102 |
| qe | 1.85 × 10−3 | β | 2.69 × 10−4 | Ki | 6.52 × 10−5 | |||
| R2 | 0.857 | R2 | 0.98635 | R2 | 0.88164 | R2 | 0.92399 | |
| 3 mg·L−1 | K1 | 0.031 | K2 | 37.8 | α | 2.33 × 103 | Ci | 0.0012 |
| qe | 2.75 × 10−3 | β | 5.13 × 10−4 | Ki | 1.22 × 10−4 | |||
| R2 | 0.907 | R2 | 0.98026 | R2 | 0.94803 | R2 | 0.95313 | |
| 4 mg·L−1 | K1 | 0.029 | K2 | 27.2 | α | 1.53 × 103 | Ci | 0.00152 |
| qe | 3.61 × 10−3 | β | 6.88 × 10−4 | Ki | 1.65 × 10−4 | |||
| R2 | 0.908 | R2 | 0.97719 | R2 | 0.93829 | R2 | 0.95237 | |

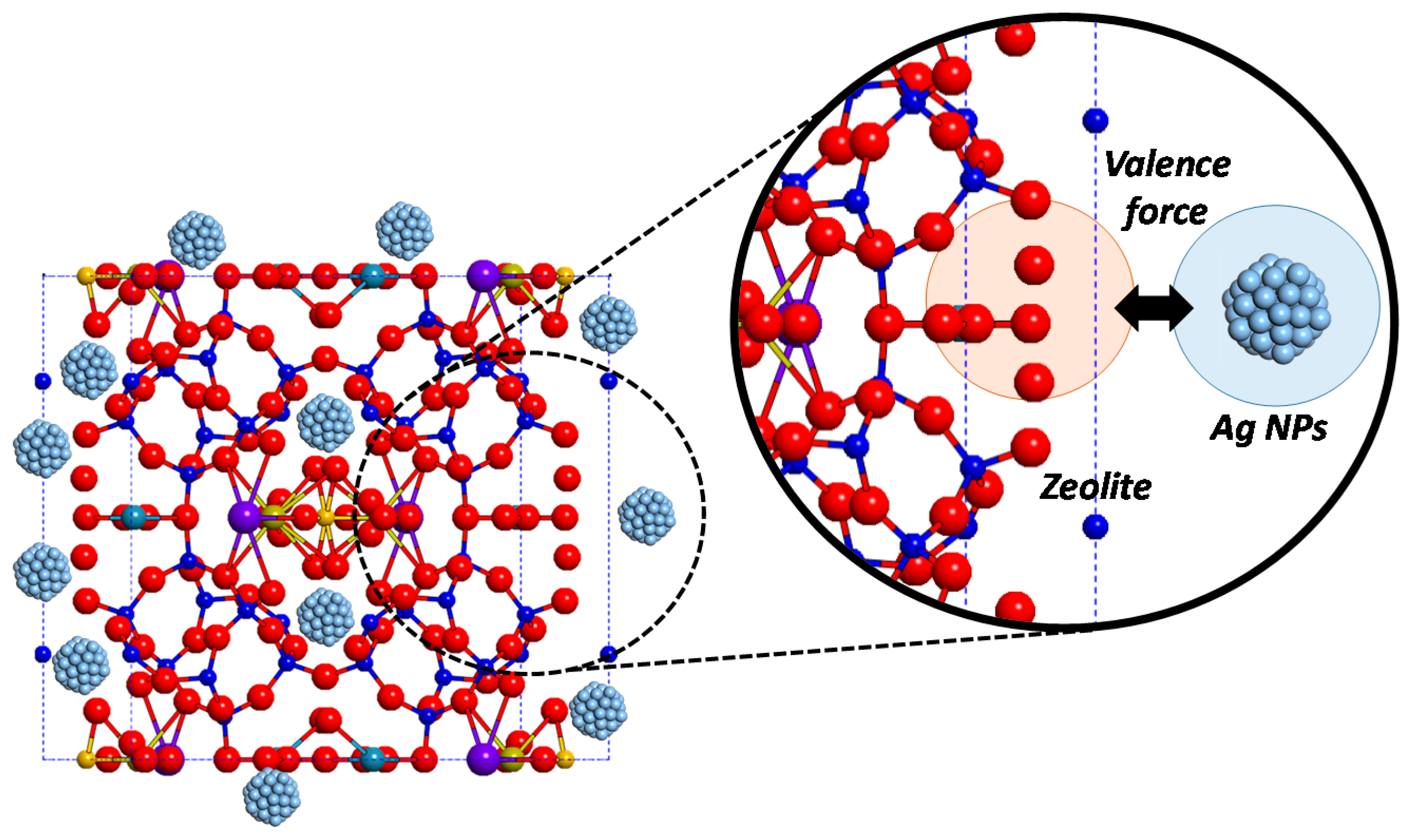
4. Conclusions
Author Contributions
Conflicts of Interest
References
- Khajeh-Talkhoncheh, S.; Haghighi, M. Syngas production via dry reforming of methane over Ni-based nanocatalyst over various supports of clinoptilolite, ceria and alumina. J. Nat. Gas Sci. Eng. 2015, 23, 16–25. [Google Scholar] [CrossRef]
- Romero, D.; Chlala, D.; Labaki, M.; Royer, S.; Bellat, J.-P.; Bezverkhyy, I.; Giraudon, J.-M.; Lamonier, J.-F. Removal of Toluene over NaX Zeolite Exchanged with Cu2+. Catalysts 2015, 5, 1479–1497. [Google Scholar] [CrossRef]
- Munthali, M.; Elsheikh, M.; Johan, E.; Matsue, N. Proton Adsorption Selectivity of Zeolites in Aqueous Media: Effect of Si/Al Ratio of Zeolites. Molecules 2014, 19, 20468–20481. [Google Scholar] [CrossRef] [PubMed]
- Ajoudanian, N.; Nezamzadeh-Ejhieh, A. Enhanced photocatalytic activity of nickel oxide supported on clinoptilolite nanoparticles for the photodegradation of aqueous cephalexin. Mater. Sci. Semicond. Process. 2015, 36, 162–169. [Google Scholar] [CrossRef]
- Donati, E.; Polcaro, C.M.; Ciccioli, P.; Galli, E. The comparative study of a laccase-natural clinoptilolite-based catalyst activity and free laccase activity on model compounds. J. Hazard. Mater. 2015, 289, 83–90. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Iznaga, I.; Petranovskii, V.; Castillón-Barraza, F.; Concepción-Rosabal, B. Copper-Silver Bimetallic System on Natural Clinoptilolite: Thermal Reduction of Cu2+ and Ag+ Exchanged. J. Nanosci. Nanotechnol. 2011, 11, 5580–5586. [Google Scholar] [CrossRef] [PubMed]
- Fateminia, F.S.; Falamaki, C. Zero valent nano-sized iron/clinoptilolite modified with zero valent copper for reductive nitrate removal. Process Saf. Environ. Prot. 2013, 91, 304–310. [Google Scholar] [CrossRef]
- Karel, F.B.; Koparal, A.S.; Kaynak, E. Development of Silver Ion Doped Antibacterial Clays and Investigation of Their Antibacterial Activity. Adv. Mater. Sci. Eng. 2015, 2015, 1–6. [Google Scholar] [CrossRef]
- Guaya, D.; Valderrama, C.; Farran, A.; Armijos, C.; Cortina, J.L. Simultaneous phosphate and ammonium removal from aqueous solution by a hydrated aluminum oxide modified natural zeolite. Chem. Eng. J. 2015, 271, 204–213. [Google Scholar] [CrossRef]
- Pourtaheri, A.; Nezamzadeh-Ejhieh, A. Enhancement in photocatalytic activity of NiO by supporting onto an Iranian clinoptilolite nano-particles of aqueous solution of cefuroxime pharmaceutical capsule. Spectrochim. Acta A Mol. Biomol. Spectrosc. 2015, 137, 338–344. [Google Scholar] [CrossRef] [PubMed]
- Zayed, M.A.; El-Begawy, S.E.M.; Hassan, H.E.S. Enhancement of stabilizing properties of double-base propellants using nano-scale inorganic compounds. J. Hazard. Mater. 2012, 227–228, 274–279. [Google Scholar] [CrossRef] [PubMed]
- Nezamzadeh-Ejhieh, A.; Shirvani, K. CdS Loaded an Iranian Clinoptilolite as a Heterogeneous Catalyst in Photodegradation of p-Aminophenol. J. Chem. 2013, 2013, 1–11. [Google Scholar] [CrossRef]
- Ortiz-Polo, A.; Richards-Uribe, R.M.; Otazo-Sánchez, E.M.; Prieto-García, F.; Hernández-Ávila, J.; Acevedo-Sandoval, O.; Gordillo-Martínez, A. New organo-inorganic materials for water contaminants remediation. In Proceedings of the Materials Research Society Symposium Proceedings, San Francisco, CA, USA, 9 April 2008; Volume 1007, pp. 129–136.
- Jiménez-Cedillo, M.J.; Olguín, M.T.; Fall, C.; Colín, A. Adsorption capacity of iron- or iron-manganese-modified zeolite-rich tuffs for As(III) and As(V) water pollutants. Appl. Clay Sci. 2011, 54, 206–216. [Google Scholar] [CrossRef]
- Top, A.; Ülkü, S. Silver, zinc, and copper exchange in a Na-clinoptilolite and resulting effect on antibacterial activity. Appl. Clay Sci. 2004, 27, 13–19. [Google Scholar] [CrossRef]
- Çoruh, S.; Şenel, G.; Ergun, O.N. A comparison of the properties of natural clinoptilolites and their ion-exchange capacities for silver removal. J. Hazard. Mater. 2010, 180, 486–492. [Google Scholar] [CrossRef] [PubMed]
- Lihareva, N.; Dimova, L.; Petrov, O.; Tzvetanova, Y. Ag+ sorption on natural and Na-exchanged clinoptilolite from Eastern Rhodopes, Bulgaria. Microporous Mesoporous Mater. 2010, 130, 32–37. [Google Scholar] [CrossRef]
- Praus, P.; Turicová, M.; Machovič, V.; Študentová, S.; Klementová, M. Characterization of silver nanoparticles deposited on montmorillonite. Appl. Clay Sci. 2010, 49, 341–345. [Google Scholar] [CrossRef]
- Fechete, I.; Vedrine, J. Nanoporous Materials as New Engineered Catalysts for the Synthesis of Green Fuels. Molecules 2015, 20, 5638–5666. [Google Scholar] [CrossRef] [PubMed]
- Pal, B.; Rana, S.; Kaur, R. Influence of Different Reducing Agents on the Ag Nanostructures and Their Electrokinetic and Catalytic Properties. J. Nanosci. Nanotechnol. 2015, 15, 2753–2760. [Google Scholar] [CrossRef] [PubMed]
- Chiericatti, C.; Basílico, J.C.; Basílico, M.L.Z.; Zamaro, J.M. Antifungal activity of silver ions exchanged in mordenite. Microporous Mesoporous Mater. 2014, 188, 118–125. [Google Scholar] [CrossRef]
- Taylor, P.; Lihareva, N.; Tzvetanova, Y.; Petrov, O.; Dimova, L. Sorption of Silver Cations by Natural and Na-Exchanged Mordenite Sorption of Silver Cations by Natural and Na-Exchanged Mordenite. Sep. Sci. Technol. 2015, 6395, 37–41. [Google Scholar]
- De la Rosa-Gómez, I.; Olguín, M.T.; Alcántara, D. Bactericides of coliform microorganisms from wastewater using silver-clinoptilolite rich tuffs. Appl. Clay Sci. 2008, 40, 45–53. [Google Scholar] [CrossRef]
- Celik, F.E.; Kim, T.-J.; Bell, A.T. Effect of zeolite framework type and Si/Al ratio on dimethoxymethane carbonylation. J. Catal. 2010, 270, 185–195. [Google Scholar] [CrossRef]
- Mihaly-Cozmuta, L.; Mihaly-Cozmuta, A.; Peter, A.; Nicula, C.; Tutu, H.; Silipas, D. Adsorption of heavy metal cations by Na-clinoptilolite: Equilibrium and selectivity studies. J. Environ. Manag. 2014, 137, 69–80. [Google Scholar] [CrossRef] [PubMed]
- Sen, S.; Bhattacharyya, K.G. Kinetics of adsorption of metal ions on inorganic materials: A review. Adv. Colloid Interface Sci. 2011, 162, 39–58. [Google Scholar]
- Goldani, E.; Moro, C.C.; Maia, S.M. A Study Employing Differents Clays for Fe and Mn Removal in the Treatment of Acid Mine Drainage. Water Air Soil Poll. 2013, 224, 1–11. [Google Scholar] [CrossRef]
- Ho, Y.S.; Mckay, G. Pseudo-second order model for sorption processes. Process Biochem. 1999, 34, 451–465. [Google Scholar] [CrossRef]
- Zhang, S.; Wang, D.; Chen, Y.C.; Zhang, X.W.; Chen, G.J. Adsorption of calcium ion from aqueous solution using Na+-conditioned clinoptilolite for hot-water softening. Huanjing Kexue 2015, 36, 744–750. [Google Scholar] [PubMed]
- Cortés-Martínez, R.; Olguín, M.T.; Solache-Ríos, M. Cesium sorption by clinoptilolite-rich tuffs in batch and fixed-bed systems. Desalination 2010, 258, 164–170. [Google Scholar] [CrossRef]
- Olad, A.; Ahmadi, S.; Rashidzadeh, A. Removal of Nickel(II) from aqueous solutions with polypyrrole modified clinoptilolite: Kinetic and isotherm studies. Desalin. Water Treat. 2013, 51, 7172–7180. [Google Scholar] [CrossRef]
- O’Carroll, D.; Sleep, B.; Krol, M.; Boparai, H.; Kocur, C. Nanoscale zero valent iron and bimetallic particles for contaminated site remediation. Adv. Water Resour. 2013, 51, 104–122. [Google Scholar] [CrossRef]
- Anari-Anaraki, M.; Nezamzadeh-Ejhieh, A. Modification of an Iranian clinoptilolite nanoparticles by hexadecyltrimethyl ammonium cationic surfactant and dithizone for removal of Pb(II) from aqueous solution. J. Colloid Interface Sci. 2015, 440, 272–281. [Google Scholar] [CrossRef] [PubMed]
- Yu, Y.; Shapter, J.G.; Popelka-Filcoff, R.; Bennett, J.W.; Ellis, A.V. Copper removal using bio-inspired polydopamine coated natural zeolites. J. Hazard. Mater. 2014, 273, 174–182. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ruíz-Baltazar, A.; Pérez, R. Kinetic Adsorption Study of Silver Nanoparticles on Natural Zeolite: Experimental and Theoretical Models. Appl. Sci. 2015, 5, 1869-1881. https://doi.org/10.3390/app5041869
Ruíz-Baltazar A, Pérez R. Kinetic Adsorption Study of Silver Nanoparticles on Natural Zeolite: Experimental and Theoretical Models. Applied Sciences. 2015; 5(4):1869-1881. https://doi.org/10.3390/app5041869
Chicago/Turabian StyleRuíz-Baltazar, Alvaro, and Ramiro Pérez. 2015. "Kinetic Adsorption Study of Silver Nanoparticles on Natural Zeolite: Experimental and Theoretical Models" Applied Sciences 5, no. 4: 1869-1881. https://doi.org/10.3390/app5041869
APA StyleRuíz-Baltazar, A., & Pérez, R. (2015). Kinetic Adsorption Study of Silver Nanoparticles on Natural Zeolite: Experimental and Theoretical Models. Applied Sciences, 5(4), 1869-1881. https://doi.org/10.3390/app5041869






