Subclinical Neck Pain Alters Gaze Stability During the Vestibulo-Ocular Reflex
Featured Application
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Eligibility
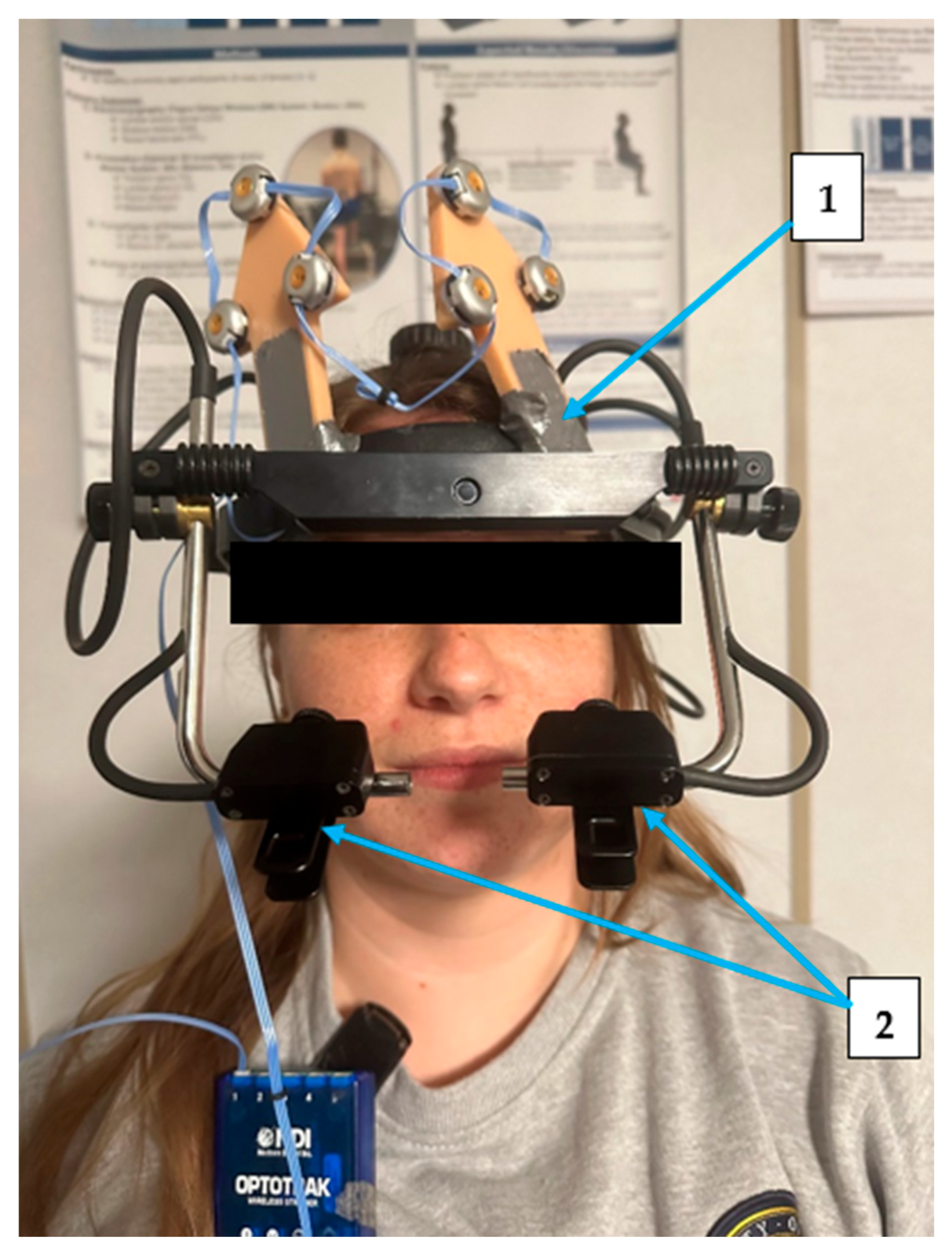
2.3. Instrumentation and Signal Acquisition
2.4. VOR Protocol
2.5. Cervical Proprioception Protocol
2.6. Data Analysis
2.7. Statistical Analysis
3. Results
3.1. Demographics
3.2. VOR Results
3.3. Saccades
3.4. Peak Head Velocity
3.5. Head Rotation
3.6. Error
3.7. Cervical Proprioception
4. Discussion
4.1. VOR Gain
4.2. Corrective Saccades
4.3. Cervical Proprioception
4.4. Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Kazeminasab, S.; Nejadghaderi, S.A.; Amiri, P.; Pourfathi, H.; Araj-Khodaei, M.; Sullman, M.J.M.; Kolahi, A.-A.; Safiri, S. Neck pain: Global epidemiology, trends and risk factors. BMC Musculoskelet. Disord. 2022, 23, 26. [Google Scholar] [CrossRef]
- Bobos, P.; MacDermid, J.; Nazari, G.; Furtado, R. Psychometric properties of the global rating of change scales in patients with neck disorders: A systematic review with meta-analysis and meta-regression. BMJ Open 2019, 9, e033909. [Google Scholar] [CrossRef]
- Ischebeck, B.K.; de Vries, J.; Janssen, M.; van Wingerden, J.P.; Kleinrensink, G.-J.; van der Geest, J.N.; Frens, M.A. Eye stabilization reflexes in traumatic and non-traumatic chronic neck pain patients. Musculoskelet. Sci. Pract. 2017, 29, 72–77. [Google Scholar] [CrossRef]
- Garcia-Perez-Juana, D.; Fernandez-de-Las-Penas, C.; Arias-Buria, J.L.; Cleland, J.A.; Plaza-Manzano, G.; Ortega-Santiago, R. Changes in Cervicocephalic Kinesthetic Sensibility, Widespread Pressure Pain Sensitivity, and Neck Pain After Cervical Thrust Manipulation in Patients With Chronic Mechanical Neck Pain: A Randomized Clinical Trial. J. Manip. Physiol. Ther. 2018, 41, 551–560. [Google Scholar] [CrossRef]
- Kendall, J.C.; French, S.D.; Hartvigsen, J.; Azari, M.F. Chiropractic treatment including instrument-assisted manipulation for non-specific dizziness and neck pain in community-dwelling older people: A feasibility randomised sham-controlled trial. Chiropr. Man. Therap 2018, 26, 14. [Google Scholar] [CrossRef]
- Reid, S.A.; Callister, R.; Katekar, M.G.; Rivett, D.A. Effects of cervical spine manual therapy on range of motion, head repositioning, and balance in participants with cervicogenic dizziness: A randomized controlled trial. Arch. Phys. Med. Rehabil. 2014, 95, 1603–1612. [Google Scholar] [CrossRef]
- Rossi, S.; della Volpe, R.; Ginanneschi, F.; Ulivelli, M.; Bartalini, S.; Spidalieri, R.; Rossi, A. Early somatosensory processing during tonic muscle pain in humans: Relation to loss of proprioception and motor ‘defensive’ strategies. Clin. Neurophysiol. 2003, 114, 1351–1358. [Google Scholar] [CrossRef] [PubMed]
- Strutton, P.H.; Theodorou, S.; Catley, M.; McGregor, A.H.; Davey, N.J. Corticospinal excitability in patients with chronic low back pain. J. Spinal Disord. Tech. 2005, 18, 420–424. [Google Scholar] [CrossRef] [PubMed]
- Waberski, T.D.; Lamberty, K.; Dieckhofer, A.; Buchner, H.; Gobbele, R. Short-term modulation of the ipsilateral primary sensory cortex by nociceptive interference revealed by SEPs. Neurosci. Lett. 2008, 435, 137–141. [Google Scholar] [CrossRef]
- Lee, H.; Nicholson, L.L.; Adams, R.D.; Bae, S.S. Proprioception and rotation range sensitization associated with subclinical neck pain. Spine 2005, 30, E60–E67. [Google Scholar] [CrossRef] [PubMed]
- Haavik, H.; Murphy, B. The role of spinal manipulation in addressing disordered sensorimotor integration and altered motor control. J. Electromyogr. Kinesiol. 2012, 22, 768–776. [Google Scholar] [CrossRef] [PubMed]
- AlDahas, A.; Devecchi, V.; Deane, J.A.; Falla, D. Measurement properties of cervical joint position error in people with and without chronic neck pain. PLoS ONE 2023, 18, e0292798. [Google Scholar] [CrossRef]
- Haavik, H.; Murphy, B. Subclinical neck pain and the effects of cervical manipulation on elbow joint position sense. J. Manip. Physiol. Ther. 2011, 34, 88–97. [Google Scholar] [CrossRef]
- Lee, H.Y.; Wang, J.D.; Yao, G.; Wang, S.F. Association between cervicocephalic kinesthetic sensibility and frequency of subclinical neck pain. Man. Ther. 2008, 13, 419–425. [Google Scholar] [CrossRef]
- Alahmari, K.A.; Reddy, R.S.; Silvian, P.; Ahmad, I.; Nagaraj, V.; Mahtab, M. Influence of chronic neck pain on cervical joint position error (JPE): Comparison between young and elderly subjects. J. Back Musculoskelet. Rehabil. 2017, 30, 1265–1271. [Google Scholar] [CrossRef] [PubMed]
- Johansson, H.; Sojka, P. Pathophysiological mechanisms involved in genesis and spread of muscular tension in occupational muscle pain and in chronic musculoskeletal pain syndromes: A hypothesis. Med. Hypotheses 1991, 35, 196–203. [Google Scholar] [CrossRef] [PubMed]
- Lund, J.P.; Donga, R.; Widmer, C.G.; Stohler, C.S. The pain-adaptation model: A discussion of the relationship between chronic musculoskeletal pain and motor activity. Can. J. Physiol. Pharmacol. 1991, 69, 683–694. [Google Scholar] [CrossRef] [PubMed]
- Beinert, K.; Englert, V.; Taube, W. After-effects of neck muscle vibration on sensorimotor function and pain in neck pain patients and healthy controls—A case-control study. Disabil. Rehabil. 2019, 41, 1906–1913. [Google Scholar] [CrossRef]
- Ranjbaran, M.; Galiana, H.L. Hybrid model of the context dependent vestibulo-ocular reflex: Implications for vergence-version interactions. Front. Comput. Neurosci. 2015, 9, 6. [Google Scholar] [CrossRef]
- De Vries, J.; Ischebeck, B.K.; Voogt, L.P.; Janssen, M.; Frens, M.A.; Kleinrensink, G.-J.; van der Geest, J.N. Cervico-ocular reflex is increased in people with nonspecific neck pain. Phys. Ther. 2016, 96, 1190–1195. [Google Scholar] [CrossRef]
- Gdowski, G.T.; Belton, T.; McCrea, R.A. The neurophysiological substrate for the cervico-ocular reflex in the squirrel monkey. Exp. Brain Res. 2001, 140, 253–264. [Google Scholar] [CrossRef]
- Cullen, K.E. The vestibular system: Multimodal integration and encoding of self-motion for motor control. Trends Neurosci. 2012, 35, 185–196. [Google Scholar] [CrossRef]
- Starkov, D.; Strupp, M.; Pleshkov, M.; Kingma, H.; van de Berg, R. Diagnosing vestibular hypofunction: An update. J. Neurol. 2021, 268, 377–385. [Google Scholar] [CrossRef] [PubMed]
- Schwarz, D.W.F.; Fredrickson, J.M. Rhesus Monkey Vestibular Cortex: A Bimodal Primary Projection Field. Science 1971, 172, 280–281. [Google Scholar] [CrossRef]
- Padoan, S.; Karlberg, M.; Fransson, P.A.; Magnusson, M. Passive sustained turning of the head induces asymmetric gain of the vestibulo-ocular reflex in healthy subjects. Acta Otolaryngol. 1998, 118, 778–782. [Google Scholar] [CrossRef]
- Andrew, D.; Yielder, P.; Haavik, H.; Murphy, B. The effects of subclinical neck pain on sensorimotor integration following a complex motor pursuit task. Exp. Brain Res. 2018, 236, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Baarbé, J.K.; Yielder, P.; Haavik, H.; Holmes, M.W.R.; Murphy, B.A. Subclinical recurrent neck pain and its treatment impacts motor training-induced plasticity of the cerebellum and motor cortex. PLoS ONE 2018, 13, e0193413. [Google Scholar] [CrossRef]
- Sağlam, M.; Lehnen, N. Gaze stabilization in chronic vestibular-loss and in cerebellar ataxia: Interactions of feedforward and sensory feedback mechanisms. J. Vestib. Res. 2014, 24, 425–431. [Google Scholar] [CrossRef] [PubMed]
- Bronstein, A.M.; Patel, M.; Arshad, Q. A brief review of the clinical anatomy of the vestibular-ocular connections—How much do we know? Eye 2015, 29, 163–170. [Google Scholar] [CrossRef]
- Johnston, J.L.; Daye, P.M.; Thomson, G.T. Inaccurate Saccades and Enhanced Vestibulo-Ocular Reflex Suppression during Combined Eye-Head Movements in Patients with Chronic Neck Pain: Possible Implications for Cervical Vertigo. Front. Neurol. 2017, 8, 23. [Google Scholar] [CrossRef]
- Leigh, R.J.; Zee, D.S. The Neurology of Eye Movements; Oxford University Press: New York, NY, USA, 2015. [Google Scholar]
- Soriano-Reixach, M.M.; Rey-Martinez, J.; Altuna, X.; Curthoys, I. Enhanced Eye Velocity With Backup Saccades in vHIT Tests of a Menière Disease Patient: A Case Report. Front. Surg. 2021, 8, 727672. [Google Scholar] [CrossRef] [PubMed]
- Ranjbaran, M.; Smith, H.L.; Galiana, H.L. Automatic Classification of the Vestibulo-Ocular Reflex Nystagmus: Integration of Data Clustering and System Identification. IEEE Trans. Biomed. Eng. 2016, 63, 850–858. [Google Scholar] [CrossRef]
- Schmidt, R.A.; Lee, T.D. Motor Control and Learning: A Behavioral Emphasis, 5th ed.; Human Kinetics: Champaign, IL, USA, 2011. [Google Scholar]
- Campbell, D.; Murphy, B.A.; Burkitt, J.; La Delfa, N.; Sanmugananthan, P.; Ambalavanar, U.; Yielder, P. Cervico-Ocular and Vestibulo-Ocular Reflexes in Subclinical Neck Pain and Healthy Individuals: A Cross-Sectional Study. Brain Sci. 2023, 13, 1603. [Google Scholar] [CrossRef] [PubMed]
- Kelders, W.; Kleinrensink, G.; Geest, J.V.D.; Schipper, I.; Feenstra, L.; Zeeuw, C.D.; Frens, M. The cervico-ocular reflex is increased in whiplash injury patients. J. Neurotrauma 2005, 22, 133–137. [Google Scholar] [CrossRef]
- Montfoort, I.; Kelders, W.P.A.; van der Geest, J.N.; Schipper, I.B.; Feenstra, L.; de Zeeuw, C.I.; Frens, M.A. Interaction between Ocular Stabilization Reflexes in Patients with Whiplash Injury. Investig. Ophthalmol. Vis. Sci. 2006, 47, 2881–2884. [Google Scholar] [CrossRef]
- Della Santina, C.C.; Cremer, P.D.; Carey, J.P.; Minor, L.B. Comparison of Head Thrust Test With Head Autorotation Test Reveals that the Vestibulo-ocular Reflex Is Enhanced During Voluntary Head Movements. Arch. Otolaryngol.—Head. Neck Surg. 2002, 128, 1044–1054. [Google Scholar] [CrossRef]
- Rogers, R.G. The effects of spinal manipulation on cervical kinesthesia in patients with chronic neck pain: A pilot study. J. Manip. Physiol. Ther. 1997, 20, 80–85. [Google Scholar]
- Feipel, V.; Salvia, P.; Klein, H.; Rooze, M. Head repositioning accuracy in patients with whiplash-associated disorders. Spine 2006, 31, E51–E58. [Google Scholar] [CrossRef]
- Heikkila, H.; Astrom, P.G. Cervicocephalic kinesthetic sensibility in patients with whiplash injury. Scand. J. Rehabil. Med. 1996, 28, 133–138. Available online: https://www.ncbi.nlm.nih.gov/pubmed/8885035 (accessed on 10 June 2024). [CrossRef]
- Heikkila, H.V.; Wenngren, B.I. Cervicocephalic kinesthetic sensibility, active range of cervical motion, and oculomotor function in patients with whiplash injury. Arch. Phys. Med. Rehabil. 1998, 79, 1089–1094. [Google Scholar] [CrossRef] [PubMed]
- Faul, F.; Erdfelder, E.; Lang, A.G.; Buchner, A. G*Power 3: A flexible statistical power analysis program for the social, behavioral, and biomedical sciences. Behav. Res. Methods 2007, 39, 175–191. [Google Scholar] [CrossRef]
- Oldfield, R.C. The assessment and analysis of handedness: The Edinburgh inventory. Neuropsychologia 1971, 9, 97–113. [Google Scholar] [CrossRef] [PubMed]
- Von Korff, M.; Ormel, J.; Keefe, F.J.; Dworkin, S.F. Grading the severity of chronic pain. Pain 1992, 50, 133–149. Available online: https://journals.lww.com/pain/fulltext/1992/08000/grading_the_severity_of_chronic_pain.2.aspx (accessed on 3 June 2024). [CrossRef] [PubMed]
- Vernon, H.T.; Humphreys, B.K.; Hagino, C.A. A systematic review of conservative treatments for acute neck pain not due to whiplash. J. Manip. Physiol. Ther. 2005, 28, 443–448. [Google Scholar] [CrossRef]
- Baarbé, J.; Yielder, P.; Daligadu, J.; Behbahani, H.; Haavik, H.; Murphy, B. A novel protocol to investigate motor training-induced plasticity and sensorimotor integration in the cerebellum and motor cortex. J. Neurophysiol. 2014, 111, 715–721. [Google Scholar] [CrossRef]
- Daligadu, J.; Haavik, H.; Yielder, P.C.; Baarbe, J.; Murphy, B. Alterations in cortical and cerebellar motor processing in subclinical neck pain patients following spinal manipulation. J. Manip. Physiol. Ther. 2013, 36, 527–537. [Google Scholar] [CrossRef]
- Stewart, V.; Mendis, M.D.; Rowland, J.; Choy, N.L. Construction and Validation of the Vestibular Screening Tool for Use in the Emergency Department and Acute Hospital Setting. Arch. Phys. Med. Rehabil. 2015, 96, 2153–2160. [Google Scholar] [CrossRef]
- Begum, M.R.; Hossain, M.A. Validity and reliability of visual analogue scale (VAS) for pain measurement. J. Med. Case Rep. Rev. 2019, 2, 11. Available online: https://jmcrr.info/index.php/jmcrr/article/view/44 (accessed on 11 June 2024).
- Bijur, P.E.; Silver, W.; Gallagher, E.J. Reliability of the visual analog scale for measurement of acute pain. Acad. Emerg. Med. 2001, 8, 1153–1157. [Google Scholar] [CrossRef]
- Dimitriadis, Z.; Strimpakos, N.; Kapreli, E.; Oldham, J. Validity of visual analog scales for assessing psychological states in patients with chronic neck pain. J. Musculoskelet. Pain 2014, 22, 242–246. [Google Scholar] [CrossRef]
- McCarthy, M.J.H.; Grevitt, M.P.; Silcocks, P.; Hobbs, G. The reliability of the Vernon and Mior neck disability index, and its validity compared with the short form-36 health survey questionnaire. Eur. Spine J. 2007, 16, 2111–2117. [Google Scholar] [CrossRef]
- Vernon, H.; Mior, S. The Neck Disability Index: A study of reliability and validity. J. Manip. Physiol. Ther. 1991, 14, 409–415. [Google Scholar]
- Gombaut, C.; Holmes, S.A. Sensorimotor Integration and Pain Perception: Mechanisms Integrating Nociceptive Processing. A Systematic Review and ALE-Meta Analysis. Front. Integr. Neurosci. 2022, 16, 931292. [Google Scholar] [CrossRef] [PubMed]
- Tashjian, R.Z.; Deloach, J.; Porucznik, C.A.; Powell, A.P. Minimal clinically important differences (MCID) and patient acceptable symptomatic state (PASS) for visual analog scales (VAS) measuring pain in patients treated for rotator cuff disease. J. Shoulder Elbow Surg. 2009, 18, 927–932. [Google Scholar] [CrossRef]
- Goh, G.S.; Yue, W.-M.; Guo, C.-M.; Tan, S.-B.; Chen, J.L. Defining threshold values on the neck disability index corresponding to a patient acceptable symptom state in patients undergoing elective surgery for degenerative disorders of the cervical spine. Spine J. 2020, 20, 1316–1326. [Google Scholar] [CrossRef]
- Borg, G.A. Psychophysical bases of perceived exertion. Med. Sci. Sports Exerc. 1982, 14, 377–381. Available online: https://www.ncbi.nlm.nih.gov/pubmed/7154893 (accessed on 12 June 2024). [CrossRef] [PubMed]
- Weber, K.P.; Aw, S.T.; Todd, M.J.; McGarvie, L.A.; Pratap, S.; Curthoys, I.S.; Halmagyi, G.M. Inter-ocular differences of the horizontal vestibulo-ocular reflex during impulsive testing. Prog. Brain Res. 2008, 171, 195–198. [Google Scholar] [CrossRef]
- Schubert, M.C.; Della Santina, C.C.; Shelhamer, M. Incremental angular vestibulo-ocular reflex adaptation to active head rotation. Exp. Brain Res. 2008, 191, 435–446. [Google Scholar] [CrossRef]
- Angelaki, D.E.; Cullen, K.E. Vestibular System: The Many Facets of a Multimodal Sense. Annu. Rev. Neurosci. 2008, 31, 125–150. [Google Scholar] [CrossRef]
- Omer, W.; Abdulhadi, K. Physiology and Diagnostic Tests of the Vestibular System. In Textbook of Clinical Otolaryngology; Al-Qahtani, A., Haidar, H., Larem, A., Eds.; Springer International Publishing: Cham, Switzerland, 2021; pp. 129–134. [Google Scholar] [CrossRef]
- Cohen, J. Statistical Power Analysis for the Behavioral Sciences; Routledge: Milton Park, UK, 1988. [Google Scholar]
- Richardson, J.T.E. Eta squared and partial eta squared as measures of effect size in educational research. Educ. Res. Rev. 2011, 6, 135–147. [Google Scholar] [CrossRef]
- Benjamini, Y.; Hochberg, Y. Controlling the False Discovery Rate: A Practical and Powerful Approach to Multiple Testing. J. R. Stat. Soc. Ser. B Methodol. 2018, 57, 289–300. [Google Scholar] [CrossRef]
- Gray, L. Vestibular System: Structure and Function. Ph.D. Thesis, McGovern Medical School at UTHealth, Houston, TX, USA, 2020. Available online: https://nba.uth.tmc.edu/neuroscience/s2/chapter10.html (accessed on 16 May 2024).
- Kandel, E.R.; Koester, J.; Mack, S.; Siegelbaum, S. Principles of Neural Science, 6th ed.; McGraw Hill: Columbus, OH, USA, 2021. [Google Scholar]
- Maire, R.; van Melle, G. Dynamic asymmetry of the vestibulo-ocular reflex in unilateral peripheral vestibular and cochleovestibular loss. Laryngoscope 2000, 110, 256–263. [Google Scholar] [CrossRef] [PubMed]
- Montfoort, I.; Van Der Geest, J.N.; Slijper, H.P.; De Zeeuw, C.I.; Frens, M.A. Adaptation of the cervico-and vestibulo-ocular reflex in whiplash injury patients. J. Neurotrauma 2008, 25, 687–693. [Google Scholar] [CrossRef]
- Perez-Fernandez, N.; Eza-Nunez, P. Normal Gain of VOR with Refixation Saccades in Patients with Unilateral Vestibulopathy. J. Int. Adv. Otol. 2015, 11, 133–137. [Google Scholar] [CrossRef]
- Brooks, J.X.; Cullen, K.E. Multimodal integration in rostral fastigial nucleus provides an estimate of body movement. J. Neurosci. 2009, 29, 10499–10511. [Google Scholar] [CrossRef]
- MacDougall, H.; Weber, K.; McGarvie, L.; Halmagyi, G.; Curthoys, I. The video head impulse test: Diagnostic accuracy in peripheral vestibulopathy. Neurology 2009, 73, 1134–1141. [Google Scholar] [CrossRef]
- Yang, C.; Lee, J.; Kang, B.; Lee, H.; Yoo, M.; Park, H. Quantitative analysis of gains and catch-up saccades of video-head-impulse testing by age in normal subjects. Clin. Otolaryngol. 2016, 41, 532–538. [Google Scholar] [CrossRef]
- Kabaya, K.; Fukushima, A.; Katsumi, S.; Minakata, T.; Iwasaki, S. Presence of corrective saccades in patients with normal vestibulo-ocular reflex gain in video head impulse test. Front. Neurol. 2023, 14, 1152052. [Google Scholar] [CrossRef]
- O’Leary, S.; Falla, D.; Elliott, J.M.; Jull, G. Muscle Dysfunction in Cervical Spine Pain: Implications for Assessment and Management. J. Orthop. Sports Phys. Ther. 2009, 39, 324–333. [Google Scholar] [CrossRef] [PubMed]
- Peng, B.; Yang, L.; Li, Y.; Liu, T.; Liu, Y. Cervical Proprioception Impairment in Neck Pain-Pathophysiology, Clinical Evaluation, and Management: A Narrative Review. Pain Ther. 2021, 10, 143–164. [Google Scholar] [CrossRef] [PubMed]
- Reddy, R.S.; Tedla, J.S.; Dixit, S.; Abohashrh, M. Cervical proprioception and its relationship with neck pain intensity in subjects with cervical spondylosis. BMC Musculoskelet. Disord. 2019, 20, 447. [Google Scholar] [CrossRef] [PubMed]
- Röijezon, U.; Clark, N.C.; Treleaven, J. Proprioception in musculoskeletal rehabilitation. Part 1: Basic science and principles of assessment and clinical interventions. Manual Ther. 2015, 20, 368–377. [Google Scholar] [CrossRef] [PubMed]
- Treleaven, J.; Peterson, G.; Ludvigsson, M.L.; Kammerlind, A.-S.; Peolsson, A. Balance, dizziness and proprioception in patients with chronic whiplash associated disorders complaining of dizziness: A prospective randomized study comparing three exercise programs. Manual Ther. 2016, 22, 122–130. [Google Scholar] [CrossRef] [PubMed]











| Healthy Control (HC) | Subclinical Neck Pain (SCNP) | |||
| Biological Sex (M:F) | 10:9 | 6:10 | ||
| Age (Years) | 22.53 ± 3.75 | 21.50 ± 3.60 | ||
| Von Kroff CPGS | Grade 0 | 19 | 0 | |
| Grade 1 | 0 | 9 | ||
| Grade 2 | 0 | 5 | ||
| Grade 3 | 0 | 2 | ||
| Grade 4 | 0 | 0 | ||
| Healthy Control (HC) | Subclinical Neck Pain (SCNP) | p-Value | ||
| NDI Score (/50) | 1.63 ± 2.03 | 5.56 ± 4.41 | 0.003 | |
| Pain VAS (/10 cm) | 0.27 ± 0.48 | 1.32 ± 0.99 | 0.104 | |
| Cervical ROM | Right | 65.40 ± 10.73 | 64.96 ±9.23 | 0.897 |
| Left | 67.54 ± 8.88 | 68.92 ± 9.85 | 0.667 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Misketis, C.; Tadayyoni, H.; Yielder, P.C.; Murphy, B. Subclinical Neck Pain Alters Gaze Stability During the Vestibulo-Ocular Reflex. Appl. Sci. 2026, 16, 247. https://doi.org/10.3390/app16010247
Misketis C, Tadayyoni H, Yielder PC, Murphy B. Subclinical Neck Pain Alters Gaze Stability During the Vestibulo-Ocular Reflex. Applied Sciences. 2026; 16(1):247. https://doi.org/10.3390/app16010247
Chicago/Turabian StyleMisketis, Christine, Hamed Tadayyoni, Paul C. Yielder, and Bernadette Murphy. 2026. "Subclinical Neck Pain Alters Gaze Stability During the Vestibulo-Ocular Reflex" Applied Sciences 16, no. 1: 247. https://doi.org/10.3390/app16010247
APA StyleMisketis, C., Tadayyoni, H., Yielder, P. C., & Murphy, B. (2026). Subclinical Neck Pain Alters Gaze Stability During the Vestibulo-Ocular Reflex. Applied Sciences, 16(1), 247. https://doi.org/10.3390/app16010247







