Simulation Study on the Single-Phase Immersion Cooling Performance of Lithium-Ion Battery Packs
Abstract
1. Introduction
2. Mathematical Model of Lithium-Ion Battery Pack
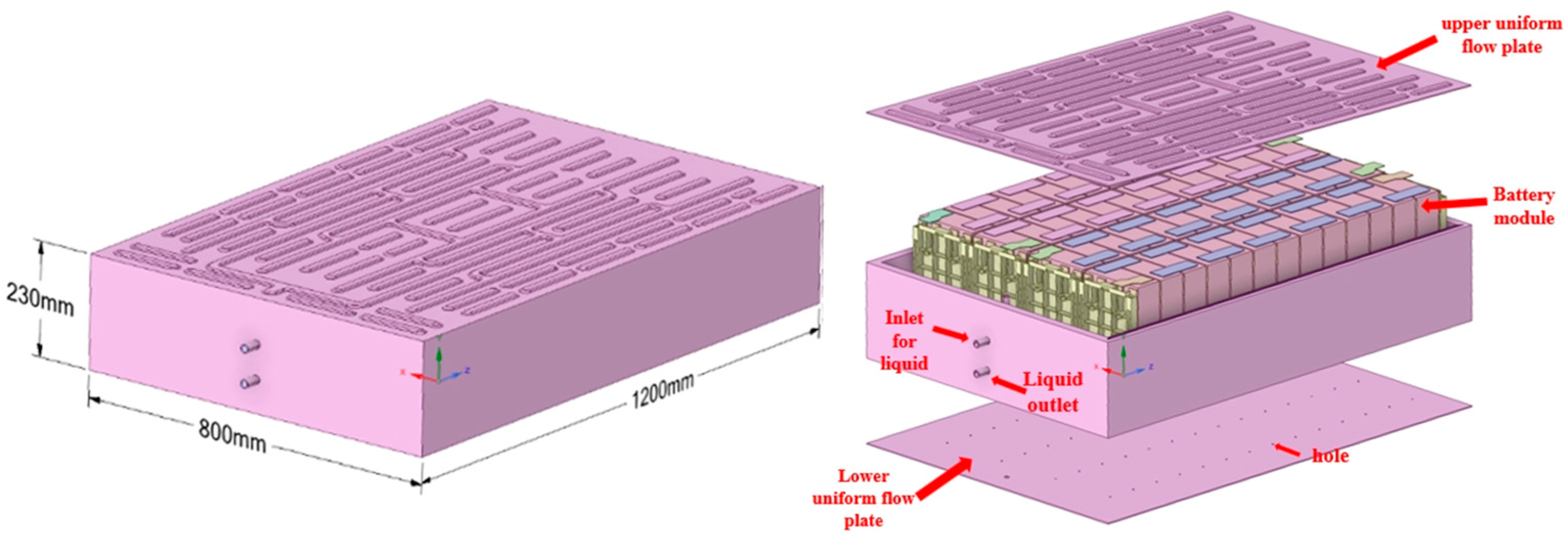
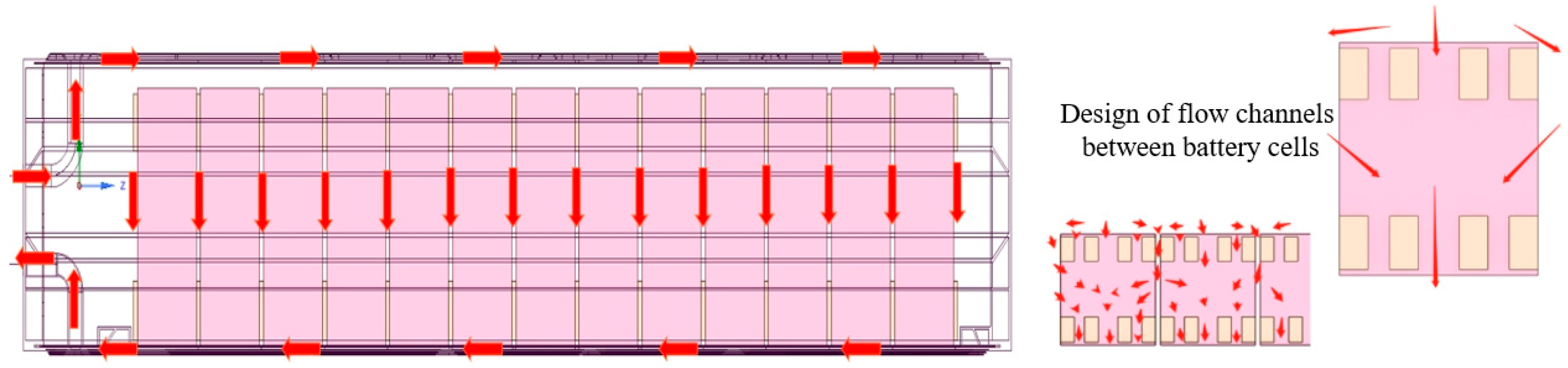
2.1. Physical Model
2.2. Mathematical Method
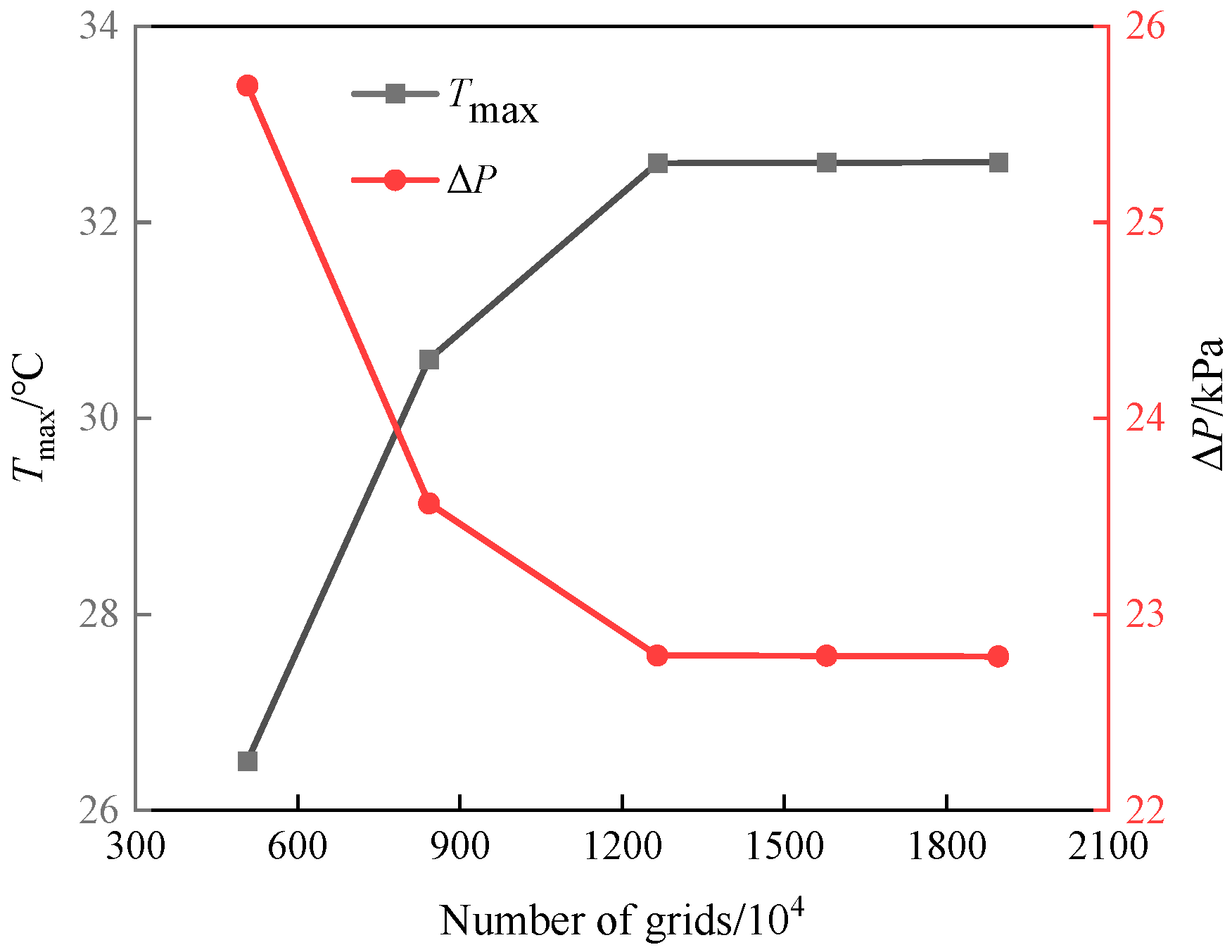
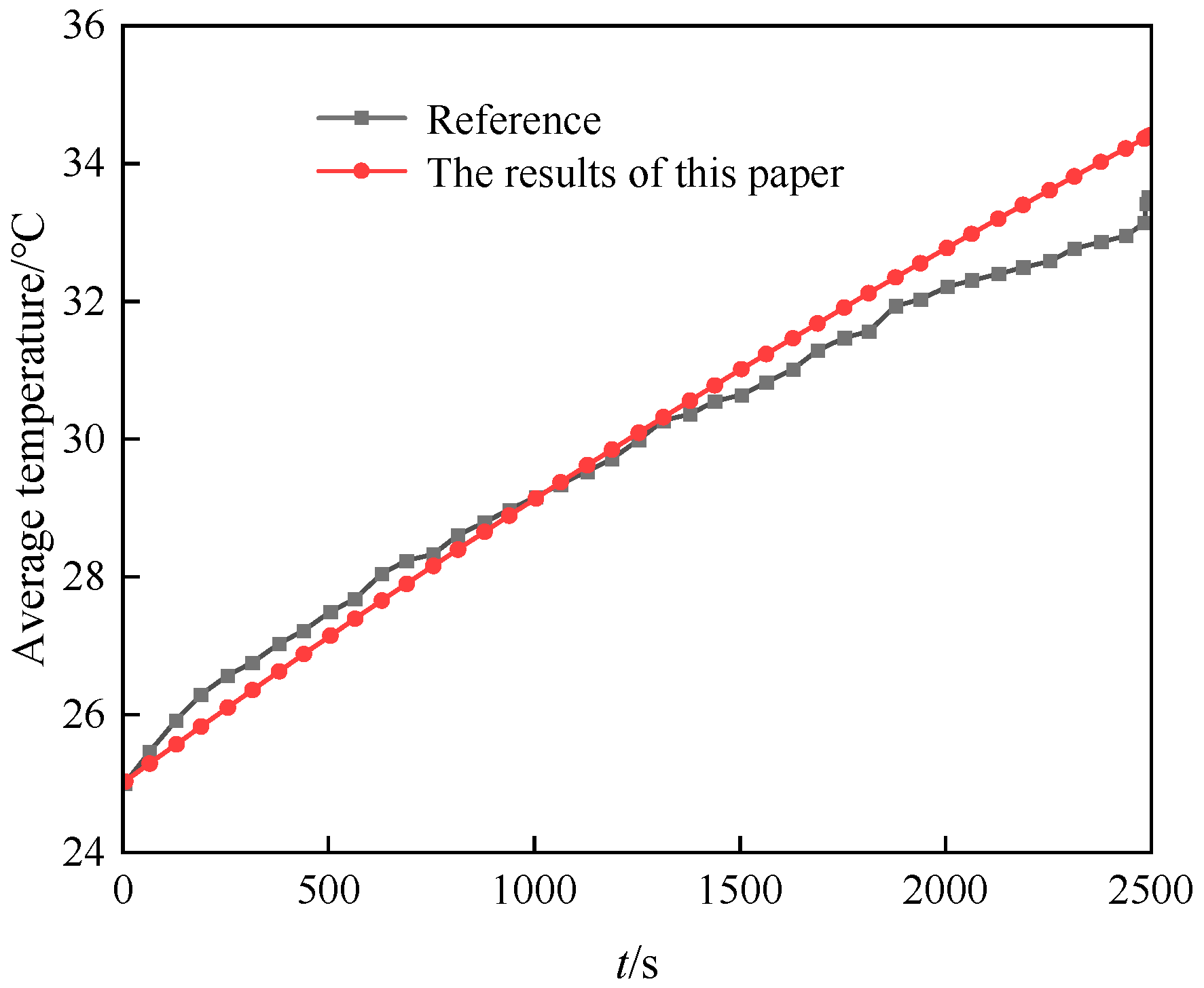
2.3. Validation of Numerical Method
3. Results and Discussion
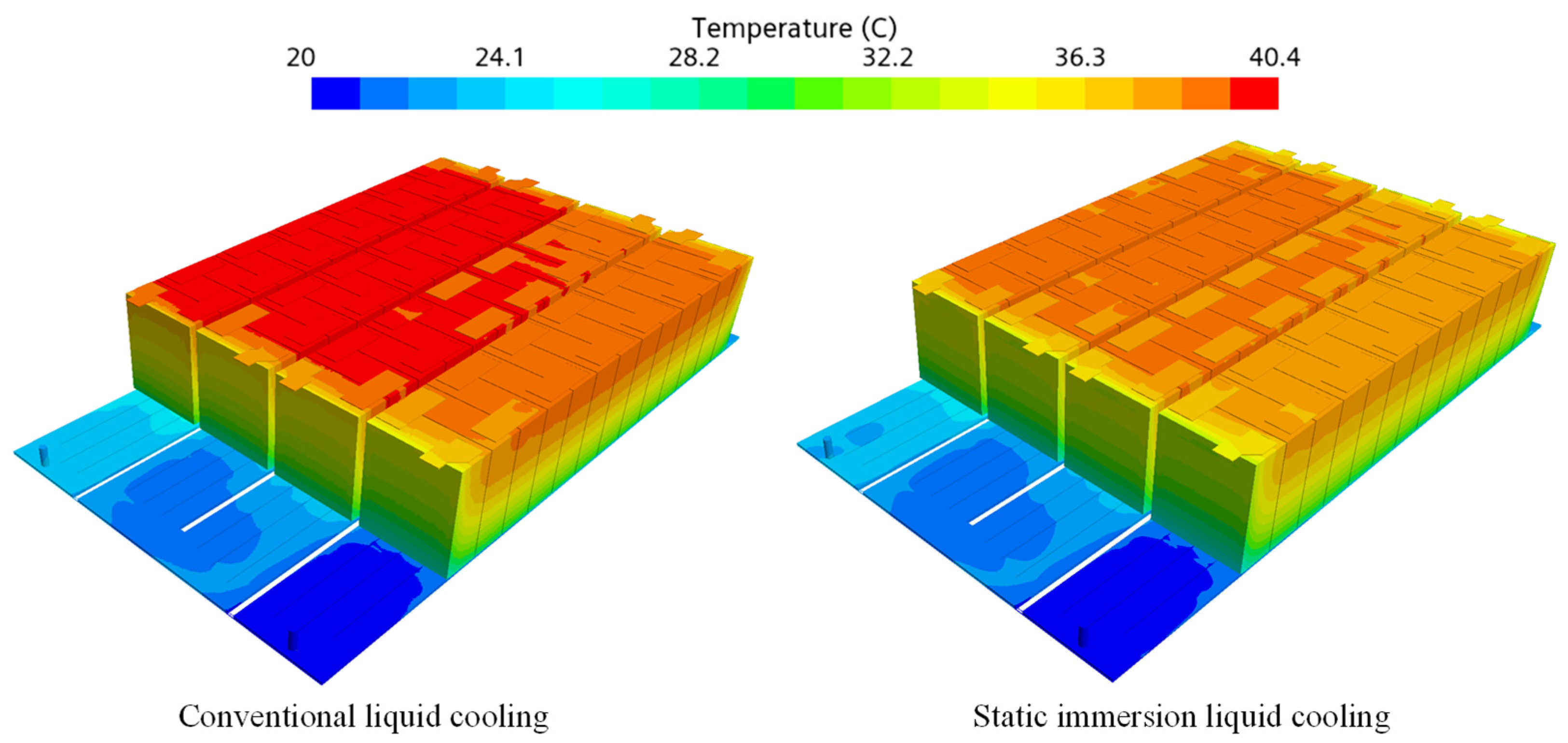
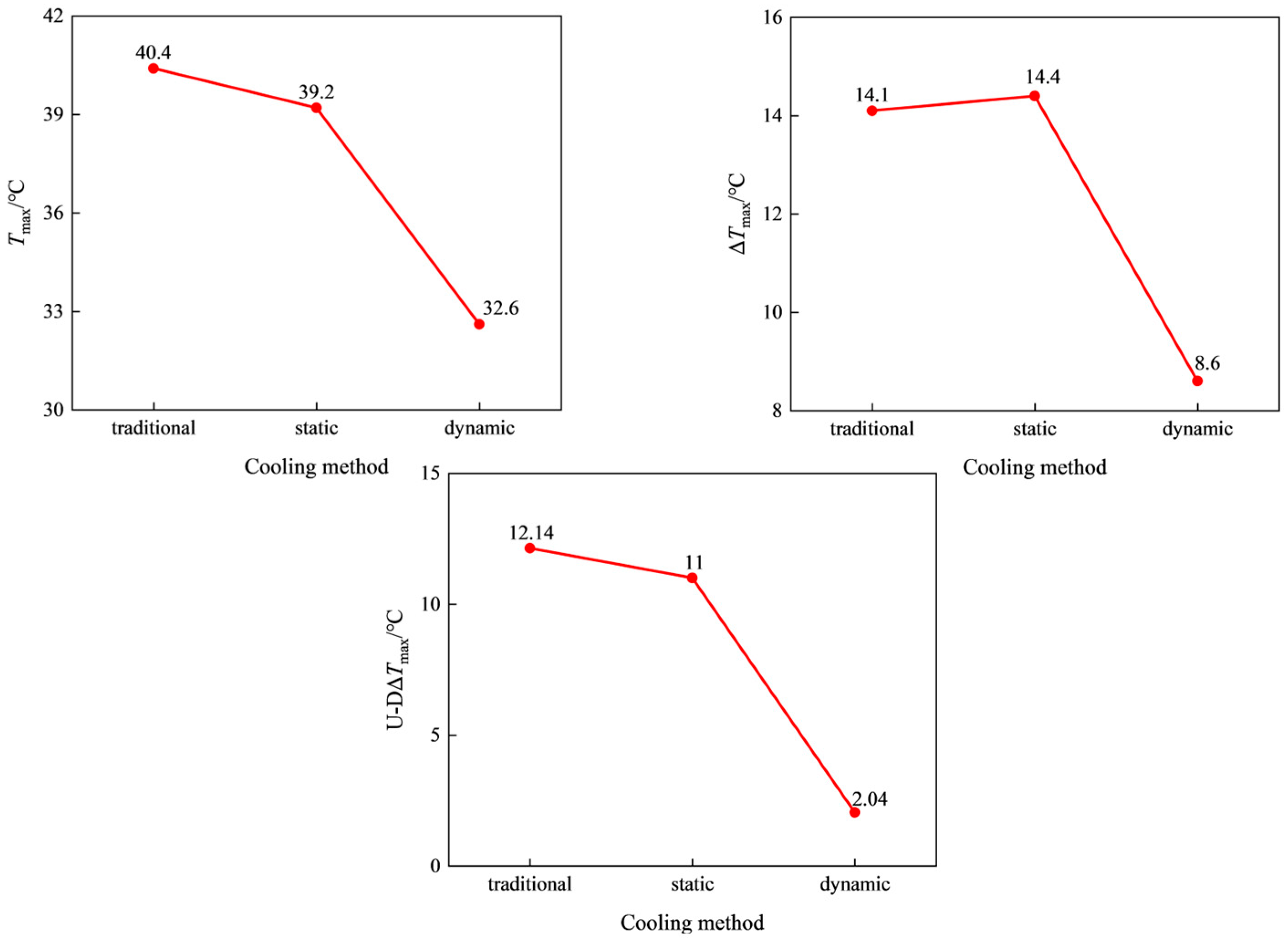
3.1. Simulation Comparison of Traditional Liquid Cooling, Static Immersion Liquid Cooling, and Dynamic Immersion Liquid Cooling
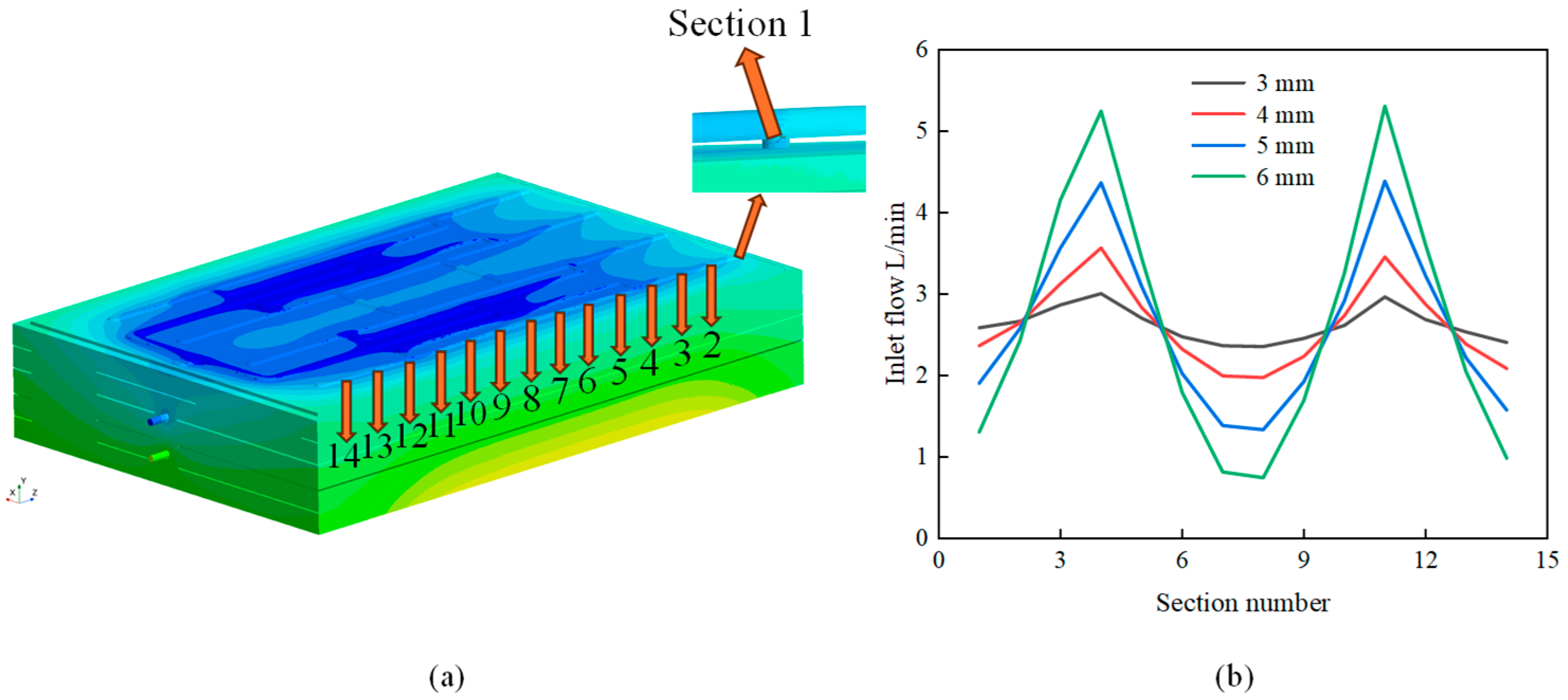
3.2. Impact of the Aperture of the Flow-Equalizing Plate on the Temperature-Rise Characteristics of the Battery Pack
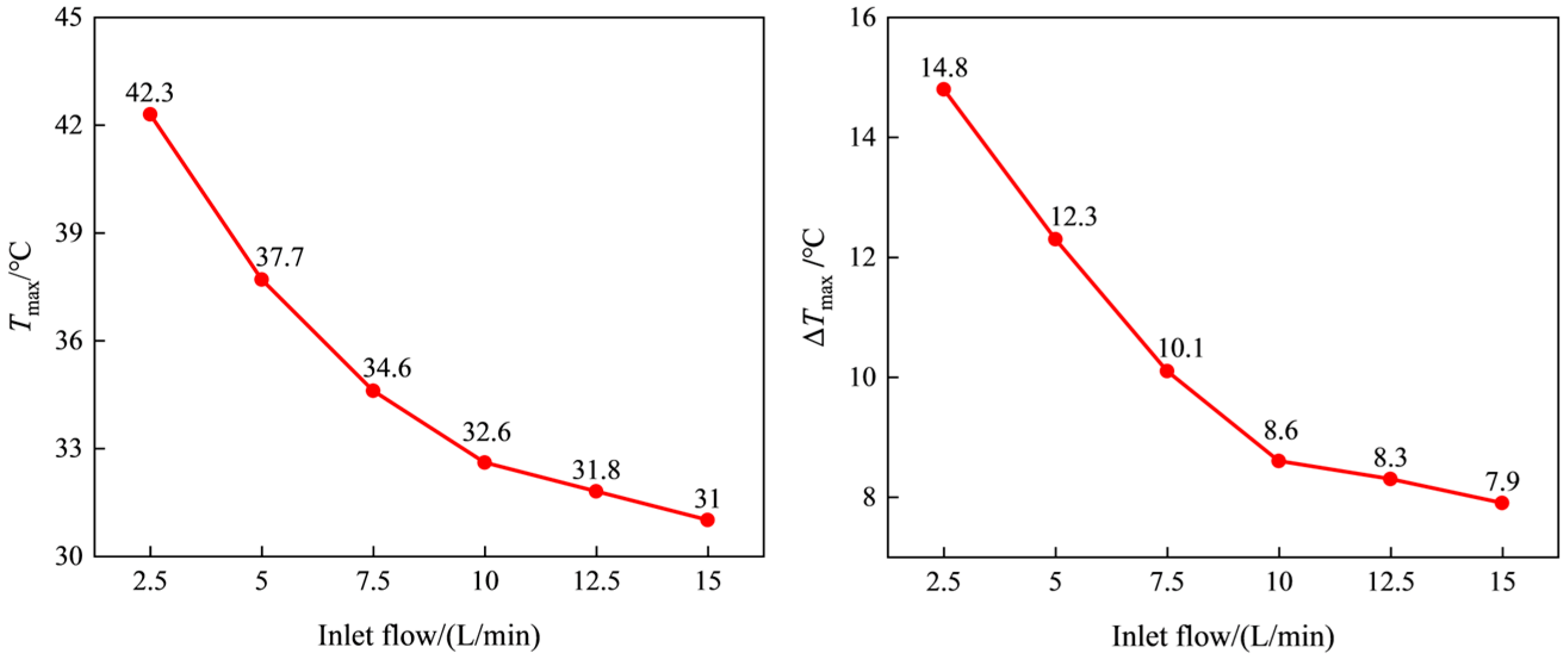
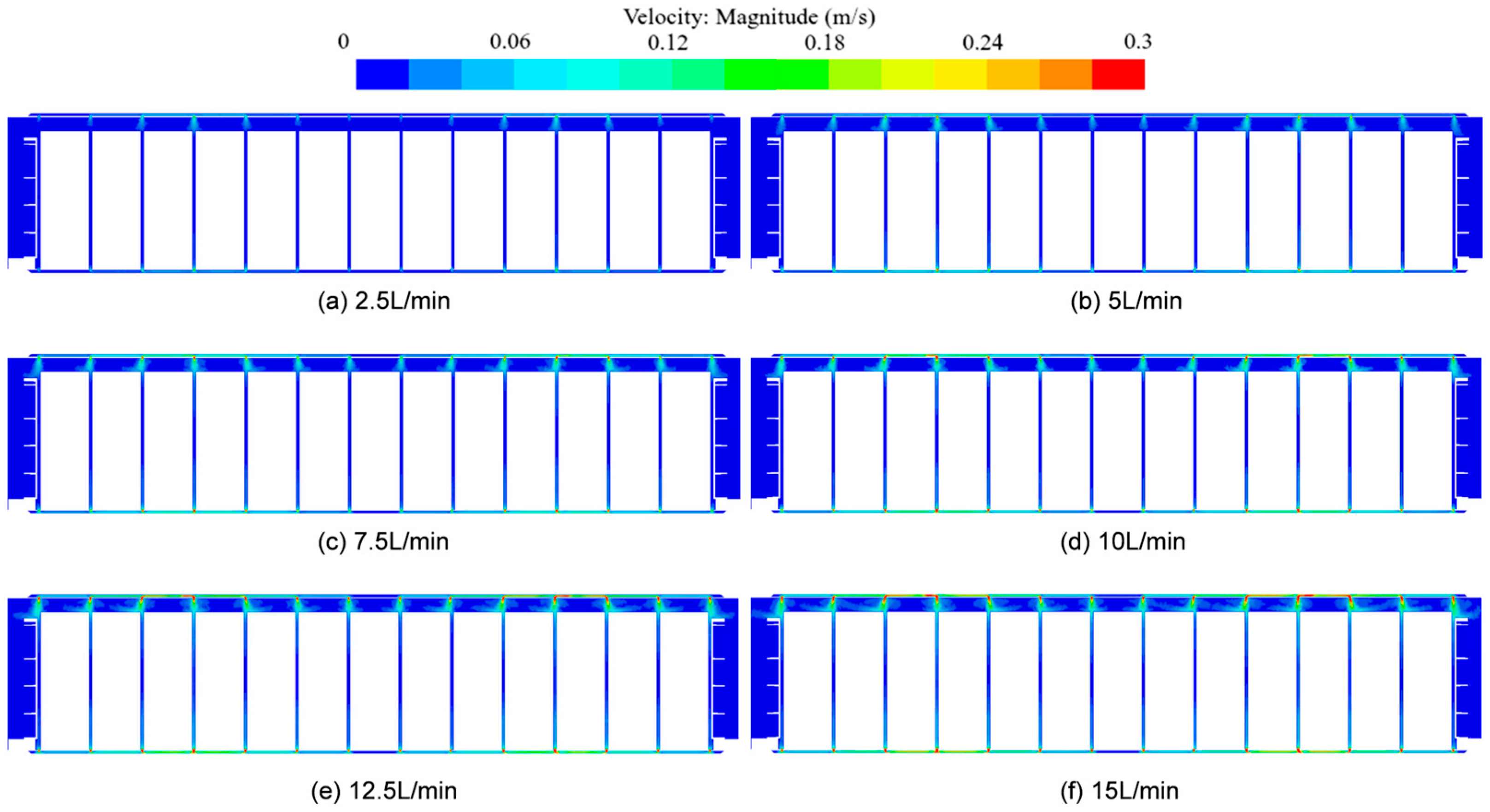
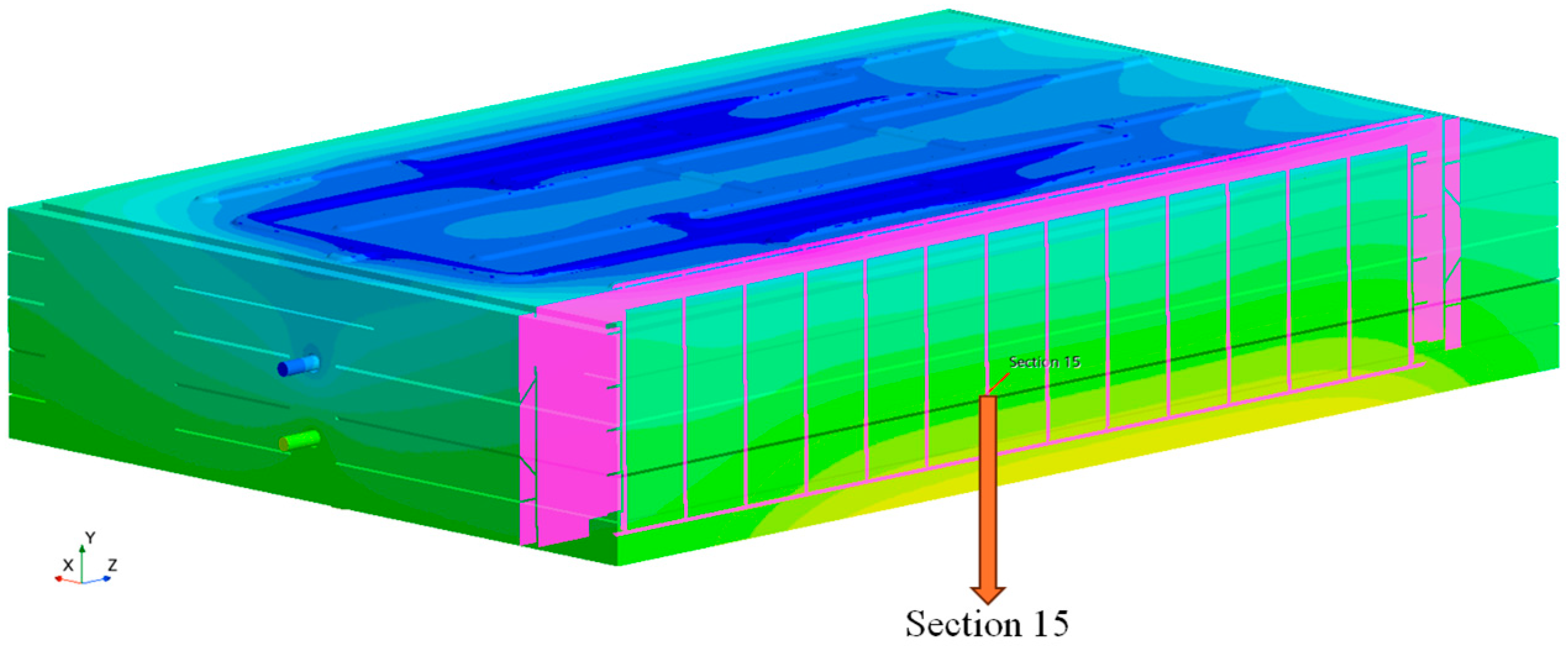
3.3. The Influence of Coolant Inlet Flow Rate on the Temperature Rise Characteristics of Battery Packs
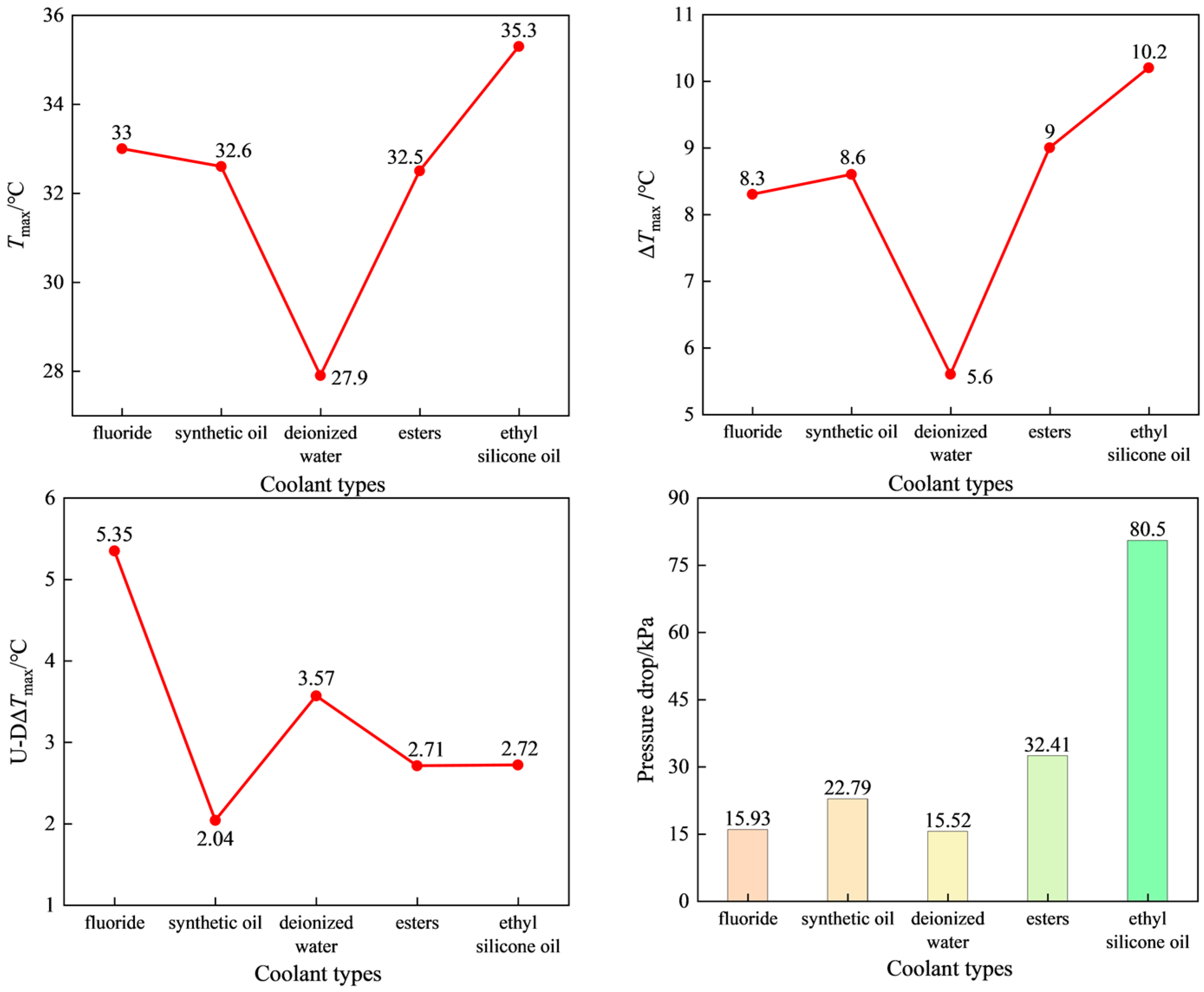
3.4. The Impact of Coolant Types on the Temperature-Rise Characteristics of Battery Packs
4. Conclusions
- (1)
- Compared with traditional liquid cooling and static immersion liquid cooling, dynamic immersion cooling can significantly reduce the temperature difference and the maximum temperature of the battery cells, which is beneficial to improving the safety and lifespan of the cells. The maximum temperature difference between the top and bottom surfaces has decreased significantly, and the problem of excessive temperature difference between the top and bottom surfaces of the cells has been greatly reduced.
- (2)
- A well-designed thermal management system can enhance the temperature uniformity within the system. For example, an appropriate aperture size positively influences the cooling performance of the battery pack. Both the maximum temperature (Tmax) and the maximum temperature difference (ΔTmax) initially decrease and subsequently increase with an increase in aperture size. The optimal cooling effect is achieved when the aperture size is 4 mm.
- (3)
- As the inlet flow rate increases, both Tmax and ΔTmax gradually decrease, and the rate of decrease gradually decreases. The influence on Tmax and ΔTmax is more obvious at low flow rates. It is indicated that the cooling flow rate has a limited impact on the temperature of the battery cells. Considering the reduction in system energy consumption, an appropriate flow rate can be selected to match the system’s design.
- (4)
- Comparing the cooling effects of five types of coolants, deionized water has the best cooling effect, and ethyl silicone oil has the worst cooling effect. The U-DΔTmax of synthetic oil is the lowest, and its uniformity is the best. At the same time, synthetic oil is widely used as the coolant in immersion liquid cooling systems due to its advantages of having a low dielectric constant, good thermal conductivity, low viscosity, and low costs.
- (5)
- Immersion liquid cooling demonstrates significant advantages in improving the uniformity and safety of heat dissipation within battery thermal management systems. However, its widespread adoption is currently hindered by several engineering challenges, including high costs, material compatibility issues, and the development of reliable sealing technologies. Future research should focus on the development of cost-effective and environmentally sustainable cooling media, the optimization of sealing processes, and the exploration of their applicability in high-energy-density configurations, such as solid-state batteries.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Goodenough, J.B.; Park, K.S. The Li-ion rechargeable battery: A perspective. J. Am. Chem. Soc 2013, 135, 1167–1176. [Google Scholar] [CrossRef]
- Wu, W.X.; Wang, S.F.; Wu, W.; Chen, K.; Hong, S.H.; Lai, Y.X. A critical review of battery thermal performance and liquid based battery thermal management. Energy Convers. Manag. 2019, 182, 262–281. [Google Scholar] [CrossRef]
- Ma, S.; Jiang, M.D.; Tao, P.; Song, C.; Wu, J.; Wang, J.; Deng, T.; Shang, W. Temperature effect and thermal impact in lithium-ion batteries: A review. Prog. Nat. Sci. Mater. Int 2018, 28, 653–666. [Google Scholar] [CrossRef]
- Ramadass, P.; Haran, B.; White, R.; Popov, B.N. Capacity fade of Sony 18650 cells cycled at elevated temperatures Part I. Cycling performance. J. Power Sources 2002, 112, 606–613. [Google Scholar] [CrossRef]
- Senyshyn, A.; Muhlbauer, M.J.; Dolotk, O.; Ehrenberg, H. Low-temperature performance of Li-ion batteries: The behavior of lithiated graphite. J. Power Sources 2015, 282, 235–240. [Google Scholar] [CrossRef]
- Wu, T.T.; Wang, C.H.; Hu, Y.X.; Liang, Z.X.; Fan, C.X. Research on electrochemical characteristics and heat generating properties of power battery based on multi-time scales. Energy 2023, 265, 126416. [Google Scholar] [CrossRef]
- Bhosale, A.J.; Deshmukh, V.N. Efficient ways of thermal management of an EV battery. Mater. Today Proc. 2023, 72, 1434–1445. [Google Scholar] [CrossRef]
- Wang, Z.C.; Du, C.Q. A comprehensive review on thermal management systems for power lithium-ion batteries. Renew. Sustain. Energy Rev. 2021, 139, 110685. [Google Scholar] [CrossRef]
- Kim, J.; Oh, J.; Lee, H. Review on battery thermal management system for electric vehicles. Appl. Therm. Eng. 2019, 149, 192–212. [Google Scholar] [CrossRef]
- Tang, X.; Guo, Q.; Li, M.; Wei, C.; Pan, Z.; Wang, Y. Performance analysis on liquid—Cooled battery thermal management for electric vehicles based on machine learning. J. Power Sources 2021, 494, 229727. [Google Scholar] [CrossRef]
- Jiaqiang, E.; Yue, M.; Chen, J.; Zhu, H.; Deng, Y.; Zhu, Y.; Zhang, F.; Wen, M.; Zhang, B.; Kang, S. Effects of the different air cooling strategies on cooling performance of a lithiumion battery module with baffle. Appl. Therm. Eng. 2018, 144, 231–241. [Google Scholar] [CrossRef]
- Dan, D.; Yao, C.N.; Zhang, Y.J.; Zhang, H.; Zeng, Z.; Xu, X. Dynamic thermal behavior of micro heart pipe array-air cooling battery thermal management system based on thermal network mode. Appl. Therm. Eng. 2019, 16, 114183. [Google Scholar] [CrossRef]
- Deng, Y.W.; Feng, C.L.; E, J.Q.; Zhu, H.; Chen, J.W.; Wen, M.; Yin, H.C. Effects of different coolants and cooling strategies on the cooling performance of the power lithium ion battery system: A review. Appl. Therm. Eng. 2018, 142, 10–29. [Google Scholar] [CrossRef]
- Jithin, K.V.; Rajesh, P.K. Numerical analysis of single-phase liquid immersion cooling for lithium-ion battery thermal management using different dielectric fluids. Int. J. Heat Mass Transf. 2022, 188, 122608. [Google Scholar] [CrossRef]
- Tan, X.; Lyu, P.; Fan, Y.; Rao, J.; Ouyang, K. Numerical investigation of the direct liquid cooling of a fast-charging lithium-ion battery pack in hydrofluoroether. Appl. Therm. Eng. 2021, 196, 117279. [Google Scholar] [CrossRef]
- Choi, H.; Lee, H.; Kim, J.; Lee, H. Hybrid single-phase immersion cooling structure for battery thermal management under fast-charging conditions. Energy Convers. Manag. 2023, 287, 117053. [Google Scholar] [CrossRef]
- Bernardi, N.; Pawlikowski, E.; Newman, J. A General Energy-Balance for Battery Systems. J. Electrochem. Soc. 1985, 132, 5–12. [Google Scholar] [CrossRef]
- Chen, Y.H.; Chen, S.; Chen, H.L.; Sun, X.Q.; Luo, Y.Q. Simulation Study on Cooling Performance of Immersion Liquid-cooled System for Energy Storage Battery Packs. Energy Storage Sci. Technol. 2025, 14, 648–658. [Google Scholar]
- Pesaran, A.A.; Keyser, M.; Burch, S. An Approach for Designing Thermal Management Systems for Electric and Hybrid Vehicle Battery Packs; Office of Scientific & Technical Information Technical Reports; National Renewable Energy Laboratory: Golden, CO, USA, 1999.
- Jarrett, A.; Kim, I.Y. Design optimization of electric vehicle battery cooling plates for thermal performance. J. Power Sources 2011, 196, 10359–10368. [Google Scholar] [CrossRef]
- Liu, Z.B.; Zhu, T.; Jiang, W.; Zhang, X.B.; Wang, J.G.; Guan, Q.Q.; Zhang, Q.S.; Zhao, Q.L. Simulation Analysis and Structure Optimization of Cooling System for Energy Storage Lithium-Ion Battery Pack. Electr. Power 2023, 56, 202–210. [Google Scholar]





| Parameter | Value |
|---|---|
| Nominal Voltage/V | 3.2 |
| Size/mm | 174 × 72 × 207 |
| Mass/g | 5600 |
| Capacity/Ah | 314 |
| Charge and Discharge Cut-off Voltage/V | 2.5–3.65 |
| Maximum Continuous Charge and Discharge Current/A | 314 |
| Density/kg/m3 | 2122 |
| Thermal Conductivity/W/(m·K) | 23.88/0.65/23.88 |
| Specific Heat Capacity/J/(kg·K) | 1058 |
| Materials | Density (kg/m3) | Thermal Conductivity W/(m·K) | Dynamic Viscosity (Pa·s) | Specific Heat Capacity J/(kg·K) |
|---|---|---|---|---|
| Synthetic oil | 920 | 0.151 | 0.008 | 1700 |
| Material Types | Materials | Density (kg/m3) | Thermal Conductivity (W/m·K) | Dynamic Viscosity (Pa·s) | Specific Heat Capacity (J/kg·K) |
|---|---|---|---|---|---|
| Water-based fluid | Deionized water | 998.2 | 0.6 | 0.001003 | 4182 |
| Fluoride | D1 | 1540 | 0.062 | 0.000544 | 1100 |
| Hydrocarbons | Synthetic oil | 920 | 0.15 | 0.008 | 1700 |
| Esters | MIVOLT-DF7 | 916 | 0.129 | 0.015 | 1907 |
| Silicone oils | Ethyl silicone oil | 970 | 0.159 | 0.0485 | 1810 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hu, J.; Yu, B.; Chen, Z.; Chen, S.; Wang, S.; Li, F. Simulation Study on the Single-Phase Immersion Cooling Performance of Lithium-Ion Battery Packs. Appl. Sci. 2025, 15, 9531. https://doi.org/10.3390/app15179531
Hu J, Yu B, Chen Z, Chen S, Wang S, Li F. Simulation Study on the Single-Phase Immersion Cooling Performance of Lithium-Ion Battery Packs. Applied Sciences. 2025; 15(17):9531. https://doi.org/10.3390/app15179531
Chicago/Turabian StyleHu, Jiajun, Bin Yu, Zhenshan Chen, Shuaikang Chen, Shuo Wang, and Fengxiang Li. 2025. "Simulation Study on the Single-Phase Immersion Cooling Performance of Lithium-Ion Battery Packs" Applied Sciences 15, no. 17: 9531. https://doi.org/10.3390/app15179531
APA StyleHu, J., Yu, B., Chen, Z., Chen, S., Wang, S., & Li, F. (2025). Simulation Study on the Single-Phase Immersion Cooling Performance of Lithium-Ion Battery Packs. Applied Sciences, 15(17), 9531. https://doi.org/10.3390/app15179531





