Optimization of Phenolic Compounds Extraction from Crataegi Fructus
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals and Reagents
2.2. Laboratory Equipment
2.3. Material and Preparation
2.4. Experimental Variants
2.5. The Determination of the Total Phenolic Content (TPC)
2.6. Determination of Total Flavonoid Content (TFC)
2.7. The Determination of the Total Tannin Content (TTC)
2.8. HPLC-DAD Analysis
2.9. Statistical Analysis
3. Results and Discussion
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Edwards, J.E.; Brown, P.N.; Talent, N.; Dickinson, T.A.; Shipley, P.R. A review of the chemistry of the genus Crataegus. Phytochemistry 2012, 79, 5–26. [Google Scholar] [CrossRef]
- Martín-García, B.; Razola-Díaz, M.D.C.; Gómez-Caravaca, A.M.; Benítez, G.; Verardo, V. Setup of an ultrasonic-assisted extraction to obtain high phenolic recovery in Crataegus monogyna leaves. Molecules 2021, 26, 4536. [Google Scholar] [CrossRef] [PubMed]
- Cheng, L.; Yang, Q.; Li, C.; Zheng, J.; Wang, Y.; Duan, B. Preparation, structural characterization, bioactivities, and applications of Crataegus spp. polysaccharides: A review. Int. J. Biol. Macromol. 2023, 253, 126671. [Google Scholar] [CrossRef] [PubMed]
- Khadivi, A.; Heidari, P.; Rezaei, M.; Safari-Khuzani, A.; Sahebi, M. Morphological variabilities of Crataegus monogyna and C. pentagyna in northeastern areas of Iran. Ind. Crops Prod. 2019, 139, 111531. [Google Scholar] [CrossRef]
- Pavlović, J.; Mitić, S.; Mitić, M.; Kocić, G.; Pavlović, A.; Tošić, S. Variation in the phenolic compounds profile and antioxidant activity in different parts of hawthorn (Crataegus pentagyna Willd.) during harvest periods. Pol. J. Food Nutr. Sci. 2019, 69, 367–378. [Google Scholar] [CrossRef]
- Wang, J.; Xiong, X.; Feng, B. Effect of Crataegus usage in cardiovascular disease prevention: An evidence-based approach. Evid.-Based Complementary Altern. Med. 2013, 2013, 149363. [Google Scholar] [CrossRef]
- Alirezalu, A.; Ahmadi, N.; Salehi, P.; Sonboli, A.; Alirezalu, K.; Mousavi Khaneghah, A.; Barba, F.; Munekata, P.; Lorenzo, J.M. Physicochemical characterization, antioxidant activity, and phenolic compounds of hawthorn (Crataegus spp.) fruits species for potential use in food applications. Foods 2020, 9, 436. [Google Scholar] [CrossRef]
- Abbas, M.; Saeed, F.; Anjum, F.M.; Afzaal, M.; Tufail, T.; Bashir, M.S.; Ishtiaq, A.; Hussain, S.; Suleria, H.A.R. Natural polyphenols: An overview. Int. J. Food Prop. 2017, 20, 1689–1699. [Google Scholar] [CrossRef]
- Jing, Y.; Han, Q.; Li, Z.; Li, Y.; Chang, X.; Hu, B.; Wu, L. Quality control and spectrum–effect relationship of Crataegus pinnatifida polysaccharide from different habitats. Nat. Prod. Res. 2025, 39. [Google Scholar] [CrossRef]
- Williamson, G. The role of polyphenols in modern nutrition. Nutr. Bull. 2017, 42, 226–235. [Google Scholar] [CrossRef]
- Chaves, J.O.; De Souza, M.C.; Da Silva, L.C.; Lachos-Perez, D.; Torres-Mayanga, P.C.; da Fonseca Machado, A.P.; Forster-Carneiro, T.; Vázquez-Espinosa, M.; González-de-Peredo, A.V.; Barbero, G.F.; et al. Extraction of flavonoids from natural sources using modern techniques. Front. Chem. 2020, 8, 507887. [Google Scholar] [CrossRef]
- Buer, C.S.; Imin, N.; Djordjevic, M.A. Flavonoids: New roles for old molecules. J. Integr. Plant Biol. 2010, 52, 98–111. [Google Scholar] [CrossRef]
- Havsteen, B.H. The biochemistry and medical significance of the flavonoids. Pharmacol. Ther. 2002, 96, 67–202. [Google Scholar] [CrossRef]
- The Pharmacopoeia Commision of the People’s Republic of China. Pharmacopoeia of the People’s Republic of China, Part 1, 8th ed.; People’s Medical Publishing House: Beijing, China, 2005; pp. 69–70, 91–92. [Google Scholar]
- American Herbal Pharmacopoeia and Therapeutic Compendium. Hawthorn Berry, Crataegus spp. Analytical, Quality Control, and Therapeutic Monograph; American Herbal Pharmacopoeia: Scotts Valley, CA, USA, 1999. [Google Scholar]
- European Directorate for the Quality of Medicines & Health Care. European Pharmacopoeia, 10th ed.; Council of Europe: Strasbourg, France, 2019; Available online: https://pheur.edqm.eu (accessed on 6 July 2025).
- United States Pharmacopeial Convention. USP–NF 2024, Issue 1; United States Pharmacopeial Convention: Rockville, MD, USA, 2024. [Google Scholar]
- The Council of Europe. European Pharmacopoeia, 5th ed.; Edqm: Strasbourg, France, 2004; pp. 1712–1715. [Google Scholar]
- Alara, O.R.; Abdurahman, N.H.; Ukaegbu, C.I. Extraction of phenolic compounds: A review. Curr. Res. Food Sci. 2021, 4, 200–214. [Google Scholar] [CrossRef]
- Sabaragamuwa, R.; Perera, C.O. Total triterpenes, polyphenols, flavonoids, and antioxidant activity of bioactive phytochemicals of Centella asiatica by different extraction techniques. Foods 2023, 12, 3972. [Google Scholar] [CrossRef]
- Lefebvre, T.; Destandau, E.; Lesellier, E. Selective extraction of bioactive compounds from plants using recent extraction techniques: A review. J. Chromatogr. A 2021, 1635, 461770. [Google Scholar] [CrossRef] [PubMed]
- Herrero, M.; Martín-Álvarez, P.J.; Señoráns, F.J.; Cifuentes, A.; Ibáñez, E. Optimization of accelerated solvent extraction of antioxidants from Spirulina platensis microalga. Food Chem. 2005, 93, 417–423. [Google Scholar] [CrossRef]
- Baiano, A.; Del Nobile, M.A. Antioxidant compounds from vegetable matrices: Biosynthesis, occurrence, and extraction systems. Crit. Rev. Food Sci. Nutr. 2016, 56, 2053–2068. [Google Scholar] [CrossRef]
- Dent, M.; Dragović-Uzelac, V.; Penić, M.; Bosiljkov, T.; Levaj, B. The effect of extraction solvents, temperature and time on the composition and mass fraction of polyphenols in Dalmatian wild sage (Salvia officinalis L.) extracts. Food Technol. Biotechnol. 2013, 51, 84–91. [Google Scholar]
- Jaime, L.; Vázquez, E.; Fornari, T.; del Carmen López-Hazas, M.; García-Risco, M.R.; Santoyo, S.; Reglero, G. Extraction of functional ingredients from spinach (Spinacia oleracea L.) using liquid solvent and supercritical CO2 extraction. J. Sci. Food Agric. 2015, 95, 722–729. [Google Scholar] [CrossRef] [PubMed]
- Wen, L.; Zhang, Z.; Rai, D.K.; Sun, D.; Tiwari, B.K. Ultrasound-assisted extraction (UAE) of bioactive compounds from coffee silverskin: Impact on phenolic content, antioxidant activity, and morphological characteristics. J. Food Process Eng. 2019, 42, e13191. [Google Scholar] [CrossRef]
- Mazilu, I.E.; Vîjan, L.E.; Cosmulescu, S. The influence of harvest moment and cultivar on variability of some chemical constituents and antiradical activity of dehydrated chokeberry pomace. Horticulturae 2022, 8, 544. [Google Scholar] [CrossRef]
- Cosmulescu, S.; Vijan, L.; Mazilu, I.C.; Badea, G. Bioactive compounds in the residue obtained from fruits of some cultivars of Lonicera caerulea. Horticulturae 2024, 10, 211. [Google Scholar] [CrossRef]
- Zhang, Y.; Cai, P.; Cheng, G.; Zhang, Y. A brief review of phenolic compounds identified from plants: Their extraction, analysis, and biological activity. Nat. Prod. Commun. 2022, 17, 1934578X2110697. [Google Scholar] [CrossRef]
- Tamayo-Vives, C.; García-Herrera, P.; Sánchez-Mata, M.C.; Cámara-Hurtado, R.M.; Pérez-Rodríguez, M.L.; Aceituno, L.; Pardo-de-Santayana, M.; Días, M.I.; Barros, L.; Morales, P. Wild fruits of Crataegus monogyna Jacq. and Sorbus aria (L.) crantz: From traditional foods to innovative sources of pigments and antioxidant ingredients for food products. Foods 2023, 12, 2427. [Google Scholar] [CrossRef]
- Janicka, P.; Płotka-Wasylka, J.; Jatkowska, N.; Chabowska, A.; Fares, M.Y.; Andruch, V.; Kaykhaii, M.; Gębicki, J. Trends in the new generation of green solvents in extraction processes. Curr. Opin. Green Sustain. Chem. 2022, 37, 100670. [Google Scholar] [CrossRef]
- Jin, Y.; Hu, D.; Chen, Q.; Shi, C.; Ye, J.; Dai, Z.; Lu, Y. Water-based green and sustainable extraction protocols for value-added compounds from natural resources. Curr. Opin. Green Sustain. Chem. 2023, 40, 100757. [Google Scholar] [CrossRef]
- Garcia-Salas, P.; Morales-Soto, A.; Segura-Carretero, A.; Fernández-Gutiérrez, A. Phenolic-compound-extraction systems for fruit and vegetable samples. Molecules 2010, 15, 8813–8826. [Google Scholar] [CrossRef] [PubMed]
- Da Rosa, G.S.; Vanga, S.K.; Garièpy, Y.; Raghavan, V. Comparison of microwave, ultrasonic and conventional techniques for extraction of bioactive compounds from olive leaves (Olea europaea L.). Innov. Food Sci. Emerg. Technol. 2019, 58, 102234. [Google Scholar] [CrossRef]
- García-Vaquero, M.; Ummat, V.; Tiwari, B.K.; Rajauria, G. Exploring ultrasound, microwave and ultrasound–microwave assisted extraction technologies to increase the extraction of bioactive compounds and antioxidants from brown macroalgae. Mar. Drugs 2020, 18, 172. [Google Scholar] [CrossRef]
- Giosanu, D.; Bărbuceanu, M.; Anghel, M.; Vîjan, L. The determination of the content of phenolic compounds from different romanian wines using Folin-Ciocîlteu method. Curr. Trends Nat. Sci. 2018, 7, 155–159. [Google Scholar]
- Giura, S.; Botu, M.; Vulpe, M.; Vîjan, L.E.; Mitrea, R. Evolution of the polyphenols, flavonoids, and tannins content in walnut leaves and green walnut husk during growing season. Not. Bot. Horti Agrobot. 2019, 47, 1264–1271. [Google Scholar] [CrossRef]
- Ye, Z.; Lu, Q.; Lv, D.; Yang, C. Enzymatic extraction of hawthorn pectin under ultrahigh pressure: Optimization and characterization. Molecules 2025, 30, 2210. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Cheng, B.; Yu, L.; Yuan, G.; Ma, Y.; Zhang, J.; Lin, F. Differential Analysis of Anthocyanins in Red and Yellow Hawthorn (Crataegus pinnatifida) Peel Based on Ultra-High Performance Liquid Chromatography-Electrospray Ionization Tandem Mass Spectrometry. Molecules 2025, 30, 1149. [Google Scholar] [CrossRef] [PubMed]
- Sezer Öztürk, F.; Gökbulut, İ.; Hayaloğlu, A.A. Evaluation of physicochemical characteristics, antioxidant activity and phenolic profile of Crataegus species in Malatya, Turkey. Emir. J. Food Agric. 2023, 35, 78–85. [Google Scholar] [CrossRef]
- Kotsou, K.; Magopoulou, D.; Chatzimitakos, T.; Athanasiadis, V.; Bozinou, E.; Sfougaris, A.I.; Lalas, S.I. Enhancing the nutritional profile of Crataegus monogyna fruits by optimizing the extraction conditions. Horticulturae 2024, 10, 564. [Google Scholar] [CrossRef]
- Paun, G.; Neagu, E.; Albu, C.; Alecu, A.; Seciu-Grama, A.-M.; Radu, G.L. Antioxidant and antidiabetic activity of Cornus mas L. and Crataegus monogyna fruit extracts. Molecules 2024, 29, 3595. [Google Scholar] [CrossRef]
- Belkhir, M.; Rebai, O.; Dhaouadi, K.; Congiu, F.; Tuberoso, C.I.G.; Amri, M.; Fattouch, S. Comparative analysis of Tunisian wild Crataegus azarolus (yellow azarole) and Crataegus monogyna (red azarole) leaf, fruit, and traditionally derived syrup: Phenolic profiles and antioxidant and antimicrobial activities of the aqueous-acetone extracts. J. Agric. Food Chem. 2013, 61, 9594–9601. [Google Scholar] [CrossRef]
- Zhang, Z.; Chang, Q.; Zhu, M.; Huang, Y.; Ho, W.K.K.; Chen, Z.-Y. Characterization of antioxidants present in hawthorn fruits. J. Nutr. Biochem. 2001, 12, 144–152. [Google Scholar] [CrossRef]
- Enescu, I.C.; Cosmulescu, S.; Giosanu, D.; Vijan, L.E. Extraction time influence on the phenolic and carotenoid level, and the dynamics of antioxidant action of chokeberry dry residue. Curr. Trends Nat. Sci. 2022, 11, 6–18. [Google Scholar] [CrossRef]
- Bamba, B.S.B.; Shi, J.; Tranchant, C.C.; Xue, S.J.; Forney, C.F.; Lim, L.T. Influence of extraction conditions on ultrasound-assisted recovery of bioactive phenolics from blueberry pomace and their antioxidant activity. Molecules 2018, 23, 1685. [Google Scholar] [CrossRef] [PubMed]
- Chandini, S.K.; Jaganmohan Rao, L.; Subramanian, R. Influence of extraction conditions on polyphenols content and cream constituents in black tea extracts. Int. J. Food Sci. Technol. 2011, 46, 879–886. [Google Scholar] [CrossRef]
- Jovanović, A.A.; Djordjević, V.B.; Petrović, P.M.; Pljevljakušić, D.S.; Zdunić, G.M.; Šavikin, K.P.; Bugarski, B.M. The influence of different extraction conditions on polyphenol content, antioxidant and antimicrobial activities of wild thyme. J. App. Res. Med. Aromat. Plants 2021, 25, 100328. [Google Scholar] [CrossRef]
- Putnik, P.; Barba, F.J.; Španić, I.; Zorić, Z.; Dragović-Uzelac, V.; Kovačević, D.B. Green extraction approach for the recovery of polyphenols from Croatian olive leaves (Olea europea). Food Bioprod. Process. 2017, 106, 19–28. [Google Scholar] [CrossRef]
- Sik, B.; Ajtony, Z.; Lakatos, E.; Székelyhidi, R. The effects of extraction conditions on the antioxidant activities, total polyphenol and monomer anthocyanin contents of six edible fruits growing wild in Hungary. Heliyon 2022, 8, e12048. [Google Scholar] [CrossRef]
- Arancibia-Avila, P.; Namiesnik, J.; Toledo, F.; Werner, E.; Martinez-Ayala, A.L.; Rocha-Guzmán, N.E.; Gallegos-Infante, J.A.; Gorinstein, S. The influence of different time durations of thermal processing on berries quality. Food Control 2012, 26, 587–593. [Google Scholar] [CrossRef]
- Muradoğlu, F.; Gürsoy, S.; Güler, E. Multivariate analysis revealed the morphological variability among Crataegus species. Yuz. Yıl Univ. J. Agric. Sci. 2021, 31, 961–972. [Google Scholar] [CrossRef]
- Liu, X.M.; Liu, Y.; Shan, C.H.; Yang, X.Q.; Zhang, Q.; Xu, N.; Song, W. Effects of five extraction methods on total content, composition, and stability of flavonoids in jujube. Food Chem. 2022, 14, 100287. [Google Scholar] [CrossRef] [PubMed]
- Messaoudi, M.; Rebiai, A.; Sawicka, B.; Atanassova, M.; Ouakouak, H.; Larkem, I.; Benchikha, N. Effect of extraction methods on polyphenols, flavonoids, mineral elements, and biological activities of essential oil and extracts of Mentha pulegium L. Molecules 2021, 27, 11. Molecules 2021, 27, 11. [Google Scholar] [CrossRef]
- Do, Q.D.; Angkawijaya, A.E.; Tran-Nguyen, P.L.; Huynh, L.H.; Soetaredjo, F.E.; Ismadji, S.; Ju, Y.H. Effect of extraction solvent on total phenol content, total flavonoid content, and antioxidant activity of Limnophila aromatica. J. Food Drug Anal. 2014, 22, 296–302. [Google Scholar] [CrossRef] [PubMed]
- Vo, T.P.; Pham, N.D.; Pham, T.V.; Nguyen, H.Y.; Tran, T.N.H.; Tran, T.N.; Nguyen, D.Q. Green extraction of total phenolic and flavonoid contents from mangosteen (Garcinia mangostana L.) rind using natural deep eutectic solvents. Heliyon 2023, 9, e14884. [Google Scholar] [CrossRef]
- Muflihah, Y.M.; Gollavelli, G.; Ling, Y.C. Correlation study of antioxidant activity with phenolic and flavonoid compounds in 12 Indonesian indigenous herbs. Antioxidants 2021, 10, 1530. [Google Scholar] [CrossRef] [PubMed]
- Movasaghi, Z.; Rehman, S.; ur Rehman, D.I. Fourier transform infrared (FTIR) spectroscopy of biological tissues. Appl. Spectrosc. Rev. 2008, 43, 134–179. [Google Scholar] [CrossRef]
- Ramos, P.M.; Gil, J.M.; Ramos Sánchez, M.C.; Gracia, L.M.N.; Navarro, S.H.; Gil, F.J.M. Vibrational and thermal characterization of seeds, pulp, leaves and seed oil of Rosa rubiginosa. Bol. Soc. Argent. Bot. 2016, 51, 429–439. [Google Scholar] [CrossRef]
- Stamin, F.D.; Vijan, L.E.; Topală, C.M.; Cosmulescu, S.N. The influence of genotype, environmental factors, and location on the nutraceutical profile of Rosa canina L. fruits. Agronomy 2024, 14, 2847. [Google Scholar] [CrossRef]
- Dovbeshko, G.I.; Gridina, N.Y.; Kruglova, E.B.; Pashchuk, O.P. FTIR spectroscopy studies of nucleic acid damage. Talanta 2000, 53, 233–246. [Google Scholar] [CrossRef] [PubMed]
- Topală, C.M.; Rusea, I. Analysis of leaves using FTIR spectroscopy and principal component analysis discrimination of different plant samples. Curr. Trends Nat. Sci. 2018, 7, 286–291. [Google Scholar]
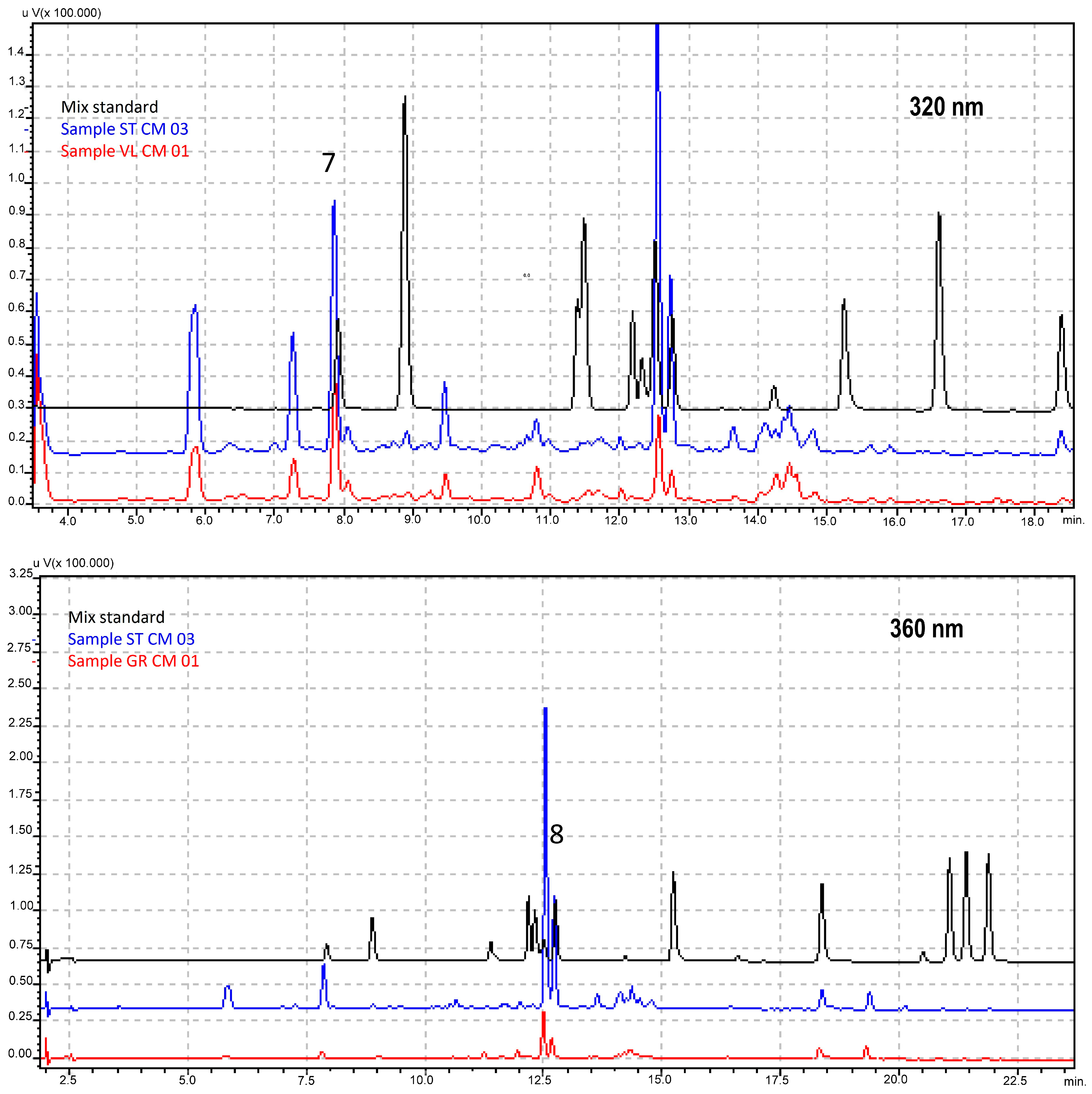
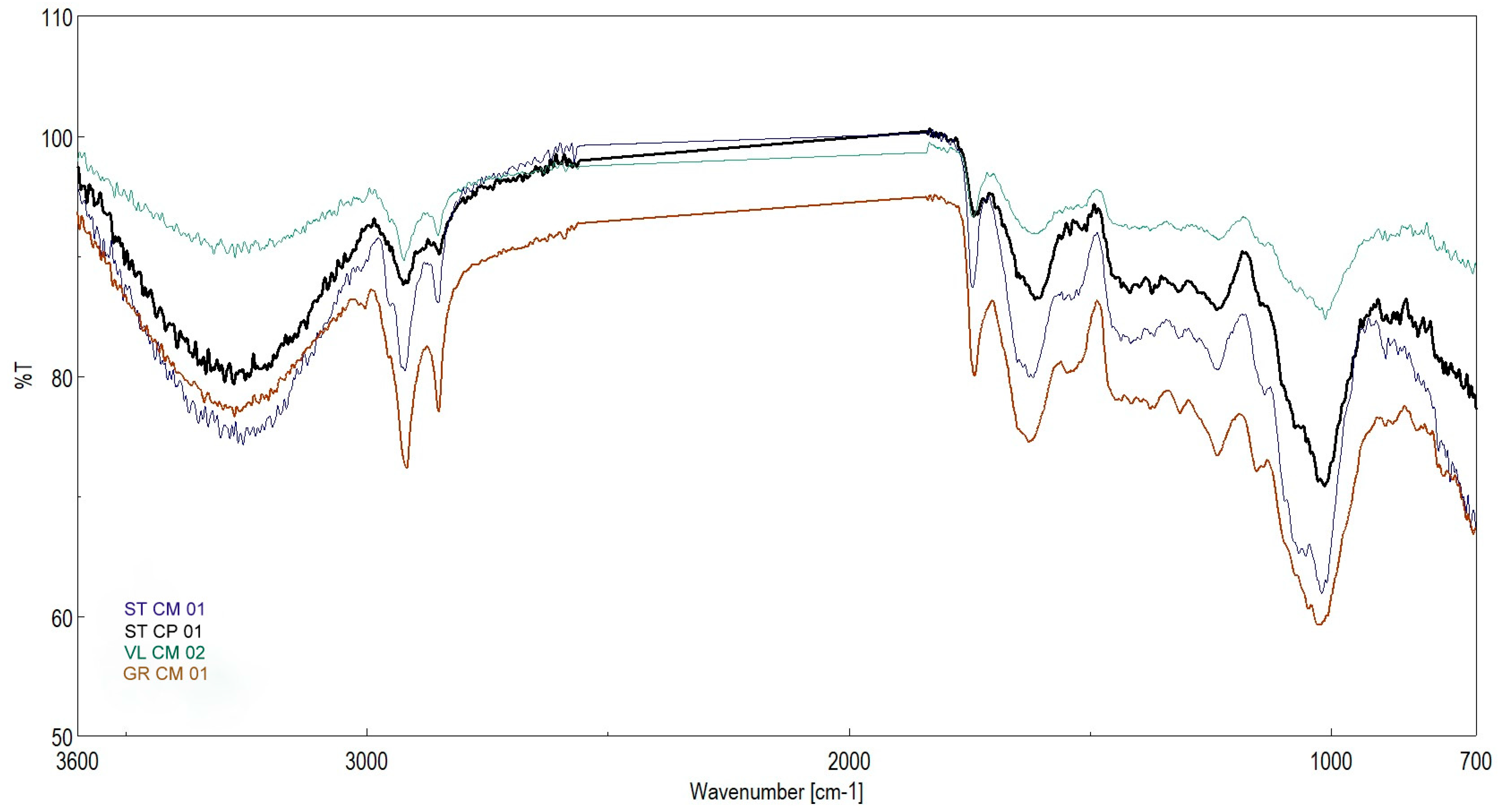
| Genotype | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 |
|---|---|---|---|---|---|---|---|---|---|---|
| GR CM 01 | 10.67 ± 0.08 h | 44.28 ± 0.27 c | 1.47 ± 0.01 d | 29.80 ± 0.05 c | 39.19 ± 0.31 c | 146.63 ± 0.06 b | 50.24 ± 0.43 c | 8.60 ± 0.04 h | 2.55 ± 0.01 g | 1.54 ± 0.02 d |
| GR CM 02 | 9.14 ± 0.07 i | 48.71 ± 0.01 b | 0.63 ± 0.02 h | 14.01 ± 0.01 i | 2.80 ± 0.03 j | 22.35 ± 0.02 h | 21.81 ± 0.01 j | 5.69 ± 0.01 k | 1.57 ± 0.01 j | 1.06 ± 0.01 h |
| GR CM 03 | 11.91 ± 0.11 f | 55.52 ± 0.42 a | 1.65 ± 0.04 c | 19.61 ± 0.15 f | 3.93 ± 0.03 i | 11.63 ± 0.08 j | 29.10 ± 0.35 h | 9.66 ± 0.14 g | 3.86 ± 0.04 d | 0.97 ± 0.01 i |
| ST CM 01 | 43.29 ± 0.32 b | 31.46 ± 0.27 d | 0.90 ± 0.02 e | 33.99 ± 0.17 a | 44.95 ± 0.27 b | 138.05 ± 0.35 c | 42.97 ± 0.33 e | 34.44 ± 0.21 c | 5.51 ± 0.04 b | 2.08 ± 0.01 b |
| ST CM 02 | 19.96 ± 0.01 e | 12.90 ± 0.01 i | 3.48 ± 0.01 a | 18.07 ± 0.03 g | 15.91 ± 0.04 d | 78.01 ± 0.12 e | 29.76 ± 0.06 g | 17.14 ± 0.03 f | 3.81 ± 0.02 | 1.74 ± 0.03 c |
| ST CM 03 | 48.05 ± 0.02 a | 28.33 ± 0.07 f | 2.19 ± 0.04 b | 31.95 ± 0.28 b | 58.29 ± 0.01 a | 187.95 ± 0.14 a | 57.12 ± 0.08 b | 37.73 ± 0.12 b | 5.13 ± 0.02 c | 1.25 ± 0.01 e |
| VL CM 01 | 11.35 ± 0.13 g | 28.48 ± 0.18 f | 0.74 ± 0.08 g | 14.73 ± 0.07 h | 11.43 ± 0.10 f | 82.97 ± 0.58 d | 28.55 ± 0.21 i | 6.77 ± 0.06 i | 1.36 ± 0.01 k | 1.14 ± 0.01 g |
| VL CM 02 | 11.89 ± 0.02 f | 17.72 ± 0.03 h | 0.21 ± 0.02 i | 7.02 ± 0.07 k | 4.82 ± 0.13 h | 27.68 ± 0.26 g | 14.65 ± 0.06 k | 6.08 ± 0.19 j | 1.84 ± 0.01 i | 1.19 ± 0.01 f |
| VL CM 03 | 26.88 ± 0.04 c | 8.59 ± 0.01 j | 0.83 ± 0.01 f | 13.02 ± 0.09 j | 13.28 ± 0.01 e | 44.06 ± 0.10 f | 34.97 ± 0.19 f | 19.80 ± 0.03 e | 9.05 ± 0.02 a | 3.00 ± 0.01 a |
| GR CP 01 | 20.65 ± 0.09 d | 20.64 ± 0.11 g | 1.63 ± 0.03 c | 23.02 ± 0.20 d | 9.93 ± 0.16 g | 21.82 ± 0.09 i | 94.46 ± 0.07 a | 54.72 ± 0.09 a | 2.44 ± 0.02 h | 0.36 ± 0.01 k |
| ST CP 01 | 10.60 ± 0.03 h | 29.26 ± 0.20 e | 0.72 ± 0.01 g | 22.33 ± 0.05 e | 4.04 ± 0.06 i | 22.50 ± 0.04 h | 44.19 ± 0.15 d | 25.04 ± 0.07 d | 3.07 ± 0.02 f | 0.41 ± 0.01 j |
| Mean | 20.40 | 29.63 | 1.31 | 20.69 | 18.96 | 71.24 | 40.71 | 20.52 | 3.65 | 1.34 |
| Median | 11.93 | 28.47 | 0.89 | 19.61 | 11.42 | 44.07 | 34.98 | 17.14 | 3.08 | 1.19 |
| Standard Deviation | 13.26 | 14.36 | 0.89 | 8.27 | 18.68 | 59.17 | 21.08 | 15.52 | 2.18 | 0.73 |
| Minimum | 9.07 | 8.58 | 0.19 | 6.96 | 2.77 | 11.56 | 14.59 | 5.68 | 1.35 | 0.36 |
| Maximum | 48.07 | 55.94 | 3.49 | 34.15 | 58.29 | 188.08 | 94.53 | 54.81 | 9.07 | 3.01 |
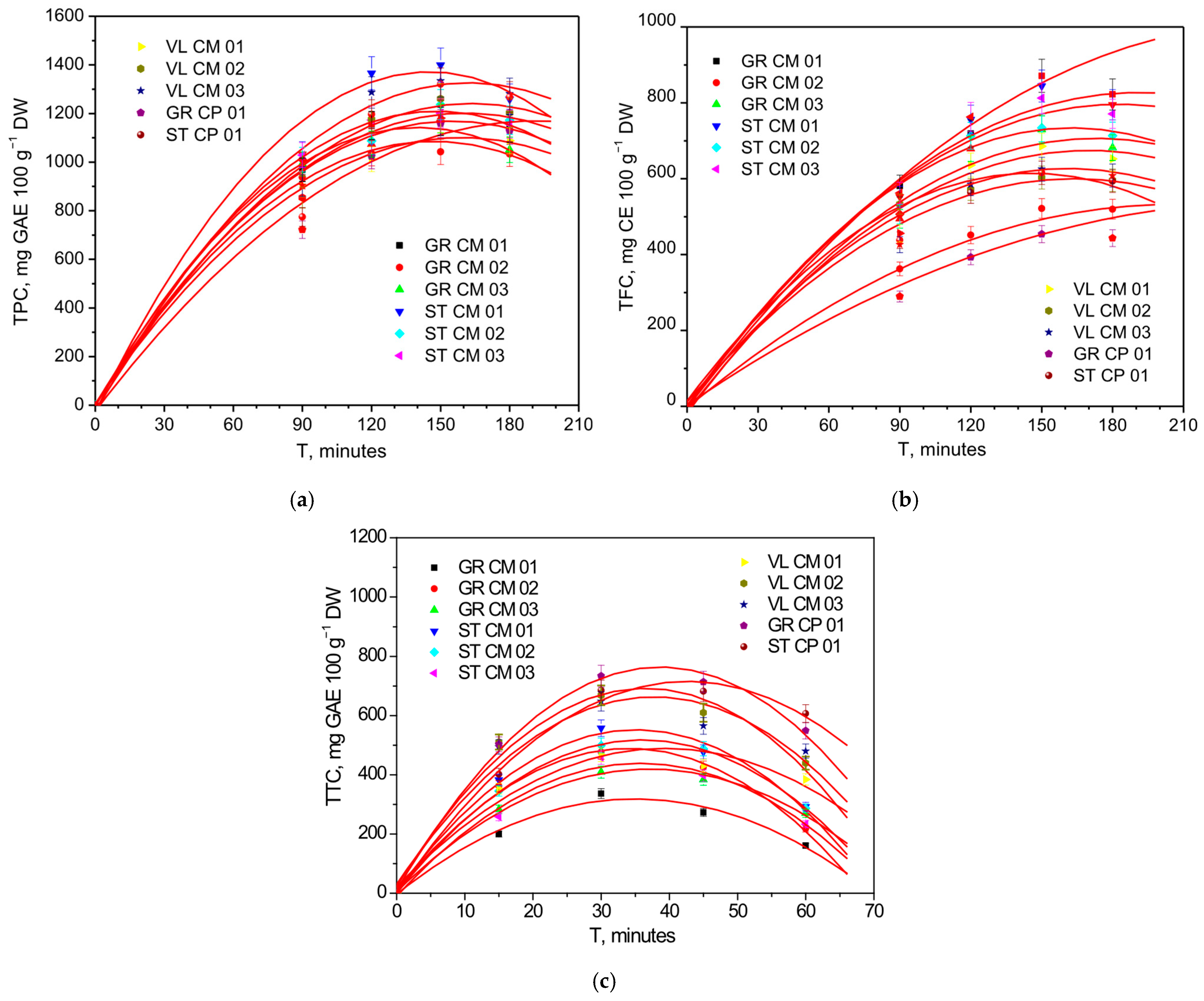
| Genotype | T90 | T120 | T150 | T180 |
|---|---|---|---|---|
| GR CM 01 | 968.47 ± 0.59 e | 1150.49 ± 0.44 f | 1167.73 ± 0.59 h | 1138.88 ± 0.10 g |
| GR CM 02 | 936.83 ± 0.66 f | 1036.23 ± 9.10 i | 1042.70 ± 0.88 j | 1033.70 ± 0.06 k |
| GR CM 03 | 992.31 ± 0.67 d | 1073.49 ± 0.12 h | 1178.86 ± 0.68 g | 1050.50 ± 0.21 j |
| ST CM 01 | 1032.45 ± 0.84 a | 1365.53 ± 2.31 a | 1399.11 ± 0.12 a | 1258.94 ± 0.46 c |
| ST CM 02 | 1009.93 ± 0.17 c | 1086.95 ± 0.42 g | 1236.69 ± 1.00 e | 1172.31 ± 0.18 e |
| ST CM 03 | 1028.87 ± 0.34 b | 1173.42 ± 0.08 e | 1200.92 ± 1.08 f | 1155.89 ± 0.06 f |
| VL CM 01 | 903.06 ± 0.96 g | 1012.29 ± 0.41 k | 1181.00 ± 0.13 g | 1088.41 ± 2.14 i |
| VL CM 02 | 854.13 ± 0.35 h | 1175.62 ± 0.67 d | 1260.51 ± 0.29 d | 1202.87 ± 0.33 d |
| VL CM 03 | 903.95 ± 0.78 g | 1286.78 ± 0.74 b | 1334.77 ± 3.42 b | 1281.49 ± 0.31 a |
| GR CP 01 | 721.79 ± 0.34 j | 1023.51 ± 0.42 j | 1158.82 ± 0.02 i | 1129.11 ± 0.07 h |
| ST CP 01 | 774.14 ± 0.34 i | 1196.67 ± 0.59 c | 1321.16 ± 0.93 c | 1268.16 ± 0.02 b |
| All genotypes | 920.58 ± 99.54 | 1143.42 ± 108.36 | 1225.40 ± 95.83 | 1161.58 ± 82.54 |
| Genotype | T90 | T120 | T150 | T180 |
|---|---|---|---|---|
| GR CM 01 | 580.74 ± 2.82 a | 719.14 ± 0.39 c | 871.13 ± 2.19 a | 822.11 ± 5.87 a |
| GR CM 02 | 262.39 ± 0.08 k | 451.33 ± 0.79 j | 551.61 ± 1.53 j | 519.58 ± 2.18 i |
| GR CM 03 | 424.22 ± 0.49 f | 709.13 ± 0.32 e | 729.32 ± 2.65 e | 681.86 ± 1.53 e |
| ST CM 01 | 552.41 ± 2.60 c | 756.26 ± 2.00 b | 844.47 ± 1.15 b | 744.89 ± 0.30 c |
| ST CM 02 | 436.14 ± 0.37 e | 712.98 ± 0.11 d | 734.04 ± 0.69 d | 713.49 ± 2.14 d |
| ST CM 03 | 562.37 ± 1.42 b | 792.70 ± 0.70 a | 812.44 ± 0.18 c | 770.81 ± 3.64 b |
| VL CM 01 | 356.05 ± 0.22 h | 635.86 ± 0.54 f | 685.60 ± 1.04 f | 652.44 ± 1.76 f |
| VL CM 02 | 532.83 ± 0.71 d | 571.55 ± 1.49 h | 602.90 ± 0.39 i | 596.13 ± 1.21 g |
| VL CM 03 | 376.40 ± 0.39 g | 584.91 ± 1.97 g | 640.94 ± 0.53 h | 588.17 ± 0.98 h |
| GR CP 01 | 289.49 ± 0.34 j | 393.07 ± 0.60 k | 453.92 ± 1.28 k | 432.20 ± 1.66 j |
| ST CP 01 | 337.93 ± 0.49 i | 563.24 ± 2.15 i | 645.42 ± 1.23 g | 593.63 ± 1.87 gh |
| All genotypes | 428.27 ± 110.61 | 626.38 ± 123.00 | 688.34 ± 123.24 | 646.85 ± 111.48 |
| Genotype | T15 | T30 | T45 | T60 |
|---|---|---|---|---|
| GR CM 01 | 200.02 ± 0.61 k | 336.74 ± 0.12 k | 273.23 ± 0.90 k | 261.13 ± 0.62 i |
| GR CM 02 | 362.24 ± 0.75 f | 476.70 ± 0.38 g | 425.19 ± 0.82 h | 217.84 ± 0.25 k |
| GR CM 03 | 283.29 ± 0.56 i | 409.27 ± 0.25 j | 383.89 ± 0.30 j | 270.31 ± 0.09 h |
| ST CM 01 | 382.96 ± 0.64 e | 557.69 ± 0.66 e | 477.58 ± 0.59 f | 292.59 ± 0.58 f |
| ST CM 02 | 345.36 ± 0.40 h | 499.33 ± 0.38 f | 488.28 ± 0.65 e | 286.93 ± 0.40 g |
| ST CM 03 | 258.46 ± 0.69 j | 457.93 ± 0.61 i | 397.88 ± 0.34 i | 233.56 ± 0.49 j |
| VL CM 01 | 353.12 ± 0.37 g | 467.92 ± 0.27 h | 430.97 ± 0.93 g | 384.51 ± 0.74 e |
| VL CM 02 | 510.93 ± 0.34 a | 668.35 ± 0.33 c | 609.80 ± 0.06 c | 439.81 ± 0.56 d |
| VL CM 03 | 494.22 ± 0.16 c | 648.31 ± 0.21 d | 565.44 ± 0.37 d | 480.12 ± 0.27 c |
| GR CP 01 | 502.93 ± 0.30 b | 733.65 ± 0.63 a | 713.77 ± 0.73 a | 549.05 ± 0.25 b |
| ST CP 01 | 401.91 ± 0.38 d | 685.57 ± 0.69 b | 682.14 ± 0.59 b | 607.00 ± 0.32 a |
| All genotypes | 372.31 ± 98.69 | 540.13 ± 123.69 | 495.29 ± 130.37 | 365.71 ± 130.51 |
| Genotype | Polynomial Equation | R2 |
|---|---|---|
| GR CM 01 | TPC = −1.6221 + 15.2944 x − 0.0497 x2 | 0.9992 |
| TFC = −6.1267 + 9.0964 x − 0.0254 x2 | 0.9939 | |
| TTC = −2.6514 + 18.3407 x − 0.2617 x2 | 0.9815 | |
| GR CM 02 | TPC = 2.2876 + 14.7186 x − 0.0502 x2 | 0.9993 |
| TFC = −5.9149 + 5.2737 x − 0.0126 x2 | 0.9959 | |
| TTC = 10.2189 + 28.1838 x − 0.4143 x2 | 0.9938 | |
| GR CM 03 | TPC = 2.8052 + 16.1058 x − 0.0572 x2 | 0.9995 |
| TFC = −23.2279 + 8.6563 x − 0.0255 x2 | 0.9913 | |
| TTC = 6.0937 + 22.2767 x − 0.3001 x2 | 0.9951 | |
| ST CM 01 | TPC = −23.5601 + 18.9416 x − 0.0646 x2 | 0.9958 |
| TFC = −23.3852 + 9.6008 x − 0.0271 x2 | 0.9924 | |
| TTC = 7.5269 + 31.0223 x − 0.4415 x2 | 0.9913 | |
| ST CM 02 | TPC = −1.9259 + 15.4288 x − 0.0492 x2 | 0.9978 |
| TFC = −2.0688 + 8.8739 x − 0.0268 x2 | 0.9924 | |
| TTC = 0.8469 + 28.7488 x − 0.3995 x2 | 0.9997 | |
| ST CM 03 | TPC = −1.9588 + 16.3405 x − 0.0551 x2 | 0.9998 |
| TFC = −7.1126 + 9.0923 x − 0.0257 x2 | 0.9909 | |
| TTC = −9.6106 + 25.0927 x − 0.3508 x2 | 0.9841 | |
| VL CM 01 | TPC = 3.4655 + 13.8322 x − 0.0433 x2 | 0.9988 |
| TFC = −10.8491 + 7.8683 x − 0.0226 x2 | 0.9931 | |
| TTC = 22.0857 + 23.7584 x − 0.3019 x2 | 0.9627 | |
| VL CM 02 | TPC = −17.7699 + 15.1208 x − 0.0454 x2 | 0.9968 |
| TFC = 15.1336 + 8.2341 x − 0.0283 x2 | 0.9945 | |
| TTC = 24.6786 + 36.5768 x − 0.5009 x2 | 0.9789 | |
| VL CM 03 | TPC = −22.8064 + 16.0956 x − 0.0481 x2 | 0.9964 |
| TFC = 5.0979 + 7.5411 x − 0.0231 x2 | 0.9977 | |
| TTC = 31.8917 + 33.4677 x − 0.4432 x2 | 0.9547 | |
| GR CP 01 | TPC = −29.7164 + 12.7193 x − 0.0353 x2 | 0.9882 |
| TFC = −3.0019 + 5.4754 x − 0.0173 x2 | 0.9948 | |
| TTC = 11.5349 + 38.9339 x − 0.5035 x2 | 0.9951 | |
| ST CP 01 | TPC = −26.8505 + 13.8338 x − 0.0372 x2 | 0.9959 |
| TFC = 1.5791 + 7.1749 x − 0.0213 x2 | 0.9969 | |
| TTC = −0.8357 + 33.6035 x − 0.3941 x2 | 0.9931 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Stamin, F.D.; Topală, C.M.; Mazilu, I.C.; Badea, G.I.; Vijan, L.E.; Cosmulescu, S. Optimization of Phenolic Compounds Extraction from Crataegi Fructus. Appl. Sci. 2025, 15, 9525. https://doi.org/10.3390/app15179525
Stamin FD, Topală CM, Mazilu IC, Badea GI, Vijan LE, Cosmulescu S. Optimization of Phenolic Compounds Extraction from Crataegi Fructus. Applied Sciences. 2025; 15(17):9525. https://doi.org/10.3390/app15179525
Chicago/Turabian StyleStamin, Florin Daniel, Carmen Mihaela Topală, Ivona Cristina Mazilu, Georgiana Ileana Badea, Loredana Elena Vijan, and Sina Cosmulescu. 2025. "Optimization of Phenolic Compounds Extraction from Crataegi Fructus" Applied Sciences 15, no. 17: 9525. https://doi.org/10.3390/app15179525
APA StyleStamin, F. D., Topală, C. M., Mazilu, I. C., Badea, G. I., Vijan, L. E., & Cosmulescu, S. (2025). Optimization of Phenolic Compounds Extraction from Crataegi Fructus. Applied Sciences, 15(17), 9525. https://doi.org/10.3390/app15179525







