1. Background
In recent years, with the widespread development of electromagnetic waves and related technologies across various fields, the issue of electromagnetic wave pollution has become increasingly severe [
1,
2]. Electromagnetic wave pollution adversely affects mobile signal reception and communication quality, posing risks to human health [
3]. Moreover, electromagnetic wave pollution can interfere with the operation of military electronic equipment and may lead to the leakage of critical electronic information [
4,
5,
6]. Therefore, considering the implications for public health, industrial production, and national security, developing cost-effective materials with excellent electromagnetic wave absorption capabilities is of great significance [
7,
8,
9,
10].
Recent reports suggest essential applications of CoFe
2O
4 as electromagnetic wave-absorbing materials [
11]. Due to its unique electromagnetic properties, this material has been extensively studied and plays a crucial role in wave-absorbing applications. CoFe
2O
4 is a magnetic material with high coercivity and saturation magnetization [
12]. In an alternating magnetic field, it undergoes repeated magnetization processes, which cause magnetic hysteresis, leading to the attenuation of electromagnetic waves [
13,
14,
15,
16]. This characteristic makes CoFe
2O
4 an effective wave-absorbing material. Additionally, CoFe
2O
4 can be combined with other materials to further enhance its wave-absorbing performance [
17]. For instance, by combining CoFe
2O
4 with nanomaterials such as graphene, the advantages of both materials can be synergistically utilized to improve the performance of electromagnetic wave absorbers [
18,
19,
20].
Biomass-derived carbon, as a novel microwave absorbing material, has garnered extensive attention and research in recent years [
21]. This material offers several advantages, including widespread availability, renewability, and environmental friendliness, presenting immense potential for various applications [
22,
23]. Zhao et al. optimized the surface structure by adding KOH and controlling the carbonization temperature, resulting in a 3D interconnected skeleton structure that is particularly favorable for electromagnetic wave absorption [
24]. Jiang et al. used waste soybean residue as a biomass carbon precursor, incorporating formaldehyde and KOH through a hydrothermal method to produce porous biomass carbon [
25].
Solidago canadensis, commonly known as Canadian goldenrod, is a widespread invasive species that has caused significant ecological issues in China. While various management strategies have been implemented, finding an effective use for this invasive biomass remains challenging [
26,
27,
28]. Recently, biomass carbon materials have gained attention in fields such as environmental protection and renewable energy, yet research on utilizing Solidago canadensis for biomass carbon is still limited. Carbonizing Solidago canadensis and combining it with nanomaterials makes it possible to produce biomass carbon with excellent electromagnetic wave absorption, catalytic activity, or energy storage properties.
This study employed Solidago canadensis as the biomass carbon precursor, and CoFe
2O
4 particles were introduced via a hydrothermal method [
29]. The resulting biomass carbon composite exhibited a larger specific surface area, better impedance matching, multiple loss mechanisms, and enhanced electromagnetic wave absorption performance [
30]. This approach addresses the ecological concerns posed by Solidago canadensis and transforms waste into valuable resources, achieving a win–win scenario for both economic and ecological benefits [
31].
3. Results and Discussions
Figure 2a,b confirm that the BC powders activated by KOH develop a porous microstructure. This high surface area design effectively enhances polarization loss and optimizes impedance matching. Additionally, the porous structure promotes multiple internal reflections and scattering of incident microwaves, further improving the material’s microwave absorption properties. In
Figure 2c–h, spherical CoFe
2O
4 particles are observed to adhere to the surface of the BC-CFO composite. As the amount of CoFe
2O
4 precursor increases, the deposition of the magnetic components on BC increases significantly. From
Figure 2g,h, it is evident that a large number of CoFe
2O
4 particles aggregate in the BC-CFO3 composite, almost covering the entire surface of the biomass carbon matrix. This destroys the porous structure crucial for microwave absorption and severely impacts impedance matching due to the excess CoFe
2O
4 nanoparticles.
Figure 3a presents the SEM image of CoFe
2O
4 particles attached to BC-CFO2, with diameters ranging from 200 nm to 300 nm.
Figure 3b–d show Fe, Co, and O elements within the CoFe
2O
4 particles, indicating that the spherical CoFe
2O
4 particles synthesized in this study possess intact morphology.
Figure 4a shows that the XRD pattern of BC exhibits two broad peaks around 23° and 43°, corresponding to the (002) and (100) planes of the graphite structure. This suggests that the BC derived from goldenrod after surface activation and vacuum calcination is typical amorphous carbon.
Figure 4b displays the XRD pattern of the BC-CFO composites, where the diffraction peaks of CoFe
2O
4 magnetic particles indicate good crystallinity. Sharp diffraction peaks appear at 2θ angles of 30.1°, 35.4°, 43.1°, 57.0°, and 62.6°, corresponding to the (220), (311), (400), (511), and (440) planes of the cubic CoFe
2O
4 phase.
XPS was used to characterize the elemental composition and valence states of the BC-CFO1 composite material.
Figure 4c shows the XPS full spectrum, which aligns with the EDS results, indicating the presence of four elements: C, O, Fe, and Co in the BC-CFO1 composite [
32]. From the high-resolution C 1s spectrum in
Figure 4d, three peaks can be observed at 284.3 eV, 285.5 eV, and 288.8 eV, corresponding to C–C/C=C bonds, C–O bonds, and C=O bonds, respectively. The O 1s spectrum in
Figure 4e reveals characteristic peaks at 530.1 eV, 531.6 eV, and 533.2 eV, which are attributed to lattice oxygen in CoFe
2O
4, O–C=O, and C–O bonds, respectively [
33]. These functional groups can serve as polarization centers, triggering dipole polarization and relaxation and enhancing electromagnetic dissipation capability.
As shown in the Fe 2p spectrum in
Figure 4f, two distinct peaks at 725.1 eV and 711.5 eV are associated with Fe 2p1/2 and Fe 2p3/2, respectively. Similarly, in the Co 2p spectrum in
Figure 4g, peaks at 797.2 eV and 781.5 eV correspond to the Co
2+ ions in the Co 2p1/2 and Co 2p3/2 states, respectively. Additionally, several satellite peaks in the Fe 2p and Co 2p spectra indicate the presence of small amounts of elemental Fe and Co, or other forms of these elements, in the BC-CFO1 composite material.
The Raman spectra in
Figure 4h show two distinct characteristic peaks at 1350 cm
−1 and 1580 cm
−1 for both BC and BC-CFO composites. The peak near 1350 cm
−1 represents the D band (ID), indicative of amorphous carbon and structural defects, while the peak near 1580 cm
−1 represents the G band (IG), associated with graphitic carbon. The intensity ratio of D to G bands (ID/IG) is commonly used to evaluate the degree of graphitization, where a lower ID/IG ratio indicates a higher degree of graphitization. The ID/IG value for BC is 1.00, indicating typical amorphous carbon, while the ID/IG values for BC-CFO1, BC-CFO2, and BC-CFO3 composites are 1.01, 1.03, and 1.05, respectively. The increasing ID/IG values suggest that the addition of CoFe
2O
4 magnetic particles disrupts the sp
2-hybridized carbon structure, introducing more structural defects and slightly reducing the degree of graphitization.
In
Figure 4i, the BC-CFO composites exhibit an S-shaped hysteresis loop, characteristic of soft magnetic materials, indicating the ferromagnetic properties of the BC-CFO composites. Additionally, the saturation magnetization of the BC-CFO composites shows an increasing trend, with the maximum saturation magnetization of 32.88 emu/g for BC-CFO3 and the minimum of 12.35 emu/g for BC-CFO1. The coercivity ranges from a minimum of 31.61 Oe for BC-CFO2 to a maximum of 251.71 Oe for BC-CFO3. The presence of saturation magnetization and coercivity enhances the magnetic loss performance of the BC-CFO composites.
The standard used for microwave absorption measurement is ASTM D4935 [
34]. The microwave absorption properties of an absorber are typically evaluated by the reflection loss (RL) value, which can be calculated using the transmission line theory through the following equations:
The parameter Zin corresponds to the input impedance, Z0 represents the impedance of free space, ƒ denotes the incident microwave frequency, d indicates the matching thickness, c represents the speed of light, while εr and μr refer to the relative complex permittivity and relative complex permeability of the absorber, respectively. A smaller RL (reflection loss) value indicates stronger microwave absorption. An RL value below −10 dB signifies that over 90% of the incident microwaves can be dissipated within the absorber, and the corresponding bandwidth is defined as the effective absorption bandwidth (EAB).
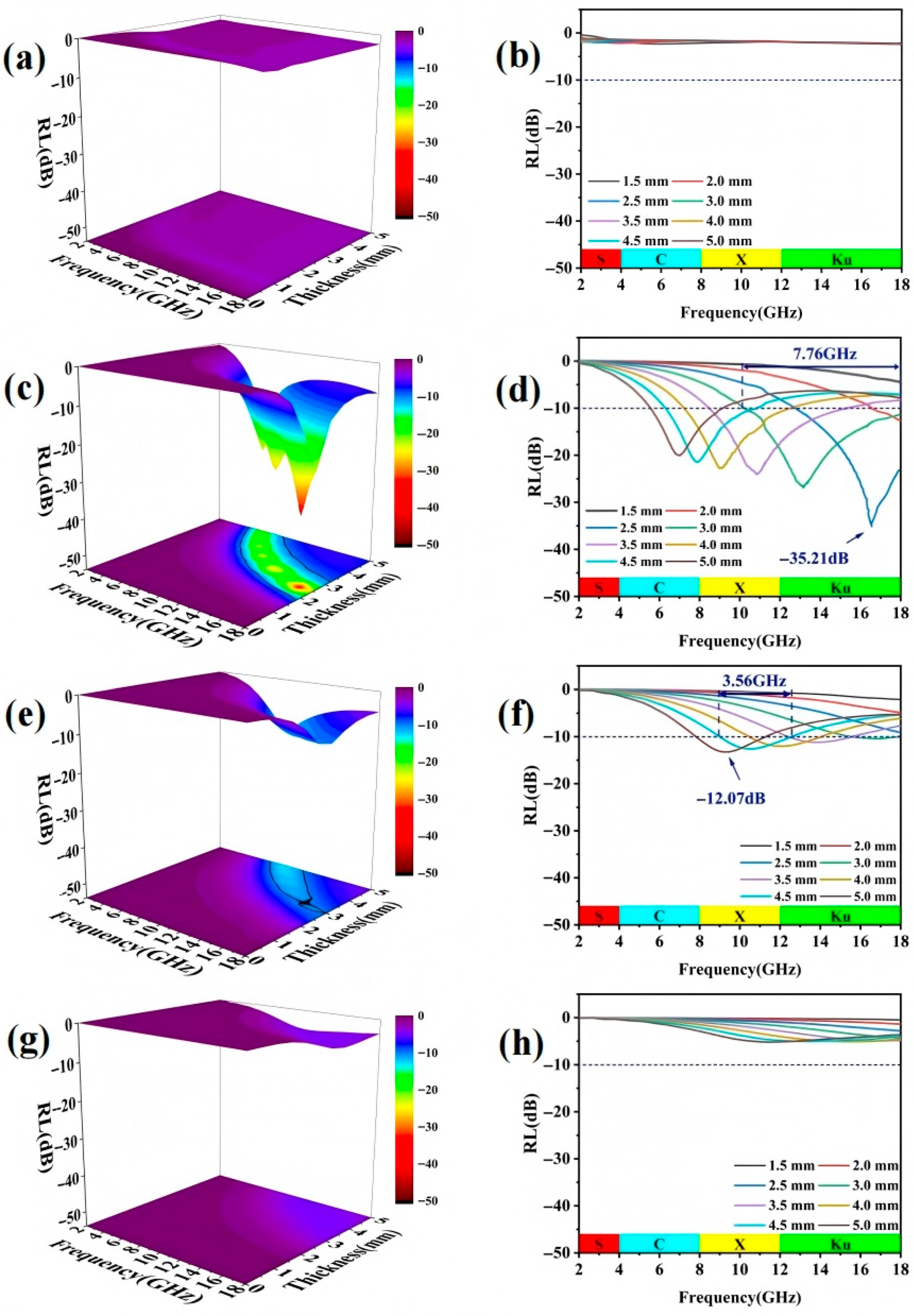
As shown in
Figure 5a,b, although BC exhibits a porous structure, its microwave absorption capability remains poor. In contrast,
Figure 5c,d reveal that the BC-CFO1 composite material demonstrates strong microwave absorption across multiple frequency bands from 5.72 GHz to 18.00 GHz, with matching thicknesses between 2.0 mm and 5.0 mm. At a frequency of 16.76 GHz and a matching thickness of 2.5 mm, the minimum RL (RLmin) value reaches −35.21 dB, and the effective absorption bandwidth (EAB) spans 7.76 GHz, covering nearly half of the X-band and the entire K
u-band. The addition of magnetic CoFe
2O
4 particles significantly enhances the microwave absorption performance.
Figure 5e,f show that the BC-CFO2 composite material exhibits an EAB in the frequency range of 7.84 GHz to 18.00 GHz. At the frequency of 7.88 GHz, with a matching thickness of 5.0 mm, the RL
min reaches −12.07 dB. However, as can be observed in
Figure 5g,h, the microwave absorption capability of BC-CFO3 is relatively weak, with no EAB observed in the frequency range of 2 GHz to 18 GHz and the matching thickness range of 1.5 mm to 5.0 mm. The poor microwave absorption performance of the BC-CFO3 composite is related to impedance matching. It is also evident from
Figure 5 that the RL
min shifts towards lower frequencies as the matching thickness increases. This can be explained by the quarter-wavelength matching model as described below:
The parameters tm and fm represent the matching thickness and frequency at the peak of reflection loss, respectively. According to the quarter-wavelength matching model, if the matching frequency and thickness satisfy the given equation, the phase difference between the incident microwaves at the air–absorber interface and the reflected electromagnetic waves at the absorber–conductor interface is 180°. This phase difference leads to destructive interference, which causes the microwave energy to dissipate completely, resulting in the strongest reflection loss.
As shown in
Figure 6a–d, the real (ε′) and imaginary (ε″) parts of the complex permittivity of BC are significantly higher than those of BC-CFO composites. This is because BC exhibits higher conductivity, thus leading to higher values of ε′ and ε″. However, the dielectric properties of CoFe
2O
4 magnetic particles are weaker, causing the real and imaginary parts of the complex permittivity of BC-CFO composites to decrease substantially with the increase of CoFe
2O
4 particle content. The values of ε′ for BC, BC-CFO1, BC-CFO2, and BC-CFO3 show a general decreasing trend, dropping from 110.33 to 21.28, 7.11 to 3.21, 4.50 to 2.83, and 2.93 to 2.15, respectively. This decrease is attributed to the lag in the response of dipoles to high-frequency electric field variations. Due to the dispersion effect, the values of ε″ for BC, BC-CFO1, BC-CFO2, and BC-CFO3 also show a downward trend, decreasing from 137.66 to 37.4, 6.32 to 2.15, 2.53 to 0.85, and 0.57 to 0.43, respectively. The ε′′ value of BC-CFO3 is nearly zero, indicating minimal influence from the dispersion effect.
In
Figure 6e,f, it can be observed that the real part of the magnetic permeability (μ′) of BC and BC-CFO composites fluctuates within the range of 0.95 to 1.15, while the imaginary part (μ″) fluctuates between −0.15 and 0.10. Compared to the real and imaginary parts of the permittivity for BC and BC-Fe composites, the values and variation of the real and imaginary parts of the magnetic permeability for BC and BC-CFO composites are relatively small. However, overall, the μ′ and μ″ values of BC-CFO composites show a significant enhancement in the frequency range of 2 GHz to 18 GHz compared to BC, which can be attributed to the introduction of the magnetic loss mechanism caused by the addition of magnetic CoFe
2O
4 particles.
Generally, the real part of the complex permittivity (ε′) reflects the material’s ability to store electrical energy, while the imaginary part (ε″) corresponds to its ability to dissipate electrical energy. The dielectric loss tangent (tan δₑ = ε″/ε′) and magnetic loss tangent (tan δμ = μ″/μ′) are commonly used to evaluate dielectric and magnetic loss capabilities, respectively. As shown in
Figure 6a–d, with increasing filler content, both ε′ and ε″ gradually decrease, displaying typical frequency dispersion behavior. This phenomenon can be attributed to the higher electrical conductivity of the carbon matrix after high-temperature calcination compared to that of the CoFe
2O
4 filler. Therefore, increasing the proportion of CoFe
2O
4 reduces the overall conductivity of the composite, leading to the observed decline in both ε′ and ε″. As shown in
Figure 6g, the dielectric loss tangent of BC is the highest, indicating that its dielectric loss ability is stronger than that of the BC-CFO composite. However, this also suggests poorer impedance matching. As the content of CoFe
2O
4 particles increases, the dielectric loss of the BC-CFO composite gradually decreases, contributing to improved impedance matching.
Figure 6h presents the magnetic loss tangent curve, showing that as the content of magnetic CoFe
2O
4 particles increases, the tan δμ value of BC-CFO increases across the 2–6 GHz range. In the 6–18 GHz range, the magnetic loss tangent of BC-CFO exceeds that of BC, suggesting that the magnetic loss capability of the BC-CFO composite is enhanced. The tan δμ values of both BC and BC-CFO follow a similar trend as μ″, indicating that magnetic loss plays a crucial role in microwave absorption. However, the dielectric loss tangent (tan δε) of both BC and BC-CFO is significantly higher than their magnetic loss tangent (tan δμ), indicating that dielectric loss dominates the microwave energy attenuation in these materials.
The dielectric loss capability of the materials is determined by polarization losses, with interfacial polarization and dipole polarization being the main contributors to polarization losses in the 2–18 GHz range. BC exhibits excellent conductivity, which facilitates electron migration and results in significant conductive losses. Additionally, the abundant defects and functional groups within BC act as polarization centers, inducing strong dipole polarization. The introduction of CoFe
2O
4 particles creates numerous heterogeneous interfaces, enhancing interfacial polarization. However, the presence of CoFe
2O
4 particles, which have lower conductivity, affects the conductive losses. When the CoFe
2O
4 particle content is low, BC’s high conductivity still ensures excellent conductive loss. However, excessive CoFe
2O
4 content completely covers the BC surface, significantly impairing its conductivity. This blocks the conductive pathways, reducing the conductive losses. Based on Debye relaxation theory, the polarization process can be described using Cole–Cole semicircles, which can be expressed by an equation relating ε′ and ε′′ as follows:
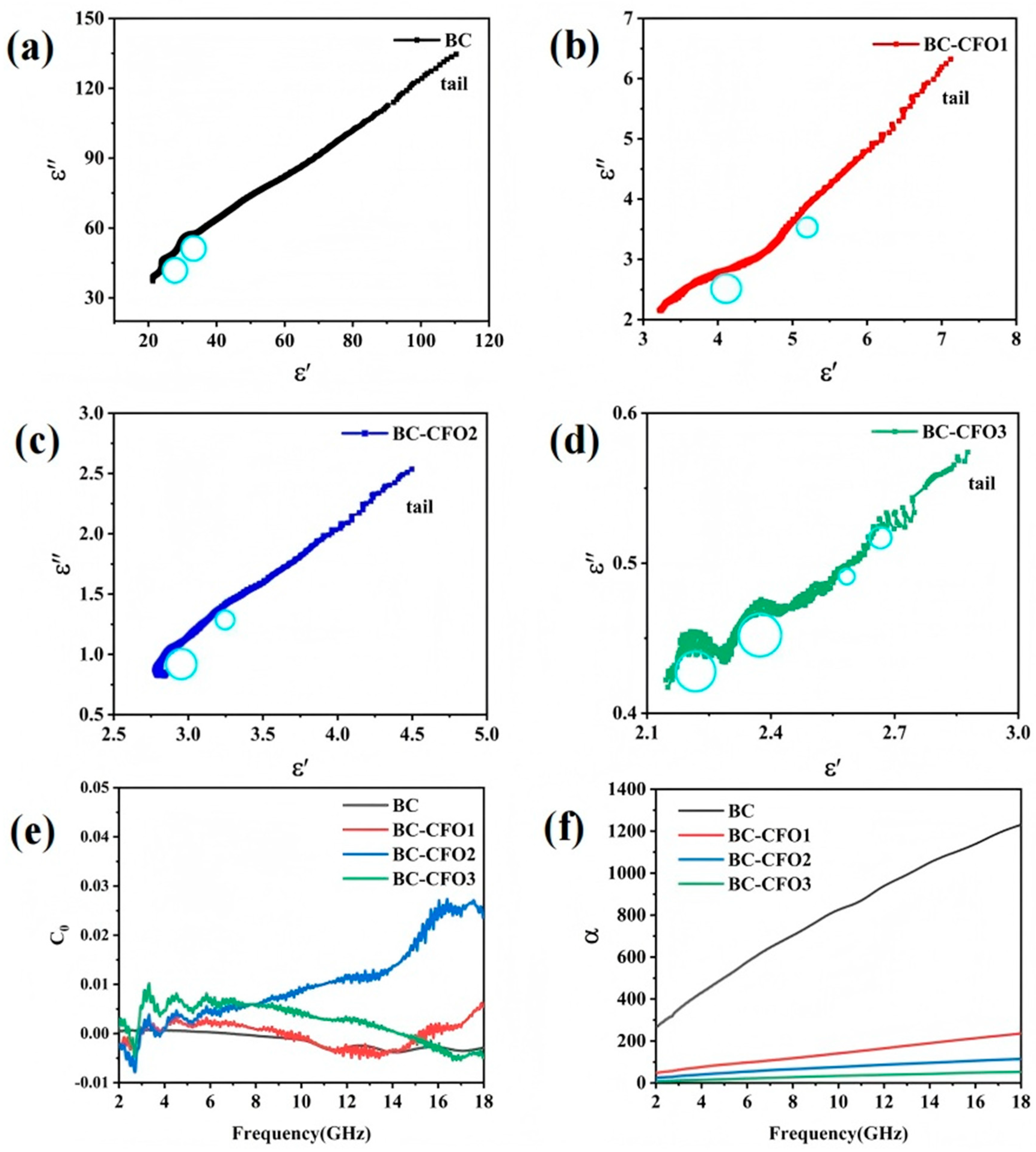
εs represents the static dielectric constant at infinite frequency, while ε∞ denotes the relative dielectric constant at infinite frequency. As illustrated in
Figure 7a–d, both BC and BC-CFO composites exhibit several semicircles with inclined tails. The presence of multiple semicircles in each sample, increasing in number, indicates the occurrence of Debye relaxation processes. The distorted semicircles suggest the presence of additional polarization mechanisms. This multi-relaxation behavior is closely related to the structure of CoFe
2O
4 particles growing on the porous BC surface. The porous structure of the composite magnetic metal particles leads to the generation of more space charge at the interfaces.
Additionally, during the composite interaction, multiple interfaces tend to form, ultimately resulting in interfacial polarization. Thus, space charge polarization and interfacial polarization are the primary contributors to dielectric loss. The polarization relaxation arises from dipole polarization caused by the inherent defects in the biomass carbon material, and from interfacial polarization between the biomass carbon and the magnetic CoFe2O4 particles. It is observed that the number of semicircles increases with the CoFe2O4 particle content, indicating that the addition of CoFe2O4 induces multiple relaxation processes and results in stronger polarization loss. The “tail” at the end of the Cole–Cole plot reflects the conductive loss due to the conductive network formed by the BC and BC-CFO composites, with the decreased conductivity of the composites leading to a reduced slope.
The primary forms of magnetic losses are exchange resonance, natural resonance, and eddy current losses. Among them, eddy current losses can be analyzed using the eddy current coefficient (C
0) as follows:
As shown in
Figure 7e, the C
0 curve of the BC-CFO composite exhibits fluctuations within the 2–10 GHz frequency range. Particularly in the 2–6 GHz range, the significant fluctuations in the C0 curve suggest the presence of exchange resonance in the magnetic loss mechanism of the BC-CFO composite. In the 10–14 GHz frequency range, the C0 values of BC-CFO1, BC-CFO2, and BC-CFO3 show pronounced fluctuations, indicating that natural resonance also contributes to the magnetic losses of the BC-CFO composite. Thus, the magnetic loss performance of the BC-CFO composite is a combined result of both natural resonance and exchange resonance mechanisms.
The attenuation constant (α) reflects the dissipation capability of an absorbing material, with larger α values indicating stronger dissipation, thereby confirming a greater absorption capacity of the material.
As shown in
Figure 7f, the α value of the BC and BC-CFO composites exhibits an increasing trend with rising frequency. Furthermore, the attenuation capability of BC-CFO composites decreases as the CoFe
2O
4 content increases. Benefiting from excellent conductive loss, BC has the highest α value, which significantly enhances its microwave absorption performance. For BC-CFO1 and BC-CFO2, while the incorporation of CoFe
2O
4 particles reduces the conductive loss and lowers the α value compared to BC, the presence of CoFe
2O
4 also provides unique magnetic loss characteristics. In contrast, the α value of BC-CFO3 drops sharply, negatively impacting its microwave absorption ability.
Impedance matching (Z) is another key parameter for evaluating electromagnetic wave absorption properties, as it plays a crucial role in determining the degree of wave attenuation in the material.
Figure 8 presents the contour plots of the impedance (Z) values for BC and BC-CFO composite materials at different thicknesses. When the impedance Z approaches 1, the material achieves better impedance matching, which allows more microwaves to enter and be attenuated. In this study, the 0.9 and 1.1 contours were selected to mark the Z values. As shown in
Figure 8a,b, the Z value of BC is around 0.2, indicating poor impedance matching. In contrast,
Figure 8c–h show that the BC-CFO composite materials, with CoFe
2O
4 particles introduced on the surface of BC, exhibit significantly higher Z values compared to BC alone. It is also observed that the Z value increases with the amount of CoFe
2O
4 particles introduced. The color band distribution in
Figure 8c,d reveals that BC-CFO1 composite material has a broad and distinct band region where the Z value fluctuates near 1 over a wide frequency range. This contributes to the excellent microwave absorption performance of BC-CFO1 across multiple frequency bands.
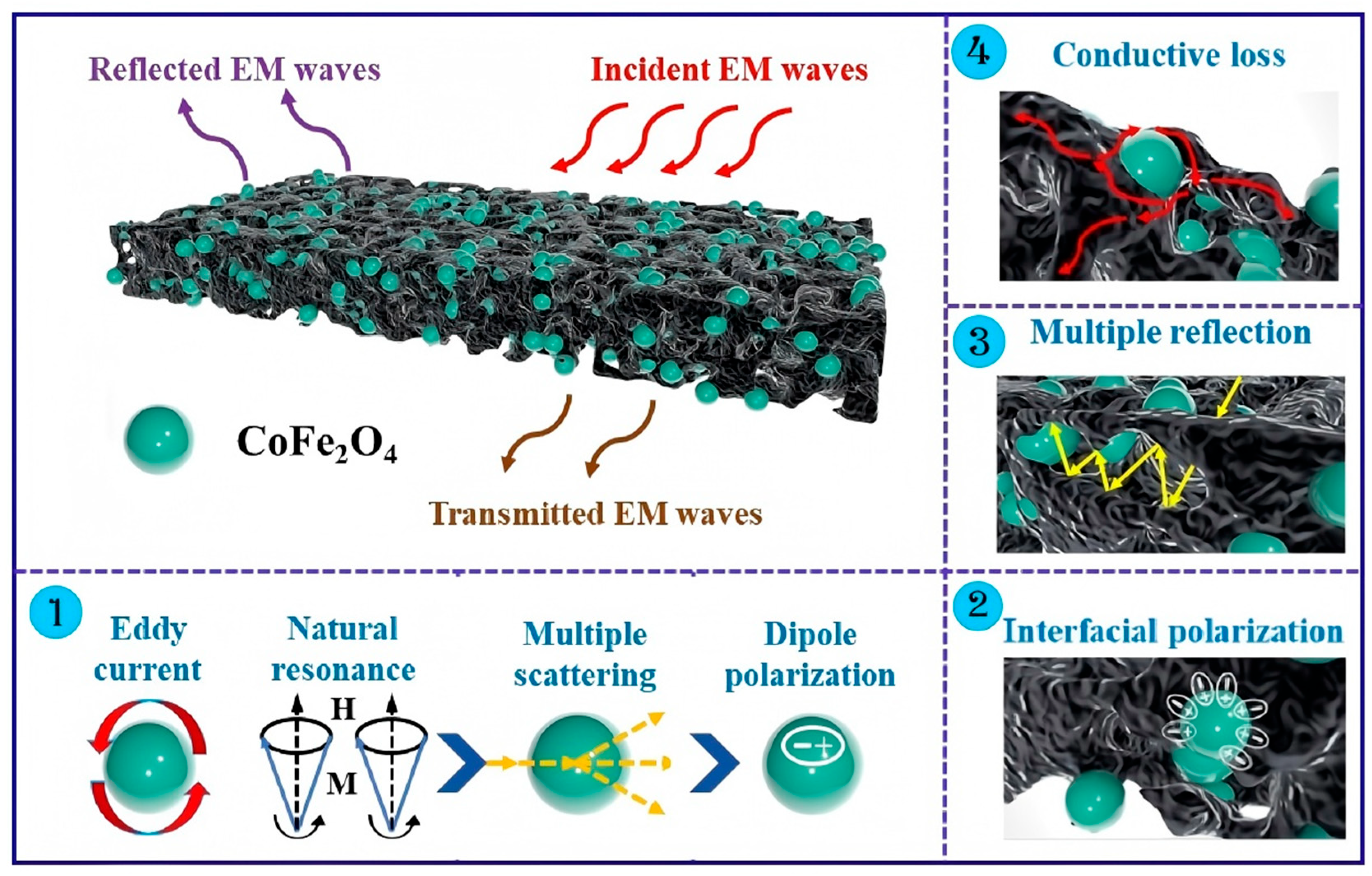
As shown in
Figure 9, the microwave absorption mechanism of the BC-CFO composite can be attributed to the following factors: (1) Introducing magnetic CoFe
2O
4 particles into the composite provides a magnetic loss mechanism. Additionally, CoFe
2O
4 particles effectively adjust the dielectric properties of the composite, enhancing dielectric loss and improving impedance matching. (2) The heterogeneous interfaces between the CoFe
2O
4 particles and BC create strong interfacial polarization, while surface functional groups and structural defects derived from BC act as polarization centers, leading to intense dipole polarization. (3) The porous structure of the BC-CFO composite facilitates multiple scattering and reflection of incident microwaves, extending the propagation path and dissipating more microwave energy. (4) The low conductivity of CoFe
2O
4 particles interrupts the conductive pathways, dissipating additional electromagnetic energy during transmission and promoting the conversion of electromagnetic energy into heat, thereby enhancing the microwave absorption performance of the material.