Olive Leaf Powder as a Potential Functional Component of Food Innovation: An In Vitro Study Evaluating Its Total Antioxidant Capacity and Phenolic Content
Abstract
1. Introduction
2. Materials and Methods
2.1. Sample Preparation
2.2. Solvent Preparation
2.3. Extraction Procedures
2.3.1. Continuous Stirring Extraction
2.3.2. Soaking Extraction
2.3.3. Ultrasound-Assisted Extraction
2.4. Sample Dilutions
2.5. Analytical Methods
2.5.1. Total Antioxidant Capacity (FRAP Assay)
2.5.2. Total Phenolic Content (Folin–Ciocalteu Assay)
2.6. Data Analysis
3. Results
3.1. Total Antioxidant Capacity (TAC)
3.1.1. Stirring Extraction
3.1.2. Soaking Extraction
3.1.3. Ultrasound-Assisted Extraction
3.2. Total Phenolic Content (TPC)
3.2.1. Stirring Extraction
3.2.2. Soaking Extraction
3.2.3. Ultrasound-Assisted Extraction
4. Discussion
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AU | Absorbance Units |
| EFSA | European Food Safety Authority |
| FDA | Food and Drug Administration |
| FRAP | Ferric Reducing Antioxidant Power |
| HPLC | High-Performance Liquid Chromatography |
| ICH | International Council for Harmonisation |
| TAC | Total Antioxidant Capacity |
| TPC | Total Phenolic Content |
| UAE | Ultrasound-Assisted Extraction |
References
- Siró, I.; Kápolna, E.; Kápolna, B.; Lugasi, A. Functional Food. Product Development, Marketing and Consumer Acceptance—A Review. Appetite 2008, 51, 456–467. [Google Scholar] [CrossRef] [PubMed]
- Daliri, E.B.-M.; Lee, B.H. Current Trends and Future Perspectives on Functional Foods and Nutraceuticals. In Beneficial Microorganisms in Food and Nutraceuticals; Liong, M.-T., Ed.; Springer International Publishing: Cham, Switzerland, 2015; pp. 221–244. ISBN 978-3-319-23177-8. [Google Scholar]
- Pistorio, E.; Chinnici, G.; Zarba, C.; Bellia, C.; Pappalardo, G. The Revolution of Functional Food: A Market Analysis (The Functional Foods Market Revolution: Market Analysis of Functional Food); Publisher STEF92 Technology: Vienna, Austria, 2023; pp. 433–441. [Google Scholar]
- Vicentini, A.; Liberatore, L.; Mastrocola, D. Functional foods: Trends and development of the global market. Ital. J. Food Sci. 2016, 28, 338–351. [Google Scholar] [CrossRef]
- Clodoveo, M.L.; Crupi, P.; Annunziato, A.; Corbo, F. Innovative Extraction Technologies for Development of Functional Ingredients Based on Polyphenols from Olive Leaves. Foods 2022, 11, 103. [Google Scholar] [CrossRef] [PubMed]
- Ronca, C.L.; Marques, S.S.; Ritieni, A.; Giménez-Martínez, R.; Barreiros, L.; Segundo, M.A. Olive Oil Waste as a Source of Functional Food Ingredients: Assessing Polyphenolic Content and Antioxidant Activity in Olive Leaves. Foods 2024, 13, 189. [Google Scholar] [CrossRef] [PubMed]
- Vogel, P.; Kasper Machado, I.; Garavaglia, J.; Zani, V.T.; de Souza, D.; Morelo Dal Bosco, S. Polyphenols Benefits of Olive Leaf (Olea europaea L.) to Human Health. Nutr. Hosp. 2014, 31, 1427–1433. [Google Scholar] [CrossRef] [PubMed]
- Erbay, Z.; Icier, F. The Importance and Potential Uses of Olive Leaves. Food Rev. Int. 2010, 26, 319–334. [Google Scholar] [CrossRef]
- Difonzo, G.; Squeo, G.; Pasqualone, A.; Summo, C.; Paradiso, V.M.; Caponio, F. The Challenge of Exploiting Polyphenols from Olive Leaves: Addition to Foods to Improve Their Shelf-Life and Nutritional Value. J. Sci. Food Agric. 2021, 101, 3099–3116. [Google Scholar] [CrossRef] [PubMed]
- Canada, H. 15. List of Permitted Solvents (Lists of Permitted Food Additives). Available online: https://www.canada.ca/en/health-canada/services/food-nutrition/food-safety/food-additives/lists-permitted/15-carrier-extraction-solvents.html (accessed on 6 July 2025).
- Generally Recognized as Safe (GRAS)|FDA. Available online: https://www.fda.gov/food/food-ingredients-packaging/generally-recognized-safe-gras (accessed on 6 July 2025).
- Sultana, B.; Anwar, F.; Ashraf, M. Effect of Extraction Solvent/Technique on the Antioxidant Activity of Selected Medicinal Plant Extracts. Molecules 2009, 14, 2167–2180. [Google Scholar] [CrossRef] [PubMed]
- Anwar, F.; Przybylski, R. Effect of Solvents Extraction on Total Phenolics and Antioxidant Activity of Extracts from Flaxseed (Linum usitatissimum L.). Acta Sci. Pol. Technol. Aliment. 2012, 11, 293–301. [Google Scholar] [PubMed]
- Extraction Solvents—European Commission. Available online: https://food.ec.europa.eu/food-safety/food-improvement-agents/extraction-solvents_en (accessed on 10 June 2025).
- Program, H.F. GRAS Notice Inventory. Available online: https://www.fda.gov/food/generally-recognized-safe-gras/gras-notice-inventory (accessed on 10 June 2025).
- Safe Food Processing: Common Standards for Extraction Solvents|EUR-Lex. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/?uri=legissum:sa0022 (accessed on 10 June 2025).
- Directive 2009/32/EC of the European Parliament and of the Council of 23 April 2009 on the Approximation of the Laws of the Member States on Extraction Solvents Used in the Production of Foodstuffs and Food Ingredients (Recast) (Text with EEA Relevance); European Union: Brussels, Belgium, 2009; Volume 141.
- ICH Official Web Site: ICH. Available online: https://www.ich.org/page/q3c-residual-solvents (accessed on 10 June 2025).
- Do, Q.D.; Angkawijaya, A.E.; Tran-Nguyen, P.L.; Huynh, L.H.; Soetaredjo, F.E.; Ismadji, S.; Ju, Y.-H. Effect of Extraction Solvent on Total Phenol Content, Total Flavonoid Content, and Antioxidant Activity of Limnophila aromatica. J. Food Drug Anal. 2014, 22, 296–302. [Google Scholar] [CrossRef] [PubMed]
- Ngo, T.V.; Scarlett, C.J.; Bowyer, M.C.; Ngo, P.D.; Vuong, Q.V. Impact of Different Extraction Solvents on Bioactive Compounds and Antioxidant Capacity from the Root of Salacia chinensis L. J. Food Qual. 2017, 2017, 9305047. [Google Scholar] [CrossRef]
- Nawaz, H.; Shad, M.A.; Rehman, N.; Andaleeb, H.; Ullah, N. Effect of Solvent Polarity on Extraction Yield and Antioxidant Properties of Phytochemicals from Bean (Phaseolus vulgaris) Seeds. Braz. J. Pharm. Sci. 2020, 56, e17129. [Google Scholar] [CrossRef]
- Luthria, D.L.; Mukhopadhyay, S.; Kwansa, A.L. A Systematic Approach for Extraction of Phenolic Compounds Using Parsley (Petroselinum crispum) Flakes as a Model Substrate. J. Sci. Food Agric. 2006, 86, 1350–1358. [Google Scholar] [CrossRef]
- Kaczorová, D.; Karalija, E.; Dahija, S.; Bešta-Gajević, R.; Parić, A.; Ćavar Zeljković, S. Influence of Extraction Solvent on the Phenolic Profile and Bioactivity of Two Achillea Species. Molecules 2021, 26, 1601. [Google Scholar] [CrossRef] [PubMed]
- Um, M.; Han, T.-H.; Lee, J.-W. Ultrasound-Assisted Extraction and Antioxidant Activity of Phenolic and Flavonoid Compounds and Ascorbic Acid from Rugosa Rose (Rosa rugosa Thunb.) Fruit. Food Sci. Biotechnol. 2017, 27, 375–382. [Google Scholar] [CrossRef] [PubMed]
- Kobus, Z.; Krzywicka, M.; Starek-Wójcicka, A.; Sagan, A. Effect of the Duty Cycle of the Ultrasonic Processor on the Efficiency of Extraction of Phenolic Compounds from Sorbus Intermedia. Sci. Rep. 2022, 12, 8311. [Google Scholar] [CrossRef] [PubMed]
- Cacique, A.P.; Barbosa, É.S.; de Pinho, G.P.; Silvério, F.O. Maceration Extraction Conditions for Determining the Phenolic Compounds and the Antioxidant Activity of Catharanthus roseus (L.) G. Don. Ciênc. Agrotec. 2020, 44, e017420. [Google Scholar] [CrossRef]
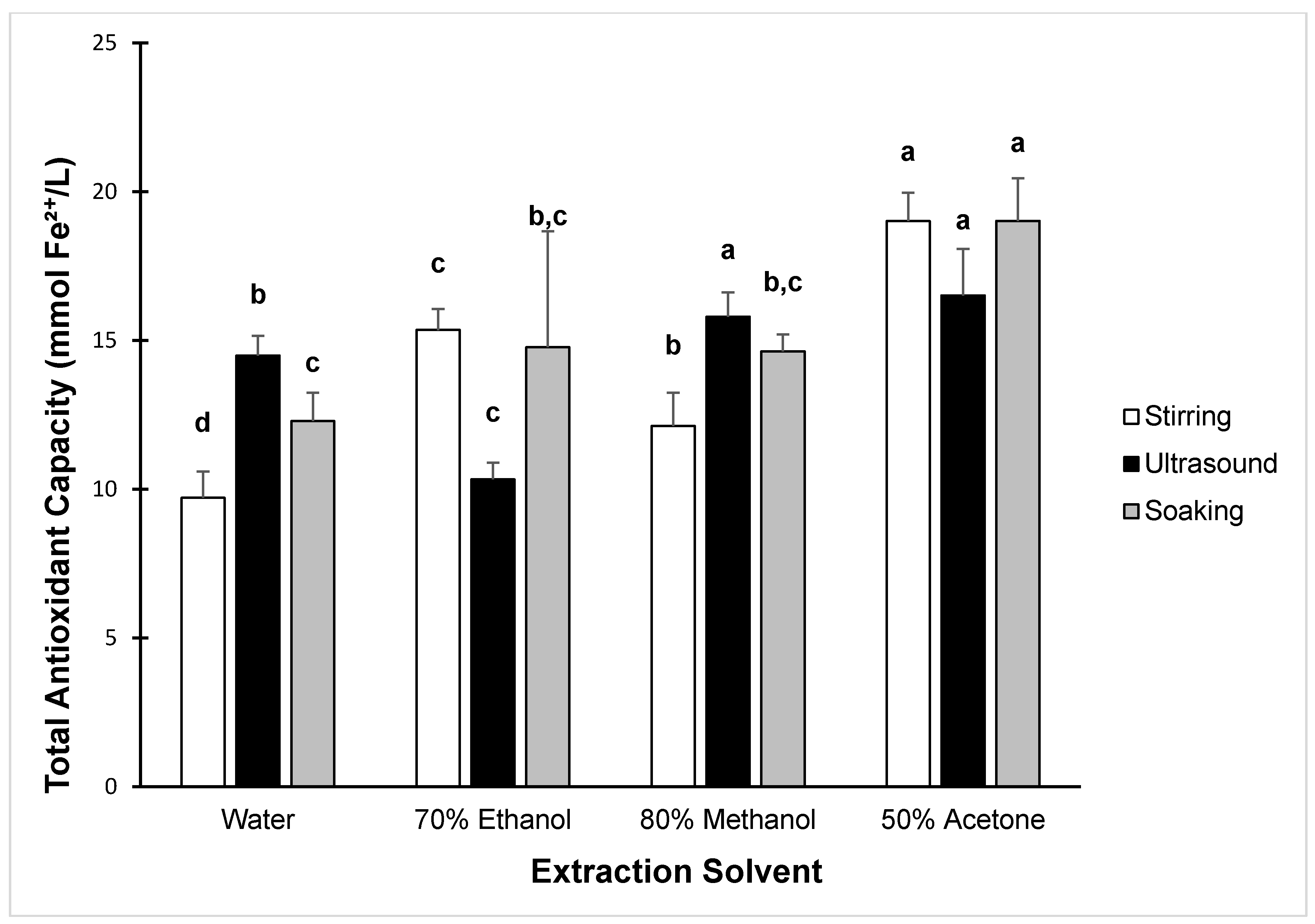

| Solvents | ||||
|---|---|---|---|---|
| Extraction Method | Water | 80% Methanol | 70% Ethanol | 50% Acetone |
| Stirring | 9.71 ± 0.88 d | 12.12 ± 1.12 c | 15.35 ± 0.71 b | 19.01 ± 0.96 a |
| Soaking | 12.29 ± 0.95 c | 14.63 ± 0.57 b,c | 14.77 ± 3.90 b,c | 19.02 ± 1.43 a |
| Ultrasound | 14.49 ± 0.67 b | 15.80 ± 0.82 a | 10.33 ± 0.56 c | 16.51 ± 1.57 a |
| Solvents | ||||
|---|---|---|---|---|
| Extraction Method | Water | 80% Methanol | 70% Ethanol | 50% Acetone |
| Stirring | 616.19 ± 113.60 c | 701.56 ± 127.60 c | 815.72 ± 119.97 b | 1152.70 ± 164.60 a |
| Soaking | 665.54 ± 67.78 c | 762.93 ± 92.38 b | 1100.07 ± 151.10 a | 1193.63 ± 145.34 a |
| Ultrasound | 794.92 ± 90.33 b | 914.08 ± 147.38 b | 1214.76 ± 190.74 a | 1289.95 ± 191.17 a |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Almpounioti, K.; Papagianni, O.; Michaloudi, P.; Tsermoula, S.K.; Potsaki, P.; Dimou, C.; Koutelidakis, A.E. Olive Leaf Powder as a Potential Functional Component of Food Innovation: An In Vitro Study Evaluating Its Total Antioxidant Capacity and Phenolic Content. Appl. Sci. 2025, 15, 9462. https://doi.org/10.3390/app15179462
Almpounioti K, Papagianni O, Michaloudi P, Tsermoula SK, Potsaki P, Dimou C, Koutelidakis AE. Olive Leaf Powder as a Potential Functional Component of Food Innovation: An In Vitro Study Evaluating Its Total Antioxidant Capacity and Phenolic Content. Applied Sciences. 2025; 15(17):9462. https://doi.org/10.3390/app15179462
Chicago/Turabian StyleAlmpounioti, Kalliopi, Olga Papagianni, Panagiota Michaloudi, Sofia Konstantina Tsermoula, Panagiota Potsaki, Charalampia Dimou, and Antonios E. Koutelidakis. 2025. "Olive Leaf Powder as a Potential Functional Component of Food Innovation: An In Vitro Study Evaluating Its Total Antioxidant Capacity and Phenolic Content" Applied Sciences 15, no. 17: 9462. https://doi.org/10.3390/app15179462
APA StyleAlmpounioti, K., Papagianni, O., Michaloudi, P., Tsermoula, S. K., Potsaki, P., Dimou, C., & Koutelidakis, A. E. (2025). Olive Leaf Powder as a Potential Functional Component of Food Innovation: An In Vitro Study Evaluating Its Total Antioxidant Capacity and Phenolic Content. Applied Sciences, 15(17), 9462. https://doi.org/10.3390/app15179462








