Potential Applications of Yeast Biomass Derived from Small-Scale Breweries
Abstract
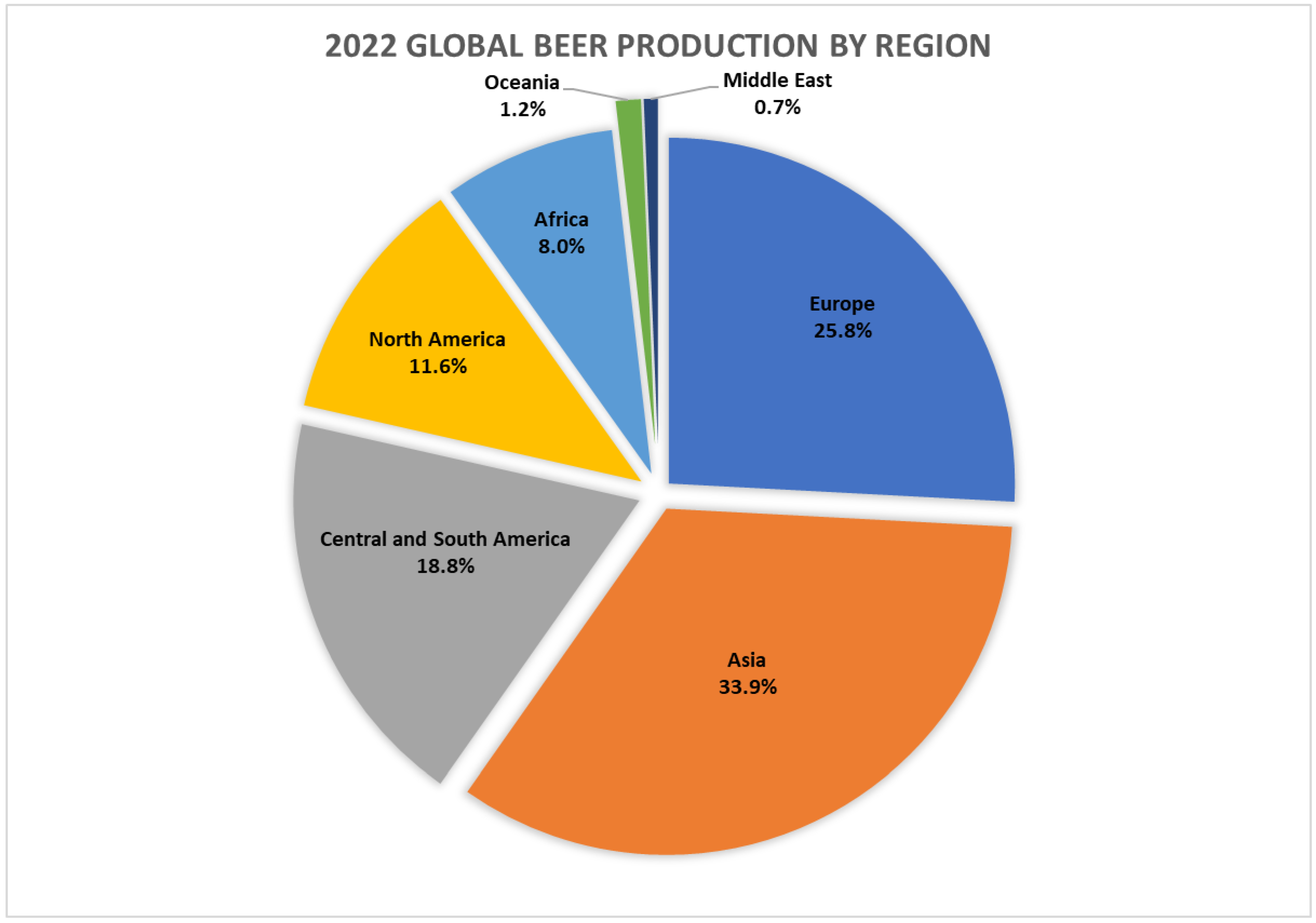
1. Beer Production Statistics
2. Brewery Industry Waste and By-Products
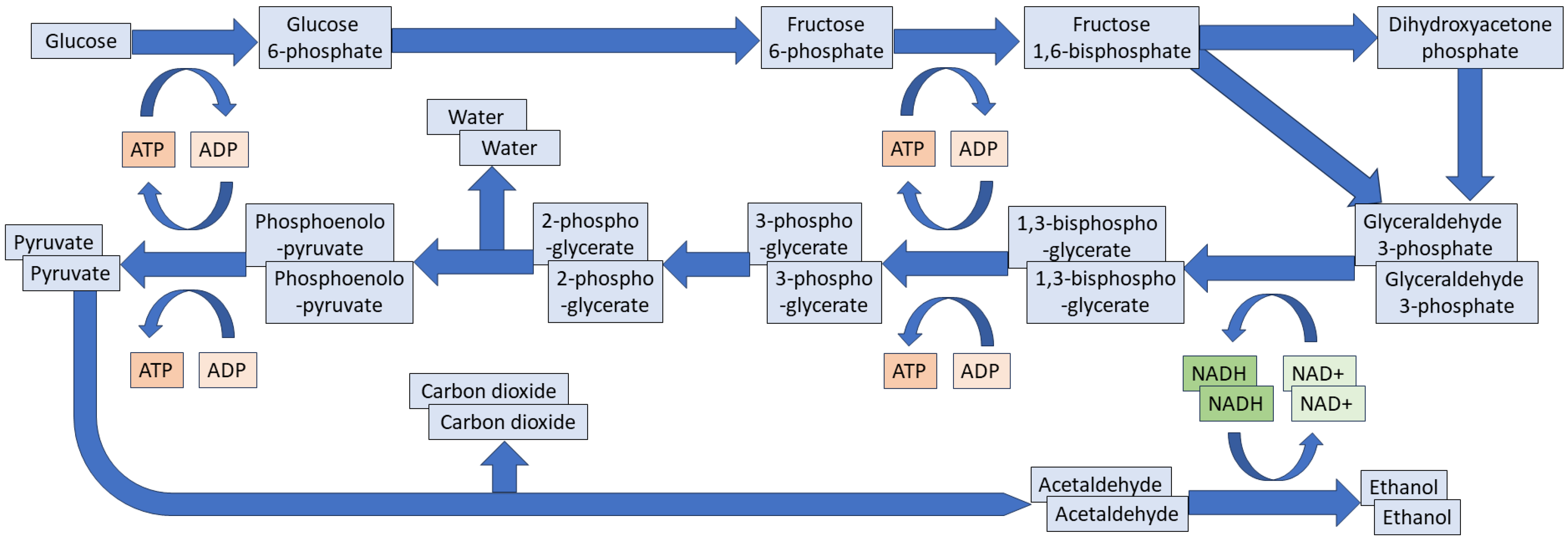
3. Fermentative Yeast Growth
4. Brewery Yeast Biomass
5. Yeast Biomass Processing Technologies
6. Spent Yeast Derivates—Potential Applications
7. Yeast Cell Walls and Their Derivates
8. Other Potential Applications
9. Conclusions and Perspectives
- -
- Beta-glucan fractions (for food fortification or diet supplementation);
- -
- Mannan fractions (suitable for animal fodder supplementation);
- -
- Food-grade protein concentrates (with a reduced content of nucleotides);
- -
- Low-mass metabolite fractions enriched with nucleotide salts derived from nucleic acid hydrolysis (suitable as a flavor enhancer).
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Meier, H. BarthHaas Report 2022/2023; BarthHaas: Nuremberg, Germany, 2023. [Google Scholar]
- Durán-Sánchez, A.; de la Cruz del Río-Rama, M.; Álvarez-García, J.; Oliveira, C. Analysis of Worldwide Research on Craft Beer. Sage Open 2022, 12, 21582440221108154. [Google Scholar] [CrossRef]
- Nilsson, I.; Reid, N. The Value of a Craft Brewery: On the Relationship between Craft Breweries and Property Values. Growth Chang. 2019, 50, 689–704. [Google Scholar] [CrossRef]
- The Union of Brewing Industry Employers Deloitte. Podsumowanie Analizy Wybranych Wskaźników Wpływu Przemysłu Piwowarskiego Na Polską Gospodarkę i Otoczenie; The Union of Brewing Industry Employers Deloitte: Waszawa, Poland, 2018. [Google Scholar]
- The Union of Brewing Industry Employers Deloitte. Podsumowanie Analizy Wybranych Wskaźników Wpływu Przemysłu Piwowarskiego Na Polską Gospodarkę i Otoczenie; The Union of Brewing Industry Employers Deloitte: Warszawa, Poland, 2021. [Google Scholar]
- Eßlinger, H.M. (Ed.) Handbook of Brewing: Processes, Technology, Markets; John Wiley & Sons: New York, NY, USA, 2009; ISBN 978-3-527-31674-8. [Google Scholar]
- European Environment Agency ETC/WMGE. Early Warning Assessment Related to the 2025 Targets for Municipal Waste and Packaging Waste—Poland; European Environment Agency ETC/WMGE: Copenhagen, Denmark, 2023. [Google Scholar]
- Warringa, G. Waste Incineration under the EU ETS—An Assessment of Climate Benefits; CE Delft: Delft, The Netherlands, 2021. [Google Scholar]
- dos Santos Mathias, T.R.; de Mello, P.M.; Eliana, F.C.S. Solid Wastes in Brewing Process: A Review. J. Brew. Distill. 2014, 5, 1–9. [Google Scholar] [CrossRef]
- Werkneh, A.A.; Beyene, H.D.; Osunkunle, A.A. Recent Advances in Brewery Wastewater Treatment; Approaches for Water Reuse and Energy Recovery: A Review. Environ. Sustain. 2019, 2, 199–209. [Google Scholar] [CrossRef]
- Lange, H.C.; Heijnen, J.J. Statistical Reconciliation of the Elemental and Molecular Biomass Composition of Saccharomyces cerevisiae. Biotechnol. Bioeng. 2001, 75, 334–344. [Google Scholar] [CrossRef] [PubMed]
- Nand, K. Debittering of Spent Brewer’s Yeast for Food Purposes. Food/Nahr. 1987, 31, 127–131. [Google Scholar] [CrossRef]
- Olajire, A.A. The Brewing Industry and Environmental Challenges. J. Clean. Prod. 2020, 256, 102817. [Google Scholar] [CrossRef]
- Rüffer, H.; Rosenwinkel, K.-H. Taschenbuch der Industrieabwasserreinigung; Vulkan-Verlag GmbH: Oldenburg, Germany, 1991; ISBN 3-486-26131-2. [Google Scholar]
- The Brewers of Europe. Guidance Note for Establishing BAT in the Brewing Industry; CMBC: Wolverhampton, UK, 2002. [Google Scholar]
- Ścisłowska, M.; Wolny, L. Charakterystyka Wybranych Gminnych Oczyszczalni Ścieków. Eng. Prot. Environ. 2010, 13, 133–146. [Google Scholar]
- Kaczor, G. Stężenia Zanieczyszczeń w Ściekach Odprowadzanych z Wiejskich Systemów Kanalizacyjnych Województwa Małopolskiego. Infrastrukt. Ekol. Teren. Wiej. 2009, 6, 97–104. [Google Scholar]
- Wartości Parametrów Ścieków Surowych Dopływających Do Oczyszczalni Wartości Ścieków Oczyszczonych w Odniesieniu Uzyskanego Pozwolenia Wodnoprawnego. Available online: https://mpwik-zywiec.pl/uploaded/parametry%20%20%C5%9Bciek%C3%B3w.pdf (accessed on 26 February 2024).
- EU. EU Council Derective of 21 May 1991 Concerning Urban Waste Water Treatment; EU: Brussels, Belgium, 1991. [Google Scholar]
- van Dijken, J.P.; Weusthuis, R.A.; Pronk, J.T. Kinetics of Growth and Sugar Consumption in Yeasts. Antonie Leeuwenhoek 1993, 63, 343–352. [Google Scholar] [CrossRef] [PubMed]
- Jones, M.; Pierce, J.S. Absorption of Amino Acids from Wort by Yeasts. J. Inst. Brew. 1964, 70, 307–315. [Google Scholar] [CrossRef]
- Burkholder, P.R. Vitamin Deficiencies in Yeasts. Am. J. Bot. 1943, 30, 206. [Google Scholar] [CrossRef]
- Wu, H.; Ito, K.; Shimoi, H. Identification and Characterization of a Novel Biotin Biosynthesis Gene in Saccharomyces cerevisiae. Appl. Environ. Microbiol. 2005, 71, 6845–6855. [Google Scholar] [CrossRef] [PubMed]
- Stewart, G.G.; Russell, I.; Anstruther, A. (Eds.) Handbook of Brewing; CRC Press: Boca Raton, FL, USA, 2017; ISBN 9781351228336. [Google Scholar]
- Nyhan, L.; Sahin, A.W.; Schmitz, H.H.; Siegel, J.B.; Arendt, E.K. Brewers’ Spent Grain: An Unprecedented Opportunity to Develop Sustainable Plant-Based Nutrition Ingredients Addressing Global Malnutrition Challenges. J. Agric. Food Chem. 2023, 71, 10543–10564. [Google Scholar] [CrossRef] [PubMed]
- Kunze, W. Technology Brewing & Malting; Hendel, O., Ed.; Versuchs- u. Lehranstalt f. Brauerei: Berlin/Heidelberg, Germany, 2019; ISBN 978-3-921690-87-1. [Google Scholar]
- Buiatti, S. Beer Composition: An Overview. In Beer in Health and Disease Prevention; Elsevier: Amsterdam, The Netherlands, 2009; pp. 213–225. [Google Scholar]
- Shotipruk, A.; Kittianong, P.; Suphantharika, M.; Muangnapoh, C. Application of Rotary Microfiltration in Debittering Process of Spent Brewer’s Yeast. Bioresour. Technol. 2005, 96, 1851–1859. [Google Scholar] [CrossRef]
- Marson, G.V.; de Castro, R.J.S.; Belleville, M.-P.; Hubinger, M.D. Spent Brewer’s Yeast as a Source of High Added Value Molecules: A Systematic Review on Its Characteristics, Processing and Potential Applications. World J. Microbiol. Biotechnol. 2020, 36, 95. [Google Scholar] [CrossRef]
- Jacob, F.F.; Striegel, L.; Rychlik, M.; Hutzler, M.; Methner, F.-J. Yeast Extract Production Using Spent Yeast from Beer Manufacture: Influence of Industrially Applicable Disruption Methods on Selected Substance Groups with Biotechnological Relevance. Eur. Food Res. Technol. 2019, 245, 1169–1182. [Google Scholar] [CrossRef]
- Pacheco, M.T.B.; Caballero-Córdoba, G.M.; Sgarbieri, V.C. Composition and Nutritive Value of Yeast Biomass and Yeast Protein Concentrates. J. Nutr. Sci. Vitaminol. 1997, 43, 601–612. [Google Scholar] [CrossRef]
- Podpora, B.; Świderski, F.; Sadowska, A.; Rakowska, R.; Wasiak-Zys, G. Spent Brewer’s Yeast Extracts as a New Component of Functional Food. Czech J. Food Sci. 2016, 34, 554–563. [Google Scholar] [CrossRef]
- World Health Organization; United Nations University. Protein and Amino Acid Requirements in Human Nutrition; World Health Organization: Geneve, Switzerland, 2007; Volume 935, ISBN 9241209356. [Google Scholar]
- Stewart, G.G. The Structure and Function of the Yeast Cell Wall, Plasma Membrane and Periplasm. In Brewing and Distilling Yeasts; Springer International Publishing: Cham, Switzerland, 2017; pp. 55–75. [Google Scholar]
- Aguilar-Uscanga, B.; Francois, J.M. A Study of the Yeast Cell Wall Composition and Structure in Response to Growth Conditions and Mode of Cultivation. Lett. Appl. Microbiol. 2003, 37, 268–274. [Google Scholar] [CrossRef]
- Stokes, J.L.; Gunness, M.; Foster, J.W. Vitamin Content of Ingredients of Microbiological Culture Media. J. Bacteriol. 1944, 47, 293–299. [Google Scholar] [CrossRef]
- McCaig, R.; Bendiak, D.S. Yeast Handling Studies. II. Temperature of Storage of Pitching Yeast. J. Am. Soc. Brew. Chem. 1985, 43, 119–122. [Google Scholar] [CrossRef]
- Reed, G.; Nagodawithana, T.W. Yeast Technology; Springer Netherlands: Dordrecht, The Netherlands, 1990; ISBN 978-94-011-9773-1. [Google Scholar]
- Mosser, M.; Kapel, R.; Chevalot, I.; Olmos, E.; Marc, I.; Marc, A.; Oriol, E. Fractionation of Yeast Extract by Nanofiltration Process to Assess Key Compounds Involved in CHO Cell Culture Improvement. Biotechnol. Prog. 2015, 31, 875–882. [Google Scholar] [CrossRef]
- Jacob, F.F.; Hutzler, M.; Methner, F.-J. Comparison of Various Industrially Applicable Disruption Methods to Produce Yeast Extract Using Spent Yeast from Top-Fermenting Beer Production: Influence on Amino Acid and Protein Content. Eur. Food Res. Technol. 2019, 245, 95–109. [Google Scholar] [CrossRef]
- Jacob, F.F.; Michel, M.; Zarnkow, M.; Hutzler, M.; Methner, F.-J. The Complexity of Yeast Extracts and Its Consequences on the Utility in Brewing: A Review. Brew. Sci. 2019, 72, 50–62. [Google Scholar] [CrossRef]
- Takalloo, Z.; Nikkhah, M.; Nemati, R.; Jalilian, N.; Sajedi, R.H. Autolysis, Plasmolysis and Enzymatic Hydrolysis of Baker’s Yeast (Saccharomyces cerevisiae): A Comparative Study. World J. Microbiol. Biotechnol. 2020, 36, 68. [Google Scholar] [CrossRef] [PubMed]
- de la Hoz, L.; Ponezi, A.N.; Milani, R.F.; Nunes da Silva, V.S.; Sonia de Souza, A.; Bertoldo-Pacheco, M.T. Iron-Binding Properties of Sugar Cane Yeast Peptides. Food Chem. 2014, 142, 166–169. [Google Scholar] [CrossRef] [PubMed]
- Hassid, W.Z.; Joslyn, M.A.; McCready, R.M. The Molecular Constitution of an Insoluble Polysaccharide from Yeast, Saccharomyces cerevisiae. J. Am. Chem. Soc. 1941, 63, 295–298. [Google Scholar] [CrossRef]
- Shokri, H.; Asadi, F.; Khosravi, A.R. Isolation of β-Glucan from the Cell Wall of Saccharomyces cerevisiae. Nat. Prod. Res. 2008, 22, 414–421. [Google Scholar] [CrossRef] [PubMed]
- Freimund, S.; Sauter, M.; Käppeli, O.; Dutler, H. A New Non-Degrading Isolation Process for 1,3-β-d-Glucan of High Purity from Baker’s Yeast Saccharomyces cerevisiae. Carbohydr. Polym. 2003, 54, 159–171. [Google Scholar] [CrossRef]
- Li, J.; Karboune, S. A Comparative Study for the Isolation and Characterization of Mannoproteins from Saccharomyces cerevisiae yeast Cell Wall. Int. J. Biol. Macromol. 2018, 119, 654–661. [Google Scholar] [CrossRef]
- Jach, M.E.; Serefko, A.; Ziaja, M.; Kieliszek, M. Yeast Protein as an Easily Accessible Food Source. Metabolites 2022, 12, 63. [Google Scholar] [CrossRef]
- Diaz-Bustamante, M.L.; Keppler, J.K.; Reyes, L.H.; Alvarez Solano, O.A. Trends and Prospects in Dairy Protein Replacement in Yogurt and Cheese. Heliyon 2023, 9, e16974. [Google Scholar] [CrossRef] [PubMed]
- Patterson, R.; Rogiewicz, A.; Kiarie, E.G.; Slominski, B.A. Yeast Derivatives as a Source of Bioactive Components in Animal Nutrition: A Brief Review. Front. Vet. Sci. 2023, 9, 1067383. [Google Scholar] [CrossRef] [PubMed]
- Mussatto, S.I. Biotechnological Potential of Brewing Industry By-Products. In Biotechnology for Agro-Industrial Residues Utilisation; Springer Netherlands: Dordrecht, The Netherlands, 2009; pp. 313–326. [Google Scholar]
- Sacakli, P.; Koksal, B.H.; Ergun, A.; Özsoy, B. Usage of Brewer’s Yeast (Saccharomyces cerevisiae) as a Replacement of Vitamin Andtrace Mineral Premix in Broiler Diets. Rev. Médecine Vétérinaire 2013, 164, 39–44. [Google Scholar]
- Tao, Z.; Yuan, H.; Liu, M.; Liu, Q.; Zhang, S.; Liu, H.; Jiang, Y.; Huang, D.; Wang, T. Yeast Extract: Characteristics, Production, Applications and Future Perspectives. J. Microbiol. Biotechnol. 2023, 33, 151–166. [Google Scholar] [CrossRef]
- Vieira, E.F.; Carvalho, J.; Pinto, E.; Cunha, S.; Almeida, A.A.; Ferreira, I.M.P.L.V.O. Nutritive Value, Antioxidant Activity and Phenolic Compounds Profile of Brewer’s Spent Yeast Extract. J. Food Compos. Anal. 2016, 52, 44–51. [Google Scholar] [CrossRef]
- Regulation (EU) No 1169/2011; Provision of Food Information to Costumers. European Parliament and Council: Strasbourg, France, 2011.
- Di Carlo, F.J.; Schultz, A.S.; Kent, A.M. Soybean Nucleic Acid. Arch. Biochem. Biophys. 1955, 55, 253–256. [Google Scholar] [CrossRef]
- Arasu, P.; Field, R.A.; Kruggel, W.G.; Miller, G.J. Nucleic Acid Content of Bovine Bone Marrow, Muscle and Mechanically Deboned Beef. J. Food Sci. 1981, 46, 1114–1116. [Google Scholar] [CrossRef]
- Jaeger, A.; Arendt, E.K.; Zannini, E.; Sahin, A.W. Brewer’s Spent Yeast (BSY), an Underutilized Brewing By-Product. Fermentation 2020, 6, 123. [Google Scholar] [CrossRef]
- Thammakiti, S.; Suphantharika, M.; Phaesuwan, T.; Verduyn, C. Preparation of Spent Brewer’s Yeast Β-glucans for Potential Applications in the Food Industry. Int. J. Food Sci. Technol. 2004, 39, 21–29. [Google Scholar] [CrossRef]
- Martins, Z.E.; Pinho, O.; Ferreira, I.M.P.L.V.O. Impact of New Ingredients Obtained from Brewer’s Spent Yeast on Bread Characteristics. J. Food Sci. Technol. 2018, 55, 1966–1971. [Google Scholar] [CrossRef] [PubMed]
- Worrasinchai, S.; Suphantharika, M.; Pinjai, S.; Jamnong, P. β-Glucan Prepared from Spent Brewer’s Yeast as a Fat Replacer in Mayonnaise. Food Hydrocoll. 2006, 20, 68–78. [Google Scholar] [CrossRef]
- da Silva Araújo, V.B.; de Melo, A.N.F.; Costa, A.G.; Castro-Gomez, R.H.; Madruga, M.S.; de Souza, E.L.; Magnani, M. Followed Extraction of β-Glucan and Mannoprotein from Spent Brewer’s Yeast (Saccharomyces Uvarum) and Application of the Obtained Mannoprotein as a Stabilizer in Mayonnaise. Innov. Food Sci. Emerg. Technol. 2014, 23, 164–170. [Google Scholar] [CrossRef]
- Amorim, M.; Marques, C.; Pereira, J.O.; Guardão, L.; Martins, M.J.; Osório, H.; Moura, D.; Calhau, C.; Pinheiro, H.; Pintado, M. Antihypertensive Effect of Spent Brewer Yeast Peptide. Process Biochem. 2019, 76, 213–218. [Google Scholar] [CrossRef]
- Guo, H.; Guo, S.; Liu, H. Antioxidant Activity and Inhibition of Ultraviolet Radiation-Induced Skin Damage of Selenium-Rich Peptide Fraction from Selenium-Rich Yeast Protein Hydrolysate. Bioorg. Chem. 2020, 105, 104431. [Google Scholar] [CrossRef]
- Amorim, M.M.; Pereira, J.O.; Monteiro, K.M.; Ruiz, A.L.; Carvalho, J.E.; Pinheiro, H.; Pintado, M. Antiulcer and Antiproliferative Properties of Spent Brewer’s Yeast Peptide Extracts for Incorporation into Foods. Food Funct. 2016, 7, 2331–2337. [Google Scholar] [CrossRef]
- Andreassen, R.C.; Pedersen, M.E.; Kristoffersen, K.A.; Beate Rønning, S. Screening of By-Products from the Food Industry as Growth Promoting Agents in Serum-Free Media for Skeletal Muscle Cell Culture. Food Funct. 2020, 11, 2477–2488. [Google Scholar] [CrossRef]
- Oliveira, A.S.; Ferreira, C.; Pereira, J.O.; Pintado, M.E.; Carvalho, A.P. Spent Brewer’s Yeast (Saccharomyces cerevisiae) as a Potential Source of Bioactive Peptides: An Overview. Int. J. Biol. Macromol. 2022, 208, 1116–1126. [Google Scholar] [CrossRef]
| Parameter | Generated in Brewery | Accepted by Treatment Plant | EUNorm for Purified Wastewater |
|---|---|---|---|
| BOD 5 [mg/L O2] | 1000–50,000 | 100–940 | 25 |
| COD [mg/L O2] | 800–38,000 | 109–1668 | 125 |
| TSS [mg/L] | 200–3000 | 78–1260 | 35 |
| Total P [mg/L] | 4–100 | 2–40 | 1–2 |
| Total N [mg/L] | 20–100 | 20–210 | 10–15 |
| Wastewater-to-beer ratio [hL/hL] | 2.2–8.7 | - | - |
| References | [13,14,15] | [16,17,18] | [19] |
| Yeast Biomass | YE, Cell Mill | YE, Sonication | YE, Autolysis | |
|---|---|---|---|---|
| Proteins/peptides | 41–49 | 32.2 | 28.5 | 5.3 |
| Free amino acids | 0.2–0.4 | 11.6 | 15.5 | 30.6 |
| Ribonucleic acids | 1.9–7.5 | 7.5 | 7 | 5 |
| Total sugars | 22–54 | 31.2 | 31.4 | 28.8 |
| Lipids | 3.9 | 1 | 1.1 | 0.5 |
| Ashes | 1.7–8.5 | 13.1 | 13.3 | 13.2 |
| Reference | [29] | [30] | [30] | [30] |
| Amino Acid | Yeast Biomass | Yeast Extract (Total) | Yeast Extract (Free Amino Acids) | WHO Amino Acid Requirements |
|---|---|---|---|---|
| Lysine | 7.13 | 7.5 | 3.2 | 4.5 |
| Histidine | 2.06 | 2.4 | 1.4 | 1.5 |
| Threonine | 6.16 | 4.6 | 2.7 | 2.3 |
| Valine | 6.2 | 6.2 | 4.0 | 3.9 |
| Leucine | 8.84 | 7.2 | 5.7 | 5.9 |
| Isoleucine | 5.64 | 5.0 | 3.4 | 3 |
| Phenylalanine | 5.3 | 4.6 | 3.5 | 3.8 |
| Tyrosine | 4.68 | 3.5 | 2.4 | |
| Tryptophan | 1.1 | 1.3 | 1.0 | 0.6 |
| Methionine | 2.5 | 1.6 | 1.2 | 1.6 |
| Cystine | 0.34 | 1.1 | 0.5 | 0.6 |
| Glutamic acid + glutamine | 13.15 | 15.8 | 5.2 | |
| Aspartic acid + aspartate | 11.98 | 10.5 | 4.0 | |
| Serine | 6.13 | 5.1 | 3.5 | |
| Proline | 4.45 | 5.8 | 3.3 | |
| Alanine | 7.07 | 7.6 | 6.0 | |
| Glycine | 4.93 | 5.2 | 2.2 | |
| Arginine | 4.11 | 5.1 | 3.0 | |
| Source material type | Brewery yeast biomass | Yeast extract obtained from brewery yeast via lysis with papain and debittering | ||
| Reference | [31] | [32] | [33] | |
| Cell wall | 11–25 | % cell DW |
| Chitin | 1.4–6.9 | % cell wall |
| Mannan | 28–67 | % cell wall |
| Total beta-glucan | 32–57 | % cell wall |
| 1–6 beta-glucan | 4.5–11.5 | % cell wall |
| Vitamin | Yeast Biomass | Yeast Extracts | ||||
|---|---|---|---|---|---|---|
| Biotin (B7) | 0.0013 | 0.5 | 1.14 | 1.28 | 1.38 | 6.19 |
| Folic acid (B9) | 0.0130 | 0.01 | 0.045 | 0.049 | 0.013 | 0.053 |
| Niacin (B3) | 0.3000 | nd | 0.94 | 1.04 | 0.68 | 0.79 |
| Pantothenic acid (B5) | 0.0700 | 0.08 | 0.2 | 0.19 | 0.16 | 0.43 |
| Riboflavin (B2) | 0.0400 | 0.13 | 0.002 | 0.002 | 0.001 | 0.011 |
| Thiamine (B1) | 0.1200 | 0.11 | 0.069 | 0.07 | 0.052 | 0.075 |
| Pyridoxine (B6) | 0.0280 | 1.76 | 0.049 | 0.051 | 0.031 | 0.059 |
| Cobalamin (B12) | 0.000001 | 0.17 | 0.0018 | 0.0012 | 0.011 | 0.0016 |
| Source material type | Brewery yeast biomass | Dedicated yeast culture, autolyzed, supplied by Bio Springer AM Corporation | Brewery yeast biomass, cell mill | Brewery yeast biomass, sonication | Brewery yeast biomass, autolysis | Saccharomyces sp. biomass, industrial autolysis |
| Reference | [38] | [39] | [30] | [30] | [30] | [30] |
| Product | Application | Advantages | Reference |
|---|---|---|---|
| Food and fodder additives | |||
| Whole yeast cell | Supplementation of broiler diet (1–5%) | Enhancement of bone health | [52] |
| Purified beta-glucan | Addition of beta-glucan to flour in bread production | Improved texture of bread | [60] |
| Purified beta-glucan | Low-fat mayonnaise | Reduced fat content, with acceptable changes in taste | [61] |
| Purified mannoprotein | Emulsifier in mayonnaise | Replacement of xanthan gum without deterioration of stability and taste | [62] |
| Medical and special applications | |||
| Peptide derived from yeast, fraction below 3 kDa | Orally applied solution (rats) | Blood pressure reduction comparable with captopril use | [63] |
| Selenium-rich peptide derived from yeast, fraction below 1 kDa | Intragastric applied solution (mice) | Protection against UV-induced skin damage | [64] |
| Yeast extract, yeast extract after additional enzymatic hydrolysis fraction below 3 kDa | Oral administration of water solution (rats) | Protection against stomach ulceration caused by 99.9% ethanol | [65] |
| Yeast extract | Addition to medium used in in vitro culture of Chinese hamster ovary cells | Growth enhancement | [39] |
| Yeast extract after additional enzymatic hydrolysis | Addition to serum-free medium used in in vitro culture of skeletal muscle cells | Restoration of cell culture growth | [66] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Łukaszewicz, M.; Leszczyński, P.; Jabłoński, S.J.; Kawa-Rygielska, J. Potential Applications of Yeast Biomass Derived from Small-Scale Breweries. Appl. Sci. 2024, 14, 2529. https://doi.org/10.3390/app14062529
Łukaszewicz M, Leszczyński P, Jabłoński SJ, Kawa-Rygielska J. Potential Applications of Yeast Biomass Derived from Small-Scale Breweries. Applied Sciences. 2024; 14(6):2529. https://doi.org/10.3390/app14062529
Chicago/Turabian StyleŁukaszewicz, Marcin, Przemysław Leszczyński, Sławomir Jan Jabłoński, and Joanna Kawa-Rygielska. 2024. "Potential Applications of Yeast Biomass Derived from Small-Scale Breweries" Applied Sciences 14, no. 6: 2529. https://doi.org/10.3390/app14062529
APA StyleŁukaszewicz, M., Leszczyński, P., Jabłoński, S. J., & Kawa-Rygielska, J. (2024). Potential Applications of Yeast Biomass Derived from Small-Scale Breweries. Applied Sciences, 14(6), 2529. https://doi.org/10.3390/app14062529







