1. Introduction
Periodontal disease is characterized by the destruction of periodontal tissues due to various local and systemic factors, such as infection and trauma. The body’s immune response to periodontal pathogens results in the loss of periodontal tissues and alveolar bone. This, in turn, impacts oral hygiene management and impedes healing. Therefore, the classification of the anatomical morphology of alveolar bone defects before the initiation of surgical periodontal interventions is crucial for treatment success.
Cortellini et al. (2000), Becker et al. (1986), and Kim et al. (2004) previously categorized the treatment prognosis of deep intrabony defects based on the type of surrounding bone tissues. Three-wall bony defects and two- and three-wall combined defects exhibited superior healing compared to one- or two-wall bony defects [
1,
2,
3].
Conventional non-surgical periodontal therapy is often insufficient for addressing periodontal tissue destruction extending from the multi-molar to the furcation region. Periodontal disease typically arises from the unchecked accumulation of pathogenic substances, such as calculus, on tooth surfaces. Involvement of the furcation area is common due to the narrow width of this region, which not only facilitates its occurrence but also complicates the treatment process. Consequently, incipient furcation involvement establishes a vicious cycle leading to the rapid progression of periodontitis.
Ross et al. (1980) previously reported a higher incidence of furcation involvement in multi-rooted teeth, with rates three times higher among maxillary molars compared to mandibular molars [
4]. Bower et al. examined furcation anatomy and noted that over half of the maxillary furcation entrances in their study were narrower than the width of a hand curette, contrasting with the situation in the mandible [
5]. Complete calculus removal poses challenges with non-surgical periodontal therapy, resulting in a particularly poor treatment prognosis for periodontal lesions extending into the periapical region.
The treatment of furcation involvement has long been recognized as challenging, often prompting periodontists to recommend extraction as the preferred therapeutic measure [
6]. However, since preserving natural teeth is a fundamental goal in periodontal therapy, various treatment approaches for furcation involvement have been developed, including guided tissue regeneration (GTR), tunneling, and root resection.
Previous studies indicate that multi-rooted teeth affected by segmental lesions can be retained for extended periods with adequate patient cooperation and effective surgical intervention. Clinical studies also suggest that furcation involvement may not be as serious a complication as initially suspected, particularly if caries development at the furcation site can be prevented [
7]. Instead, relatively straightforward periodontal treatment measures can be employed to maintain the functionality of these teeth over the long term.
Various surgical treatment measures for furcation involvement have been proposed over time. Farrar et al. were pioneers in 1884, reporting the resolution of a periodontal abscess in a multi-rooted tooth through root excision [
8]. Another technique, tunneling, involves exposing the affected furcation and was developed to enhance oral hygiene management. Longitudinal studies have shown its particular effectiveness in mandibular molars, with no associated loss of attachment or periapical or proximal alveolar bone reported [
9]. However, this method is less commonly used nowadays due to its association with complications such as root caries [
10].
In 1986, Gottlow et al. proposed GTR, aiming to achieve balanced attachment of bone, dentin, connective, and epithelial tissues through periodontal tissue regeneration [
11,
12]. The development of surgical guidelines and the increasing availability of clinical evidence on the efficacy of GTR have made this procedure popular for treating furcation involvement [
13]. Currently, GTR stands as the most commonly employed treatment measure for furcation involvement. Despite controversial evidence regarding its prognosis, previous studies have suggested that this procedure is effective in treating grade II furcation lesions in maxillary molars and grade III furcation lesions in mandibular molars [
14,
15].
In these segmental foci, GTR consistently exhibited superior results when compared to open flap debridement, particularly in reducing open horizontal furcation depth, enhancing horizontal and vertical attachment levels, and decreasing pocket depth in mandibular or maxillary class II furcation involvement lesions [
16].
GTR has the capacity to induce significant tissue regeneration, even in intrabony defects, and the treatment prognosis often hinges on the anatomical morphology of the lesion, such as the presence of a residual bone wall. The regenerated tissue is notably influenced by the size of the initial defect, with deeper and narrower defects exhibiting higher levels of clinical attachment.
Moreover, the angles between the bone wall and the base of the intrabony defects play a crucial role. Gentle angles necessitate lower degrees of bone filling compared to narrower and deeper defects, which, in turn, require proportionally greater bone regeneration [
17].
While numerous studies have compared graft and membrane types, few have focused on the morphology of the defect, and none have directly compared the amount of bone fill after GTR in intrabony defects and furcation involvement. Hence, the objective of this study is to compare the radiographic bone fill after GTR in mandibular and maxillary periodontal defects. Additionally, the study aims to assess the amount of radiographic bone fill after GTR in defects with distinct morphologic and etiologic characteristics, such as intrabony defects and furcation involvement. This aims to determine if GTR might be more effective depending on the morphology or location of the bone defect.
2. Materials and Methods
2.1. Patients and Treatment
The current retrospective study included patients who underwent GTR at Kyungpook National University Dental Hospital since 2012. The exclusion criteria were as follows: (1) the presence of uncontrolled endocrine systemic disease; (2) GTR performed on an implant rather than a natural tooth; (3) the impossibility of radiological evaluation of the operated tooth both pre- and post-operatively and during long-term follow-up; (4) use of other surgical techniques (e.g., the use of a pin for membrane fixation) and (5) patients who had not quit smoking for 2 months before and after surgery.
The study protocol was reviewed and accepted by the Research Ethics Committee at Kyungpook National University (KNUDH 2022-12-01-00).
All surgical treatments commenced with the elevation of the mucoperiosteal flap under thorough local anesthesia at the lesion site. Debridement of calculus, granulation tissue, and pathologic cementum located at the osseous defect was carried out, and root conditioning with doxycycline or ethylene-diamine-tetraacetic acid was performed. Subsequently, following the insertion of the bone graft or bone substitute graft, a collagen absorbable membrane (Bio-Gide® (Geistlich, Wolhusen, Switzerland) or OSSIX® PLUS (Datum Dental Biotech, Lod, Israel)) was placed to cover the defect up to 3 mm of surrounding healthy bone. The mucoperiosteal flap was closed with nylon or non-resorbable polytetrafluoroethylene sutures without exposing the membrane.
After surgical treatment, patients were instructed to gargle twice daily with a 0.12% chlorhexidine solution for 2 weeks and to take clindamycin or amoxicillin and non-steroidal anti-inflammatory drugs three times daily for at least 5 days. All patients visited the hospital once every 4 to 6 months for over a year after surgery to undergo full mouth scaling and examination for signs of infection, such as suppuration or swelling at the surgical site.
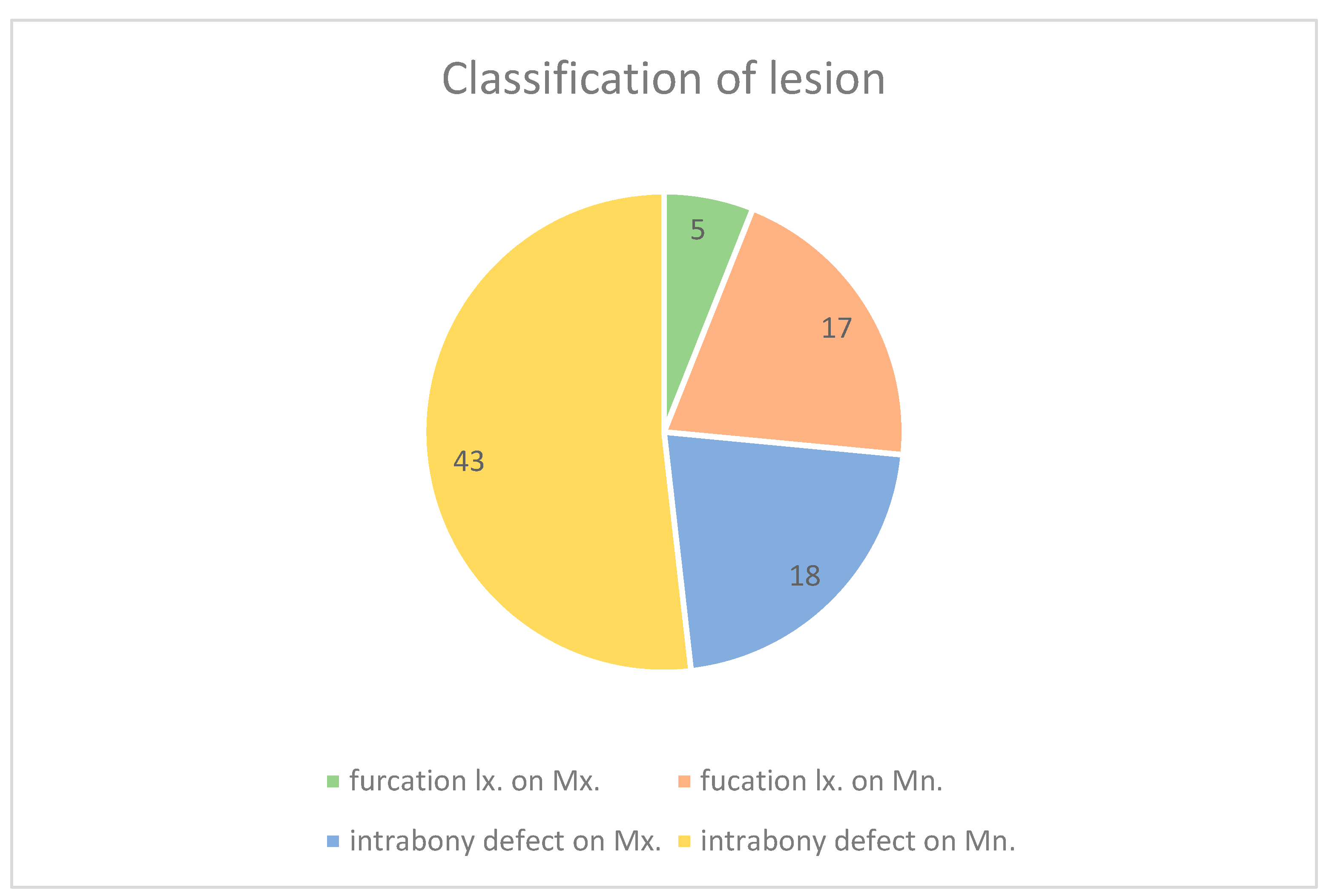
The final study sample comprised 80 patients, including 47 females and 33 males. One female patient underwent treatment for two teeth (maxillary second left molar, mandibular right first molar) over multiple appointments, while another underwent treatment for two separate lesions (mesial intrabony defects in mandibular first and second right molars) simultaneously. All other subjects received treatment for a single lesion. All defects classified as intrabony defects were 2- or 3-wall defects.
GTR was performed using various bone graft materials, including allograft (demineralized freeze-dried bone allograft), alloplast (MBCP® (Biomatlante Sarl, France): hydroxyapatite and beta-tricalcium phosphate), and xenograft from bovine (Bio-Oss® (Geistlich, Wolhusen, Switzerland)) or porcine (Legograft® (Purgo Biologics, Seongnam, Republic of Korea)) sources.
2.2. Radiographic Measurements
Panoramic radiographs, taken at a minimum vertical angle, were utilized to measure the depth of the bone defect one month before surgery and again between one year eight months and two years six months after surgery. The reference point was established as the distance between the deepest point of the bone defect and the cemento-enamel junction. This measurement was calculated using a system (INFINITT® (Infinitt Healthcare Co., Seoul, Republic of Korea)) that determines the actual clinical length of the radiograph using the same landmarks in the same case. Measurements were conducted independently by two periodontal trainees to minimize errors. Remeasurement was carried out if inter-examiner differences exceeded 0.5 mm.
Pre-operative radiographs were employed to assess the location (maxillary/mandibular) and nature (intrabony defect or alveolar bone loss associated with furcation involvement) of the lesion. The study sample was then divided into four groups: maxillary furcation involvement, mandibular furcation involvement, maxillary intrabony defect, and mandibular intrabony defect.
The amount of regenerated bone was measured using a radiograph and expressed as a proportion of the lesion depth.
2.3. Statistical Analysis
The statistical analysis comprised an equality of variance test to ensure uniform variances in all groups, followed by a t-test to compare the mean volume of regenerated bone after GTR between the groups. A p-value of <0.05 was deemed statistically significant.
4. Discussion
In this study, 82 periodontal lesions with bone defects in 80 patients were analyzed radiographically. The significance of the location of the lesion in the jawbone, the type of graft material, and the type of intraosseous defect were examined.
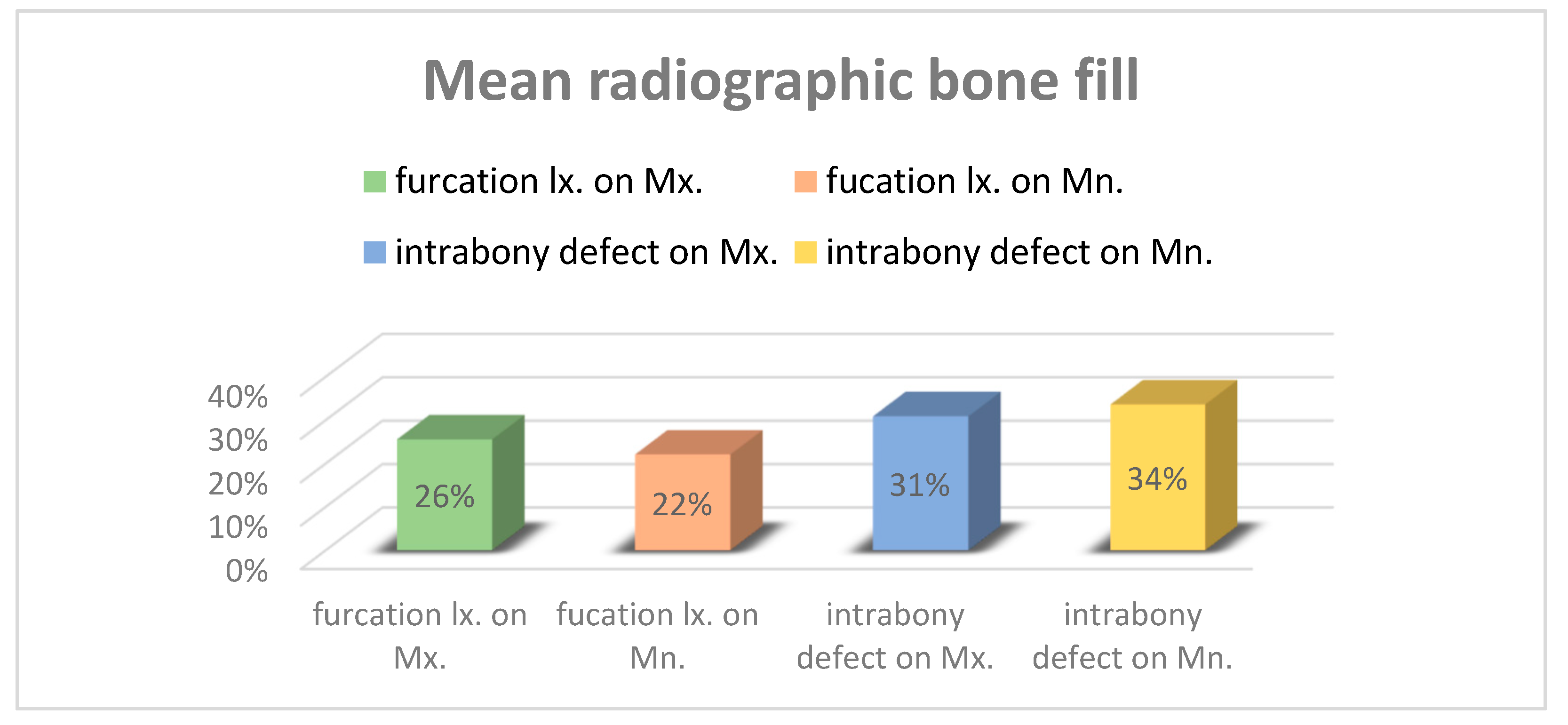
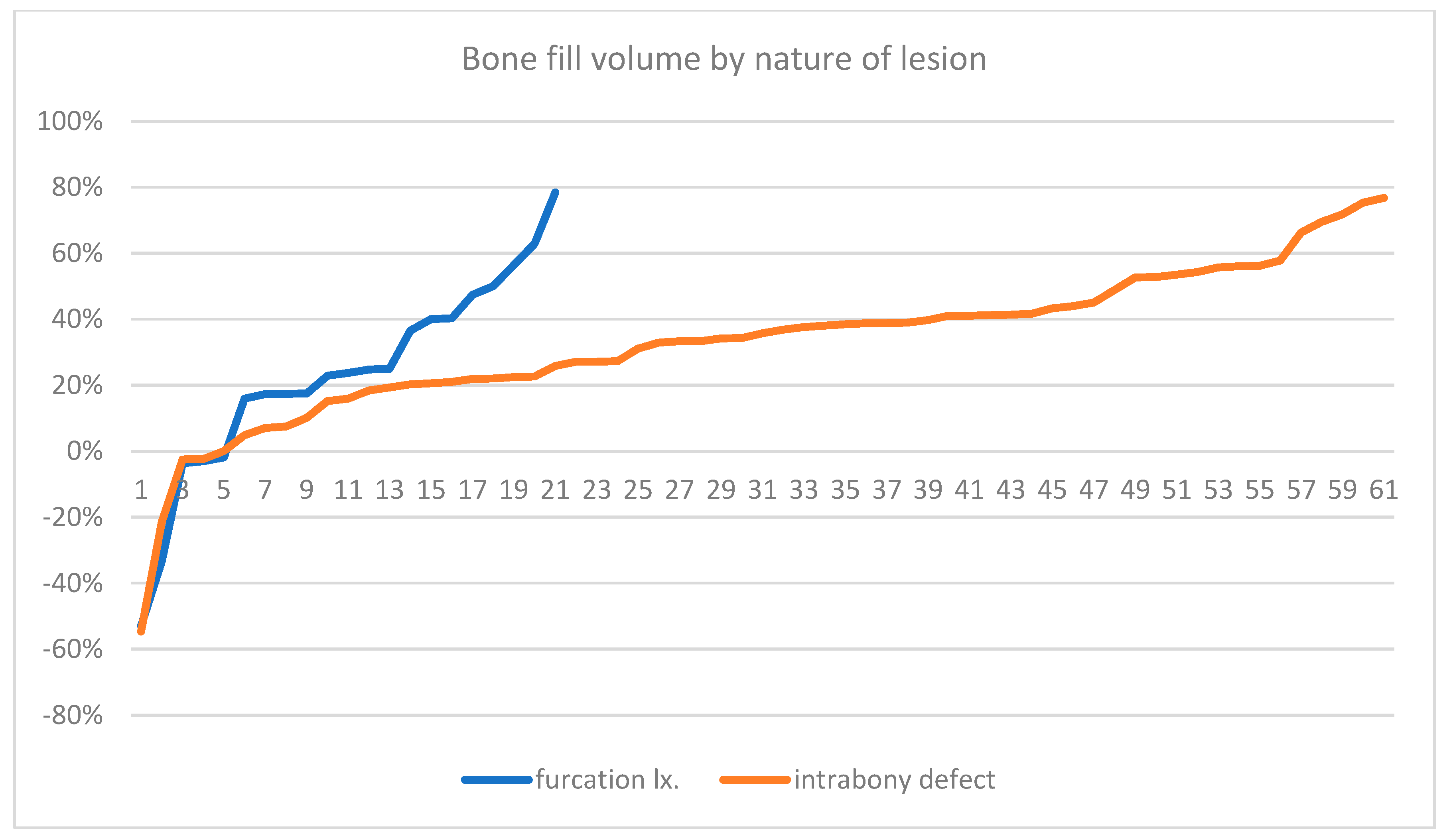
The results revealed no significant difference in bone volume between the furcation involvement group and the intrabony defect group. Therefore, a t-test assuming equal variance indicated that there was no statistically significant difference in the amount of bone growth based on the type of intraosseous defect.
Additionally, when the location of the intraosseous lesion was divided into two groups, there was no difference in variance between the groups. A t-test assuming equal variance also demonstrated no statistically significant difference in the amount of bone fill between the two groups.
There was not sufficient variation in bone graft materials or an adequate number of cases for each graft material to make a significant comparison. However, no graft material yielded significantly different results. This challenges the widely held theory that bone grafts or bone substitute grafts, such as demineralized freeze-dried bone allograft, possess unique osteoinductivity. Instead, it seems these materials primarily enhance wound stability within the defect, serving as a provisional matrix or scaffold.
In a 1999 study by CR Richardson et al. [
18], GTR in humans using Bio-Oss
®, a bovine-derived xenograft, and demineralized freeze-dried bone allograft showed no statistically significant difference in radiographic bone fill between the two graft groups. Numerous bone substitute grafts have been compared until recently. In a 2019 study [
19] by S Oh. et al., GTR with deproteinized bovine bone mineral (DBBM) and collagen membrane, and periodontal regeneration with collagen-containing DBBM alone, produced similar radiographic bone fill results. In 2023, Lee, J.H. et al. [
20] compared GTR for severe class II furcation defects in the mandibular molar using porcine-derived soft block bone and porcine-derived particulate bone substitutes, respectively, and found no significant difference in radiographic bone fill.
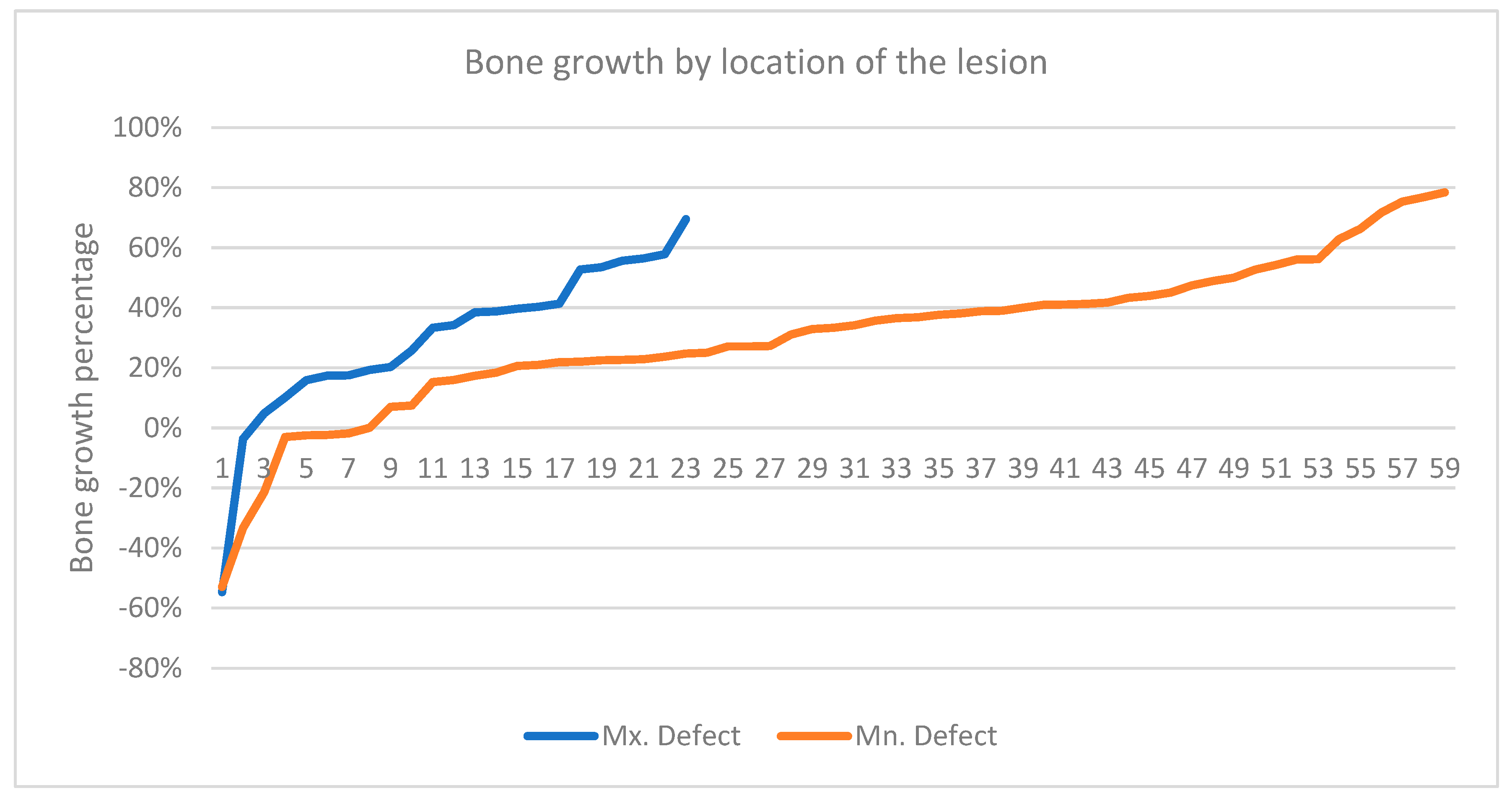
For furcation involvement, the
t-test also demonstrated no significant difference in the amount of bone fill when the location of the lesion was dichotomized into the upper and lower jaw. This finding contradicts a previous study by Tonetti et al. (1993), which suggested that maxillary furcation lesions would not respond more effectively to GTR than mandibular furcation lesions [
17].
The hypotheses positing a greater bone regeneration effect in GTR in the mandible than in the maxilla and a greater bone-filling effect in periodontal lesions without furcation area involvement than in lesions with furcation involvement, as advocated in the 1971 article by Ellegaard et al. and the 1993 article by Tonetti et al., were rejected in this study [
17,
21].
However, these results would be applicable if the internal debridement and root surface treatment of the defect were performed to the same degree in each case. The anatomy of maxillary molars is more likely to be trifurcated compared to mandibular molars, greatly limiting instrumental access to the furcation entrance [
22]. Successful regeneration of periodontal tissue occurs only in the absence of residual epithelial or granulation tissue or a complete couch of calculus debris and pathologic chalky material that may interfere with regeneration. To achieve this, preoperative computed tomography is recommended to determine the morphology of the defect and predict the flap design and necessary instrumentation.
During the course of this study, only radiologic evaluation of the teeth was possible, making it impossible to classify the Glickman or Hamp grade of the furcation lesion. Pontoriero’s study [
23] demonstrated that 100% horizontal defect fill could be achieved in mandibular Glickman grade 2 furcation involvement lesions. However, in class 3 furcation lesions, 100% fill was observed only when the width of the furcation entrance was within 4 mm. Therefore, it should not be overlooked that the difference in bone fill between maxillary and mandibular locations was not significant in this study, and the prognosis may vary depending on the morphology of individual teeth.
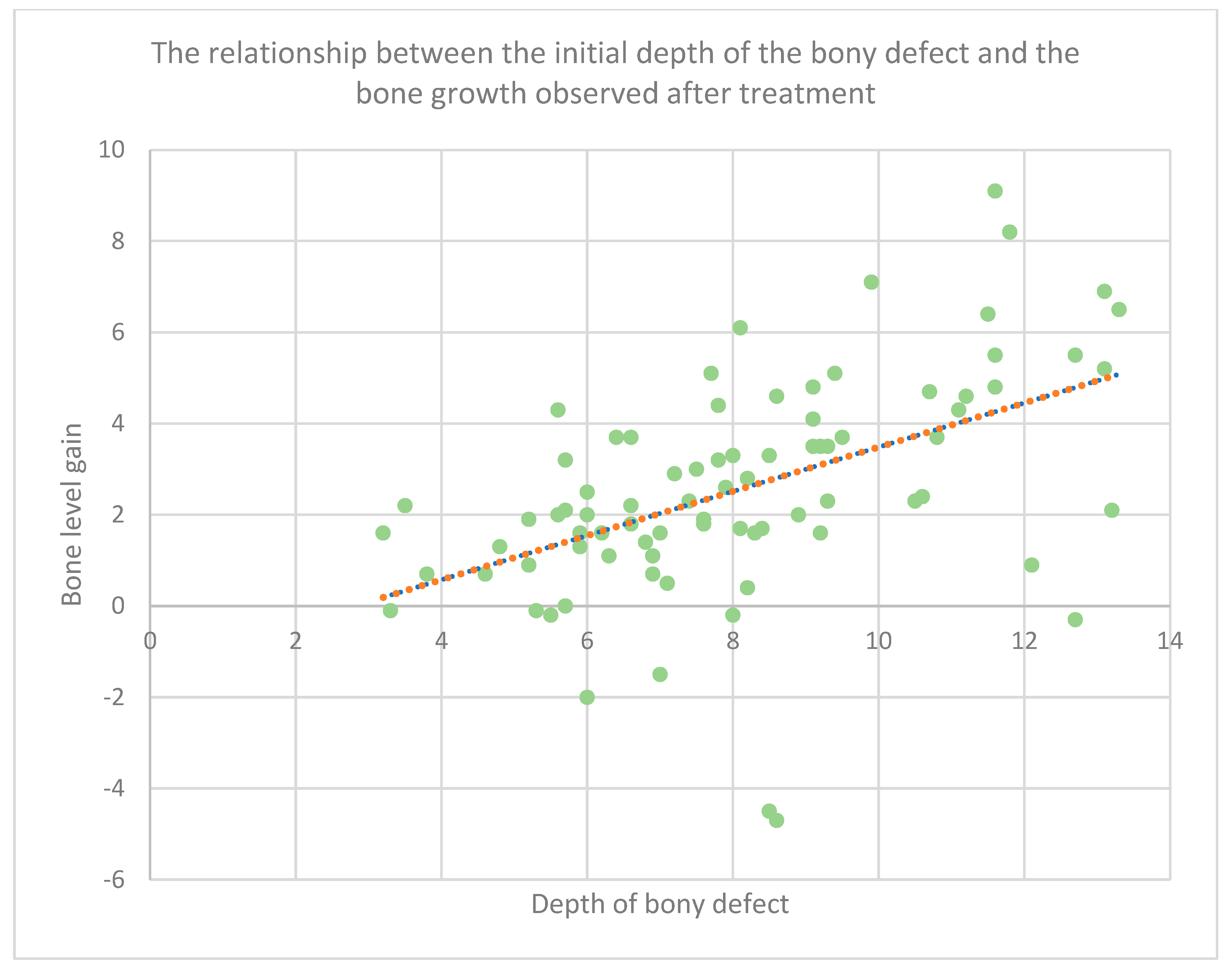
We conducted a comparative analysis of each case and found that, in intrabony defects with furcation involvement, as well as mesio-distal and interproximal defects, deeper defects did not result in a lower defect fill percentage. Consequently, we performed additional experiments during statistical analysis. Anticipating that the outcome might differ depending on the extent of the intraosseous defect, we analyzed the correlation between the initial amount of bone loss and the amount of bone filling.
The correlation between the distance (in mm) from the cemento-enamel junction to the lowest point of the bone defect lesion immediately before GTR and the amount of bone mass gain approximately 2 years after GTR was examined. A clear quantitative correlation was found. This supports findings from other previous studies, such as the one conducted by Tonetti et al. in 1993 [
20], suggesting that the greater the amount of bone loss, the greater the amount of bone gain.
This is likely attributed to the deeper and narrower nature of the bone defect and the larger volumetric measurements of the focal area, resulting in a more abundant supply of surrounding bone tissue and periodontal progenitor cells or originating growth factors, as supported by the literature [
24,
25]. Additionally, the vertical depth implies that the height of the marginal bone is higher than the base of the lesion, potentially preventing the collapse of the graft material or membrane itself and improving soft tissue stability for the operator.
Therefore, if volumetric stability of the bone graft and primary wound closure can be achieved, bone filling can be explored through GTR even in lesions with deep bone defects.
A limitation of this retrospective study is that the prognostic judgment of the surgeon was added to the decision of GTR in each case, and only cases with high predictive value were recruited. Nevertheless, it is noteworthy that similar amounts of bone filling were found in the maxilla, mandible, segmental lesions, and intraosseous defects. As revealed in this study, the type of defect and the alveolar location of the lesion do not significantly impact the differentiation of indications in the surgical treatment of lesions with alveolar bone defects. Therefore, it is worthwhile for clinicians to consider GTR more aggressively without being bound by traditional indication differentiation criteria.
When performing the GTR procedure, the choice of membrane is crucial. In this study, absorbable membranes were primarily used, but it is possible that the membrane resorption process is accompanied by a cellular inflammatory phase, which may have an adverse effect on healing. Expanded polytetrafluoroethylene (ePTFE) membranes, once considered the gold standard, or high-density polytetrafluoroethylene (dPTFE) membranes, commonly used in a wide range of hard tissue regeneration, may also be considered.
In the case of Capetillo, J.E. et al. [
26], GTR of a mixed Hamp grade 1 and 2 wall intrabony defect using a high-density polytetrafluoroethylene (dPTFE) membrane and a mixed graft of mineralized freeze-dried bone allograft and demineralized freeze-dried bone allograft resulted in successful bone regeneration despite the exposure of the membrane. If GTR is performed in large and challenging-to-repair defects, as this study aims to do, a releasing vertical incision will be performed due to the management of the periodontal flap, securing the vision, and efforts for primary closure. This sometimes results in inadequate nutrient or blood supply to the upper part of the flap, leading to necrosis or shrinkage, and ultimately membrane exposure. Exposure of the membrane can lead to bacterial colonization of the exposed area and infiltration into the membrane, interfering with the healing of the alveolar bone defect and resulting in an inadequate amount of bone fill after GTR [
27].
Furthermore, GTR has been actively applied to the treatment of peri-implantitis, which has been on the rise in recent years. Although the application of the GTR concept in general chronic bacterial peri-implantitis has not yielded successful results such as osseointegration or sustainable bone regeneration, it has produced positive outcomes in early implant failure. GTR not only prevents further loss of hard tissue but also achieves the regeneration of lost bone. In a case report by Lehmann, B. et al. [
28], GTR using an expanded polytetrafluoroethylene (ePTFE) membrane without bone graft re-addition was performed for early implant failure observed 2 months after implantation, and both radiographic and clinical findings at re-entry showed complete bone fill. Thus, it would be valuable to expand the use of GTR, considered one of the most predictive regenerative procedures, to include not only natural teeth but also the peri-implant area.
Another limitation of this study is that the vertical angle of the radiographs (panoramic or periapical radiographs) taken at each time point in each case was not consistent, potentially introducing errors in the measurement of bone level. There are variations in the time periods during which the radiographs were taken. Additionally, because this is a retrospective study, we cannot guarantee that all cases underwent surgery under the same conditions, as the treatment was not performed according to the purpose of this study. An additional limitation is that the evaluation was conducted beforehand, which may have influenced the results.
Due to the nature of GTR, postoperative bone filling or bone regeneration may be sensitive to differences in a person’s recovery ability, bone density, operator’s experience, etc. Therefore, prospective studies using a split-mouth design under the same conditions for various types and locations of bone defect lesions may be necessary in the future.