Distribution of Arabinoxylans and Their Relationship with Physiochemical and Rheological Properties in Wheat Flour Mill Streams as an Effective Way to Predict Flour Functionality
Abstract
:1. Introduction
2. Materials and Methods
3. Results
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Brütsch, L.; Huggler, I.; Kuster, S.; Windhab, E.J. Industrial roller milling process characterisation for targeted bread quality optimization. Food Bioprocess. Technol. 2017, 10, 710–719. [Google Scholar] [CrossRef]
- Prabhasankar, P.; Sudha, M.L.; Rao, P.H. Quality characteristics of wheat flour milled streams. Food Res. Int. 2000, 33, 381–386. [Google Scholar] [CrossRef]
- Banu, I.; Stoenescu, G.; Ionescu, V.; Aprodu, I. Physicochemical and rheological analysis of flour mill streams. Cereal Chem. 2010, 87, 112–117. [Google Scholar] [CrossRef]
- Vukić, M.; Janić Hajnal, E.; Mastilović, J.; Vujadinović, D.; Ivanović, M.; Šoronja-Simović, D. Application of solvent retention capacity tests for prediction of rheological parameters of wheat flour mill streams. Hem. Ind. 2020, 74, 37–49. [Google Scholar] [CrossRef]
- Ahmed, R.; Ali, R.; Saeed, S.A.; Ghufran Saeed, S.M.; Mobin, L. Impact of distinct compositional variations in flours of various milled streams on dough behavior and end quality of baked products. Pak. J. Bot. 2017, 49, 383–387. [Google Scholar]
- Ramseyer, D.; Bettge, A.; Morris, C.F. Distribution of total, water-unextractable, and water-extractable arabinoxylans in wheat flour mill streams. Cereal Chem. 2011, 88, 209–216. [Google Scholar] [CrossRef]
- Indrani, D.; Rajiv, J.; Prabhasankar, P.; Rao, G. Chemical, rheological and parotta making characteristics of flourmill streams. Eur. Food Res. Technol. 2003, 217, 219–223. [Google Scholar] [CrossRef]
- Gómez, M.; Ruiz-París, E.; Oliete, B. Influence of flour mill streams on cake quality. Int. J. Food Sci. Technol. 2010, 45, 1794–1800. [Google Scholar] [CrossRef]
- Liu, Y.; Ohm, J.B.; Hareland, G.; Wiersma, J.; Kaiser, D. Sulfur, protein size distribution, and free amino acids in flour mill streams and their relationship to dough rheology and breadmaking traits. Cereal Chem. 2011, 88, 109–116. [Google Scholar] [CrossRef]
- Pojić, M.; Hadnađev, M.; Dapčević Hadnađev, T. Gelatinization properties of wheat flour as determined by empirical and fundamental rheometric method. Eur. Food Res. Technol. 2013, 237, 299–307. [Google Scholar] [CrossRef]
- Pojić, M.; Spasojević, N.; Atlas, M. Chemometric approach to characterization of flour mill streams: Chemical and rheological properties. Food Bioprocess. Technol. 2014, 7, 1298–1309. [Google Scholar] [CrossRef]
- Every, D.; Simmons, L.; Al-Hakkak, J.; Hawkins, S.; Ross, M. Amylase, falling number, polysaccharide, protein and ash relationships in wheat millstreams. Euphytica 2002, 126, 135–142. [Google Scholar] [CrossRef]
- Iqbal, Z.; Pasha, I.; Abrar, M.; Hanif, M.S.; Arif, A.M.; Masih, S. Protein concentration, composition and distribution in wheat flour mill streams. Annals. Food Sci. Technol. 2015, 16, 104–114. [Google Scholar]
- Sutton, K.H.; Simmons, L.D. Molecular level protein composition of flour mill streams from a pilot-scale flour mill and its relationship to product quality. Cereal Chem. 2006, 83, 52–56. [Google Scholar] [CrossRef]
- Wang, Y.G.; Khan, K.; Hareland, G.; Nygard, G. Distribution of protein composition in bread wheat flour mill streams and relationship to breadmaking quality. Cereal Chem. 2007, 84, 271–275. [Google Scholar] [CrossRef]
- Dornez, E.; Gebruers, K.; Wiame, S.; Delcour, J.; Courtin, C. Insight into the distribution of arabinoxylans, endoxylanases, and endoxylanase inhibitors in industrial wheat roller mill streams. J. Agric. Food Chem. 2006, 54, 8521–8529. [Google Scholar] [CrossRef]
- Every, D.; Simmons, L.; Ross, M. Distribution of redox enzymes in millstreams and relationships to chemical and baking properties of flour. Cereal Chem. 2006, 83, 62–68. [Google Scholar] [CrossRef]
- Gebruers, K.; Courtin, C.; Goesaert, H.; Campenhout, S.; Delcour, J. Endoxylanase inhibition activity in different european wheat cultivars and milling fractions. Cereal Chem. 2002, 79, 613–616. [Google Scholar] [CrossRef]
- Rani, K.; Prasada Rao, U.J.S.; Leelavathi, K.; Haridas Rao, P. Distribution of enzymes in wheat flour mill streams. J. Cereal Sci. 2001, 34, 233–242. [Google Scholar] [CrossRef]
- Abdel-Haleem, A. Influence of heat treatment for some wheat milling fractions on fino bread quality. J. Food Sci. Technol. 2019, 56, 2639–2650. [Google Scholar] [CrossRef]
- Wang, M.; Sapirstein, H.D.; Machet, A.S.; Dexter, J.E. Composition and distribution of pentosans in millstreams of different hard spring wheats. Cereal Chem. 2006, 83, 161–168. [Google Scholar] [CrossRef]
- Engelsen, M.M.; Hansen, Å. Tocopherol and tocotrienol content in commercial wheat mill streams. Cereal Chem. 2009, 86, 499–502. [Google Scholar] [CrossRef]
- Izydorczyk, M.; Biliaderis, C.; Bushuk, W. Physical properties of water-soluble pentosans from different wheat varieties. Cereal Chem. 1991, 68, 145–150. [Google Scholar]
- Zannini, E.; Bravo Núñez, Á.; Sahin, A.W.; Arendt, E.K. Arabinoxylans as functional food ingredients: A Review. Foods 2022, 11, 1026. [Google Scholar] [CrossRef] [PubMed]
- Delcour, J.; Vanhamel, S.; Hoseney, R. Physicochemical and functional properties of rye nonstarch polysaccharides. II. Impact of a fraction containing water-soluble pentosans and proteins on gluten-starch loaf volumes. Cereal Chem. 1991, 68, 72–76. [Google Scholar]
- Michniewicz, J.; Biliaderis, C.; Bushuk, W. Effect of added pentosans on some properties of wheat bread. Food Chem. 1992, 43, 251–257. [Google Scholar] [CrossRef]
- Labat, E.; Morel, M.H.; Rouau, X. Effects of laccase and ferulic acid on wheat flour doughs. Cereal Chem. 2000, 77, 823–828. [Google Scholar] [CrossRef]
- Wang, M.W. Effect of Pentosans on Gluten Formation and Properties. Ph.D. Thesis, Wageningen University, Wageningen, The Netherlands, 2003. [Google Scholar]
- Jelaca, S.L.; Hlynca, I. Water binding capacity of wheat flour crude pentosans and their relation to mixing characteristics of dough. Cercal Chem. 1971, 48, 211–222. [Google Scholar]
- Patil, S.K.; Tsen, C.C.; Lineback, D.R. Water soluble pentosans of wheat flour. l. Viscosity properties and molecular weights estimated by gel filtration. Cereal Chem. 1975, 52, 44–56. [Google Scholar]
- Hoseney, R.C.; Faubion, J.M. A mechanism for the oxidative gelation of wheat flour water-soluble pentosans. Cereal Chem. 1981, 58, 421–424. [Google Scholar]
- Izydorczyk, M.S.; Biliaderis, C.G.; Bushuk, W. Physical properties of water-soluble pentosans from wheat. J. Cereal Sci. 1990, 11, 153–169. [Google Scholar] [CrossRef]
- Rouau, X.; EI-Hayek, M.L.; Moreau, D. Effect of an enzyme preparation containing pentosanases on the bread making quality of flours in relation to changes in pentosans properties. J. Cereal Sci. 1994, 19, 259–272. [Google Scholar] [CrossRef]
- Jelaca, S.L.; Hlynka, I. Effect of wheat-flour pentosans in dough, gluten, and bread. Cereal Chem. 1972, 49, 489–495. [Google Scholar]
- Kim, S.K.; D’Appolonia, B.L. Bread staling studies. I. Effect of protein content on staling rate and bread crumb pasting properties. Cereal Chem. 1977, 54, 207–215. [Google Scholar]
- Delcour, J.; Win, H.; Grobet, P. Distribution and structural variation of arabinoxylans in common wheat mill streams. J. Agric. Food Chem. 1999, 47, 271–275. [Google Scholar] [CrossRef] [PubMed]
- ICC. Standard Methods, on-line version; International Association for Cereal Science and Technology: Vienna, Austria, 2018. [Google Scholar]
- AACC. Approved Methods of Analysis; American Association of Cereal Chemists: St. Paul, MN, USA, 2009. [Google Scholar]
- Codină, G.G.; Mironeasa, S.; Bordei, D.; Leahu, A. Mixolab versus Alveograph and Falling Number. Czech J. Food Sci. 2010, 28, 185–191. [Google Scholar] [CrossRef]
- Jødal, A.S.S.; Larsen, K.L. Investigation of the relationships between the alveograph parameters. Sci. Rep. 2021, 11, 5349. [Google Scholar] [CrossRef] [PubMed]
- Szafrańska, A.; Rachoń, L.; Szumiło, G. Estimation of protein-starch complex of selected wheat species depending on production technology intensity. Zesz. Probl. Postępów Nauk Rol. 2015, 582, 81–90. [Google Scholar]
- Dubat, A. A New AACC International Approved Method to measure rheological properties of a dough sample. Cereal Foods World 2010, 55, 150–153. [Google Scholar] [CrossRef]
- Dubat, A.; Rosell, C.M.; Gallagher, E. Mixolab: A New Approach to Rheology, 1st ed.; American Associate of Cereal Chemists International: St. Paul, MN, USA, 2013. [Google Scholar]
- Guzmán, C.; Posadas-Romano, G.; Hernández-Espinosa, N.; Morales-Dorantes, A.; Peña, R.J. A new standard water absorption criteria based on solvent retention capacity (SRC) to determine dough mixing properties, viscoelasticity, and bread-making quality. J. Cereal Sci. 2015, 66, 59–65. [Google Scholar] [CrossRef]
- Kweon, M.; Slade, L.; Levine, H. Solvent Retention Capacity (SRC) testing of wheat flour: Principles and value in predicting flour functionality in different wheat-based food processes and in wheat breeding-A Review. Cereal Chem. 2011, 88, 537–552. [Google Scholar] [CrossRef]
- Englyst, H.; Cummings, J. Simplified method for the measurement of total non-starch polysaccharides by gas-liquid chromatography of constituent sugars as alditol acetates. Analyst 1984, 109, 937–942. [Google Scholar] [CrossRef]
- AOAC. Official Methods of Analysis, 15th ed.; Herlich, K., Ed.; The Association of Official Analytical Chemists: Arlington, VA, USA, 1990. [Google Scholar]
- Sakhare, S.D.; Inamdar, A.A. The cumulative ash curve: A best tool to evaluate complete mill performance. J. Food Sci. Technol. 2014, 51, 795–799. [Google Scholar] [CrossRef] [PubMed]
- Hoseney, C. Principles of Cereal Science and Technology, 2nd ed.; American Association of Cereal Chemists: St. Paul, MN, USA, 1994. [Google Scholar]
- Curić, D.; Karlović, D.; Tušak, D.; Petrović, B.; Dugum, J. Gluten as a standard of wheat flour quality. Food Technol. Biotechnol. 2001, 39, 353–361. [Google Scholar]
- He, J.; Penson, S.; Powers, S.; Hawes, C.; Shewry, P.R.; Tosi, P. Spatial patterns of gluten protein and polymer distribution in wheat grain. J. Agric. Food Chem. 2013, 61, 6207–6215. [Google Scholar] [CrossRef]
- Shewry, P.R.; Wan, Y.; Hawkesford, M.J.; Tosi, P. Spatial distribution of functional components in the starchy endosperm of wheat grains. J. Cereal Sci. 2020, 91, 102869. [Google Scholar] [CrossRef]
- Banu, I.; Aprodu, I. Association of physicochemical with technological properties of wheat. Int. J. Food Sci. Technol. 2015, 5, 1644–1650. [Google Scholar] [CrossRef]
- Tian, X.; Wang, Z.; Wang, X.; Ma, S.; Sun, B.; Wang, F. Mechanochemical effects on the structural properties of wheat starch during vibration ball milling of wheat endosperm. Int. J. Biol. Macromol. 2022, 206, 306–312. [Google Scholar] [CrossRef]
- Rheological and enzyme analyses. In Mixolab Applications Handbook; Chopin Applications Laboratory: Villeneuve-la-Garenne, France, 2012.
- van Vliet, T. Strain hardening as an indicator of bread-making performance: A review with discussion. J. Cereal Sci. 2008, 48, 1–9. [Google Scholar] [CrossRef]
- Dobraszczyk, B.J.; Salmanowicz, B.P. Comparison of predictions of baking volume using large deformation rheological properties. J. Cereal Sci. 2008, 47, 292–301. [Google Scholar] [CrossRef]
- Sroan, B.; Bean, S.; Macritchie, F. Mechanism of gas cell stabilization in bread making. I. The primary gluten—starch matrix. J. Cereal Sci. 2009, 49, 32–40. [Google Scholar] [CrossRef]
- Di Stasio, M.; Vacca, P.; Piciocchi, N.; Meccariello, C.; Volpe, M.G. Particle size distribution and starch damage in some soft wheat cultivars. Int. J. Food Sci. Technol. 2007, 42, 246–250. [Google Scholar] [CrossRef]
- Collar, C.; Bollain, C.; Rosell, C.M. Rheological behaviour of formulated bread doughs during mixing and heating. Food Sci. Technol. Int. 2007, 13, 99–107. [Google Scholar] [CrossRef]
- Rosell, C.M.; Collar, C.; Haros, M. Assessment of hydrocolloid effects on the thermo-mechanical properties of wheat using the Mixolab. Food Hydrocoll. 2007, 21, 452–462. [Google Scholar] [CrossRef]
- Fustier, P.; Castaigne, F.; Turgeon, S.L.; Biliaderis, C.G. Impact of commercial soft wheat flour streams on dough rheology and quality attributes of cookies. J. Food Eng. 2009, 90, 228–237. [Google Scholar] [CrossRef]
- Haros, M.; Ferrer, A.; Rosell, M.C. Rheological behaviour of whole wheat flour. In Proceedings of the IUFoST 13th World Congress of Food Sciences Technology, Nantes, France, 17–21 September 2006; pp. 1139–1148. [Google Scholar] [CrossRef]
- Lindgren, A.; Simsek, S. Evaluation of hard red spring wheat mill stream fractions using solvent retention capacity test. J. Food Process Preserv. 2016, 40, 131–139. [Google Scholar] [CrossRef]
- Li, Y.; Wang, L.; Wang, H.; Li, Z.; Qiu, J.; Wang, L. Correlation of microstructure, pore characteristics and hydration properties of wheat bran modified by airflow impact mill. Innov. Food Sci. Emerg. Technol. 2022, 77, 102977. [Google Scholar] [CrossRef]
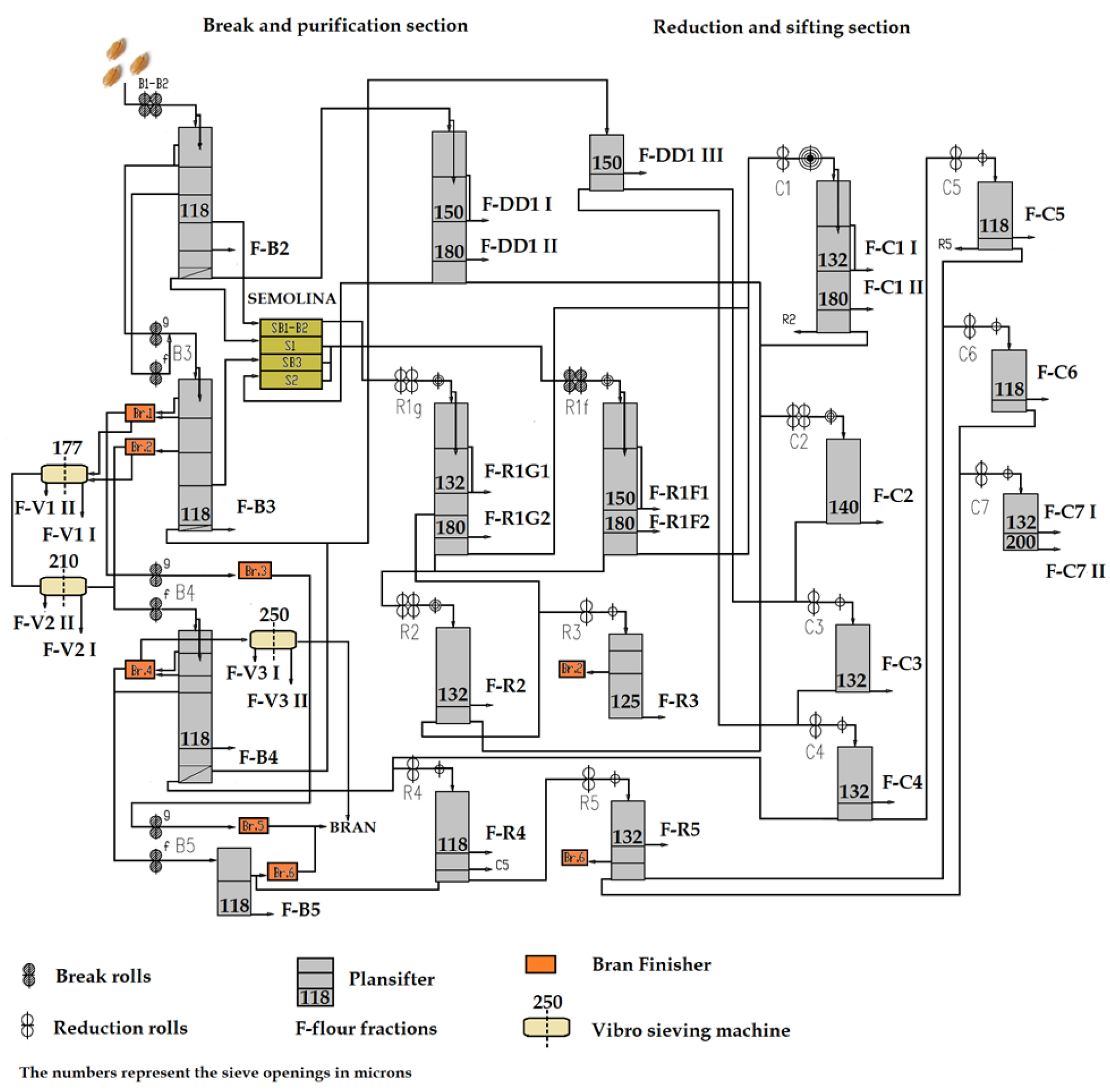
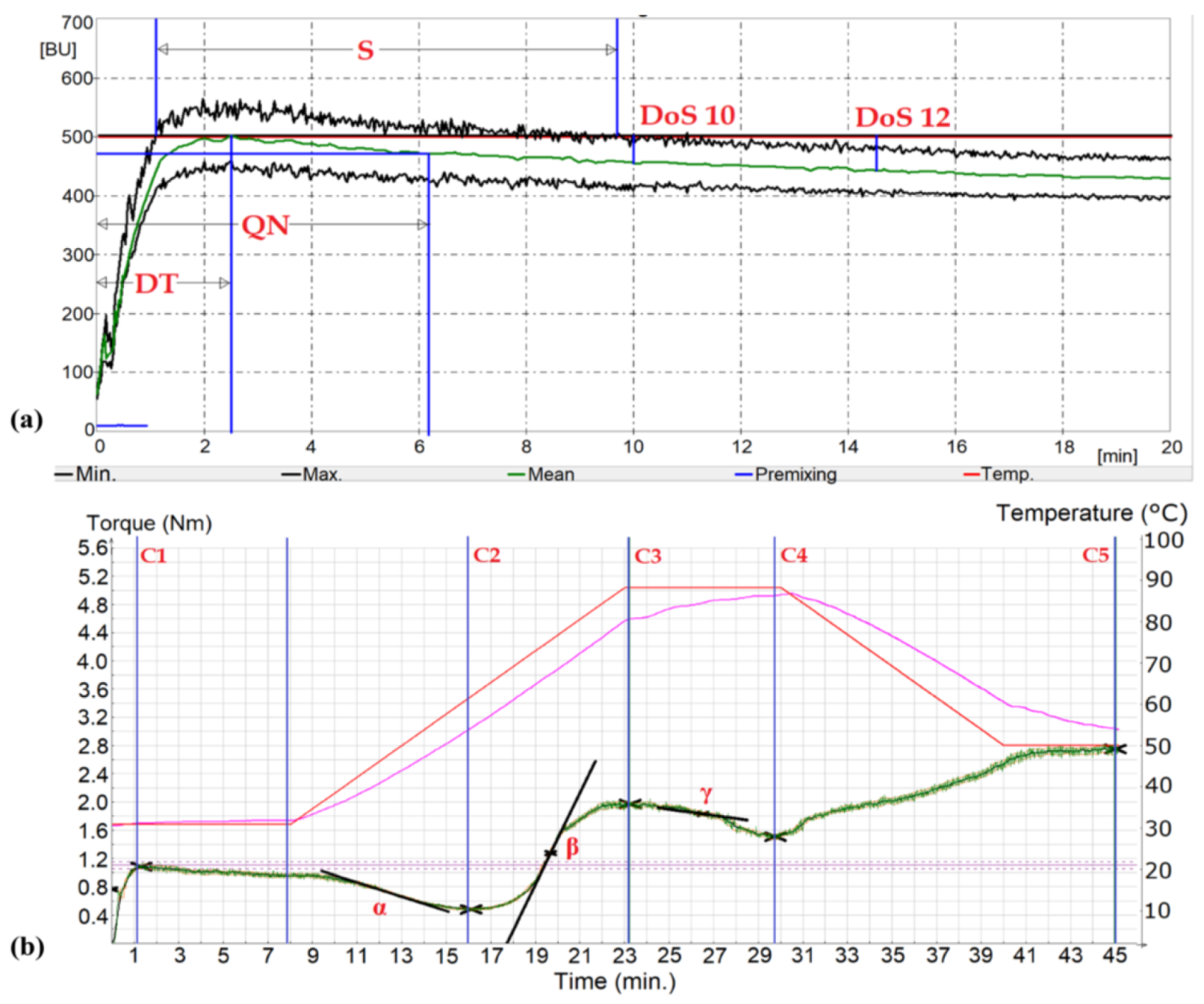
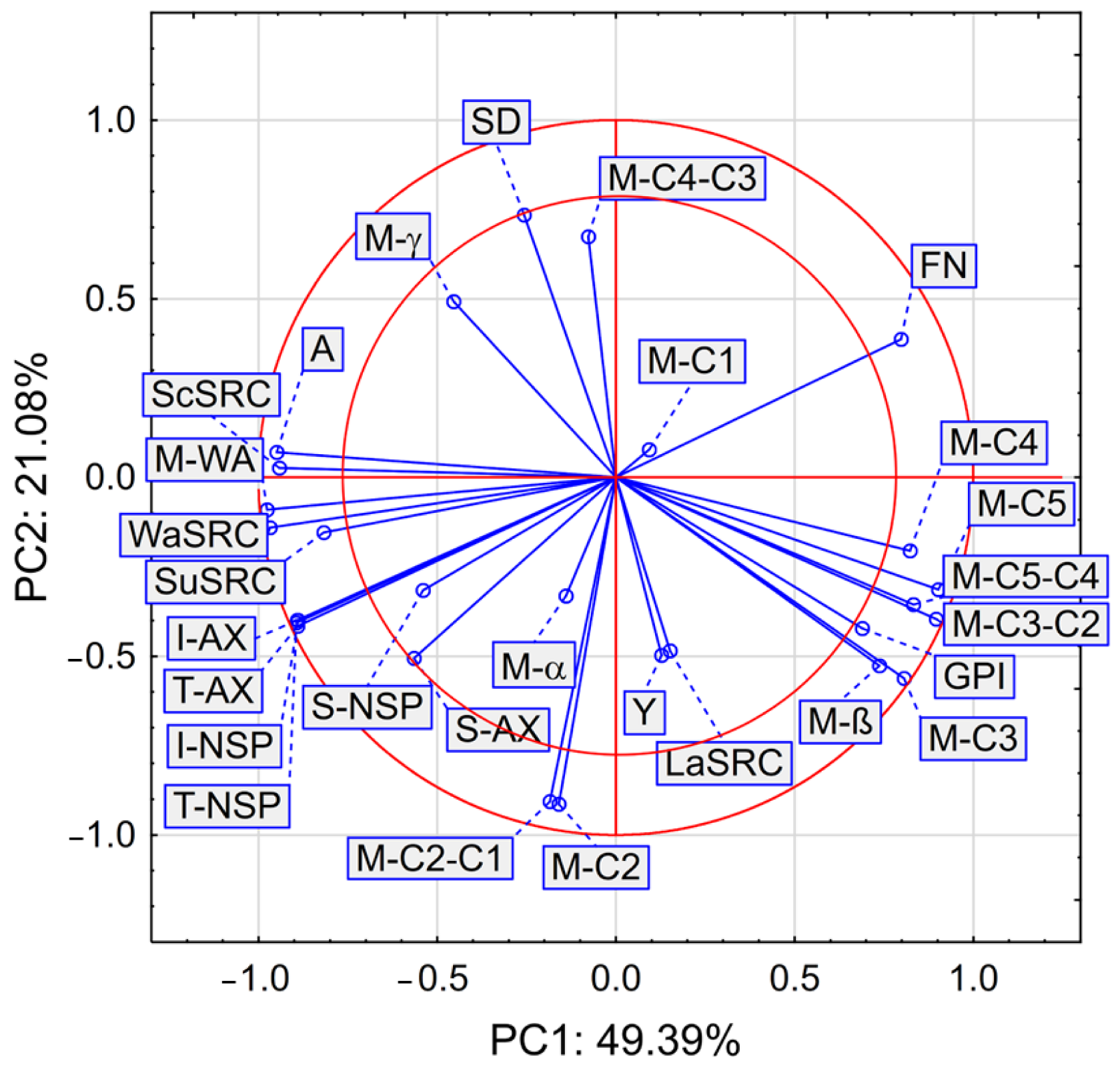
| Flour Stream | A (%) | G (%) | GI (-) | FN (s) | SD (%) |
|---|---|---|---|---|---|
| B2 | 0.671 ± 0.026 g | 35.99 ± 1.67 j,k | 94.8 ± 2.2 e,f | 388 ± 14 d,e | 93.01 ± 0.39 i |
| B3 | 0.620 ± 0.009 f | 38.78 ± 0.59 l | 96.3 ± 0.9 f,g,h,i | 423 ± 26 h,j,k,l | 92.90 ± 0.23 i |
| B4 | 0.936 ± 0.002 i | 36.62 ± 0.27 k | 95.5 ± 0.5 e,f,g,h | 410 ± 4 e,f,g,h,i,j,k | 94.00 ± 0.16 j |
| B5 | 1.282 ± 0.002 m | 43.56 ± 0.16 m,n | 86.5 ± 0.5 b | 446 ± 4 l,m | 95.84 ± 0.02 m |
| C1I | 0.440 ± 0.009 a,b | 30.86 ± 0.04 f,g | 97.5 ± 0.5 g,h,i | 430 ± 2 j,k,l | 92.30 ± 0.10 h |
| C1II | 0.465 ± 0.003 b,c | 29.53 ± 0.12 c,d,e,f | 99.0 ± 0.0 i | 446 ± 2 l,m | 85.09 ± 0.03 b |
| C2 | 0.466 ± 0.015 b,c | 28.65 ± 0.25 b,c,d,e | 98.5 ± 0.5 h,i | 359 ± 1 c | 89.32 ± 0.07 h |
| C3 | 0.489 ± 0.012 c | 32.10 ± 0.09 g,h | 99.0 ± 0.0 i | 410 ± 5 e,f,g,h,i,j,k | 90.65 ± 0.03 f |
| C4 | 0.733 ± 0.006 h | 29.59 ± 0.15 c,d,e,f | 94.0 ± 1.0 d,e,f | 419 ± 5 g,h,i,j,k,l | 96.00 ± 0.01 m,n |
| C5 | 0.755 ± 0.001 h | 27.59 ± 0.07 b,c | 98.5 ± 0.5 h,i | 436 ± 5 k,l,m | 95.91 ± 0.06 m,n |
| C6 | 1.461 ± 0.003 o | 23.84 ± 0.06 a | 93.0 ± 0.0 d,e | 429 ± 4 j,k,l | 95.69 ± 0.05 l,m |
| C7I | 2.352 ± 0.002 p | ND | ND | 410 ± 4 e,f,g,h,i,j,k | 97.20 ± 0.03 p |
| C7II | 3.931 ± 0.002 s | ND | ND | 74 ± 4 a | 87.60 ± 0.12 d |
| DD1I | 0.584 ± 0.007 d,e | 32.96 ± 0.26 h,i | 99.0 ± 0.0 i | 444 ± 2 l,m | 90.73 ± 0.11 f |
| DD1II | 0.579 ± 0.005 d,e | 30.66 ± 0.17 e,f,g | 96.5 ± 2.5 f,g,h,i | 394 ± 1 d,e,f,g | 86.83 ± 0.03 c |
| DD1III | 0.565 ± 0.001 d | 37.54 ± 0.12 k,l | 96.0 ± 0.0 e,f,g,h,i | 463 ± 3 m | 90.33 ± 0.05 f |
| R1F1 | 0.471 ± 0.007 b,c | 30.26 ± 0.10 d,e,f,g | 97.0 ± 0.0 f,g,h,i | 385 ± 1 c,d,e,f | 93.84 ± 0.15 j |
| R1G1 | 0.486 ± 0.010 c | 28.54 ± 0.25 b,c,d | 91.5 ± 0.5 c,d | 428 ± 5 j,k,l | 96.71 ± 0.05 o |
| R1G2 | 0.428 ± 0.001 a,c | 27.05 ± 0.07 b | 98.0 ± 0.0 g,h,i | 388 ± 4 c,d,e,f | 91.51 ± 0.06 g |
| R1F2 | 0.418 ± 0.003 a | 29.69 ± 0.17 d,e,f | 97.5 ± 0.5 g,h,i | 405 ± 5 c,d,e,f | 81.97 ± 0.04 a |
| R2 | 0.589 ± 0.002 d,e | 29.57 ± 0.42 c,d,e,f | 98.0 ± 0.0 g,h,i | 424 ± 4 h,i,j,k,l | 92.12 ± 0.13 h |
| R3 | 0.609 ± 0.004 e,f | 28.67 ± 0.21 b,c,d,e | 95.5 ± 0.5 e,f,g,h | 421 ± 5 g,h,i,j,k,l | 95.89 ± 0.06 m,n |
| R4 | 1.063 ± 0.001 k | 29.33 ± 0.18 c,d,e,f | 96.0 ± 0.0 e,f,g,h,i | 412 ± 2 e,f,g,h,i,j,k | 96.38 ± 0.06 n,o |
| R5 | 2.633 ± 0.006 r | ND | ND | 301 ± 3 b | 98.09 ± 0.07 r |
| V1I | 1.008 ± 0.014 j | 36.39 ± 0.04 j,k | 94.0 ± 0.0 d,e,f | 377 ± 3 c,d | 95.21 ± 0.04 k,l |
| V1II | 1.012 ± 0.003 j | 37.12 ± 0.06 k,l | 88.5 ± 0.5 b,c | 362 ± 6 c | 97.20 ± 0.06 p |
| V2I | 1.041 ± 0.01 j,k | 34.40 ± 0.24 i,j | 95.0 ± 0.0 e,f,g | 403 ± 5 d,e,f,g,h,i,j | 90.39 ± 0.04 f |
| V2II | 1.284 ± 0.008 m | 28.64 ± 0.16 b,c,d | 59.0 ± 0.0 a | 398 ± 4 d,e,f,g,h,i | 91.56 ± 0.06 g |
| V3I | 1.397 ± 0.007 n | 41.79 ± 0.18 m | 87.0 ± 0.0 b | 414 ± 4 f,g,h,i,j,k | 96.61 ± 0.01 r |
| V3II | 1.218 ± 0.006 l | 44.62 ± 0.17 n | 87.0 ± 1.0 b | 395 ± 4 d,e,f,g,i | 94.96 ± 0.07 k |
| Flour Stream | P (mm) | L (mm) | W (J 10−4) | P/L | Ie (%) | SH Index |
|---|---|---|---|---|---|---|
| B2 | 66.4 ± 0.9 b | 146.7 ± 11.1 m | 289.4 ± 12.5 k,m | 0.46 ± 0.04 a,b | 58.53 ± 0.96 m,n | 1.81 ± 0.02 n |
| B3 | 62.3 ± 1.5 a | 173.8 ± 19.8 n | 309.5 ± 21.5 m,n | 0.36 ± 0.04 a | 60.23 ± 0.63 n | 1.82 ± 0.03 m,n |
| B4 | 83.3 ± 1.2 d,e,f | 107.7 ± 12.0 j,k,l | 210.3 ± 14.5 d,e | 0.78 ± 0.09 b,c,d | 41.93 ± 0.93 f,g | 1.33 ± 0.04 b,c,d |
| B5 | 75.7 ± 1.2 c | 119.0 ± 6.2 l | 191.0 ± 7.8 b,c,d | 0.64 ± 0.03 a,b,c,d | 38.83 ± 0.15 e | 1.30 ± 0.04 b,c |
| C1I | 81.0 ± 0.0 c,d,e | 113.0 ± 2.0 k,l | 283.3 ± 6.1 j,k,l,m | 0.72 ± 0.02 a,b,c,d | 55.93 ± 0.21 l,m | 1.79 ± 0.01 l,m,n |
| C1II | 90.0 ± 1.7 g,h,i | 88.3 ± 4.0 g,h,i,j,k | 256.0 ± 10.1 g,h,i,j | 1.02 ± 0.04 d,e,f | 52.97 ± 0.23 j,k | 1.74 ± 0.02 j,k,l,m,n |
| C2 | 82.7 ± 1.5 d,e,f | 94.3 ± 4.2 h,i,j,k,l | 261.3 ± 8.5 g,h,i,j,l | 0.88 ± 0.04 c,d,e | 57.20 ± 0.36 m,n | 1.82 ± 0.00 m,n |
| C3 | 89.7 ± 2.1 g,h | 106.3 ± 1.2 j,k,l | 290.7 ± 7.0 k,l,m | 0.84 ± 0.02 c,d,e | 54.20 ± 0.17 k,l | 1.74 ± 0.01 j,k,l,m,n |
| C4 | 103.7 ± 0.6 l,m | 66.0 ± 2.0 d,e,f,g | 211.0 ± 2.6 d,e,f | 1.57 ± 0.06 h,i,j | 40.93 ± 0.47 e,f | 1.51 ± 0.03 e,f,g,h |
| C5 | 95.7 ± 1.5 i,j,k | 60.7 ± 1.5 c,d,e,f | 171.7 ± 6.0 b,c | 1.57 ± 0.02 h,i,j | 34.67 ± 0.74 d | 1.38 ± 0.02 b,c,d,e |
| C6 | 111.7 ± 2.5 n,o | 40.0 ± 1.0 a,b,c | 106.3 ± 10.5 a | 2.79 ± 0.12 l | 6.45 ± 0.50 a | ND |
| C7I | 93.3 ± 2.3 h,i,j | 31.0 ± 6.6 a,b | 80.3 ± 4.9 a | 3.11 ± 0.71 l | ND | ND |
| C7II | ND | ND | ND | ND | ND | ND |
| DD1I | 80.3 ± 2.1 c,d | 104.0 ± 1.7 i,j,k,l | 270.0 ± 4.6 h,i,j,k,l | 0.78 ± 0.03 b,c,d | 57.77 ± 0.21 m,n | 1.79 ± 0.01 l,m,n |
| DD1II | 86.7 ± 1.5 e,f,g | 88.3 ± 3.1 g,h,i,j,k | 245.0 ± 6.9 f,g,h,i | 0.98 ± 0.02 d,e,f | 52.90 ± 0.20 j,k | 1.72 ± 0.03 j,k,l,m,n |
| DD1III | 68.7 ± 1.5 b | 148.3 ± 9.5 m | 293.0 ± 4.4 k,l,m | 0.46 ± 0.04 a,b,c | 57.27 ± 0.84 m,n | 1.79 ± 0.02 l,m,n |
| R1F1 | 109.3 ± 2.1 m,n | 87.3 ± 4.0 g,h,i,j | 330.0 ± 14.4 n | 1.26 ± 0.06 e,f,g,h | 57.57 ± 0.06 m,n | 1.83 ± 0.01 m,n |
| R1G1 | 121.3 ± 0.6 p | 68.3 ± 6.1 d,e,f,g | 296.7 ± 16.1 g,i,j,k,l,m | 1.79 ± 0.17 i,j,k | 53.73 ± 0.21 k,l | 1.81 ± 0.02 m,n |
| R1G2 | 107.7 ± 3.1 m,n | 72.0 ± 2.6 d,e,f,g,h | 272.3 ± 3.5 k,m,n | 1.50 ± 0.10 g,h,i | 54.17 ± 0.67 k,l | 1.78 ± 0.03 k,l,m,n |
| R1F2 | 86.3 ± 0.6 e,f,g | 81.3 ± 4.2 e,f,g,h,i | 235.7 ± 8.3 e,f,g | 1.06 ± 0.06 d,e,f,g | 54.30 ± 0.44 k,l | 1.76 ± 0.02 k,l,m,n |
| R2 | 101.3 ± 1.2 k,l | 75.3 ± 1.5 d,e,f,g,h | 238.0 ± 2.0 e,f,g,h | 1.35 ± 0.04 f,g,h,i | 46.23 ± 0.06 h | 1.63 ± 0.01 h,i,j,k |
| R3 | 87.3 ± 0.6 f,g | 108.3 ± 3.8 j,k,l | 265.0 ± 6.1 g,h,i,j,k,l | 0.81 ± 0.04 b,c,d,e | 49.93 ± 0.51 i | 1.64 ± 0.02 h,i,j,k,l |
| R4 | 98.7 ± 0.6 j,k,l | 56.7 ± 2.5 c,d,e | 160.7 ± 1.5 b | 1.74 ± 0.09 i,j | 29.20 ± 0.20 b | 1.23 ± 0.07 b |
| R5 | 90.0 ± 1.0 g,h,i | 25.0 ± 1.0 a | 85.7 ± 8.1 a | 3.60 ± 0.13 m | ND | 1.05 ± 0.34 a |
| V1I | 143.7 ± 6.0 r | 80.0 ± 5.0 e,f,g,h,i | 365.3 ± 22.0 o | 1.79 ± 0.10 i,j,k | 50.30 ± 0.27 i | 1.67 ± 0.01 i,j,k,l,m |
| V1II | 151.3 ± 1.2 s | 77.0 ± 4.6 e,f,g,h | 382.3 ± 9.5 o | 1.97 ± 0.13 j,k | 51.27 ± 0.25 i,j | 1.72 ± 0.03 j,k,l,m,n |
| V2I | 112.7 ± 2.3 n,o | 50.7 ± 2.1 b,c,d | 184.3 ± 4.0 b,c,d | 2.23 ± 0.13 k | 31.57 ± 2.45 c | 1.42 ± 0.04 c,d,e,f |
| V2II | 79.3 ± 2.5 c,d | 83.7 ± 0.6 f,g,h,i,j | 198.7 ± 7.0 c,d | 0.95 ± 0.03 d,e,f | 47.23 ± 0.21 h | 1.59 ± 0.03 g,h,i,j |
| V3I | 115.7 ± 2.3 o,p | 78.3 ± 5.1 e,f,g,h | 259.3 ± 3.8 g,h,i,j,l | 1.48 ± 0.12 g,h,i | 43.23 ± 0.60 g | 1.47 ± 0.02 d,e,f,g |
| V3II | 103.7 ± 1.5 l,m | 75.7 ± 3.5 e,f,g,h | 238.0 ± 3.5 e,f,g,h | 1.37 ± 0.08 f,g,h,i | 46.10 ± 0.00 h | 1.57 ± 0.03 f,g,h,i |
| Flour Stream | WA (%) | DT (min) | S (min) | DoS 10 (BU) | DoS 12 (BU) | QN (-) |
|---|---|---|---|---|---|---|
| B2 | 56.68 ± 0.63 d,e | 7.4 ± 1.0 g | 16.2 ± 0.9 j | 11.67 ± 3.43 a | 48.17 ± 3.37 a,b,c | 136.89 ± 6.35 l,m |
| B3 | 57.43 ± 0.16 f,g | 7.0 ± 1.1 e,f,g | 17.9 ± 0.3 k | 8.33 ± 3.50 a | 42.40 ± 7.86 a,b | 155.67 ± 8.14 n |
| B4 | 62.90 ± 0.00 k | 5.5 ± 0.4 d,e | 6.3 ± 0.2 d | 38.67 ± 1.53 e,f,g | 77.33 ± 2.08 i,j,k | 88.33 ± 3.79 e,f,g |
| B5 | 66.27 ± 0.06 m,n | 6.2 ± 0.5 d,e,f,g | 5.9 ± 0.3 c,d | 36.00 ± 6.08 d,e,f | 69.00 ± 6.00 g,h,i,j | 92.33 ± 5.51 f,g,h |
| C1I | 56.47 ± 0.21 c,d,e | 3.2 ± 0.3 a,b,c | 10.5 ± 0.9 g,h | 35.33 ± 7.57 d,e,f | 53.33 ± 5.13 b,c,d,e | 87.67 ± 18.04 e,f,g |
| C1II | 55.53 ± 0.06 b | 2.4 ± 0.4 a | 9.7 ± 0.6 g | 39.67 ± 2.52 e,f,g | 54.00 ± 3.00 b,c,d,e,f | 75.67 ± 3.22 d,e,f |
| C2 | 55.67 ± 0.06 b,c | 2.9 ± 0.5 a,b | 12.2 ± 0.8 h,i | 34.67 ± 4.04 d,e,f | 48.00 ± 2.65 a,b,c,d | 86.67 ± 4.16 e,f,g |
| C3 | 57.20 ± 0.10 e,f,g | 2.7 ± 0.2 a | 9.2 ± 0.4 f,g | 42.67 ± 1.53 f,g | 58.67 ± 1.53 c,d,e,f,g,h | 72.67 ± 2.52 d,e,f |
| C4 | 61.70 ± 0.00 j | 4.8 ± 0.9 b,c,d | 7.5 ± 0.1 d,e,f | 40.33 ± 2.08 e,f,g | 77.00 ± 4.00 i,j,k | 88.33 ± 2.52 e,f,g |
| C5 | 61.83 ± 0.06 j | 4.9 ± 0.3 c,d | 6.2 ± 0.1 d | 43.33 ± 2.08 f,g | 81.67 ± 0.58 j,k | 83.67 ± 2.31 d,e,f |
| C6 | 69.13 ± 0.06 o | 5.6 ± 0.1 d,e,f | 4.2 ± 0.5 a,b,c | 61.67 ± 4.16 h,i | 87.33 ± 5.51 k | 76.00 ± 2.00 d,e,f |
| C7I | 69.83 ± 0.06 o | 5.7 ± 0.3 d,e,f | 2.7 ± 0.4 a | 116.67 ± 6.35 j | 182.33 ± 5.86 l | 74.00 ± 1.73 d,e,f |
| C7II | ND | ND | ND | ND | ND | ND |
| DD1I | 56.23 ± 0.06 b,c,d | 4.7 ± 0.7 b,c,d | 15.2 ± 1.2 j | 18.33 ± 5.03 a,b,c | 40.00 ± 0.00 a | 129.33 ± 10.26 k,l,m |
| DD1II | 55.67 ± 0.06 b,c | 2.2 ± 0.3 a | 9.3 ± 0.5 f,g | 45.67 ± 2.08 f,g | 56.33 ± 2.08 c,d,e,f,g | 69.00 ± 3.00 c,d,e |
| DD1III | 57.07 ± 0.06 e,f | 7.8 ± 0.3 g | 16.5 ± 0.6 j,k | 8.67 ± 2.52 a | 50.00 ± 0.00 a,b,c,d,e | 144.33 ± 7.77 m,n |
| R1F1 | 57.90 ± 0.10 g,h | 2.7 ± 0.3 a | 8.8 ± 0.3,f,g | 44.00 ± 2.65 f,g | 55.00 ± 3.46 c,d,e,f | 64.00 ± 2.65 b,c,d |
| R1G1 | 61.47 ± 0.06 j | 2.1 ± 0.2 a | 3.9 ± 0.8 a,b | 66.00 ± 4.58 i | 81.33 ± 6.03 j,k | 43.33 ± 7.10 a |
| R1G2 | 57.00 ± 0.20 d,e,f | 2.3 ± 0.2 a | 6.0 ± 0.7 c,d | 50.00 ± 5.57 g,h | 63.67 ± 3.22 e,f,g,h | 50.67 ± 4.62 a,b,c |
| R1F2 | 54.30 ± 0.17 a | 2.2 ± 0.2 a | 7.1 ± 0.9 d,e | 50.67 ± 3.79 g,h | 61.00 ± 2.65 d,e,f,g,h | 49.00 ± 6.56 a,b |
| R2 | 59.03 ± 0.06 i | 2.5 ± 0.2 a | 7.4 ± 0.7 d,e,f | 49.00 ± 6.56 g | 69.00 ± 6.25 g,h,i,j | 71.00 ± 7.00 d,e |
| R3 | 58.67 ± 0.06 h,i | 2.5 ± 0.2 a | 8.8 ± 0.1 e,f,g | 42.33 ± 1.53 f,g | 61.33 ± 1.53 e,f,g,h | 81.67 ± 3.06 d,e |
| R4 | 64.50 ± 0.10 l | 5.1 ± 0.4 c,d | 5.9 ± 0.2 b,c,d | 36.67 ± 3.06 e,f | 71.00 ± 1.73 h,i,j | 92.33 ± 3.79 f,g,h |
| R5 | 71.40 ± 0.10 p | 6.1 ± 0.2 d,e,f,g | 2.8 ± 0.1 b | 109.67 ± 4.16 j | 208.67 ± 1.53 m | 80.33 ± 1.53 d,e,f |
| V1I | 66.23 ± 0.35 m | 5.2 ± 0.9 d | 8.9 ± 0.8 e,f,g | 24.33 ± 3.51 b,c,d | 71.33 ± 3.51 h,i,j | 108.33 ± 6.51 h,i,j |
| V1II | 67.07 ± 0.15 n | 6.2 ± 0.4 d,e,f,g | 10.0 ± 0.5 g | 18.00 ± 2.00 a,b,c | 66.67 ± 7.02 f,g,h,i | 119.00 ± 7.94 i,j,k |
| V2I | 59.17 ± 0.15 i | 4.8 ± 0.4 b,c,d | 12.1 ± 0.3 h,i | 18.00 ± 2.65 a,b,c | 55.33 ± 1.53 c,d,e,f | 122.00 ± 3.61 i,j,k,l |
| V2II | 62.17 ± 0.38 j,k | 5.1 ± 0.2 c,d | 9.3 ± 0.1 f,g | 28.33 ± 2.08 c,d,e | 64.67 ± 3.79 e,f,g,h,i | 104.00 ± 2.65 g,h,i |
| V3I | 65.77 ± 0.29 m | 6.3 ± 0.9 d,e,f,g | 13.2 ± 0.8 i | 15.33 ± 1.53 a,b | 55.67 ± 2.52 c,d,e,f | 127.67 ± 2.52 j,k,l,m |
| V3II | 65.70 ± 0.10 m | 7.4 ± 0.3 f,g | 13.2 ± 0.8 i | 11.67 ± 1.16 a | 57.67 ± 2.52 c,d,e,f,g | 133.33 ± 6.66 k,l,m |
| Flour Stream | M-WA (%) | M-C2 (Nm) | M-C3 (Nm) | M-C4 (Nm) | M-C5 (Nm) | M-α | M-β | M-γ |
|---|---|---|---|---|---|---|---|---|
| B2 | 56.97 ± 0.70 d,e | 0.463 ± 0.007 j | 2.542 ± 1.992 m | 1.689 ± 0.031 m | 2.810 ± 0.033 h,i | −0.099 ± 0.016 a | 0.576 ± 0.167 h,i,j,k,l,m | −0.050 ± 0.030 f,g |
| B3 | 57.80 ± 0.11 f | 0.471 ± 0.009 j,k,l | 1.839 ± 0.017 l | 1.627 ± 0.045 k,l | 2.853 ± 0.052 i | −0.084 ± 0.008 a,b | 0.576 ± 0.136 g,h,i,j,k,l,m | −0.056 ± 0.028 f,g |
| B4 | 62.07 ± 0.15 j,k | 0.392 ± 0.006 c | 1.590 ± 0.009 g | 1.373 ± 0.005 e,f | 2.273 ± 0.017 f | −0.117 ± 0.011 a | 0.510 ± 0.018 d,e,f,g,h,i,j,k,l | −0.030 ± 0043 f,g |
| B5 | 65.77 ± 0.50 m,n | 0.365 ± 0.017 b | 1.303 ± 0.013 b | 1.073 ± 0.023 b,c | 1.850 ± 0.045 c | −0.127 ± 0.002 a | 0.368 ± 0.040 a,b,c,d,e,f | −0.042 ± 0.022 f,g |
| C1I | 56.53 ± 0.06 c,d | 0.490 ± 0.003 m,n,o,p,r | 2.038 ± 0.020 o | 1.505 ± 0.046 g,h,i | 3.131 ± 0.048 k,l | −0.102 ± 0.007 a,b | 0.719 ± 0.054 l,m,n,o | −0.207 ± 0.064 a,b,c |
| C1II | 55.90 ± 0.10 a,b,c | 0.507 ± 0.003 r | 2.203 ± 0.013 r | 1.682 ± 0.015 l,m | 3.262 ± 0.014 m,n | −0.095 ± 0.001 a,b | 0.829 ± 0.019 o | −0.177 ± 0.032 a,b,c,d |
| C2 | 56.20 ± 0.10 b,c | 0.491 ± 0.002 n,o,p,r | 2.149 ± 0.014 p | 1.634 ± 0.014 k,l,m | 3.322 ± 0.020 n | −0.082 ± 0.003 a,b | 0.788 ± 0.003 n,o | −0.215 ± 0.090 a,b |
| C3 | 57.50 ± 0.10 e,f | 0.487 ± 0.002 l,m,n,o,p | 2.013 ± 0.008 o | 1.527 ± 0.012 h,i,j | 3.051 ± 0.018 j,k | −0.103 ± 0.005 a,b | 0.693 ± 0.031 k,l,m,n,o | −0.115 ± 0.008 b,c,d,e,f,g |
| C4 | 61.30 ± 0.00 i,j | 0.457 ± 0.005 i,j | 1.693 ± 0.009 h | 1.435 ± 0.025 f,g | 2.489 ± 0.039 g | −0.106 ± 0.006 a | 0.465 ± 0.039 c,d,e,f,g,h,i,j,k | −0.063 ± 0.010 e,f,g |
| C5 | 61.50 ± 0.00 i,j,k | 0.438 ± 0,004 h,i | 1.666 ± 0.004 h | 1.152 ± 0.005 c,d | 2.462 ± 0.035 g | −0.101 ± 0.007 a,b | 0.431 ± 0.006 b,c,d,e,f,g,h,i | −0.101 ± 0.049 c,d,e,f,g |
| C6 | 66.10 ± 0.10 n | 0.435 ± 0.004 g,h | 1.475 ± 0.015 e | 1.137 ± 0.015 c,d | 1.968 ± 0.028 d | −0.085 ± 0.003 a,b | 0.337 ± 0.041 a,b,c,d,e | −0.061 ± 0.011 e,f,g |
| C7I | 67.13 ± 0.06 o,p | 0.355 ± 0.003 b | 1.315 ± 0.012 b | 1.049 ± 0.002 b | 1.785 ± 0.021 c | −0.078 ± 0.005 a,b | 0.250 ± 0.019 a,b,c | −0.053 ± 0.022 f,g |
| C7II | 100.10 ± 0.0 r | 0.697 ± 0.013 s | 1.177 ± 0.029 a | 0.647 ± 0.033 a | 0.880 ± 0.028 a | −0.070 ± 0.039 a,b | 0.202 ± 0.012 a,b | −0.028 ± 0,005 f,g |
| DD1I | 56.50 ± 0.00 c,d | 0.495 ± 0.004 n,o,p,r | 2.013 ± 0.007 o | 1.865 ± 0.023 n | 3.174 ± 0.033 l,m | −0.101 ± 0.004 a,b | 0.720 ± 0.018 l,m,n,o | −0.057 ± 0.026 f,g |
| DD1II | 55.60 ± 0.00 a,b | 0.502 ± 0.005 o,p,r | 2.150 ± 0.003 p | 2.000 ± 0.025 o | 3.349 ± 0.041 n,o | −0.099 ± 0.011 a,b | 0.750 ± 0.060 m,n,o | −0.054 ± 0.030 f,g |
| DD1III | 57.50 ± 0.00 e,f | 0.471 ± 0.003 j,k,l,m | 1.958 ± 0.008 n | 1.425 ± 0.024 f,g | 3.140 ± 0.007 k,l | −0.088 ± 0.009 a,b | 0.671 ± 0.062 j,k,l,m,n,o | −0.170 ± 0.031 a,b,c,d,e |
| R1F1 | 58.20 ± 0.00 f,g | 0.480 ± 0.003 k,l,m,n | 1.968 ± 0.003 n | 1.566 ± 0.060 i,j,k | 2.781 ± 0.032 h,i | −0.030 ± 0.122 b | 0.632 ± 0.025 h,i,j,k,l,m,n,o | −0.079 ± 0.038 d,e,f,g |
| R1G1 | 61.10 ± 0.17 i | 0.465 ± 0.005 j,k | 1.801 ± 0.010 j,k | 1.529 ± 0.017 h,i,j | 2.341 ± 0.021 f | −0.109 ± 0.007 a | 0.473 ± 0.055 c,d,e,f,g,h,i,j,k | −0.070 ± 0011 d,e,f,g |
| R1G2 | 57.50 ± 0.00 e,f | 0.505 ± 0.002 p,r | 2.053 ± 0.007 o | 1.480 ± 0.019 g,h | 3.158 ± 0.015 l | −0.105 ± 0.005 a,b | 0.661 ± 0.008 i,j,k,l,m,n,o | −0.191 ± 0.065 a,b,c |
| R1F2 | 55.07 ± 0.06 a | 0.483 ± 0.009 k,l,m,n,o | 2.253 ± 0.011 s | 1.714 ± 0.039 m | 3.432 ± 0.034 o | −0.100 ± 0.007 a,b | 0.858 ± 0.026 o | −0.277 ± 0.031 a |
| R2 | 58.10 ± 0.20 f,g | 0.482 ± 0.005 k,l,m,n | 1.941 ± 0.014 n | 1.439 ± 0.006 f,g | 2.813 ± 0.047 h,i | −0.111 ± 0.011 a | 0.653 ± 0.005 i,j,k,l,m,n,o | −0.131 ± 0.038 b,c,d,e,f |
| R3 | 58.60 ± 0.00 g | 0.465 ± 0.003 j,k | 1.834 ± 0.012 k,l | 1.332 ± 0.029 e | 2.748 ± 0.046 h | −0.089 ± 0.002 a,b | 0.568 ± 0.027 e,f,g,h,i,j,k,l,m,n | −0.222 ± 0.029 a,b |
| R4 | 63.13 ± 0.00 g | 0.425 ± 0.007 e,f,g,h | 1.541 ± 0.010 f | 1.338 ± 0.010 e | 2.300 ± 0.027 f | −0.103 ± 0.008 a,b | 0.369 ± 0.006 a,b,c,d,e,f | −0.047 ± 0.050 f,g |
| R5 | 67.97 ± 0.21 p | 0.319 ± 0.003 a | 1.159 ± 0.008 a | 0.992 ± 0.003 b | 1.676 ± 0.022 b | −0.069 ± 0.008 a,b | 0.164 ± 0.016 a | −0.025 ± 0.010 f,g |
| V1I | 66.27 ± 0.15 n,o | 0.414 ± 0.004 d,e,f | 1.421 ± 0.002 d | 1.220 ± 0.006 d | 2.120 ± 0.014 e | −0.127 ± 0.013 a | 0.375 ± 0.014 a,b,c,d,e,f,g | −0.027 ± 0.020 f,g |
| V1II | 67.10 ± 0.10 o,p | 0.397 ± 0.005 c,d | 1.377 ± 0.005 c | 1.173 ± 0.006 d | 2.023 ± 0.020 d,e | −0.123 ± 0.005 a | 0.354 ± 0.034 a,b,c,d,e,f | −0.034 ± 0.011 f,g |
| V2I | 59.90 ± 0.10 h | 0.433 ± 0.004 f,g,h | 1.771 ± 0.009 i,j | 1.700 ± 0.012 m | 3.173 ± 0.025 l,m | −0.100 ± 0.012 a,b | 0.277 ± 0.005 a,b,c,d | −0.014 ± 0.016 g |
| V2II | 62.20 ± 0.10 k | 0.418 ± 0.004 e,f,g | 1.739 ± 0.006 i | 1.609 ± 0.014 j,k,l | 2.954 ± 0.043 j | −0.108 ± 0.005 a | 0.571 ± 0.025 f,g,h,i,j,k,l,m,n | −0.035 ± 0.043 f,g |
| V3I | 65.70 ± 0.20 m,n | 0.411 ± 0.005 c,d,e | 1.456 ± 0.006 d,e | 1.333 ± 0.016 e | 2.350 ± 0.020 f | −0.122 ± 0.005 a | 0.401 ± 0.014 b,c,d,e,f,g,h | −0.057 ± 0.024 f,g |
| V3II | 64.90 ± 0.10 m | 0.427 ± 0.003 e,f,g,h | 1.471 ± 0.002 e | 1.385 ± 0.016 e,f | 2.480 ± 0.018 g | −0.113 ± 0.014 a | 0.441 ± 0.020 c,d,e,f,g,h,i,j | −0.042 ± 0.008 f,g |
| Flour Stream | M-C2−C1 (Nm) | M-C3−C2(Nm) | M-C4−C3 (Nm) | M-C5−C4 (Nm) |
|---|---|---|---|---|
| B2 | −0.635 ± 0.013 e,f,g,h | 1.411 ± 0.019 m | −0.186 ± 0.017 f,g,h,i | 1.120 ± 0.035 i |
| B3 | −0.622 ± 0.006 f,i | 1.368 ± 0.017 l | −0.212 ± 0.045 e,f,g | 1.225 ± 0.060 j,k |
| B4 | −0.709 ± 0.013 b,c | 1.198 ± 0.008 i | −0,216 ± 0.005 e,f,g,h | 0.900 ± 0.020 e,f |
| B5 | −0.758 ± 0.018 a | 0.938 ± 0.005 c | −0,229 ± 0.015 e,f | 0.777 ± 0.022 b,c,d |
| C1I | −0.604 ± 0.017 i,j | 1.549 ± 0.018 p | −0.533 ± 0.027 a,b | 1.626 ± 0.036 r,s |
| C1II | −0.615 ± 0.011 f,i,j | 1.696 ± 0.013 s | −0.520 ± 0.028 a,b | 1.580 ± 0.029 p,r |
| C2 | −0.589 ± 0.004 j | 1.657 ± 0.012 r | −0.514 ± 0.024 a,b | 1.687 ± 0.026 s |
| C3 | −0.598 ± 0.005 i,j | 1.527 ± 0.008 p | −0.486 ± 0.018 b | 1.524 ± 0.008 n,p |
| C4 | −0.657 ± 0.018 d,e,g | 1.236 ± 0.004 j | −0.258 ± 0.026 e | 1.054 ± 0.036 g,h,i |
| C5 | −0.646 ± 0.012 e,f,g,h | 1.229 ± 0.006 i,j | −0.514 ± 0.007 a,b | 1.310 ± 0.030 k,l |
| C6 | −0.688 ± 0.005 c,d | 1.040 ± 0.011 f,g | −0.338 ± 0.002 c,d | 0.830 ± 0,014 c,d,e |
| C7I | −0.736 ± 0.010 a,b | 0.960 ± 0.010 c,d | −0.266 ± 0.010 d,e | 0.736 ± 0.019 b,c |
| C7II | −0.394 ± 0.007 k | 0.480 ± 0.021 a | −0.530 ± 0.010 a,b | 0.233 ± 0.023 a |
| DD1I | −0.595 ± 0.010 i,j | 1,518 ± 0.005 o,p | −0.148 ± 0.016 h,i,j,k | 1.309 ± 0.014 k,l |
| DD1II | −0.602 ± 0.011 i,j | 1,648 ± 0.006 r | −0.150 ± 0.028 g,h,i,j,k | 1.349 ± 0.042 l,m |
| DD1III | −0.618 ± 0.019 f,h,i,j | 1.487 ± 0.009 m,o | −0.533 ± 0.030 a,b | 1.715 ± 0.019 s |
| R1F1 | −0.604 ± 0.004 i,j | 1.488 ± 0.002 m,o | −0.402 ± 0.061 c | 1.214 ± 0.028 j,k |
| R1G1 | −0.655 ± 0.015 d,e,g | 1.337 ± 0.012 k,l | −0.273 ± 0.007 d,e | 0.813 ± 0.005 c,d,e |
| R1G2 | −0.622 ± 0.012 f,g,h,i,j | 1.548 ± 0.005 p | −0.573 ± 0.014 a | 1.678 ± 0.032 s |
| R1F2 | −0.612 ± 0.015 f,i,j | 1.769 ± 0.006 t | −0.538 ± 0.030 a,b | 1.718 ± 0.006 s |
| R2 | −0.654 ± 0.005 d,e,g,h | 1.459 ± 0.012 n | −0.502 ± 0.013 a,b | 1.374 ± 0.042 l,m |
| R3 | −0.642 ± 0.005 e,f,g,h | 1.369 ± 0.009 l | −0.501 ± 0.021 a,b | 1.415 ± 0.027 m,n |
| R4 | −0.699 ± 0.013 c | 1.116 ± 0.003 h | −0.204 ± 0.008 e,f,g,h | 0.962 ± 0.018 f,g |
| R5 | −0.757 ± 0.013 b | 0.840 ± 0.006 b | −0.166 ± 0.006 f,g,h,i,j | 0.684 ± 0.019 b |
| V1I | −0.713 ± 0.005 b,c | 1.007 ± 0.002 e,f | −0.201 ± 0.008 e,f,g,h,i | 0.900 ± 0.020 e,f |
| V1II | −0.689 ± 0.009 c,d | 0.980 ± 0.009 d,e | −0.204 ± 0.008 e,f,g,h | 0.849 ± 0.020 d,e |
| V2I | −0.656 ± 0.019 d,e,g | 1.338 ± 0.007 k,l | −0.070 ± 0.004 l | 1.473 ± 0.014 n,o |
| v2II | −0.703 ± 0.009 b,c | 1.321 ± 0.005 k | −0.130 ± 0.009 i,j,k,l | 1.345 ± 0.042 l,m |
| V3I | −0.656 ± 0.008 d,e,g | 1.045 ± 0.001 g | −0.123 ± 0.016 j,k,l | 1.017 ± 0.028 g,h |
| V3II | −0.659 ± 0.006 d,e | 1.044 ± 0.004 g | −0.086 ± 0.015 k,l | 1.095 ± 0.002 h,i |
| Flour Stream | WaSRC (%) | SuSRC (%) | LaSRC (%) | ScSRC (%) | GPI (-) |
|---|---|---|---|---|---|
| B2 | 62.642 ± 0.593 a,b | 115.695 ± 1.534 e,f,g,h,i | 161.558 ± 1.010 n | 79.567 ± 0.704 d | 0.827 ± 0.007 m |
| B3 | 63.017 ± 0.58 a,b | 120.274 ± 1,153 i,j,k,l | 180.035 ± 1.701 s | 78.150 ± 0.692 c | 0.907 ± 0.007 o |
| B4 | 71.675 ± 0.574 g | 119.569 ± 0.640 h,i,j,k,l | 134.362 ± 0.501 h | 94.163 ± 0.304 j | 0.629 ± 0.005 e |
| B5 | 75.508 ± 0.302 h | 124.692 ± 1370 k,l,m | 129.109 ± 0.412 g | 97.265 ± 0.412 k | 0.582 ± 0.003 d |
| C1I | 65.164 ± 0.523 c,d | 114.885 ± 0.570 d,e,f,g,h,i,j | 162.432 ± 0.114 n,o | 86.106 ± 0.198 e,f,g | 0.808 ± 0.002 l |
| C1II | 63.932 ± 0.228 a,b,c | 94.796 ± 0.747 a | 128.096 ± 0.228 g | 78.541 ± 0.228 c,d | 0.739 ± 0.004 i |
| C2 | 63.009 ± 0.302 a,b | 104.997 ± 1.951 b | 147.579 ± 0.685 k | 80.345 ± 0.198 d | 0.796 ± 0.011 k,l |
| C3 | 64.177 ± 0.302 b,c | 114.794 ± 2.205 d,e,f,g,h,i,j | 154.734 ± 0.457 m | 84.806 ± 0.498 e | 0.775 ± 0.008 j,k |
| C4 | 75.233 ± 0.115 h | 121.145 ± 0,574 h,i,j,k,l | 143.074 ± 1.033 j | 100.673 ± 0.500 l | 0.645 ± 0.005 e,f |
| C5 | 73.052 ± 0.496 g | 114.671 ± 0.911 d,e,f,g,h,i | 138.012 ± 1.532 i | 102.573 ± 0.711 m | 0.635 ± 0.006 e |
| C6 | 86.747 ± 0.856 j | 113.323 ± 1.262 c,d,e,f,g,h | 100.100 ± 0.340 b | 125.105 ± 0.631 p | 0.420 ± 0.003 a |
| C7I | 87.798 ± 0.490 j | 107.871 ± 1.369 b,c,d | 114.951 ± 0.585 d,e | 136.063 ± 0.981 r | 0.471 ± 0.002 b |
| C7II | 159.775 ± 2.833 k | 178.502 ± 12.648 n | 163.080 ± 1.323 n,o | 193.406 ± 0.683 t | 0.439 ± 0.014 a |
| DD1I | 62.096 ± 0.307 a,b | 110.142 ± 1.686 b,c,d,e,f,g | 149.610 ± 0.307 k,l | 77.642 ± 0.580 b,c | 0.797 ± 0.008 l |
| DD1II | 61.867 ± 0.464 a | 105.465 ± 1.508 b,c | 128.637 ± 0.876 g | 76.065 ± 0.307 a,b | 0.709 ± 0.002 h |
| DD1III | 62.331 ± 0.463 a,b | 115.751 ± 1.219 d,e,f,g,h,i,j | 166.634 ± 0.530 p,r | 77.422 ± 0.116 b,c | 0.863 ± 0.008 n |
| R1F1 | 69.124 ± 0.115 f | 120.538 ± 1.966 h,i,j,k,l | 164.314 ± 1.852 o,p | 95.562 ± 0.230 j,k | 0.760 ± 0.013 j |
| R1G1 | 78.190 ± 0.576 i | 117.884 ± 0.984 g,h,i,j,k,l | 169.014 ± 1.282 r | 113.562 ± 0.461 o | 0.730 ± 0.004 i |
| R1G2 | 66.829 ± 0.230 d,e | 109.832 ± 0.305 b,c,d,e,f | 152.437 ± 1.055 l,m | 87.699 ± 0.115 f,g,h | 0.772 ± 0.006 j |
| R1F2 | 62.023 ± 0.691 a | 92.951 ± 0.399 a | 121.951 ± 0.610 f | 74.394 ± 0.399 a | 0.729 ± 0.004 h,i |
| R2 | 68.044 ± 0.115 e,f | 109.922 ± 1.490 b,c,d,e,f | 131.159 ± 0.413 g,h | 87.958 ± 0.198 g,h | 0.663 ± 0.005 f,g |
| R3 | 69.252 ± 0.527 f | 121.621 ± 0.996 i,j,k,l | 154.675 ± 0.690 m | 90.160 ± 0.527 g,h | 0.730 ± 0.006 i |
| R4 | 77917 ± 0.198 i | 122.578 ± 0.457 j,k,l,m | 118.158 ± 0.749 e | 103.645 ± 0.198 m | 0.522 ± 0.004 c |
| R5 | 90.464 ± 0.297 k | 129.955 ± 0.971 m | 117.460 ± 0.624 e | 148.082 ± 1.866 s | 0.422 ± 0.002 a |
| V1I | 86.505 ± 0.606 j | 109.513 ± 0.499 b,c,d,e | 146.734 ± 1.301 k | 110.835 ± 0.525 n | 0.666 ± 0.006 f,g |
| V1II | 87.365 ± 0.458 i | 110.637 ± 0.595 b,c,d,e,f,g | 151.362 ± 1.196 l | 114.008 ± 0.344 o | 0.674 ± 0.004 g |
| V2I | 66.753 ± 0.231 d,e | 117.492 ± 0.924 f,g,h,i,j,k | 113.692 ± 1.677 d | 79.888 ± 0.115 d | 0.576 ± 0.011 d |
| V2II | 72.040 ± 0.528 g | 115.615 ± 0.502 d,e,f,g,h,i,j | 96.788 ± 0.599 a | 86.011 ± 0.200 e,f | 0.480 ± 0.005 b |
| V3I | 79.754 ± 0.499 i | 125.271 ± 0.794 k,l,m | 106.614 ± 0.198 c | 99.271 ± 0.397 l | 0.475 ± 0.002 b |
| V3II | 73.711 ± 0.303 g,h | 126.770 ± 1.952 l,m | 116.065 ± 1.300 d,e | 88.975 ± 0.499 h,i | 0.538 ± 0.004 c |
| Flour Stream | T-NSP (%) | S-NSP (%) | I-NSP (%) | T-AX (%) | I-AX (%) | S-AX (%) |
|---|---|---|---|---|---|---|
| B2 | 3.80 ± 0.00 i,j,k | 1.72 ± 0.05 g,h,i | 2.08 ± 0.04 g,h | 2.05 ± 0.01 g,h,i,j | 1.30 ± 0.02 f,g | 0.75 ± 0.03 e,f,g,h,i |
| B3 | 2.70 ± 0.00 a | 1.29 ± 0.04 a | 1.41 ± 0.04 a | 1.48 ± 0.00 a | 0.93 ± 0.04 a | 0.55 ± 0.04 a |
| B4 | 4.17 ± 0.12 n,o | 1.85 ± 0.05 i,j | 2.31 ± 0.07 i | 2.42 ± 0.05 m,n | 1.61 ± 0.04 j,k | 0.81 ± 0.01 h,i,j |
| B5 | 3.89 ± 0.04 j,k,l,m | 1.57 ± 0.03 d,e,f | 2.32 ± 0.07 i | 2.29 ± 0.07 l,m,n | 1.61 ± 0.07 i,j,k | 0.68 ± 0.00 c,d,e,f |
| C1I | 3.09 ± 0.02 b,c | 1.57 ± 0.01 d,e,f | 1.52 ± 0.03 a,b | 1.71 ± 0.08 b,c | 1.01 ± 0.08 a,b,c | 0.71 ± 0.01 d,e,f,g |
| C1II | 4.12 ± 0.08 m,n | 1.41 ± 0.04 a,b,c | 2.70 ± 0.11 j,k | 2.23 ± 0.03 k,l,m | 1.58 ± 0.05 i,j | 0.65 ± 0.02 b,c,d,e |
| C2 | 3.35 ± 0.03 d,e,f | 1.55 ± 0.02 d,e,f | 1.80 ± 0.01 c,d,e | 1.86 ± 0.04 b,c,d,e | 1.13 ± 0.01 c,d,e | 0.74 ± 0.05 e,f,g,h,i |
| C3 | 3.29 ± 0.08 c,d,e | 1.63 ± 0.04 e,f,g | 1.66 ± 0.04 b,c | 1.79 ± 0.01 b,c,d | 1.08 ± 0.02 b,c,d | 0.71 ± 0.01 d,e,f,g |
| C4 | 3.64 ± 0.09 g,h,i | 1.71 ± 0.03 g,h | 1.93 ± 0.06 e,f,g | 1.95 ± 0.05 d,e,f,g,h,i | 1.25 ± 0.08 e,f,g | 0.70 ± 0.03 d,e,f,g |
| C5 | 3.66 ± 0.02 g,h,i,j | 1.71 ± 0.03 g,h | 1.95 ± 0.02 e,f,g | 2.03 ± 0.05 f,g,h,i,j | 1.27 ± 0.08 e,f,g | 0.77 ± 0.03 f,g,h,i,j |
| C6 | 6.47 ± 0.14 t | 2.07 ± 0.07 k | 4.40 ± 0.07 m | 3.75 ± 0.11 r | 2.83 ± 0.09 n | 0.91 ± 0.02 k,l |
| C7I | 7.98 ± 0.04 v | 2.15 ± 0.05 k | 5.83 ± 0.09 o | 4.63 ± 0.06 t | 3.68 ± 0.03 p | 0.94 ± 0.03 k,l |
| C7II | 24.70 ± 0.16 w | 2.48 ± 0.03 l | 22.22 ± 0.19 p | 16.45 ± 0.10 u | 15.07 ± 0.09 r | 1.37 ± 0.01 n |
| DD1I | 4.66 ± 0.12 r | 1.82 ± 0.06 h,i,j | 2.84 ± 0.06 k | 2.61 ± 0.00 o | 1.84 ± 0.04 l | 0.77 ± 0.03 f,g,h,i,j |
| DD1II | 3.74 ± 0.06 h,i,j,k | 1.65 ± 0.01 e,f,g | 2.08 ± 0.05 g,h | 2.19 ± 0.04 j,k,l | 1.34 ± 0.01 g,h | 0.85 ± 0.03 j,k |
| DD1III | 3.28 ± 0.12 c,d,e | 1.876 ± 0.066 j | 1.41 ± 0.05 a | 1.90 ± 0.10 d,e,f,g | 0.95 ± 0.05 a,b | 0.95 ± 0.05 l |
| R1F1 | 4.39 ± 0.06 o,p | 2.59 ± 0.02 l | 1.807 ± 0.035 c,d,e | 2.37 ± 0.07 m,n | 1.17 ± 0.04 d,e,f | 1.20 ± 0.04 m |
| R1G1 | 2.95 ± 0.01 b | 1.40 ± 0.05 a,b,c | 1.55 ± 0.03 a,b | 1.51 ± 0.01 a | 0.94 ± 0.03 a,b | 0.56 ± 0.02 a,b |
| R1G2 | 3.18 ± 0.13 b,c,d | 1.60 ± 0.06 e,f,g | 1.58 ± 0.06 a,b | 1.69 ± 0.05 b | 1.00 ± 0.03 a,b,c | 0.69 ± 0.02 c,d,e,f,g |
| R1F2 | 3.67 ± 0.01 g,h,i,j | 1.46 ± 0.01 b,c,d | 2.21 ± 0.00 h,i | 1.92 ± 0.04 d,e,f,g,h | 1.30 ± 0.01 f,g | 0.61 ± 0.03 a,b,c,d |
| R2 | 4.04 ± 0.07 l,m,n | 1.68 ± 0.03 f,g,h | 2.36 ± 0.04 i | 2.28 ± 0.01 l,m,n | 1.47 ± 0.03 h,i | 0.81 ± 0.04 i,j |
| R3 | 3.26 ± 0.06 c,d,e | 1.59 ± 0.04 d,e,f,g | 1.68 ± 0.02 b,c,d | 1.87 ± 0.06 c,d,e,f | 1.09 ± 0.01 b,c,d | 0.78 ± 0.05 g,h,i,j |
| R4 | 3.96 ± 0.00 k,l,m,n | 1.33 ± 0.03 a,b | 2.63 ± 0.03 j | 2.32 ± 0.03 l,m,n | 1.74 ± 0.04 k,l | 0.59 ± 0.01 a,b |
| R5 | 6.96 ± 0.06 u | 1.52 ± 0.02 c,d,e | 5.44 ± 0.04 n | 4.09 ± 0.09 s | 3.48 ± 0.03 o | 0.61 ± 0.05 a,b,c,d |
| V1I | 3.82 ± 0.02 i,j,k,l | 1.93 ± 0.00 j | 1.89 ± 0.02 d,e,f,g | 2.08 ± 0.03 h,i,j,k | 1.30 ± 0.04 f,g | 0.78 ± 0.01 g,h,i,j |
| V1II | 3.452 ± 0.081 e,f,g | 1.59 ± 0.05 e,f,g | 1.86 ± 0.03 c,d,e,f | 1.99 ± 0.03 e,f,g,h,i | 1.28 ± 0.01 f,g | 0.71 ± 0.04 e,f,g,h |
| V2I | 3.56 ± 0.06 f,g,h | 1.52 ± 0.01 c,d,e | 2.04 ± 0.08 f,g,h | 1.89 ± 0.01 d,e,f,g | 1.29 ± 0.02 f,g | 0.60 ± 0.01 a,b,c |
| V2II | 5.53 ± 0.01 s | 1.72 ± 0.07 g,h,i | 3.81 ± 0.07 l | 3.23 ± 0.03 p | 2.49 ± 0.00 m | 0.74 ± 0.03 e,f,g,h,i |
| V3I | 4.43 ± 0.01 s | 1.72 ± 0.07 g,h,i | 2.71 ± 0.15 j,k | 2.44 ± 0.00 n,o | 1.74 ± 0.04 k,l | 0.70 ± 0.04 d,e,f,g |
| V3II | 3.69 ± 0.01 h,i,j | 1.36 ± 0.00 a,b | 2.33 ± 0.01 i | 2.11 ± 0.00 i,j,k | 1.53 ± 0.00 i,j | 0.57 ± 0.00 a,b |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lewko, P.; Wójtowicz, A.; Gancarz, M. Distribution of Arabinoxylans and Their Relationship with Physiochemical and Rheological Properties in Wheat Flour Mill Streams as an Effective Way to Predict Flour Functionality. Appl. Sci. 2023, 13, 5458. https://doi.org/10.3390/app13095458
Lewko P, Wójtowicz A, Gancarz M. Distribution of Arabinoxylans and Their Relationship with Physiochemical and Rheological Properties in Wheat Flour Mill Streams as an Effective Way to Predict Flour Functionality. Applied Sciences. 2023; 13(9):5458. https://doi.org/10.3390/app13095458
Chicago/Turabian StyleLewko, Piotr, Agnieszka Wójtowicz, and Marek Gancarz. 2023. "Distribution of Arabinoxylans and Their Relationship with Physiochemical and Rheological Properties in Wheat Flour Mill Streams as an Effective Way to Predict Flour Functionality" Applied Sciences 13, no. 9: 5458. https://doi.org/10.3390/app13095458
APA StyleLewko, P., Wójtowicz, A., & Gancarz, M. (2023). Distribution of Arabinoxylans and Their Relationship with Physiochemical and Rheological Properties in Wheat Flour Mill Streams as an Effective Way to Predict Flour Functionality. Applied Sciences, 13(9), 5458. https://doi.org/10.3390/app13095458







