Abstract
Neuromyelitis optica (NMO) is a rare autoimmune inflammatory disorder affecting the central nervous system (CNS), specifically the optic nerve and the spinal cord, with severe clinical manifestations, including optic neuritis (ON) and transverse myelitis. Initially, NMO was wrongly understood as a condition related to multiple sclerosis (MS), due to a few similar clinical and radiological features, until the discovery of the AQP4 antibody (NMO-IgG/AQP4-ab). Various etiological factors, such as genetic-environmental factors, medication, low levels of vitamins, and others, contribute to the initiation of NMO pathogenesis. The autoantibodies against AQP4 target the AQP4 channel at the blood–brain barrier (BBB) of the astrocyte end feet, which leads to high permeability or leakage of the BBB that causes more influx of AQP4-antibodies into the cerebrospinal fluid (CSF) of NMO patients. The binding of AQP4-IgG onto the AQP4 extracellular epitopes initiates astrocyte damage through complement-dependent cytotoxicity (CDC) and antibody-dependent cellular cytotoxicity (ADCC). Thus, a membrane attack complex is formed due to complement cascade activation; the membrane attack complex targets the AQP4 channels in the astrocytes, leading to astrocyte cell damage, demyelination of neurons and oligodendrocytes, and neuroinflammation. The treatment of NMOSD could improve relapse symptoms, restore neurological functions, and alleviate immunosuppression. Corticosteroids, apheresis therapies, immunosuppressive drugs, and B cell inactivating and complement cascade blocking agents have been used to treat NMOSD. This review intends to provide all possible recent studies related to molecular mechanisms, clinical perspectives, and treatment methodologies of the disease, particularly focusing on recent developments in clinical criteria and therapeutic formulations.
1. Introduction
Autoimmune diseases (ADs) are chronic conditions initiated due to loss of immunological tolerance or immune responses toward self-antigens. ADs are a heterogeneous group of disorders, having multifactorial origins and affecting specific organs or multiple organs of the body [1,2]. Genetics, drugs, immune responses initiated by environmental agents and microbiota, air pollution, stress, low levels of vitamin D, medications, vaccination, sex hormones, and the interplay between gene-environment context are the possible risk factors of ADs, in addition to cigarette smoking, alcohol, and coffee consumption [1,3].
Neurological autoimmune disease (NAD) is an organ-specific AD that particularly affects the central nervous system (CNS) and peripheral nervous system (PNS). Certain autoimmune disorders, such as multiple sclerosis (MS), acute disseminated encephalomyelitis (ADEM), anti-NMDAR encephalitis, longitudinally extensive transverse myelitis, Guillain-Barré Syndrome (GBS), Myasthenia Gravis (MG), Sjogren’s syndrome (SS), immunoglobulin G4 related diseases, limbic encephalitis, opsoclonus-myoclonus, Miller-Fisher syndrome, Stiff-person syndrome, Morvan syndrome, sensory ganglionopathy, Lambert-Eaton myasthenic syndrome, and neuromyelitis optica (NMO), are categorized as NADs. Among these NADs, MS is the most prevalent worldwide, while other NADs are less common [4]. NMO is one of the immune-mediated inflammatory disorders of the CNS. NMO predominantly causes astrocyte loss and dysfunction resulting in secondary myelination and neurodegeneration, particularly affecting the optical nerves and spinal cord with concurrent inflammation and demyelination of ON and the spinal cord (myelitis) [5]. Astrocytic lesions, caused by the binding of IgG to aquaporin 4 (AQP4) channels, and subsequent deposition of complement factors resulting in lytic reactions in astrocytes lead to astrocyte damage. Human astrocytes are more complex than rodent astrocytes. Detailed immunohistochemical, molecular, and histopathological studies of astrocytes in optic nerves, brain, and spinal cord are obligatory in studying NMO patients to understand the neuropsychological symptoms in NMO cases [6].
NMO was expanded to NMO spectrum disorder (NMOSD) because of its varied clinical phenotypes. NMOSD is characterized by the activation of the complement cascade, granulocyte, eosinophil and lymphocyte infiltration, oligodendrocyte, and astrocyte injury, demyelination, and neuronal loss [7]. Most NMOSD patients exhibit acute inflammatory responses activated by autoantibodies of IgG against AQP4 in the optic nerve and spinal cord [8], CNS attack relapses, ON, and encephalitis of the diencephalon, area postrema, and brain stem [9]. Classically, about 30% of NMOSD patients have been observed with severe neurological disability, ON, worse clinical outcomes, and the presence of oligoclonal bands in the CSF [10]. This review focuses on the history, clinical perspectives, treatment, and possible molecular mechanisms of NMOSD.
2. Methods
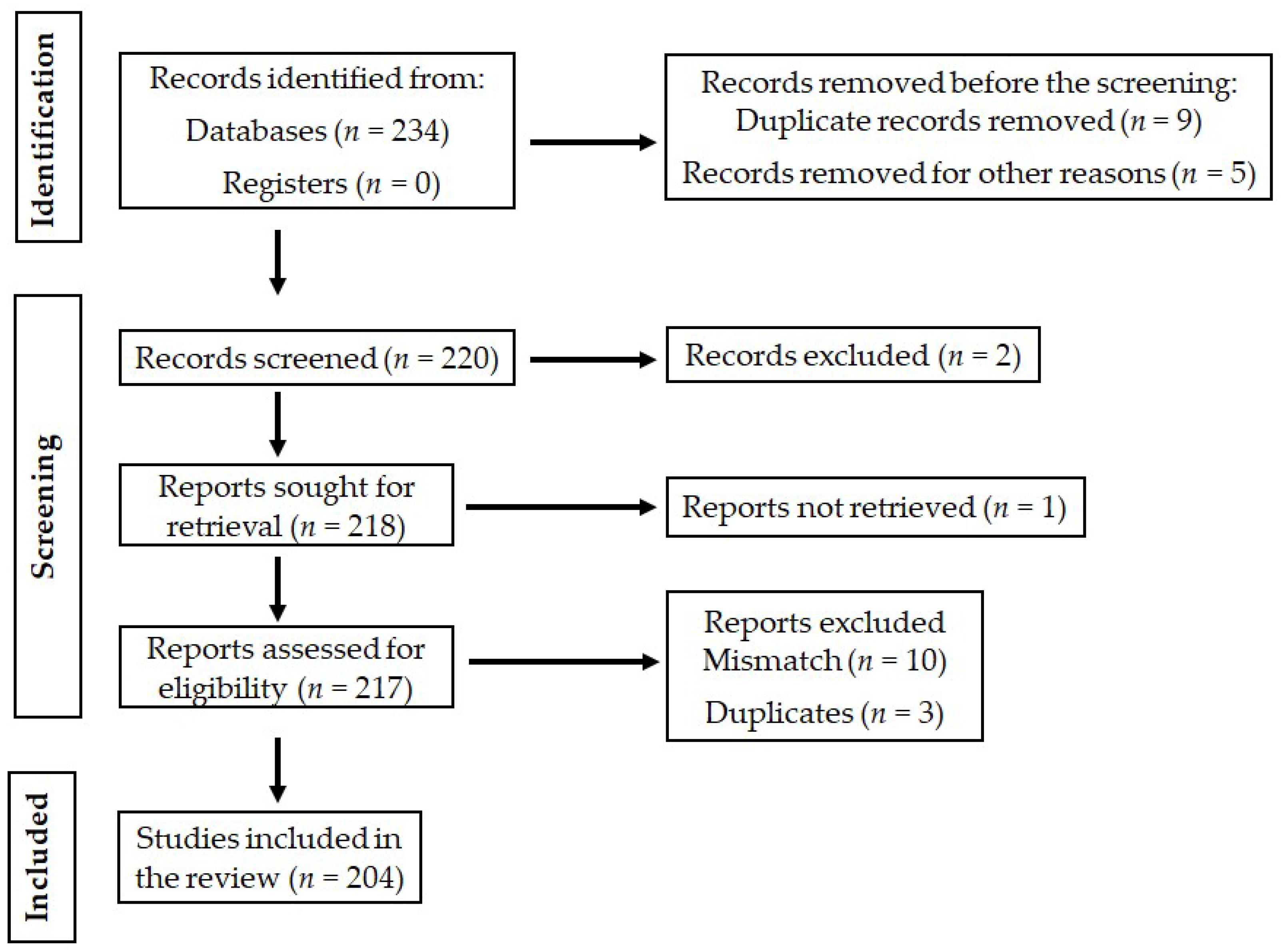
The descriptive review was conducted to evaluate the molecular pathogenesis of NMOSD and immunological responses, symptoms, and diagnoses, as well as the treatment methodologies of the disease. A wide range of keywords was searched, including “neurological autoimmune diseases”, neuromyelitis optica”, “neuromyelitis optica symptoms and diagnosis”, “molecular mechanism of neuromyelitis optica”, “neuromyelitis optica treatment methods”, “optic neuritis”, “transverse myelitis”, “longitudinally extensive transverse myelitis”, “multiple sclerosis”, “astrocytopathy”, “Sjogrens’s syndrome”, “Systemic Lupus Erythematosus”, “autoimmune neuropathies”, “autoimmune encephalitis”, “autoimmune limbic encephalitis”, and “acute disseminated encephalomyelitis”. All English research articles, reviews, and abstracts were searched on the Scopus, PubMed Central, Medline, and Google Scholar platforms. Studies related to our study of interest were inspected and selected for the preparation of the manuscript. The PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) chart explains the selection criteria of the collected articles (Figure 1).
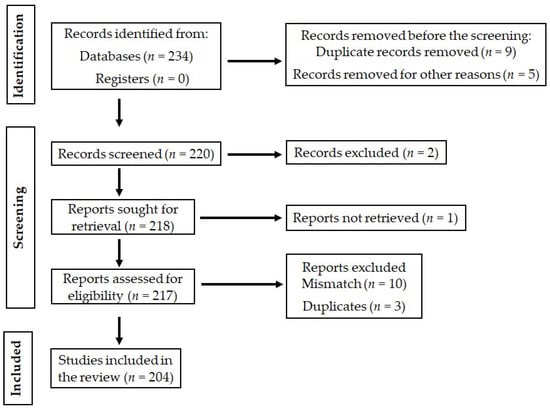
Figure 1.
Schematic representation of the PRISMA chart explaining the selection of studies.
3. History of Neuromyelitis Optica (NMO)
The term ‘neuromyelitis optica’ was first used by Eugène Devic and Fernand Gault in 1894 while working with patients who suffered bilateral ON and myelitis. NMO shares some clinical features with MS; therefore, NMO was long considered another clinical form of MS [11]. The term ‘neuromyelitis optica acuta’, translated from the French term ‘neuromyélite optique aiguë,’ was first used by Eugène Devic in 1894. Hence, NMO is also known as Devic syndrome [11].
Over the next decades, different criteria were discussed with regard to the clinical feature of NMO, and the term NMO was changed over the years. NMO has also been referred to as “autoimmune AQP4 channelopathy” or “autoimmune AQP4 disease” [12], as well as AQP4-ab associated encephalomyelitis [11]. Initially, NMO was associated with the NMO-IgG antibody; later, NMO-IgG became AQP4-IgG, which binds with AQP4 and is identified as the target antigen. AQP4-IgG is recognized as a potential biomarker of NMO pathogenesis [13,14]. Identifying AQP4 seronegative cases with NMO pathogenesis led to the discovery of another antigen called MOG [15]. Alternatively, the AQP4 seronegative NMO phenotype harbors the MOG-IgG antibodies and exhibits different disorders called MOG-associated disorders (MOGAD) [16]. Patients with AQP4-IgG seronegative showed the clinical phenotype of MOGAD with ON, encephalitis with brain lesions, or myelitis, cumulatively termed as MOG-IgG-associated ON, Encephalitis, and Myelitis [15].
Wingerchuk and his team have described how the presence of NMO-IgG differentiates the neuromyelitis optica from multiple sclerosis. The discovery of a new class of NMO-IgG and its pathogenicity determines knowledge about neuromyelitis optica pathogenesis in treatment trials [12]. Later, the international panel for NMO diagnosis, which was assembled in 2015, revised the diagnostic criteria for NMO. The panel described the core clinical characteristics required for NMO patients with or without AQP4-IgG. In addition to serological testing, the panel developed additional clinical criteria with neuroimaging techniques to diagnose NMO [17].
4. Etiology of NMO
The genetic risks for NMO are the class I and II alleles of the human leukocyte antigen (HLA). Demyelinated lesions found in the cervical spinal cord showed that these lesions were associated with HLA alleles DRB1 and DQB1 [18]. HLA plays an important role in genetic risk for NMO, MS, and other demyelinating diseases, facilitating understanding of its pathogenesis and differential diagnosis [19]. The contribution of HLA alleles in NMO pathogenesis and the profile of HLA in NMOSD patients have been detailed by Gontika and Anagnostouli (2014) [19]. Different specific alleles were present in the different populations of people from various countries; such as the HLA-DRB1*03 allele marked for NMO in French, Brazilian, and Afro-Caribbean populations [20,21], the HLA-DRB1*05:01 allele for NMO in African-American, Latino, and Japanese populations [19,22,23], and DRB1*1602 for NMO in Chinese populations [24]. The single nucleotide polymorphism (SNP) at several sites of the gene AQP4 is the risk factor for NMO in the Chinese population [25]. In NMO AQP4 seropositive cases, the interleukin 17 (IL-17) gene polymorphism was found compared to healthy controls. Increased production of inflammatory cytokines IL-17, IL-6, and IL-32 in the plasma was found [26]. Polymorphism in the allele programmed death-1 receptor is highly present in NMO conditions [27]. An expression study comparing NMO cases and normal controls showed more than 700 mRNAs differentially expressed in NMO subjects. The genes responsible for IL-23, INF-γ, NFκB, and T cell-related genes showed changes in NMO subjects [28]. Small RNAseq analysis from the whole blood of rituximab-responsive NMO patients before and after six months following treatment and healthy controls showed that 32 and 14 miRNAs were downregulated and upregulated, respectively, after rituximab treatment. In comparison, untreated NMO patients and healthy controls showed that 17 and 25 miRNAs were upregulated and downregulated, respectively. Thus, miRNAs could be a potential circulating biomarker for NMO [29].
Understanding the complexity of NMO requires knowledge about the biomarkers. Proteomic and metabolomic evaluations were used to avoid misdiagnosis and to differentiate NMOSD from other diseases such as multiple sclerosis, longitudinally extensive transverse myelitis (LETM), and relapsing-remitting multiple sclerosis (RRMS). The discovery of NMOSD biomarkers would support diagnosing and differentiating seropositive and seronegative AQP4-ab and MOG-Ab NMO cases and other related disorders [30]. The involvement of T follicular helper (Tfh) cells and chemokine CXC receptor 5 (CXCR5) in reducing the disease progression and reoccurrence of NMOSD was studied. The function of Tfh cells on B cell mediated humoral immunity is modulated by the serum C-X-C motif ligand 13 (CXCL-13). The correlation between gut microbiome-CXCL-13 and Tfh could facilitate understanding NMOSD [31,32]. NMO patients with high recurrence rates showed higher levels of CXCR5, and NMO patients with low annual relapse rates showed reduced CXCL13 in their serum and CSF. The results indicated that CXCL13 and CXCR5 in the CSF are directly related to NMO disease-related nerve defects [31]. The effects of gut microbiota and its metabolites in causing NMO pathology are described by certain studies [33,34].
AQP4 is an osmotically driven water channel present in the glial cells, responsible for the extracellular osmolality stabilisation, glutamate homeostasis, and balance of the metabolic load in the blood–brain barrier (BBB) [35]; thus, regulating the CNS in response to injury could influence synaptic plasticity as well as behaviour [36]. AQP4-ab causes inflammation and complement-dependent immune cytotoxicity [37]. AQP4 stabilizes the extracellular osmolality during neuronal activity, thus maintaining the BBB’s glutamate homeostasis, energy, and metabolic loads [35]. AQP4-NMOSD pathogenesis involves loss of APQ4 in astrocytes, infiltration of neutrophils, macrophage, and microglial cells, eosinophil degranulation, and loss of myelin in neurons [38].
NMO selectively insults the CNS tissue more than the other AQP4-expressing tissues because the aggregation of AQP4 is greater in the spinal cord [39]. The two isoforms AQP4 M1 and M23 play a prudent role in NMOSD pathogenesis; their expression and distribution in the CNS determine the NMOSD stages in patients of different age groups [40,41]. The M23 isoform activates the immune complement pathway and creates orthogonal arrays of particles (OAPs), which initiate NMOSD pathogenesis [42]. Matiello and his team evaluated the genetic analysis of AQP4 by evaluating AQP4 single nucleotide polymorphisms and their relation to NMO. Their results showed that two missense allelic mutations at the Arg 19 site of AQP4 produce changes in AQP4 proteins that lead to NMO [43]. AQP4 M1 proteins can be internalized completely, whereas M23 resists internalization, activating the immune complement system more competently than M1 [44]. Another important NMO pathoetiology is that brain lesions occur due to macrophage-mediated inflammatory responses because of the high expression of NFκB and Blimp-1 genes [45].
Myelin oligodendrocyte glycoprotein (MOG) is located on the outer surface of oligodendrocytes and myelin sheaths. Autoantibodies targeting myelin oligodendrocyte glycoprotein (MOG-IgG) were observed in AQP4 seronegative NMO patients [46]. The MOG-Ig-related NMO showed different features than AQP4-Ig cases in parameters such as sex distribution and disease outcomes [47]. MOG-Ig-related pathology is characterised by perivenous demyelination, gliosis in the white and grey matter, demyelinated lesions in intracortical regions, and no change in AQP4 expression, whereas AQP4-Ig pathology is characterised by a loss of AQP4 protein, astrocyte dystrophy, and the absence of cortical demyelination [48]. In AQP4-IgG NMOSD, serum antibodies affect the astrocytes and cause astrocytopathy. In MOG-IgG NMOSD, the antibodies affect the oligodendrocytes of myelin sheaths and produce demyelinating diseases [49]. Thus, the AQP4-IgG and MOG-IgG NMOSD are two different entities with distinct pathophysiological patterns.
5. Symptoms of NMOSD
Generally, NMOSD and MS show proximity and are misdiagnosed often, as their symptoms look similar. CNS lesions, concomitant attacks with breaks, vision impairment, trouble walking, fatigue, spasticity, bladder dysfunction, anxiety, and depression are seen in both NMOSD and MS. NMOSD includes specific pathological and immunological variations, with more severe attacks than MS conditions. NMOSD affects the optic nerves and spinal cord, while MS affects other brain regions [50,51]. Symptoms found in NMOSD patients include neuropathic pain [52], neuropathic pruritis [53], paroxysmal tonic spasms [54], lesions in the brain that can cause nausea, vomiting and hiccups [55], and other clinical features such as hypersomnolence and narcolepsy [56]. The cognitive impairment found in NMOSD patients is similar to that found in MS patients; however, in advanced MS conditions, cognitive dysfunction is highly common and is not prevalent in NMOSD [51]. Major cortical pathology and lifetime depression are more dominant in NMOSD than MS [57]. NMOSD patients showed disturbances in processing speed, executing function, attention, memory, and verbal frequency [58]. Recurrent attacks may sometimes result in severe ON conditions, transverse myelitis, or even permanent disability or complete blindness tetraplegia and could be fatal in some cases [58].
5.1. Imaging and Diagnosis of NMOSD
The diagnosis of NMOSD predominantly relies on identifying AQP4-IgG in the patient’s serum. According to the international consensus diagnostic criteria of NMOSD, seropositive AQP4 IgG patients exhibit at least one of the clinical characteristics, such as ON or area postrema syndrome and the presence of acute myelitis, brainstem syndrome, acute diencephalic clinical syndrome with lesions, or symptomatic cerebral syndrome with lesions [17].
Diagnostic criteria for seronegative AQP4-IgG patients are stringent with neuroimaging analysis. Additionally, the patients must have one of the previously mentioned seropositive clinical characteristics such as ON and transverse myelitis with area postrema syndrome [17]. The AQP4 seronegative patients were further diagnosed using cell-based assays repeatedly more than two times within 6 to 9 months [59]. Compared with MS, the relapses in NMOSD are more severe; ON induces inflammation, pain, and visual loss. The spinal cord damage triggers inflammation, which causes fatigue, neuropathic pain, bowel and bladder dysfunction [60], alterations in consciousness, respiratory failure, paraplegia, and even death [9].
Diagnosing and differentiating MS and NMOSD is difficult for neurologists in AQP4-IgG seronegative cases. A differential diagnosis method is required to facilitate accurate diagnosis of autoimmune CNS disorders. Imaging the brain, optic nerve, spinal cord, and retina could favour the understanding of the disease progression and necessary measures for treating autoimmune CNS disorders. Magnetic resonance imaging (MRI) and retinal optical coherence tomography (OCT) are non-invasive imaging techniques that play an important role in diagnosing and differentiating MS and NMOSD. OCT helps visualize the retinal neuronal axons, retinal ganglion cells, and neuroaxonal retinal damages [61]. Retinal OCT, brain, spinal cord, and optic nerve MRI methods are useful in differential diagnosis, screening, and monitoring the degree of CNS damage [62]. MRI studies in patients with ON caused by NMOSD and MS revealed that NMO patients showed bilateral optic nerve and chiasmal enhancement [63]. Crane et al. developed a live cell and single molecule imaging strategy for visualizing AQP4 diffusion and formation of OAPs by comparing the AQP4 transfected cells and AQP4 knockout mice [64].
Quantum dot-labelled AQP4 proteins were tracked through the reflection of the green fluorescent protein and fluorophore-labelled AQP4 using total internal reflection fluorescence microscopy. Biophysical methods such as photobleaching and fluorescence correlation spectroscopy, the physiology of AQP4 isoforms, I interactions, site of OAP formations, size, and stability can also be studied [64]. The presence of inflammation in the visual pathways and cloud-like and pencil-thin ependymal enhancement have been reported using a contrast-enhanced MRI [65,66,67]. An increase in the thickness of the anterior visual pathways and optic T2 signal hypersensitivity during ON is a marker of neuroinflammation [68]. For the past two decades, imaging research has attained peculiar development in structural and functional imaging at subcellular resolution and has contributed a lot to understanding the tissue damage mechanisms, disease progression sites, and degree of pathology, monitoring the disease patterns in individual patients. Although imaging methods have been technically developed, limitations exist, such as a need for standard protocols and imaging techniques to understand and differentiate neuroinflammation in NMOSD and other neuroautoimmune diseases.
Clinical MRI examination is important in suspected demyelinating myelitis to prevent misdiagnosis. MRI examinations provide necessary cues for differential diagnosis of different myelopathies and support proper treatment decisions [69]. AQP4+ NMOSD subjects showed massive inflammation, complete spinal cord impairment, and other motor deficits such as sensory loss, para or quadriparesis, and sphincter and erectile dysfunction [70]. Few patients suffer from respiratory failure due to cervical myelitis [71]. Up to 4% of patients with acute myelitis also suffer bilateral ON and brainstem manifestations with symptomatic brain lesions [72]. MRI analysis revealed that long cord lesions with central necrosis and cavitation in the 3rd or 4th vertebral segments over weeks or months are the most prominent clinical characteristic of NMOSD [73]. Thus, MRI diagnosis accurately understands specific clinical outcomes of the inflammatory myelopathies and would support required relapse preventive treatment methods for the disease [69].
Numerous studies help distinguish MOGAD from MS and AQP4+ NMOSD. The clinical manifestations of MOGAD are heterogenous, including ON, myelitis, and multifocal demyelination in the CNS, which can often resemble acute disseminated encephalomyelitis (ADEM). MRI results differentiate MOGAD from MS and AQP4+ NMOSD with perineural optical nerve enhancement. Other possible methods of differentiating MOGAD diagnosis from MS and AQP4+ NMOSD include the cell-based assay and antibody titer for MOG-IgG [74]. Differential diagnosis of ON was characterized in MOGAD and AQP4+ NMOSD. AQP4-ON shows long, unilateral lesions mostly affecting the posterior optic nerves through T1 gadolinium enhancement [63]. MOG-ON shows longer, bilateral lesions mostly affecting the anterior of the optic nerve with perineural T2 hypersensitivity and gadolinium enhancement [75]. MOG and AQP4 optic lesions extend into the intracranial and pre-chiasmic segments of the optic nerve compared to MS [76,77]. Bilateral ON and longitudinal optic nerve lesions are common in MOGAD and AQP4+ NMOSD, respectively.
MRI studies with different MOGAD cohorts revealed localized lesions in the deep white matter and area postrema [78]. In another study, lesions were observed in the supratentorial deep white matter [79]. These MRI studies help distinguish the MOG and AQP4 seropositive patients through the key indicator that the AQP4 cohort preferentially showed no deep white matter lesions compared to the MOG type. Another unique feature in MOGAD is the leptomeningeal enhancement and cortical/juxtacortical lesions, which are absent in AQP4+ NMOSD patients [80]. AQP4+ NMOSD displayed distinctive lesions in anatomical locations such as the peri ependymal. Periaqueductal and hypothalamic areas showed no imaging abnormalities in the MOGAD cohort [81]. Brain lesions are more commonly reported in pediatric-onset NMOSD than adult-onset, making it difficult to differentiate ADEM and NMOSD. Pediatric ADEM presents with longitudinally extensive transverse myelitis such as NMOSD. Therefore, myelitis is not considered a reliable sign or predictor of NMOSD in pediatrics [82]. Pediatric NMOSD and ADEM can be differentiated through thalamic involvement. Thalamic involvement in NMOSD is infrequent, with a limited non-specific lesion, whereas thalamic involvement in ADEM is more frequent with a large, asymmetric lesion. Thus, the thalamic lesion location favors differentiating NMOSD and ADEM [82]. Zhang et al. reported that brain lesions in the putamen and hypothalamus favor the diagnosis of ADEM and NMOSD, respectively [83]. NMOSD is occasionally associated with primary SS and systemic lupus erythematosus (SLE). Non-infectious inflammatory myelitis was reported in paraneoplastic myelitis and SS [84]. Most myelitis cases are related to SS, having longitudinally extensive spinal cord involvement that has been used as a diagnostic criterion for NMOSD. Critically, the clinical, radiological, and prognostic spectra have not differentiated SS from NMOSD, yet AQP4-ab is presented only in NMOSD patients [85]. SLE patients were also reported to have CNS lupus with headache, seizure, and memory impairment. Myelitis and optic neuritis were also found in a few SLE patients. Although the symptoms are the same, CNS lupus manifestation in SLE is distinct from that in NMOSD [86]. Thus, the diagnosis of NMOSD is critical in cases exhibiting similarities with regard to certain symptoms and signs of other NADs and of conditions such as multiple autoimmune disorders.
Detection of antibodies against MOG were found in a subset of patients with ADEM. The clinical spectrum of MOGAD showed monophasic acute ON, brainstem encephalitis, and transverse myelitis [87]. In contrast, the clinical spectrum of ADEM showed chronic relapsing inflammatory ON and LETM [88]. The MRI studies of MOGAD encephalitis are coherent with ADEM patterns and with abnormalities in the cortical grey matter, subcortical white matter, and deep grey and white matter [89]. Non-MOGAD ADEM conditions mimic MOGAD, which involves the brain, spine, and optic nerve. However, brain lesions are asymmetric, bilateral, large, and involve the brainstem, cerebellar white matter, thalamus, and basal ganglia [90,91].
5.2. Metabolomic Studies to Differentiate NMOSD and Other Related Disorders
Barring the discovery of specific biomarkers for early diagnosis of AQP4-IgG+, AQP4-IgG−, MOG-IgG+, and MOG-IgG−, the differentiation of NMOSD and other related disorders remains impassable. Although AQP4-IgG antibody titers represent the clinical presentation and immune response associated with NMOSD, similar AQP4-IgG titers values may occur due to complex immunoinflammatory processes [92,93]. This could complicate the similarities and variations between disease and non-disease conditions. Studies evaluated serum samples of MS patients using metabolomic profiling to differentiate the pathophysiology of MS from healthy subjects. Mass spectrometry and nuclear magnetic resonance (NMR) spectroscopic studies revealed increased levels of sphingomyelin, phosphatidyl-inositol, phosphatidylcholine, phosphatidyl-ethanolamine, glutamate, and other amino acids [94,95,96,97]. Further studies with ultra-high-performance liquid chromatography-mass spectrometry and ultra-high-performance liquid chromatography-time of flight-mass spectrometry determined the possible biomarkers of MS, which help differentiate MS from healthy conditions and other diseases such as NMOSD [30]. A lipid profiling study in MS patients revealed increased serum lysine and histidine and decreased HDL particles, unsaturated lipid, and alanine, which helps to differentiate MS from NMOSD [98].
A serological comparative study in NMOSD and RRMS patients using a proton high-resolution magic angle spinning NMR spectroscopy (1H-HRMAS-NMR) evidenced that the astrocytic metabolites scyllo-inositol and acetate are promising biomarkers of MS and NMO, respectively, with more than 90% sensitivity and specificity [99]. The NMR analysis of urine samples of MS patients and healthy subjects revealed that 27 metabolites were altered in MS patients and were associated with fatty acid metabolism, mitochondrial activity, and the gut microbiome [100]. CSF metabolites of MS, NMO, and idiopathic transverse myelitis patients and healthy controls were reported. The study specified that CSF metabolomic profiling could enhance the accuracy of CNS inflammatory demyelinating diseases. The lipids (1-monopalmitin and 1-monostearin) were common in MS, NMO, and idiopathic transverse myelitis cases [101]. Coherently, the metabolomic studies of blood, plasma, serum, urine, and CSF showed that several metabolites were distinctive and that such changes directly describe their invincible role in disease progression or regression.
6. Molecular Mechanism of NMOSD
6.1. AQP4 Dependent Pathology
AQP4 is a bidirectional transmembrane protein highly present in the astrocyte end feet of the BBB and the ependymal cells of the CNS [60]. AQP4 has been found to be necessary for the functioning of the BBB and important for maintaining CNS water homeostasis [60]. BBB disruption is the hallmark pathological sign of NMOSD, resulting in BBB breakdown and an influx of serum AQP4 IgG and complement factors into the CNS compartment, followed by the entry of B cells into the CNS and the formation of lesions [102]. Many histological studies in NMOSD patients have revealed the loss of AQP4 and CNS lesions, with or without astrocyte loss [103]. The presence of the circulating serum autoantibody against the AQP4 water channel (AQP4-IgG) induces neuroinflammation by breaching the BBB, releasing proinflammatory cytokines into the CNS, and producing acute CNS attacks, CNS pathologies, area postrema syndrome, astrocyte inactivation, and astrocyte loss through complement inactivation, BBB disruption, and neural injury [103,104,105].
Besides the astrocytes, the retinal muller cells are highly packed with AQP4 and inwardly rectifying potassium (Kir4.1) channels [106]. The binding of AQP4-ab with AQP4 in the astrocyte initiates immune complement cascade signals, which results in the infiltration of granulocytes, lymphocytes, and eosinophils, leading to astrocyte and oligodendrocyte injury and demyelination, myelin loss, neurodegeneration [5], and thickened hyalinization blood [35].
The high expression of AQP4 in the periventricular zone and particularly in the area postrema make these regions more prone to NMOSD pathology [51]. The international consensus diagnostic criteria for NMOSD suggest that AQP4-IgG plays a major causative role in NMOSD. AQP4-IgG seropositive individuals showed six affected regions in the CNS, including the optic nerves, spinal cord, area postrema, brain stem, and cerebrum. The spinal cord and optic nerves are affected in AQP4-IgG seronegative individuals [17]. The astrocyte-specific, glial fibrillary acidic protein (GFAP) necessary for maintaining the structural integrity of the BBB was significantly higher in the CSF of NMOSD patients [107]. A cohort study in North American patients with meningeal encephalomyelitis revealed that GFAP autoantibodies could act as the serum biomarker for CNS autoimmune diseases and potentially initiate complement-mediated immune responses and cause astrocyte damage [108,109].
Aquaporins are the transmembrane proteins essential for water transport and homeostasis in the CNS. Among the thirteen aquaporins in mammals, AQP1, AQP4, and AQP9 are in the brain, especially AQP4, which is present in the CNS–blood and CNS–CSF interfaces in the subarachnoid, subependymal, and pericapillary spaces [110,111]. The AQP4 protein is also expressed more in the retina, optic nerves, and optic tract [112]. AQP4 exists as two dominant isoforms, M1 and M23, due to the extra 22 amino acids in the intracellular N terminus of M1 isoforms. AQP4 monomers consist of six transmembrane helices and two helices with asparagine–proline–alanine motifs that form the pore for transporting water [113]. Both the AQP4 isoforms are highly selective, allowing only the water molecules and excluding other ions or molecules. AQP4 involves potassium reuptake, which is essential for maintaining BBB integrity, synaptic plasticity, and spatial memory [114,115]. The absence of AQP4 in the AQP4 −/− mice showed hyperpermeability of the BBB [7]. A study on autoimmune encephalitis AQP4 −/− mice revealed reduced demyelination, CNS inflammation, motor dysfunction, and cytokine production [116].
The antibodies of AQP4 (AQP4-ab), also known as NMO-IgG, are the prominent serum markers of autoimmune NMO. AQP4-mediated complement pathways and internalization of AQP4 are important reasons for NMO disease [117]. Most NMOSD patients are seropositive for circulating AQP4 immunoglobulin G (AQP4-IgG) antibodies, which target various extracellular epitopes of the AQP4 water channel [8,14]. AQP4-IgG autoantibodies possess a polyclonal ability that recognizes and binds to various epitopes and domains of the AQP4 channel [118]. Anti-AQP4-IgG produced by the plasma cells in peripheral blood enters the CNS through endothelial transcytosis and a permeable BBB and binds with the AQP4 channel. The binding of AQP-IgG with AQP4 activates the complement factors leading to astrocytic injuries through CDC and ADCC. The CDC and ADCC induce further inflammatory signals downstream and the migration of macrophages, neutrophils, and eosinophils to the inflammation site, resulting in BBB interruptions, loss of myelin, and neuronal injury [116,119]. Increased levels of IL-6 are correlated with increased anti-AQP4-IgG and glial fibrillary acidic protein and increased damage in NMOSD [120]. AQP4 crosses the BBB, and the Fc domain of AQP4-IgG binds to the AQP4 in the astrocyte end feet, thereby employing complement activation and causing complement-dependent cytotoxicity (CDC). CDC is activated by AQP4 binding with C1q complement protein. The formation of OAPs by clustering AQP4 results in adding more AQP4-IgGs, which enhances the multivalent binding of C1q onto the grouped AQP4-IgGs over the OAPs [42]. The binding of AQ4-IgG causes endocytosis of AQP4 and reduces water transport across the plasma membrane [120].
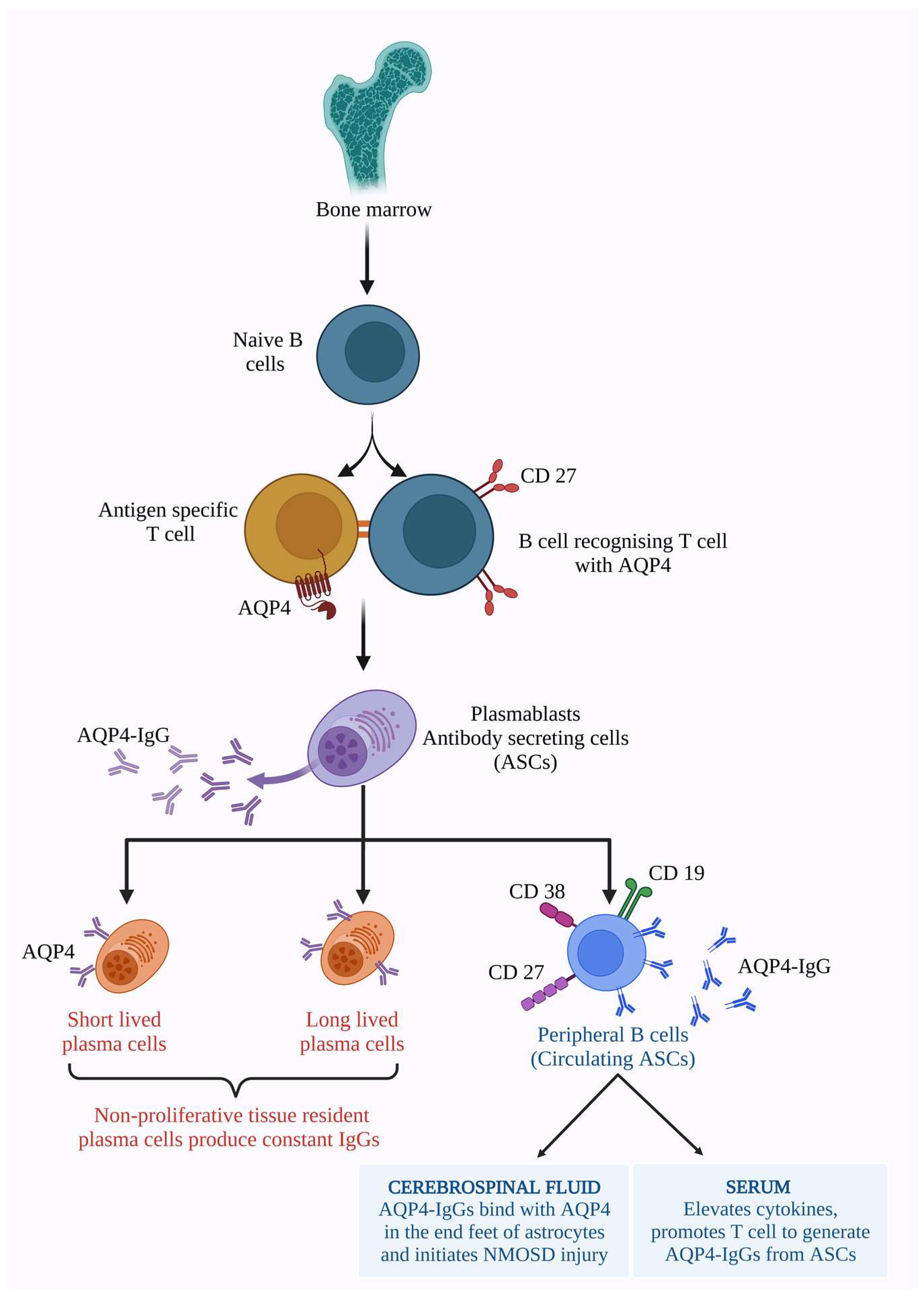
To develop memory for AQP4 antigen, B cells require AQP4-specific T cells [53]. AQP4-IgG-generating plasma cells were originally developed by the recognition of T cells with AQP4, which led to the expansion of B cell differentiation into plasmablasts [121]. Identifying the subsets of B cells that produce AQP4-IgG helps in targeted immunotherapies. Antibody-secreting cells (ASCs) are the proliferative plasmablasts that emerge from bone marrow. Initially they are naive B cells, later developing antigen memory, acquiring CD 27, and switching the IgD/IgM expression into IgG expression on its surface [119,122]. Wilson et al. studied the capacity of B cells to produce AQP4-IgGs. The circulating B cells produce AQP4 antibodies in the absence of antigens. Few other factors induce AQP4-IgGs without the cytokines. Hence, it was found that antigens are not necessary for AQP4-IgG production [121]. The production cycle and availability of AQP4-IgGs in the peripheral nervous system and the CNS, and their function in the serum and CNS, are described in Figure 2.
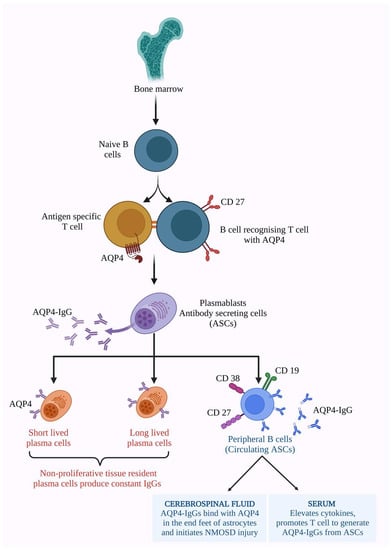
Figure 2.
The AQP4-IgG antibodies are produced by antibody-secreting cells (ASCs). Precursors of ASCs secreted from bone marrow as naive B cells. Naive B cells acquire memory antigen CD 27 on their surface along with AQP4 antigen-specific T cells and switch their IgM/IgD expression into IgG. Thus, developed antibody-secreting plasma differentiates into short, long-lived (these are tissue-resident plasma cells that produce constant IgGs), and peripheral circulating B cells secreting AQP4-IgG antibodies with other cell surfaces markers such as CD 19, CD 27, and CD 38 on its surface. Thus, produced AQP4-IgGs are available in the serum and enter the CNS, thereby inducing cytokine production, developing the inflammation cycle, and promoting T cells to generate more ASCs. In the CNS, the AQP4-IgGs bind with AQP4 present in the end feet of astrocytes and initiate NMOSD injury and lesions [109,121,122]. (Figure created using BioRender.com; accessed on 7 February 2023).
The binding of AQP4-IgG to AQP4 induces endocytosis, which results in the loss or internalization of the excitatory amino acid transporter 2 (EAAT2) [123]. The internalization of glutamate receptors reduces the astrocytic enzyme glutamate synthase activity, causing glutamate accumulation [123]. More accretion of glutamate stimulates the production of various immunological factors, chemokines, cytokines, and complement factors [124]. The production of immunological components activates immune cells such as eosinophils, neutrophils, and macrophages, which damage neurons’ myelin [125,126]. The complement factors bind to the Fc region of the AQP4-IgG antibody; in that way, CDC and ADCC effector mechanisms are initiated by activating the multivalent complement factors such as C1q, C3a, C5a, and MAC over the AQP4-IgG [127,128]. Thus, the activated complement cascade recruits other factors such as C3a and C5a.
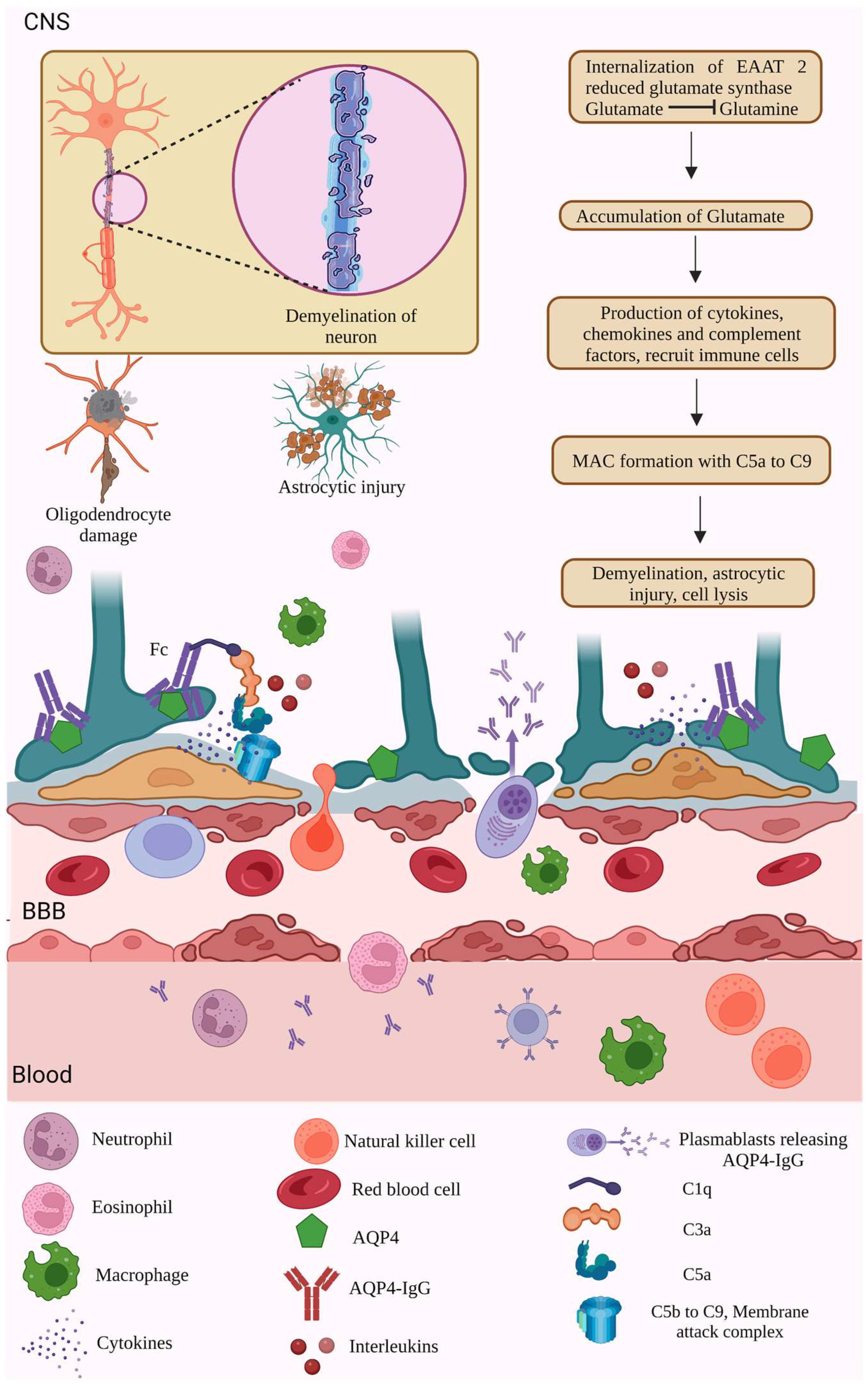
The binding of C5a attracts complement factors C5 b to 9, leading to the formation of a membrane attack complex (MAC), which further leads to cell lysis. In contrast, complement activation is modulated by regulatory proteins such as CD59. ADCC allows the binding of AQP4-IgG onto the Fc receptors and activates the ADCC pathway and degranulation. Thus, the inflammatory niche produced by the CDC and ADCC results in the infiltration of neutrophils, macrophages, and eosinophils, astrocytic and oligodendrocyte injury, BBB disruption and leakage, and demyelination and neuronal injury [127,128]. In addition to the neurons and oligodendrocytes, the bystander cells that do not express the AQP4 are also subjected to the inflammatory cytokine storm produced due to astrocytic injuries [127] (Figure 3).
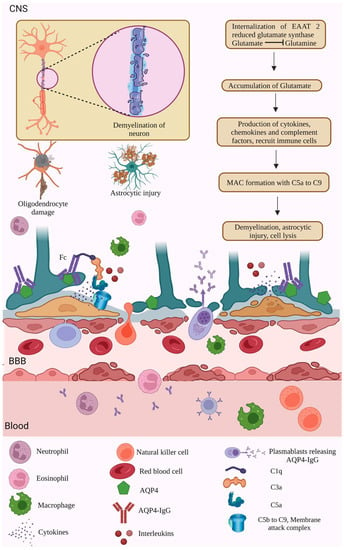
Figure 3.
AQP4- dependent NMOSD pathology. NMOSD pathogenesis is initiated through various etiological factors such as genetic reasons, environmental factors, genetic-environment context, low levels of vitamins, medications, and so on. NMOSD pathogenesis involves the synthesis of autoantibodies against AQP4, a water channel transmembrane protein present in the astrocyte end feet of the BBB. AQP4 antibodies moved from the plasma into the CSF through the BBB. The presence of more AQP4 autoantibodies circulating in the plasma results in inflammation and causes the production of pro-inflammatory cytokines, interleukins, and complement factors. Thus, produced cytokines, complement factors, and granulocytes breach into the BBB and CSF of NMOSD patients. The cytokines activate the complement cascade factors, activating NK cells and the MAC. The MAC targets the astrocytes’ AQP4 channels, leading to astrocyte damage and neuroinflammation [123,124,125,126,127,128]. (Figure created using BioRender.com; accessed on 12 February 2023).
6.2. MOG-IgG-Dependent Pathology
Myelin oligodendrocyte glycoprotein (MOG) is present on the outer surface of myelin sheaths [47] that consist of 218 amino acid residues spanning twice across the cell membrane and having IgG-like domains in the extracellular N-terminal-end [129]. In AQP4-ab seronegative NMO cases, autoantibodies against MOG (MOG-IgG) have been detected. The AQP4-IgG type of NMOSD primarily affects astrocytes and causes astrocytopathy, whereas MOG-IgG causes demyelination, as it expresses mostly in the oligodendrocytes and outermost myelin sheaths [49]. AQP4-ab and MOG-IgG are the two different target-mediated entities of NMOSD. The characteristics of this two-antibody-mediated NMOSD are different. AQP4-ab positive cases showed loss of AQP4 and dystrophic astrocytes. In the MOG-IgG type of NMOSD, the pathology is differentiated by the expression of AQP4, perivenous and primary demyelination, reactive gliosis in the white and grey matter, and numerous lesions in cortical regions [48]. The clinical features of MOGAD differ from AQP4-IgG seropositive NMOSD. Even though ON is a common feature in AQP4-IgG NMOSD and MOGAD, the phenotypic difference can be seen in the length of optic nerve involvement, optical disc morphology, and the ganglion cell inner plexiform layer pattern in OCT [130,131].
The clinical characteristics of MOGAD and AQP4+ NMOSD were compared with CSF analysis and peripheral T/B lymphocytes during active and chronic phases. The results showed that the protein level in MOGAD was significantly lower than in AQP4+ NMOSD. The level of myelin basic protein was significantly lower in MOGAD. Furthermore, the albumin and IgG reflecting BBB permeability and intrathecal IgG production were significantly lower in MOGAD patients compared to AQP4+ NMOSD patients. The increased permeability of the BBB in AQP4+ NMOSD was observed more than in MOGAD. Concurrently, the BBB allows AQP4-IgG to promote IL-6 production from AQP4-positive astrocytes. T/B lymphocyte subset plasmablasts are also normal in MOGAD and higher in AQP4+ NMOSD, and the B cell subsets differ in both conditions. Transitional B cells were higher in MOGAD than AQP4+ NMOSD and healthy controls, but there was no significant difference in memory B cells [132].
In MS, MOGAD, and AQP4+ NMOSD, the primary phenomenon is the neurodegeneration of myelin deficient retina. Analyzing the degeneration of retinal neurons is a frequently used diagnostic tool in MOGAD and NMOSD. The retinal nerve fibre layer was significantly reduced in NMOSD patients, and visual impairment was observed in MOGAD [131]. The inflammation and visual system degeneration were evaluated using spectral domain OCT and electroretinography in transgenic spontaneous opticospinal encephalomyelitis mice. The results revealed that the damage to the retina and optical nerve, which could affect the retinal functions, caused inflammation, complement factors activation, and degeneration (Figure 4). Further histological analysis of retinal and optic nerve degeneration is only capable in postmortem tissues [131].
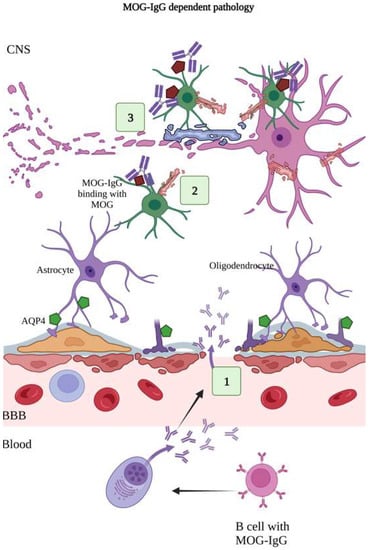
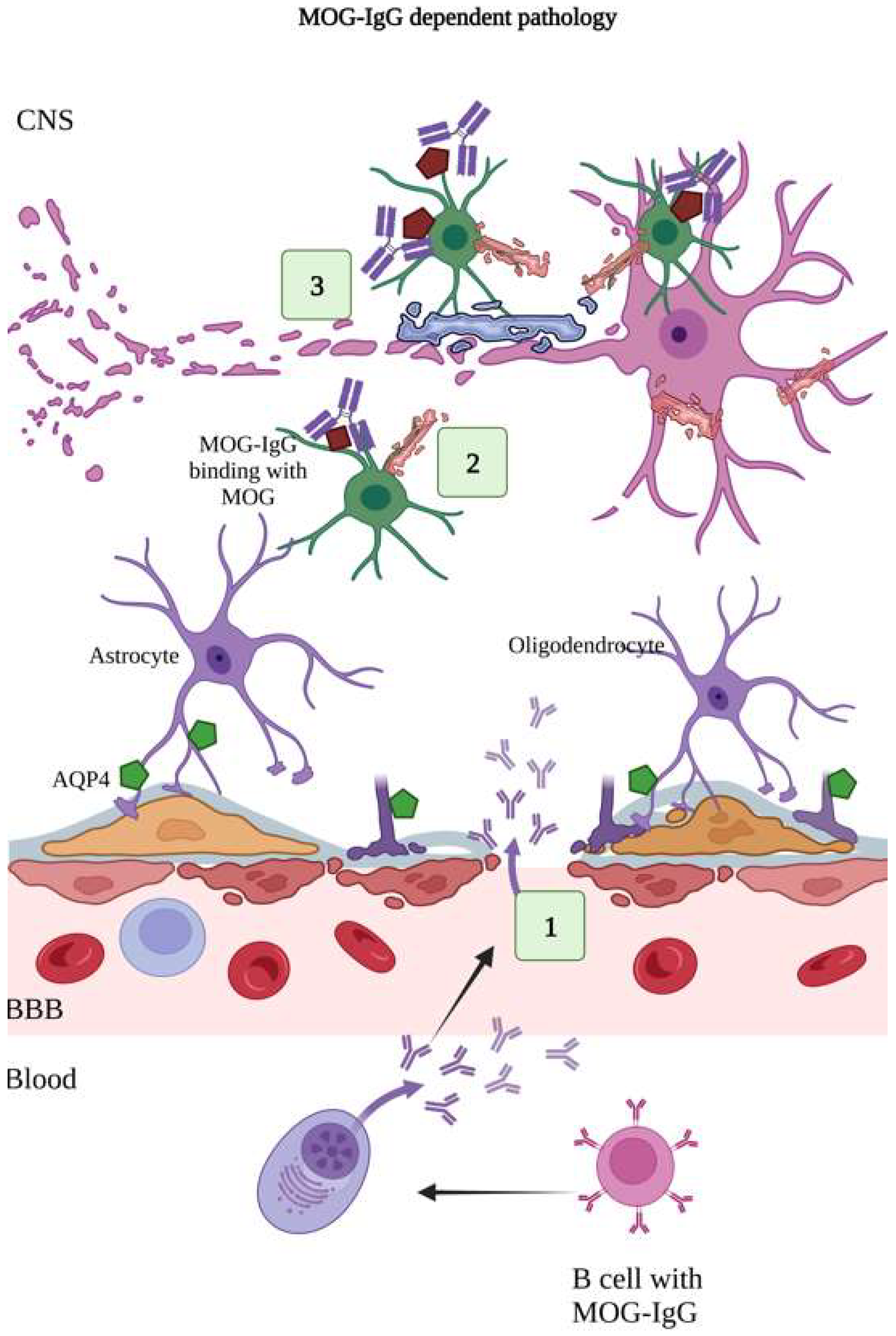
Figure 4.
MOG-IgG dependent pathology. MOG-IgG antibodies are specific for myelin oligodendrocyte glycoprotein present on the myelin-forming oligodendrocytes and myelin sheath, which insulates the neuronal cell. (1) The MOG-IgG antibodies produced by plasma cells cross the BBB. (2) MOG-IgG antibodies bind with MOG antigens. (3) Binding of MOG-IgG onto the MOG causes release of myelin basic proteins, resulting in demyelination, oligodendrocyte damage, and loss of neuronal cells [129,130,131]. (Figure created using BioRender.com; accessed on 12 February 2023).
6.3. IL-6 Pathophysiology
IL-6 is an important pathogenic factor of NMOSD, a pleiotropic cytokine and regulator of acute and chronic inflammation, and is responsible for the survival of plasmablasts, which is essential for AQP4-IgG production [133]. The survival of plasmablasts and B memory cell maturation are modulated by IL-6 signalling. Alternatively, IL-6 inhibits the differentiation of Th17 cells [134]. IL-6 initiates the pathophysiological processes and disease activity of NMOSD by stimulating AQP4-IgG secretion, disturbing BBB integrity, enhancing proinflammatory T cell differentiation and activation [135], and regulating the balance between Th-17 and regulatory T (Treg) cells. Studies have revealed that enhanced IL-6 levels inhibit regulatory T cell differentiation and induce B cell differentiation in NMOSD conditions. Levels of IL-6 and soluble IL-6 receptors are increased more in the CSF and serum of NMOSD patients during relapse than during the remission period [136,137].
IL-6 enhances BBB permeability, enhances the entry of antibodies into the CNS, and induces pro-inflammatory cell infiltration, leading to the binding of AQP4-IgG with AQP4 [135]. It was previously reported that NMO-IgG enhances the expression of the IL-6 gene [136]. The production of IL-6 in astrocytes depends on the Janus Kinase/Signal transducer and activator of the transcription 3 protein (JAK/STAT3) pathway and NFκB pathway [136,138]. Under usual circumstances, the IL-6 level is low in the CNS. CNS lesions could stimulate IL-6. T cells, B cells, endothelial cells, monocytes, and glial cells can produce IL-6 in different situations such as inflammation, antigen-specific immune responses, and during the use of host defence mechanisms [139]. In multiple neuroinflammatory diseases, the injured neurons, astrocytes, and microglial cells produce IL-6, which is helpful in differential diagnosis [140], oligodendrocyte and axonal injury, and demyelination [135].
The levels of IL-2 and interferon-γ (INF- γ) in the CSF remain unchanged in NMOSD [141,142]. The cytokines IL-1β, IL-6, IL-17, and TNF-α and other complement factors C1s, C3a, C4a, C4d, and C5 are elevated in NMOSD [30]. NMOSD pathogenesis can be promoted through IL-6-dependent Th-17 cells and Th-17-associated cytokines [143]. IL-6 induces the naive T cells and differentiates them into Th-17 inflammatory cells, which initiates further inflammation and disturbs BBB integrity. IL-6 also induces B cells to differentiate into plasmablasts and produce AQP4-IgG antibodies [144]. The increased IL-6 levels in the serum and CSF correlate with expanded the disability status scale (EDSS) score [137,139,140] and relate to the levels of AQP4-IgG and GFAP, which are directly proportional to the degree of astrocyte damage [141]. Thus, high levels of IL-6 in serum and the CSF can be marked as an indication of NMOSD relapse. Blocking the IL-6 receptors with the help of monoclonal antibodies might inhibit IL-6-associated humoral immune response mechanisms, T cell activation and pathways, BBB dysfunctions, recruitment and activation of the complement system, and macrophage activities [135].
Pro-inflammatory cytokine IL-6 levels remain high in the serum of NMOSD patients, which remains an important prognostic biomarker and a potential target for NMOSD therapy because the IL-6 initiates the production and maintenance of Th17, a kind of CD4+ T helper cell, which further induces other pro-inflammatory markers, neutrophil deposition, and organ-specific autoimmunity, enhancing AQP4-IgG secretion even more [135,137]. In addition to IL-6, the granulocyte-macrophage stimulating colony-forming factor (GM-CSF) could stimulate the function of Th17 cells [145]. Apart from IL-6, the other cytokines IL-1, IL-8, IL-10, IL-13, IL-17, IL-21, and IL-23 were found in higher levels in the serum and CSF of NMOSD patients [146].
6.4. Complement-Mediated Pathology
Other immune pathways, such as complement-dependent cytotoxicity (CDC), complement-dependent cell-mediated cytotoxicity (CDCC), and antibody-dependent cellular cytotoxicity (ADCC), also initiate astrocyte injuries. CDC produces a membrane attack complex (MAC) by triggering the complement pathway [127,128]. The complement cascade is activated by binding AQP4-IgG onto AQP4, which recruits the complement factors and forms the membrane attack complex formation that attacks the astrocytes, oligodendrocytes, and neurons, leading to demyelination and neuronal cell death [127]. The complement factor C3a is quite elevated in NMOSD patients and is found to be responsible for the disease activity, presence of neurologic disabilities, and AQP4-IgG [147]. ADCC mainly involves binding neutrophils, macrophages, and NK cells with the Fc region of the AQP4-IgG antibody [148]. CDC pathways induce membrane permeability, cause AQP4-IgG antibody influx into the BBB, and accelerate inflammation [149]. Another MAC inhibitory protein, CD59, constrains the accumulation of MACs on the AQP4-expressing tissues. A study demonstrated that rats lacking CD59 are highly prone to NMO pathology, and that CD59 is responsible for protecting AQP4 tissues. In CD59 −/− rats, the MAC gets activated, targets the AQP4 channels, and causes astrocyte damage, inflammation, and injury. CD59, the regulatory complement protein, protects AQP4 seropositive NMOSD cases [150].
The levels of complement factors C3 and C4 in the plasma of patients with MS, MOGAD, and AQP4-IgG+ NMOSD and healthy controls were examined. AQP4+ NMOSD and MOGAD patients have the lowest C3 and C4 levels compared to other participants, suggesting that C3 and C4 might be diagnostic markers for AQP4-IgG+, NMOSD, and MOGAD [143]. Another study demonstrated the association of the levels of complement factors C3 and C4 with AQP4-IgG titers, indicating the involvement of AQP4-IgG clones in activating the complement [149]. Purinergic receptors (P2Rs) were used to treat inflammatory diseases that protect against complement-mediated cell injury. P2Rs interact with AQP4-IgG and cause misfolding of antibodies, which disrupts the complement involvement and activation. The interaction of AQP4-IgG antibodies is of vital clinical importance because its interactions determine the complement activation and BBB permeability [151]. In particular, C5 initiates the terminal cascade reactions, and a C5 inhibitor (eculizumab) can neutralize C5 and inhibit MAC formation by blocking the CDC [152].
Complement activation requires aggregation of AQP4 monomers (OAPs). AQP4-IgGs bind to OAPs and C1q, a multiple interaction reaction in which a single C1q molecule binds to six AQP4-IgG molecules [42]. The binding of C1q to AQP4-IgG activates the CDC, recruits pro-inflammatory anaphylatoxins, forms a MAC, and induces cellular injury. In ADCC, the cascade gets activated by binding effector leukocytes onto the Fcγ region of AQP4-IgG. This can cause further cellular injury by degranulation and local cell death by infiltrating NK cells, neutrophils, eosinophils, and macrophages [148]. Thus, the injury creates an inflammatory response in astrocytes and produces other cytokines, disturbing the BBB and damaging oligodendrocytes, microglial cells, and nearby neurons [37,153]. The cell types involved in ADCC also participate in CDCC through enhanced phagocytosis and anaphylatoxin-induced degranulation of cells. The anaphylatoxins produced by NMO lesions act as chemoattractants for the nearby circulating eosinophils, neutrophils, monocytes, and macrophages [154]. Höftberger and her team histopathologically analysed autopsies and brain biopsies of patients with MOGAD. They observed that MOG-IgG activates the complement and complement deposition in all white matter lesions and that MOGAD pathology is dominated by perivenous and confluent white matter demyelination. MOG-IgG binds to the outer myelin surface, then the myelin is destroyed by complement and ADCC phagocytosis due to the endocytic internalization of MOG antigens into the cell [48].
6.5. Involvement of Gut Microbiome and NMOSD
Numerous microbial populations inhabit the gastrointestinal tract. They are involved in various functions in the host, such as digestion, absorption, protection, energy balance, immune response, intestinal permeability, enteric nervous system activity, and brain functions such as emotions, pain, behaviour, stress responses, etc. The gut is linked with the neural networks through vagus nerves and organizes the exchange of metabolites [155]. The gut microbiota (GM) is vital in developing neuroimmunological disorders. Recent studies signify the association between GM dysbiosis and NMOSD through intestinal mucosal barrier destruction and intestinal and peripheral immunity.
Any abnormality in GM functions may cause the release of endotoxins, microbial products, and proinflammatory cells and cytokines, which affects the intestinal mucosal barrier function, allowing the substances to enter the bloodstream, thereby damaging the BBB and producing demyelination, axonal loss, and CNS damage and initiating NMOSD pathology [33]. Th17 cells involved in organ-specific immunity originate from the gut and are abundant in the intestinal lamina propria. The differentiation or migration of Th17 cells to the lymphoid-rich sites depends on the gut commensals [156]. The magnitude and frequency of T cell proliferation are higher in NMOSD patients than in healthy controls in response to AQP4 determinants. The adenosine triphosphate-binding cassette transporter permease (ABC-TP) of Clostridium perfringens has 90% similarity to AQP4 p61–80 peptide. AQP4 p61–80 peptide binds to T cells, but T cells mimic the homologous sequence of C. perfringens, bind with the ABC-TP transporter peptide, and cause cross-reaction and Th17 polarization. Other AQP4 sequences, such as p63–76, also exhibit 60–70% homology with the ABC-TP of other commensal and pathogenic species of Clostridium, such as C. scindens, C. hylemonae and C. sporogenes. Thus, Clostridium species stimulate proinflammatory immune responses by influencing the balance between Th17 and Treg cells [157]. C. perfrigens has dual functions; i.e., promoting the polarization of proinflammatory Th17 cells and exposing ABC-TP homologous determinants that cross-react with AQP4-producing reactive T cells and regulate the balance between T cells and Th17 cells, eventually resulting in immunologic dysfunctions and NMOSD [158].
Cree and his team examined faecal microbiota in NMOSD patients for the first time. The study revealed that microbial diversity was reduced, and C. perfringens abundance was increased significantly in NMOSD patients, suggesting that C. perfringens could be involved in NMOSD pathogenesis [159]. C. perfringens is also implicated in MS and NMO. Other members, such as Fibrobacteres, were over-presented in NMO patients’ gut microbiota. Th-17 cells proliferate in response to the ABC peptide of C. perfringens, which shows the link between NMO and gut microbiota [159].
The abundance of butyrate-producing bacteria such as Faecalibacterium, Roseburia, Ruminococcus, and Coprococcus was reduced in AQP4+ and AQP4− groups. Bacteroides and Parabacteroides were predominant in AQP4− groups [160]. Investigation of possible foreign proteins that possess structural homology with AQP4 revealed that transmembrane proteins from Klebsiella pneumoniae were involved in cross-reactivity in place of AQP4 in the case of NMO [161]. A Chinese cohort study revealed the presence of a high abundance of Streptococcus in NMOSD patients. The high abundance of Streptococcus was associated with reduced short-chain fatty acids and increased CD4+ T cells promoting inflammatory reactions in NMOSD patients [162]. Another study in NMOSD patients showed that distributions of Streptococcus, Shigella, and Faecalibacterium were positively associated with NMOSD severity. Some species of Streptococcus, including S. oralis, S. salivarius, S. parasanguinis, S. pneumonia, and S. mitis, varied significantly between healthy subjects and NMOSD patients [163]. Studies showed that Bacteroides, Firmicutes, and Proteobacteria were more abundant, and other phyla such as Tenericutes and Actinobacteria were less abundant in NMOSD patients and healthy subjects [34]. Phyla Proteobacteria and Verrucomicrobia were significantly higher in AQP4+ and AQP4− NMOSD patients. Members of genera Clostridium, Streptococcus, Megamonas, Enterobacteriaceae, and Bilophila were predominant in AQP4+ cases, whereas members of Bacteroides, Megamonas, Phascolarctobacterium, and Akkermansia were prominent in AQP4− cases. At the species level, AQP4+ NMOSD patients had Clostridium boltae and Flavonifractor. In the case of AQP4− NMOSD patients, Megamonas funiformis and Clostridium ramosum were significantly abundant. Megamonas funiformis, Phascolarctobacterium faecium, Flavonifractor plautii, and Clostridium ramosum were described in both AQP4 seropositive and seronegative patients [34].
Like the C. perfringens ABC peptide, peptide 59–71 from C. boltae also showed homology with the AQP4 p91–110 peptide, was highly reactive to AQP4-specific T cells [34], and initiated disease pathogenesis. C. boltae caused immunopathogenesis in NMOSD patients through Th17, which induces inflammatory responses and influences the balance between Th17 and Treg cells [34]. NMOSD patients with high annual reoccurrence rates have a high abundance of Actinomyces and Sphingomonas and lower abundances of Veillomonas, Atopobium, and Haemophilus [31].
The studies on gut microbiota and NMOSD explain the correlation between gut dysbiosis and NMOSD by signifying the abundance of pathogenic bacteria and reduction in commensal organisms, which cause abnormal metabolism and metabolic signals in the pathogenesis of autoimmune diseases such as NMOSD. The exposure of intestinal epithelium to microbial pathogens provokes an inflammatory response. The transported pathogens and certain toxins are endocytosed in the lamina propria [164]. Still, failed defense mechanisms in the mucosal immune system damage gut integrity, which can elicit inflammatory responses [165] and disturb BBB integrity in NMOSD patients [166].
Streptococcus induces inflammatory factors, causes damage to the intestinal mucosal barrier, and affects immune function [33]. Once the tight junction in the intestinal barrier is affected, it increases gut leakage. The T helper1 cells and Th17 and Treg cells are stimulated by cytokines such as IL12 and IL18; later, this cascade induces interferon-γ production and results in inflammation and damage in the intestine [167]. A high abundance of Streptococcus might indicate the inducing of neuroinflammation and BBB degradation through Th17 activation [33]. Certain microbial antigens stimulate immunomodulation in autoimmune diseases of the CNS. A few bacterial species, such as Fusobacterium nucleatum and members of Clostridium, contain potent encephalitogenic peptides that closely mimic the myelin basic protein and myelin oligodendrocyte protein that trigger autoimmunity [168]. The immunomodulation of the CNS in autoimmune diseases needs to be further studied to reveal the involvement of the gut microbiome and its products.
6.6. Models of NMOSD Study
The clinical features of NMOSD and ON were evaluated in some of the studies. Animal models of NMOSD developed by introducing the NMO-IgG antibody into mice with experimental autoimmune encephalomyelitis displayed a few features of NMOSD, such as inflammation and loss of AQP4 and GFAP [169,170,171]. Injecting AQP4-IgG can stimulate pathogenicity [172]. In another study, NMO-IgG and complement factors were injected directly into the murine brain, stimulating pathology around the injection site [173]. In such methods of generating animal models of NMOSD, the doses of NMO-IgG to be injected determine the pathology and the dosages need to be clarified further. Luo et al. developed an NMOSD animal model by delivering NMO-IgG and complement factors into the brain intravenously after reversing the BBB opening using a microbubble-enhanced low-frequency ultrasound (MELFUS). The mice generated lesions of NMOSD with ON and myelitis. The mice also showed optic nerve and spinal cord inflammation associated with loss of AQP4 and GFAP expression in the brain, spinal cord, and optic nerve [174].
Many of the experimental animal models of NMOSD were established by passive administration of AQP4-IgG and T cells to rodent models using various methods [175]. AQP4 is an important drug target in NMOSD pathogenesis, and the assembly of AQP4 in OAPs is prudent for AQP4-IgG pathogenicity [176]. NMO animal models were developed in animals with pre-existing experimental autoimmune encephalomyelitis (EAE) with similar CNS inflammation and BBB injury pathogenicity.
Myelin basic protein immunization induced EAE in rats. Other possible methods of generating NMO models involve intraperitoneal injections of AQP4-IG that have been purified from the NMO patient serum and retrobulbar administration of recombinant-AQP4-IgG in rats with EAE [170], intrathecal administration of AQP4-IgG at the atlanto-occipital joint [177], continuous intraventricular perfusion of AQP4-IgG by osmotic-mini pump in mice for 7 days [38,178], continuous intracerebral infusion of AQP4-IgG and human complement factors [173], and continuous intracranial infusion of AQP4-IgG into the optic chiasm [38]. Though the development of animal models for NMOSD pathogenesis has improved in recent years, some issues still need to be rectified, such as developing a model without spontaneous AQP4 autoimmunity, possibly due to the different neuron-to-astrocyte ratios in rodents and differential AQP4 expression and availability [175].
In addition to animal models, culture models of CNS tissues and retinal cultures were also developed to understand NMO mechanisms. The expression of AQP4 was reduced after AQP4-IgG exposure in retinal culture [179]. Ex vivo spinal cord slice cultures were made by culturing vibratome-cut spinal cord slices from adult mice on transwell porous supports. The spinal cord cultures exposed to AQP4-IgG and the complement for 1 to 3 days showed significant loss of GFAP, AQP4, and myelin. The NMO pathology in these cultures was enhanced while incubated with macrophages, neutrophils, and inflammatory factors such as IL-6, TNF-α, IL-1β, and interferon-γ (IFN- γ) [170]. Likewise, several other cultures were established to study NMOSD pathology, including the cultures of optic nerves, hippocampal slices, and cerebellar slice cultures [180].
Marignier and his team developed a new model of NMOSD by prolonged infusion of AQP4-IgG into the CSF of live rats. The brain, spinal cord, and optic nerves alter astrocytes, myelin, and loss of AQP4 expression in the rats’ optic nerves and spinal cord. Reduced expression of myelin binding protein was observed in the grey and white matter and certain motor deficits were observed. However, the NMO rat model showed exclusions such as the absence of immune cell infiltration, microglial activation, and complement deposition in the CNS [178].
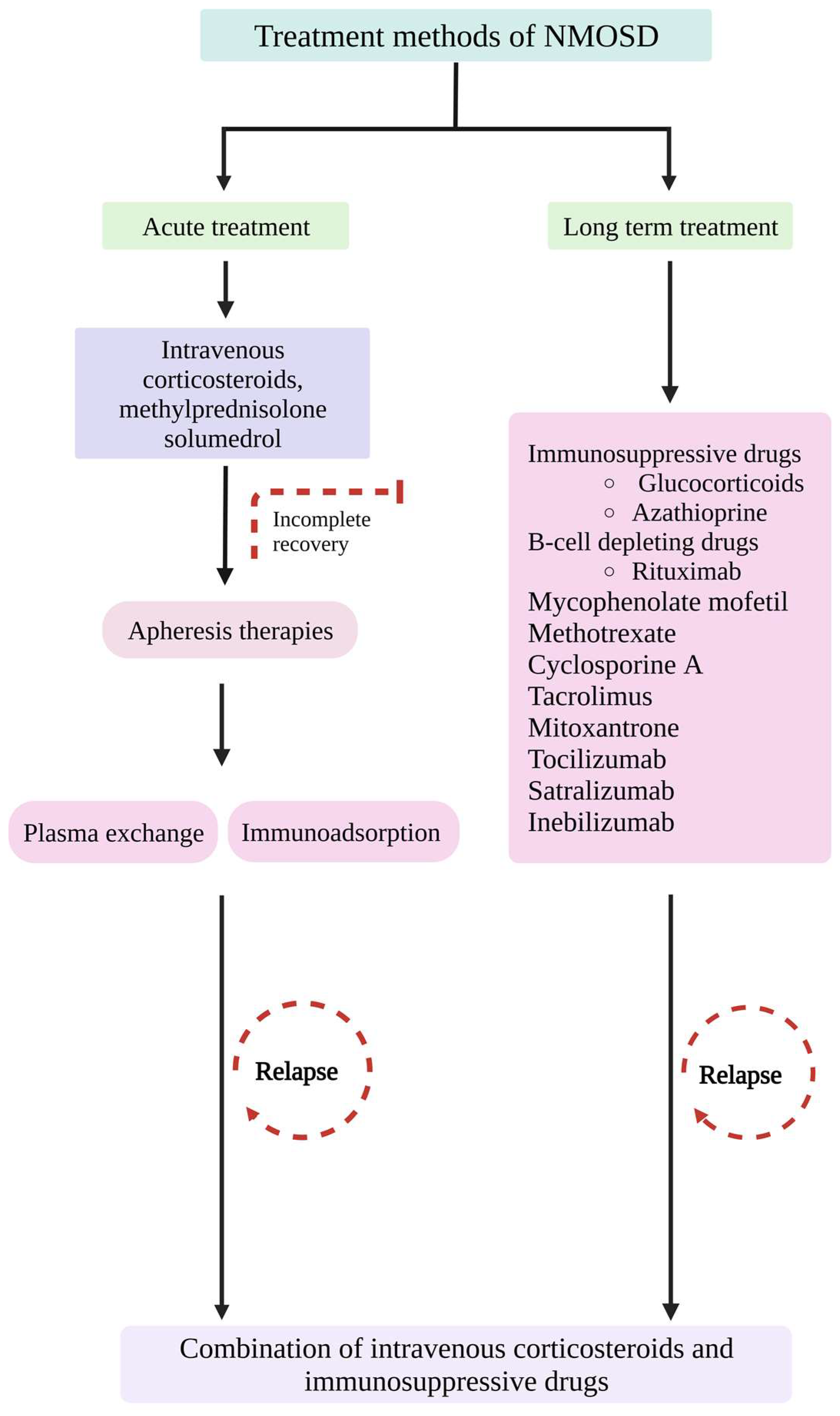
7. Treatment Methods
Intravenous methylprednisolone (IVMP) treatment inhibits inflammatory cascade reactions by inhibiting pro-inflammatory cytokine production, increasing anti-inflammatory cytokine levels, suppressing T cell activation by inducing T cell apoptosis, and causing impairment of granulocyte migration and downregulation of expression of cell adhesion molecules, receptors, and matrix metalloproteinases [181]. IVMP could significantly improve visual acuity and facilitate retention of the thickness of the retinal nerve fibre [7,182].
Corticosteroids are used to treat acute inflammation in NMO. A high dose of corticosteroids suppresses the acute phase of the inflammatory response [183]. A high dose of corticosteroid, solumedrol, is given to NMO patients with acute demyelination. Corticosteroids can cause immunosuppression and anti-inflammation by reducing peripheral lymphocytes and inflammatory cytokines [184].
In cases of poor or minimal response to steroid pulse therapy, apheresis therapies are recommended, wherein the pathogenic antibodies and proteins are removed from the patient’s blood circulation [185,186]. Plasma exchange (PLEX) and immunoadsorption (IA) are the two major apheresis therapies used in NMOSD. PLEX involves the removal of AQP4 autoantibodies and proinflammatory cytokines and regulates the proliferation and function of lymphocytes [187]. PLEX has side effects, including infections due to invasive techniques, hypotension, coagulopathies, and electrolyte imbalances [9]. In combination with IVMP, PLEX therapy confers better improvement in cases of severe spinal impairment and visual acuity in ON [188]. In cases such as corticosteroid-resistant relapses and NMO patients, PLEX treatment can be given in addition to IVMP to suppress severe spinal attacks [189] and to improve visual acuity in ON [190].
IA is an alternative apheresis technique that deals with albumin and clotting factors [191]. Both the PLEX and IA are effective treatments for NMOSD with myelitis [192]. Studies indicate that in case of severe relapse of NMOSD, the combination of corticosteroids and apheresis therapy exhibits better outcomes [193,194]. Thus, instead of providing a single therapy at a time, combinatorial therapies might be considered a better treatment procedure in case of an acute attack of NMOSD.
Apart from the treatment mentioned above, steroid therapy, apheresis therapies, and the intravenous immunoglobulins (IVIg) method can be used to treat NMOSD attacks. IVIg exerts immunomodulatory effects such as modulation of immune effector cells, blocking cellular receptors, and neutralizing cytokines, complementing molecules and autoantibodies [195]. IVIg reduces the relapse rate in NMOSD and can be used as a treatment procedure for NMOSD patients with repeated infections [9]. In NMOSD patients, IVIg treatment has proved promising as it could reduce AQP4 antibody levels [196] and neutralize the B cell activating factor at the onset of NMOSD [197]. Furthermore, IVIg interferes with administered IgG through antigen recognition, suppression of T cell activation, and downregulation of cytokine networks and adhesion molecules. To conclude, when there is low or insignificant remission in the case of corticosteroid therapy and apheresis therapies, IVIg could be an alternative treatment option [198].
7.1. Immunosuppressive Treatment Methods
Several drugs have been used in treating NMOSD, including immunosuppressant drugs such as glucocorticoids and azathioprine (AZA) and B cell-depleting drugs such as rituximab (RTX), mycophenolate mofetil (MMF), methotrexate, cyclosporine A, tacrolimus, mitoxantrone (MTX), IL-6 signalling blocking agents (tocilizumab and satralizumab), B cell inactivating agents (ocrelizumab and inebilizumab), and complement pathway blocking drugs (eculizumab and IVIgs) [9,199]. A randomized clinical phase 2 trial was conducted among 118 NMOSD patients with anti-IL-6 receptor antibodies tocilizumab (TCZ) and AZA. The results revealed that TCZ was effective and safe compared to AZA in preventing relapses [200].
B cells are involved in NMOSD pathogenesis. B cells and CD19, CD23, and CD38 may increase peripheral blood circulation and reduce disease relapse in NMOSD patients [121]. AZA, a purine analogue, could effectively reduce relapse and restore neurological functions in NMOSD patients by inhibiting B and T cell proliferation and the induction of anti-inflammatory actions. AZA could be used along with corticosteroids for better results [201]. RTX is a monoclonal antibody that targets CD20 by displaying pre, mature, and memory B cells, resulting in the depletion of CD20+ B cells and a reduction in pathogenic antibody production [199].
MMF improves clinical symptoms and suppresses disease activities and relapse. MMF inhibits monophosphate dehydrogenase, which involves guanosine nucleotide synthesis, and suppresses T and B cell synthesis and proliferation. MMF is an effective therapy compared to RTX and AZA, but has limitations such as teratogenicity and malignancy risk that need more clarification. Viral and bacterial infections are the side effects of MMF therapy [9]. MMF is more specific in targeting guanosine than AZA. The production and incorporation of thioguanosine into DNA results in treatment-induced lymphoma, as was reported in some AZA-treated cases. Thus, MMF is safe and efficient compared to AZA [202]. After three years of treatment with AZA, RTX, and MMF treatments, NMOSD patients revealed that RTX was more effective than MMF. AZA and RTX showed similar effects in reducing relapses and antibody levels [199]. Another potent immunosuppressive drug, MTX, acts as a DNA intercalating agent and causes structural disruption of DNA. It also negatively modulates DNA repair by inhibiting the repair enzyme topoisomerase II. MTX inhibits the proliferation of lymphocytes and macrophages [203,204,205].
Methotrexate is another common drug used for NMOSD and can be prescribed to long-term immunosuppressive patients. Methotrexate reduces acute relapses and is considered a safe drug for long-term usage. It suppresses the immune response in AQP4-IgG-seropositive patients by inhibiting dihydrofolate reductase (DHFR), which is critical in synthesizing nucleic acid precursors and causes T cell apoptosis [206,207].
One more pathological feature of NMOSD is the terminal deposits of complement factors as lesions. This can result in CNS inflammation and astrocyte damage. Eculizumab treatment significantly reduced the number of relapses in NMOSD patients [208]. However, eculizumab treatment promotes meningococcal and upper respiratory tract infections and pulmonary empyema [208].
The report says that the humanized monoclonal antibody satralizumab targets the IL-6 receptor but does not reduce pain and fatigue in NMOSD patients [209]. Satralizumab treatment in seropositive and seronegative AQP4 patients showed that it was ineffective in AQP4 seronegative patients [210]. and potently active in seropositive patients [210]. Other drugs, including inebilizumab and ocrelizumab, were also used to treat NMOSD. Several studies reported that antibodies targeting B cell and IL-6 receptors (such as RTX, inebilizumab, ocrelizumab, tocilizumab, and satralizumab) and the complement inhibitor (Eculizumab) diminish the progress of the disease [211].
Targeting B cell and IL-6 receptor and complement factors are included in biological therapies. The treatment gateways of NMOSD are described in the flowchart (Figure 5). The immunosuppressive drugs affect multiple events in the immune cascade, which could cause deleterious effects on the immune signals and be fatal in some cases. Thus, more specific therapeutic agents targeting B cells, plasma cells, and downstream effectors are important for treating NMOSD with increased specificity and reduced toxicity.
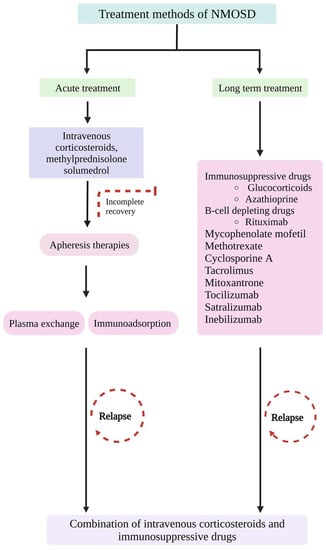
Figure 5.
The illustration describes the pharmacological treatment methods for neuromyelitis optical spectrum disorder (NMOSD). (Figure created using BioRender.com; accessed on 11 April 2023).
7.2. Blocking and Inactivation of AQP4, IL-6, and Complement Factors
The single target antibody AQP4 could be used to treat NMOSD. Blocking the cell surface polyclonal AQP4 antibodies with engineered, high-affinity antibodies prevents downstream cytotoxicity and reduces NMO lesions [212]. The binding of pathogenic AQP4-IgG with the AQP4 channel on the astrocyte initiates pathogenesis in AQP4 seropositive NMOSD cases. Inactivation, deletion, or blocking of the AQP4-IgG binding with its respective antigen using a small molecule can be used as a therapeutic method. Engineered monoclonal antibodies block AQP4-IgG from binding to AQP4 with high efficacy in NMOSD seropositive animal models [212,213].
Bacterial enzymes are used to inactivate AQP4-IgG. Endoglycosidase S (Endo S) and IgG degrading enzyme (IdeS) from Streptococcus pyogenes help digest asparagine-linked glycans on heavy chains of IgG [214] and cleave IgG antibodies into non-pathogenic Fc and F(ab) fragments, which inhibits AQP4-IgG from binding with AQP4 [215]. The administration of Endo S and IdeS through the apheresis method would prevent the disease. A high-affinity antibody (aquaporumab) blocked AQP4-IgG and prevented CDC in seropositive NMOSD patients. Thus, intravenous antibody-blocking therapy could reduce NMO pathology and the frequency of exacerbations [212]. Targeting the neonatal Fc receptor (FcRn) of AQP4 by its inhibitor (rozanolixizumab) reduced IgG concentration in human subjects [216]. AQP4 immunoadsorption-based removal of circulatory AQP4-IgG by AQP4-affinity resin, tryptophan, or phenylalanine-linked polyvinyl alcohol needs to be further studied before use [217,218].
Targeting IL-6 and blocking its functions through the use of tocilizumab, an antibody used to block the IL-6 receptor, reduces the frequency of relapse in MOGAD cases that did not respond to other immunotherapies [219]. Satralizumab is another drug engineered to dissociate IL-6 from its receptor in a pH-dependent manner, which promotes lysosomal degradation of the IL-6 receptor [220]. However, satralizumab was ineffective in AQP4-IgG seronegative NMOSD cases [209,210]. Upper respiratory tract infection, nasopharyngitis, and headaches are the adverse side effects of satralizumab [221]. Therefore, certain pretreatment procedures are needed before satralizumab treatment, including screening for the hepatitis B virus, tuberculosis, liver transaminases, and serum bilirubin [221].
Blocking the IL-6 receptor through the use of tocilizumab has been approved for treating rheumatoid arthritis and NMOSD [222]. Tocilizumab could bind to IL-6R in the astrocyte or T cell membrane and initiate downstream signalling cascades such as JAK/STAT, mitogen-activated protein kinase, and the phosphatidylinositol 3 kinase (PI3k) pathway [223].
7.3. Stem Cell Therapy
The administration of corticosteroids and immunosuppressants has not provided a complete cure for NMOSD, and long-time drug intake might invite serious side effects. Alternative therapies, such as stem cell therapies, are becoming increasingly popular. Stem cell therapy has improved motor functions in animal models of Parkinson’s disease [224]. Self-renewable neural stem cells derived from fetal tissues and embryonic stem cells as replacement strategies are being researched in relation to spinal injuries. Transplanting human-induced pluripotent stem cells improved spinal cord injuries in a mouse model by recovering the motor activities of the hind limbs [225].
Treatment using hematopoietic stem cell transplantation (HSCT) is being considered for certain autoimmune disorders such as MS and NMO. Autologous peripheral hematopoietic stem cell transplantation in NMO patients showed improved clinical remission, reduced frequent attacks, or no relapse and reduced disability. Additionally, composed MRI signals revealed neurological development after HSCT [226]. HSCT inhibits the cytotoxic CD4+ T cells and enhances other T cell subgroups such as CD3+ and CD8+. The study showed beneficial visual acuity in NMO patients [226]. However, earlier diagnosis and treatment with stem cell grafts might help facilitate a successful recovery. Zhang and Liu performed a meta-analysis of the treatment results of autologous HSCT on MS and NMOSD, and they concluded that AHSCT imparts long-term effects and is highly safe for use in both MS and NMOSD patients [227]. The peripheral blood stem cell translation improved the health status of about 80% of AQP4 seropositive and seronegative NMOSD patients [228]. Treatment with stem cell transplantation or neural grafting has limitations, such as the limited supply of donor cells, surgical difficulties, and graft rejection-related side effects [224].
8. Conclusions and Future Perspectives
Genetic predispositions or any unfavourable epigenetic modulations might trigger NADs. The possible interplay between previous infections or morbidities has also been considered. The immune response to an autoantibody requires information about cascade signalling and successive metamorphic changes in the numerous immune targets such as B cells, T cells, interleukins, and other proteins. The identification of unique biomarkers strengthens NMOSD and MS disease differentiation. Proteomic analysis of serum samples of NMO, NMOSD, and MS subjects suggests the use of composite biomarkers to discriminate among these diseases. Metabolomic studies reveal NMOSD’s pathophysiology and aid in its diagnosis and treatments.
Due to differences in the serotypes and clinical manifestations of NMOSD, developing therapy methods is complicated, and further in-depth studies are necessary. Except for the pathogenesis due to autoantibodies against AQP4, the autoantibodies against the MOG antigen and other blockers of interleukins might also be given more importance clinically to achieve therapeutic efficacies. In addition to the immunosuppressive therapies targeting the complement cascade system, B cells, interleukins, other inflammatory cytokines, and anti-AQP4 and anti-MOG antibodies, more scientific evidence is needed for personalized therapies for NMOSD. Protecting the BBB functions through enhancing immune tolerance in immunosuppressive patients could facilitate the development of neuroprotection and prevent the invasion of inflammatory cells from the CNS. Studies on combinatorial therapies using immunosuppressive drugs, plasmapheresis, or complement inhibition in animal models might reveal potent treatment strategies to manage NMOSD. In addition to the clinical application of advanced stem cell therapies, the pros and cons of stem cell transplantation and the key problems arising due to different sources of donor cells are also tough strategies. Thus, optimized stem cell therapy might be one of the prominent future treatment options for NMOSD.
Damage to the intestinal barrier, gut dysbiosis, and inflammation are the primary pathological events in NMOSD. The imbalance in the mucosal microbiota produces inflammatory responses, damaging the gut barrier and allowing pathogens, endotoxins, and microbial metabolites to cross the barrier and reach the BBB. Understanding the role of gut microbiota in NMOSD might provide insights into employing microbial intervention (for example, probiotics) as an added therapeutic strategy for NMOSD.
Author Contributions
Conceptualization, C.C., B.S.S. and P.K.; methodology, S.T. and B.S.S.; validation, C.C., B.S.S. and P.K.; formal analysis, S.T. and P.K.; investigation, S.T. and B.S.S.; resources, C.C.; data curation, S.T. and A.R.; writing—original draft preparation, S.T., B.S.S., A.R., P.K. and C.C.; writing—review and editing, S.T., B.S.S., P.K. and C.C.; supervision, C.C. and B.S.S.; project administration, C.C.; funding acquisition, C.C. All authors have read and agreed to the published version of the manuscript.
Funding
This project was supported by Chiang Mai University, Chiang Mai, Thailand.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Acknowledgments
The authors gratefully acknowledge Chiang Mai University, Chiang Mai, for the support. S.T. thankfully acknowledges the CMU post-doctoral fellowship for its support.
Conflicts of Interest
The authors declare no conflict of interest.
Abbreviations
| NMO | Neuromyelitis optica |
| CNS | Central nervous system |
| PNS | Peripheral nervous system |
| ON | Optic neuritis |
| MS | Multiple sclerosis |
| AQP4 | Aquaporin 4 |
| AQP4-ab | Aquaporin 4- antibody |
| CSF | Cerebrospinal fluid |
| BBB | Blood–brain barrier |
| CDC | Complement dependent cytotoxicity |
| ADCC | Antibody-dependent cellular cytotoxicity |
| MAC | Membrane attack complex |
| AD | Autoimmune diseases |
| NAD | Neurological autoimmune diseases |
| ADEM | Acute disseminated encephalomyelitis |
| LTEM | Longitudinally extensive transverse myelitis |
| SLE | Systemic lupus erythematosus |
| GBS | Guillain-Barré syndrome |
| MG | Myasthenia gravis |
| SS | Sjogren’s syndrome |
| NMOSD | Neuromyelitis optica spectrum disorder |
| Tfh | T follicular helper cells |
| MOGAD | Myelin oligodendrocyte glycoprotein-associated disorders |
| CXCR5 | Chemokine C-X-C receptor 5 |
| CXCL13 | C-X-C motif ligand 13 |
| OAP | Orthogonal array proteins |
| MRI | Magnetic resonance imaging |
| NMR | Nuclear magnetic resonance |
| HDL | High-density lipoprotein |
| GFAP | Glial fibrillary acidic protein |
| ASC | Antibody secreting cell |
| NK | Natural killer cell |
| IL | Interleukin |
| JAK/STAT3 | Janus kinase/Signal transducer and activator of the transcription 3 protein |
| NFkB | Nuclear factor kappa-light-chain-enhancer of activated B cells |
| INF γ | Interferon-gamma |
| TNF α | Tumour necrosis factor-alpha |
| Th17 | T helper cell 17 |
| Treg | Regulatory T cell |
| P2R | Purinergic receptors |
| GM | Gut microbiota |
| ABC-TP | Adenosine triphosphate-binding cassette transporter permease |
| EAE | Experimental autoimmune encephalomyelitis |
| IVMP | Intravenous methylprednisolone |
| PLEX | Plasma exchange |
| IA | Immunoadsorption |
| IVIg | Intravenous immunoglobulins |
| AZA | Azathioprine |
| RTX | Rituiximab |
| MTX | Mitoxantrone |
| TCZ | Tocilizumab |
| MMF | Mycophenolate mofetil |
| DHFR | Dihydrofolate reductase |
| FcRn | Neonatal Fc receptor |
| HSCT | Hematopoietic stem cell transplantation |
| AHSCT | Autologous hematopoietic stem cell transplantation |
| P13k | Phosphatidylinositol 3 kinase |
References
- Anaya, J.M.; Ramirez-Santana, C.; Alzate, M.A.; Molano-Gonzalez, N.; Rojas-Villarraga, A. The Autoimmune Ecology. Front. Immunol. 2016, 7, 139. [Google Scholar] [CrossRef]
- Acosta-Ampudia, Y.; Monsalve, D.M.; Ramírez-Santana, C. Identifying the culprits in neurological autoimmune diseases. J. Transl. Autoimmun. 2019, 2, 100015. [Google Scholar] [CrossRef]
- Pierrot-Deseilligny, C.; Souberbielle, J.C. Vitamin D and multiple sclerosis: An update. Mult. Scler. Relat. Disord. 2017, 14, 35–45. [Google Scholar] [CrossRef] [PubMed]
- Rubin, D.B.; Batra, A.; Vaitkevicius, H.; Vodopivec, I. Autoimmune Neurologic Disorders. Am. J. Med. 2018, 131, 226–236. [Google Scholar] [CrossRef]
- Kawachi, I.; Lassmann, H. Neurodegeneration in multiple sclerosis and neuromyelitis optica. J. Neurol. Neurosurg. Psychiatry 2017, 88, 137–145. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.; Lennon, V.A.; Parisi, J.E.; Popescu, B.; Vasquez, C.; Pittock, S.J.; Howe, C.L.; Lucchinetti, C.F. Spectrum of sub lytic astrocytopathy in neuromyelitis optica. Brain 2022, 145, 1379–1390. [Google Scholar] [CrossRef]
- Carnero Contentti, E.; Correale, J. Neuromyelitis optica spectrum disorders: From pathophysiology to therapeutic strategies. J. Neuroinflamm. 2021, 18, 208. [Google Scholar] [CrossRef]
- Papadopoulos, M.C.; Verkman, A.S. Aquaporin 4 and Neuromyelitis Optica. Lancet Neurol. 2012, 11, 535–544. [Google Scholar] [CrossRef]
- Chan, K.H.; Lee, C.Y. Treatment of Neuromyelitis Optica Spectrum Disorders. Int. J. Mol. Sci. 2021, 22, 8638. [Google Scholar] [CrossRef] [PubMed]
- Wingerchuk, D.M. Neuromyelitis optica: New findings on pathogenesis. Int. Rev. Neurobiol. 2007, 79, 665–688. [Google Scholar] [PubMed]
- Jarius, S.; Wildemann, B. The history of neuromyelitis optica. J. Neuroinflamm. 2013, 10, 797. [Google Scholar] [CrossRef] [PubMed]
- Wingerchuk, D.M.; Lennon, V.A.; Lucchinetti, C.F.; Pittock, S.J.; Weinshenker, B.G. The spectrum of neuromyelitis optica. Lancet Neurol. 2007, 6, 805–815. [Google Scholar] [CrossRef] [PubMed]
- Lennon, V.A.; Wingerchuk, D.M.; Kryzer, T.J.; Pittock, S.J.; Lucchinetti, C.F.; Fujihara, K.; Nakashima, I.; Weinshenker, B.G. A serum autoantibody marker of neuromyelitis optica: Distinction from multiple sclerosis. Lancet 2004, 364, 2106–2112. [Google Scholar] [CrossRef]
- Lennon, V.A.; Kryzer, T.J.; Pittock, S.J.; Verkman, A.S. Hinson SR. IgG marker of optic-spinal multiple sclerosis binds to the aquaporin-4 water channel. J. Exp. Med. 2005, 202, 473–477. [Google Scholar] [CrossRef] [PubMed]
- Dos Passos, G.R.; Oliveira, L.M.; da Costa, B.K.; Apostolos-Pereira, S.L.; Callegaro, D.; Fujihara, K.; Sato, D.K. MOG-IgG-Associated Optic Neuritis, Encephalitis, and Myelitis: Lessons Learned From Neuromyelitis Optica Spectrum Disorder. Front. Neurol. 2018, 9, 217. [Google Scholar] [CrossRef] [PubMed]
- Spiezia, A.L.; Carotenuto, A.; Iovino, A.; Moccia, M.; Gastaldi, M.; Iodice, R.; Tedeschi, E.; Petracca, M.; Lavorgna, L.; d’Ambrosio, A.; et al. AQP4-MOG Double-Positive Neuromyelitis Optica Spectrum Disorder: Case Report with Central and Peripheral Nervous System Involvement and Review of Literature. Int. J. Mol. Sci. 2022, 23, 14559. [Google Scholar] [CrossRef]
- Wingerchuk, D.M.; Banwell, B.; Bennett, J.L.; Cabre, P.; Carroll, W.; Chitnis, T.; de Seze, J.; Fujihara, K.; Greenberg, B.; Jacob, A.; et al. International consensus diagnostic criteria for neuromyelitis optica spectrum disorder. Neurology 2015, 85, 177–189. [Google Scholar] [CrossRef]
- Mirsattari, S.M.; Johnston, J.B.; McKenna, R.; Del Bigio, M.R.; Orr, P.; Ross, R.T.; Power, C. Aboriginals with multiple sclerosis: HLA types and predominance of neuromyelitis optica. Neurology 2001, 56, 317–323. [Google Scholar] [CrossRef]
- Gontika, M.P.; Anagnostouli, M.C. Human leukocyte antigens-immunogenetics of neuromyelitis optica or Devic’s disease and the impact on the immunopathogenesis, diagnosis and treatment: A critical review. Neuroimmunol. Neuroinflamm. 2014, 1, 44–50. [Google Scholar] [CrossRef][Green Version]
- Brum, D.G.; Barreira, A.A.; dos Santos, A.C.; Kaimen-Maciel, D.R.; Matiello, M.; Costa, R.M.; Deghaide, N.H.; Costa, L.S.; Louzada-Junior, P.; Diniz, P.R.; et al. HLA-DRB association in neuromyelitis optica is different from that observed in multiple sclerosis. Mult. Scler. 2010, 16, 21–29. [Google Scholar] [CrossRef]
- Deschamps, R.; Paturel, L.; Jeannin, S.; Chausson, N.; Olindo, S.; Béra, O.; Bellance, R.; Smadja, D.; Césaire, D.; Cabre, P. Different HLA class II (DRB1 and DQB1) alleles determine either susceptibility or resistance to NMO and multiple sclerosis among the French Afro-Caribbean population. Mult. Scler. 2011, 17, 24–31. [Google Scholar] [CrossRef]
- Collongues, N.; Marignier, R.; Zéphir, H.; Papeix, C.; Blanc, F.; Ritleng, C.; Tchikviladzé, M.; Outteryck, O.; Vukusic, S.; Fleury, M. Neuromyelitis optica in France: A multicenter study of 125 patients. Neurology 2010, 74, 736–742. [Google Scholar] [CrossRef] [PubMed]
- Yoshimura, S.; Isobe, N.; Yonekawa, T.; Matsushita, T.; Masaki, K.; Sato, S.; Kawano, Y.; Yamamoto, K.; Kira, J. South Japan Multiple Sclerosis Genetics Consortium. Genetic and infectious profiles of Japanese multiple sclerosis patients. PLoS ONE 2012, 7, e48592. [Google Scholar] [CrossRef] [PubMed]
- Isobe, N.; Oksenberg, J.R. Genetic studies of multiple sclerosis and neuromyelitis optica: Current status in European, African American and Asian populations. Clin. Exp. Neuroimmunol. 2014, 5, 61–68. [Google Scholar] [CrossRef][Green Version]
- Wei, Q.; Chang, Y.; Rui, L.; Caixia, L.; Long, Y.; Huang, J.; Mai, W.; Sun, X.; Xu, W.; Chen, Y.; et al. Human aquaporin 4 gene polymorphisms in Chinese patients with neuromyelitis optica. J. Neuroimmunol. 2014, 274, 192–196. [Google Scholar] [CrossRef]
- Wang, H.; Zhong, X.; Wang, K.; Qiu, W.; Li, J.; Dai, Y.; Hu, X. Interleukin 17 gene polymorphism is associated with anti-aquaporin 4 antibody-positive neuromyelitis optica in the Southern Han Chinese–A case control study. J. Neurol. Sci. 2012, 314, 26–28. [Google Scholar] [CrossRef]
- Asgari, N.; Nielsen, C.; Stenager, E.; Kyvik, K.O.; Lillevang, S.T. HLA, PTPN22 and PD-1 associations as markers of autoimmunity in neuromyelitis optica. Mult. Scler. 2012, 18, 23–30. [Google Scholar] [CrossRef]
- Xu, J.; Zhang, F.; Gao, C.; Ma, X.; Peng, X.; Kong, D.; Hao, J. Microarray Analysis of lncRNA and mRNA Expression Profiles in Patients With Neuromyelitis Optica. Mol. Neurobiol. 2017, 54, 2201–2208. [Google Scholar] [CrossRef]
- Vaknin-Dembinsky, A.; Charbit, H.; Brill, L.; Abramsky, O.; Gur-Wahnon, D.; Ben-Dov, I.Z.; Lavon, I. Circulating microRNAs as Biomarkers for Rituximab Therapy, in Neuromyelitis Optica (NMO). J. Neuroinflamm. 2016, 13, 179. [Google Scholar] [CrossRef]
- Thoman, M.E.; McKarns, S.C. Metabolomic Profiling in Neuromyelitis Optica Spectrum Disorder Biomarker Discovery. Metabolites 2020, 10, 374. [Google Scholar] [CrossRef]
- Cheng, X.; Zhou, L.; Li, Z.; Shen, S.; Zhao, Y.; Liu, C.; Zhong, X.; Chang, Y.; Kermode, A.G.; Qiu, W. Gut Microbiome and Bile Acid Metabolism Induced the Activation of CXCR5+ CD4+ T Follicular Helper Cells to Participate in Neuromyelitis Optica Spectrum Disorder Recurrence. Front. Immunol. 2022, 13, 827865. [Google Scholar] [CrossRef] [PubMed]
- Fan, X.; Jiang, Y.; Han, J.; Liu, J.; Wei, Y.; Jiang, X.; Jin, T. Circulating Memory T Follicular Helper Cells in Patients with Neuromyelitis Optica/Neuromyelitis Optica Spectrum Disorders. Mediat. Inflamm. 2016, 2016, 3678152. [Google Scholar] [CrossRef] [PubMed]
- Cui, C.; Ruan, Y.; Qiu, W. Potential role of the gut microbiota in neuromyelitis optica spectrum disorder: Implication for intervention. J. Clin. Neurosci. 2020, 82, 193–199. [Google Scholar] [CrossRef]
- Pandit, L.; Cox, L.M.; Malli, C.; D’Cunha, A.; Rooney, T.; Lokhande, H.; Willocq, V.; Saxena, S.; Chitnis, T. Clostridium bolteae is elevated in neuromyelitis optica spectrum disorder in India and shares sequence similarity with AQP4. Neurol. Neuroimmunol. Neuroinflamm. 2020, 8, e907. [Google Scholar] [CrossRef] [PubMed]
- Roemer, S.F.; Parisi, J.E.; Lennon, V.A.; Benarroch, E.E.; Lassmann, H.; Bruck, W.; Mandler, R.N.; Weinshenker, B.G.; Pittock, S.J.; Wingerchuk, D.M.; et al. Pattern-Specific Loss of Aquaporin-4 Immunoreactivity Distinguishes Neuromyelitis Optica from Multiple Sclerosis. Brain 2007, 130, 1194–1205. [Google Scholar] [CrossRef]
- Scharfman, H.E.; Binder, D.K. Aquaporin-4 water channels and synaptic plasticity in the hippocampus. Neurochem. Int. 2013, 63, 702–711. [Google Scholar] [CrossRef]
- Vincent, T.; Saikali, P.; Cayrol, R.; Roth, A.D.; Bar-Or, A.; Prat, A.; Antel, J.P. Functional consequences of neuromyelitis optica-IgG astrocyte interactions on blood-brain barrier permeability and granulocyte recruitment. J. Immunol. 2008, 181, 5730–5737. [Google Scholar] [CrossRef]
- Asavapanumas, N.; Ratelade, J.; Papadopoulos, M.C.; Bennett, J.L.; Levin, M.H.; Verkman, A.S. Experimental Mouse Model of Optic Neuritis with Inflammatory Demyelination Produced by Passive Transfer of Neuromyelitis Optica-Immunoglobulin, G. J. Neuroinflamm. 2014, 11, 16. [Google Scholar] [CrossRef]
- Matiello, M.; Schaefer-Klein, J.; Sun, D.; Weinshenker, B.G. Aquaporin 4 Expression and Tissue Susceptibility to Neuromyelitis Optica. JAMA Neurol. 2013, 70, 1118–1125. [Google Scholar] [CrossRef]
- Abe, Y.; Yasui, M. Aquaporin-4 in Neuromyelitis Optica Spectrum Disorders: A Target of Autoimmunity in the Central Nervous System. Biomolecules 2022, 12, 591. [Google Scholar] [CrossRef]
- Mckeon, A.; Lennon, V.A.; Lotze, T.; Tenenbaum, S.; Ness, J.M.; Rensel, M.; Kuntz, N.L.; Fryer, J.P.; Homburger, H.; Hunter, J.; et al. CNS Aquaporin-4 Autoimmunity in Children. Neurology 2008, 71, 93–100. [Google Scholar] [CrossRef] [PubMed]
- Phuan, P.W.; Ratelade, J.; Rossi, A.; Tradtrantip, L.; Verkman, A.S. Complement-Dependent Cytotoxicity in Neuromyelitis Optica Requires Aquaporin-4 Protein Assembly in Orthogonal Arrays. J. Biol. Chem. 2012, 287, 13829–13839. [Google Scholar] [CrossRef]
- Matiello, M.; Schaefer-Klein, J.L.; Hebrink, D.D.; Kingsbury, D.J.; Atkinson, E.J.; Weinshenker, B.G. NMO Genetics Collaborators. Genetic analysis of aquaporin-4 in neuromyelitis optica. Neurology 2011, 77, 1149–1155. [Google Scholar] [CrossRef]
- Hinson, S.R.; Romero, M.F.; Popescu, B.F.; Lucchinetti, C.F.; Fryer, J.P.; Wolburg, H.; Fallier-Becker, P.; Noell, S.; Lennon, V.A. Molecular Outcomes of Neuromyelitis Optica (NMO)-IgG Binding to Aquaporin-4 in Astrocytes. Proc. Natl. Acad. Sci. USA 2012, 109, 124–1250. [Google Scholar] [CrossRef]
- Satoh, J.; Obayashi, S.; Misawa, T.; Tabunoki, H.; Yamamura, T.; Arima, K.; Konno, H. Neuromyelitis Optica/Devic’s Disease: Gene Expression Profiling of Brain Lesions. Neuropathology 2008, 28, 561–576. [Google Scholar] [CrossRef] [PubMed]
- Rostásy, K.; Mader, S.; Hennes, E.M.; Schanda, K.; Gredler, V.; Guenther, A.; Blaschek, A.; Korenke, C.; Pritsch, M.; Pohl, D. Persisting myelin oligodendrocyte glycoprotein antibodies in aquaporin-4 antibody negative pediatric neuromyelitis optica. Mult. Scler. 2013, 19, 1052–1059. [Google Scholar] [CrossRef] [PubMed]
- Luppe, S.; Robertson, N.P. MOG-IgG in neuromyelitis optica. J. Neurol. 2014, 261, 640–642. [Google Scholar] [CrossRef] [PubMed]
- Höftberger, R.; Guo, Y.; Flanagan, E.P.; Lopez-Chiriboga, A.S.; Endmayr, V.; Hochmeister, S.; Joldic, D.; Pittock, S.J.; Tillema, J.M.; Gorman, M.; et al. The pathology of central nervous system inflammatory demyelinating disease accompanying myelin oligodendrocyte glycoprotein autoantibody. Acta Neuropathol. 2020, 139, 875–892. [Google Scholar] [CrossRef] [PubMed]
- Mader, S.; Gredler, V.; Schanda, K.; Rostasy, K.; Dujmovic, I.; Pfaller, K.; Lutterotti, A.; Jarius, S.; Di Pauli, F.; Kuenz, B.; et al. Complement activating antibodies to myelin oligodendrocyte glycoprotein in neuromyelitis optica and related disorders. J. Neuroinflamm. 2011, 8, 184. [Google Scholar] [CrossRef]
- Ruiz-Gaviria, R.; Baracaldo, I.; Castañeda, C.; Ruiz-Patiño, A.; Acosta-Hernandez, A.; Rosselli, D. Specificity and sensitivity of aquaporin 4 antibody detection tests in patients with neuromyelitis optica: A meta-analysis. Mult. Scler. Relat. Dis. 2015, 4, 345–349. [Google Scholar] [CrossRef]
- Whittam, D.; Wilson, M.; Hamid, S.; Keir, G.; Bhojak, M.; Jacob, A. What’s new in neuromyelitis optica? A short review for the clinical neurologist. J. Neurol. 2017, 264, 2330–2344. [Google Scholar] [CrossRef] [PubMed]
- Zhao, S.; Mutch, K.; Elsone, L.; Nurmikko, T.; Jacob, A. Neuropathic pain in neuromyelitis optica affects activities of daily living and quality of life. Mult. Scler. 2014, 20, 1658–1661. [Google Scholar] [CrossRef] [PubMed]
- Elsone, L.; Townsend, T.; Mutch, K.; Das, K.; Boggild, M.; Nurmikko, T.; Jacob, A. Neuropathic pruritis (itch) in neuromyelitis optica. Mult. Scler. 2012, 19, 475–479. [Google Scholar] [CrossRef]
- Kim, S.M.; Go, M.J.; Sung, J.J.; Park, K.S.; Lee, K.W. Painful tonic spasm in neuromyelitis optica: Incidence, diagnostic utility, and clinical characteristics. Arch. Neurol. 2012, 69, 1026–1031. [Google Scholar] [CrossRef]
- Sato, D.; Fukihara, K. Atypical presentations of neuromyelitis optica. Arq. Neuropsiquiatr. 2011, 69, 824–828. [Google Scholar] [CrossRef] [PubMed]
- Baba, T.; Nakashima, I.; Kanbayashi, T.; Konno, M.; Takahashi, T.; Fujihara, K.; Misu, T.; Takeda, A.; Shiga, Y.; Ogawa, H.; et al. Narcolepsy as an initial manifestation of neuromyelitis optica with antiaquaporin-4 antibody. J. Neurol. 2009, 256, 287–288. [Google Scholar] [CrossRef]
- Moore, P.; Methley, A.; Pollard, C.; Mutch, K.; Hamid, S.; Elsone, L.; Jacob, A. Cognitive and psychiatric comorbidities in neuromyelitis optica. J. Neurol. Sci. 2016, 360, 4–9. [Google Scholar] [CrossRef]
- Meng, H.; Xu, J.; Pan, C.; Cheng, J.; Hu, Y.; Hong, Y.; Shen, Y.; Dai, H. Cognitive dysfunction in adult patients with neuromyelitis optica: A systematic review and meta-analysis. J. Neurol. 2017, 264, 1549–1558. [Google Scholar] [CrossRef]
- Zekeridou, A.; Lennon, V.A. Aquaporin-4 autoimmunity. Neurol. Neuroimmunol. Neuroinflamm. 2015, 2, e110. [Google Scholar] [CrossRef]
- Pereira, W.L.; Reiche, E.M.; Kallaur, A.P.; Kaimen-Maciel, D.R. Epidemiological, clinical, and immunological characteristics of neuromyelitis optica: A review. J. Neurol Sci. 2015, 355, 7–17. [Google Scholar] [CrossRef]
- Pfueller, C.F.; Paul, F. Imaging the visual pathway in neuromyelitis optica. Mult. Scler. Int. 2011, 2011, 869814. [Google Scholar] [CrossRef]
- Lin, T.Y.; Chien, C.; Lu, A.; Paul, F.; Zimmermann, H.G. Retinal optical coherence tomography and magnetic resonance imaging in neuromyelitis optica spectrum disorders and MOG-antibody associated disorders: An updated review. Expert. Rev. Neurother. 2021, 21, 1101–1123. [Google Scholar] [CrossRef]
- Khanna, S.; Sharma, A.; Huecker, J.; Gordon, M.; Naismith, R.T.; Van Stavern, G.P. Magnetic Resonance Imaging of Optic Neuritis in Patients with Neuromyelitis Optica Versus Multiple Sclerosis. J. Neuro. Ophthalmol. 2012, 32, 216–220. [Google Scholar] [CrossRef] [PubMed]
- Crane, J.M.; Tajima, M.; Verkman, A.S. Live-cell imaging of aquaporin-4 diffusion and interactions in orthogonal arrays of particles. Neuroscience 2010, 168, 892–902. [Google Scholar] [CrossRef]
- Youl, B.D.; Turano, G.; Miller, D.H.; Towell, A.D.; MacManus, D.G.; Moore, S.G.; Jones, S.J.; Barrett, G.; Kendall, B.E.; Moseley, I.F.; et al. The pathophysiology of acute optic neuritis. An association of gadolinium leakage with clinical and electrophysiological deficits. Brain 1991, 114, 2437–2450. [Google Scholar] [CrossRef]
- Banker, P.; Sonni, S.; Kister, I.; Loh, J.P.; Lui, Y.W. Pencil-thin ependymal enhancement in neuromyelitis optica spectrum disorders. Mult. Scler. 2012, 18, 1050–1053. [Google Scholar] [CrossRef]
- Ito, S.; Mori, M.; Makino, T.; Hayakawa, S.; Kuwabara, S. ‘Cloud-like enhancement’ is a magnetic resonance imaging abnormality specific to neuromyelitis optica. Ann. Neurol. 2009, 66, 425–428. [Google Scholar] [CrossRef] [PubMed]
- Youl, B.D.; Turano, G.; Towell, A.D.; Barrett, G.; MacManus, D.G.; Moore, S.G.; Miller, D.H.; Jones, S.J.; du Boulay, E.P.; Kendall, B.E.; et al. Optic neuritis: Swelling and atrophy. Electroencephalogr. Clin. Neurophysiol. Suppl. 1996, 46, 173–179. [Google Scholar] [PubMed]
- Fadda, G.; Flanagan, E.P.; Cacciaguerra, L.; Jitprapaikulsan, J.; Solla, P.; Zara, P.; Sechi, E. Myelitis features and outcomes in CNS demyelinating disorders: Comparison between multiple sclerosis, MOGAD, and AQP4-IgG-positive NMOSD. Front. Neurol. 2022, 13, 1011579. [Google Scholar] [CrossRef] [PubMed]
- Ayzenberg, I.; Richter, D.; Henke, E.; Asseyer, S.; Paul, F.; Trebst, C.; Hümmert, M.W.; Havla, J.; Kümpfel, T.; Ringelstein, M.; et al. Pain, depression, and quality of life in neuromyelitis optica spectrum disorder: A crosssectional study of 166 AQP4 antibody-seropositive patients. Neurol. Neuroimmunol. Neuroinflamm. 2021, 8, e985. [Google Scholar] [CrossRef] [PubMed]
- Zhao-Fleming, H.H.; Valencia Sanchez, C.; Sechi, E.; Inbarasu, J.; Wijdicks, E.F.; Pittock, S.J.; Chen, J.J.; Wingerchuk, D.M.; Weinshenker, B.G.; Lopez-Chiriboga, S.; et al. CNS demyelinating attacks requiring ventilatory support with myelin oligodendrocyte glycoprotein or aquaporin-4 antibodies. Neurology 2021, 97, e1351–e1358. [Google Scholar] [CrossRef]
- Mariano, R.; Messina, S.; Kumar, K.; Kuker, W.; Leite, M.I.; Palace, J. Comparison of clinical outcomes of transverse myelitis among adults with myelin oligodendrocyte glycoprotein antibody vs. aquaporin-4 antibody disease. JAMA Netw. Open 2019, 2, e1912732. [Google Scholar] [CrossRef]
- Wingerchuk, D.M.; Lennon, V.A.; Pittock, S.J.; Lucchinetti, C.F.; Weinshenker, B.G. Revised diagnostic criteria for neuromyelitis optica. Neurology 2006, 66, 1485–1489. [Google Scholar] [CrossRef]
- Sechi, E.; Cacciaguerra, L.; Chen, J.J.; Mariotto, S.; Fadda, G.; Dinoto, A.; Lopez-Chiriboga, A.S.; Pittock, S.J.; Flanagan, E.P. Myelin Oligodendrocyte Glycoprotein Antibody-Associated Disease (MOGAD): A Review of Clinical and MRI Features, Diagnosis, and Management. Front. Neurol. 2022, 13, 885218. [Google Scholar] [CrossRef]
- Chen, J.J.; Flanagan, E.P.; Jitprapaikulsan, J.; Lopez-Chiriboga, A.S.S.; Fryer, J.P.; Leavitt, J.A.; Weinshenker, B.G.; Mckeon, A.; Tillema, J.M.; Lennon, V.A.; et al. Myelin Oligodendrocyte Glycoprotein Antibody-Positive Optic Neuritis: Clinical Characteristics, Radiologic Clues, and Outcome. Am. J. Ophthalmol. 2018, 195, 8–15. [Google Scholar] [CrossRef] [PubMed]
- Mealy, M.A.; Whetstone, A.; Orman, G.; Izbudak, I.; Calabresi, P.A.; Levy, M. Longitudinally extensive optic neuritis as an MRI biomarker distinguishes neuromyelitis optica from multiple sclerosis. J. Neurol. Sci. 2015, 355, 59–63. [Google Scholar] [CrossRef]
- Cobo-Calvo, A.; Ruiz, A.; Maillart, E.; Audoin, B.; Zephir, H.; Bourre, B.; Ciron, J.; Collongues, N.; Brassat, D.; Cotton, F. Clinical spectrum and prognostic value of CNS MOG autoimmunity in adults: The MOGADOR study. Neurology 2018, 90, e1858–e1869. [Google Scholar] [CrossRef]
- Jarius, S.; Ruprecht, K.; Kleiter, I.; Borisow, N.; Asgari, N.; Pitarokoili, K.; Pache, F.; Stich, O.; Beume, L.A.; Hümmert, M.W. MOG-IgG in NMO and related disorders: A multicenter study of 50 patients. Part 2: Epidemiology, clinical presentation, radiological and laboratory features, treatment responses, and long-term outcome. J. Neuroinflamm. 2016, 13, 280. [Google Scholar] [CrossRef] [PubMed]
- Jurynczyk, M.; Geraldes, R.; Probert, F.; Woodhall, M.R.; Waters, P.; Tackley, G.; DeLuca, G.; Chandratre, S.; Leite, M.I.; Vincent, A.; et al. Distinct brain imaging characteristics of autoantibody-mediated CNS conditions and multiple sclerosis. Brain 2017, 140, 617–627. [Google Scholar] [CrossRef]
- Salama, S.; Khan, M.; Shanechi, A.; Levy, M.; Izbudak, I. MRI differences between MOG antibody disease and AQP4 NMOSD. Mult. Scler. 2020, 26, 1854–1865. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Zhang, C.; Jia, D.; Fan, M.; Li, T.; Tian, D.C.; Liu, Y.; Shi, F.D. The Occurrence of Myelin Oligodendrocyte Glycoprotein Antibodies in Aquaporin-4-Antibody Seronegative Neuromyelitis Optica Spectrum Disorder: A Systematic Review and Meta-Analysis. Mult. Scler. Relat. Disor. 2021, 53, 103030. [Google Scholar] [CrossRef] [PubMed]
- Bulut, E.; Karakaya, J.; Salama, S.; Levy, M.; Huisman, T.A.G.M.; Izbudak, I. Brain MRI Findings in Pediatric-Onset Neuromyelitis Optica Spectrum Disorder: Challenges in Differentiation from Acute Disseminated Encephalomyelitis. Am. J. Neuroradiol. 2019, 40, 726–731. [Google Scholar] [CrossRef]
- Zhang, L.; Wu, A.; Zhang, B.; Chen, S.; Men, X.; Lin, Y.; Lu, Z. Comparison of deep gray matter lesions on magnetic resonance imaging among adults with acute disseminated encephalomyelitis, multiple sclerosis, and neuromyelitis optica. Mult. Scler. 2014, 20, 418–423. [Google Scholar] [CrossRef]
- Jayarangaiah, A.; Sehgal, R.; Epperla, N. Sjögren’s syndrome and neuromyelitis optica spectrum disorders (NMOSD)-a case report and review of literature. BMC Neurol. 2014, 14, 200. [Google Scholar] [CrossRef]
- Kim, S.M.; Waters, P.; Vincent, A.; Kim, S.Y.; Kim, H.J.; Hong, Y.H.; Park, K.S.; Min, J.H.; Sung, J.J.; Lee, K.W. Sjogren’s syndrome myelopathy: Spinal cord involvement in Sjogren’s syndrome might be a manifestation of neuromyelitis optica. Mult. Scler. 2009, 15, 1062–1068. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.M.; Kim, S.J.; Lee, H.J.; Kuroda, H.; Palace, J.; Fujihara, K. Differential diagnosis of neuromyelitis optica spectrum disorders. Ther. Adv. Neurol. Disord. 2017, 10, 265–289. [Google Scholar] [CrossRef] [PubMed]
- Jarius, S.; Paul, F.; Aktas, O.; Asgari, N.; Dale, R.C.; de Seze, J.; Franciotta, D.; Fujihara, K.; Jacob, A.; Kim, H.J. MOG encephalomyelitis: International recommendations on diagnosis and antibody testing. J. Neuroinflamm. 2018, 15, 134. [Google Scholar] [CrossRef] [PubMed]
- López-Chiriboga, A.S.; Majed, M.; Fryer, J.; Dubey, D.; McKeon, A.; Flanagan, E.P.; Jitprapaikulsan, J.; Kothapalli, N.; Tillema, J.M.; Chen, J. Association of MOG-IgG serostatus with relapse after acute disseminated encephalomyelitis and proposed diagnostic criteria for MOG-IgG-associated disorders. JAMA Neurol. 2018, 75, 1355–1363. [Google Scholar] [CrossRef]
- Salama, S.; Khan, M.; Pardo, S.; Izbudak, I.; Levy, M. MOG antibody-associated encephalomyelitis/encephalitis. Mult. Scler. 2019, 25, 1427–1433. [Google Scholar] [CrossRef] [PubMed]
- Tenembaum, S.N. Acute disseminated encephalomyelitis. Handb. Clin. Neurol. 2013, 112, 1253–1262. [Google Scholar]
- Pohl, D.; Alper, G.; Van Haren, K.; Kornberg, A.J.; Lucchinetti, C.F.; Tenembaum, S.; Belman, A.L. Acute disseminated encephalomyelitis: Updates on an inflammatory CNS syndrome. Neurology 2016, 87, S38–S45. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, T.; Fujihara, K.; Nakashima, I.; Misu, T.; Miyazawa, I.; Nakamura, M.; Watanabe, S.; Shiga, Y.; Kanaoka, C.; Fujimori, J.; et al. Anti-Aquaporin-4 Antibody Is Involved in the Pathogenesis of NMO: A Study on Antibody Titre. Brain 2007, 130, 1235–1243. [Google Scholar] [CrossRef] [PubMed]
- Schmetzer, O.; Lakin, E.; Roediger, B.; Duchow, A.; Asseyer, S.; Paul, F.; Siebert, N. Anti-Aquaporin 4 IgG Is Not Associated with Any Clinical Disease Characteristics in Neuromyelitis Optica Spectrum Disorder. Front. Neurol. 2021, 12, 635419. [Google Scholar] [CrossRef] [PubMed]
- Mehrpour, M.; Kyani, A.; Tafazzoli, M.; Fathi, F.; Joghataie, M.T. A metabonomics investigation of multiple sclerosis by nuclear magnetic resonance. Magn. Reson. Chem. 2013, 51, 102–109. [Google Scholar] [CrossRef]
- Villoslada, P.; Alonso, C.; Agirrezabal, I.; Kotelnikova, E.; Zubizarreta, I.; Pulido-Valdeolivas, I.; Saiz, A.; Comabella, M.; Montalban, X.; Villar, L.; et al. Metabolomic signatures associated with disease severity in multiple sclerosis. Neurol. Neuroimmunol. Neuroinflamm. 2017, 4, e321. [Google Scholar] [CrossRef] [PubMed]
- Del Boccio, P.; Rossi, C.; di Ioia, M.; Cicalini, I.; Sacchetta, P.; Pieragostino, D. Integration of metabolomics and proteomics in multiple sclerosis: From biomarkers discovery to personalized medicine. Proteom. Clin. Appl. 2016, 10, 470–484. [Google Scholar] [CrossRef] [PubMed]
- Pieragostino, D.; D’Alessandro, M.; di Ioia, M.; Rossi, C.; Zucchelli, M.; Urbani, A.; Di Ilio, C.; Lugaresi, A.; Sacchetta, P.; Del Boccio, P. An integrated metabolomics approach for the research of new cerebrospinal fluid biomarkers of multiple sclerosis. Mol. Biosyst. 2015, 11, 1563–1572. [Google Scholar] [CrossRef]
- Jurynczyk, M.; Probert, F.; Yeo, T.; Tackley, G.; Claridge, T.D.W.; Cavey, A.; Woodhall, M.R.; Arora, S.; Winkler, T.; Schiffer, E.; et al. Metabolomics reveals distinct, antibody-independent, molecular signatures of MS, AQP4-antibody and MOG-antibody disease. Acta. Neuropathol. Commun. 2017, 5, 95. [Google Scholar] [CrossRef]
- Moussallieh, F.M.; Elbayed, K.; Chanson, J.B.; Rudolf, G.; Piotto, M.; De Seze, J.; Namer, I.J. Serum analysis by 1H nuclear magnetic resonance spectroscopy: A new tool for distinguishing neuromyelitis optica from multiple sclerosis. Mult. Scler. 2014, 20, 558–565. [Google Scholar] [CrossRef]
- Gebregiworgis, T.; Nielsen, H.H.; Massilamany, C.; Gangaplara, A.; Reddy, J.; Illes, Z.; Powers, R. A Urinary Metabolic Signature for Multiple Sclerosis and Neuromyelitis Optica. J. Proteome Res. 2016, 15, 659–666. [Google Scholar] [CrossRef]
- Park, S.J.; Jeong, I.H.; Kong, B.S.; Lee, J.E.; Kim, K.H.; Lee, D.Y.; Kim, H.J. Disease Type- and Status-Specific Alteration of CSF Metabolome Coordinated with Clinical Parameters in Inflammatory Demyelinating Diseases of CNS. PLoS ONE 2016, 11, e0166277. [Google Scholar] [CrossRef]
- Kowarik, M.C.; Dzieciatkowska, M.; Wemlinger, S.; Ritchie, A.M.; Hemmer, B.; Owens, G.P.; Bennett, J.L. The cerebrospinal fluid immunoglobulin transcriptome and proteome in neuromyelitis optica reveals central nervous system-specific B cell populations. J. Neuroinflamm. 2015, 12, 19. [Google Scholar] [CrossRef]
- Lucchinetti, C.F.; Guo, Y.; Popescu, B.F.G.; Fujihara, K.; Itoyama, Y.; Misu, T. The Pathology of an Autoimmune Astrocytopathy: Lessons Learned from Neuromyelitis Optica. Brain. Pathol. 2014, 24, 83–97. [Google Scholar] [CrossRef] [PubMed]
- Pittock, S.J.; Lucchinetti, C.F. Neuromyelitis optica and the evolving spectrum of autoimmune aquaporin-4 channelopathies: A decade later. Ann. N. Y. Acad. Sci. 2016, 1366, 20–39. [Google Scholar] [CrossRef]
- Popescu, B.; Lennon, V.A.; Parisi, J.E.; Howe, C.L.; Weigand, S.D.; Cabrera-Gomez, J.A.; Newell, K.; Mandler, R.N.; Pittock, S.J.; Weinshenker, B.G.; et al. Neuromyelitis optica unique area postrema lesions: Nausea, vomiting, and pathogenic implications. Neurology 2011, 76, 1229–1237. [Google Scholar] [CrossRef] [PubMed]
- Katoozi, S.; Rao, S.B.; Skauli, N.; Froehner, S.C.; Ottersen, O.P.; Adams, M.E.; Amiry-Moghaddam, M. Functional specialization of retinal Müller cell endfeet depends on an interplay between two syntrophin isoforms. Mol. Brain 2020, 13, 40. [Google Scholar] [CrossRef] [PubMed]
- Takano, R.; Misu, T.; Takahashi, T.; Sato, S.; Fujihara, K.; Itoyama, Y. Astrocytic damage is far more severe than demyelination in NMO: A clinical CSF biomarker study. Neurology 2010, 75, 208–216. [Google Scholar] [CrossRef]
- Storoni, M.; Verbeek, M.M.; Illes, Z.; Marignier, R.; Teunissen, C.E.; Grabowska, M.; Confavreux, C.; Plant, G.T.; Petzold, A. Serum GFAP levels in optic neuropathies. J. Neurol. Sci. 2012, 317, 117–122. [Google Scholar] [CrossRef]
- Fang, B.; McKeon, A.; Hinson, S.R.; Kryzer, T.J.; Pittock, S.J.; Aksamit, A.J.; Lennon, V.A. Autoimmune glial fibrillary acidic protein astrocytopathy: A novel meningo encephalomyelitis. JAMA Neurol. 2016, 73, 1297–1307. [Google Scholar] [CrossRef]
- Jarius, S.; Aboul-Enein, F.; Waters, P.; Kuenz, B.; Hauser, A.; Berger, T.; Lang, W.; Reindl, M.; Vincent, A.; Kristoferitsch, W. Antibody to aquaporin-4 in the long-term course of neuromyelitis optica. Brain 2008, 131, 3072–3080. [Google Scholar] [CrossRef]
- Ho, J.D.; Yeh, R.; Sandstrom, A.; Chorny, I.; Harries, W.E.; Robbins, R.A.; Miercke, L.J.; Stroud, R.M. Crystal structure of human aquaporin 4 at 1.8 a and its mechanism of conductance. Proc. Natl. Acad. Sci. USA 2009, 106, 7437–7442. [Google Scholar] [CrossRef] [PubMed]
- Xu, M.; Xiao, M.; Li, S.; Yang, B. Aquaporins in Nervous System. Adv. Exp. Med. Biol. 2017, 969, 81–103. [Google Scholar] [PubMed]
- Jung, J.S.; Bhat, R.V.; Preston, G.M.; Guggino, W.B.; Baraban, J.M.; Agre, P. Molecular characterization of an aquaporin cDNA from brain: Candidate osmoreceptor and regulator of water balance. Proc. Natl. Acad. Sci. USA 1994, 91, 13052–13056. [Google Scholar] [CrossRef]
- Zhang, S.; Shang, D.; Shi, H.; Teng, W.; Tian, L. Function of Astrocytes in Neuroprotection and Repair after Ischemic Stroke. Eur. Neurol. 2021, 84, 426–434. [Google Scholar] [CrossRef] [PubMed]
- Szu, J.I.; Binder, D.K. The Role of Astrocytic Aquaporin-4 in Synaptic Plasticity and Learning and Memory. Front. Integr. Neurosci. 2016, 24, 10:18. [Google Scholar] [CrossRef]
- Li, L.; Zhang, H.; Verkman, A.S. Greatly attenuated experimental autoimmune encephalomyelitis in aquaporin-4 knockout mice. BMC Neurosci. 2009, 10, 94. [Google Scholar] [CrossRef]
- Netti, V.; Fernández, J.; Melamud, L.; Garcia-Miranda, P.; Di Giusto, G.; Ford, P.; Echevarría, M.; Capurro, C. Aquaporin-4 Removal from the Plasma Membrane of Human Müller Cells by AQP4-IgG from patients with neuromyelitis optica induces changes in cell volume homeostasis: The first step of retinal injury? Mol. Neurobiol. 2021, 58, 5178–5193. [Google Scholar] [CrossRef]
- Jarius, S.; Wildemann, B.; Paul, F. Neuromyelitis optica: Clinical features, immunopathogenesis and treatment. Clin. Exp. Immunol. 2014, 176, 149–164. [Google Scholar] [CrossRef]
- Dalakas, M.C. B cells as therapeutic targets in autoimmune neurological disorders. Nat. Clin. Pract. 2008, 4, 557–567. [Google Scholar] [CrossRef]
- Chihara, N.; Aranami, T.; Sato, W.; Miyazaki, Y.; Miyake, S.; Okamoto, T.; Ogawa, M.; Toda, T.; Yamamura, T. Interleukin 6 signaling promotes anti-aquaporin 4 autoantibody production from plasmoblasts in neuromyelitis optica. Proc. Nat. Acad. Sci. USA 2011, 108, 3701–3706. [Google Scholar] [CrossRef]
- Wilson, R.; Makuch, M.; Kienzler, A.K.; Varley, J.; Taylor, J.; Woodhall, M.; Palace, J.; Leite, M.I.; Waters, P.; Irani, S.R. Condition-dependent generation of aquaporin-4 antibodies from circulating B cells in neuromyelitis optica. Brain 2018, 141, 1063–1074. [Google Scholar] [CrossRef] [PubMed]
- LeBien, T.W.; Tedder, T.F. B lymphocytes: How they develop and function. Blood 2008, 112, 1570. [Google Scholar] [CrossRef] [PubMed]
- Hinson, S.R.; Roemer, S.F.; Lucchinetti, C.F.; Fryer, J.P.; Kryzer, T.J.; Chamberlain, J.L.; Howe, C.L.; Pittock, S.J.; Lennon, V.A. Aquaporin-4-binding autoantibodies in patients with neuromyelitis optica impair glutamate transport by down-regulating EAAT2. J. Exp. Med. 2008, 205, 2473–2481. [Google Scholar] [CrossRef]
- Marignier, R.; Nicolle, A.; Watrin, C.; Touret, M.; Cavagna, S.; Varrin-Doyer, M.; Cavillon, G.; Rogemond, V.; Confavreux, C.; Honnorat, J.; et al. Oligodendrocytes are damaged by neuromyelitis optica immunoglobulin G via astrocyte injury. Brain 2010, 133, 2578–2591. [Google Scholar] [CrossRef] [PubMed]
- Howe, C.L.; Kaptzan, T.; Magaña, S.M.; Ayers-Ringler, J.R.; LaFrance-Corey, R.G.; Lucchinetti, C.F. Neuromyelitis optica IgG stimulates an immunological response in rat astrocyte cultures. Glia 2014, 62, 692–708. [Google Scholar] [CrossRef]
- Zhang, H.; Verkman, A.S. Eosinophil pathogenicity mechanisms and therapeutics in neuromyelitis optica. J. Clin. Investig. 2013, 123, 2306–2316. [Google Scholar] [CrossRef]
- Tradtrantip, L.; Yao, X.; Su, T.; Smith, A.J.; Verkman, A.S. Bystander mechanism for complement-initiated early oligodendrocyte injury in neuromyelitis optica. Acta. Neuropathol. 2017, 134, 35–44. [Google Scholar] [CrossRef]
- Duan, T.; Smith, A.J.; Verkman, A.S. Complement-dependent bystander injury to neurons in AQP4-IgG seropositive neuromyelitis optica. J. Neuroinflamm. 2018, 15, 294. [Google Scholar] [CrossRef]
- John, T.G.; Bernard, C.A.C. The structure and function of myelin oligodendrocyte glycoprotein. J. Neurochem. 2002, 72, 1–9. [Google Scholar] [CrossRef]
- De Lott, L.B.; Bennett, J.L.; Costello, F. The Changing Landscape of Optic Neuritis: A Narrative Review. J. Neurol. 2022, 269, 111–124. [Google Scholar] [CrossRef]
- Petrikowski, L.; Reinehr, S.; Haupeltshofer, S.; Deppe, L.; Graz, F.; Kleiter, I.; Dick, H.B.; Gold, R.; Faissner, S.; Joachim, S.C. Progressive Retinal and Optic Nerve Damage in a Mouse Model of Spontaneous Opticospinal Encephalomyelitis. Front. Immunol. 2022, 12, 759389. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, S.; Hashimoto, B.; Izaki, S.; Oji, S.; Fukaura, H.; Nomura, K. Clinical and Immunological Differences between Mog Associated Disease and Anti Aqp4 Antibody-Positive Neuromyelitis Optica Spectrum Disorders: Blood-Brain Barrier Breakdown and Peripheral Plasmablasts. Mult. Scler. Rela. Disord. 2020, 41, 102005. [Google Scholar] [CrossRef] [PubMed]
- Ataie-Kachoie, P.; Pourgholami, M.H.; Morris, D.L. Inhibition of the IL-6 signaling pathway: A strategy to combat chronic inflammatory diseases and cancer. Cytokine Growth Factor Rev. 2013, 24, 163–173. [Google Scholar] [CrossRef] [PubMed]
- Van Wagoner, N.J.; Benveniste, E.N. Interleukin-6 expression and regulation in astrocytes. J. Neuroimmunol. 1999, 100, 124–139. [Google Scholar] [CrossRef]
- Fujihara, K.; Bennett, J.L.; de Seze, J.; Haramura, M.; Kleiter, I.; Weinshenker, B.G.; Kang, D.; Mughal, T.; Yamamura, T. Interleukin-6 in neuromyelitis optica spectrum disorder pathophysiology. Neurol. Neuroimmunol. Neuroinflamm. 2020, 7, e841. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Zhang, J.; Chang, H.; Wang, H.; Xu, W.; Cong, H.; Zhang, X.; Liu, J.; Yin, L. NMO-IgG Induce Interleukin-6 Release via Activation of the NF-κB Signaling Pathway in Astrocytes. Neuroscience 2022, 496, 96–104. [Google Scholar] [CrossRef]
- Wang, H.; Wang, K.; Zhong, X.; Dai, Y.; Qiu, W.; Wu, A.; Hu, X. Notable increased cerebrospinal fluid levels of soluble interleukin-6 receptors in neuromyelitis optica. Neuroimmunomodulation 2012, 19, 304–308. [Google Scholar] [CrossRef] [PubMed]
- Du, L.; Chang, H.; Xu, W.; Wei, Y.; Wang, Y.; Yin, L.; Zhang, X. Effect of NMO-IgG on the interleukin-6 cascade in astrocytes via activation of the JAK/STAT3 signaling pathway. Life Sci. 2020, 258, 118217. [Google Scholar] [CrossRef]
- Garbers, C.; Heink, S.; Korn, T.; Rose-John, S. Interleukin-6: Designing specific therapeutics for a complex cytokine. Nat. Rev. Drug Discov. 2018, 17, 395–412. [Google Scholar] [CrossRef]
- Uzawa, A.; Mori, M.; Ito, M.; Uchida, T.; Hayakawa, S.; Masuda, S.; Kuwabara, S. Markedly increased CSF interleukin-6 levels in neuromyelitis optica, but not in multiple sclerosis. J. Neurol. 2009, 256, 2082–2084. [Google Scholar] [CrossRef] [PubMed]
- Uzawa, A.; Mori, M.; Arai, K.; Sato, Y.; Hayakawa, S.; Masuda, S.; Taniguchi, J.; Kuwabara, S. Cytokine and Chemokine Profiles in Neuromyelitis Optica: Significance of Interleukin-6. Mult. Scler. 2010, 16, 1443–1452. [Google Scholar] [CrossRef] [PubMed]
- Zipfel, P.F.; Skerka, C. Complement regulators and inhibitory proteins. Nat. Rev. Immunol. 2009, 9, 729–740. [Google Scholar] [CrossRef] [PubMed]
- Pache, F.; Ringelstein, M.; Aktas, O.; Kleiter, I.; Jarius, S.; Siebert, N.; Bellmann-Strobl, J.; Paul, F.; Ruprecht, K. C3 and C4 complement levels in AQP4-IgG-positive NMOSD and in MOGAD. J. Neuroimmunol. 2021, 360, 577699. [Google Scholar] [CrossRef] [PubMed]
- Maciak, K.; Pietrasik, S.; Dziedzic, A.; Redlicka, J.; Saluk-Bijak, J.; Bijak, M.; Włodarczyk, T.; Miller, E. Th17-Related Cytokines as Potential Discriminatory Markers between Neuromyelitis Optica (Devic’s Disease) and Multiple Sclerosis—A Review. Int. J. Mol. Sci. 2021, 22, 8946. [Google Scholar] [CrossRef] [PubMed]
- Uzawa, A.; Mori, M.; Kuwabara, S. Cytokines and chemokines in neuromyelitis optica: Pathogenetic and therapeutic implications. Brain Pathol. 2014, 24, 67–73. [Google Scholar] [CrossRef] [PubMed]
- Hou, M.M.; Li, Y.F.; He, L.L.; Li, X.Q.; Zhang, Y.; Zhang, S.X.; Li, X.Y. Proportions of Th17 Cells and Th17-Related Cytokines in Neuromyelitis Optica Spectrum Disorders Patients: A Meta-Analysis. Int. Immunopharmacol. 2019, 75, 105793. [Google Scholar] [CrossRef]
- Nytrova, P.; Potlukova, E.; Kemlink, D.; Woodhall, M.; Horakova, D.; Waters, P.; Havrdova, E.; Zivorova, D.; Vincent, A.; Trendelenburg, M. Complement activation in patients with neuromyelitis optica. J. Neuroimmunol. 2014, 274, 185–191. [Google Scholar] [CrossRef]
- Asavapanumas, N.; Tradtrantip, L.; Verkman, A.S. Targeting the complement system in neuromyelitis optica spectrum disorder. Expert. Opin. Biol. Ther. 2021, 21, 1073–1086. [Google Scholar] [CrossRef]
- Soltys, J.; Liu, Y.; Ritchie, A.; Wemlinger, S.; Schaller, K.; Schumann, H.; Owens, G.P.; Bennett, J.L. Membrane assembly of aquaporin-4 autoantibodies regulates classical complement activation in neuromyelitis optica. J. Clin. Investig. 2019, 129, 2000–2013. [Google Scholar] [CrossRef]
- Yao, X.; Verkman, A.S. Complement regulator CD59 prevents peripheral organ injury in rats made seropositive for neuromyelitis optica immunoglobulin G. Acta. Neuropathol. Commun. 2017, 5, 57. [Google Scholar] [CrossRef] [PubMed]
- Kalluri, S.R.; Srivastava, R.; Kenet, S.; Tanti, G.K.; Dornmair, K.; Bennett, J.L.; Misgeld, T.; Hemmer, B.; Wyss, M.T.; Herwerth, M. P2R Inhibitors Prevent Antibody-Mediated Complement Activation in an Animal Model of Neuromyelitis Optica: P2R Inhibitors Prevent Autoantibody Injury. Neurotherapeutics 2022, 19, 1603–1616. [Google Scholar] [CrossRef]
- Pittock, S.J.; Lennon, V.A.; McKeon, A.; Mandrekar, J.; Weinshenker, B.G.; Lucchinetti, C.F.; O’Toole, O.; Wingerchuk, D.M. Eculizumab in AQP4-IgG-positive relapsing neuromyelitis optica spectrum disorders: An open-label pilot study. Lancet Neurol. 2013, 12, 554–562. [Google Scholar] [CrossRef]
- Bennett, J.L.; Lam, C.; Kalluri, S.R.; Saikali, P.; Bautista, K.; Dupree, C.; Glogowska, M.; Case, D.; Antel, J.P.; Owens, G.P.; et al. Intrathecal pathogenic antiaquaporin-4 antibodies in early neuromyelitis optica. Ann. Neurol. 2009, 66, 617–629. [Google Scholar] [CrossRef] [PubMed]
- Klos, A.; Tenner, A.J.; Johswich, K.O.; Ager, R.R.; Reis, E.S.; Kohl, J. The role of the anaphylatoxins in health and disease. Mol. Immunol. 2009, 46, 2753–2766. [Google Scholar] [CrossRef] [PubMed]
- Thangaleela, S.; Sivamaruthi, B.S.; Kesika, P.; Bharathi, M.; Chaiyasut, C. Role of the Gut-Brain Axis, Gut Microbial Composition, Diet, and Probiotic Intervention in Parkinson’s Disease. Microorganisms 2022, 10, 1544. [Google Scholar] [CrossRef] [PubMed]
- Ivanov, I.I.; Atarashi, K.; Manel, N.; Brodie, E.L.; Shima, T.; Karaoz, U.; Wei, D.; Goldfarb, K.C.; Santee, C.A.; Lynch, S.V.; et al. Induction of intestinal Th17 cells by segmented filamentous bacteria. Cell 2009, 139, 485–498. [Google Scholar] [CrossRef]
- Varrin-Doyer, M.; Spencer, C.M.; Schulze-Topphoff, U.; Nelson, P.A.; Stroud, R.M.; Cree, B.A.; Zamvil, S.S. Aquaporin 4-specific T cells in neuromyelitis optica exhibit a Th17 bias and recognize Clostridium ABC transporter. Ann. Neurol. 2012, 72, 53–64. [Google Scholar] [CrossRef]
- Zamvil, S.S.; Spencer, C.M.; Baranzini, S.E.; Cree, B.A.C. The Gut Microbiome in Neuromyelitis Optica. Neurotherapeutics 2018, 15, 92–101. [Google Scholar] [CrossRef] [PubMed]
- Cree, B.A.; Spencer, C.M.; Varrin-Doyer, M.; Baranzini, S.E.; Zamvil, S.S. Gut microbiome analysis in neuromyelitis optica reveals overabundance of Clostridium perfringens. Ann. Neurol. 2016, 80, 443–447. [Google Scholar] [CrossRef]
- Zhang, J.; Xu, Y.F.; Wu, L.; Li, H.F.; Wu, Z.Y. Characteristic of gut microbiota in southeastern Chinese patients with neuromyelitis optica spectrum disorders. Mult. Scler. Relat. Disor. 2020, 44, 102217. [Google Scholar] [CrossRef]
- Jarius, S.; Wandinger, K.P.; Platzer, S.; Wildemann, B. Homology between Klebsiella pneumoniae and human aquaporin-4: No evidence for cross-reactivity in neuromyelitis optica. A study on 114 patients. J. Neurol. 2011, 258, 929–931. [Google Scholar] [CrossRef]
- Gong, J.; Qiu, W.; Zeng, Q.; Liu, X.; Sun, X.; Li, H.; Yang, Y.; Wu, A.; Bao, J.; Wang, Y.; et al. Lack of short-chain fatty acids and overgrowth of opportunistic pathogens define dysbiosis of neuromyelitis optica spectrum disorders: A Chinese pilot study. Mult. Scler. 2019, 25, 1316–1325. [Google Scholar] [CrossRef] [PubMed]
- Katz, S.I.; Zhu, Y.; Ntranos, A.; Clemente, J.C.; Cekanaviciute, E.; Brandstadter, R.; Crabtree-Hartman, E.; Singh, S.; Bencosme, Y.; Debelius, J.; et al. Disease-modifying therapies alter gut microbial composition in MS. Neurol. Neuroimmunol. Neuroinflamm. 2018, 6, e517. [Google Scholar] [CrossRef] [PubMed]
- Okumura, R.; Takeda, K. Maintenance of intestinal homeostasis by mucosal barriers. Inflamm. Regen. 2018, 38, 5. [Google Scholar] [CrossRef] [PubMed]
- Nagpal, R.; Yadav, H. Bacterial Translocation from the Gut to the Distant Organs: An Overview. Ann. Nutr. Metab. 2017, 71, 11–16. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Zhu, M.; Liu, C.; Han, J.; Lang, W.; Gao, Y.; Lu, C.; Wang, S.; Hou, S.; Zheng, N. Blood brain barrier permeability could be a biomarker to predict severity of neuromyelitis optica spectrum disorders: A retrospective analysis. Front. Neurol. 2018, 9, 648. [Google Scholar] [CrossRef] [PubMed]
- Schmitz, H.; Barmeyer, C.; Fromm, M.; Runkel, N.; Foss, H.D.; Bentzel, C.J.; Riecken, E.O.; Schulzke, J.D. Altered tight junction structure contributes to the impaired epithelial barrier function in ulcerative colitis. Gastroenterology 1999, 116, 301–309. [Google Scholar] [CrossRef]
- Sterlin, D.; Larsen, M.; Fadlallah, J.; Parizot, C.; Vignes, M.; Autaa, G.; Dorgham, K.; Juste, C.; Lepage, P.; Aboab, J.; et al. Perturbed microbiota/immune homeostasis in multiple sclerosis. Neurol. Neuroimmunol. Neuroinflamm. 2021, 8, e997. [Google Scholar] [CrossRef]
- Asavapanumas, N.; Verkman, A.S. Neuromyelitis optica pathology in rats following intraperitoneal injection of NMO-IgG and intracerebral needle injury. Acta. Neuropathol. Commun. 2014, 2, 48. [Google Scholar] [CrossRef]
- Kinoshita, M.; Nakatsuji, Y.; Kimura, T.; Moriya, M.; Takata, K.; Okuno, T.; Kumanogoh, A.; Kajiyama, K.; Yoshikawa, H.; Sakoda, S. Neuromyelitis optica: Passive transfer to rats by human immunoglobulin. Biochem. Biophys. Res. Commun. 2009, 386, 623–627. [Google Scholar] [CrossRef]
- Saini, H.; Rifkin, R.; Gorelik, M.; Huang, H.; Ferguson, Z.; Jones, M.V.; Levy, M. Passively transferred human NMO-IgG exacerbates demyelination in mouse experimental autoimmune encephalomyelitis. BMC Neurol. 2013, 13, 104. [Google Scholar] [CrossRef] [PubMed]
- Hillebrand, S.; Schanda, K.; Nigritinou, M.; Tsymala, I.; Böhm, D.; Peschl, P.; Takai, Y.; Fukihara, K.; Nakashima, I.; Misu, T. Circulated AQP4-specific auto-antibodies alone can induce neuromyelitis optica spectrum disorder in the rat. Acta Neuropathol. 2019, 137, 467–485. [Google Scholar] [CrossRef] [PubMed]
- Saadoun, S.; Waters, P.; Bell, B.A.; Vincent, A.; Verkman, A.S.; Papadopoulos, M.C. Intra-cerebral injection of neuromyelitis optica immunoglobulin G and human complement produces neuromyelitis optica lesions in mice. Brain 2010, 133, 349–361. [Google Scholar] [CrossRef] [PubMed]
- Luo, J.; Xie, C.; Zhang, W.; Cai, Y.; Ding, J.; Wang, Y.; Hao, Y.; Zhang, Y.; Guan, Y. Experimental mouse model of NMOSD produced by facilitated brain delivery of NMO-IgG by microbubble-enhanced low-frequency ultrasound in experimental allergic encephalomyelitis mice. Mult. Scler. Relat. Disor. 2020, 46, 102473. [Google Scholar] [CrossRef]
- Duan, T.; Verkman, A.S. Experimental animal models of aquaporin-4-IgG-seropositive neuromyelitis optica spectrum disorders: Progress and shortcomings. Brain Pathol. 2020, 30, 13–25. [Google Scholar] [CrossRef] [PubMed]
- Crane, J.M.; Lam, C.; Rossi, A.; Gupta, T.; Bennett, J.L.; Verkman, A.S. Binding affinity and specificity of neuromyelitis optica autoantibodies to aquaporin-4 M1/M23 isoforms and orthogonal arrays. J. Biol. Chem. 2011, 286, 16516–16524. [Google Scholar] [CrossRef]
- Geis, C.; Ritter, C.; Ruschil, C.; Weishaupt, A.; Grunewald, B.; Stoll, G.; Holmoy, T.; Misu, T.; Fujihara, K.; Hemmer, B.; et al. The intrinsic pathogenic role of autoantibodies to aquaporin 4 mediating spinal cord disease in a rat passive-transfer model. Exp. Neurol. 2015, 265, 8–21. [Google Scholar] [CrossRef]
- Marignier, R.; Ruiz, A.; Cavagna, S.; Nicole, A.; Watrin, C.; Touret, M.; Parrot, S.; Malleret, G.; Peyron, C.; Benetollo, C. Neuromyelitis optica study model based on chronic infusion of autoantibodies in rat cerebrospinal fluid. J. Neuroinflamm. 2016, 13, 111. [Google Scholar] [CrossRef]
- Felix, C.M.; Levin, M.H.; Verkman, A.S. Complement-independent retinal pathology produced by intravitreal injection of neuromyelitis optica immunoglobulin G. J. Neuroinflamm. 2016, 13, 275. [Google Scholar] [CrossRef]
- Zhang, H.; Bennett, J.L.; Verkman, A.S. Ex vivo spinal cord slice model of neuromyelitis optica reveals novel immunopathogenic mechanisms. Ann. Neurol. 2011, 70, 943–954. [Google Scholar] [CrossRef]
- Nakamura, M.; Nakazawa, T.; Doi, H.; Hariya, T.; Omodaka, K.; Misu, T.; Takahashi, T.; Fujihara, K.; Nishida, K. Early high-dose intravenous methylprednisolone is effective in preserving retinal nerve fiber layer thickness in patients with neuromyelitis optica. Graefe’s Arch. Clin. Exp. Ophthalmol. 2010, 248, 1777–1785. [Google Scholar]
- Stiebel-Kalish, H.; Hellmann, M.A.; Mimouni, M.; Paul, F.; Bialer, O.; Bach, M.; Lotan, I. Does time equal vision in the acute treatment of a cohort of AQP4 and MOG optic neuritis? Neurol. Neuroimmunol. Neuroinflam. 2019, 6, e572. [Google Scholar] [CrossRef] [PubMed]
- Coutinho, A.E.; Chapman, K.E. The anti-inflammatory and immunosuppressive effects of glucocorticoids, recent developments and mechanistic insights. Mol. Cell. Endocrinol. 2011, 335, 2–13. [Google Scholar] [CrossRef] [PubMed]
- Kimbrough, D.; Fujihara, K.; Jacob, A.; Lana-Peixoto, M.A.; Leite, M.I.; Levy, M.; Marignier, R.; Nakashima, I.; Palace, J.; de Seze, J.; et al. Treatment of neuromyelitis optica: Review and recommendations. Mult. Scler. Relat. Disord. 2012, 1, 180–187. [Google Scholar] [CrossRef] [PubMed]
- Klingele, M.; Allmendinger, C.; Thieme, S.; Baerens, L.; Fliser, D.; Jan, B. Therapeutic apheresis within immune-mediated neurological disorders: Dosing and its effectiveness. Sci. Rep. 2020, 10, 7925. [Google Scholar] [CrossRef]
- Reeves, H.M.; Winters, J.L. The mechanisms of action of plasma exchange. Br. J. Haematol. 2014, 164, 342–351. [Google Scholar] [CrossRef]
- Kessler, R.A.; Mealy, M.A.; Levy, M. Treatment of neuromyelitis optica spectrum disorder: Acute, preventative, and symptomatic. Curr. Treat. Options Neurol. 2016, 18, 2. [Google Scholar] [CrossRef]
- Bonnan, M.; Valentino, R.; Olindo, S.; Mehdaoui, H.; Smadja, D.; Cabre, P. Plasma exchange in severe spinal attacks associated with neuromyelitis optica spectrum disorder. Mult. Scler. 2009, 15, 487–492. [Google Scholar] [CrossRef]
- Merle, H.; Olindo, S.; Jeannin, S.; Valentino, R.; Mehdaoui, H.; Cabot, F.; Donnio, A.; Hage, R.; Richer, R.; Smadja, D.; et al. Treatment of Optic Neuritis by Plasma Exchange (Add-On) in Neuromyelitis Optica. Arch. Ophthalmol. 2012, 130, 858–862. [Google Scholar] [CrossRef]
- Braun, N.; Risler, T. Immunoadsorption as a Tool for the Immunomodulation of the Humoral and Cellular Immune System in Autoimmune Disease. Ther. Apher. 1999, 3, 240–245. [Google Scholar] [CrossRef]
- Kleiter, I.; Gahlen, A.; Borisow, N.; Fischer, K.; Wernecke, K.D.; Hellwig, K.; Pache, F.; Ruprecht, K.; Havla, J.; Kümpfel, T. Apheresis therapies for NMOSD attacks: A retrospective study of 207 therapeutic interventions. Neurol. Neuroimmunol. Neuroinflamm. 2018, 5, e504. [Google Scholar] [CrossRef] [PubMed]
- Kleiter, I.; Gahlen, A.; Borisow, N.; Fischer, K.; Wernecke, K.-D.; Wegner, B.; Hellwig, K.; Pache, F.; Ruprecht, K.; Havla, J.; et al. Neuromyelitis optica: Evaluation of 871 attacks and 1153 treatment courses. Ann. Neurol. 2016, 79, 206–216. [Google Scholar] [CrossRef]
- Kumawat, B.L.; Choudhary, R.; Sharma, C.M.; Jain, D.; Hiremath, A. Plasma Exchange as a First Line Therapy in Acute Attacks of Neuromyelitis Optica Spectrum Disorders. Ann. Indian Acad. Neurol. 2019, 22, 389–394. [Google Scholar] [PubMed]
- Bhatia, R.; Pedapati, R.; Chopra, S. Plasmapheresis for NMOSD: Not a Rescue Therapy Anymore!? Ann. Indian Acad. Neurol. 2019, 22, 371–372. [Google Scholar]
- Lunemann, J.; Nimmerjahn, F.; Dalakas, M.C. Intravenous immunoglobulin in neurology—Mode of action and clinical efficacy. Nat. Rev. Neurol. 2015, 11, 80–89. [Google Scholar] [CrossRef]
- Yu, Z.; Lennon, V.A. Mechanism of intravenous immune globulin therapy in antibody-mediated autoimmune diseases. N. Engl. J. Med. 1999, 340, 227–228. [Google Scholar] [CrossRef] [PubMed]
- Okada, K.; Matsushita, T.; Kira, J.; Tsuji, S. B-cell activating factor of the TNF family is upregulated in neuromyelitis optica. Neurology 2012, 74, 177–178. [Google Scholar] [CrossRef]
- Altunrende, B.; Akdal, G.; Bajin, M.S.; Yaman, A.; Kocaslan, M.; Nalbantoğlu, M.; Ertaşoğlu, H.; Akman, G. Intravenous Immunoglobulin Treatment for Recurrent Optic Neuritis. Noro. Psikiyatr. Ars. 2019, 56, 3–6. [Google Scholar]
- Shi, M.; Chu, F.; Jin, T.; Zhu, J. Progress in treatment of neuromyelitis optica spectrum disorders (NMOSD): Novel insights into therapeutic possibilities in NMOSD. CNS Neurosci. Ther. 2022, 28, 981–991. [Google Scholar] [CrossRef]
- Graf, J.; Mares, J.; Barnett, M.; Aktas, O.; Albrecht, P.; Zamvil, S.S.; Hartung, H.P. Targeting B cells to modify MS, NMOSD, and MOGAD: Part 1. Neurol. Neuroimmunol. Neuroinflamm. 2021, 8, e919. [Google Scholar] [CrossRef]
- Zhang, C.; Zhang, M.; Qiu, W.; Ma, H.; Zhang, X.; Zhu, Z.; Yang, C.S.; Jia, D.; Zhang, T.X.; Yuan, M.; et al. TANGO Study Investigators. Safety and efficacy of tocilizumab versus azathioprine in highly relapsing neuromyelitis optica spectrum disorder (TANGO): An open-label, multicentre, randomised, phase 2 trial. Lancet Neurol. 2020, 19, 391–401. [Google Scholar] [CrossRef] [PubMed]
- Houssiau, F.A.; D’Cruz, D.; Sangle, S.; Remy, P.; Vasconcelos, C.; Petrovic, R.; Fiehn, C.; de Ramon Garrido, E.; Gilboe, I.M.; Tektonidou, M.; et al. Azathioprine versus mycophenolate mofetil for long-term immunosuppression in lupus nephritis: Results from the maintain nephritis trial. Ann. Rheum. Dis. 2010, 69, 2083–2089. [Google Scholar] [CrossRef]
- Poupart, J.; Giovannelli, J.; Deschamps, R.; Audoin, B.; Ciron, J.; Maillart, E.; Papeix, C.; Collongues, N.; Bourre, B.; Cohen, M.; et al. Evaluation of efficacy and tolerability of first-line therapies in NMOSD. Neurology 2020, 94, e1645–e1656. [Google Scholar] [CrossRef]
- Weinstock-Guttman, B.; Ramanathan, M.; Lincoff, N.; Napoli, S.Q.; Sharma, J.; Feichter, J.; Bakshi, R. Study of mitoxantrone for the treatment of recurrent neuromyelitis optica (Devic’s disease). Arch. Neurol. 2006, 63, 957–963. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.H.; Kim, W.; Park, M.S.; Sohn, E.H.; Li, X.F.; Kim, H.J. Efficacy and safety of mitoxantrone in patients with highly relapsing neuromyelitis optica. Arch. Neurol. 2011, 68, 473–479. [Google Scholar] [CrossRef] [PubMed]
- Cabre, P.; Olindo, S.; Marignier, R.; Jeannin, S.; Merle, H.; Smadja, D. Efficacy of mitoxantrone in neuromyelitis optica spectrum: Clinical and neuroradiological study. J. Neurol. Neurosurg. Psychiatry 2013, 84, 511–516. [Google Scholar] [CrossRef]
- Kitley, J.; Elsone, L.; George, J.; Waters, P.; Woodhall, M.; Vincent, A.; Jacob, A.; Leite, M.I.; Palace, J. Methotrexate is an alternative to azathioprine with aquaporin-4 antibodies. J. Neurol. Neurosurg. Psychiatry 2013, 84, 918–921. [Google Scholar] [CrossRef]
- Ramanathan, R.S.; Malhotra, K.; Scott, T. Treatment of neuromyelitis optica/neuromyelitis optica spectrum disorders with methotrexate. BMC Neurol. 2014, 14, 51. [Google Scholar] [CrossRef] [PubMed]
- Yamamura, T.; Kleiter, I.; Fujihara, K.; Palace, J.; Greenberg, B.; Zakrzewska-Pniewska, B.; Patti, F.; Tsai, C.P.; Saiz, A.; Yamazaki, H.; et al. Trial of satralizumab in neuromyelitis optica spectrum disorder. N. Engl. J. Med. 2019, 381, 2114–2124. [Google Scholar] [CrossRef]
- Traboulsee, A.; Greenberg, B.M.; Bennett, J.L.; Szczechowski, L.; Fox, E.; Shkrobot, S.; Yamamura, T.; Terada, Y.; Kawata, Y.; Wright, P.; et al. Safety and efficacy of satralizumab monotherapy in neuromyelitis optica spectrum disorder: A randomised, double-blind, multicentre, placebo-controlled phase 3 trial. Lancet Neurol. 2020, 19, 402–412. [Google Scholar] [CrossRef]
- Holmøy, T.; Høglund, R.A.; Illes, Z.; Myhr, K.M.; Torkildsen, Ø. Recent progress in maintenance treatment of neuromyelitis optica spectrum disorder. J. Neurol. 2020, 268, 4522–4536. [Google Scholar] [CrossRef] [PubMed]
- Tradtrantip, L.; Zhang, H.; Saadoun, S.; Phuan, P.W.; Lam, C.; Papadopoulos, M.C.; Bennett, J.L.; Verkman, A.S. Anti-aquaporin-4 monoclonal antibody blocker therapy for neuromyelitis optica. Ann. Neurol. 2012, 71, 314–322. [Google Scholar] [CrossRef] [PubMed]
- Tradtrantip, L.; Zhang, H.; Anderson, M.O.; Saadoun, S.; Phaun, P.W.; Papadopoulos, M.C.; Bennett, J.L.; Verkman, A.S. Small-molecule inhibitors of NMO-IgG binding to aquaporin-4 reduce astrocyte cytotoxicity in neuromyelitis optica. FASEB J. 2012, 26, 2197–2208. [Google Scholar] [CrossRef]
- Collin, M.; Olsen, A. Endo S, a novel secreted protein from Streptococcus pyogenes with endoglycosidase activity on human IgG. EMBO J. 2001, 20, 3046–3055. [Google Scholar] [CrossRef] [PubMed]
- Tradtrantip, L.; Asavapanumas, N.; Verkman, A.S. Therapeutic cleavage of anti-aquaporin-4 autoantibody in neuromyelitis optica by an IgG-selective proteinase. Mol. Pharmacol. 2013, 83, 1268–1275. [Google Scholar] [CrossRef]
- Kiessling, P.; Lledo-Garcia, R.; Watanabe, S.; Langdon, G.; Tran, D.; Bari, M.; Christodoulou, L.; Jones, E.; Price, G.; Smith, B.; et al. The FcRn inhibitor rozanolixizumab reduces human serum IgG concentration: A randomized phase 1 study. Sci. Transl. Med. 2017, 9, eaan1208. [Google Scholar] [CrossRef] [PubMed]
- Lipphardt, M.; Mühlhausen, J.; Kitze, B.; Heigl, F.; Mauch, E.; Helms, H.J.; Müller, G.A.; Koziolek, M.J. Immunoadsorption or plasma exchange in steroid-refractory multiple sclerosis and neuromyelitis optica. J. Clin. Apher. 2019, 34, 381–391. [Google Scholar] [CrossRef]
- Faissner, S.; Nikolayczik, J.; Chan, A.; Gold, R.; Yoon, M.S.; Haghikia, A. Immunoadsorption in patients with neuromyelitis optica spectrum disorder. Ther. Adv. Neurol. Disord. 2016, 9, 281–286. [Google Scholar] [CrossRef]
- Elsbernd, P.M.; Hoffman, W.R.; Carter, J.L.; Wingerchuk, D.M. Interleukin-6 inhibition with tocilizumab for relapsing MOG-IgG associated disorder (MOGAD): A case-series and review. Mult. Scler. Relat. Disord. 2021, 48, 102696. [Google Scholar] [CrossRef]
- Paton, D.M. Satralizumab: An interleukin-6 (IL-6) receptor antagonist for the treatment of neuromyelitis optica spectrum disorders. Drugs Today 2021, 57, 209–218. [Google Scholar] [CrossRef]
- Enspryng (Satralizumab-Mwge) Injection [Prescribing Information]. Food and Drug Administration (FDA). 2020. Available online: https://www.accessdata.fda.gov/drugsatfda_docs/label/2020/761149s000lbl.pdf. (accessed on 12 December 2022).
- Kieseier, B.C.; Stüve, O.; Dehmel, T.; Goebels, N.; Leussink, V.I.; Mausberg, A.K.; Ringelstein, M.; Turowski, B.; Aktas, O.; Antoch, G.; et al. Disease amelioration with tocilizumab in a treatment-resistant patient with neuromyelitis optica: Implication for cellular immune responses. JAMA Neurol. 2013, 70, 390–393. [Google Scholar] [CrossRef] [PubMed]
- Gruol, D.L. IL-6 regulation of synaptic function in the CNS. Neuropharmacology 2015, 96, 42–54. [Google Scholar] [CrossRef]
- Morizane, A.; Li, J.Y.; Brundin, P. From bench to bed: The potential of stem cells for the treatment of Parkinson’s disease. Cell Tissue. Res. 2008, 331, 323–336. [Google Scholar] [CrossRef] [PubMed]
- Fujimoto, Y.; Abematsu, M.; Falk, A.; Tsujimura, K.; Sanosaka, T.; Juliandi, B.; Semi, K.; Namihira, M.; Komiya, S.; Smith, A.; et al. Treatment of a mouse model of spinal cord injury by transplantation of human induced pluripotent stem cell-derived long-term self-renewing neuroepithelial-like stem cells. Stem Cells 2012, 30, 1163–1173. [Google Scholar] [CrossRef] [PubMed]
- Peng, F.; Qiu, W.; Li, J.; Hu, X.; Huang, R.; Lin, D.; Bao, J.; Jiang, Y.; Bian, L. A preliminary result of treatment of neuromyelitis optica with autologous peripheral hematopoietic stem cell transplantation. Neurologist 2010, 16, 375–378. [Google Scholar] [CrossRef]
- Zhang, P.; Liu, B. Effect of autologous hematopoietic stem cell transplantation on multiple sclerosis and neuromyelitis optica spectrum disorder: A PRISMA-compliant meta-analysis. Bone Marrow Transplant. 2020, 55, 1928–1934. [Google Scholar] [CrossRef] [PubMed]
- Burt, R.K.; Balabanov, R.; Han, X.; Burns, C.; Gastala, J.; Jovanovic, B.; Helenowski, I.; Jitprapaikulsan, J.; Fryer, J.P.; Pittock, S.J. Autologous nonmyeloablative hematopoietic stem cell transplantation for neuromyelitis optica. Neurology 2019, 93, e1732–e1741. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).