Development of a Simple Protocol for Zymogram-Based Isolation and Characterization of Gingipains from Porphyromonas gingivalis: The Causative Agent of Periodontitis
Abstract
Featured Application
Abstract
1. Introduction
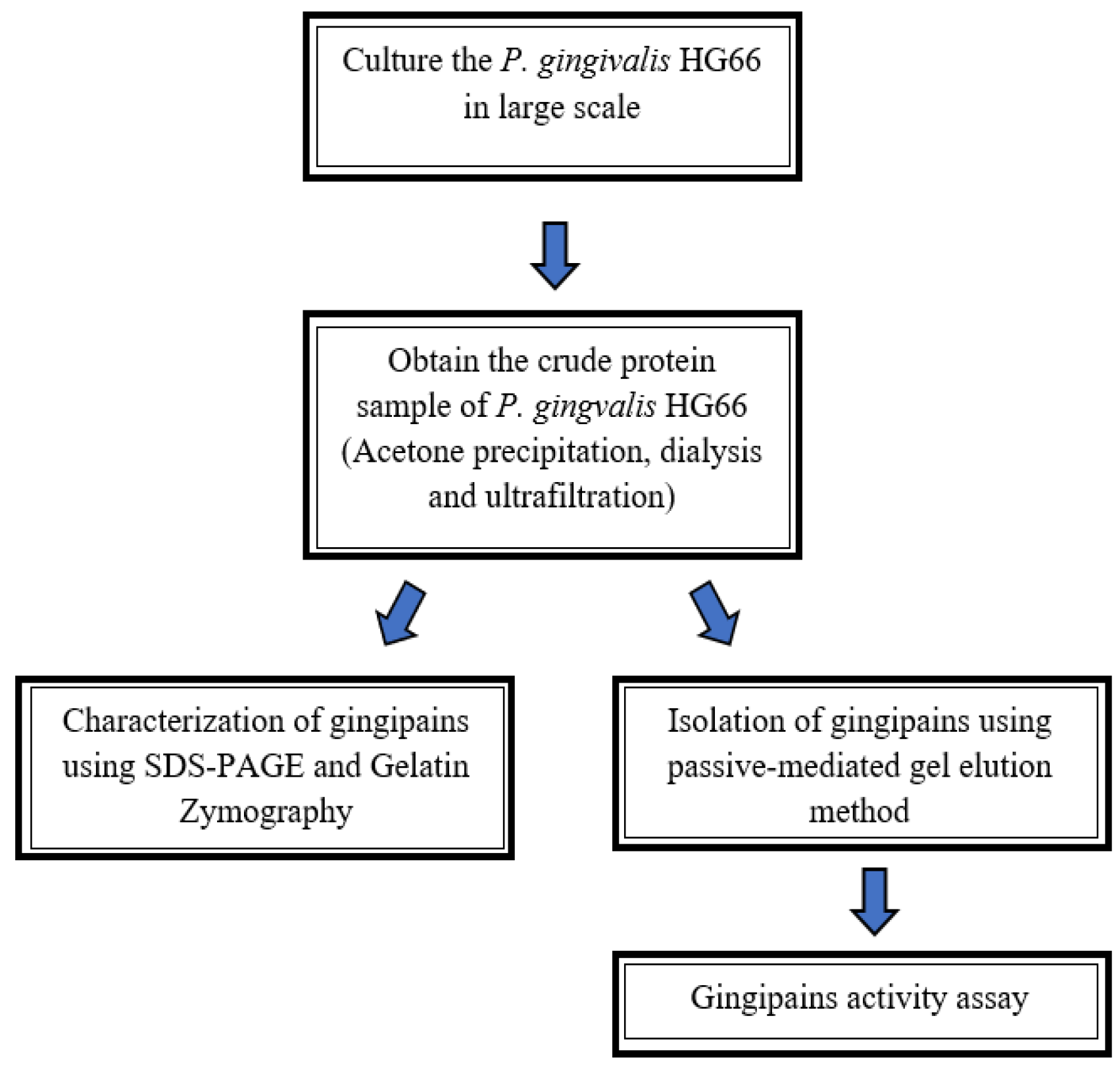
2. Materials and Methods
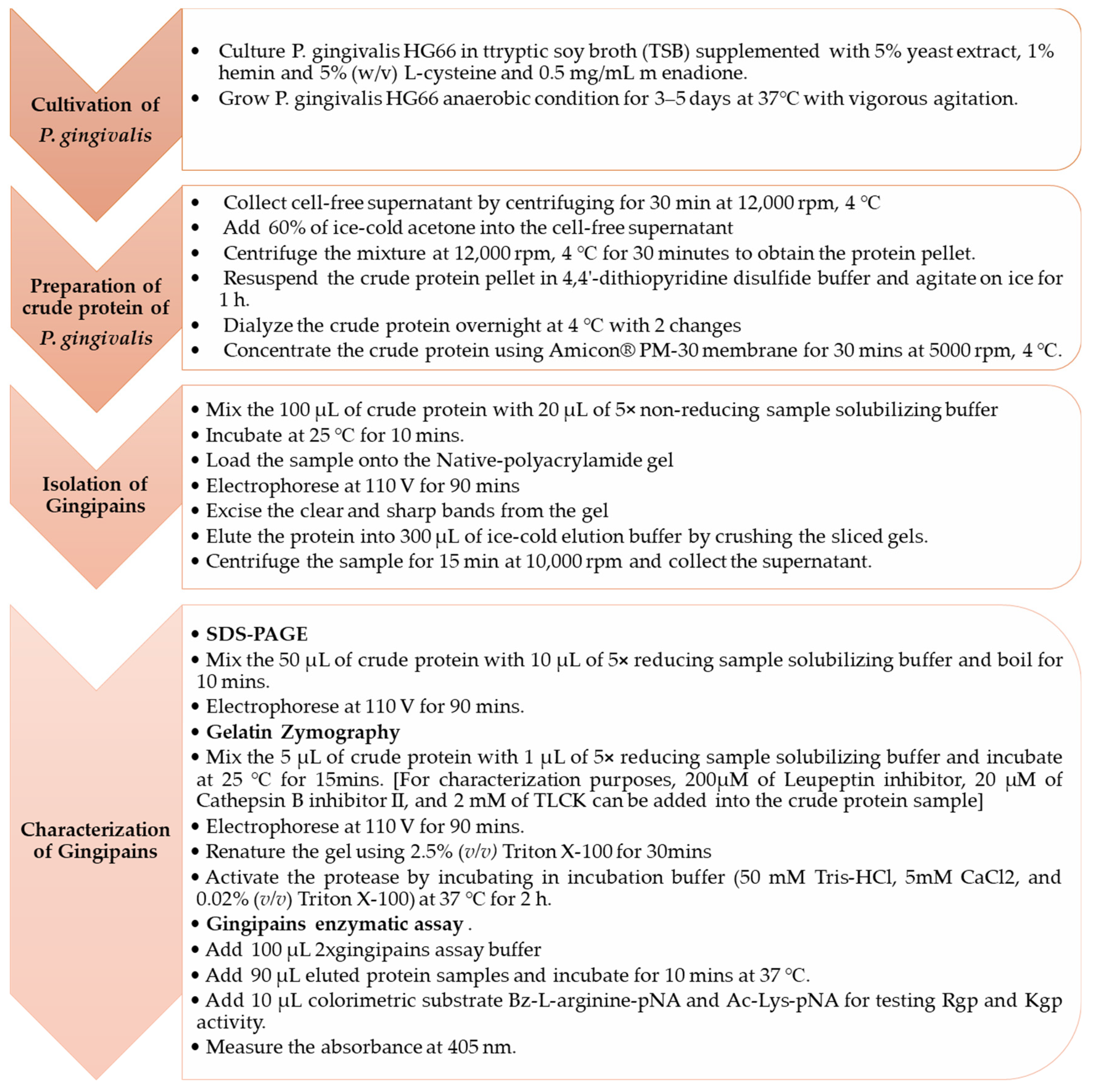
2.1. Cultivation of P. gingivalis HG66 Strain
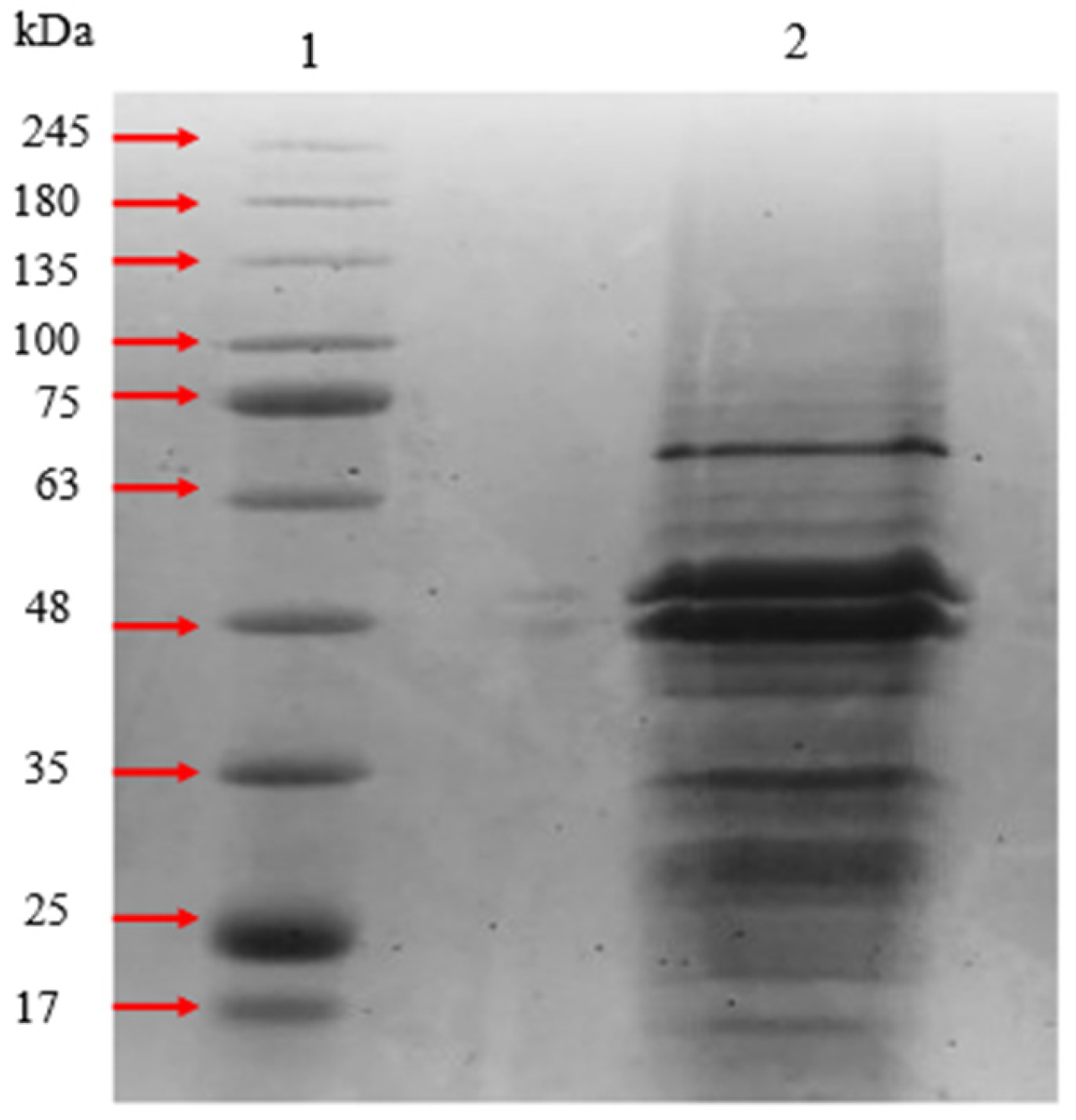
2.2. Preparation of Crude Protein of P. gingivalis HG66
2.3. Isolation of Gingipains Using Passive Diffusion-Mediated Gel Elution Method
2.3.1. Sample Preparation
2.3.2. Native-PAGE Electrophoresis
2.3.3. Elution of Protein
2.3.4. Gingipains Activity Assay
2.4. Characterization of Gingipains
2.4.1. SDS-PAGE
2.4.2. Gelatin Zymography
Sample Preparation
Gelatin Zymography
Gel Renaturation and Incubation
Gel Staining
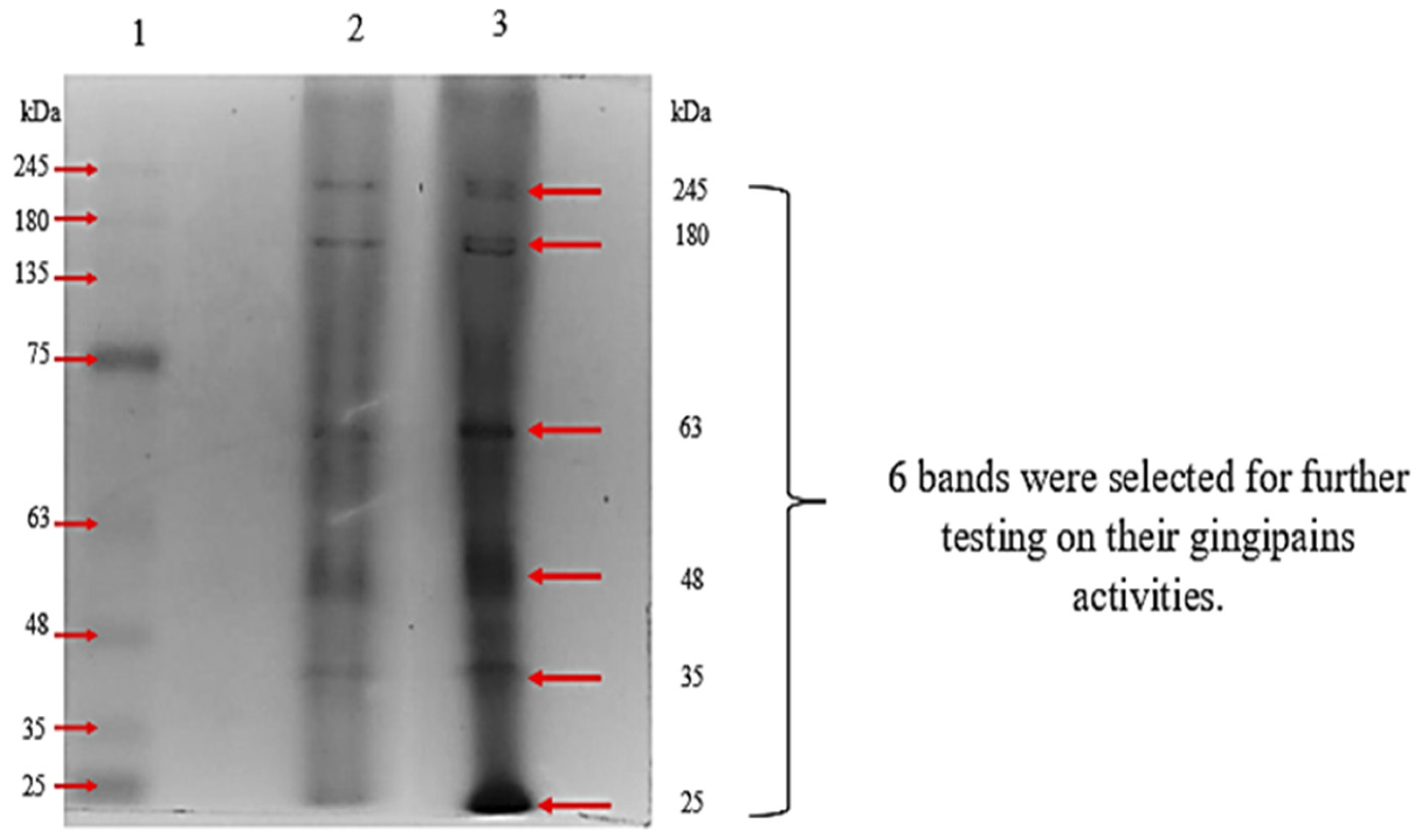
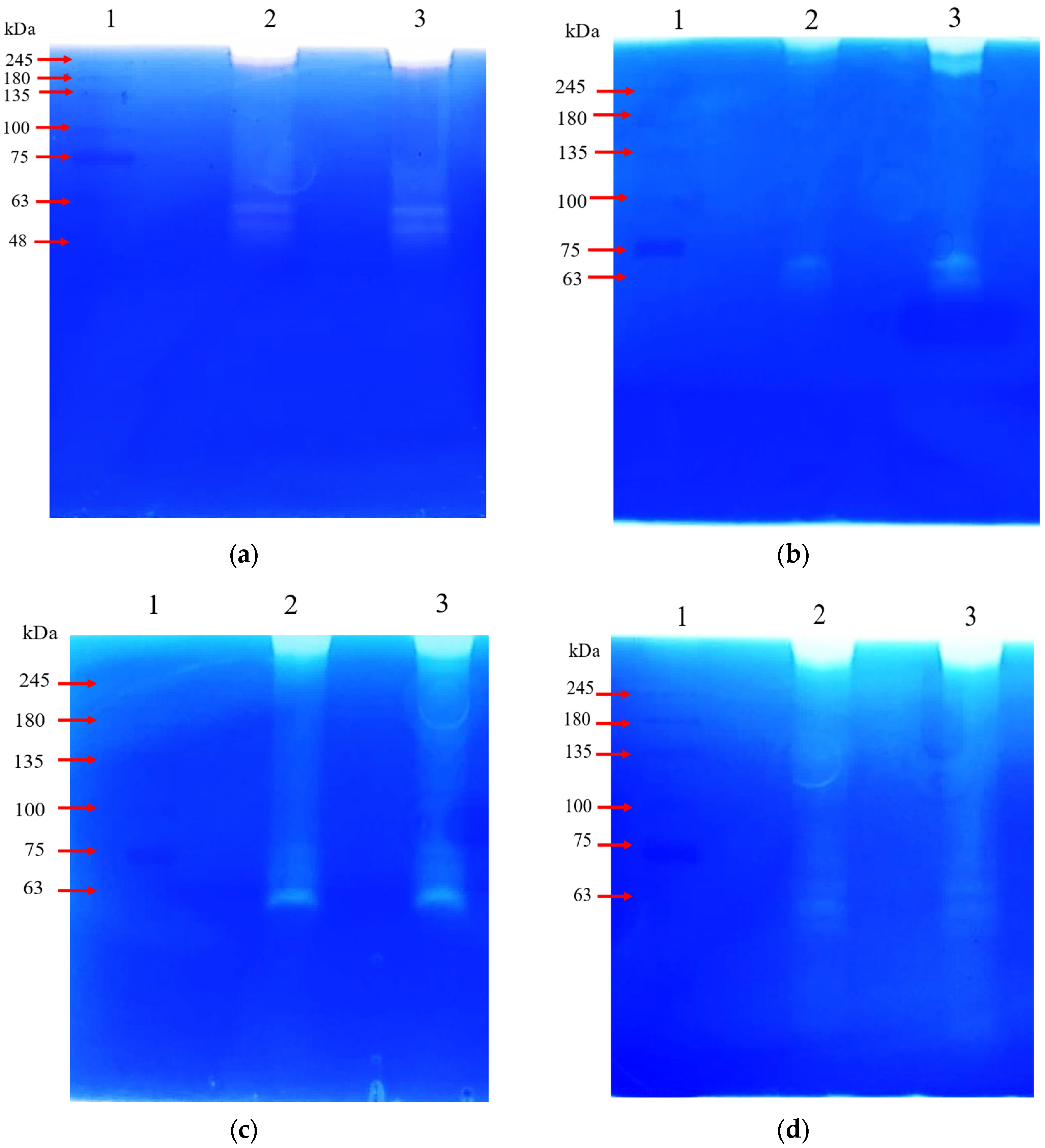
3. Results
3.1. Purification: Passive-Diffusion Mediated Elution Method
3.2. Characterization of Gingipains
3.2.1. SDS-PAGE
3.2.2. Gelatin Zymography
4. Discussion
4.1. Developing and Detailing a Protocol for Isolation and Characterization of Gingipains
4.1.1. Gingipains Preparation
- 1.
- Prepare 500 mL of P. gingivalis HG66 cell-free culture supernatant by centrifuging it for 30 min at 12,000 rpm, 4 °C. Discard the pellet and preserve the supernatant on ice.
- 2.
- Add 750 mL of ice-cold acetone into the brownish supernatant slowly with vigorous agitation.
- 3.
- Obtain the protein pellet by centrifugation for 30 min at 12,000 rpm, 4 °C.
- 4.
- Resuspend the protein pellet with 10 mL of ice-cold 4,4′-dithiopyridine disulfide buffer (DTDS buffer) and incubate on ice for 1 h with agitation.
- 5.
- Transfer the protein into a dialysis bag (MWCO 12,000 to 14,000) and dialyze overnight at 4 °C with 2 changes.
- 6.
- Concentrate the protein with ultrafiltration using Amicon PM-30 membrane for 30 min at 5000 rpm, 4 °C.
- 7.
- Keep the protein sample at −20 °C for future use.
4.1.2. Isolation of Gingipains Using Passive-Diffusion Mediated Gel Elution Method
- Sample preparation
- 1.
- Transfer 100 µL of gingipains sample into the sterile 1.5 mL microcentrifuge tube.
- 2.
- Mix the gingipain sample with 20 µL of 5× non-reducing solubilizing sample buffer (without SDS and β-mercaptoethanol)
- 3.
- Load samples onto Native-polyacrylamide gel.
- Perform Gel electrophoresis
- Elution of protein
- 1.
- Cut the electrophoresed gel into 3 strips with a sterile blade.
- 2.
- Stain the protein ladder and one sample gel strip with staining buffer for 30 min with gentle agitation
- 3.
- Cut the target protein bands using the sterile blade and transfer them into the sterile 1.5 mL microcentrifuge tube.
- 4.
- Add 300 µL of ice-cold elution buffer.
- 5.
- Crush the sliced gel with a disposable homogenizer and incubate for overnight, at 4 °C.
- 6.
- Centrifuge the sample for 15 min at 10,000 rpm and collect the supernatant.
- 7.
- Test the eluted proteins with a gingipains activity assay.
- Gingipain activity assay
- 1.
- Prepare the gingipains sample of 20 µL and 50 µL for Rgp and Kgp activity assay, respectively, and dilute the samples with deionized water, making the total volume of 90 µL.
- 2.
- Add 100 µL of 2× gingipain assay buffer to the 96-wells flat bottom microtiter plates.
- 3.
- Add 90 µL of prepared gingipains sample as test group; add 90 µL of deionized water as positive control; add 100 µL of deionized water as blank.
- 4.
- Mix the samples well by pipetting them up and down gently.
- 5.
- Incubate the samples for 10 min at 37 °C.
- 6.
- Add 10 µL colorimetric substrate, Bz-L-arginine-pNA (L-BAPNA), and Ac-Lys-pNA for Rgp and Kgp activity assay respectively.
- 7.
- Measure the absorbance at 405 nm.
4.1.3. Characterization of Gingipains
SDS-PAGE
- Sample preparation
- 1.
- Transfer 50 µL of gingipains sample into the sterile 1.5 mL microcentrifuge tube.
- 2.
- Mix the gingipain sample with 10 µL of 5× non-reducing sample solubilizing buffer and 6 µL of 10% β-mercaptoethanol.
- 3.
- Boil the sample for 10 min at 100 °C.
- 4.
- Load the samples onto SDS-polyacrylamide gel.
- Perform Gel electrophoresis
- Gel staining
- 1.
- Stain the gel with Coomassie gel staining solution for 30 min at room temperature while agitating it gently.
- 2.
- De-stain the gel with the de-staining solution for 30 min until the clear and sharp, blue-colored bands are visible.
- Gel viewing
Gelatin Zymography
- Sample preparation
- 1.
- Transfer 5 µL of gingipains sample into the sterile 1.5 mL microcentrifuge tube.
- 2.
- Mix the gingipain sample with 1 µL of 5× non-reducing sample solubilizing buffer (without β-mercaptoethanol)
- 3.
- Add 200 µM of Leupeptin inhibitor (Rgp selective inhibitor), 20 µM of Cathepsin B inhibitor II (Kgp selective inhibitor), and 2 mM of TLCK (general inhibitor) into gingipains samples separately.
- 4.
- Incubate the samples for 15 min at room temperature.
- 5.
- Load samples onto SDS-gelatin-polyacrylamide gel.
- Perform Gel electrophoresis
- Gel Renaturation/Incubation
- 1.
- After electrophoresis, remove the gel from the electrophoretic gel cassette carefully.
- 2.
- Place the gel in a clean plastic container and rinse it with distilled water twice for 15 min with gentle agitation.
- 3.
- Equilibrate the gel with 100 mL of renaturation buffer for 30 min at room temperature while agitating it gently.
- 4.
- Incubate and activate the proteases present in the gel with 100 mL of incubation buffer for 2 h at 37 °C.
- Gel Staining
- 1.
- Stain the gel with Coomassie gel staining solution for 30 min at room temperature while agitating it gently.
- 2.
- De-stain the gel with de-staining buffer for 30 min until the transparent and sharp bands are observed.
- Gel viewing
4.1.4. Chemicals
4.1.5. Equipment
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Nazir, M.A. Prevalence of Periodontal Disease, Its Association with Systemic Diseases and Prevention. Int. J. Health Sci. 2017, 11, 72–80. [Google Scholar]
- Fitzpatrick, R.E.; Wijeyewickrema, L.C.; Pike, R.N. The Gingipains: Scissors and Glue of the Periodontal Pathogen, Porphyromonas gingivalis. Future Microbiol. 2009, 4, 471–487. [Google Scholar] [CrossRef]
- Dominy, S.S.; Lynch, C.; Ermini, F.; Benedyk, M.; Marczyk, A.; Konradi, A.; Nguyen, M.; Haditsch, U.; Raha, D.; Griffin, C.; et al. Porphyromonas gingivalis in Alzheimer’s Disease Brains: Evidence for Disease Causation and Treatment with Small-Molecule Inhibitors. Sci. Adv. 2019, 5, eaau3333. [Google Scholar] [CrossRef]
- Peng, H.-Y.; Chen, S.-Y.; Siao, S.-H.; Chang, J.T.; Xue, T.-Y.; Lee, Y.-H.; Jan, M.-S.; Tsay, G.J.; Zouali, M. Targeting a Cysteine Protease from a Pathobiont Alleviates Experimental Arthritis. Arthritis Res. Ther. 2020, 22, 114. [Google Scholar] [CrossRef]
- Gómez-Bañuelos, E.; Mukherjee, A.; Darrah, E.; Andrade, F. Rheumatoid Arthritis-Associated Mechanisms of Porphyromonas gingivalis and Aggregatibacter Actinomycetemcomitans. J. Clin. Med. 2019, 8, 1309. [Google Scholar] [CrossRef]
- Haraszthy, V.; Zambon, J.; Trevisan, M.; Zeid, M.; Genco, R. Identification of Periodontal Pathogens in Atheromatous Plaques. J. Periodontol. 2000, 71, 1554–1560. [Google Scholar] [CrossRef]
- Szulc, M.; Kustrzycki, W.; Janczak, D.; Michalowska, D.; Baczynska, D.; Radwan-Oczko, M. Presence of Periodontopathic Bacteria DNA in Atheromatous Plaques from Coronary and Carotid Arteries. BioMed Res. Int. 2015, 2015, e825397. [Google Scholar] [CrossRef]
- Bostanci, N.; Belibasakis, G.N. Porphyromonas gingivalis: An Invasive and Evasive Opportunistic Oral Pathogen. FEMS Microbiol. Lett. 2012, 333, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Nakayama, K. Porphyromonas gingivalis and Related Bacteria: From Colonial Pigmentation to the Type IX Secretion System and Gliding Motility. J. Periodontal Res. 2014, 50, 12255. [Google Scholar] [CrossRef]
- Gruss, A.; Borezée-Durant, E.; Lechardeur, D. Chapter Three—Environmental Heme Utilization by Heme-Auxotrophic Bacteria. In Advances in Microbial Physiology; Poole, R.K., Ed.; Advances in Bacterial Respiratory Physiology; Academic Press: Cambridge, MA, USA, 2012; Volume 61, pp. 69–124. [Google Scholar]
- Zhang, Z.; Liu, D.; Liu, S.; Zhang, S.; Pan, Y. The Role of Porphyromonas gingivalis Outer Membrane Vesicles in Periodontal Disease and Related Systemic Diseases. Front. Cell. Infect. Microbiol. 2021, 10, 585917. [Google Scholar] [CrossRef]
- Singhrao, S.K.; Harding, A.; Poole, S.; Kesavalu, L.; Crean, S. Porphyromonas gingivalis Periodontal Infection and Its Putative Links with Alzheimer’s Disease. Mediat. Inflamm. 2015, 2015, 137357. [Google Scholar] [CrossRef]
- Kavitha, R.; Sa’ad, M.A.; Fuloria, S.; Fuloria, N.K.; Ravichandran, M.; Lalitha, P. Synthesis, Characterization, Cytotoxicity Analysis and Evaluation of Novel Heterocyclic Derivatives of Benzamidine against Periodontal Disease Triggering Bacteria. Antibiotics 2023, 12, 306. [Google Scholar] [CrossRef]
- Bao, K.; Belibasakis, G.N.; Thurnheer, T.; Aduse-Opoku, J.; Curtis, M.A.; Bostanci, N. Role of Porphyromonas gingivalis Gingipains in Multi-Species Biofilm Formation. BMC Microbiol. 2014, 14, 258. [Google Scholar] [CrossRef]
- Imamura, T. The Role of Gingipains in the Pathogenesis of Periodontal Disease. J. Periodontol. 2003, 74, 111–118. [Google Scholar] [CrossRef]
- Kadowaki, T. Enzymatic Characteristics and Activities of Gingipains from Porphyromonas gingivalis. Methods Mol. Biol. 2021, 2210, 97–112. [Google Scholar] [CrossRef]
- Veillard, F.; Sztukowska, M.; Nowakowska, Z.; Mizgalska, D.; Thøgersen, I.B.; Enghild, J.J.; Bogyo, M.; Potempa, B.; Nguyen, K.-A.; Potempa, J. Proteolytic Processing and Activation of Gingipain Zymogens Secreted by T9SS of Porphyromonas gingivalis. Biochimie 2019, 166, 161–172. [Google Scholar] [CrossRef]
- Sugawara, S.; Nemoto, E.; Tada, H.; Miyake, K.; Imamura, T.; Takada, H. Proteolysis of Human Monocyte CD14 by Cysteine Proteinases (Gingipains) from Porphyromonas gingivalis Leading to Lipopolysaccharide Hyporesponsiveness. J. Immunol. 2000, 165, 411–418. [Google Scholar] [CrossRef]
- Jia, L.; Han, N.; Du, J.; Guo, L.; Luo, Z.; Liu, Y. Pathogenesis of Important Virulence Factors of Porphyromonas gingivalis via Toll-Like Receptors. Front. Cell. Infect. Microbiol. 2019, 9, 262. [Google Scholar] [CrossRef]
- Chen, Z.; Potempa, J.; Polanowski, A.; Wikstrom, M.; Travis, J. Purification and Characterization of a 50-KDa Cysteine Proteinase (Gingipain) from Porphyromonas gingivalis. J. Biol. Chem. 1992, 267, 18896–18901. [Google Scholar] [CrossRef] [PubMed]
- Potempa, J.; Nguyen, K.-A. Purification and Characterization of Gingipains. Curr. Protoc. Protein. Sci. 2007, 21, Unit 21.20. [Google Scholar] [CrossRef]
- McGraw, W.T.; Potempa, J.; Farley, D.; Travis, J. Purification, Characterization, and Sequence Analysis of a Potential Virulence Factor from Porphyromonas gingivalis, Peptidylarginine Deiminase. Infect. Immun. 1999, 67, 3248–3256. [Google Scholar] [CrossRef]
- Pavloff, N.; Pemberton, P.A.; Potempa, J.; Chen, W.C.; Pike, R.N.; Prochazka, V.; Kiefer, M.C.; Travis, J.; Barr, P.J. Molecular Cloning and Characterization of Porphyromonas gingivalis Lysine-Specific Gingipain. A New Member of an Emerging Family of Pathogenic Bacterial Cysteine Proteinases. J. Biol. Chem. 1997, 272, 1595–1600. [Google Scholar] [CrossRef]
- Margetts, M.B.; Barr, I.G.; Webb, E.A. Overexpression, Purification, and Refolding of a Porphyromonas gingivalis Cysteine Protease from Escherichia Coli. Protein Expr. Purif. 2000, 18, 262–268. [Google Scholar] [CrossRef]
- Arndt, C.; Koristka, S.; Feldmann, A.; Bachmann, M. Native Polyacrylamide Gels. In Electrophoretic Separation of Proteins: Methods and Protocols; Kurien, B.T., Scofield, R.H., Eds.; Methods in Molecular Biology; Springer: New York, NY, 2019; pp. 87–91. ISBN 978-1-4939-8793-1. [Google Scholar]
- Burgess, R.R. Elution of Proteins from Gels. Methods Enzym. 2009, 463, 565–572. [Google Scholar] [CrossRef]
- Toth, M.; Fridman, R. Assessment of Gelatinases (MMP-2 and MMP-9) by Gelatin Zymography. Methods Mol. Med. 2001, 57, 163. [Google Scholar] [CrossRef]
- Hashimoto, Y. Gelatin Zymography Using Leupeptin for the Detection of Various Cathepsin L Forms. Methods Mol. Biol. 2017, 1594, 243–254. [Google Scholar] [CrossRef]
- Potempa, J.; Mikolajczyk-Pawlinska, J.; Brassell, D.; Nelson, D.; Thøgersen, I.B.; Enghild, J.J.; Travis, J. Comparative Properties of Two Cysteine Proteinases (Gingipains R), the Products of Two Related but Individual Genes of Porphyromonas Gingivalis. J. Biol. Chem. 1998, 273, 21648–21657. [Google Scholar] [CrossRef]
- Platt, M.O.; Ankeny, R.F.; Jo, H. Laminar Shear Stress Inhibits Cathepsin L Activity in Endothelial Cells. Arter. Thromb. Vasc. Biol. 2006, 26, 1784–1790. [Google Scholar] [CrossRef]
- Jung, Y.-J.; Jun, H.-K.; Choi, B.-K. Porphyromonas Gingivalis Suppresses Invasion of Fusobacterium Nucleatum into Gingival Epithelial Cells. J. Oral Microbiol. 2017, 9, 1320193. [Google Scholar] [CrossRef]
- Mei, F.; Xie, M.; Huang, X.; Long, Y.; Lu, X.; Wang, X.; Chen, L. Porphyromonas gingivalis and Its Systemic Impact: Current Status. Pathogens 2020, 9, 944. [Google Scholar] [CrossRef] [PubMed]
- Reyes, L. Porphyromonas gingivalis. Trends Microbiol. 2021, 29, 376–377. [Google Scholar] [CrossRef]
- Leonard, A.K.; Loughran, E.A.; Klymenko, Y.; Liu, Y.; Kim, O.; Asem, M.; McAbee, K.; Ravosa, M.J.; Stack, M.S. Chapter 4—Methods for the Visualization and Analysis of Extracellular Matrix Protein Structure and Degradation. In Methods in Cell Biology; Mecham, R.P., Ed.; Methods in Extracellular Matrix Biology; Academic Press: Cambridge, MA, USA, 2018; Volume 143, pp. 79–95. [Google Scholar]
- Ciborowski, P.; Nishikata, M.; Allen, R.D.; Lantz, M.S. Purification and Characterization of Two Forms of a High-Molecular-Weight Cysteine Proteinase (Porphypain) from Porphyromonas gingivalis. J. Bacteriol. 1994, 176, 4549–4557. [Google Scholar] [CrossRef]
- Hočevar, K.; Vizovišek, M.; Wong, A.; Kozieł, J.; Fonović, M.; Potempa, B.; Lamont, R.J.; Potempa, J.; Turk, B. Proteolysis of Gingival Keratinocyte Cell Surface Proteins by Gingipains Secreted From Porphyromonas gingivalis—Proteomic Insights Into Mechanisms Behind Tissue Damage in the Diseased Gingiva. Front. Microbiol. 2020, 11, 722. [Google Scholar] [CrossRef]
- Potempa, J.; Pike, R.; Travis, J. The Multiple Forms of Trypsin-like Activity Present in Various Strains of Porphyromonas gingivalis Are Due to the Presence of Either Arg-Gingipain or Lys-Gingipain. Infect. Immun. 1995, 63, 1176–1182. [Google Scholar] [CrossRef]
- Sa’ad, M.A.; Kavitha, R.; Fuloria, N.K.; Fuloria, S.; Ravichandran, M.; Lalitha, P. The virulence system of Porphyromonas gingivalis: Genes, mechanism and potential role of gingipains inhibitors. Malays. J. Microbiol. 2021, 17, 212–226. [Google Scholar] [CrossRef]
- Curtis, M.A.; Aduse-Opoku, J.; Rangarajan, M. Cysteine Proteases of Porphyromonas gingivalis. Crit. Rev. Oral Biol. Med. 2001, 12, 192–216. [Google Scholar] [CrossRef]
- Kim, R. Native Agarose Gel Electrophoresis of Multiprotein Complexes. Cold Spring Harb. Protoc. 2011, 2011, 884–887. [Google Scholar] [CrossRef] [PubMed]
- Kurien, B.T.; Scofield, R.H. Extraction of Proteins from Gels—A Brief Review. Methods Mol. Biol. 2012, 869, 403–405. [Google Scholar] [CrossRef]
- Hosn, K.N.; Jefferson, M.M.; Leding, C.; Shokouh-Amiri, S.; Thomas, E.L. Inhibitors of Bacterial Protease Enzymes for Periodontal Therapy. Clin. Exp. Dent. Res. 2015, 1, 18–25. [Google Scholar] [CrossRef]
- Grenier, D.; Imbeault, S.; Plamondon, P.; Grenier, G.; Nakayama, K.; Mayrand, D. Role of Gingipains in Growth of Porphyromonas gingivalis in the Presence of Human Serum Albumin. Infect. Immun. 2001, 69, 5166–5172. [Google Scholar] [CrossRef] [PubMed]
- Grudkowska, M.; Lisik, P.; Rybka, K. Two-Dimensional Zymography in Detection of Proteolytic Enzymes in Wheat Leaves. Acta Physiol. Plant 2013, 35, 3477–3482. [Google Scholar] [CrossRef]
| Samples | Positive Control 1 | ~25 kDa 2 | ~35 kDa 2 | ~48 kDa 2 | ~63 kDa 2 | ~180 kDa 2 | ~245 kDa 2 |
|---|---|---|---|---|---|---|---|
| Absorbance (OD405nm) 3 | |||||||
| Rgp activity | 0.073 | 0.074 | 0.064 | 0.078 | 0.075 | 0.101 | 0.073 |
| Kgp activity | 0.079 | 0.085 | 0.085 | 0.061 | 0.081 | 0.091 | 0.080 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wei, E.S.; Kavitha, R.; Sa’ad, M.A.; Lalitha, P.; Fuloria, N.K.; Ravichandran, M.; Fuloria, S. Development of a Simple Protocol for Zymogram-Based Isolation and Characterization of Gingipains from Porphyromonas gingivalis: The Causative Agent of Periodontitis. Appl. Sci. 2023, 13, 4314. https://doi.org/10.3390/app13074314
Wei ES, Kavitha R, Sa’ad MA, Lalitha P, Fuloria NK, Ravichandran M, Fuloria S. Development of a Simple Protocol for Zymogram-Based Isolation and Characterization of Gingipains from Porphyromonas gingivalis: The Causative Agent of Periodontitis. Applied Sciences. 2023; 13(7):4314. https://doi.org/10.3390/app13074314
Chicago/Turabian StyleWei, Eng Sze, Ramasamy Kavitha, Mohammad Auwal Sa’ad, Pattabhiraman Lalitha, Neeraj Kumar Fuloria, Manickam Ravichandran, and Shivkanya Fuloria. 2023. "Development of a Simple Protocol for Zymogram-Based Isolation and Characterization of Gingipains from Porphyromonas gingivalis: The Causative Agent of Periodontitis" Applied Sciences 13, no. 7: 4314. https://doi.org/10.3390/app13074314
APA StyleWei, E. S., Kavitha, R., Sa’ad, M. A., Lalitha, P., Fuloria, N. K., Ravichandran, M., & Fuloria, S. (2023). Development of a Simple Protocol for Zymogram-Based Isolation and Characterization of Gingipains from Porphyromonas gingivalis: The Causative Agent of Periodontitis. Applied Sciences, 13(7), 4314. https://doi.org/10.3390/app13074314







