Abstract
Africa, particularly sub-Sharan Africa (SSA), faces major challenges in respect to chronic kidney disease (CKD). There is a rising prevalence due to the combined effects of hypertension, diabetes, and human immunodeficiency virus (HIV) (and the interaction between them) and the effect of apolipoprotein L1 (APOL1) variants on the susceptibility to CKD. Epidemiological data on the prevalence of CKD are of low-to-medium quality, and reliable data are urgently needed for health planning. Furthermore, there are important deficiencies in creatinine-based equations in underestimating the prevalence of CKD in Africa, and evidence suggests that cystatin C based equations are more reliable. There is a changing spectrum of HIV related CKD with the greater availability of antiretroviral treatment. Major clinical trials using SGLT2 inhibitors have signalled a major advance in the treatment of CKD, especially in relation to type 2 diabetes, but the affordability, availability, and relevance to the African population is not established. The importance of the effects of hypertension in pregnancy and pregnancy related acute kidney injury on CKD and the newer concept of CKD of unknown cause (CKDu) are highlighted. Hypertension remains a dominant cause of CKD in Africa, and newer information suggests that the most appropriate treatment to control blood pressure and thus prevent CKD is the combination of either amlodipine plus a thiazide diuretic or angiotensin converting enzyme (ACE) inhibitor.
1. Introduction
At a global level, 7 of the 10 leading causes of deaths in 2019 were noncommunicable diseases and accounted for 74% of deaths globally [1]. Chronic kidney disease (CKD) is increasingly recognised as an important and rising contributor to mortality. In 1990, there were 601,307 deaths due to CKD, which was ranked 16th as a cause of global deaths. In 2019, there were 1.43 million deaths, adjusting the rank to 10th, representing a 2.37-fold increase.
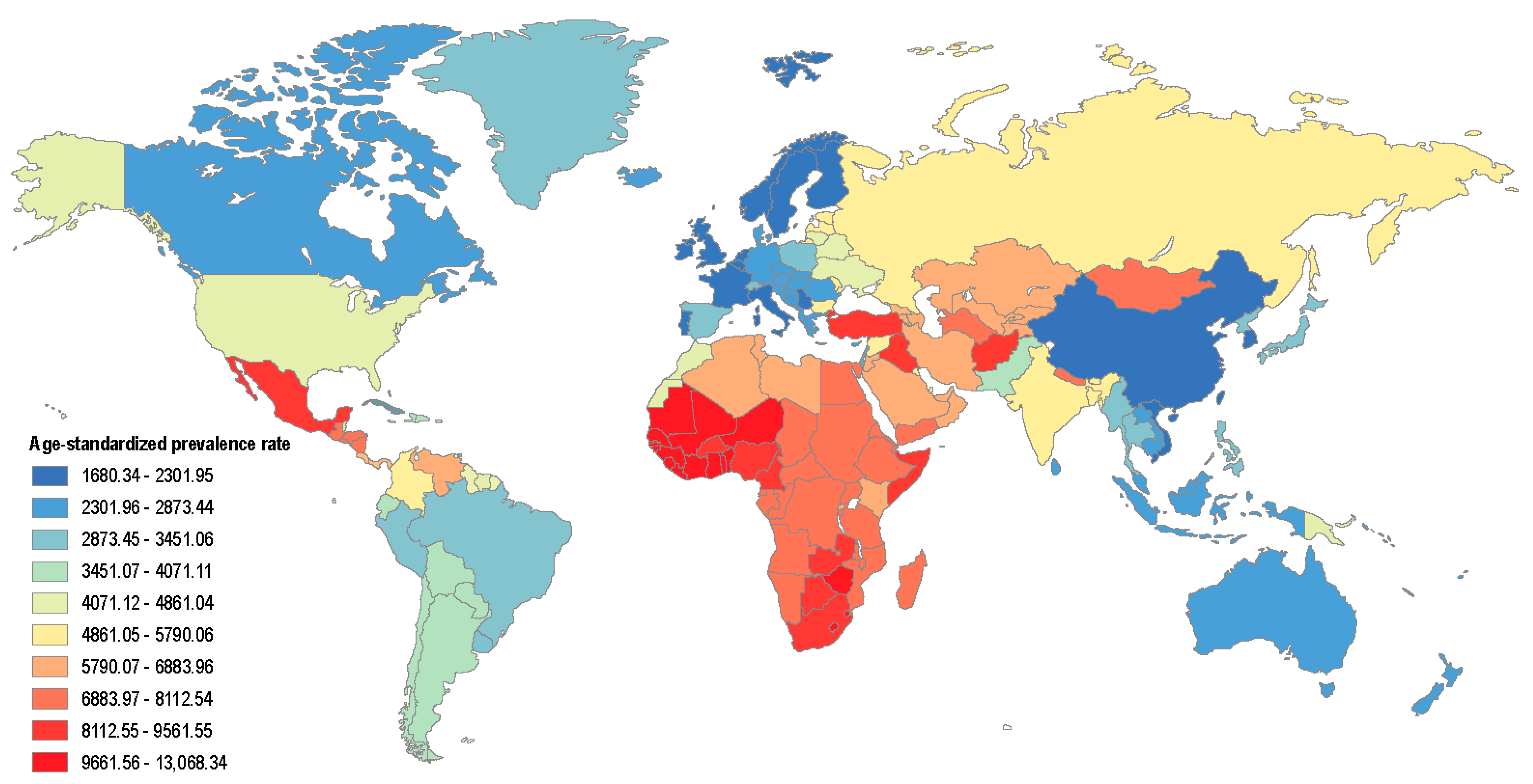
In 2017, 697.5 million cases of all-stage CKD were recorded with a global prevalence of 9.1%, representing a 29.3% increase in CKD [2]. CKD resulted in 35.8 million disability-adjusted life years (DALYs) in 2017, with diabetic nephropathy accounting for almost a third of them. Most of the burden of CKD was concentrated in the three lowest quintiles of the socio-demographic index, but in several regions, particularly Oceania, sub-Saharan Africa (SSA), and Latin America, the burden of CKD was much higher than expected for the level of development. The age-adjusted global prevalence of CKD is shown in Figure 1, demonstrating a high prevalence in Africa compared to the rest of the world [3] (Figure 1). Additionally, 1.4 million cardiovascular (CV) disease-related deaths and 25.3 million CV disease DALYs were attributed to CKD.
Figure 1.
The worldwide age-standardised global prevalence of CKD per 100,000 population in 2016 (adapted from reference [3]).
The regional prevalence of CKD in SSA ranges from 12.2 to 16.5%, based on a recent systematic review, but these estimates are based on mainly low-quality studies [4]. Nevertheless, this is considerably higher than the global prevalence of 9.1%. It is, therefore, timely to discuss the challenges and advances in CKD in SSA. This review will focus on advances related to estimating glomerular filtration rate (GFR) in Africans, HIV related CKD, diabetic kidney disease, hypertension, CKD after hypertension in pregnancy, the interaction of HIV with diabetes/hypertension, and other topical issues.
2. Challenges of Estimating GFR in SSA
Estimating GFR (eGFR) based on the serum creatinine is the most practical way of determining the prevalence of CKD. The most common methods used are the Modification of Diet in Renal Disease (MDRD) and Chronic Kidney Disease Epidemiology Collaboration (CKD-EPI) equations, which have been developed in high income countries and have not been validated against measured GFR (mGFR) in other regions, including SSA. Furthermore, both MDRD and CKD-EPI use a race-based correction factor for African Americans, which has become particularly controversial. In African Americans living with HIV, the racial adjustment adversely affected GFR estimates [5]. A new equation developed for both creatinine and cystatin C without racial adjustment appears to work reasonably well. However, eGFR was underestimated compared to mGFR in Blacks (median, 3.6 mL per minute per 1.73 m2) and overestimated compared to mGFR in non-Blacks (median, 3.9 mL per minute per 1.73 m2) [6].
Whether this correction factor is applicable to Africans living in Africa is also controversial as no major study has been performed comparing eGFR to mGFR in SSA. As a result, the true prevalence of CKD in Africa may be either overestimated or, more likely, underestimated. Accurately determining the prevalence of CKD in any country or region has major implications for health planning, given the high costs of treating CKD and the high associated morbidity and mortality.
In a recent study by Fabian et al. conducted in Uganda, Malawi, and South Africa, 3025 subjects underwent determination of mGFR using the slope-intercept method for iohexol plasma clearance, and this was compared to eGFR using a variety of creatinine and cystatin C based equations with or without the correction factor for African Americans [7]. Complete data and adequate quality mGFR were available from 2578 participants. This demonstrated that creatinine-based equations overestimated kidney function compared to mGFR, which was worsened by correcting for ethnicity. The greatest bias occurred with reduced kidney function. The proportion of subjects with a mGFR < 60 mL/min was more than double compared to a creatinine based eGFR. Cystatin C equations outperformed all creatinine-based equations. The imputed prevalence of CKD based on mGFR was more than two-times higher than creatinine-based eGFR in six countries in Africa [7]. The over estimation of GFR and under estimation of CKD impacts both clinical care and public health in SSA.
The reasons for the poor performance of creatinine based eGFR is not known. Non-GFR determinants of creatinine, rather than ethnicity, might underlie greater inaccuracy. Lower muscle mass with less creatinine generation due to growth stunting, wasting from chronic infection or inflammation (e.g., tuberculosis (TB) and HIV), lower dietary protein, and undiagnosed liver disease may all contribute to these inaccuracies. Renal tubular handling of creatinine might be affected by antiretroviral therapy (ART) and by variants in genes affecting creatinine production and tubular secretion.
These findings have highly relevant implications for detecting CKD in SSA. Cystatin C measurement is not generally available in Africa and, in addition, costs significantly more than measuring creatinine. Increasing the accessibility of cystatin C and driving the cost down seems the only practical method of overcoming this problem, although further research for other biomarkers is needed.
3. The Importance of APOL1 and CKD
Despite probable under-reporting, there are data that suggest that Africa has a higher prevalence of CKD and that genetic risk plays an important role in CKD prevalence. The APOL1 gene encodes for the protein apolipoprotein L1. Historically, this gene grants innate immunity against most strains of Trypanosoma brucei. Two gain-of-function Apolipoprotein L1 (APOL1) variants G1 or G2 were first determined among individuals of African descent with a high prevalence found in Western Africa [8,9]. The African diaspora led to the widespread distribution of these coding variants, reaching frequencies of 21% for G1 and 13% for G2 in African Americans [10]. APOL1 risk variants have been strongly associated with an increased risk of developing focal segmental glomerulosclerosis (OR 10.5–16.9), HIV-associated nephropathy (HIVAN) (OR 29.2), and, possibly, increasing the risk of developing other causes of CKD, such as hypertensive nephrosclerosis (OR 7.3) [11]. APOL1 risk variants have also been strongly associated with HIVAN in Black South Africans [12]. Possessing two APOL1 risk alleles results in up to a 50% lifetime risk in those with HIVAN [13]. However, 20 to 30% of African Americans with HIVAN have 0 to 1 APOL1 risk alleles, suggesting the role of other environmental and viral factors or other genetic contributions [14]. Differentiating hypertensive nephrosclerosis from other forms of CKD may be difficult, and further research is required linking APOL1 variants to the high prevalence of hypertension-associated CKD.
There are also evolving targeted therapies, these include APOL1 small molecule inhibitors, APOL1 antisense oligonucleotides (ASO), and inhibitors of APOL1-associated inflammatory pathways [15]. The novel small molecule APOL1 inhibitors bind recombinant APOL1 proteins, thereby inhibiting its biological function, providing the potential to treat APOL-1 mediated kidney diseases.
4. Diabetic Kidney Disease
Approximately 40% of people with type 2 diabetes (T2D) will develop diabetic kidney disease (DKD), accounting for approximately 50% of cases of end-stage kidney disease (ESKD) in the developed world [16,17]. Furthermore, CV disease is the leading cause of death [18]. However, epidemiological data from SSA regarding CKD in people with diabetes is extremely limited and, at best, of low-to-medium quality.
In a systematic review conducted in Africa, the prevalence of CKD in people with type 1 and type 2 diabetes varied from 11% to 83.7% [19]. Incident event rates were 94.9% for proteinuria at 10 years of follow-up, 34.7% for ESKD at 5 years of follow-up and 18.4% for mortality from nephropathy at 20 years. Duration of diabetes, blood pressure (BP), advancing age, obesity, and glucose control were the common determinants of kidney disease. These figures strongly suggest a far greater incidence of DKD in Africa and higher incidence of morbidity and mortality.
There is also a significant interaction with HIV, which is common, particularly in Southern Africa. In 2019, a retrospective study of 653 patients with diabetes from KwaZulu-Natal found a significant interaction with HIV on the prevalence of CKD [20]. Nephropathy, defined by proteinuria, was present in 25.66% vs. 15.43% of the HIV infected vs. non-infected patients, respectively. This finding was confirmed by a systematic review and meta-analysis conducted by Ekrikpo et al. [21].
Most guidelines have advocated a multi-faceted approach to prevent DKD, entailing strict BP and diabetic control, the intensive treatment of dyslipidaemia, the use of ACE inhibitors/angiotensin receptor blockers (ARBs), aspirin, and lifestyle intervention. This was based on the STENO-2 study that was associated with a 20% reduction in death and 47% reduction in nephropathy in the intensively treated group compared to standard treatment [22].
However, this approach has changed dramatically with the publication of clinical trials involving SGLT2 inhibitors. A full review of this is beyond the scope of this article, but, briefly, there was a striking reduction in CV death, heart failure, and “hard” kidney outcomes, including kidney failure, in a trial involving patients with diabetes with underlying atherosclerotic CV disease [23]. Several other studies reported similar results [24,25]. Four dedicated CKD studies (two of which included patients without underlying diabetes) [26,27,28,29] reported a consistent reduction in hard kidney end points, heart failure, and CV death, even in those without underling diabetes. In a recent meta-analysis of 13 trials involving 90,409 participants (82.7% with diabetes and 17.3% without diabetes), allocation to an SGLT2 inhibitor reduced the risk of progressive kidney disease by 37% with no difference in benefits in patients with and without diabetes [30]. In the four CKD trials, risk reductions were similar, irrespective of primary CKD diagnosis.
It is evident from the systematic reviews that the greatest benefit is in those with eGFR < 60 mL/min with albuminuria and/or underlying CV disease and most major guidelines recommend SGLT2 inhibitors for treatment and prevention of DKD [31]. However, the problem with all these major studies were that only 4–5% of patients entering these studies were identified as Black. In an analysis of the outcomes across geographic regions, there were consistent reductions in the composite endpoint of 50% reduction in eGFR, ESKD, death related to kidney failure, or CV cause. The reduction in hazard ratio ranged from 0.70 in Asia, 0.6 in Europe, 0.61 in Latin America, and 0.51 in North America [32]. There were no data from SSA, but the available data are reassuring that Africans are likely to benefit from SGLT2 inhibitors.
In regard to GLP-1 receptor agonists, there has been no dedicated CKD study. However, eight CV outcomes trials included participants with an eGFR as low as 15 mL/min/1.73 m2. In a meta-analysis of these studies, GLP-1 receptor agonists significantly reduced the risk for a composite CKD outcome, mainly driven by the reduction in albuminuria [33]. As a result, the consensus report by the American Diabetes Association (ADA) and Kidney Disease Improving Global Outcomes (KDIGO) only recommends GLP-1 receptors agonists for additional risk-based management and glycaemic control in patients with CKD already treated with metformin and SGLT2 inhibitors [34].
These very important advances in the treatment and prevention of DKD are difficult to translate into SSA. Currently, there are no generic formulations of these drugs, and they remain expensive, particularly the GLP-1 receptor agonists. It, therefore, remains unlikely that most people with DKD in SSA are likely to benefit from these drugs, due to affordability and availability.
5. Hypertension and CKD
In 2019, high systolic BP was the leading risk factor for DALYs worldwide, accounting for 9.3% of all DALYs [35]. In Africa, the proportion of people affected by hypertension is estimated to be between 15 and 70% [36], and the estimated direct costs of hypertension from a South African study amount to USD 0.711 billion, with societal costs of USD 2.08 billion [37]. However, an Ethiopian study reported that treating hypertension according to the International Society of Hypertension 2020 guidelines could result in USD 9574 million in savings [38]. The cost saving is due to the decrease in hypertensive mediated organ damage, including CKD. This highlights the need for ensuring optimal blood pressure control in SSA. The CREOLE trial, performed in SSA, demonstrated excellent blood pressure control from initiating antihypertensives with the combination of a calcium channel blocker in combination with a thiazide diuretic or ACE inhibitor and is the preferred choice of initial treatment of Africans [38,39]. However, access to optimal therapies is lacking, particularly the shortage of single pill combinations.
A systematic review of community-based data shows that the prevalence of CKD in SSA ranges from 2–14% and, in patients with hypertension from Africa, CKD was present in 13–51%. Vascular disease/hypertension was the most common (16%) cause of CKD, followed by diabetes, glomerulonephritis, and other causes [4]. The presence of severe hypertension was associated with a higher risk of complications. Data from a community-based study (including both rural and urban sites) from the Cote d’Ivoire found that severe hypertension was present in 14.1% of screened subjects and was almost double in urbanised regions [40]. In the setting of non-diabetic-CKD, hypertensive retinopathy was detected in 70.1% of patients from a Tanzanian tertiary centre, with grade III at 19.6% and grade IV at 13.8%, suggesting that severe/malignant hypertension is present in ±33% of people with stages 3-5 CKD [41]. Whether severe hypertension or malignant hypertension is the cause or a result of CKD remains to be determined, but it is of concern that this is a common feature detected in the region.
Modifiable risk factors for CKD are a rising concern in Africa. The Global Burden of Disease report from 2019 demonstrates the rising CV and CKD risk factors, particularly high body mass index, high systolic blood pressure, and elevated plasma glucose [34]. A community-based screening study in Nigeria identified 32.3% of subjects with prehypertension and 43.4% with hypertension. The identified modifiable risk factors included diabetes, cigarette smoking, and the use of non-steroidal anti-inflammatories and herbal substances [42]. Similarly, in other community-based studies, hypertension was found to be the most common risk factor for CKD, with 21.8% in Ethiopia and 55.4% in Ghana [43,44]. Obesity is an independent risk factor for CKD and was identified in 21.4% of participants in the Ghana report. However, screening for CKD in patients with pre-existing risk factors provides a higher yield of CKD diagnoses and is a better indicator of the need to change treatment when compared with population-based screening for CKD [45].
The development of CKD needs to be considered as a lifelong risk and determining the presence of risk factors in childhood is essential for prevention programmes and CV risk tracking. A report from Ghana found that hypertension was present in 3.5% of school children (average age 8.9 years) and obesity in 9.8%, similar to other countries [46]. Similarly, in South Africa, obesity/overweight was identified in 20% of supposedly healthy 5–9 year olds, pre-hypertension in 14%, and hypertension in 22% [47]. In this report, adiposity and elevated BP was more common in the White and Black ethnic groups, respectively. Salt intake is a further concern that has been found to contribute to hypertension and CKD. It was estimated that sodium intake contributed to 1.5% of deaths in 2012 and 0.7% of DALYs in South Africa [48]. Salt legislation has, subsequently, been introduced and found to lower sodium intake in South Africa [49].
6. Pregnancy Related AKI and Its Causes—A Growing Concern of CKD in Africa
Pregnancy-related acute kidney injury (PRAKI) is defined as AKI occurring during pregnancy, labour, delivery, and/or the post-partum period. The reported incidence of PRAKI in developing nations is higher than developed countries (4–26% vs. 1–2.8%, respectively) [50,51,52]. PRAKI in Africa is estimated to occur in 1 in 1000 deliveries [53]. It also accounts for a significant proportion of AKI in adults in Africa (5–27%) [53]. This is 20–100-fold higher than in developed countries. [54]. In low–low-middle income countries, pre-eclampsia [PET], post-partum haemorrhage, and sepsis are the leading causes of PRAKI [50,51,52]. Severe or repeated episodes of AKI are risk factors for CKD, and PRAKI is increasingly recognized as a risk for developing future CKD in women. This progression to CKD adds an additional load onto the substantial health care burden in African countries.
Kidney recovery after PRAKI is poorly described in literature from Africa. A systematic review and meta-analysis on PRAKI in SSA found only 9 of the 14 studies reported on kidney recovery data [53]. Non recovery ranged from 0.4–36.5%. Table 1 demonstrates the data from Africa with kidney recovery data following PRAKI in different cohorts [51,55,56,57,58,59,60,61,62,63,64,65,66,67,68]. Full recovery ranged from 39–84%, and dialysis dependence ranged from 0.4–35% of those not recovering. Literature from other developing countries reported that 1.5–2.5% of patients with PRAKI progressed to ESKD, and 4–9% of women remain dialysis-dependent at 6 months post-partum in severe PRAKI [69,70,71,72,73]. Studies have demonstrated that the risk for CKD progressively increases with the severity of AKI. The adjusted HR was 2.0 for mild AKI and increased to 28.2 for severe AKI [74]. An additional concern is the high rate of loss to follow up in post-partum women. This raises major concerns for (1) gaps in the health care system, (2) lack of awareness of the need for follow-up after AKI, and (3) the known rates of incomplete recovery post PET are likely being vastly underestimated. South Africa reported a high risk of PRAKI (17%) in women with preeclampsia. Of those women that survived, 1/3 had not recovered kidney function at discharge, and half did not have a follow-up kidney function performed [65].

Table 1.
Studies related to PRAKI in Obstetric cohorts or referral to Nephrology.
7. Pre-Eclampsia as a Future Risk for CKD
The last decade has demonstrated that the long-term health consequences resulting from pre-eclampsia are marked. These included sustained hypertension (three-fold increased risk), two-fold increased risk of CV disease and stroke, increased risk of metabolic syndrome, and, more recently, studies showed a 6× increased risk of ESKD [75,76,77,78]. The risk of progression to CKD or ESKD is not due to the PRAKI alone. The aetiology is thought to be an interplay between endotheliosis and podocyte damage during PET, followed by the future development of co-morbidities, particularly diabetes, hypertension, metabolic syndrome, and, possibly, persistent endothelial dysfunction [79]. This is an important contributing risk for CKD. The World Health Organisation estimates the incidence of PET in developing countries is seven times higher than in developed countries [80]. Hypertensive disorders in pregnancy (HDP) are the second leading maternal cause of death in SSA [81].
8. Newer Findings into the Pathogenesis of PET
Advances in research of genetic and inflammatory system involvement in PET in the African setting has increased in recent years. The inflammatory system’s role in the pathogenesis of PET has been studied. Tumour necrosis factor alpha (TNF-α) polymorphisms, altering the regulatory function and leading to increased transcription and production, has been associated with PET. Recent research suggests polymorphisms, particularly 1031C/A and the haplotype CA, contribute to an increased risk of PET in Tunisian women [82]. Other TNF-α SNPs, mostly the −308 G/A, have been studied in different ethnic cohorts (European and Asian) with varying results [83,84]. Research into genetic predispositions for pre-eclampsia is being studied. APOL1 G1 risk allele in pregnant women of African origin may contribute to the development of PET and early-onset PET [85]. International data have shown that maternal–foetal APOL1 genotype discordance and foetal high-risk APOL1 genotypes independently contribute to PET in African American mothers [86]. In a study reviewing the association of the infant APOL1 genotype and PET, it was found that the infant APOL1 genotype was significantly associated with preeclampsia in a dominant inheritance pattern with an odds ratio of 1.41 (p = 0.029, 95% CI 1.037, 1.926) [87]. In patients of African origin, the R536Q gene mutation is found in a higher proportion of women with pre-eclampsia versus the controls [88]. This mutation alters the beta subunit of the epithelial sodium channel and is a genetic cause for hypertension in Africans. By understanding the association of these genetic variants and inflammatory components with PET, the development of future useful screening tests may assist in predicting at-risk pregnancies and targeted interventions to improve pregnancy outcomes.
9. What Is New in HIV and CKD in SSA
In 2020, human immunodeficiency virus (HIV) affected 37.7 million people globally, with 1.5 million new cases during that year. Eastern and SSA had the highest disease burden, with SSA accounting for almost 60% of new infections [89,90]. In a systematic review and meta-analysis of CKD in an adult cohort with HIV, the overall global prevalence was 6.4%, with the highest prevalence in SSA (7.9%) [21]. The improved longevity on ART has extended the causes of ESKD by increasing the prevalence of traditional CKD risk factors such as diabetes and hypertension [91].
With improved HIV viral suppression, the survival of people with HIV (PWH) receiving kidney replacement therapy (KRT) has been described as being comparable to HIV-negative cohorts, although there have been some conflicting data [92,93]. In Cameroon, the one-year survival of PWH on maintenance haemodialysis was reported to be lower compared to their HIV negative counterparts, with a two-fold increased risk of death [94]. Registry data throughout SSA are scarce; however, South Africa reported that 13.5% of patients on chronic KRT were PWH in 2019 [95]. This is an underestimate of the true prevalence, as unsuppressed HIV is often an exclusion criterion for maintenance dialysis in the state sector, due to imposed resource constraints.
There have now been several studies reporting on a change in the spectrum of kidney disease in PWH since the ART era [96,97,98]. As early as 1998, HIVAN was reported to decline in regions with early ART initiatives [99]. South Africa lagged woefully behind, with large-scale roll-out only occurring from 2004. Since then, published biopsy data from South Africa reported a decline in HIVAN, which corresponds with an increase in the proportion of PWH receiving ART at the time of biopsy and an increase in median CD4 cell count [96,97,100]. Two recent biopsy series from South Africa also reported an increase in the proportion of biopsies with tubulointerstitial disease [97,101]. This increase was likely influenced by HIV/TB coinfection, sepsis, and tenofovir exposure, as well as other insults.
TB is the most common opportunistic infection seen in PWH. It can affect the kidney in two major ways: firstly, by causing genitourinary TB, and, secondly, in the form of granulomatous interstitial nephritis (GIN). Unfortunately, the COVID-19 pandemic displaced TB improvement targets globally, with an increase in TB deaths [102]. In SSA, the overall estimate of HIV prevalence in TB patients was 31.8% [103]. Autopsy series describing TB involvement in the kidney of PWH in the pre-ART from India, Mexico, and the Cote d’Ivoire reported a frequency of 59%, 23%, and 60%, respectively [104,105,106].
ART has also seen the additional challenge of TB immune reconstitution inflammatory syndrome (IRIS) in the kidney in the form of TB-GIN and acute interstitial nephritis [107]. ART re-establishes the host’s ability to form granulomata, often resulting in an intense “paradoxical” reaction [108]. The role of the kidney biopsy is particularly important in this cohort due to the multiple pathologies that often coexist providing a role in assisting management decisions.
TAF is the next generation tenofovir (TDF) prodrug whose composition has improved stability and allows superior penetrance into cells. Tenofovir diphosphate (TFV-DP) is the active intracellular metabolite for TDF and TAF [109]. Compared with 300 mg TDF, TAF 25 mg showed more potent antiviral activity, higher peripheral blood mononuclear cell (PBMC) intracellular TFV-DP concentrations, and lower plasma tenofovir exposures. This study also demonstrated less proximal tubular injury and a smaller decrease in eGFR compared with TDF [110].
Dolutegravir (DTG) is a pivotal ART that has become the backbone of several HIV programmes, especially in SSA. It inhibits the organic cation transporter 2, thereby inducing an increase in serum creatinine of up to 0.4 mg/dL (44 mmol/L) and a drop in eGFR of 10–15 mL/min per 1.73 m2 without altering the actual GFR [111,112].
Most regions in SSA have no formal transplantation programs or, if available, are focused on living-related donation, with the operation occurring outside of their own country. PWH were initially not enrolled into transplantation programmes due to the perceived poor outcomes [113]. There is now robust evidence supporting positive outcomes in both HIV-negative donors to HIV-positive recipients and positive donors to positive recipients. South Africa reported on 15 years of experience of HIV-positive donors to HIV-positive recipients. The 5-year overall survival and graft survival of 83.3% and 78.7%, respectively, are similar to the 3-year overall survival [114] and graft survival observed among HIV-positive patients who received an organ from an HIV-negative donor in the United States (88.2% and 73.7%, respectively) [115]. However, there is the additional complexity of drug interactions. Managing ART and immunosuppression can make drug level monitoring challenging. There is also increased risk of rejection, similar to that described in the HIV-negative to HIV positive cohort. The South African cohort recently described a concerning signal for increased rejection rates in those on a protease-inhibitor-based regimen [116].
A heterogeneous spectrum of immune complex-mediated glomerular diseases has been reported in PWH [117,118,119]. These include mesangiocapillary GN (MCGN), membranous GN, post-infectious GN, “lupus-like GN”, and IgA nephropathy. The pathogenesis and natural history of these GNs is not currently clarified. Key questions remain including the role of HIV infection in the pathogenesis, the efficacy of ART in the prevention and treatment, and the difference in the outcomes between those without HIV. A study from Cape Town aimed to determine the kidney outcomes of MCGN in PWH, and, without HIV, there was no difference in the composite outcome of either doubling of the serum creatinine or ESKD between PWH and HIV negative patients 45.0% versus 25.4%, respectively; p = 0.10 [120]. There was also no difference in mortality between the groups. Due to late presentation, kidney outcomes remained poor, regardless of the HIV status.
Due to the increased genetic, infection, and drug risks for kidney disease in PWH, the monitoring of kidney function is essential. This is recommended at the ART initiation and when modifying ART, as well as yearly after that. However, those high-risk groups, including those of the Black ethnicity, CD4 cell counts < 200 cells/L, viral loads > 4000 copies/mL, or those with additional comorbid conditions such as diabetes mellitus, hypertension, or hepatitis C virus coinfection, require more frequent monitoring. Due to the changing spectrum of kidney disease since the ART rollout, additional attention needs to be given to traditional risk factors of CKD, ART, and opportunistic infections, particularly TB in high-risk regions. The overall positive outcomes of PWH with ESKD should not influence candidacy for KRT.
10. CKD Unknown (CKDu) in Africa
While hypertension, diabetes, GN, and HIV are considered the major contributors to CKD globally and in Africa, there is a large proportion of people with CKD who do not have a clear cause for their kidney dysfunction, now termed CKD unknown (CKDu). In a South African rural population, CKDu was found in 32% of those surveyed [121]. A scoping review reports CKDu in Cameroon was detected in 13.5–17% of cases [122]. However, 3.4% of sugarcane plantation workers had CKD, and CKD was detected in 15.9% of first-degree relatives of people with CKD. Herbal medication and self-medication increased the odds of having CKD.
A common theme for those detected with CKDu is employment in the agricultural sector, such as sugarcane plantations. This raises the hypothesis that heat stress, dehydration, strenuous labour, and the exposure to agricultural chemicals could be contributing to CKD in these people. Other theories include exposure to heavy metals, excess trace elements and pollutants in water or agricultural products [123]. In support of some of these theories, data from Africa have demonstrated that heat may contribute to CKD in sugarcane plantation workers [124], crop farming with hexachlorocyclohexane [125], and chemical exposure in cassava farmers where the use of unsafe farming practices increased the risk [126]. Contrary to the findings from other African countries, a study in Malawi reported a very low prevalence of CKDu (5%), with no added risk in those from a rural setting or from agricultural work [127].
In Africa, it is essential to consider infectious diseases as potential contributing factors to the development of CKD. HIV is well described, but other infectious diseases need to be considered. A study from Tanzania demonstrated that a history of TB or schistosomiasis was associated with albuminuria, and prior TB was associated with impaired kidney function, aside from the usual causes [128]. In Uganda, a study in children with complicated malaria (cerebral malaria or with severe anaemia) found that AKI was a risk factor for CKD at 1 year. Severe AKI was associated with a four-fold increased risk of CKD [129]. AKI may be underestimated as a contributing factor to CKDu if recurrent episodes are undetected, particularly in non-hospitalised infectious diseases and agricultural workers [129].
Genetics may also contribute to CKDu in Africa [130]. APOL1 is well described, but other genetic variants may be implicated, such as polymorphisms of the ectonucleotide pyrophosphatase/phosphodiesterase 1 [131], transforming growth factor beta 1 (particularly in HIV) [132], heme oxygenase, glycine amidinotransferase, and haemoglobin beta [133].
11. Conclusions
Africa, particularly SSA, faces a major challenge in respect to CKD. There is a rising prevalence due to the combined effects of hypertension, diabetes, HIV (and interaction between them), and the effect of APOL1 variants on the susceptibility to CKD. Epidemiological data on the prevalence of CKD is of low-to-medium quality, and reliable data are urgently needed. Furthermore, there are important deficiencies in creatinine-based equations in estimating the prevalence of CKD. The review highlights several important advances and understanding of CKD in Africa in terms of hypertension, diabetes, HIV, CKDu, and the long-term effects of hypertension in pregnancy and PRAKI.
Author Contributions
All authors contributed to the writing of the original draft preparation, reviewing and editing. All authors have read and agreed to the published version of the manuscript.
Funding
The research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
No original data was used in publishing this article.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Available online: https://ourworldindata.org/causes-of-death (accessed on 15 February 2023).
- Bikbov, B.; Purcell, C.A.; Levey, A.S.; Smith, M.; Abdoli, A.; Abebe, M.; Adebayo, O.M.; Afarideh, M.; Agarwal, S.K.; GBD Chronic Kidney Disease Collaboration; et al. Global, regional, and national burden of chronic kidney disease, 1990–2017: A systematic analysis for the Global Burden of Disease Study 2017. Lancet 2020, 395, 709–733. [Google Scholar] [CrossRef] [PubMed]
- Xie, Y.; Bowe, B.; Mokdad, A.H.; Xian, H.; Yan, Y.; Li, T.; Maddukuri, G.; Tsai, C.-Y.; Floyd, T.; Al-Aly, Z. Analysis of the Global Burden of Disease study highlights the global, regional, and national trends of chronic kidney disease epidemiology from 1990 to 2016. Kidney Int. 2018, 94, 567–581. [Google Scholar] [CrossRef] [PubMed]
- Elhafeez, S.A.; Bolignano, D.; D’Arrigo, G.; Dounousi, E.; Tripepi, G.; Zoccali, C. Prevalence and burden of chronic kidney disease among the general population and high-risk groups in Africa: A systematic review. BMJ Open 2018, 8, e015069. [Google Scholar] [CrossRef] [PubMed]
- Atta, M.G.; Zook, K.; Brown, T.T.; Vaidya, D.; Tao, X.; Maier, P.; Schwartz, G.J.; Lucas, G.M. Racial Adjustment Adversely Affects Glomerular Filtration Estimates in Black Americans Living with HIV. J. Am. Soc. Nephrol. 2021, 32, 2143–2147. [Google Scholar] [CrossRef] [PubMed]
- Inker, L.A.; Eneanya, N.D.; Coresh, J.; Tighiouart, H.; Wang, D.; Sang, Y.; Crews, D.C.; Doria, A.; Estrella, M.M.; Froissart, M.; et al. New Creatinine- and Cystatin C–Based Equations to Estimate GFR without Race. N. Engl. J. Med. 2021, 385, 1737–1749. [Google Scholar] [CrossRef]
- Fabian, J.; Kalyesubula, R.; Mkandawire, J.; Hansen, C.H.; Nitsch, D.; Musenge, E.; Nakanga, W.P.; E Prynn, J.; Dreyer, G.; Snyman, T.; et al. Measurement of kidney function in Malawi, South Africa, and Uganda: A multicentre cohort study. Lancet Glob. Health 2022, 10, e1159–e1169. [Google Scholar] [CrossRef]
- Genovese, G.; Friedman, D.J.; Ross, M.D.; Lecordier, L.; Uzureau, P.; Freedman, B.I.; Bowden, D.W.; Langefeld, C.D.; Oleksyk, T.K.; Knob, A.L.U.; et al. Association of Trypanolytic ApoL1 Variants with Kidney Disease in African Americans. Science 2010, 329, 841–845. [Google Scholar] [CrossRef]
- Hung, R.K.; Binns-Roemer, E.; Booth, J.W.; Hilton, R.; Harber, M.; Santana-Suarez, B.; Campbell, L.; Fox, J.; Ustianowski, A.; Cosgrove, C.; et al. Genetic Variants of APOL1 Are Major Determinants of Kidney Failure in People of African Ancestry With HIV. Kidney Int. Rep. 2022, 7, 786–796. [Google Scholar] [CrossRef]
- Limou, S.; Nelson, G.W.; Kopp, J.B.; Winkler, C.A. APOL1 Kidney Risk Alleles: Population Genetics and Disease Associations. Adv. Chronic Kidney Dis. 2014, 21, 426–433. [Google Scholar] [CrossRef]
- Genovese, G.; Friedman, D.J.; Pollak, M.R. APOL1 variants and kidney disease in people of recent African ancestry. Nat. Rev. Nephrol. 2013, 9, 240–244. [Google Scholar] [CrossRef]
- Kasembeli, A.N.; Duarte, R.; Ramsay, M.; Mosiane, P.; Dickens, C.; Dix-Peek, T.; Limou, S.; Sezgin, E.; Nelson, G.W.; Fogo, A.B.; et al. APOL1 Risk Variants Are Strongly Associated with HIV-Associated Nephropathy in Black South Africans. J. Am. Soc. Nephrol. 2015, 26, 2882–2890. [Google Scholar] [CrossRef] [PubMed]
- Dummer, P.D.; Limou, S.; Rosenberg, A.Z.; Heymann, J.; Nelson, G.; Winkler, C.A.; Kopp, J.B. APOL1 Kidney Disease Risk Variants: An Evolving Landscape. Semin. Nephrol. 2015, 35, 222–236. [Google Scholar] [CrossRef] [PubMed]
- Atta, M.G.; Estrella, M.M.; Kuperman, M.; Foy, M.C.; Fine, D.M.; Racusen, L.C.; Lucas, G.M.; Nelson, G.W.; Warner, A.C.; Winkler, C.A.; et al. HIV-associated nephropathy patients with and without apolipoprotein L1 gene variants have similar clinical and pathological characteristics. Kidney Int. 2012, 82, 338–343. [Google Scholar] [CrossRef] [PubMed]
- Friedman, D.J.; Ma, L.; Freedman, B.I. Treatment potential in APOL1-associated nephropathy. Curr. Opin. Nephrol. Hypertens. 2022, 31, 442–448. [Google Scholar] [CrossRef]
- Alicic, R.Z.; Rooney, M.T.; Tuttle, K.R. Diabetic Kidney Disease: Challenges, Progress, and Possibilities. Clin. J. Am. Soc. Nephrol. 2017, 12, 2032–2045. [Google Scholar] [CrossRef]
- Tuttle, K.R.; Bakris, G.L.; Bilous, R.W.; Chiang, J.L.; de Boer, I.H.; Goldstein-Fuchs, J.; Hirsch, I.B.; Kalantar-Zadeh, K.; Narva, A.S.; Navaneethan, S.D.; et al. Diabetic Kidney Disease: A Report From an ADA Consensus Conference. Diabetes Care 2014, 37, 2864–2883. [Google Scholar] [CrossRef]
- Pálsson, R.; Patel, U.D. Cardiovascular Complications of Diabetic Kidney Disease. Adv. Chronic Kidney Dis. 2014, 21, 273–280. [Google Scholar] [CrossRef]
- Noubiap, J.J.N. Diabetic nephropathy in Africa: A systematic review. World J. Diabetes 2015, 6, 759–773. [Google Scholar] [CrossRef]
- Pillay, S.; Aldous, C.; Mahomed, F. A deadly combination—HIV and diabetes mellitus: Where are we now? South Afr. Med. J. 2016, 106, 378. [Google Scholar] [CrossRef]
- Ekrikpo, U.E.; Kengne, A.P.; Bello, A.K.; Effa, E.E.; Noubiap, J.J.; Salako, B.L.; Rayner, B.L.; Remuzzi, G.; Okpechi, I.G. Chronic kidney disease in the global adult HIV-infected population: A systematic review and meta-analysis. PLoS ONE 2018, 13, e0195443. [Google Scholar] [CrossRef]
- Gæde, P.; Lund-Andersen, H.; Parving, H.-H.; Pedersen, O. Effect of a Multifactorial Intervention on Mortality in Type 2 Diabetes. N. Engl. J. Med. 2008, 358, 580–591. [Google Scholar] [CrossRef]
- Zinman, B.; Wanner, C.; Lachin, J.M.; Fitchett, D.; Bluhmki, E.; Hantel, S.; Mattheus, M.; Devins, T.; Johansen, O.E.; Woerle, H.J.; et al. Empagliflozin, Cardiovascular Outcomes, and Mortality in Type 2 Diabetes. N. Engl. J. Med. 2015, 373, 2117–2128. [Google Scholar] [CrossRef] [PubMed]
- Neal, B.; Perkovic, V.; Mahaffey, K.W.; de Zeeuw, D.; Fulcher, G.; Erondu, N.; Shaw, W.; Law, G.; Desai, M.; Matthews, D.R.; et al. Canagliflozin and Cardiovascular and Renal Events in Type 2 Diabetes. N. Engl. J. Med. 2017, 377, 644–657. [Google Scholar] [CrossRef]
- Wiviott, S.D.; Raz, I.; Bonaca, M.P.; Mosenzon, O.; Kato, E.T.; Cahn, A.; Silverman, M.G.; Zelniker, T.A.; Kuder, J.F.; Murphy, S.A.; et al. Dapagliflozin and Cardiovascular Outcomes in Type 2 Diabetes. N. Engl. J. Med. 2019, 380, 347–357. [Google Scholar] [CrossRef]
- Perkovic, V.; Jardine, M.J.; Neal, B.; Bompoint, S.; Heerspink, H.J.L.; Charytan, D.M.; Edwards, R.; Agarwal, R.; Bakris, G.; Bull, S.; et al. Canagliflozin and Renal Outcomes in Type 2 Diabetes and Nephropathy. N. Engl. J. Med. 2019, 380, 2295–2306. [Google Scholar] [CrossRef] [PubMed]
- Heerspink, H.J.L.; Stefánsson, B.V.; Correa-Rotter, R.; Chertow, G.M.; Greene, T.; Hou, F.-F.; Mann, J.F.E.; McMurray, J.J.V.; Lindberg, M.; Rossing, P.; et al. Dapagliflozin in Patients with Chronic Kidney Disease. N. Engl. J. Med. 2020, 383, 1436–1446. [Google Scholar] [CrossRef] [PubMed]
- The EMPA-KIDNEY Collaborative Group; Herrington, W.G.; Staplin, N.; Wanner, C.; Green, J.B.; Hauske, S.J.; Emberson, J.R.; Preiss, D.; Judge, P.; Mayne, K.J.; et al. Empagliflozin in Patients with Chronic Kidney Disease. N. Engl. J. Med. 2023, 388, 117–127. [Google Scholar] [CrossRef]
- Bhatt, D.L.; Szarek, M.; Pitt, B.; Cannon, C.P.; Leiter, L.A.; McGuire, D.K.; Lewis, J.B.; Riddle, M.C.; Inzucchi, S.E.; Kosiborod, M.N.; et al. Sotagliflozin in Patients with Diabetes and Chronic Kidney Disease. N. Engl. J. Med. 2021, 384, 129–139. [Google Scholar] [CrossRef]
- Baigent, C.; Emberson, J.; Haynes, R.; Herrington, W.G.; Judge, P.; Landray, M.J.; Mayne, K.J.; Ng, S.Y.; Preiss, D.; Roddick, A.J.; et al. Impact of diabetes on the effects of sodium glucose co-transporter-2 inhibitors on kidney outcomes: Collaborative meta-analysis of large placebo-controlled trials. Lancet 2022, 400, 1788–1801. [Google Scholar] [CrossRef]
- de Boer, I.H.; Caramori, M.L.; Chan, J.C.; Heerspink, H.J.; Hurst, C.; Khunti, K.; Liew, A.; Michos, E.D.; Navaneethan, S.D.; Kidney Disease: Improving Global Outcomes (KDIGO) Diabetes Work Group; et al. KDIGO 2020 Clinical Practice Guideline for Diabetes Management in Chronic Kidney Disease. Kidney Int. 2020, 98, S1–S115. [Google Scholar] [CrossRef]
- Vart, P.; Correa-Rotter, R.; Hou, F.F.; Jongs, N.; Chertow, G.M.; Langkilde, A.M.; McMurray, J.J.; Rossing, P.; Sjöström, C.D.; Stefansson, B.V.; et al. Efficacy and Safety of Dapagliflozin in Patients With CKD across Major Geographic Regions. Kidney Int. Rep. 2022, 7, 699–707. [Google Scholar] [CrossRef] [PubMed]
- Sattar, N.; Lee, M.M.Y.; Kristensen, S.L.; Branch, K.R.H.; Del Prato, S.; Khurmi, N.S.; Lam, C.S.P.; Lopes, R.D.; McMurray, J.J.V.; Pratley, R.E.; et al. Cardiovascular, mortality, and kidney outcomes with GLP-1 receptor agonists in patients with type 2 diabetes: A systematic review and meta-analysis of randomised trials. Lancet Diabetes Endocrinol. 2021, 9, 653–662. [Google Scholar] [CrossRef]
- de Boer, I.H.; Khunti, K.; Sadusky, T.; Tuttle, K.R.; Neumiller, J.J.; Rhee, C.M.; Rosas, S.E.; Rossing, P.; Bakris, G. Diabetes management in chronic kidney disease: A consensus report by the American Diabetes Association (ADA) and Kidney Disease: Improving Global Outcomes (KDIGO). Kidney Int. 2022, 102, 974–989. [Google Scholar] [CrossRef] [PubMed]
- Murray, C.J.L.; Abbafati, C.; Abbas, K.M.; Abbasi, M.; Abbasi-Kangevari, M.; Abd-Allah, F.; Abdollahi, M.; Abedi, P.; Abedi, A.; GBD 2019 Viewpoint Collaborators; et al. Five insights from the Global Burden of Disease Study 2019. Lancet 2020, 396, 1135–1159. [Google Scholar] [CrossRef]
- Ataklte, F.; Erqou, S.; Kaptoge, S.; Taye, B.; Echouffo-Tcheugui, J.B.; Kengne, A.P. Burden of Undiagnosed Hypertension in Sub-Saharan Africa: A systematic review and meta-analysis. Hypertension 2015, 65, 291–298. [Google Scholar] [CrossRef]
- Kohli-Lynch, C.N.; Erzse, A.; Rayner, B.; Hofman, K.J. Hypertension in the South African public healthcare system: A cost-of-illness and burden of disease study. BMJ Open 2022, 12, e055621. [Google Scholar] [CrossRef]
- Davari, M.; Sorato, M.M.; Kebriaeezadeh, A.; Sarrafzadegan, N. Cost-effectiveness of hypertension therapy based on 2020 International Society of Hypertension guidelines in Ethiopia from a societal perspective. PLoS ONE 2022, 17, e0273439. [Google Scholar] [CrossRef] [PubMed]
- Comparison of Dual Therapies for Lowering Blood Pressure in Black Africans; Ojji, D.B.; Mayosi, B.; Francis, V.; Badri, M.; Cornelius, V.; Smythe, W.; Kramer, N.; Barasa, F.; Damasceno, A.; et al. Comparison of Dual Therapies for Lowering Blood Pressure in Black Africans. N. Engl. J. Med. 2019, 380, 2429–2439. [Google Scholar] [CrossRef]
- Gaye, B.; Janeczek, A.-L.; Narayanan, K.; N’Guetta, R.; Vignac, M.; Gallardo, V.; Jouven, X.; Luu, D.; Marijon, E. Prevalence of severe hypertension in a Sub-Saharan African community. Int. J. Cardiol. Hypertens. 2019, 2, 100016. [Google Scholar] [CrossRef]
- Chillo, P.; Ismail, A.; Sanyiwa, A.; Ruggajo, P.; Kamuhabwa, A. Hypertensive retinopathy and associated factors among nondiabetic chronic kidney disease patients seen at a tertiary hospital in Tanzania: A cross-sectional study. Int. J. Nephrol. Renov. Dis. 2019, 12, 79–86. [Google Scholar] [CrossRef]
- Akinbodewa, A.A.; Adejumo, A.O.; Koledoye, O.V.; Kolawole, J.O.; Akinfaderin, D.; Lamidi, A.O.; Gbakinro, G.O.; Ogunduyile, C.; Osungbemiro, W.B. Community screening for pre-hypertension, traditional risk factors and markers of chronic kidney disease in Ondo State, South-Western Nigeria. Niger. Postgrad. Med. J. 2017, 24, 25–30. [Google Scholar] [CrossRef] [PubMed]
- Kebede, K.M.; Abateneh, D.D.; Teferi, M.B.; Asres, A. Chronic kidney disease and associated factors among adult population in Southwest Ethiopia. PLoS ONE 2022, 17, e0264611. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Owusu, I.K.; Geng, Q.; Folson, A.A.; Zheng, Z.; Adu-Boakye, Y.; Dong, X.; Wu, W.; Agyekum, F.; Fei, H.; et al. Cardiometabolic Risk Factors and Preclinical Target Organ Damage among Adults in Ghana: Findings from a National Study. J. Am. Heart Assoc. 2020, 9, e017492. [Google Scholar] [CrossRef] [PubMed]
- Tonelli, M.; Tiv, S.; Anand, S.; Mohan, D.; Garcia, G.G.; Padilla, J.A.G.; Klarenbach, S.; Blackaller, G.N.; Seck, S.; Wang, J.; et al. Diagnostic Yield of Population-Based Screening for Chronic Kidney Disease in Low-Income, Middle-Income, and High-Income Countries. JAMA Netw. Open 2021, 4, e2127396. [Google Scholar] [CrossRef] [PubMed]
- Ajaegbu, O.C.; Ezeonwu, B.U.; Emeagui, O.D.; Okafor, H.U. Modifiable Risk Factors for Chronic Kidney Disease in Adulthood seen among School Children in Asaba. West Afr. J. Med. 2021, 38, 674–678. [Google Scholar]
- Kruger, R.; Kruger, H.S.; Monyeki, M.A.; Pienaar, A.E.; Roux, S.B.-L.; Gafane-Matemane, L.F.; Smith, W.; Mels, C.M.C.; Lammertyn, L.; Brits, J.S.; et al. A demographic approach to assess elevated blood pressure and obesity in prepubescent children: The ExAMIN Youth South Africa study. J. Hypertens. 2021, 39, 2190–2199. [Google Scholar] [CrossRef]
- Nojilana, B.; Abdelatif, N.; Cois, A.; E Schutte, A.; Wentzel-Viljoen, E.; Turuwa, E.B.; A Roomaney, R.; Awotiwon, O.F.; Neethling, I.; Pacella, R.; et al. Estimating the changing burden of disease attributable to high sodium intake in South Africa for 2000, 2006 and 2012. South Afr. Med. J. 2022, 112, 627–638. [Google Scholar] [CrossRef]
- Strauss-Kruger, M.; Wentzel-Viljoen, E.; Ware, L.J.; Van Zyl, T.; Charlton, K.; Ellis, S.; Schutte, A.E. Early evidence for the effectiveness of South Africa’s legislation on salt restriction in foods: The African-PREDICT study. J. Hum. Hypertens. 2023, 37, 42–49. [Google Scholar] [CrossRef]
- Vinturache, A.; Popoola, J.; Watt-Coote, I. The Changing Landscape of Acute Kidney Injury in Pregnancy from an Obstetrics Perspective. J. Clin. Med. 2019, 8, 1396. [Google Scholar] [CrossRef]
- Bentata, Y.; Housni, B.; Mimouni, A.; Azzouzi, A.; Abouqal, R. Acute kidney injury related to pregnancy in developing countries: Etiology and risk factors in an intensive care unit. J. Nephrol. 2012, 25, 764–775. [Google Scholar] [CrossRef]
- Najar, M.S.; Shah, A.R.; Wani, I.A.; Reshi, A.R.; Banday, K.A.; Bhat, M.A.; Saldanha, C.L. Pregnancy related acute kidney injury: A single center experience from the Kashmir Valley. Indian J. Nephrol. 2008, 18, 159–161. [Google Scholar] [CrossRef] [PubMed]
- Shalaby, A.S.; Shemies, R.S. Pregnancy-related acute kidney injury in the African continent: Where do we stand? A systematic review. J. Nephrol. 2022, 35, 2175–2189. [Google Scholar] [CrossRef] [PubMed]
- Organization WHO (2015) Trends in Maternal Mortality: 1990–2015: Estimates from WHO, UNICEF, UNFPA, World Bank Group and the United Nations Population Division: World Health Organization. Available online: https://www.unfpa.org/publications/trends-maternal-mortality-1990-2015 (accessed on 14 February 2023).
- Gaber, T.Z.; Shemies, R.S.; Baiomy, A.A.; Aladle, D.A.; Mosbah, A.; Abdel-Hady, E.S.; Sayed-Ahmed, N.; Sobh, M. Acute kidney injury during pregnancy and puerperium: An Egyptian hospital-based study. J. Nephrol. 2021, 34, 1611–1619. [Google Scholar] [CrossRef] [PubMed]
- Adejumo, O.; Akinbodewa, A.; Enikuomehin, O.; Lawal, O.; Abolarin, O.; Alli, O. Pregnancy-related acute kidney injury: Etiologies and short-term outcomes in a tertiary hospital in Southwest Nigeria. Saudi J. Kidney Dis. Transplant. 2019, 30, 1423–1430. [Google Scholar] [CrossRef]
- Awowole, I.O.; Omitinde, O.S.; Arogundade, F.A.; Bola-Oyebamiji, S.B.; Adeniyi, O.A. Pregnancy-related acute kidney injury requiring dialysis as an indicator of severe adverse maternal morbidity at a tertiary center in Southwest Nigeria. Eur. J. Obstet. Gynecol. Reprod. Biol. 2018, 225, 205–209. [Google Scholar] [CrossRef]
- Cooke, W.R.; Hemmilä, U.K.; Craik, A.L.; Mandula, C.J.; Mvula, P.; Msusa, A.; Dreyer, G.; Evans, R. Incidence, aetiology and outcomes of obstetric-related acute kidney injury in Malawi: A prospective observational study. BMC Nephrol. 2018, 19, 25. [Google Scholar] [CrossRef]
- Kabbali, N.; Tachfouti, N.; Arrayhani, M.; Harandou, M.; Tagnaouti, M.; Bentata, Y.; Laouad, I.; Ramdani, B.; Bayahia, R.; Oualim, Z.; et al. Outcome assessment of pregnancy-related acute kidney injury in Morocco: A national prospective study. Saudi J. Kidney Dis. Transplant. 2015, 26, 619–624. [Google Scholar] [CrossRef]
- Arrayhani, M.; El Youbi, R.; Sqalli, T. Pregnancy-Related Acute Kidney Injury: Experience of the Nephrology Unit at the University Hospital of Fez, Morocco. ISRN Nephrol. 2012, 2013, 109034. [Google Scholar] [CrossRef]
- Abdelkader, F.; Conte, A.B.; Saleh, A.M. Insuffisance rénale aigue du post partum: À propos de 102 cas au centre hospitalier National de Nouakchott, Mauritanie. PAMJ Clin. Med. 2020, 4, 48. [Google Scholar] [CrossRef]
- Msehli, M.; Jbali, H.; Ikram, M.; Ben Kaab, B.; Ben Hamida, F.; Rais, L.; BEN Fatma, L.; Karim, Z. Pregnancy-related acute kidney injury in Tunisia: A clinical challenge. Nephrol. Dial. Transplant. 2021, 36, gfab082-0024. [Google Scholar] [CrossRef]
- Elshinnawy, H.A.; Aref, H.M.; Rezk, K.M.; Elkotb, A.M.; Mohamed, A.Y. Study of Pregnancy related AKI in Egyptian patients: Incidence, Risk factors and Outcome. QJM Int. J. Med. 2020, 113, hcaa052-016. [Google Scholar] [CrossRef]
- Muhammad, A.S.; Usman, M.; Garba, B.I.; Abdullahi, U.; Mohammed, B.A.; Garba, S.; Liman, H.M.; Muhammad, A.M.; Bello, K.S. Pregnancy Related Acute Kidney Injury, Clinical profile and Outcome of management: An experience from 3 years retrospective review in a specialist hospital in Gusau, North-Western Nigeria. Trop. J. Nephrol. 2017, 12, 17–21. [Google Scholar]
- Conti-Ramsden, F.I.; Nathan, H.L.; De Greeff, A.; Hall, D.R.; Seed, P.T.; Chappell, L.; Shennan, A.H.; Bramham, K. Pregnancy-Related Acute Kidney Injury in Preeclampsia: Risk Factors and Renal Outcomes. Hypertension 2019, 74, 1144–1151. [Google Scholar] [CrossRef] [PubMed]
- Akuse, R.M.; Bosan, I.B.; Mohammed, H.H.; Eno, H. Outcome of pregnancy related acute kidney injury requiring haemodialysis in a Nigerian teaching hospital. Ther. RRT 2013, 8, 11. [Google Scholar]
- Bouaziz, M.; Chaari, A.; Turki, O.; Dammak, H.; Chelly, H.; Ammar, R.; Nasri, A.; Ben Algia, N.; Bahloul, M.; Ben Hamida, C. Acute renal failure and pregnancy: A seventeen-year experience of a Tunisian intensive care unit. Ren. Fail. 2013, 35, 1210–1215. [Google Scholar] [CrossRef]
- Oluseyi, A.; Ayodeji, A.; Ayodeji, F. Aetiologies and Short-term Outcomes of Acute Kidney Injury in a Tertiary Centre in Southwest Nigeria. Ethiop. J. Health Sci. 2016, 26, 37–44. [Google Scholar] [CrossRef]
- Liu, Y.; Ma, X.; Zheng, J.; Liu, X.; Yan, T. Pregnancy outcomes in patients with acute kidney injury during pregnancy: A systematic review and meta-analysis. BMC Pregnancy Childbirth 2017, 17, 235. [Google Scholar] [CrossRef]
- Prasad, N.; Gupta, A.; Bhadauria, D.; Kaul, A.; Sharma, R.; Kapoor, D.; Singh, R.; Krishna, A. Maternal, fetal and renal outcomes of pregnancy-associated acute kidney injury requiring dialysis. Indian J. Nephrol. 2015, 25, 77–81. [Google Scholar] [CrossRef]
- Prakash, J.; Pant, P.; Prakash, S.; Sivasankar, M.; Vohra, R.; Doley, P.; Pandey, L.; Singh, U. Changing picture of acute kidney injury in pregnancy: Study of 259 cases over a period of 33 years. Indian J. Nephrol. 2016, 26, 262–267. [Google Scholar] [CrossRef]
- Belayev, L.Y.; Palevsky, P.M. The link between acute kidney injury and chronic kidney disease. Curr. Opin. Nephrol. Hypertens. 2014, 23, 149–154. [Google Scholar] [CrossRef]
- Parr, S.K.; Matheny, M.E.; Abdel-Kader, K.; Greevy, R.A.; Bian, A.; Fly, J.; Chen, G.; Speroff, T.; Hung, A.M.; Ikizler, T.A.; et al. Acute kidney injury is a risk factor for subsequent proteinuria. Kidney Int. 2018, 93, 460–469. [Google Scholar] [CrossRef] [PubMed]
- Coca, S.G.; Singanamala, S.; Parikh, C.R. Chronic kidney disease after acute kidney injury: A systematic review and meta-analysis. Kidney Int. 2012, 81, 442–448. [Google Scholar] [CrossRef] [PubMed]
- Brown, M.C.; Best, K.E.; Pearce, M.S.; Waugh, J.; Robson, S.C.; Bell, R. Cardiovascular disease risk in women with pre-eclampsia: Systematic review and meta-analysis. Eur. J. Epidemiol. 2013, 28, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Barrett, P.M.; McCarthy, F.P.; Evans, M.; Kublickas, M.; Perry, I.J.; Stenvinkel, P.; Khashan, A.S.; Kublickiene, K. Hypertensive disorders of pregnancy and the risk of chronic kidney disease: A Swedish registry-based cohort study. PLoS Med. 2020, 17, e1003255. [Google Scholar] [CrossRef]
- Covella, B.; Vinturache, A.E.; Cabiddu, G.; Attini, R.; Gesualdo, L.; Versino, E.; Piccoli, G.B. A systematic review and meta-analysis indicates long-term risk of chronic and end-stage kidney disease after preeclampsia. Kidney Int. 2019, 96, 711–727. [Google Scholar] [CrossRef] [PubMed]
- Khashan, A.S.; Evans, M.; Kublickas, M.; McCarthy, F.P.; Kenny, L.C.; Stenvinkel, P.; Fitzgerald, T.; Kublickiene, K. Preeclampsia and risk of end stage kidney disease: A Swedish nationwide cohort study. PLoS Med. 2019, 16, e1002875. [Google Scholar] [CrossRef] [PubMed]
- Kattah, A. Preeclampsia and Kidney Disease: Deciphering Cause and Effect. Curr. Hypertens. Rep. 2020, 22, 91. [Google Scholar] [CrossRef]
- Say, L.; Chou, D.; Gemmill, A.; Tunçalp, Ö.; Moller, A.-B.; Daniels, J.; Gülmezoglu, A.M.; Temmerman, M.; Alkema, L. Global causes of maternal death: A WHO systematic analysis. Lancet Glob. Health 2014, 2, E323–E333. [Google Scholar] [CrossRef]
- Musarandega, R.; Nyakura, M.; Machekano, R.; Pattinson, R.; Munjanja, S.P. Causes of maternal mortality in Sub-Saharan Africa: A systematic review of studies published from 2015 to 2020. J. Glob. Health 2021, 11, 04048. [Google Scholar] [CrossRef]
- Raguema, N.; Gannoun, M.B.A.; Zitouni, H.; Ben Letaifa, D.; Seda, O.; Mahjoub, T.; Lavoie, J.L. Contribution of -1031T/C and -376G/A tumor necrosis factor alpha polymorphisms and haplotypes to preeclampsia risk in Tunisia (North Africa). J. Reprod. Immunol. 2022, 149, 103461. [Google Scholar] [CrossRef]
- Bombell, S.; McGUIRE, W. Tumour necrosis factor (-308A) polymorphism in pre-eclampsia: Meta-analysis of 16 case-control studies. Aust. N. Z. J. Obstet. Gynaecol. 2008, 48, 547–551. [Google Scholar] [CrossRef] [PubMed]
- Heiskanen, J.; Romppanen, E.-L.; Hiltunen, M.; Iivonen, S.; Mannermaa, A.; Punnonen, K.; Heinonen, S. Polymorphism in the Tumor Necrosis Factor-α Gene in Women with Preeclampsia. J. Assist. Reprod. Genet. 2002, 19, 220–223. [Google Scholar] [CrossRef] [PubMed]
- Thakoordeen-Reddy, S.; Winkler, C.; Moodley, J.; David, V.; Binns-Roemer, E.; Ramsuran, V.; Naicker, T. Maternal variants within the apolipoprotein L1 gene are associated with preeclampsia in a South African cohort of African ancestry. Eur. J. Obstet. Gynecol. Reprod. Biol. 2020, 246, 129–133. [Google Scholar] [CrossRef]
- Hong, X.; Rosenberg, A.Z.; Zhang, B.; Binns-Roemer, E.; David, V.; Lv, Y.; Hjorten, R.C.; Reidy, K.J.; Chen, T.K.; Wang, G.; et al. Joint Associations of Maternal-Fetal APOL1 Genotypes and Maternal Country of Origin with Preeclampsia Risk. Am. J. Kidney Dis. 2021, 77, 879–888.e1. [Google Scholar] [CrossRef] [PubMed]
- Miller, A.; Azhibekov, T.; O’Toole, J.F.; Sedor, J.R.; Williams, S.M.; Redline, R.W.; Bruggeman, L.A. Association of preeclampsia with infant APOL1 genotype in African Americans. BMC Med. Genet. 2020, 21, 110. [Google Scholar] [CrossRef] [PubMed]
- Dhanjal, M.; Owen, E.; Anthony, J.; Davidson, J.; Rayner, B. Short communication: Association of pre-eclampsia with the R563Q mutation of the β-subunit of the epithelial sodium channel. BJOG Int. J. Obstet. Gynaecol. 2006, 113, 595–598. [Google Scholar] [CrossRef] [PubMed]
- Centers for Disease Control and Prevention. Basic Statistics: HIV 2022. Available online: https://www.cdc.gov/hiv/basics/statistics.html (accessed on 21 June 2022).
- The Global HIV/AIDS Epidemic. Available online: https://www.hiv.gov/hiv-%20basics/overview/dataand-trends/global-%20statistics (accessed on 6 August 2022).
- Wong, C.; Gange, S.J.; Buchacz, K.; Moore, R.D.; Justice, A.C.; Horberg, M.A.; Gill, M.J.; Koethe, J.R.; Rebeiro, P.F.; Silverberg, M.J.; et al. First Occurrence of Diabetes, Chronic Kidney Disease, and Hypertension among North American HIV-Infected Adults, 2000–2013. Clin. Infect. Dis. 2016, 64, 459–467. [Google Scholar] [CrossRef]
- Ahuja, T.S.; Grady, J.; Khan, S. Changing Trends in the Survival of Dialysis Patients with Human Immunodeficiency Virus in the United States. J. Am. Soc. Nephrol. 2002, 13, 1889–1893. [Google Scholar] [CrossRef]
- Fabian, J.; Maher, H.A.; Clark, C.; Naicker, S.; Becker, P.; Venter, W.D.F. Morbidity and mortality of black HIV-positive patients with end-stage kidney disease receiving chronic haemodialysis in South Africa. South Afr. Med. J. 2015, 105, 110–114. [Google Scholar] [CrossRef]
- Halle, M.P.; Edjomo, A.M.; Fouda, H.; Djantio, H.; Essomba, N.; Ashuntantang, G.E. Survival of HIV infected patients on maintenance hemodialysis in Cameroon: A comparative study. BMC Nephrol. 2018, 19, 166. [Google Scholar] [CrossRef]
- Davids, M.R.; Jardine, R.; Marais, N.; Sebastian, S.; Davids, T.; Jacobs, J.C. South African Renal Registry Annual Report 2019. Afr. J. Nephrol. 2021, 24, 95–106. [Google Scholar] [CrossRef]
- Bookholane, H.; Wearne, N.; Surapaneni, A.; Ash, S.; Berghammer-Böhmer, R.; Omar, A.; Spies, R.; Grams, M.E. Predictors and Prognosis of HIV-Associated Nephropathy on Kidney Biopsy in South Africa. Kidney Int. Rep. 2020, 5, 1799–1804. [Google Scholar] [CrossRef] [PubMed]
- Diana, N.E.; Davies, M.; Mosiane, P.; Vermeulen, A.; Naicker, S. Clinicopathological correlation of kidney disease in HIV infection pre- and post- ART rollout. PLoS ONE 2022, 17, e0269260. [Google Scholar] [CrossRef] [PubMed]
- Kudose, S.; Santoriello, D.; Bomback, A.S.; Stokes, M.B.; Batal, I.; Markowitz, G.S.; Wyatt, C.M.; D’Agati, V.D. The spectrum of kidney biopsy findings in HIV-infected patients in the modern era. Kidney Int. 2020, 97, 1006–1016. [Google Scholar] [CrossRef] [PubMed]
- Berliner, A.R.; Fine, D.M.; Lucas, G.M.; Rahman, M.H.; Racusen, L.C.; Scheel, P.J.; Atta, M.G. Observations on a Cohort of HIV-Infected Patients Undergoing Native Renal Biopsy. Am. J. Nephrol. 2008, 28, 478–486. [Google Scholar] [CrossRef]
- Wearne, N.; Swanepoel, C.R.; Boulle, A.; Duffield, M.S.; Rayner, B.L. The spectrum of renal histologies seen in HIV with outcomes, prognostic indicators and clinical correlations. Nephrol. Dial. Transplant. 2012, 27, 4109–4118. [Google Scholar] [CrossRef]
- Wearne, N.; Manning, K.; Price, B.; Rayner, B.L.; Davidson, B.; Jones, E.S.; Spies, R.; Cunningham, C.; Omar, A.; Ash, S.; et al. The Evolving Spectrum of Kidney Histology in HIV-Positive Patients in South Africa. Kidney Int. Rep. 2023, in press. [Google Scholar] [CrossRef]
- Available online: https://www.who.int/news/item/27-10-2022-tuberculosis-deaths-and-disease-increase-during-the-covid-19-pandemic (accessed on 14 February 2023).
- Gelaw, Y.A.; Williams, G.; Magalhães, R.J.S.; Gilks, C.F.; Assefa, Y. HIV Prevalence Among Tuberculosis Patients in Sub-Saharan Africa: A Systematic Review and Meta-analysis. AIDS Behav. 2019, 23, 1561–1575. [Google Scholar] [CrossRef]
- Lanjewar, D.N.; Duggal, R. Pulmonary pathology in patients with AIDS: An autopsy study from Mumbai. HIV Med. 2001, 2, 266–271. [Google Scholar] [CrossRef]
- Soriano-Rosas, J.; Avila-Casado, M.; Carrera-Gonzalez, E.; Chavez-Mercado, L.; Cruz-Ortiz, H.; Rojo, J. AIDS-associated Nephropathy: 5-year Retrospective Morphologic Analysis of 87 Cases. Pathol. Res. Pract. 1998, 194, 567–570. [Google Scholar] [CrossRef]
- Wearne, N.; Hung, R.; Bohmer, R.; Spies, R.; Omar, A.; Ash, S.; Ibrahim, F.; Miller, R.F.; Booth, J.W.; Lucas, S.B.; et al. Kidney disease in Africans with HIV and tuberculosis. Aids 2019, 33, 1207–1213. [Google Scholar] [CrossRef] [PubMed]
- Davidson, B.; Nel, D.; Jones, E.S.W.; Manning, K.; Spies, R.; Bohmer, R.; Omar, A.; Ash, S.; Wearne, N. Granulomatous interstitial nephritis on renal biopsy in human immunodeficiency virus positive patients: Prevalence and causes in Cape Town, South Africa. Nephrology 2019, 24, 681–688. [Google Scholar] [CrossRef] [PubMed]
- Lawn, S.D.; Bekker, L.-G.; Miller, R.F. Immune reconstitution disease associated with mycobacterial infections in HIV-infected individuals receiving antiretrovirals. Lancet Infect. Dis. 2005, 5, 361–373. [Google Scholar] [CrossRef] [PubMed]
- Lee, W.A.; He, G.-X.; Eisenberg, E.; Cihlar, T.; Swaminathan, S.; Mulato, A.; Cundy, K.C. Selective Intracellular Activation of a Novel Prodrug of the Human Immunodeficiency Virus Reverse Transcriptase Inhibitor Tenofovir Leads to Preferential Distribution and Accumulation in Lymphatic Tissue. Antimicrob. Agents Chemother. 2005, 49, 1898–1906. [Google Scholar] [CrossRef]
- E Sax, P.; Wohl, D.; Yin, M.T.; Post, F.; DeJesus, E.; Saag, M.; Pozniak, A.; Thompson, M.; Podzamczer, D.; Molina, J.M.; et al. Tenofovir alafenamide versus tenofovir disoproxil fumarate, coformulated with elvitegravir, cobicistat, and emtricitabine, for initial treatment of HIV-1 infection: Two randomised, double-blind, phase 3, non-inferiority trials. Lancet 2015, 385, 2606–2615. [Google Scholar] [CrossRef]
- Atta, M.G.; De Seigneux, S.; Lucas, G.M. Clinical Pharmacology in HIV Therapy. Clin. J. Am. Soc. Nephrol. 2019, 14, 435–444. [Google Scholar] [CrossRef]
- Walmsley, S.L.; Antela, A.A.; Clumeck, N.; Duiculescu, D.; Eberhard, A.A.; Gutiérrez, F.; Hocqueloux, L.L.; Maggiolo, F.F.; Sandkovsky, U.U.; Granier, C.C.; et al. Dolutegravir plus Abacavir–Lamivudine for the Treatment of HIV-1 Infection. N. Engl. J. Med. 2013, 369, 1807–1818. [Google Scholar] [CrossRef]
- Muller, E.; Barday, Z. HIV-Positive Kidney Donor Selection for HIV-Positive Transplant Recipients. J. Am. Soc. Nephrol. 2018, 29, 1090–1095. [Google Scholar] [CrossRef]
- Muller, E.; Barday, Z.; Mendelson, M.; Kahn, D. HIV-Positive–to–HIV-Positive Kidney Transplantation—Results at 3 to 5 Years. N. Engl. J. Med. 2015, 372, 613–620. [Google Scholar] [CrossRef]
- Selhorst, P.; Combrinck, C.E.; Manning, K.; Botha, F.C.; Labuschagne, J.P.; Anthony, C.; Matten, D.L.; Breaud, A.; Clarke, W.; Quinn, T.C.; et al. Longer-Term Outcomes of HIV-Positive–to–HIV-Positive Renal Transplantation. N. Engl. J. Med. 2019, 381, 1387–1389. [Google Scholar] [CrossRef]
- Barday, Z.; Manning, K.; Freercks, R.; Bertels, L.; Wearne, N.; Muller, E. Retrospective Review of ART Regimens in HIV-Positive to HIV-Positive Kidney Transplant Recipients. Kidney Int. Rep. 2022, 7, 2039–2046. [Google Scholar] [CrossRef] [PubMed]
- Nobakht, E.; Cohen, S.D.; Rosenberg, A.Z.; Kimmel, P.L. HIV-associated immune complex kidney disease. Nat. Rev. Nephrol. 2016, 12, 291–300. [Google Scholar] [CrossRef] [PubMed]
- Swanepoel, C.R.; Atta, M.G.; D’Agati, V.D.; Estrella, M.M.; Fogo, A.B.; Naicker, S.; Post, F.A.; Wearne, N.; Winkler, C.A.; Cheung, M.; et al. Kidney disease in the setting of HIV infection: Conclusions from a Kidney Disease: Improving Global Outcomes (KDIGO) Controversies Conference. Kidney Int. 2018, 93, 545–559. [Google Scholar] [CrossRef]
- Booth, J.W.; Hamzah, L.; Jose, S.; Horsfield, C.; O’Donnell, P.; McAdoo, S.; Kumar, E.A.; Turner-Stokes, T.; Khatib, N.; Das, P.; et al. Clinical characteristics and outcomes of HIV-associated immune complex kidney disease. Nephrol. Dial. Transplant. 2016, 31, 2099–2107. [Google Scholar] [CrossRef] [PubMed]
- Inusah, A.-J.; Coetzee, L.; Bates, W.; Chothia, M.-Y. Kidney outcomes of immune-complex associated mesangiocapillary glomerulonephritis in patients with and without HIV. J. Nephropathol. 2022, 11, e17269. [Google Scholar] [CrossRef]
- Fabian, J.; Gondwe, M.; Mayindi, N.; Chipungu, S.; Khoza, B.; Gaylard, P.; Wade, A.N.; Gómez-Olivé, F.X.; A Tomlinson, L.; Ramsay, M.; et al. Chronic kidney disease (CKD) and associated risk in rural South Africa: A population-based cohort study. Wellcome Open Res. 2022, 7, 236. [Google Scholar] [CrossRef]
- Aseneh, J.B.; Kemah, B.-L.A.; Mabouna, S.; Njang, M.E.; Ekane, D.S.M.; Agbor, V.N. Chronic kidney disease in Cameroon: A scoping review. BMC Nephrol. 2020, 21, 409. [Google Scholar] [CrossRef] [PubMed]
- Abdissa, D. Purposeful Review to Identify Risk Factors, Epidemiology, Clinical Features, Treatment and Prevention of Chronic Kidney Disease of Unknown Etiology. Int. J. Nephrol. Renov. Dis. 2020, 13, 367–377. [Google Scholar] [CrossRef]
- Magombo, M.; Barregard, L.; Kgalamono, S.; George, J.; Naicker, S.; Dorkin, E.; Made, F.; Rees, D. O4D.4 Changes in kidney function among sugarcane cutters on a moderately hot sugar plantation in South Africa. Occup. Environ. Med. 2019, 76, A38. [Google Scholar] [CrossRef]
- Ajayi, S.; Raji, Y.R.; Michael, O.S.; Adewole, D.; Akande, T.; Abiola, B.; Aminu, S.; Olugbenga-Bello, A.; Arije, A. Exposure to Agrochemicals and Markers of Kidney Damage among Farmers in Rural Communities in Southwestern Nigeria. West Afr. J. Med. 2021, 38, 48–53. [Google Scholar]
- Olowogbon, T.S.; O Babatunde, R.; Asiedu, E.; Yoder, A.M. Agrochemical Health Risks Exposure and Its Determinants: Empirical Evidence among Cassava Farmers in Nigeria. J. Agromed. 2020, 26, 199–210. [Google Scholar] [CrossRef] [PubMed]
- Hamilton, S.A.; Nakanga, W.P.; Prynn, J.E.; Crampin, A.C.; Fecht, D.; Vineis, P.; Caplin, B.; Pearce, N.; Nyirenda, M.J. Prevalence and risk factors for chronic kidney disease of unknown cause in Malawi: A cross-sectional analysis in a rural and urban population. BMC Nephrol. 2020, 21, 387. [Google Scholar] [CrossRef] [PubMed]
- Hodel, N.C.; Hamad, A.; Praehauser, C.; Mwangoka, G.; Kasella, I.M.; Reither, K.; Abdulla, S.; Hatz, C.F.R.; Mayr, M. The epidemiology of chronic kidney disease and the association with non-communicable and communicable disorders in a population of sub-Saharan Africa. PLoS ONE 2018, 13, e0205326. [Google Scholar] [CrossRef] [PubMed]
- Conroy, A.L.; Opoka, R.O.; Bangirana, P.; Idro, R.; Ssenkusu, J.M.; Datta, D.; Hodges, J.S.; Morgan, C.; John, C.C. Acute kidney injury is associated with impaired cognition and chronic kidney disease in a prospective cohort of children with severe malaria. BMC Med. 2019, 17, 98. [Google Scholar] [CrossRef] [PubMed]
- George, C.; Yako, Y.Y.; Okpechi, I.G.; E Matsha, T.; Folefack, F.J.K.; Kengne, A.P. An African perspective on the genetic risk of chronic kidney disease: A systematic review. BMC Med. Genet. 2018, 19, 187. [Google Scholar] [CrossRef] [PubMed]
- Cave, E.M.; Prigge, K.L.; Crowther, N.J.; George, J.A.; Padoa, C.J. A Polymorphism in the Gene Encoding the Insulin Receptor Binding Protein ENPP-1 Is Associated with Decreased Glomerular Filtration Rate in an Under-Investigated Indigenous African Population. Kidney Blood Press. Res. 2020, 45, 1009–1017. [Google Scholar] [CrossRef] [PubMed]
- Ekrikpo, U.E.; Mnika, K.; Effa, E.E.; Ajayi, S.O.; Okwuonu, C.; Waziri, B.; Bello, A.; Dandara, C.; Kengne, A.P.; Wonkam, A.; et al. Association of Genetic Polymorphisms of TGF-β1, HMOX1, and APOL1 With CKD in Nigerian Patients With and Without HIV. Am. J. Kidney Dis. 2020, 76, 100–108. [Google Scholar] [CrossRef]
- Fatumo, S.; Chikowore, T.; Kalyesubula, R.; Nsubuga, R.N.; Asiki, G.; Nashiru, O.; Seeley, J.; Crampin, A.C.; Nitsch, D.; Smeeth, L.; et al. Discovery and fine-mapping of kidney function loci in first genome-wide association study in Africans. Hum. Mol. Genet. 2021, 30, 1559–1568. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).