Investigation of the Photoinduced Antimicrobial Properties of N-Doped TiO2 Nanoparticles under Visible-Light Irradiation on Salmonella Typhimurium Biofilm
Abstract
1. Introduction
2. Materials and Methods
2.1. Synthesis of N-Doped TiO2 NPs
2.2. Characterization
2.3. Antimicrobial Testing
3. Results and Discussion
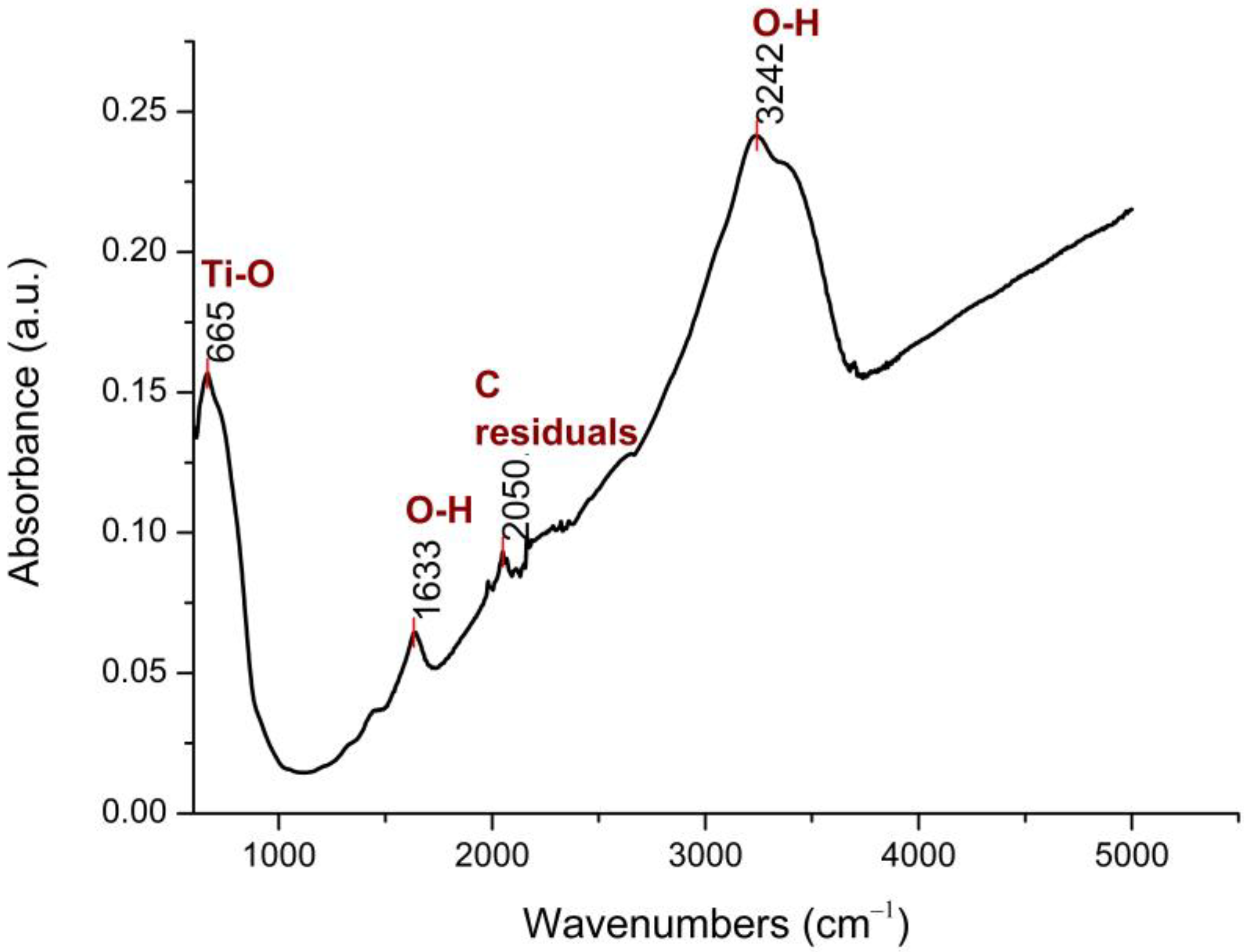
3.1. FTIR Analysis
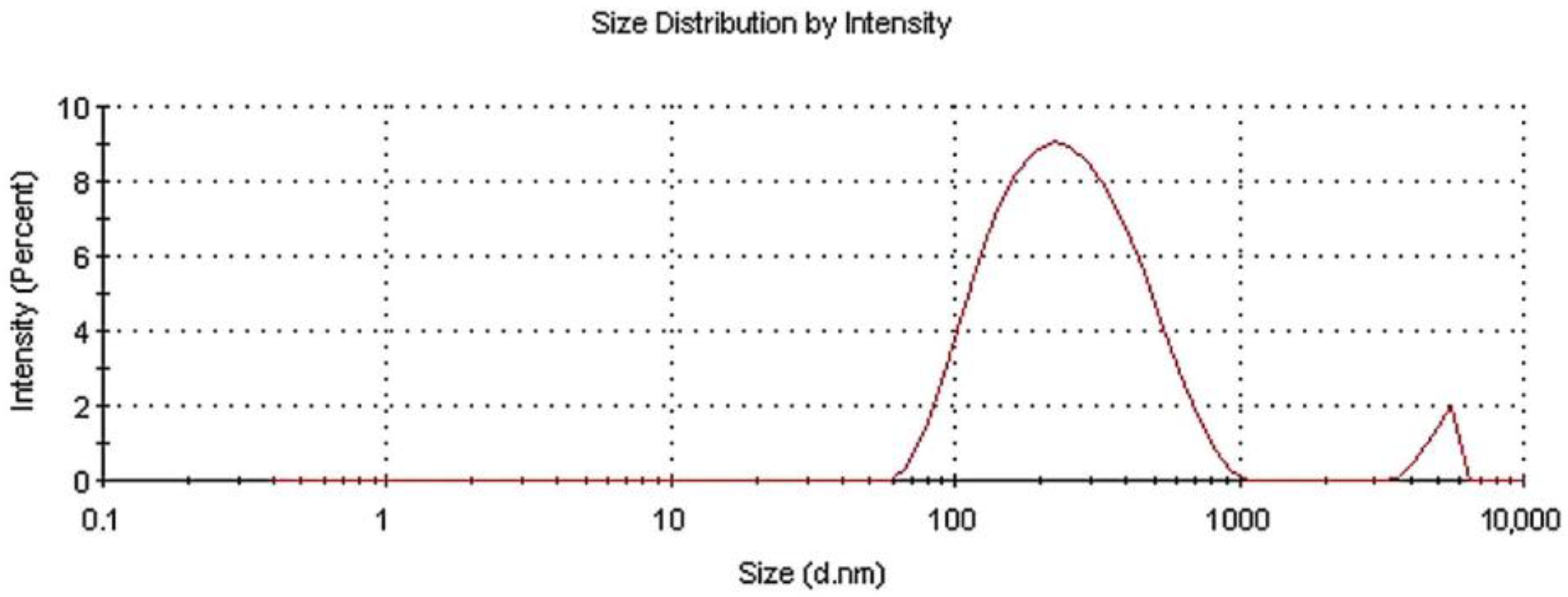
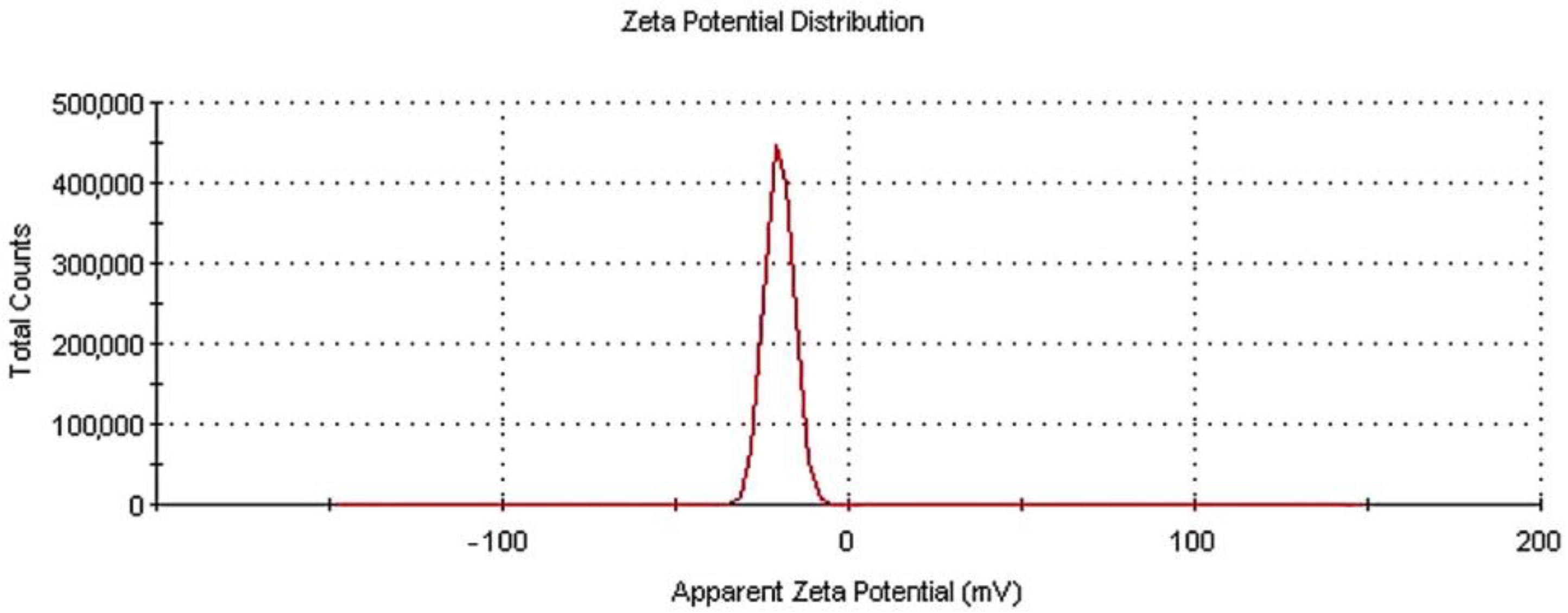
3.2. DLS Analysis
3.3. BET Analysis
3.4. SEM/EDS and TEM Analysis
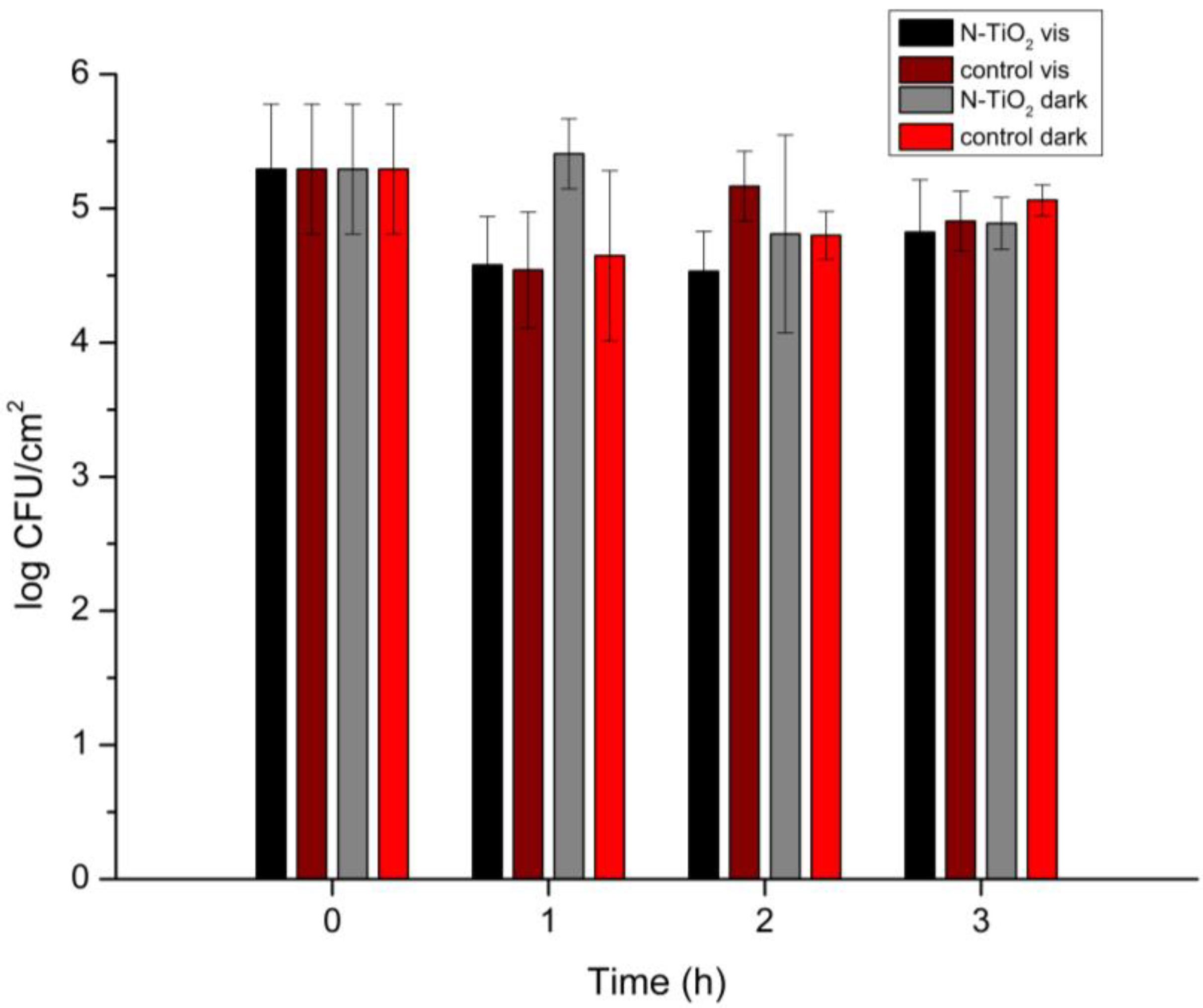
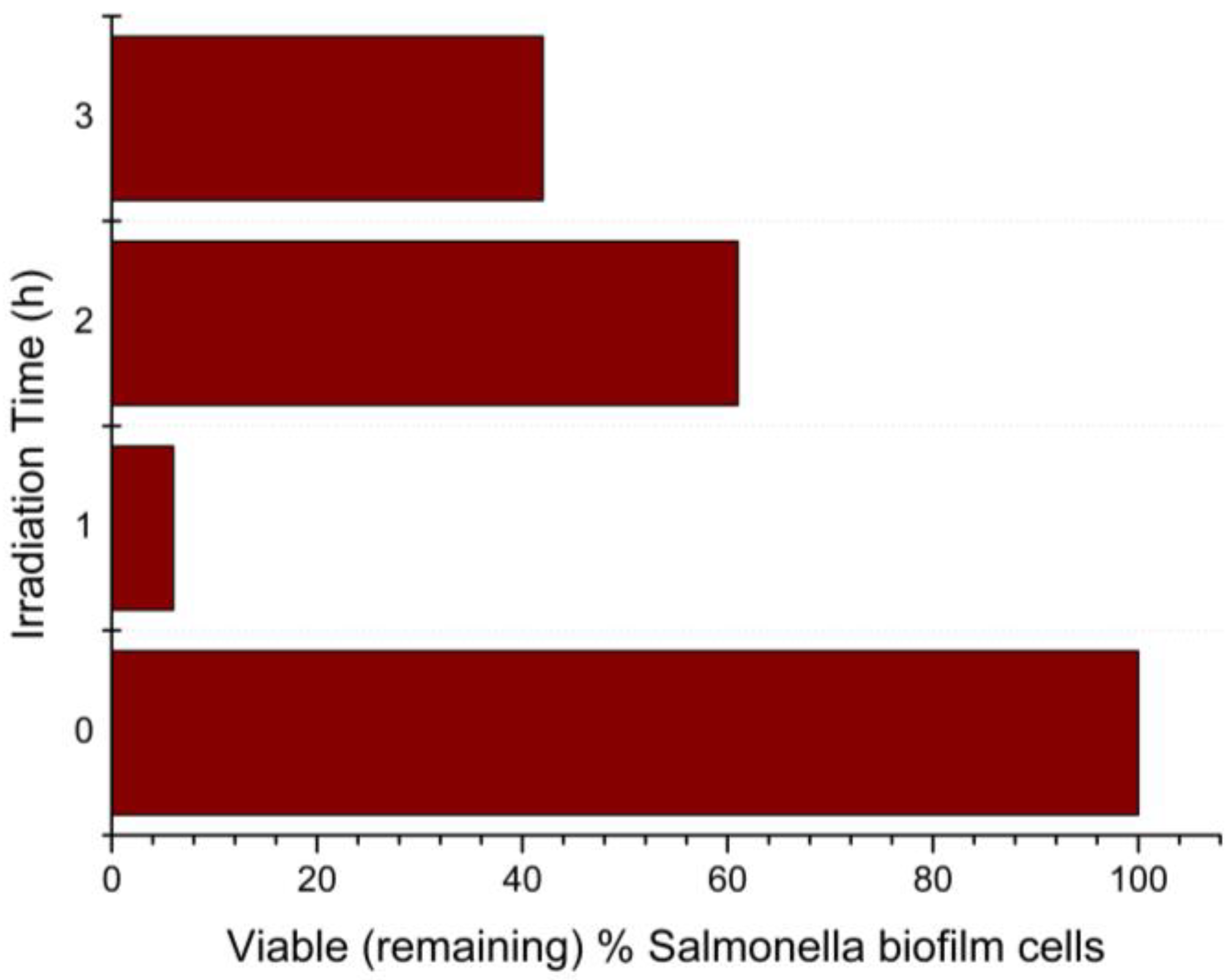
3.5. Antimicrobial Assessment
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Hall-Stoodley, L.; Costerton, J.W.; Stoodley, P. Bacterial biofilms: From the Natural environment to infectious diseases. Nat. Rev. Genet. 2004, 2, 95–108. [Google Scholar] [CrossRef]
- Ceramella, J.; Iacopetta, D.; Catalano, A.; Cirillo, F.; Lappano, R.; Sinicropi, M.S. A Review on the Antimicrobial Activity of Schiff Bases: Data Collection and Recent Studies. Antibiotics 2022, 11, 191. [Google Scholar] [CrossRef]
- Wang, L.; Hu, C.; Shao, L. The antimicrobial activity of nanoparticles: Present situation and prospects for the future. Int. J. Nanomed. 2017, 12, 1227–1249. [Google Scholar] [CrossRef] [PubMed]
- Dizaj, S.M.; Mennati, A.; Jafari, S.; Khezri, K.; Adibkia, K. Review. Antimicrobial activity of carbon-based nanoparticles. Adv. Pharm. Bull. 2015, 1, 19–23. [Google Scholar]
- Dizaj, S.M.; Lotfipour, F.; Barzegar-Jalali, M.; Zarrintan, M.H.; Adibkia, K. Review. Antimicrobial activity of the metals and metal oxide nanoparticles. Mater. Sci. Eng. C 2014, 44, 278–284. [Google Scholar] [CrossRef] [PubMed]
- Aziz, A.A.; Khatun, F.; Monir, M.U.; Ching, S.L.; Hon, L.K. TiO2: A Semiconductor Photocatalyst. In Titanium Dioxide—Advances and Applications; IntechOpen: London, UK, 2021; pp. 1–16. [Google Scholar]
- Yadav, S.; Jaiswar, G. Review on Undoped/Doped TiO2 Nanomaterial; Synthesis and Photocatalytic and Antimicrobial Activity. J. Chin. Chem. Soc. 2017, 64, 103–116. [Google Scholar] [CrossRef]
- Wold, A. Photocatalytic Properties of TiO2. Chem. Mater. 1993, 5, 280–283. [Google Scholar] [CrossRef]
- Bavykin, D.; Friedrich, J.; Walsh, F. Protonated Titanates and TiO2 Nanostructured Materials: Synthesis, Properties and Applications. Adv. Mater. 2006, 18, 2807–2824. [Google Scholar] [CrossRef]
- Hashimoto, K.; Irie, H.; Fujishima, A. TiO2 Photocatalysis: A Historical Overview and Future Prospects. Jpn. J. Appl. Phys. 2005, 44, 8269–8285. [Google Scholar] [CrossRef]
- Al-Mamun, M.; Kader, S.; Islam, M.; Khan, M. Photocatalytic activity improvement and application of UV-TiO2 photocatalysis in textile wastewater treatment: A review. J. Environ. Chem. Eng. 2019, 7, 103248. [Google Scholar] [CrossRef]
- Ansari, S.A.; Khan, M.M.; Ansari, M.O.; Cho, M.H. Nitrogen-doped titanium dioxide (N-doped TiO2) for visible light photocatalysis. New J. Chem. 2016, 40, 3000–3009. [Google Scholar] [CrossRef]
- Mathew, S.; Ganguly, P.; Rhatigan, S.; Kumaravel, V.; Byrne, C.; Hinder, S.J.; Bartlett, J.; Nolan, M.; Pillai, S.C. Cu-Doped TiO2: Visible Light Assisted Photocatalytic Antimicrobial Activity. Appl. Sci. 2018, 8, 2067. [Google Scholar] [CrossRef]
- Kubacka, A.; Muñoz-Batista, M.J.; Ferrer, M.; Fernández-García, M. UV and visible light optimization of anatase TiO2 antimicrobial properties: Surface deposition of metal and oxide (Cu, Zn, Ag) species. Appl. Catal. B Environ. 2013, 140–141, 680–690. [Google Scholar] [CrossRef]
- He, R.-L.; Wei, Y.; Cao, W.-B. Preparation of (Fe, N)-Doped TiO2 Powders and Their Antibacterial Activities Under Visible Light Irradiation. J. Nanosci. Nanotechnol. 2009, 9, 1094–1097. [Google Scholar] [CrossRef]
- Ananpattarachai, J.; Boonto, Y.; Kajitvichyanukul, P. Visible light photocatalytic antibacterial activity of Ni-doped and N-doped TiO2 on Staphylococcus aureus and Escherichia coli bacteria. Environ. Sci. Pollut. Res. 2016, 23, 4111–4119. [Google Scholar] [CrossRef]
- Yu, B.; Lau, W.M.; Yang, J. Preparation and characterization of N–TiO2 photocatalyst with high crystallinity and enhanced photocatalytic inactivation of bacteria. Nanotechnology 2013, 24, 335705. [Google Scholar] [CrossRef]
- Seneviratne, K.L.; Munaweera, I.; Peiris, S.E.; Peiris, C.N.; Kottegoda, N. Recent Progress in Visible-Light Active (VLA) TiO2 Nano-Structures for Enhanced Photocatalytic Activity (PCA) and Antibacterial Properties: A Review. Iran. J. Catal. 2021, 11, 217–245. [Google Scholar]
- Yan, Y.; Kuang, W.; Shi, L.; Ye, X.; Yang, Y.; Xie, X.; Shi, Q.; Tan, S. Carbon quantum dot-decorated TiO2 for fast and sustainable antibacterial properties under visible-light. J. Alloys Compd. 2019, 777, 234–243. [Google Scholar] [CrossRef]
- Raut, A.; Yadav, H.; Gnanamani, A.; Pushpavanam, S.; Pawar, S. Synthesis and characterization of chitosan-TiO2: Cu nanocomposite and their enhanced antimicrobial activity with visible light. Colloids Surf. B Biointerfaces 2016, 148, 566–575. [Google Scholar] [CrossRef]
- Wong, M.-S.; Chu, W.-C.; Sun, D.-S.; Huang, H.-S.; Chen, J.-H.; Tsai, P.-J.; Lin, N.-T.; Yu, M.-S.; Hsu, S.-F.; Wang, S.-L.; et al. Visible-Light-Induced Bactericidal Activity of a Nitrogen-Doped Titanium Photocatalyst against Human Pathogens. Appl. Environ. Microbiol. 2006, 72, 6111–6116. [Google Scholar] [CrossRef]
- Nagay, B.E.; Dini, C.; Cordeiro, J.M.; Ricomini-Filho, A.P.; de Avila, E.D.; Rangel, E.C.; da Cruz, N.C.; Barao, V.A.R. Visible-Light-Induced Photocatalytic and Antibacterial Activity of TiO2 Codoped with Nitrogen and Bismuth: New Perspectives to Control Implant-Biofilm-Related Diseases. ACS Appl. Mater. Interfaces 2019, 11, 18186–18202. [Google Scholar] [CrossRef] [PubMed]
- Cao, S.; Liu, B.; Fan, L.; Yue, Z.; Liu, B.; Cao, B. Highly antibacterial activity of N-doped TiO2 thin films coated onstainless steel brackets under visible light irradiation. Appl. Surf. Sci. 2014, 309, 119–127. [Google Scholar] [CrossRef]
- Lee, H.U.; Lee, S.C.; Choi, S.; Son, B.; Lee, S.M.; Kim, H.J.; Lee, J. Efficient visible-light induced photocatalysis on nanoporous nitrogen-doped titanium dioxide catalysts. Chem. Eng. J. 2013, 228, 756–764. [Google Scholar] [CrossRef]
- Prakash, B.; Veeregowda, B.M.; Krishnappa, G. Biofilms: A Survival Strategy of Bacteria. Curr. Sci. 2003, 85, 1299–1307. [Google Scholar]
- Kostaki, M.; Chorianopoulos, N.; Braxou, E.; Nychas, G.-J.; Giaouris, E. Differential Biofilm Formation and Chemical Disinfection Resistance of Sessile Cells of Listeria monocytogenes Strains under Monospecies and Dual-Species (with Salmonella enterica) Conditions. Appl. Environ. Microbiol. 2012, 78, 2586–2595. [Google Scholar] [CrossRef]
- Steenackers, H.; Hermans, K.; Vanderleyden, J.; De Keersmaecker, S.C. Salmonella biofilms: An overview on occurrence, structure, regulation and eradication. Food Res. Int. 2012, 45, 502–531. [Google Scholar] [CrossRef]
- Gkana, E.N.; Giaouris, E.D.; Doulgeraki, A.I.; Kathariou, S.; Nychas, G.-J.E. Biofilm formation by Salmonella Typhimurium and Staphylococcus aureus on stainless steel under either mono- or dual-species multi-strain conditions and resistance of sessile communities to sub-lethal chemical disinfection. Food Control. 2017, 73, 838–846. [Google Scholar] [CrossRef]
- Joseph, B.; Otta, S.; Karunasagar, I. Biofilm formation by Salmonella spp. on food contact surfaces and their sensitivity to sanitizers. Int. J. Food Microbiol. 2001, 64, 367–372. [Google Scholar] [CrossRef] [PubMed]
- Giaouris, E.; Chorianopoulos, N.; Nychas, G.-J. Effect of Temperature, pH, and Water Activity on Biofilm Formation by Salmonella enterica Enteritidis PT4 on Stainless Steel Surfaces as Indicated by the Bead Vortexing Method and Conductance Measurements. J. Food Prot. 2005, 68, 2149–2154. [Google Scholar] [CrossRef]
- Chorianopoulos, N.G.; Giaouris, E.D.; Kourkoutas, Y.; Nychas, G.-J.E. Inhibition of the Early Stage of Salmonella enterica Serovar Enteritidis Biofilm Development on Stainless Steel by Cell-Free Supernatant of a Hafnia alvei Culture. Appl. Environ. Microbiol. 2010, 76, 2018–2022. [Google Scholar] [CrossRef] [PubMed]
- Kassalia, M.-E.; Nikolaou, Z.; Pavlatou, E.A. Photocatalytic Testing Protocol for N-Doped TiO2 Nanostructured Particles under Visible Light Irradiation Using the Statistical Taguchi Experimental Design. Appl. Sci. 2023, 13, 774. [Google Scholar] [CrossRef]
- Galata, E.; Georgakopoulou, E.A.; Kassalia, M.-E.; Papadopoulou-Fermeli, N.; Pavlatou, E.A. Development of Smart Composites Based on Doped-TiO2 Nanoparticles with Visible Light Anticancer Properties. Materials 2019, 12, 2589. [Google Scholar] [CrossRef]
- Tan, L.; Ong, W.; Chai, S.; Mohamed, A. Reduced graphene oxide-TiO2 nanocomposite as a promising visible-light-active photocatalyst for the conversion of carbon dioxide. Nanoscale Res. Lett. 2013, 8, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Qiu, X.; Burda, C. The Effects of Sintering on the Photocatalytic Activity of N-Doped TiO2 Nanoparticles. Chem. Mater. 2008, 20, 2629–2636. [Google Scholar] [CrossRef]
- Sahu, M.; Biswas, P. Single-step processing of copper-doped titania nanomaterials in a flame aerosol reactor. Nanoscale Res. Lett. 2011, 6, 441. [Google Scholar] [CrossRef]
- Moulahoum, H.; Ghorbanizamani, F.; Sakarya, S.; Timur, S. Lightless catalytic layered chitosan coating film using doped TiO2@metal ions nanoparticles for highly efficient dye degradation in aqueous media and disinfection applications. Prog. Org. Coat. 2022, 169, 106923. [Google Scholar] [CrossRef]
- Bhattacharjee, S. DLS and zeta potential—What they are and what they are not? J. Control. Release 2016, 235, 337–351. [Google Scholar] [CrossRef]
- Bhatkhande, D.S.; Pangarkar, V.G.; Beenackers, A.A. Photocatalytic degradation for environmental applications—A review. J. Chem. Technol. Biotechnol. 2002, 77, 102–116. [Google Scholar] [CrossRef]
- Hussain, M.; Ceccarelli, R.; Marchisio, D.; Fino, D.; Russo, N.; Geobaldo, F. Synthesis, characterization, and photocatalytic application of novel TiO2 nanoparticles. Chem. Eng. J. 2010, 157, 45–51. [Google Scholar] [CrossRef]
- Thommes, M.; Kaneko, K.; Neimark, A.V.; Olivier, J.P.; Rodriguez-Reinoso, F.; Rouquerol, J.; Sing, K.S.W. Physisorption of Gases, With Special Reference to the Evaluation of Surface Area and Pore Size Distribution (IUPAC Technical Report). Pure Appl. Chem. 2015, 87, 1051. [Google Scholar] [CrossRef]
- de la Calle, I.; Menta, M.; Klein, M.; Maxit, B.; Séby, F. Towards routine analysis of TiO2 (nano-)particle size in consumer products: Evaluation of potential techniques. Spectrochim. Acta Part B At. Spectrosc. 2018, 147, 28–42. [Google Scholar] [CrossRef]
- Souza, T.G.F.; Ciminelli, V.S.T.; Mohallem, N.D.S. A comparison of TEM and DLS methods to characterize size distribution of ceramic nanoparticles. J. Phys. Conf. Ser. 2016, 733, 012039. [Google Scholar] [CrossRef]
- Ma, W.; Li, J.; Liu, Y.; Ren, X.; Gu, Z.-G.; Xie, Z.; Liang, J. Preparation and characterization of excellent antibacterial TiO2/N-halamines nanoparticles. Colloids Surf. A Physicochem. Eng. Asp. 2016, 506, 284–290. [Google Scholar] [CrossRef]
- Iwatsu, M.; Kanetaka, H.; Mokudai, T.; Ogawa, T.; Kawashita, M.; Sasaki, K. Visible light-induced photocatalytic and antibacterial activity of N-doped TiO2. J. Biomed. Mater. Res. 2020, 108B, 451–459. [Google Scholar] [CrossRef] [PubMed]
- Kumaravel, V.; Nair, K.M.; Mathew, S.; Bartlett, J.; Kennedy, J.E.; Manning, H.G.; Whelan, B.J.; Leyland, N.S.; Pillai, S.C. Antimicrobial TiO2 nanocomposite coatings for surfaces, dental and orthopaedic implants. Chem. Eng. J. 2021, 416, 129071. [Google Scholar] [CrossRef] [PubMed]
- Gkana, E.N.; Doulgeraki, A.I.; Chorianopoulos, N.G.; Nychas, G.-J.E. Anti-adhesion and Anti-biofilm Potential of Organosilane Nanoparticles against Foodborne Pathogens. Front. Microbiol. 2017, 8, 1295. [Google Scholar] [CrossRef]



| Sample | BET Surf. Area σ (m2 g−1) (Micropore Surf. Area) | V1.7–300 nm (cm3 g−1) | Av. Pore Diam. (nm). |
|---|---|---|---|
| N-TiO2 | 82 (6) | 0.3 | 13 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kassalia, M.-E.; Chorianopoulos, N.; Nychas, G.-J.; Pavlatou, E.A. Investigation of the Photoinduced Antimicrobial Properties of N-Doped TiO2 Nanoparticles under Visible-Light Irradiation on Salmonella Typhimurium Biofilm. Appl. Sci. 2023, 13, 4498. https://doi.org/10.3390/app13074498
Kassalia M-E, Chorianopoulos N, Nychas G-J, Pavlatou EA. Investigation of the Photoinduced Antimicrobial Properties of N-Doped TiO2 Nanoparticles under Visible-Light Irradiation on Salmonella Typhimurium Biofilm. Applied Sciences. 2023; 13(7):4498. https://doi.org/10.3390/app13074498
Chicago/Turabian StyleKassalia, Maria-Emmanouela, Nikos Chorianopoulos, George-John Nychas, and Evangelia A. Pavlatou. 2023. "Investigation of the Photoinduced Antimicrobial Properties of N-Doped TiO2 Nanoparticles under Visible-Light Irradiation on Salmonella Typhimurium Biofilm" Applied Sciences 13, no. 7: 4498. https://doi.org/10.3390/app13074498
APA StyleKassalia, M.-E., Chorianopoulos, N., Nychas, G.-J., & Pavlatou, E. A. (2023). Investigation of the Photoinduced Antimicrobial Properties of N-Doped TiO2 Nanoparticles under Visible-Light Irradiation on Salmonella Typhimurium Biofilm. Applied Sciences, 13(7), 4498. https://doi.org/10.3390/app13074498







