The Material Matters: Sorption/Desorption Study of Selected Estrogens on Common Tubing or Sampling Materials Used in Water Sampling, Handling, Analysis or Treatment Technologies
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals and Materials
2.2. Short-Term Material Exposure
2.3. Long-Term EPDM Exposure
2.4. LC-MS/MS Analysis
2.4.1. Solid Phase Extraction and Derivatization
2.4.2. Instrumental Analysis
2.5. Data Analysis and Quality Control
2.6. Statistical Evaluation
3. Results and Discussion
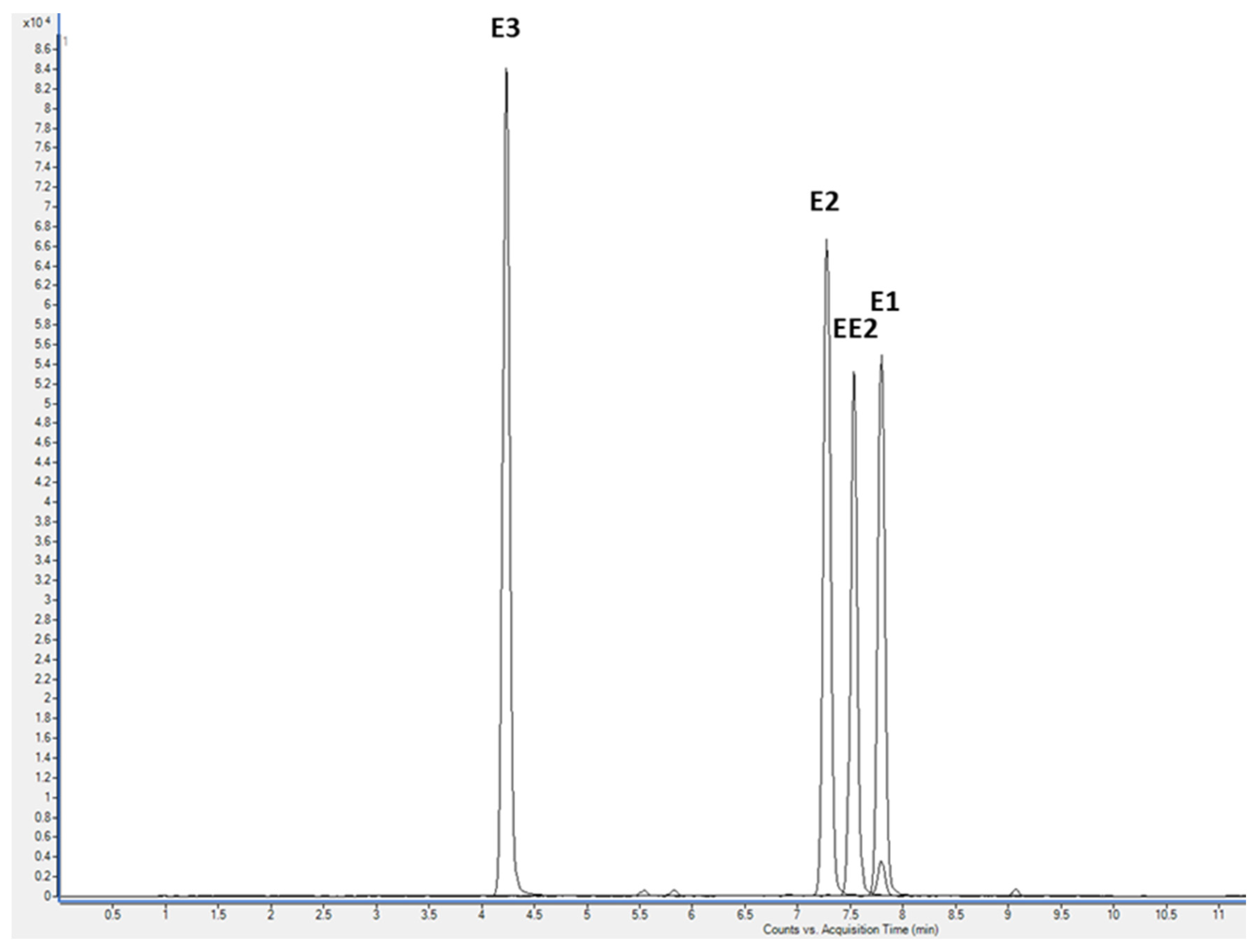
3.1. LC-MS/MS Analysis
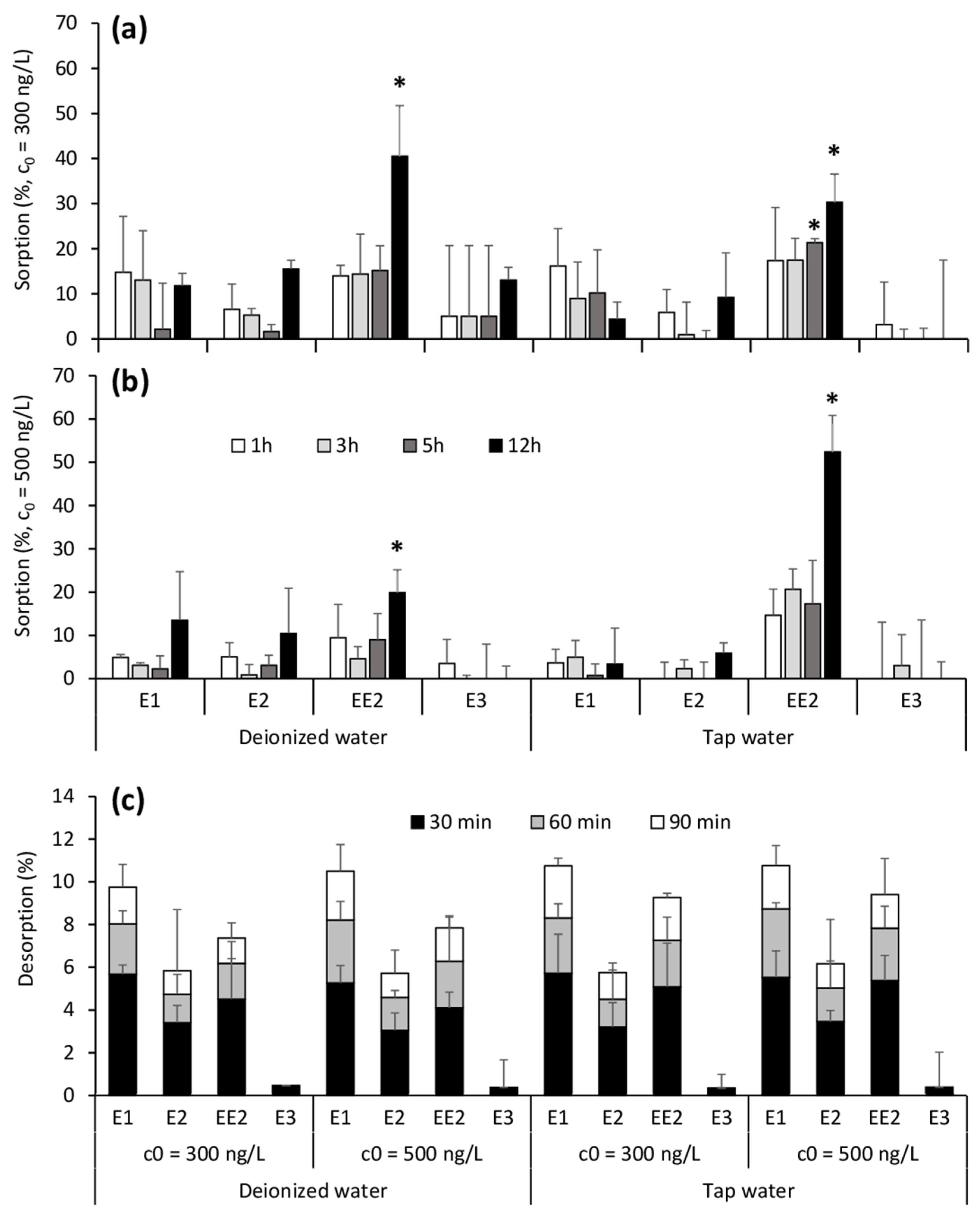
3.2. Short-Term Material Exposure
3.3. Long-Term Material Exposure
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Graur, V.; Mukherjee, A.; Sebakhy, K.O.; Bose, R.K. Initiated Chemical Vapor Deposition (iCVD) of Bio-Based Poly(tulipalin A) Coatings: Structure and Material Properties. Polymers 2022, 14, 3993. [Google Scholar] [CrossRef]
- Miklos, D.B.; Remy, C.; Jekel, M.; Linden, K.G.; Drewes, J.E.; Hubner, U. Evaluation of advanced oxidation processes for water and wastewater treatment—A critical review. Water Res. 2018, 139, 118–131. [Google Scholar] [CrossRef] [PubMed]
- Gogate, P.R.; Patil, P.N. Combined treatment technology based on synergism between hydrodynamic cavitation and advanced oxidation processes. Ultrason. Sonochemistry 2015, 25, 60–69. [Google Scholar] [CrossRef] [PubMed]
- Kanakaraju, D.; Glass, B.D.; Oelgemoller, M. Advanced oxidation process-mediated removal of pharmaceuticals from water: A review. J. Environ. Manag. 2018, 219, 189–207. [Google Scholar] [CrossRef]
- Walker, C.W.; Watson, J.E. Adsorption of Estrogens on Laboratory Materials and Filters during Sample Preparation. J. Environ. Qual. 2010, 39, 744–748. [Google Scholar] [CrossRef]
- Chai, Y.F.; Chen, H.P.; Gao, G.W.; Liu, X.; Lu, C.Y. Identification of new interferences leached from plastic microcentrifuge tubes in electrospray ionization mass spectrometry. Rapid Commun. Mass Spectrom. 2019, 33, 969–977. [Google Scholar] [CrossRef]
- Schafer, A.I.; Akanyeti, I.; Semiao, A.J.C. Micropollutant sorption to membrane polymers: A review of mechanisms for estrogens. Adv. Colloid Interface Sci. 2011, 164, 100–117. [Google Scholar] [CrossRef]
- Li, Q.; Han, C.B.; Horton, S.R.; Fuentes-Cabrera, M.; Sumpter, B.G.; Lu, W.C.; Bernholc, J.; Maksymovych, P.; Pan, M. Supramolecular Self-Assembly of pi-Conjugated Hydrocarbons via 2D Cooperative CH/pi Interaction. ACS Nano 2012, 6, 566–572. [Google Scholar] [CrossRef]
- E.U. Ec. Commission Implementing Decision (EU) 2015/495 of 20 March 2015 establishing a watch list of substances for Union-wide monitoring in the field of water policy pursuant to Directive 2008/105/EC of the European Parliament and of the Council. Available online: https://eur-lex.europa.eu/eli/dec_impl/2020/1161/oj (accessed on 7 June 2019).
- USEP Agency. Environmental Protection Agency (EPA), Analytical Methods: Contaminants of Emerging Concern Pharmaceuticals and Personal Care Products (PPCPs). Available online: http://www.epa.gov/cwa-methods/cwa-analytical-methods-contaminants-emerging-concern (accessed on 7 June 2019).
- Yang, Y.; Zhang, X.R.; Jiang, J.Y.; Han, J.R.; Li, W.X.; Li, X.Y.; Leung, K.M.Y.; Snyder, S.A.; Alvarez, P.J. Which Micropollutants in Water Environments Deserve More Attention Globally? Environ. Sci. Technol. 2022, 56, 13–29. [Google Scholar] [CrossRef]
- Zhou, S.B.; Di Paolo, C.; Wu, X.; Shao, Y.; Seiler, T.B.; Hollert, H. Optimization of screening-level risk assessment and priority selection of emerging pollutants—The case of pharmaceuticals in European surface waters. Environ. Int. 2019, 128, 1–10. [Google Scholar] [CrossRef]
- Auriol, M.; Filali-Meknassi, Y.; Tyagi, R.D.; Adams, C.D.; Surampalli, R.Y. Endocrine disrupting compounds removal from wastewater, a new challenge. Process Biochem. 2006, 41, 525–539. [Google Scholar] [CrossRef]
- Zhang, Y.P.; Zhou, J.L. Removal of estrone and 17 beta-estradiol from water by adsorption. Water Res. 2005, 39, 3991–4003. [Google Scholar] [CrossRef] [PubMed]
- Atkinson, S.K.; Marlatt, V.L.; Kimpe, L.E.; Lean, D.R.S.; Trudeau, V.L.; Blais, J.M. The occurrence of steroidal estrogens in south-eastern Ontario wastewater treatment plants. Sci. Total Environ. 2012, 430, 119–125. [Google Scholar] [CrossRef]
- Pessoa, G.P.; de Souza, N.C.; Vidal, C.B.; Alves, J.A.C.; Firmino, P.I.M.; Nascimento, R.F.; dos Santos, A.B. Occurrence and removal of estrogens in Brazilian wastewater treatment plants. Sci. Total Environ. 2014, 490, 288–295. [Google Scholar] [CrossRef] [PubMed]
- Kuster, M.; Lopez, M.J.; de Alda, M.J.L.; Barcelo, D. Analysis and distribution of estrogens and progestogens in sewage sludge, soils and sediments. Trac-Trends Anal. Chem. 2004, 23, 790–798. [Google Scholar] [CrossRef]
- Kuster, M.; de Alda, M.J.; Hernando, M.D.; Petrovic, M.; Martin-Alonso, J.; Barcelo, D. Analysis and occurrence of pharmaceuticals, estrogens, progestogens and polar pesticides in sewage treatment plant effluents, river water and drinking water in the Llobregat river basin (Barcelona, Spain). J. Hydrol. 2008, 358, 112–123. [Google Scholar] [CrossRef]
- Lei, K.; Lin, C.Y.; Zhu, Y.; Chen, W.; Pan, H.Y.; Sun, Z.; Sweetman, A.; Zhang, Q.; He, M.-C. Estrogens in municipal wastewater and receiving waters in the Beijing-Tianjin-Hebei region, China: Occurrence and risk assessment of mixtures. J. Hazard. Mater. 2020, 389, 121891. [Google Scholar] [CrossRef] [PubMed]
- Lei, B.L.; Huang, S.B.; Zhou, Y.Q.; Wang, D.H.; Wang, Z.J. Levels of six estrogens in water and sediment from three rivers in Tianjin area, China. Chemosphere 2009, 76, 36–42. [Google Scholar] [CrossRef]
- Gorga, M.; Insa, S.; Petrovic, M.; Barcelo, D. Occurrence and spatial distribution of EDCs and related compounds in waters and sediments of Iberian rivers. Sci. Total Environ. 2015, 503, 69–86. [Google Scholar] [CrossRef]
- Matthiessen, P.; Allen, Y.; Bamber, S.; Craft, J.; Hurst, M.; Hutchinson, T.; Feist, S.; Katsiadaki, I.; Kirby, M.; Robinson, C.; et al. The impact of oestrogenic and androgenic contamination on marine organisms in the United Kingdom—Summary of the EDMAR programme. Mar. Environ. Res. 2002, 54, 645–649. [Google Scholar] [CrossRef]
- Sumpter, J.P.; Jobling, S. The occurrence, causes, and consequences of estrogens in the aquatic environment. Environ. Toxicol. Chem. 2013, 32, 249–251. [Google Scholar] [CrossRef] [PubMed]
- Gabet-Giraud, V.; Miege, C.; Jacquet, R.; Coquery, M. Impact of wastewater treatment plants on receiving surface waters and a tentative risk evaluation: The case of estrogens and beta blockers. Environ. Sci. Pollut. Res. 2014, 21, 1708–1722. [Google Scholar] [CrossRef]
- Gutjahr-Gobell, R.E.; Zaroogian, G.E.; Horowitz, D.J.B.; Gleason, T.R.; Mills, L.J. Individual effects of estrogens on a marine fish, Cunner (Tautogolabrus adspersus), extrapolated to the population level. Ecotoxicol. Environ. Saf. 2006, 63, 244–252. [Google Scholar] [CrossRef] [PubMed]
- Adeel, M.; Song, X.M.; Wang, Y.Y.; Francis, D.; Yang, Y.S. Environmental impact of estrogens on human, animal and plant life: A critical review. Environ. Int. 2017, 99, 107–119. [Google Scholar] [CrossRef] [PubMed]
- Han, J.; Qiu, W.; Gao, W. Adsorption of estrone in microfiltration membrane filters. Chem. Eng. J. 2010, 165, 819–826. [Google Scholar] [CrossRef]
- Schafer, A.I.; Nghiem, L.D.; Waite, T.D. Removal of the natural hormone estrone from aqueous solutions using nanofiltration and reverse osmosis. Environ. Sci. Technol. 2003, 37, 182–188. [Google Scholar] [CrossRef]
- Chang, S.; Waite, T.D.; Schafer, A.I.; Fane, A.G. Adsorption of the endocrine-active compound estrone on microfiltration hollow fiber membranes. Environ. Sci. Technol. 2003, 37, 3158–3163. [Google Scholar] [CrossRef]
- Tong, X.; Jiang, L.S.; Li, Y.X.; Chen, X.C.; Zhao, Y.; Hu, B.Y.; Zhang, F. Function of agricultural waste montmorillonite-biochars for sorptive removal of 17 beta-estradiol. Bioresour. Technol. 2020, 296, 122368. [Google Scholar] [CrossRef]
- Zhang, P.; Liu, S.B.; Tan, X.F.; Liu, Y.G.; Zeng, G.M.; Yin, Z.H.; Ye, S.; Zeng, Z. Microwave-assisted chemical modification method for surface regulation of biochar and its application for estrogen removal. Process. Saf. Environ. Protect. 2019, 128, 329–341. [Google Scholar] [CrossRef]
- Kuczynska, A. Presence of pharmaceutical compounds in groundwater with respect to land use in the vicinity of sampling sites. Geologos 2019, 25, 231–240. [Google Scholar] [CrossRef]
- Jedruchniewicz, K.; Ok, Y.S.; Oleszczuk, P. COVID-19 discarded disposable gloves as a source and a vector of pollutants in the environment. J. Hazard. Mater. 2021, 417, 125938. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.M.; Xu, J.; Zhao, Y.P.; Shi, H.H.; Huang, C.H. Hydrophobic sorption behaviors of 17 beta-Estradiol on environmental microplastics. Chemosphere 2019, 226, 726–735. [Google Scholar] [CrossRef] [PubMed]
- Hu, B.Y.; Li, Y.X.; Jiang, L.S.; Chen, X.C.; Wang, L.; An, S.Y.; Zhang, F. Influence of microplastics occurrence on the adsorption of 17 beta-estradiol in soil. J. Hazard. Mater. 2020, 400, 123325. [Google Scholar] [CrossRef]
- Sadilek, J.; Spalovska, P.; Vrana, B.; Vavrova, M.; Marsalek, B.; Simek, Z. Comparison of extraction techniques for isolation of steroid oestrogens in environmentally relevant concentrations from sediment. Int J. Environ. Anal. Chem. 2016, 96, 1022–1037. [Google Scholar] [CrossRef]
- McKeen, L. Introduction to Plastics and Polymers. In Film Properties of Plastics and Elastomers, 3rd ed.; PDL Handbook Series; Elsevier Science B.V.: Amsterdam, The Netherlands, 2012; pp. 1–18. [Google Scholar]
- Jonker, M.T.O.; Muijs, B. Using solid phase micro extraction to determine salting-out (Setschenow) constants for hydrophobic organic chemicals. Chemosphere 2010, 80, 223–227. [Google Scholar] [CrossRef] [PubMed]
- Campbell, C.G.; Borglin, S.E.; Green, F.B.; Grayson, A.; Wozei, E.; Stringfellow, W.T. Biologically directed environmental monitoring, fate, and transport of estrogenic endocrine disrupting compounds in water: A review. Chemosphere 2006, 65, 1265–1280. [Google Scholar] [CrossRef]

| Material | Characteristics a | Applications |
|---|---|---|
| Tygon S3™ E-3603 | non-DEHP PVC, non-toxic, non-contaminating, chemical resistance, non-oxidizing, glassy–smooth inner bore | analytical instruments, general laboratory, peristaltic and vacuum pumps, biopharmaceutical, incubators, desiccators, gas lines, food, beverages |
| Polyvinyl chloride (PVC) | resistant to weathering, chemical rotting, corrosion, shock and abrasion; self-extinguishing | water, gas, sewage, industrial process, irrigation, medical devices, blood storage bags |
| Teflon (PTFE) | chemical inertness, high resistance to ageing, temperature resistance | conveying pressures and temperatures, fluids, corrosive fluids, steam, push–pull cables, seals, gaskets |
| Kynar® (PVDF) | outstanding resistance to UV exposure, tremendous chemical resistance to a wide range of aggressive chemicals, resistance to chemical products, soluble in aprotic solvents | contact surface for the production, storage and transfer of corrosive fluids, used in mechanical components, fabricated vessels, tanks, pumps, valves, filters, heat exchangers, tower packing, piping systems, seals, gaskets |
| Ethylene propylene diene monomer (EPDM) | resistant to ozone and weather, abrasion resistant, UV resistant, electrically conductive, cloth impression | suitable for use as a discharge hose in the chemical industry and raw materials extraction |
| Styrene butadiene rubber (SBR) | good resilience and tensile strength, outstanding resistance to abrasion and fatigue; water, organic acid, ketone, chemical, alcohol, and aldehyde resistance; low resistance to ozone | industrial applications, adhesives, rubber/mechanical goods, car tires |
| Compound | Precursor Ion (m/z) | Quantitation/Qualification Ion (m/z) | Collision Energy (V) | Fragmentor Voltage (V) |
|---|---|---|---|---|
| E1 | 504 | 171/156 | 140 | 38 |
| E2 | 506 | 171/156 | 140 | 42 |
| EE2 | 530 | 171/156 | 140 | 40 |
| E3 | 522 | 171/156 | 140 | 42 |
| E2-d4 | 510 | 171/156 | 140 | 42 |
| EE2-d4 | 534 | 171/156 | 140 | 40 |
| Compound | MDL (ng/L) | MQL (ng/L) | CV a (%) Inter-Day | CV a (%) Intra-Day | CV b (%) Inter-Day | CV b (%) Intra-Day | CV c (%) Inter-Day | CV c (%) Intra-Day |
|---|---|---|---|---|---|---|---|---|
| E1 | 0.2 | 0.5 | 7.54 | 10.41 | 12.22 | 11.59 | 11.18 | 9.97 |
| E2 | 0.2 | 0.5 | 4.47 | 7.33 | 13.03 | 13.08 | 5.29 | 4.51 |
| EE2 | 0.1 | 0.4 | 3.69 | 7.65 | 8.88 | 8.76 | 5.62 | 4.50 |
| E3 | 0.2 | 0.6 | 6.43 | 3.84 | 13.25 | 13.28 | 13.94 | 15.09 |
| Concentration | E1 | E2 | EE2 | E3 |
|---|---|---|---|---|
| 100 ng/L a | 97.06 | 100.35 | 99.03 | 123.84 |
| 300 ng/L a | 73.15 | 83.34 | 88.52 | 94.11 |
| 300 ng/L b | 88.47 | 92.43 | 92.91 | 95.17 |
| 500 ng/L a | 114.33 | 104.61 | 111.48 | 83.62 |
| 500 ng/L b | 96.80 | 107.75 | 103.07 | 113.09 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Odehnalová, K.; Přibilová, P.; Zezulka, Š.; Maršálek, B. The Material Matters: Sorption/Desorption Study of Selected Estrogens on Common Tubing or Sampling Materials Used in Water Sampling, Handling, Analysis or Treatment Technologies. Appl. Sci. 2023, 13, 3328. https://doi.org/10.3390/app13053328
Odehnalová K, Přibilová P, Zezulka Š, Maršálek B. The Material Matters: Sorption/Desorption Study of Selected Estrogens on Common Tubing or Sampling Materials Used in Water Sampling, Handling, Analysis or Treatment Technologies. Applied Sciences. 2023; 13(5):3328. https://doi.org/10.3390/app13053328
Chicago/Turabian StyleOdehnalová, Klára, Petra Přibilová, Štěpán Zezulka, and Blahoslav Maršálek. 2023. "The Material Matters: Sorption/Desorption Study of Selected Estrogens on Common Tubing or Sampling Materials Used in Water Sampling, Handling, Analysis or Treatment Technologies" Applied Sciences 13, no. 5: 3328. https://doi.org/10.3390/app13053328
APA StyleOdehnalová, K., Přibilová, P., Zezulka, Š., & Maršálek, B. (2023). The Material Matters: Sorption/Desorption Study of Selected Estrogens on Common Tubing or Sampling Materials Used in Water Sampling, Handling, Analysis or Treatment Technologies. Applied Sciences, 13(5), 3328. https://doi.org/10.3390/app13053328






