The Influence of Emotional and Cognitive Factors on Limb Laterality Discrimination in Women with Fibromyalgia Syndrome: A Cross-Sectional Study Secondary Analysis
Abstract
1. Introduction
2. Materials and Methods
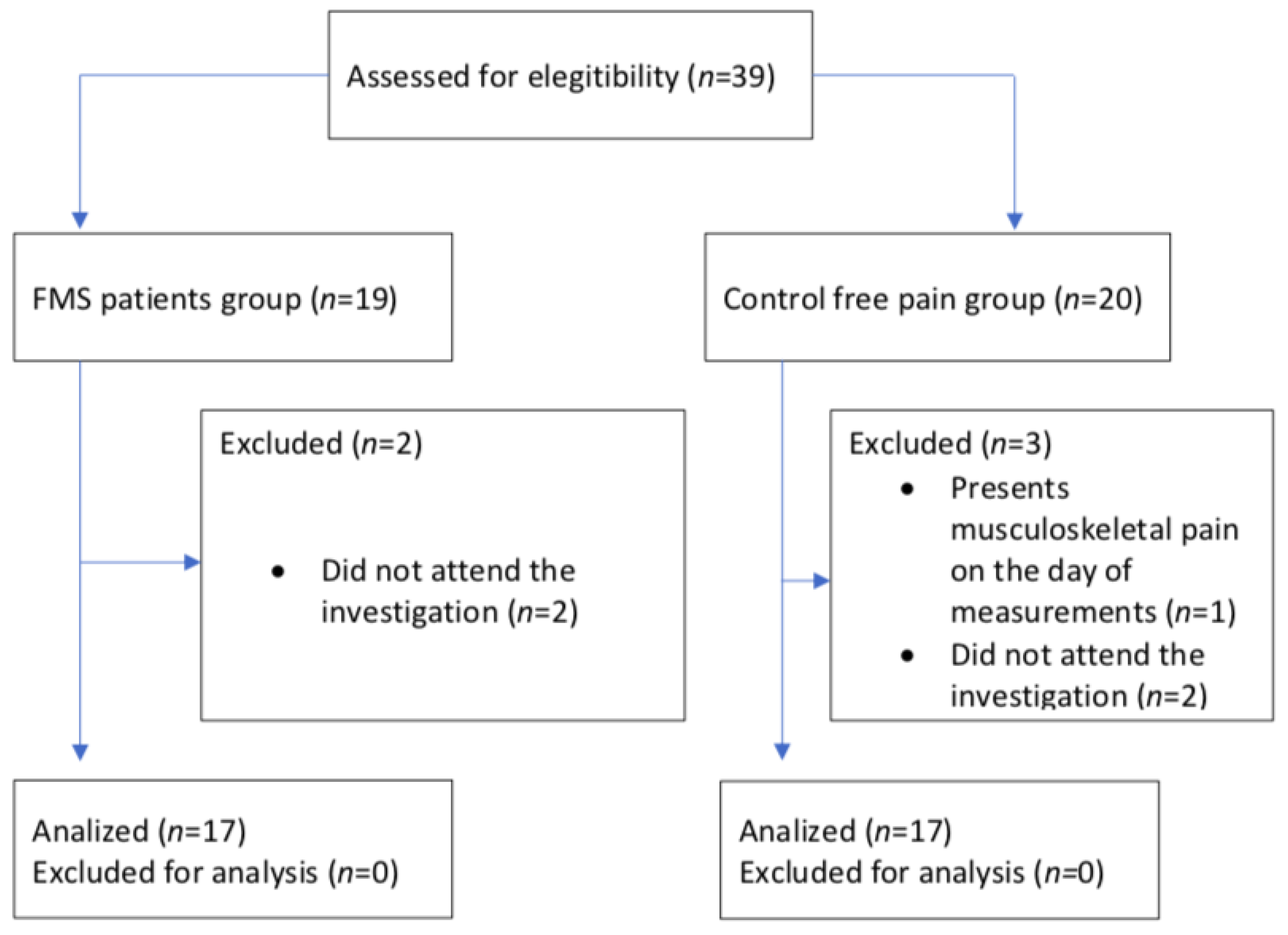
2.1. Participants
2.2. Pain and Clinical Status
2.3. Left/Right Judgment Test (LRJT)
2.4. Anxiety
2.5. Depression
2.6. Pain Catastrophizing
- Pain catastrophizing was measured with the validated Spanish version of the Pain Catastrophizing Scale (PCS). This is a self-administered scale consisting of 13 items with a score ranging from 0 “Not at all” to 4 “All the time.” It presents three dimensions: (1) helplessness corresponds to questions 1 to 5 and 12 and refers to the person’s belief about their capability to influence their pain; (2) magnification corresponds to questions 6, 7, and 13 and refers to exaggeration of the threatening properties of the painful stimulus; (3) rumination corresponds to questions 8 to 11 and refers to the patients’ inability to stop thinking of their pain and to avoid the idea.
2.7. Fear-Related Movement (Kinesiophobia)
- The Spanish version of Tampa Kinesiophobia scale (TSK-11) was used to measure the fear that a patient has of movement. It consists of 11 items, each with 1 of 4 response options, where “strongly disagree” scores 1 point and “strongly agree” scores 4 points. Therefore, the total score will vary between a minimum of 11 and a maximum of 44. A high score means a greater fear of movement/injury, that is, high levels of fear-related movement [66]. The TSK-11 has been shown to be consistent, reliable, and appropriate to assess fear of movement in patients with FMS within a clinical context (ICC = 0.85) [66,67].
2.8. Data Analysis
3. Results
3.1. Baseline Clinical Status of FMS Patients
3.2. Impairment of Limb Laterality Discrimination
3.3. Emotional and Cognitive Factors
3.4. Correlation Analysis
4. Discussion
4.1. Limb Laterality Discrimination and Psychological Variables in Fibromyalgia Pain Syndrome
4.2. Why Do Psychological Variables Not Influence Limb Laterality Discrimination? A Neurophysiological Explanation
4.3. Limitations
4.4. Future Research Lines
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Merskey, H.; Bogduk, N. Classification of Chronic Pain. Descriptions of Chronic Pain Syndromes and Definitions of Pain Terms. Prepared by the International Association for the Study of Pain, Subcommittee on Taxonomy. Pain Suppl. 1986, 3, S1–S226. [Google Scholar]
- Melzack, R. From the Gate to the Neuromatrix. Pain 1999, 82 (Suppl. 6), S121–S126. [Google Scholar] [CrossRef]
- International Association for the Study of Pain (IASP) IASP Terminology (Internet). 2017. Available online: https://www.iasp-pain.org/resources/terminology/ (accessed on 1 October 2022).
- Knudsen, L.; Petersen, G.L.; Nørskov, K.N.; Vase, L.; Finnerup, N.; Jensen, T.S.; Svensson, P. Review of Neuroimaging Studies Related to Pain Modulation. Scand. J. Pain 2018, 2, 108–120. [Google Scholar] [CrossRef] [PubMed]
- Bushnell, M.C.; Čeko, M.; Low, L.A. Cognitive and Emotional Control of Pain and Its Disruption in Chronic Pain. Nat. Rev. Neurosci. 2013, 14, 502–511. [Google Scholar] [CrossRef]
- Malfliet, A.; Coppieters, I.; van Wilgen, P.; Kregel, J.; de Pauw, R.; Dolphens, M.; Ickmans, K. Brain Changes Associated with Cognitive and Emotional Factors in Chronic Pain: A Systematic Review. Eur. J. Pain 2017, 21, 769–786. [Google Scholar] [CrossRef]
- Wolfe, F.; Brähler, E.; Hinz, A.; Häuser, W. Fibromyalgia Prevalence, Somatic Symptom Reporting, and the Dimensionality of Polysymptomatic Distress: Results from a Survey of the General Population. Arthritis Care Res. 2013, 65, 777–785. [Google Scholar] [CrossRef] [PubMed]
- Wolfe, F.; Ross, K.; Anderson, J.; Russell, I.J.; Hebert, L. The Prevalence and Characteristics of Fibromyalgia in the General Population. Arthritis Rheum. 1995, 38, 19–28. [Google Scholar] [CrossRef]
- Wolfe, F.; Smythe, H.A.; Yunus, M.B.; Bennett, R.M.; Bombardier, C.; Goldenberg, D.L.; Tugwell, P.; Campbell, S.M.; Abeles, M.; Clark, P.; et al. The American College of Rheumatology 1990 Criteria for the Classification of Fibromyalgia. Arthritis Rheum. 1990, 33, 160–172. [Google Scholar] [CrossRef] [PubMed]
- Sarzi-Puttini, P.; Buskila, D.; Carrabba, M.; Doria, A.; Atzeni, F. Treatment Strategy in Fibromyalgia Syndrome: Where Are We Now? Semin. Arthritis Rheum. 2008, 37, 353–365. [Google Scholar] [CrossRef] [PubMed]
- Staud, R.; Domingo, M. Evidence for Abnormal Pain Processing in Fibromyalgia Syndrome. Pain Med. 2001, 2, 208–215. [Google Scholar] [CrossRef] [PubMed]
- Silverman, S.L.; Harnett, J.; Zlateva, G.; Mardekian, J. Identifying Fibromyalgia-Associated Symptoms and Conditions from a Clinical Perspective: A Step toward Evaluating Healthcare Resource Utilization in Fibromyalgia. Pain Pract. 2010, 10, 520–529. [Google Scholar] [CrossRef] [PubMed]
- Serra, J.; Collado, A.; Solà, R.; Antonelli, F.; Torres, X.; Salgueiro, M.; Quiles, C.; Bostock, H. Hyperexcitable C Nociceptors in Fibromyalgia. Ann. Neurol. 2014, 75, 196–208. [Google Scholar] [CrossRef] [PubMed]
- Desmeules, J.A.; Cedraschi, C.; Rapiti, E.; Baumgartner, E.; Finckh, A.; Cohen, P.; Dayer, P.; Vischer, T.L. Neurophysiologic Evidence for a Central Sensitization in Patients with Fibromyalgia. Arthritis Rheum. 2003, 48, 1420–1429. [Google Scholar] [CrossRef] [PubMed]
- Gracely, R.H.; Ambrose, K.R. Neuroimaging of Fibromyalgia. Best Pract. Res. Clin. Rheumatol. 2011, 25, 271–284. [Google Scholar] [CrossRef] [PubMed]
- Kaplan, C.M.; Schrepf, A.; Vatansever, D.; Larkin, T.E.; Mawla, I.; Ichesco, E.; Kochlefl, L.; Harte, S.E.; Clauw, D.J.; Mashour, G.A.; et al. Functional and Neurochemical Disruptions of Brain Hub Topology in Chronic Pain. Pain 2019, 160, 973–983. [Google Scholar] [CrossRef]
- Staud, R.; Robinson, M.E.; Vierck, C.J.; Price, D.D. Diffuse Noxious Inhibitory Controls (DNIC) Attenuate Temporal Summation of Second Pain in Normal Males but Not in Normal Females or Fibromyalgia Patients. Pain 2003, 101, 167–174. [Google Scholar] [CrossRef]
- Harris, R.E.; Clauw, D.J.; Scott, D.J.; McLean, S.A.; Gracely, R.H.; Zubieta, J.-K. Decreased Central Mu-Opioid Receptor Availability in Fibromyalgia. J. Neurosci. 2007, 27, 10000–10006. [Google Scholar] [CrossRef]
- O’Brien, A.T.; Deitos, A.; Triñanes Pego, Y.; Fregni, F.; Carrillo-de-la-Peña, M.T. Defective Endogenous Pain Modulation in Fibromyalgia: A Meta-Analysis of Temporal Summation and Conditioned Pain Modulation Paradigms. J. Pain 2018, 19, 819–836. [Google Scholar] [CrossRef]
- Jones, K.D.; Horak, F.B.; Winters-Stone, K.; Irvine, J.M.; Bennett, R.M. Fibromyalgia Is Associated with Impaired Balance and Falls. J. Clin. Rheumatol. 2009, 15, 16–21. [Google Scholar] [CrossRef]
- Burgunder, J.M. Pathophysiology of Akinetic Movement Disorders: A Paradigm for Studies in Fibromyalgia? Z. Rheumatol. 1998, 57 (Suppl. 2), 27–30. [Google Scholar] [CrossRef]
- Lee, Y.C.; Nassikas, N.J.; Clauw, D.J. The Role of the Central Nervous System in the Generation and Maintenance of Chronic Pain in Rheumatoid Arthritis, Osteoarthritis and Fibromyalgia. Arthritis Res. Ther. 2011, 13, 211. [Google Scholar] [CrossRef] [PubMed]
- Valenzuela-Moguillansky, C.; Reyes-Reyes, A.; Gaete, M.I. Exteroceptive and Interoceptive Body-Self Awareness in Fibromyalgia Patients. Front. Hum. Neurosci. 2017, 11, 117. [Google Scholar] [CrossRef]
- Yunus, M.B.; Aldag, J.C. Restless Legs Syndrome and Leg Cramps in Fibromyalgia Syndrome: A Controlled Study. BMJ 1996, 312, 1339. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Protsenko, E.; Lazaridou, A.; Franceschelli, O.; Ellingsen, D.M.; Mawla, I.; Isenburg, K.; Berry, M.P.; Galenkamp, L.; Loggia, M.L.; et al. Encoding of Self-Referential Pain Catastrophizing in the Posterior Cingulate Cortex in Fibromyalgia. Arthritis Rheumatol. 2018, 70, 1308–1318. [Google Scholar] [CrossRef] [PubMed]
- Bernik, M.; Sampaio, T.P.A.; Gandarela, L. Fibromyalgia Comorbid with Anxiety Disorders and Depression: Combined Medical and Psychological Treatment. Curr. Pain Headache Rep. 2013, 17, 358. [Google Scholar] [CrossRef]
- Giesecke, T.; Williams, D.A.; Harris, R.E.; Cupps, T.R.; Tian, X.; Tian, T.X.; Gracely, R.H.; Clauw, D.J. Subgrouping of Fibromyalgia Patients on the Basis of Pressure-Pain Thresholds and Psychological Factors. Arthritis Rheum. 2003, 48, 2916–2922. [Google Scholar] [CrossRef]
- Leon-Llamas, J.L.; Murillo-Garcia, A.; Villafaina, S.; Domínguez-Muñoz, F.J.; Morenas, J.; Gusi, N. Relationship between Kinesiophobia and Mobility, Impact of the Disease, and Fear of Falling in Women with and without Fibromyalgia: A Cross-Sectional Study. Int. J. Environ. Res. Public Health 2022, 19, 8257. [Google Scholar] [CrossRef]
- Koçyiğit, B.F.; Akaltun, M.S. Kinesiophobia Levels in Fibromyalgia Syndrome and the Relationship Between Pain, Disease Activity, Depression. Arch. Rheumatol. 2020, 35, 214–219. [Google Scholar] [CrossRef]
- Haider, S.; Janowski, A.J.; Lesnak, J.B.; Hayashi, K.; Dailey, D.L.; Chimenti, R.; Frey-Law, L.A.; Sluka, K.A.; Berardi, G. A Comparison of Pain, Fatigue, and Function between Post-COVID-19 Condition, Fibromyalgia, and Chronic Fatigue Syndrome: A Survey Study. Pain 2022, 164, 385–401. [Google Scholar] [CrossRef]
- Lazaridou, A.; Paschali, M.; Vilsmark, E.S.; Wilkins, T.; Napadow, V.; Edwards, R. The Impact of COVID-19 Pandemic on Mental and Physical Wellbeing in Women with Fibromyalgia: A Longitudinal Mixed-Methods Study. BMC Womens Health 2022, 22, 267. [Google Scholar] [CrossRef]
- Martínez, E.; Aira, Z.; Buesa, I.; Aizpurua, I.; Rada, D.; Azkue, J.J. Embodied Pain in Fibromyalgia: Disturbed Somatorepresentations and Increased Plasticity of the Body Schema. PLoS ONE 2018, 13, e0194534. [Google Scholar] [CrossRef]
- Lotze, M.; Moseley, G.L. Role of Distorted Body Image in Pain. Curr. Rheumatol. Rep. 2007, 9, 488–496. [Google Scholar] [CrossRef] [PubMed]
- Parsons, L.M. Integrating Cognitive Psychology, Neurology and Neuroimaging. Acta Psychol. 2001, 107, 155–181. [Google Scholar] [CrossRef] [PubMed]
- Haggard, P.; Iannetti, G.D.; Longo, M.R. Spatial Sensory Organization and Body Representation in Pain Perception. Curr. Biol. 2013, 23, R164–R176. [Google Scholar] [CrossRef]
- Tsay, A.; Allen, T.J.; Proske, U.; Giummarra, M.J. Sensing the Body in Chronic Pain: A Review of Psychophysical Studies Implicating Altered Body Representation. Neurosci. Biobehav. Rev. 2015, 52, 221–232. [Google Scholar] [CrossRef] [PubMed]
- Wand, B.M.; Parkitny, L.; O’Connell, N.E.; Luomajoki, H.; McAuley, J.H.; Thacker, M.; Moseley, G.L. Cortical Changes in Chronic Low Back Pain: Current State of the Art and Implications for Clinical Practice. Man. Ther. 2011, 16, 15–20. [Google Scholar] [CrossRef]
- Anderson, W.S.; Weiss, N.; Lawson, H.C.; Ohara, S.; Rowland, L.; Lenz, F.A. Demonstration of Motor Imagery Movement and Phantom Movement-Related Neuronal Activity in Human Thalamus. Neuroreport 2011, 22, 88–92. [Google Scholar] [CrossRef]
- Nijs, J.; Daenen, L.; Cras, P.; Struyf, F.; Roussel, N.; Oostendorp, R.A.B. Nociception Affects Motor Output: A Review on Sensory-Motor Interaction with Focus on Clinical Implications. Clin. J. Pain 2012, 28, 175–181. [Google Scholar] [CrossRef]
- Moseley, G.L. Why Do People with Complex Regional Pain Syndrome Take Longer to Recognize Their Affected Hand? Neurology 2004, 62, 2182–2186. [Google Scholar] [CrossRef] [PubMed]
- Moseley, G.L.; Flor, H. Targeting Cortical Representations in the Treatment of Chronic Pain. Neurorehabil. Neural Repair 2012, 26, 646–652. [Google Scholar] [CrossRef]
- Bray, H.; Moseley, G.L. Disrupted Working Body Schema of the Trunk in People with Back Pain. Br. J. Sports Med. 2011, 45, 168–173. [Google Scholar] [CrossRef]
- Bowering, K.J.; Butler, D.S.; Fulton, I.J.; Moseley, G.L. Motor Imagery in People with a History of Back Pain, Current Back Pain, Both, or Neither. Clin. J. Pain 2014, 30, 1070–1075. [Google Scholar] [CrossRef]
- von Piekartz, H.; Wallwork, S.B.; Mohr, G.; Butler, D.S.; Moseley, G.L. People with Chronic Facial Pain Perform Worse than Controls at a Facial Emotion Recognition Task, but It Is Not All about the Emotion. J. Oral Rehabil. 2015, 42, 243–250. [Google Scholar] [CrossRef]
- Wallwork, S.B.; Butler, D.S.; Fulton, I.; Stewart, H.; Darmawan, I.; Moseley, G.L. Left/Right Neck Rotation Judgments Are Affected by Age, Gender, Handedness and Image Rotation. Man. Ther. 2013, 18, 225–230. [Google Scholar] [CrossRef] [PubMed]
- Breckenridge, J.D.; McAuley, J.H.; Butler, D.S.; Stewart, H.; Moseley, G.L.; Ginn, K.A. The Development of a Shoulder Specific Left/Right Judgement Task: Validity & Reliability. Musculoskelet. Sci. Pract. 2017, 28, 39–45. [Google Scholar] [PubMed]
- Schmid, A.B.; Coppieters, M.W. Left/Right Judgment of Body Parts Is Selectively Impaired in Patients with Unilateral Carpal Tunnel Syndrome. Clin. J. Pain 2012, 28, 615–622. [Google Scholar] [CrossRef] [PubMed]
- Reinersmann, A.; Haarmeyer, G.S.; Blankenburg, M.; Frettlöh, J.; Krumova, E.K.; Ocklenburg, S.; Maier, C. Left Is Where the L Is Right. Significantly Delayed Reaction Time in Limb Laterality Recognition in Both CRPS and Phantom Limb Pain Patients. Neurosci. Lett. 2010, 486, 240–245. [Google Scholar] [CrossRef] [PubMed]
- Linder, M.; Michaelson, P.; Röijezon, U. Laterality Judgments in People with Low Back Pain—A Cross-Sectional Observational and Test-Retest Reliability Study. Man. Ther. 2016, 21, 128–133. [Google Scholar] [CrossRef]
- Ravat, S.; Olivier, B.; Gillion, N.; Lewis, F. Laterality Judgment Performance between People with Chronic Pain and Pain-Free Individuals. A Systematic Review and Meta-Analysis. Physiother. Theory Pract. 2020, 36, 1279–1299. [Google Scholar] [CrossRef]
- Elsig, S.; Luomajoki, H.; Sattelmayer, M.; Taeymans, J.; Tal-Akabi, A.; Hilfiker, R. Sensorimotor Tests, Such as Movement Control and Laterality Judgment Accuracy, in Persons with Recurrent Neck Pain and Controls. A Case-Control Study. Man. Ther. 2014, 19, 555–561. [Google Scholar] [CrossRef]
- Lee, W.J.; Choi, S.-H.; Jang, J.H.; Moon, J.Y.; Kim, Y.C.; Noh, E.; Shin, J.E.; Shin, H.; Kang, D.-H. Different Patterns in Mental Rotation of Facial Expressions in Complex Regional Pain Syndrome Patients. Medicine 2017, 96, e7990. [Google Scholar] [CrossRef] [PubMed]
- Pedler, A.; Motlagh, H.; Sterling, M. Laterality Judgments Are Not Impaired in Patients with Chronic Whiplash Associated Disorders. Man. Ther. 2013, 18, 72–76. [Google Scholar] [CrossRef] [PubMed]
- Martínez, E.; Guillen, V.; Buesa, I.; Azkue, J.J. A Distorted Body Schema and Susceptibility to Experiencing Anomalous Somatosensory Sensations in Fibromyalgia Syndrome. Clin. J. Pain 2019, 35, 887–893. [Google Scholar] [CrossRef] [PubMed]
- Riquelme-Aguado, V.; Gil-Crujera, A.; Fernández-Carnero, J.; Cuenca-Martínez, F.; Gómez Esquer, F. Limb Laterality Discrimination, Evoked Sensations and Somatosensory Behavior in Fibromyalgia Syndrome: A Cross-Sectional Study. Appl. Sci. 2022, 12, 7495. [Google Scholar] [CrossRef]
- Rivera, J.; González, T. The Fibromyalgia Impact Questionnaire: A Validated Spanish Version to Assess the Health Status in Women with Fibromyalgia. Clin. Exp. Rheumatol. 2004, 22, 554–560. [Google Scholar]
- Casanueva, B.; García-Fructuoso, F.; Belenguer, R.; Alegre, C.; Moreno-Muelas, J.V.; Hernández, J.L.; Pina, T.; González-Gay, M.Á. The Spanish Version of the 2010 American College of Rheumatology Preliminary Diagnostic Criteria for Fibromyalgia: Reliability and Validity Assessment. Clin. Exp. Rheumatol. 2016, 34, S55–S58. [Google Scholar]
- Saimpont, A.; Pozzo, T.; Papaxanthis, C. Aging Affects the Mental Rotation of Left and Right Hands. PLoS ONE 2009, 4, e6714. [Google Scholar] [CrossRef]
- Ryde-Brandt, B. Anxiety and Depression in Mothers of Children with Psychotic Disorders and Mental Retardation. Br. J. Psychiatry 1990, 156, 118–121. [Google Scholar] [CrossRef]
- Herrero, M.J.; Blanch, J.; Peri, J.M.; de Pablo, J.; Pintor, L.; Bulbena, A. A Validation Study of the Hospital Anxiety and Depression Scale (HADS) in a Spanish Population. Gen. Hosp. Psychiatry 2003, 25, 277–283. [Google Scholar] [CrossRef]
- Sanz, J. Adaptación Española Del Inventario Para La Depresión de Beck-II (BDI-II): 3. Propiedades Psicométricas En Pacientes Con Trastornos Psicológicos. Clín. Salud 2005, 16, 121–142. [Google Scholar]
- Olaya-Contreras, P.; Persson, T.; Styf, J. Comparison between the Beck Depression Inventory and Psychiatric Evaluation of Distress in Patients on Long-Term Sick Leave Due to Chronic Musculoskeletal Pain. J. Multidiscip. Healthc. 2010, 3, 161–167. [Google Scholar] [CrossRef]
- Choi, Y.; Mayer, T.G.; Williams, M.J.; Gatchel, R.J. What Is the Best Screening Test for Depression in Chronic Spinal Pain Patients? Spine J. 2014, 14, 1175–1182. [Google Scholar] [CrossRef]
- Wiebe, J.S.; Penley, J.A. A Psychometric Comparison of the Beck Depression Inventory-II in English and Spanish. Psychol. Assess. 2005, 17, 481–485. [Google Scholar] [CrossRef]
- García Campayo, J.; Rodero, B.; Alda, M.; Sobradiel, N.; Montero, J.; Moreno, S. Validación de La Versión Española de La Escala de La Catastrofización Ante El Dolor (Pain Catastrophizing Scale) En La Fibromialgia. Med. Clin. 2008, 131, 487–493. [Google Scholar] [CrossRef]
- Larsson, C.; Ekvall Hansson, E.; Sundquist, K.; Jakobsson, U. Kinesiophobia and Its Relation to Pain Characteristics and Cognitive Affective Variables in Older Adults with Chronic Pain. BMC Geriatr 2016, 16, 128. [Google Scholar] [CrossRef]
- Salvador, E.M.E.S.; Franco, K.F.M.; Miyamoto, G.C.; Dos Santos Franco, Y.R.; Cabral, C.M.N. Analysis of the Measurement Properties of the Brazilian-Portuguese Version of the Tampa Scale for Kinesiophobia-11 in Patients with Fibromyalgia. Braz. J. Phys. Ther. 2021, 25, 168–174. [Google Scholar] [CrossRef]
- Lami, M.J.; Martínez, M.P.; Miró, E.; Sánchez, A.I.; Guzmán, M.A. Catastrophizing, Acceptance, and Coping as Mediators Between Pain and Emotional Distress and Disability in Fibromyalgia. J. Clin. Psychol. Med. Settings 2018, 25, 80–92. [Google Scholar] [CrossRef]
- Ellingson, L.D.; Stegner, A.J.; Schwabacher, I.J.; Lindheimer, J.B.; Cook, D.B. Catastrophizing Interferes with Cognitive Modulation of Pain in Women with Fibromyalgia. Pain Med. 2018, 19, 2408–2422. [Google Scholar] [CrossRef]
- Morishita, T.; Miki, K.; Inoue, T. Penfield Homunculus and Recent Advances in Brain Mapping. World Neurosurg. 2020, 134, 515–517. [Google Scholar] [CrossRef]
- Ghimire, P.; Lavrador, J.P.; Baig Mirza, A.; Pereira, N.; Keeble, H.; Borri, M.; Furlanetti, L.; Brogna, C.; Jarosz, J.; Gullan, R.; et al. Intraoperative Mapping of Pre-Central Motor Cortex and Subcortex: A Proposal for Supplemental Cortical and Novel Subcortical Maps to Penfield’s Motor Homunculus. Brain Struct. Funct. 2021, 226, 1601–1611. [Google Scholar] [CrossRef]
- Timmers, I.; López-Solà, M.; Heathcote, L.C.; Heirich, M.; Rush, G.Q.; Shear, D.; Borsook, D.; Simons, L.E. Amygdala Functional Connectivity Mediates the Association between Catastrophizing and Threat-Safety Learning in Youth with Chronic Pain. Pain 2022, 163, 719–728. [Google Scholar] [CrossRef]
- Terry, E.L.; Tanner, J.J.; Cardoso, J.S.; Sibille, K.T.; Lai, S.; Deshpande, H.; Deutsch, G.; Price, C.C.; Staud, R.; Goodin, B.R.; et al. Associations between Pain Catastrophizing and Resting-State Functional Brain Connectivity: Ethnic/Race Group Differences in Persons with Chronic Knee Pain. J. Neurosci. Res. 2022, 100, 1047–1062. [Google Scholar] [CrossRef] [PubMed]
- Barber Foss, K.D.; Slutsky-Ganesh, A.B.; Diekfuss, J.A.; Grooms, D.R.; Simon, J.E.; Schneider, D.K.; Jayanthi, N.; Lamplot, J.D.; Hill, D.; Pombo, M.; et al. Brain Activity During Experimental Knee Pain and Its Relationship With Kinesiophobia in Patients With Patellofemoral Pain: A Preliminary Functional Magnetic Resonance Imaging Investigation. J. Sport Rehabil. 2022, 31, 589–598. [Google Scholar] [CrossRef] [PubMed]
- Ihara, N.; Wakaizumi, K.; Nishimura, D.; Kato, J.; Yamada, T.; Suzuki, T.; Hashiguchi, S.; Terasawa, Y.; Kosugi, S.; Morisaki, H. Aberrant Resting-State Functional Connectivity of the Dorsolateral Prefrontal Cortex to the Anterior Insula and Its Association with Fear Avoidance Belief in Chronic Neck Pain Patients. PLoS ONE 2019, 14, e0221023. [Google Scholar] [CrossRef] [PubMed]
- Etkin, A.; Egner, T.; Kalisch, R. Emotional Processing in Anterior Cingulate and Medial Prefrontal Cortex. Trends Cogn. Sci. 2011, 15, 85–93. [Google Scholar] [CrossRef]
| Measures | FMS (n = 30) | Pain-Free Controls (n = 30) | p Value Independent Samples Student’s t-Test |
|---|---|---|---|
| Age | 51.29 ± 7.22 | 49.71 ± 8.50 | 0.56 |
| Pain (NPRS) | 6.00 ± 2.00 | - | |
| FIQ | 86.88 ± 3.28 | - |
| Measures | FMS (n = 17) | Pain-Free Controls (n = 17) | p Value Samples Student’s t-test |
|---|---|---|---|
| ACC hands (20 images) | 84.85 ± 9.57 | 87.64 ± 6.92 | 0.377 |
| ACC hands (50 images) | 84.41 ± 7.11 | 88.82 ± 6.82 | 0.074 |
| RT hands (20 images) | 2.80 ± 1.15 | 2.11 ± 0.54 | 0.031 * |
| RT hands (50 images) | 3.01 ± 0.90 | 2.37 ± 0.45 | 0.013 * |
| ACC feet (20 images) | 89.55 ± 7.76 | 89.85 ± 4.87 | 0.896 |
| ACC feet (50 images) | 87.11 ± 8.12 | 89.29 ± 6.59 | 0.397 |
| RT feet (20 images) | 2.58 ± 1.27 | 2.00 ± 0.41 | 0.087 |
| RT feet (50 images) | 2.72 ± 1.14 | 2.25 ± 0.58 | 0.137 |
| Measures | FMS (n = 17) | Pain-Free Controls (n = 17) | p Value Independent Samples Student’s t-Test |
|---|---|---|---|
| Anxiety | 11.12 ± 2.54 | 7.88 ± 2.97 | 0.002 * |
| Depression | 38.35 ± 8,01 | 9.76 ± 2.19 | 0.000 * |
| Pain catastrophizing | 33.82 ± 3.43 | 14.76 ± 4.33 | 0.000 * |
| Helplessness | 16.06 ± 2.19 | 7.00 ± 2.44 | 0.000 * |
| Magnification | 6.65 ± 1.69 | 3.47 ± 1.54 | 0.000 * |
| Rumination | 11.12 ± 1.53 | 4.29 ± 2.31 | 0.000 * |
| Fear-related movement | 31.76 ± 3.73 | 14.53 ± 10.25 | 0.000 * |
| Measures | Anxiety | Depression | Pain Catastrophizing | Fear-Related Movement |
|---|---|---|---|---|
| ACC hands (20 images) | −0.43 | −0.42 | −0.08 | 0.02 |
| ACC hands (50 images) | −0.33 | −0.20 | 0.19 | 0.18 |
| RT hands (20 images) | −0.48 | −0.30 | −0.19 | −0.45 |
| RT hands (50 images) | −0.28 | −0.32 | −0.22 | 0.43 |
| ACC feet (20 images) | −0.03 | −0.04 | 0.06 | −0.04 |
| ACC feet (50 images) | −0.37 | −0.01 | −0.02 | 0.28 |
| RT feet (20 images) | −0.13 | −0.40 | 0.03 | −0.41 |
| RT feet (50 images) | 0.07 | −0.28 | 0.00 | −0.24 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Riquelme-Aguado, V.; Gil-Crujera, A.; Fernández-Carnero, J.; Cuenca-Martínez, F.; Klett, G.B.; Esquer, F.G. The Influence of Emotional and Cognitive Factors on Limb Laterality Discrimination in Women with Fibromyalgia Syndrome: A Cross-Sectional Study Secondary Analysis. Appl. Sci. 2023, 13, 1894. https://doi.org/10.3390/app13031894
Riquelme-Aguado V, Gil-Crujera A, Fernández-Carnero J, Cuenca-Martínez F, Klett GB, Esquer FG. The Influence of Emotional and Cognitive Factors on Limb Laterality Discrimination in Women with Fibromyalgia Syndrome: A Cross-Sectional Study Secondary Analysis. Applied Sciences. 2023; 13(3):1894. https://doi.org/10.3390/app13031894
Chicago/Turabian StyleRiquelme-Aguado, Víctor, Antonio Gil-Crujera, Josué Fernández-Carnero, Ferran Cuenca-Martínez, Guillermo Baviano Klett, and Francisco Gómez Esquer. 2023. "The Influence of Emotional and Cognitive Factors on Limb Laterality Discrimination in Women with Fibromyalgia Syndrome: A Cross-Sectional Study Secondary Analysis" Applied Sciences 13, no. 3: 1894. https://doi.org/10.3390/app13031894
APA StyleRiquelme-Aguado, V., Gil-Crujera, A., Fernández-Carnero, J., Cuenca-Martínez, F., Klett, G. B., & Esquer, F. G. (2023). The Influence of Emotional and Cognitive Factors on Limb Laterality Discrimination in Women with Fibromyalgia Syndrome: A Cross-Sectional Study Secondary Analysis. Applied Sciences, 13(3), 1894. https://doi.org/10.3390/app13031894








