Dentin Hypersensitivity: Etiology, Diagnosis and Contemporary Therapeutic Approaches—A Review in Literature
Abstract
:1. Introduction
2. Etiology of Dentin Hypersensitivity
3. Pain Mechanism of Dentin Hypersensitivity
4. Diagnosis of Dentin Hypersensitivity
4.1. Clinical Examination
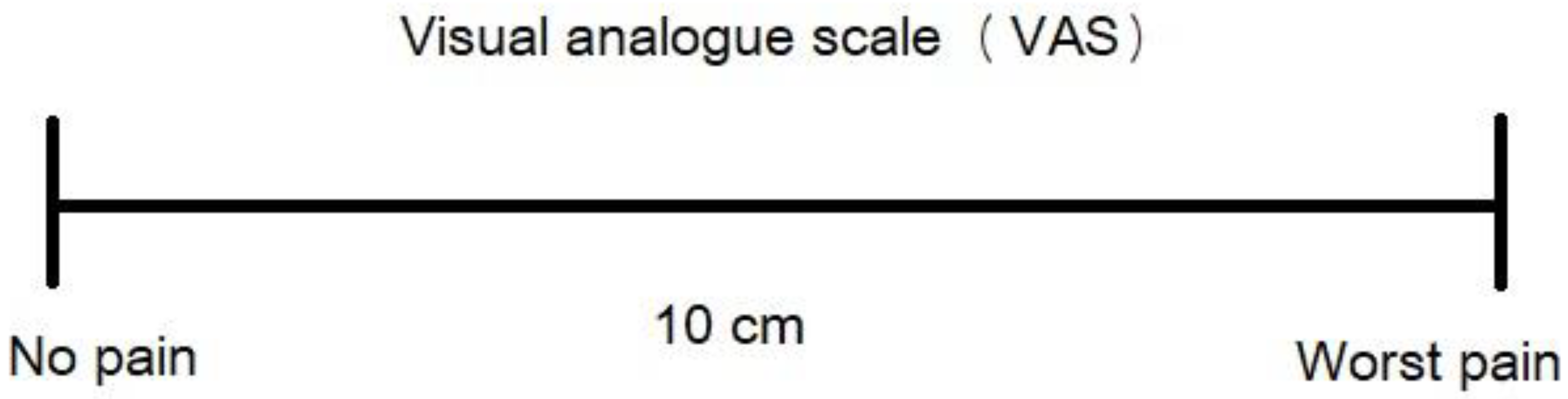
4.2. Assessment and Recording of Dentin Hypersensitivity
5. Dentin Hypersensitivity Assessment Methods
5.1. Response-Based Methods
- 0—The patient displays no response to the air stimulus.
- 1—The patient reacts to the air stimulus without requesting it to cease.
- 2—The patient reacts to the air stimulus and either requests it to stop or moves away from it.
- 3—The patient responds to the air stimulus, deeming it painful, and explicitly asks for it to stop.
5.2. Stimulus-Based Methods
6. Therapeutic Approaches of Dentin Hypersensitivity
- Accurate diagnosis subsequent to recording the patient’s dental history and initial clinical examination.
- Identification of etiological and predisposing factors, with particular attention to dietary and brushing habits and their potential impact on erosion and abrasion.
- Differential diagnosis to rule out other conditions presenting similar pain symptoms.
- Therapeutic intervention for any coexisting conditions manifesting symptoms similar to DH.
- Removal or mitigation of etiological and predisposing factors, accompanied by guidance on appropriate dietary habits and oral hygiene practices.
- Dispensing patient instructions or implementing office-based treatments in accordance with the patient’s requirements.
6.1. At-Home Therapeutic Treatments of Dentin Hypersensitivity
6.1.1. Potassium Salts
6.1.2. Sodium Fluoride
6.1.3. Stannous Fluoride
6.1.4. Arginine
6.1.5. Nano-Hydroxyapatite
6.1.6. Bioactive Glasses
6.1.7. Casein Phosphopeptide-Amorphous Calcium Phosphate (CPP-ACP)
6.1.8. Strontium Chloride
6.2. In-Office Therapeutic Treatments of Dentin Hypersensitivity
- Those that hinder nerve impulse transmission;
- Those that occlude dentin tubules (Figure 2).
| In-Office Treatments of Dentin Hypersensitivity | |||
|---|---|---|---|
| Active Agents | Form | Mechanism of Action | Duration of Therapy |
| Potassium salts | Gel | Impeding nerve impulse transmission | 2–3 times during a two-week period |
| Low-level laser irradiation (LLLT) | Irradiation | Impeding nerve impulse transmission | Case-sensitive |
| Sodium fluoride | Varnish or gel | Occlusion of dentin tubules | Once |
| Silver diamine fluoride (SDF) | Solution | Occlusion of dentin tubules | Once |
| Adhesive agents | Solution | Occlusion of dentin tubules | Once |
| Bioactive glass powder | Air abrasion | Occlusion of dentin tubules | Once |
| Portland cement | Paste | Occlusion of dentin tubules | Once |
| Oxalate salts | Gel or solution | Occlusion of dentin tubules | Once |
| Laser irradiation | Irradiation | Occlusion of dentin tubules | Once |
6.2.1. Potassium Salts Gels
6.2.2. Low-Level Laser Therapy
- Increased blood flow and microcirculation: LLLT has been shown to promote vasodilation and improve microcirculation in the treated area. This enhanced blood flow can facilitate the delivery of nutrients and oxygen to the tissues, aiding in their healing and reducing inflammation. Improved circulation can also help remove waste products and inflammatory mediators that contribute to pain.
- Stimulation of cellular activity: LLLT is thought to stimulate cellular activity, including the activation of mitochondria, the cell’s energy-producing organelles. This increased cellular energy can accelerate tissue repair and regeneration, promoting healing and reducing pain.
- Anti-inflammatory effects: LLLT is known to have anti-inflammatory effects by modulating immune responses and reducing the release of inflammatory cytokines. This can lead to a decrease in inflammation, which often accompanies pain in conditions like DH.
- Neurological modulation: LLLT may influence nerve function and sensitivity. It could modulate nerve conduction and reduce the transmission of pain signals. Additionally, LLLT might affect nerve endings and receptors, altering their response to stimuli.
- Endorphin release: LLLT might trigger the release of endorphins, which are natural pain-relieving chemicals produced by the body. Endorphins can help block pain signals and promote a sense of well-being.
- Stimulation of dentin remineralization: LLLT might encourage the remineralization of dentin, potentially sealing or occluding the tubules and reducing their sensitivity.
6.2.3. Sodium Fluoride
6.2.4. Silver Diamine Fluoride
6.2.5. Adhesive Agents
6.2.6. Air Abrasion with Bioactive Glass Particles
6.2.7. Portland Cement
6.2.8. Oxalate Salts
6.2.9. Laser Irradiation Treatments
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Dowell, P.; Addy, Μ.; Dummer, Ρ. Dentine hypersensitivity: Aetiology, differential diagnosis and management. Br. Dent. J. 1985, 158, 97–98. [Google Scholar] [CrossRef] [PubMed]
- Canadian Advisory Board on Dentin Hypersensitivity. Consensus-based recommendations for the diagnosis and management of dentin hypersensitivity. J. Can. Dent. Assoc. 2003, 69, 221–226. [Google Scholar]
- Holland, G.R.; Narhi, M.N.; Addy, M.; Gangarosa, L.; Orchardson, R. Guidelines for the design and conduct of clinical trials on dentine hypersensitivity. J. Clin. Periodontol. 1997, 24, 808–813. [Google Scholar] [CrossRef] [PubMed]
- Porto, I.C.; Andrade, A.K.; Montes, M.A. Diagnosis and treatment of dentinal hypersensitivity. J. Oral Sci. 2009, 51, 323–332. [Google Scholar] [CrossRef]
- Brännström, M. Etiology of dentin hypersensitivity. Proc. Finn. Dent. Soc. 1992, 88 (Suppl. 1), 7–14. [Google Scholar] [PubMed]
- Ciucchi, B.; Bouillaguet, S.; Holz, J.; Pashley, D.H. Dentinal fluid dynamics in human teeth, in vivo. J. Endod. 1995, 21, 191–194. [Google Scholar] [CrossRef]
- Gernhardt, C.R. How valid and applicable are current diagnostic criteria and assessment methods for dentin hypersensitivity? An overview. Clin. Oral Investig. 2013, 17 (Suppl. 1), S31–S40. [Google Scholar] [CrossRef]
- West, N.X. Dentine hypersensitivity: Preventive and therapeutic approaches to treatment. Periodontology 2000, 48, 31–41. [Google Scholar] [CrossRef]
- Trushkowsky, R.D.; Oquendo, A. Treatment of dentin hypersensitivity. Dent. Clin. N. Am. 2011, 55, 599–608. [Google Scholar] [CrossRef] [PubMed]
- Bak, S.; Kim, J.Y.; Chung, B.; Kim, J.; Cho, M.J.; Ha, W.; Lee, K.H.; Jung, Y. A randomized controlled clinical evaluation of desensitization efficacy of a newly developed toothpaste with highly stabilized SnF2. Am. J. Dent. 2023, 36, 183–187. [Google Scholar]
- Jang, J.H.; Oh, S.; Kim, H.J.; Kim, D.S. A randomized clinical trial for comparing the efficacy of desensitizing toothpastes on the relief of dentin hypersensitivity. Sci. Rep. 2023, 13, 5271. [Google Scholar] [CrossRef] [PubMed]
- Arshad, S.; Zaidi, S.J.A.; Farooqui, W.A. Comparative efficacy of BioMin-F, Colgate Sensitive Pro-relief and Sensodyne Rapid Action in relieving dentin hypersensitivity: A randomized controlled trial. BMC Oral Health 2021, 21, 498. [Google Scholar] [CrossRef] [PubMed]
- West, N.X. Dentine hypersensitivity. Monogr. Oral Sci. 2006, 20, 173–189. [Google Scholar] [PubMed]
- Addy, M.; West, Ν. Etiology, mechanisms, and management of dentine hypersensitivity. Curr. Opin. Periodontol. 1994, 1, 71–77. [Google Scholar]
- Absi, E.G.; Addy, M.; Adams, D. Dentine hypersensitivity. A study of the patency of dentinal tubules in sensitive and non-sensitive cervical dentine. J. Clin. Periodontol. 1987, 14, 280–284. [Google Scholar] [CrossRef]
- Absi, E.G.; Addy, M.; Adams, D. Dentine hypersensitivity. The development and evaluation of a replica technique to study sensitive and non-sensitive cervical dentine. J. Clin. Periodontol. 1989, 16, 190–195. [Google Scholar] [CrossRef]
- Zero, D.T.; Lussi, A. Erosion—chemical and biological factors of importance to the dental practitioner. Int. Dent. J. 2005, 55, 285–290. [Google Scholar] [CrossRef]
- Pashley, D.H.; Tay, F.R. Pulpodentin complex. In Seltzer and Bender’s Dental Pulp, 2nd ed.; Hargreaves, K.M., Goodis, H.E., Tay, F.R., Eds.; Quintessence Publishing Co.: Chicago, IL, USA, 2012; pp. 47–66. [Google Scholar]
- Pashley, D.H.; Livingston, M.J.; Greenhill, J.D. Regional resistances to fluid flow in human dentine in vitro. Arch. Oral Biol. 1978, 23, 807–810. [Google Scholar] [CrossRef]
- Pashley, D.H. How can sensitive dentine become hypersensitive and can it be reversed? J. Dent. 2013, 41, S49–S55. [Google Scholar] [CrossRef]
- Carrilho, M.R.; Tay, F.R.; Sword, J.; Donnelly, A.; Agee, K.A.; Nishitani, Y.; Sadek, F.T.; Carvalho, R.M.; Pashley, D.H. Dentine sealing provided by smear layer/smear plugs vs. adhesive resins/resin tags. Eur. J. Oral Sci. 2007, 115, 321–329. [Google Scholar] [CrossRef]
- West, N.X.; Lussi, A.; Seong, J.; Hellwig, E. Dentin hypersensitivity: Pain mechanisms and aetiology of exposed cervical dentin. Clin. Oral Investig. 2013, 17 (Suppl. 1), S9–S19. [Google Scholar] [CrossRef] [PubMed]
- Nähri, M.; Jyväsjärvi, E.; Virtannen, A. Role of intradental A and C- type fibres in dental pain mechanisms. Proc. Finn. Dent. Soc. 1992, 88 (Suppl. 1), 507–516. [Google Scholar]
- Matthews, B.; Andrew, D.; Wanachantararak, S. Biology of the Dental Pulp with Special Reference to Its Vasculature and Innervation. In Tooth Wear and Sensitivity; Embery, G., Edgar, W.M., Orchardson, R., Addy, M., Eds.; Taylor and Francis: London, UK, 2000; pp. 39–51. [Google Scholar]
- Michelich, V.; Schuster, G.S.; Pashley, D.H. Bacterial penetration of human dentin in vitro. J. Dent. Res. 1980, 59, 1398–1403. [Google Scholar] [CrossRef]
- Pashley, D.H.; Matthew, W.G. The effects of outward forced corrective flow on inward diffusion in human dentin in vitro. Arch. Oral Biol. 1993, 38, 577–582. [Google Scholar] [CrossRef]
- Kerns, D.G.; Scheidt, M.J.; Pashley, D.H.; Homer, J.A.; Strong, S.L.; Van Dyke, T.E. Dentinal tubule occlusion and root hypersensitivity. J. Periodontol. 1991, 62, 421–428. [Google Scholar] [CrossRef]
- Caviedes-Bucheli, J.; Munoz, H.R.; Azuero-Holguin, M.M.; Ulate, E. Neuropeptides in dental pulp: The silent protagonists. J. Endod. 2008, 34, 773–788. [Google Scholar] [CrossRef] [PubMed]
- Byers, M.; Närhi, M.V.O. Dental injury models: Experimental tools for understanding neuroinflammatory nociceptor functions. Crit. Rev. Oral Biol. Med. 1999, 10, 4–39. [Google Scholar] [CrossRef] [PubMed]
- Byers, M.; Henry, M.A.; Närhi, M.V.O. Dental innervation and its response to tooth injury. In Seltzer and Bender’s Dental Pulp, 2nd ed.; Hargreaves, K.M., Goodis, H.E., Tay, F.R., Eds.; Quintessence Publishing Co.: Chicago, IL, USA, 2012; pp. 133–157. [Google Scholar]
- Renton, T.; Yiangou, Y.; Plumpton, C.; Tate, S.; Bountra, C.; Anand, P. Sodium channel Nav1.8 immunoreactivity in painful human dental pulp. BMC Oral Health 2005, 5, 5. [Google Scholar] [CrossRef]
- Lundy, T.; Stanley, H.R. Correlation and pulpal histopathology and clinical symptoms in human teeth subjected to experimental irritation. Oral Surg. Oral Med. Oral Pathol. 1969, 27, 187–201. [Google Scholar] [CrossRef]
- Pashley, D.H.; Kepler, E.E.; Williams, E.C.; Okabe, A. Progressive decrease in dentine permeability following cavity preparation. Arch. Oral Biol. 1983, 28, 853–858. [Google Scholar] [CrossRef]
- Pashley, D.H.; Galloway, S.E.; Stewart, F.P. Effects of fibrinogen in vivo on dentin permeability in the dog. Arch. Oral Biol. 1984, 29, 725–728. [Google Scholar] [CrossRef] [PubMed]
- Hahn, C.L.; Overton, B. The effects of immunoglobulins on the convective permeability of human dentine in vitro. Arch. Oral Biol. 1997, 42, 835–843. [Google Scholar] [CrossRef]
- Gillam, D.G.; Orchardson, R.; Närhi, M.V.O.; Kontturi-Närhi, V. Present and future methods for the evaluation of pain associated with dentine hypersensitivity. In Tooth Wear and Sensitivity; Addy, M., Embery, G., Edgar, W.M., Orchardson, R., Eds.; Martin Dunitz Ltd.: London, UK, 2000; pp. 283–497. [Google Scholar]
- Miglani, S.; Aggarwal, V.; Ahuja, Β. Dentin hypersensitivity: Recent trends in management. J. Conserv. Dent. 2010, 13, 218–224. [Google Scholar] [CrossRef]
- O’Toole, S.; Bartlett, D. The relationship between dentine hypersensitivity, dietary acid intake and erosive tooth wear. J. Dent. 2017, 67, 84–87. [Google Scholar] [CrossRef]
- Bamise, C.T.; Olusile, A.O.; Oginni, A.O. An analysis of the etiological and predisposing factors related to dentin hypersensitivity. J. Contemp. Dent. Pract. 2008, 9, 52–59. [Google Scholar]
- Boiko, O.V.; Baker, S.R.; Gibson, B.J.; Locker, D.; Sufi, F.; Barlow, A.P.S.; Robinson, P.G. Construction and validation of the quality of life measure for dentine hypersensitivity (DHEQ). J. Clin. Periodontol. 2010, 37, 973–980. [Google Scholar] [CrossRef]
- Slade, G.D.; Spencer, A.J. Development and evaluation of the Oral Health Impact Profile. Community Dent. Health 1994, 11, 3–11. [Google Scholar] [PubMed]
- John, M.T.; Miglioretti, D.L.; LeResche, L.; Koepsell, T.D.; Hujoel, P.; Micheelis, W. German short forms of the Oral Health Impact Profile. Community Dent. Oral Epidemiol. 2006, 34, 277–288. [Google Scholar] [CrossRef]
- Hassel, A.J.; Rolko, C.; Koke, U.; Leisen, J.; Rammelsberg, Ρ. A German version of the GOHAI. Community Dent. Oral Epidemiol. 2008, 36, 34–42. [Google Scholar] [CrossRef] [PubMed]
- Gillam, D.G.; Newman, H.N. Assessment of pain in cervical dentinal sensitivity studies. A review. J. Clin. Periodontol. 1993, 20, 383–394. [Google Scholar] [CrossRef]
- Ernst, Ε. Placebo: New insights into an old enigma. Drug. Discov. Today 2007, 12, 413–418. [Google Scholar] [CrossRef] [PubMed]
- McCarney, R.; Warner, J.; Iliffe, S.; van Haselen, R.; Griffin, M.; Fisher, P. The Hawthorne Effect: A randomized, controlled trial. BMC Med. Res. Μethodol. 2007, 7, 30. [Google Scholar] [CrossRef]
- Addy, M.; West, N.X.; Barlow, A.; Smith, S. Dentine hypersensitivity: Is there both stimulus and placebo responses in clinical trials? Int. J. Dent. Hyg. 2007, 5, 53–59. [Google Scholar] [CrossRef]
- Barsky, A.J.; Saintfort, R.; Rogers, M.P.; Borus, J.F. Nonspecific medication side effects and the nocebo phenomenon. JAMA 2002, 287, 622–627. [Google Scholar] [CrossRef] [PubMed]
- Cunha-Cruz, J.; Stout, J.R.; Heaton, L.J.; Wataha, J.C. Dentin hypersensitivity and oxalates: A systematic review. J. Dent. Res. 2011, 90, 304–310. [Google Scholar] [CrossRef]
- Gillam, D.G.; Aris, A.; Bulman, J.S.; Newman, H.N.; Ley, F. Dentine hypersensitivity in subjects recruited for clinical trials: Clinical evaluation prevalence and intra-oral distribution. J. Oral Rehabil. 2002, 29, 226–231. [Google Scholar] [CrossRef]
- Schiff, T.; Delgado, E.; Zhang, Y.P.; Cummins, D.; DeVizio, W.; Mateo, L.R. Clinical evaluation of the efficacy of an in-office desensitizing paste containing 8% arginine and calcium carbonate in providing instant and lasting relief of dentin hypersensitivity. Am. J. Dent. 2009, 22, 8A–15A. [Google Scholar]
- Kobler, A.; Kuss, O.; Schaller, H.G.; Gernhardt, C.R. Clinical effectiveness of a strontium chloride-containing desensitizing agent over 6 months: A randomized, double-blind, placebo-controlled study. Quintessence Int. 2008, 39, 321–325. [Google Scholar] [PubMed]
- Orchardson, R.; Coffins, W.J. Thresholds of hypersensitive teeth to 2 forms of controlled stimulation. J. Clin. Periodontol. 1987, 14, 68–73. [Google Scholar] [CrossRef] [PubMed]
- Hicks, C.L.; von Baeyer, C.L.; Spafford, P.A.; van Korlaar, I.; Goodenough, Β. The Faces Pain Scale-Revised: Toward a common metric in pediatric pain measurement. Pain 2001, 93, 173–183. [Google Scholar] [CrossRef] [PubMed]
- Melzack, R. The short-form McGill Pain Questionnaire. Pain 1987, 30, 191–197. [Google Scholar] [CrossRef]
- Schiff, T.; Delgado, E.; Zhang, Y.P.; DeVizio, W.; Cummins, D.; Mateo, L.R. The clinical effect of a single direct topical application of a dentifrice containing 8.0% arginine, calcium carbonate, and 1450 ppm fluoride on dentin hypersensitivity: The use of a cotton swab applicator versus the use of a fingertip. J. Clin. Dent. 2009, 20, 131–136. [Google Scholar]
- Petrou, I.; Heu, R.; Stranick, M.; Lavender, S.; Zaidel, L.; Cummins, D.; Sullivan, R.J.; Hsueh, C.; Gimzewski, J.K. A breakthrough therapy for dentin hypersensitivity: How dental products containing 8% arginine and calcium carbonate work to deliver effective relief of sensitive teeth. J. Clin. Dent. 2009, 20, 23–31. [Google Scholar] [PubMed]
- Hamlin, D.; Milliams, K.P.; Delgado, E.; Zhang, Y.P.; DeVizio, W.; Mateo, L.R. Clinical evaluation of the efficacy of a desensitizing paste containing 8% arginine and calcium carbonate for the in-office relief of dentin hypersensitivity associated with dental prophylaxis. Am. J. Dent. 2009, 22, 16A–20A. [Google Scholar] [PubMed]
- Walline, B.W.; Wagner, J.G.; Marx, D.B.; Reinhardt, R.A. Comparison of methods for measuring root and mucogingival sensitivity. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. 2000, 90, 641–646. [Google Scholar] [CrossRef]
- Dionysopoulos, D.; Papageorgiou, S.; Papadopoulos, C.; Davidopoulou, S.; Konstantinidis, A.; Tolidis, K. Effect of whitening toothpastes with different active agents on the abrasive wear of dentin following tooth brushing simulation. J. Funct. Biomater. 2023, 14, 268. [Google Scholar] [CrossRef]
- Eisenburger, M.; Addy, M. Erosion and attrition of human enamel in vitro part I: Interaction effects. J. Dent. 2002, 30, 341–347. [Google Scholar] [CrossRef] [PubMed]
- Amaechi, B.T.; Mathews, S.M.; Mensinkai, P.K. Effects of theobromine-containing toothpaste on dentin tubule occlusion in situ. Clin. Oral Investig. 2015, 19, 109–116. [Google Scholar] [CrossRef] [PubMed]
- Zhong, Y.; Liu, J.; Li, X.; Yin, W.; He, Τ.; Hu, D.; Liao, Y.; Yao, X.; Wang, Y. Effect of a novel bioactive glass-ceramic on dental tubule occlusion: An in vitro study. Aust. Dent. J. 2015, 60, 96–103. [Google Scholar] [CrossRef]
- Magno, M.B.; Nascimento, G.C.; Da Penha, N.K.; Pessoa, O.F.; Loretto, B.C.; Maia, L.C. Difference in effectiveness between strontium acetate and arginine-based toothpastes to relieve dentin hypersensitivity. A systematic review. Am. J. Dent. 2015, 28, 40–44. [Google Scholar]
- Cummins, D. Recent advances in dentin hypersensitivity: Clinically proven treatments for instant and lasting sensitivity relief. Am. J. Dent. 2010, 23, 3A–13A. [Google Scholar]
- Kopycka-Kedzierawski, D.T.; Meyerowitz, C.; Litaker, M.S.; Heft, M.W.; Tasgaonkar, N.; Day, M.R.; Porter-Williams, A.; Gordan, V.V.; Yardic, R.L.; Lawhorn, T.M.; et al. National Dental PBRN Collaborative Group. Management of dentin hypersensitivity by practitioners in The National Dental Practice-Based Research Network. J. Am. Dent. Assoc. 2017, 148, 728–736. [Google Scholar] [CrossRef]
- Peacock, J.M.; Orchardson, R. Effects of potassium ions on action potential conduction in A- and C-fibers of rat spinal nerves. J. Dent. Res. 1995, 74, 634–641. [Google Scholar] [CrossRef] [PubMed]
- Orchardson, R.; Gillam, D.G. Managing dentin hypersensitivity. J. Am. Dent. Assoc. 2006, 137, 990–998. [Google Scholar] [CrossRef]
- Aranha, A.C.; Pimenta, L.A.; Marchi, G.M. Clinical evaluation of desensitizing treatments for cervical dentin hypersensitivity. Braz. Oral Res. 2009, 23, 333–339. [Google Scholar] [CrossRef]
- Chu, C.H.; Lo, E.C.M. Dentin hypersensitivity: A review. Hong Kong Dent. J. 2010, 7, 15–22. [Google Scholar]
- Sharma, D.; McGuire, J.A.; Gallob, J.T.; Amini, P. Randomized clinical efficacy trial of potassium oxalate mouthrinse in relieving dentinal sensitivity. J. Dent. 2013, 41 (Suppl. 4), S40–S48. [Google Scholar] [CrossRef] [PubMed]
- Sowinski, J.A.; Battista, G.W.; Petrone, M.E.; Chaknis, P.; Zhang, Y.P.; DeVizio, W.; Volpe, A.R.; Proskin, H.M. A new desensitizing dentifrice-an 8-week clinical investigation. Compend. Contin. Educ. Dent. Suppl. 2000, 27, 11–16. [Google Scholar]
- Faller, R.V.; Eversole, S.L. Protective effects of SnF2—Part III. Mechanism of barrier layer attachment. Int. Dent. J. 2014, 64, 16–21. [Google Scholar] [CrossRef]
- Faller, R.V.; Eversole, S.L.; Tzeghai, G.E. Enamel protection: A comparison of marketed dentifrice performance against dental erosion. Am. J. Dent. 2011, 24, 205–210. [Google Scholar]
- Hines, D.; Xu, S.; Stranick, M.; Lavender, S.; Pilch, S.; Zhang, Y.P.; Sullivan, R.; Montesani, L.; Montesani, L.; Mateo, L.R.; et al. Effect of a stannous fluoride toothpaste on dentinal hypersensitivity: In vitro and clinical evaluation. J. Am. Dent. Assoc. 2019, 150, S47–S59. [Google Scholar] [CrossRef] [PubMed]
- Papazisi, N.; Dionysopoulos, D.; Naka, O.; Strakas, D.; Davidopoulou, S.; Tolidis, K. Efficiency of various tubular occlusion agents in human dentin after in-office tooth bleaching. J. Funct. Biomater. 2023, 14, 430. [Google Scholar] [CrossRef] [PubMed]
- Yan, B.; Yi, J.; Li, Y.; Chen, Y.; Shi, Z. Arginine-containing toothpastes for dentin hypersensitivity: Systematic review and meta-analysis. Quintessence Int. 2013, 44, 709–723. [Google Scholar] [PubMed]
- França, I.L.; Sallum, E.A.; Do Vale, H.F.; Casati, M.Z.; Sallum, A.W.; Stewart, B. Efficacy of a combined in office/home-use desensitizing system containing 8% arginine and calcium carbonate in reducing dentin hypersensitivity: An 8 week randomized clinical study. Am. J. Dent. 2015, 28, 45–50. [Google Scholar] [PubMed]
- Bae, J.H.; Kim, Y.K.; Myung, S.K. Desensitizing toothpaste versus placebo for dentine hypersensitivity: A systematic review and meta-analysis. J. Clin. Periodontol. 2015, 42, 131–141. [Google Scholar] [CrossRef] [PubMed]
- Dionysopoulos, D. New strategies for caries prevention. The role of alkali production in dental biofilms. ARC J. Dent. Sci. 2016, 1, 19–21. [Google Scholar]
- Champaiboon, C.; Ongphichetmetha, N.; Lertpimonchai, A. Bioactive glass and arginine dentifrices reduce root sensitivity during daily activities following non-surgical periodontal therapy: A randomized controlled trial. J. Periodontol. 2023; online ahead of print. [Google Scholar] [CrossRef] [PubMed]
- Low, S.B.; Allen, E.P.; Kontogiorgos, E.D. Reduction in dental hypersensitivity with nano-hydroxyapatite, potassium nitrate, sodium monoflurophosphate and antioxidans. Open Dent. J. 2015, 9, 92–97. [Google Scholar] [CrossRef]
- Gopinath, N.M.; John, J.; Nagappan, N.; Prabhu, S.; Kumar, E.S. Evaluation of dentifrice containing nano-hydroxyapatite for dentinal hypersensitivity: A randomized controlled trial. J. Int. Oral Health 2015, 7, 118–122. [Google Scholar]
- Vano, M.; Derchi, G.; Barone, A.; Covani, U. Effectiveness of nano-hydroxypatite toothpaste in reducing dentin hypersensitivity: A double-blind randomized controlled trial. Quintessence Int. 2014, 45, 703–711. [Google Scholar]
- Yuan, P.; Liu, S.; Lv, Y.; Liu, W.; Ma, W.; Xu, P. Effect of a dentifrice containing different particle sizes of hydroxyapatite on dentin tubule occlusion and aqueous Cr (VI) sorption. Int. J. Nanomed. 2019, 14, 5243–5256. [Google Scholar] [CrossRef]
- de Melo Alencar, C.; de Paula, B.L.F.; Guanipa Ortiz, M.I.; Baraúna Magno, M.; Martins Silva, C.; Cople Maia, L. Clinical efficacy of nano-hydroxyapatite in dentin hypersensitivity: A systematic review and meta-analysis. J. Dent. 2019, 82, 11–21. [Google Scholar] [CrossRef] [PubMed]
- Hill, R.G.; Chen, X.; Gillam, D.G. In vitro ability of a novel nanohydroxyapatite oral rinse to occlude dentine tubules. Int. J. Dent. 2015, 2015, 153284. [Google Scholar] [CrossRef] [PubMed]
- Lin, X.; Xie, F.; Ma, X.; Hao, Y.; Qin, H.; Long, J. Fabrication and characterization of dendrimer-functionalized nano-hydroxyapatite and its application in dentin tubule occlusion. J. Biomater. Sci. Polym. Ed. 2017, 28, 846–863. [Google Scholar] [CrossRef] [PubMed]
- Petrović, D.; Galić, D.; Seifert, D.; Lešić, N.; Smolić, M. Evaluation of bioactive glass treatment for dentin hypersensitivity: A systematic review. Biomedicines 2023, 11, 1992. [Google Scholar] [CrossRef]
- Cai, F.; Shen, P.; Morgan, M.V.; Reynolds, E.C. Remineralization of enamel subsurface lesions in situ by sugar-free lozenges containing casein phosphopeptide-amorphous calcium phosphate. Aust. Dent. J. 2003, 48, 240–243. [Google Scholar] [CrossRef]
- Ranjitkar, S.; Kaidonis, J.A.; Richards, L.C.; Townsend, G.C. The effect of CPPACP on enamel wear under severe erosive conditions. Arch. Oral Biol. 2009, 54, 527–532. [Google Scholar] [CrossRef]
- Poggio, C.; Lombardini, M.; Dagna, A.; Chiesa, M.; Bianchi, S. Protective effect on enamel demineralization of a CPP-ACP paste: An AFM in vitro study. J. Dent. 2009, 37, 949–954. [Google Scholar] [CrossRef]
- Pompeu, D.dS.; de Paula, B.L.F.; Barros, A.P.O.; Nunes, S.C.; Carneiro, A.M.P.; Araujo, J.L.N.; Silva, C.M. Combination of strontium chloride and photobiomodulation in the control of tooth sensitivity post-bleaching: A split-mouth randomized clinical trial. PLoS ONE 2021, 16, e0250501. [Google Scholar] [CrossRef]
- Durgalakshmi, D.; Ajay Rakkesh, R.; Kesavan, M.; Ganapathy, S.; Ajithkumar, T.G.; Karthikeyan, S.; Balakumar, S. Highly reactive crystalline-phase-embedded strontium-bioactive nanorods for multimodal bioactive applications. Biomater. Sci. 2018, 6, 1764–1776. [Google Scholar] [CrossRef]
- Saeki, K.; Marshall, G.W.; Gansky, S.A.; Parkinson, C.R.; Marshall, S.J. Strontium effects on root dentin tubule occlusion and nanomechanical properties. Dent. Mater. 2016, 32, 240–251. [Google Scholar] [CrossRef] [PubMed]
- Huang, M.; Hill, R.G.; Rawlinson, S.C. Strontium (Sr) elicits odontogenic differentiation of human dental pulp stem cells (hDPSCs): A therapeutic role for Sr in dentine repair? Acta Biomater. 2016, 38, 201–211. [Google Scholar] [CrossRef] [PubMed]
- Dedhiya, M.G.; Young, F.; Higuchi, W.I. Mechanism for the retardation of the acid dissolution rate of hydroxapatite by strontium. J. Dent. Res. 1973, 52, 1097–1109. [Google Scholar] [CrossRef]
- Ross, M.R. Hypersensitive teeth: Effect of strontium chloride in a compatible dentifrice. J. Periodontol. 1961, 32, 49–53. [Google Scholar] [CrossRef]
- Majji, P.; Murthy, K.R. Clinical efficacy of four interventions in the reduction of dentinal hypersensitivity: A 2-month study. Indian J. Dent. Res. 2016, 27, 477–482. [Google Scholar] [CrossRef] [PubMed]
- Lochaiwatana, Y.; Poolthong, S.; Hirata, I.; Okazaki, M.; Swasdison, S.; Vongsavan, N. The synthesis and characterization of a novel potassium chloride fluoridated hydroxyapatite varnish for treating dentin hypersensitivity. Dent. Mater. J. 2015, 34, 31–40. [Google Scholar] [CrossRef] [PubMed]
- Bamise, C.T.; Esan, T.A. Mechanisms and treatment approaches of dentine hypersensitivity: A literature review. Oral Health Prev. Dent. 2011, 9, 353–367. [Google Scholar]
- Orchardson, R.; Gillam, D.G. The efficacy of potassium salts as agents for treating dentin hypersensitivity. J. Orofac. Pain 2000, 14, 9–19. [Google Scholar]
- Papadopoulou, A.; Vourtsa, G.; Tolidis, K.; Koliniotou-Koumpia, E.; Gerasimou, P.; Strakas, D.; Kouros, P. Clinical evaluation of a fluoride gel, a low-level laser, and a resin varnish at the treatment of dentin hypersensitivity. Lasers Dent. Sci. 2019, 3, 129–135. [Google Scholar] [CrossRef]
- Lopes, A.O.; de Paula Eduardo, C.; Aranha, A.C.C. Evaluation of different treatment protocols for dentin hypersensitivity: An 18-month randomized clinical trial. Lasers Med. Sci. 2017, 32, 1023–1030. [Google Scholar] [CrossRef]
- Tabibzadeh, Z.; Fekrazad, R.; Esmaeelnejad, A.; Shadkar, M.M.; Khalili Sadrabad, Z.; Ghojazadeh, M. Effect of combined application of high- and low-intensity lasers on dentin hypersensitivity: A randomized clinical trial. J. Dent. Res. Dent. Clin. Dent.Prospects 2018, 12, 49–55. [Google Scholar] [CrossRef]
- Tolentino, A.B.; Zeola, L.F.; Fernandes, M.R.U.; Pannuti, C.M.; Soares, P.V.; Aranha, A.C.C. Photobiomodulation therapy and 3% potassium nitrate gel as treatment of cervical dentin hypersensitivity: A randomized clinical trial. Clin. Oral Investig. 2022, 26, 6985–6993. [Google Scholar] [CrossRef] [PubMed]
- Pandey, R.; Koppolu, P.; Kalakonda, B.; Lakshmi, B.V.; Mishra, A.; Reddy, P.K.; Bollepalli, A.C. Treatment of dentinal hypersensitivity using low-level laser therapy and 5% potassium nitrate: A randomized, controlled, three arm parallel clinical study. Int. J. Appl. Basic Med. Res. 2017, 7, 63–66. [Google Scholar] [CrossRef] [PubMed]
- Shan, Z.; Ji, J.; McGrath, C.; Gu, M.; Yang, Y. Effects of low-level light therapy on dentin hypersensitivity: A systematic review and meta-analysis. Clin. Oral Investig. 2021, 25, 6571–6595. [Google Scholar] [CrossRef] [PubMed]
- Machado, A.C.; Viana, Í.E.L.; Farias-Neto, A.M.; Braga, M.M.; de Paula Eduardo, C.; de Freitas, P.M.; Aranha, A.C.C. Is photobiomodulation (PBM) effective for the treatment of dentin hypersensitivity? A systematic review. Lasers Med. Sci. 2018, 33, 745–753. [Google Scholar] [CrossRef] [PubMed]
- Shah, F.A. Fluoride-containing bioactive glasses: Glass design, structure, bioactivity, cellular interactions and recent developments. Mater. Sci. Eng. C Mater. Biol. Appl. 2016, 58, 1279–1289. [Google Scholar] [CrossRef]
- Han, L.; Okiji, T. Dentin tubule occluding ability of dentin desensitizers. Am. J. Dent. 2015, 28, 90–94. [Google Scholar] [PubMed]
- Antoniazzi, R.P.; Machado, M.E.; Grellmann, A.P.; Santos, R.C.; Zanatta, F.B. Effectiveness of a desensitizing agent for topical and home use for dentin hypersensitivity: A randomized clinical trial. Am. J. Dent. 2014, 27, 251–257. [Google Scholar]
- Chen, C.L.; Parolia, A.; Pau, A.; Celerino de Moraes Porto, I.C. Comparative evaluation of effectiveness of desensitizing agents in dentine tubule occlusion using scanning electron microscopy. Aust. Dent. J. 2015, 60, 65–72. [Google Scholar] [CrossRef]
- Markowitz, K. A new treatment alternative for sensitive teeth: A desensitizing oral rinse. J. Dent. 2013, 41 (Suppl. 1), S1–S11. [Google Scholar] [CrossRef]
- Chaiwat, A.; Chunhacheevachaloke, E.; Kidkhunthod, P.; Pakawanit, P.; Ajcharanukul, O. Enamel remineralization and crystallization after fluoride iontophoresis. J. Dent. Res. 2023, 102, 402–411. [Google Scholar] [CrossRef]
- Chan, A.K.Y.; Tsang, Y.C.; Jiang, C.M.; Leung, K.C.M.; Lo, E.C.M.; Chu, C.H. Treating hypersensitivity in older adults with silver diamine fluoride: A randomised clinical trial. J. Dent. 2023, 136, 104616. [Google Scholar] [CrossRef]
- Kiesow, A.; Menzel, M.; Lippert, F.; Tanzer, J.M.; Milgrom, P. Dentin tubule occlusion by a 38% silver diamine fluoride gel: An in vitro investigation. BDJ Open 2022, 8, 1. [Google Scholar] [CrossRef] [PubMed]
- Piovesan, É.T.A.; Alves, J.B.; Ribeiro, C.D.P.V.; Massignan, C.; Bezerra, A.C.B.; Leal, S.C. Is silver diamine fluoride effective in reducing dentin hypersensitivity? A systematic review. J. Dent. Res. Dent. Clin. Dent. Prospects 2023, 17, 63–70. [Google Scholar] [CrossRef]
- Craig, G.G.; Knight, G.M.; McIntyre, J.M. Clinical evaluation of diamine silver fluoride/potassium iodide as a dentine desensitizing agent. A pilot study. Aust. Dent. J. 2012, 57, 308–311. [Google Scholar] [CrossRef] [PubMed]
- Duran, I.; Sengun, A. The long-term effectiveness of five current desensitizing products on cervical dentine sensitivity. J. Oral Rehabil. 2004, 31, 351–356. [Google Scholar] [CrossRef]
- Dondi dall’Orologio, G.; Lone, A.; Finger, W.J. Clinical evaluation of the role of glutardialdehyde in a one-bottle adhesive. Am. J. Dent. 2002, 15, 330–334. [Google Scholar]
- Forsback, A.P.; Areva, S.; Salonen, J.I. Mineralization of dentin induced by treatment with bioactive glass S53P4 in vitro. Acta Odontol. Scand. 2004, 62, 14–20. [Google Scholar] [CrossRef]
- Vollenweider, M.; Brunner, T.J.; Knecht, S.; Grass, R.N.; Zehnder, M.; Imfeld, T.; Stark, W.J. Remineralization of human dentin using ultrafine bioactive glass particles. Acta Biomater. 2007, 3, 936–943. [Google Scholar] [CrossRef] [PubMed]
- Bakry, A.S.; Marghalani, H.Y.; Amin, O.A.; Tagami, J. The effect of a bioglass paste on enamel exposed to erosive challenge. J. Dent. 2014, 42, 1458–1463. [Google Scholar] [CrossRef]
- Karaoulani, K.; Dionysopoulos, D.; Tolidis, K.; Kouros, P.; Konstantinidis, A.; Hill, R. Effect of air-abrasion pretreatment with three bioactive materials on enamel susceptibility to erosion by artificial gastric juice. Dent. Mater. 2022, 38, 1218–1231. [Google Scholar] [CrossRef]
- Lopez, T.C.; Diniz, I.M.; Ferreira, L.S.; Marchi, J.; Borges, R.; de Cara, S.P.; D’Almeida-Couto, R.; Marques, M.M. Bioactive glass plus laser phototherapy as promise candidates for dentine hypersensitivity treatment. J. Biomed. Mater. Res. B Appl. Biomater. 2017, 105, 107–116. [Google Scholar] [CrossRef] [PubMed]
- Chen, Q.; Thompson, I.; Boccaccini, A. 45S5 Bioglass®-derived glass–ceramic scaffolds for bone tissue engineering. Biomaterials 2006, 27, 2414–2425. [Google Scholar] [CrossRef]
- Behzadi, S.; Mohammadi, Y.; Rezaei-Soufi, L.; Farmany, A. Occlusion effects of bioactive glass and hydroxyapatite on dentinal tubules: A systematic review. Clin. Oral Investig. 2022, 26, 6061–6078. [Google Scholar] [CrossRef]
- Gandolfi, M.G.; Farascioni, S.; Pashley, D.H.; Gasparotto, G.; Prati, C. Calcium silicate coating derived from Portland cement as treatment for hypersensitive dentine. J. Dent. 2008, 36, 565–578. [Google Scholar] [CrossRef] [PubMed]
- Pillon, F.L.; Romani, I.G.; Schmidt, E.R. Effect of a 3% potassium oxalate topical application on dentinal hypersensitivity after subgingival scaling and root planning. J. Periodontol. 2004, 75, 1461–1464. [Google Scholar] [CrossRef] [PubMed]
- Vieira, A.H.; Passos, V.F.; de Assis, J.S.; Mendonça, J.S.; Santiago, S.L. Clinical evaluation of a 3% potassium oxalate gel and a GaAlAs laser for the treatment of dentinal hypersensitivity. Photomed. Laser Surg. 2009, 27, 807–812. [Google Scholar] [CrossRef] [PubMed]
- Pashley, D.H.; Andringa, H.J.; Eichmiller, F. Effects of ferric and aluminum oxalates on dentin permeability. Am. J. Dent. 1991, 4, 123–126. [Google Scholar] [PubMed]
- Peacock, J.M.; Orchardson, R. Action potential conduction block of nerves in vitro by potassium citrate, potassium tartrate and potassium oxalate. J. Clin. Periodontol. 1999, 26, 33–37. [Google Scholar] [CrossRef] [PubMed]
- Bader, J.; Balevi, B.; Farsai, P.; Flores-Mir, C.; Gunsolley, J.; Mathews, D.; Vig, K.; Zahrowsky, J. Laser may reduce pain arising from dentin hypersensitivity. J. Am. Dent. Assoc. 2014, 145, e1–e2. [Google Scholar] [CrossRef]
- Lopes, A.O.; Eduardo Cde, P.; Aranha, A.C. Clinical evaluation of low-power laser and a desensitizing agent on dentin hypersensitivity. Lasers Med. Sci. 2015, 30, 823–829. [Google Scholar] [CrossRef]
- Yilmaz, H.G.; Bayindir, H. Clinical and scanning electron microscopy of the Er,Cr:YSGG laser therapy for treating dentin hypersensitivity: A short-term, randomized, controlled study. J. Oral Rehabil. 2014, 41, 392–398. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.Y.; Jung, H.I.; Jung, B.Y.; Cho, Y.S.; Kwon, H.K.; Kim, B.I. Desensitizing efficacy of nano-carbonate apatite dentifrice and Er,Cr:YSGG laser: A randomized clinical trial. Photomed. Laser Surg. 2015, 33, 9–14. [Google Scholar] [CrossRef]
- Tunar, O.L.; Gürsoy, H.; Çakar, G.; Kuru, B.; Ipci, S.D.; Yilmaz, S. Evaluation of the effect of Er:YAG laser and desensitizing part containing 8% arginine and calcium carbonate and their combinations on human dentine tubules: A scanning electron microscopic analysis. Photomed. Laser Surg. 2014, 32, 540–545. [Google Scholar] [CrossRef]
- He, S.; Wang, Y.; Li, X.; Hu, D. Effectiveness of laser ther¬apy and topical desensitising agents in treating dentine hypersensitivity: A systematic review. J. Oral Rehabil. 2011, 38, 348–358. [Google Scholar] [CrossRef] [PubMed]
- Meng, Y.; Huang, F.; Wang, S.; Huang, X.; Lu, Y.; Li, Y.; Dong, Y.; Pei, D. Evaluation of dentinal tubule occlusion and pulp tissue response after using 980-nm diode laser for dentin hypersensitivity treatment. Clin. Oral Investig. 2023, 27, 4843–4854. [Google Scholar] [CrossRef]
- Khoubrouypak, Z.; Hasani Tabatabaei, M.; Chiniforush, N.; Moradi, Z. Evaluation of the effects of 810 nm diode laser alone and in combination with Gluma© and chromophore on dentinal tubule occlusion: A scanning electron microscopic analysis. J. Lasers Med. Sci. 2020, 11, 268–273. [Google Scholar] [CrossRef] [PubMed]
- Solati, M.; Fekrazad, R.; Vahdatinia, F.; Farmany, A.; Farhadian, M.; Hakimiha, N. Dentinal tubule blockage using nanobioglass in the presence of diode (980 nm) and Nd:YAG lasers: An in vitro study. Clin. Oral Investig. 2022, 26, 2975–2981. [Google Scholar] [CrossRef]
- Landmayer, K.; da Silva, J.C.V.; Anhesini, B.H.; Iatarola, B.O.; Aranha, A.C.C.; Francisconi-Dos-Rios, L.F. Effect of Nd:YAG laser irradiation, used as a desensitizing strategy, on bond strength to simulated hypersensitive dentin. Clin. Oral Investig. 2022, 26, 4109–4116. [Google Scholar] [CrossRef]
- Zhuang, H.; Liang, Y.; Xiang, S.; Li, H.; Dai, X.; Zhao, W. Dentinal tubule occlusion using Er:YAG Laser: An in vitro study. J. Appl. Oral Sci. 2021, 29, e20200266. [Google Scholar] [CrossRef]
- Forouzande, M.; Rezaei-Soufi, L.; Yarmohammadi, E.; Ganje-Khosravi, M.; Fekrazad, R.; Farhadian, M.; Farmany, A. Effect of sodium fluoride varnish, Gluma, and Er,Cr:YSGG laser in dentin hypersensitivity treatment: A 6-month clinical trial. Lasers Med. Sci. 2022, 37, 2989–2997. [Google Scholar] [CrossRef]
- Gholami, G.A.; Fekrazad, R.; Esmaiel-Nejad, A.; Kalhori, K.A. An evaluation of the occluding effects of Er;Cr:YSGG, Nd:YAG, CO₂ and diode lasers on dentinal tubules: A scanning electron microscope in vitro study. Photomed. Laser Surg. 2011, 29, 115–121. [Google Scholar] [CrossRef] [PubMed]
- Belal, M.H.; Yassin, A. A comparative evaluation of CO2 and erbium-doped yttrium aluminium garnet laser therapy in the management of dentin hypersensitivity and assessment of mineral content. J. Periodontal. Implant Sci. 2014, 44, 227–234. [Google Scholar] [CrossRef] [PubMed]
- Schwarz, F.; Arweiler, N.; Georg, T.; Reich, E. Desensitizing effects of an Er:YAG laser on hypersensitive dentine. J. Clin. Periodontol. 2002, 29, 211–215. [Google Scholar] [CrossRef] [PubMed]
- Aranha, A.C.; Eduardo Cde, P. Effects of Er:YAG and Er,Cr:YSGG lasers on dentine hypersensitivity. Short-term clinical evaluation. Lasers Med. Sci. 2012, 27, 813–818. [Google Scholar] [CrossRef]
- Aranha, A.C.; Eduardo Cde, P. In vitro effects of Er,Cr:YSGG laser on dentine hypersensitivity. Dentine permeability and scanning electron microscopy analysis. Lasers Med. Sci. 2012, 27, 827–834. [Google Scholar] [CrossRef] [PubMed]
- Femiano, F.; Femiano, L.; Grassia, V.; D’Apuzzo, F.; Nucci, L.; Femiano, R. Evaluation of pulp necrosis of a tooth with extensive restoration in absence of microleakages. J. Biol. Regul. Homeost. Agents 2019, 33, 1941–1944. [Google Scholar] [PubMed]


| At-Home Treatments for Dentin Hypersensitivity | |||
|---|---|---|---|
| Active Agents | Form | Mechanism of Action | Duration of Therapy |
| Potassium salts | Toothpaste–mouthwash | Impeding nerve impulse transmission | 2 times daily for 2–4 weeks |
| Sodium fluoride | Toothpaste–mouthwash | Occlusion of dentin tubules | 2 times daily for 2–4 weeks |
| Stannous fluoride | Toothpaste–gel | Occlusion of dentin tubules | 2 times daily up to 2 weeks |
| Arginine | Toothpaste–mouthwash | Occlusion of dentin tubules | 2 times daily for 2–4 weeks |
| Hydroxyapatite or nano-hydroxyapatite | Toothpaste–mouthwash | Occlusion of dentin tubules | 2 times daily for 2–4 weeks |
| Bioactive glass | Toothpaste–mouthwash | Occlusion of dentin tubules | 2 times daily for 2–4 weeks |
| CPP-ACP | Toothpaste–mousse | Occlusion of dentin tubules | 2 times daily for 2–4 weeks |
| Strontium chloride | Toothpaste–mouthwash | Occlusion of dentin tubules | 2 times daily for 2–4 weeks |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dionysopoulos, D.; Gerasimidou, O.; Beltes, C. Dentin Hypersensitivity: Etiology, Diagnosis and Contemporary Therapeutic Approaches—A Review in Literature. Appl. Sci. 2023, 13, 11632. https://doi.org/10.3390/app132111632
Dionysopoulos D, Gerasimidou O, Beltes C. Dentin Hypersensitivity: Etiology, Diagnosis and Contemporary Therapeutic Approaches—A Review in Literature. Applied Sciences. 2023; 13(21):11632. https://doi.org/10.3390/app132111632
Chicago/Turabian StyleDionysopoulos, Dimitrios, Olga Gerasimidou, and Charis Beltes. 2023. "Dentin Hypersensitivity: Etiology, Diagnosis and Contemporary Therapeutic Approaches—A Review in Literature" Applied Sciences 13, no. 21: 11632. https://doi.org/10.3390/app132111632
APA StyleDionysopoulos, D., Gerasimidou, O., & Beltes, C. (2023). Dentin Hypersensitivity: Etiology, Diagnosis and Contemporary Therapeutic Approaches—A Review in Literature. Applied Sciences, 13(21), 11632. https://doi.org/10.3390/app132111632








