HPLC with Post-Column Derivatization with Alizarin for Determination of OATD-02, an Anticancer Arginase Inhibitor in Clinical Development
Abstract
1. Introduction
2. Materials and Methods
2.1. Reagents
2.2. Instruments
2.3. HPLC and Derivatization Conditions
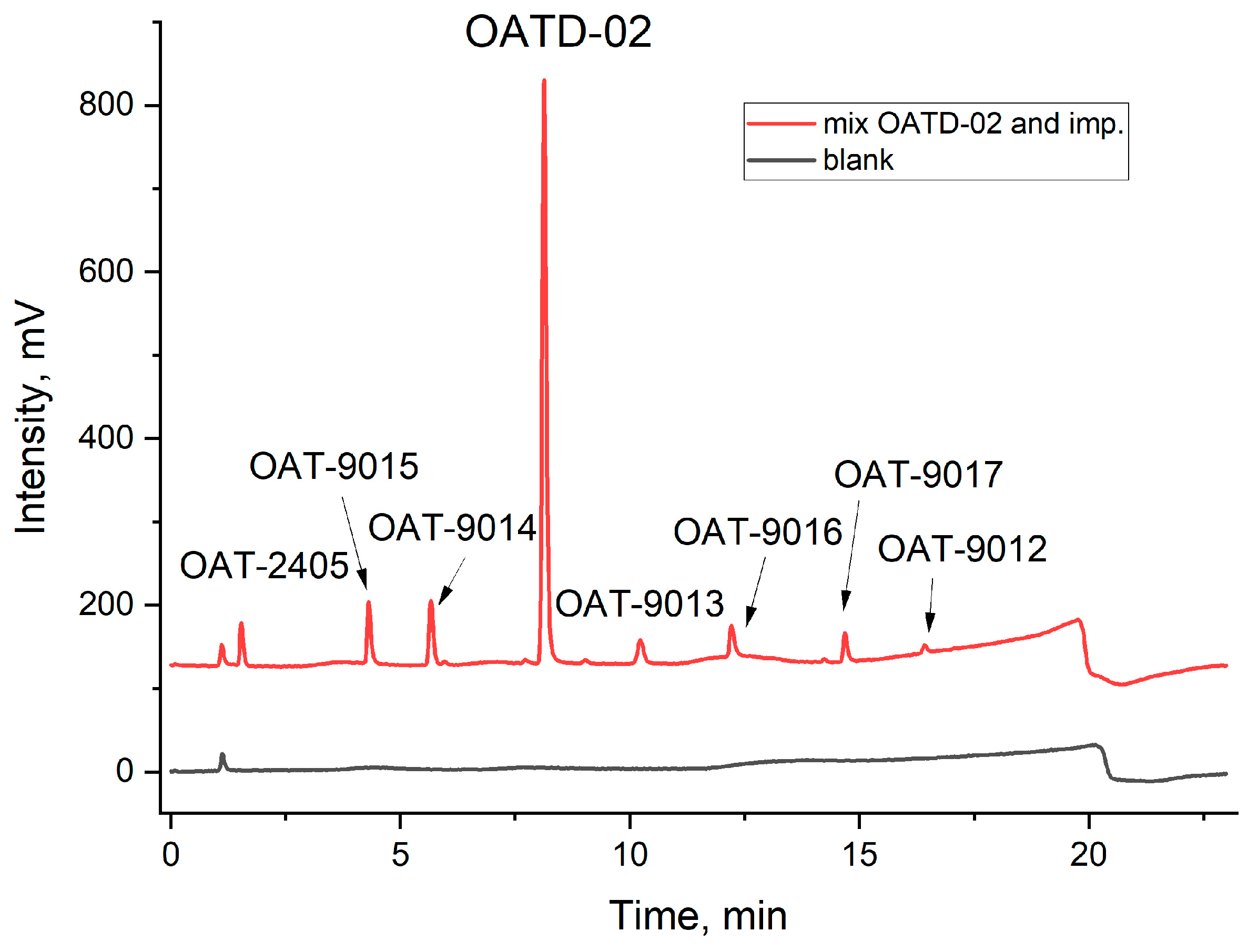
2.4. Method Development
2.5. Preparation of Sample Solution
2.6. Method Validation
3. Results
3.1. Optimal Conditions for HPLC
3.1.1. Effect of Mobile Phase pH on Retention of OATD-02
3.1.2. Optimization of Detection and Derivatization
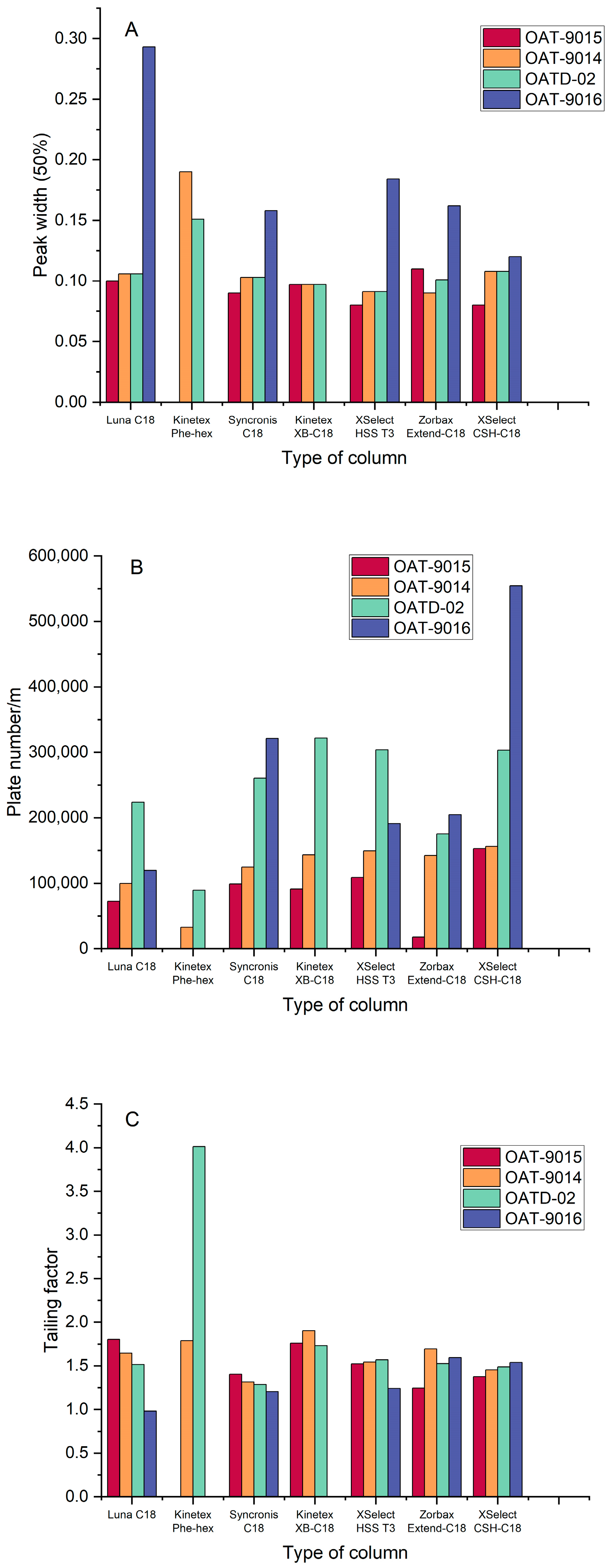
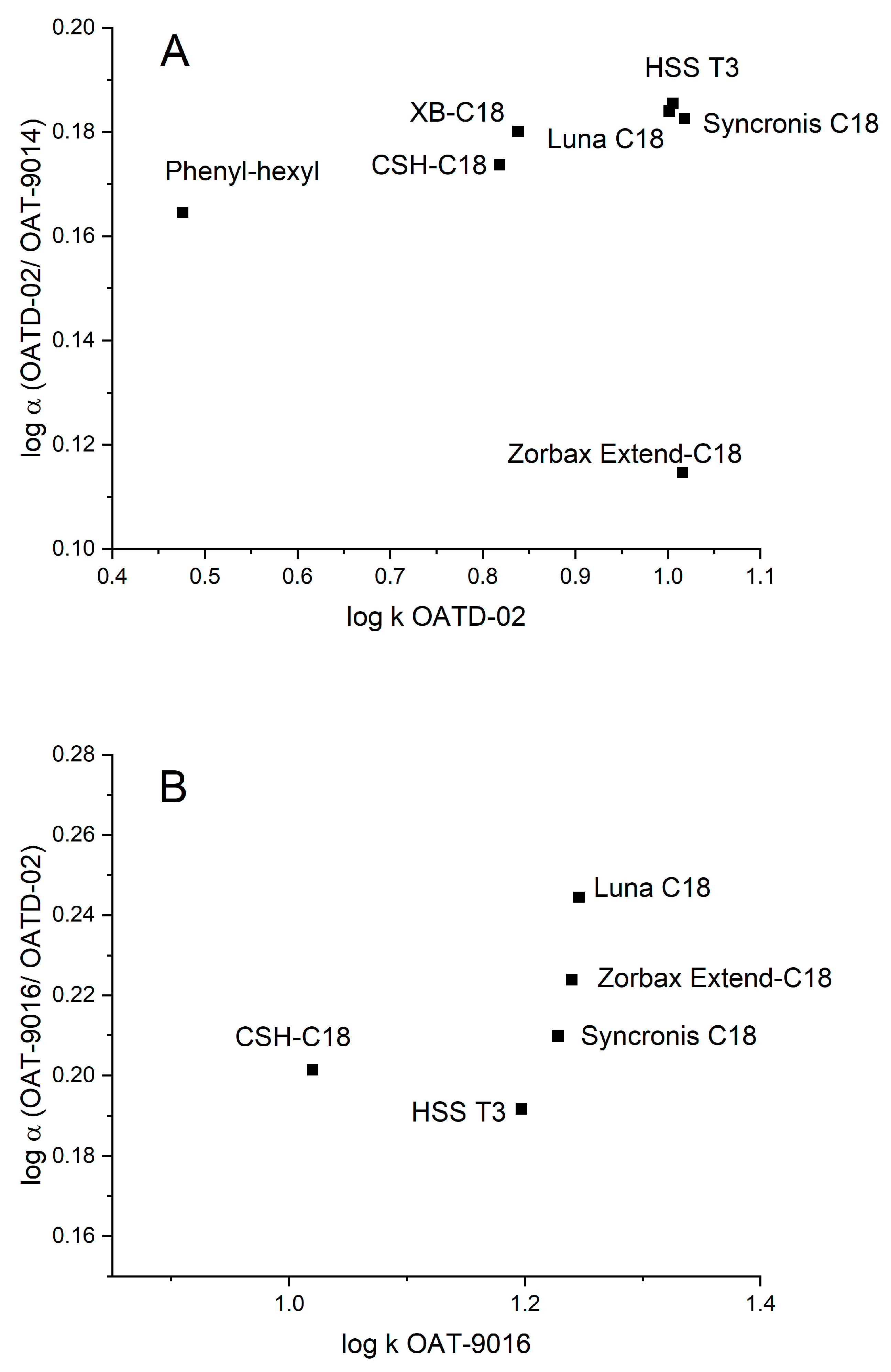
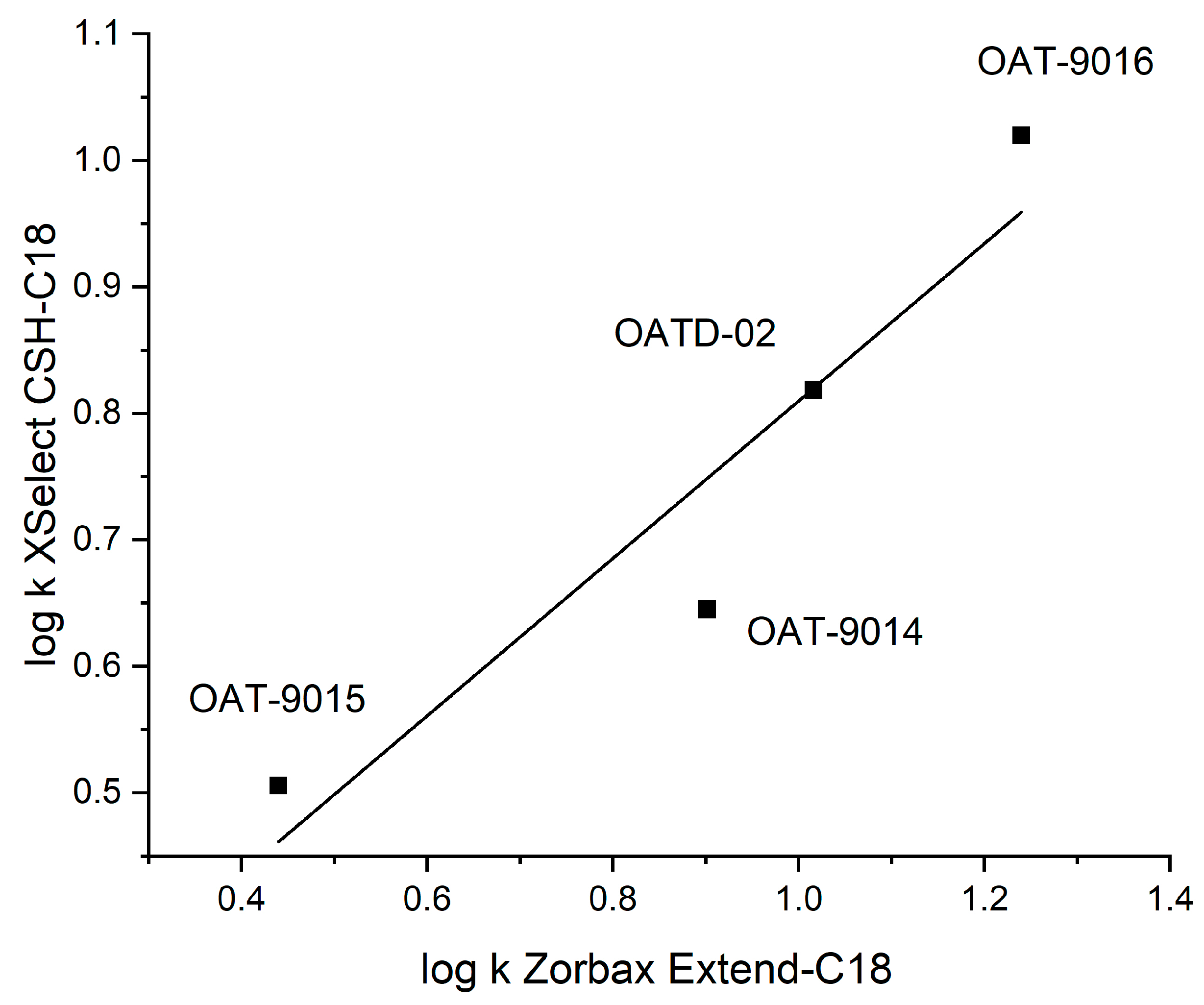
3.1.3. Influence of the Stationary Phase
3.2. Method Validation
4. Discussion
Derivatization Mechanism
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- ICH Harmonised Guideline, Analytical Procedure Development Q14. Available online: https://database.ich.org/sites/default/files/ICH_Q14_Document_Step2_Guideline_2022_0324.pdf (accessed on 29 March 2023).
- ICH Harmonised Tripartite Guideline, Validation of Analytical Procedures: Text and Methodology Q2(R1). Available online: https://database.ich.org/sites/default/files/Q2%28R1%29%20Guideline.pdf (accessed on 29 March 2023).
- Borek, B.; Nowicka, J.; Gzik, A.; Dziegielewski, M.; Jedrzejczak, K.; Brzezinska, J.; Grzybowski, M.; Stanczak, P.; Pomper, P.; Zagozdzon, A.; et al. Arginase 1/2 inhibitor OATD-02: From discovery to first-in-man setup in cancer immunotherapy. Mol. Cancer Ther. 2023, 22, 807–817. [Google Scholar] [CrossRef] [PubMed]
- Grzybowski, M.M.; Stańczak, P.S.; Pomper, P.; Błaszczyk, R.; Borek, B.; Gzik, A.; Nowicka, J.; Jędrzejczak, K.; Brzezińska, J.; Rejczak, T.; et al. OATD-02 Validates the Benefits of Pharmacological Inhibition of Arginase 1 and 2 in Cancer. Cancers 2022, 14, 3967. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, E.; Niijima, J.; Asakawa, N. Selective determination of potential impurities in an active pharmaceutical ingredient using HPLC-SPE-HPLC. J. Pharm. Biomed. Anal. 2013, 84, 41–47. [Google Scholar] [CrossRef] [PubMed]
- Wolfender, J.-L. HPLC in Natural Product Analysis: The Detection Issue. Planta Med. 2009, 75, 719–734. [Google Scholar] [CrossRef]
- Holzgrabe, U.; Nap, C.-J.; Beyer, T.; Almeling, S. Alternatives to amino acid analysis for the purity control of pharmaceutical grade L-alanine. J. Sep. Sci. 2010, 33, 2402–2410. [Google Scholar] [CrossRef]
- Liu, D.Q.; Sun, M.; Kord, A.S. Recent advances in trace analysis of pharmaceutical genotoxic impurities. J. Pharm. Biomed. Anal. 2010, 51, 999–1014. [Google Scholar] [CrossRef]
- McCrossen, S.; Bryant, D.; Cook, B.; Richards, J. Comparison of LC detection methods in the investigation of non-UV detectable organic impurities in a drug substance. J. Pharm. Biomed. Anal. 1998, 17, 455–471. [Google Scholar] [CrossRef]
- Bhateria, M.; Ramakrishna, R.; Pakala, D.B.; Bhatta, R.S. Development of an LC–MS/MS method for simultaneous determination of memantine and donepezil in rat plasma and its application to pharmacokinetic study. J. Chromatogr. B 2015, 1001, 131–139. [Google Scholar] [CrossRef] [PubMed]
- Hsieh, Y.; Brisson, J.-M.; Wang, G.; Ng, K.; Korfmacher, W.A. Simultaneous fast HPLC-MS/MS analysis of drug candidates and hydroxyl metabolites in plasma. J. Pharm. Biomed. Anal. 2003, 33, 251–261. [Google Scholar] [CrossRef]
- Wang, J.; Hu, X.; Tu, Y.; Ni, K. Determination of spectinomycin hydrochloride and its related substances by HPLC–ELSD and HPLC–MSn. J. Chromatogr. B 2006, 834, 178–182. [Google Scholar] [CrossRef]
- Abid, F.; Youssef, S.H.; Song, Y.; Parikh, A.; Trott, D.; Page, S.W.; Garg, S. Development and validation of a new analytical method for estimation of narasin using refractive index detector and its greenness evaluation. Microchem. J. 2022, 175, 107149. [Google Scholar] [CrossRef]
- Chepyala, D.; Tsai, I.-L.; Sun, H.-Y.; Lin, S.-W.; Kuo, C.-H. Development and validation of a high-performance liquid chromatography-fluorescence detection method for the accurate quantification of colistin in human plasma. J. Chromatogr. B 2015, 980, 48–54. [Google Scholar] [CrossRef] [PubMed]
- Cintrón, J.M.; Risley, D.S. Hydrophilic interaction chromatography with aerosol-based detectors (ELSD, CAD, NQAD) for polar compounds lacking a UV chromophore in an intravenous formulation. J. Pharm. Biomed. Anal. 2013, 78–79, 14–18. [Google Scholar] [CrossRef] [PubMed]
- Clarot, I.; Regazzeti, A.; Auzeil, N.; Laadani, F.; Citton, M.; Netter, P.; Nicolas, A. Analysis of neomycin sulfate and framycetin sulfate by high-performance liquid chromatography using evaporative light scattering detection. J. Chromatogr. A 2005, 1087, 236–244. [Google Scholar] [CrossRef] [PubMed]
- Gunsch, M.J.; Schwalm, E.L.; Ouimet, C.M.; Halsey, H.M.; Hamilton, S.E.; Bernardoni, F.; Jo, J. Development and validation of ion-pairing HPLC-CAD chromatography for measurement of Islatravir’s phosphorylated intermediates. J. Pharm. Biomed. Anal. 2022, 213, 114684. [Google Scholar] [CrossRef] [PubMed]
- Shen, X.; Yang, M.; Tomellini, S.A. Liquid Chromatographic Analysis of Glucosamine in Commercial Dietary Supplements Using Indirect Fluorescence Detection. J. Chromatogr. Sci. 2007, 45, 70–75. [Google Scholar] [CrossRef]
- Douša, M.; Břicháč, J.; Tkadlecová, M.; Man, S.; Zezula, J.; Hájíček, J.; Pekárek, T. A novel approach for HPLC determination of 2-cynaoacetamide using derivatization procedure with 2-hydroxyacetophenone as a new useful derivatization reagent. J. Pharm. Biomed. Anal. 2016, 128, 391–397. [Google Scholar] [CrossRef]
- Vervoort, N.; Daemen, D.; Török, G. Performance evaluation of evaporative light scattering detection and charged aerosol detection in reversed phase liquid chromatography. J. Chromatogr. A 2008, 1189, 92–100. [Google Scholar] [CrossRef]
- Dixon, R.W.; Peterson, D.S. Development and Testing of a Detection Method for Liquid Chromatography Based on Aerosol Charging. Anal. Chem. 2002, 74, 2930–2937. [Google Scholar] [CrossRef]
- Huang, Y.; Lu, H.; Li, Z.; Zeng, Y.; Xu, Q.; Wu, Y. Development of HPLC-CAD method for simultaneous quantification of nine related substances in ursodeoxycholic acid and identification of two unknown impurities by HPLC-Q-TOF-MS. J. Pharm. Biomed. Anal. 2023, 229, 115357. [Google Scholar] [CrossRef]
- Gamache, P.H.; McCarthy, R.S.; Freeto, S.M.; Asa, D.J.; Woodcock, M.J.; Laws, K.; Cole, R.O. HPLC Analysis of non-volatile analytes using charged aerosol detection. LC GC Europe 2005, 18, 345–354. [Google Scholar]
- Kulkarni, S.; Sawant, A.; Zinjad, P.; Sanatan, P.; Bhope, S.; Padmanabhan, S. Development and validation of green chemistry HPLC-RI method for the simultaneous estimation of glucosamine and chondroitin sulfate from drug products. J. Appl. Biotechnol. Bioeng. 2022, 9, 67–74. [Google Scholar] [CrossRef]
- Lee, K.W.Y.; Porter, C.J.H.; Boyd, B.J. A simple quantitative approach for the determination of long and medium chain lipids in bio-relevant matrices by high performance liquid chromatography with refractive index detection. Am. Assoc. Pharm. Sci. 2013, 14, 927–934. [Google Scholar] [CrossRef] [PubMed]
- Silva, M.P.; Saraiva, L.; Pinto, M.; Sousa, M.E. Boronic Acids and Their Derivatives in Medicinal Chemistry: Synthesis and Biological Applications. Molecules 2020, 25, 4323. [Google Scholar] [CrossRef] [PubMed]
- Duval, F.; Beek, T.A.V.; Zuilhof, H. Sensitive Thin-Layer Chromatography Detection of Boronic Acids Using Alizarin. Synlett 2012, 23, 1751–1754. [Google Scholar] [CrossRef]
- Duval, F.; Wardani, P.A.; Zuilhof, H.; van Beek, T.A. Selective on-line detection of boronic acids and derivatives in high-performance liquid chromatography eluates by post-column reaction with alizarin. J. Chromatogr. A 2015, 1417, 57–63. [Google Scholar] [CrossRef]
- Temova Rakuša, Ž.; Grobin, A.; Roškar, R. A comprehensive approach for the simultaneous analysis of all main water-soluble vitamins in multivitamin preparations by a stability-indicating HPLC-DAD method. Food Chem. 2021, 337, 127768. [Google Scholar] [CrossRef]
- Petruczynik, A.; Wróblewski, K.; Dzioba, K.; Waksmundzka-Hajnos, M. Retention, separation selectivity and system efficiency of selected basic psychotropic drugs on different RPLC columns. Open Chem. 2015, 13, 943–950. [Google Scholar] [CrossRef]
- Alden, B.; Hudalla, C.; Wyndham, K.; Walter, T.; Lawrence, N.; Bouvier, E.; Walsh, D.; Iraneta, P. Synthesis and Applications of BEH Particles in Liquid Chromatography. LCGC Suppl. 2012, 30, 20–29. [Google Scholar]
- Neue, U.D.; Cheng, Y.-F.; Lu, Z.; Alden, B.A.; Iraneta, R.C.; Phoebe, C.H.; Van Tran, K. Properties of Reversed Phase Packings with an Embedded Polar Group. Chromatographia 2001, 54, 169–177. [Google Scholar] [CrossRef]
- Kadlecová, Z.; Kalíková, K.; Ansorge, M.; Gilar, M.; Tesařová, E. The effect of particle and ligand types on retention and peak shape in liquid chromatography. Microchem. J. 2020, 159, 105466. [Google Scholar] [CrossRef]
- Snydera, L.R.; Dolan, J.W.; Carr, P.W. The hydrophobic-subtraction model of reversed-phase column selectivity. J. Chromatogr. A 2004, 1060, 77–116. [Google Scholar] [CrossRef]
- Yang, W.; Gao, X.; Wang, B. Boronic Acid Compounds as Potential Pharmaceutical Agents. Med. Res. Rev. 2003, 23, 346–368. [Google Scholar] [CrossRef] [PubMed]
- Prashanthi, Y.; Ahmed, F.; Rao, T.N.; Parvatamma, B. Method development and validation of ixazomib drug by RP-HPLC in bulk and pharmaceutical dosage form. Indian Drugs 2021, 58, 28–34. [Google Scholar] [CrossRef]
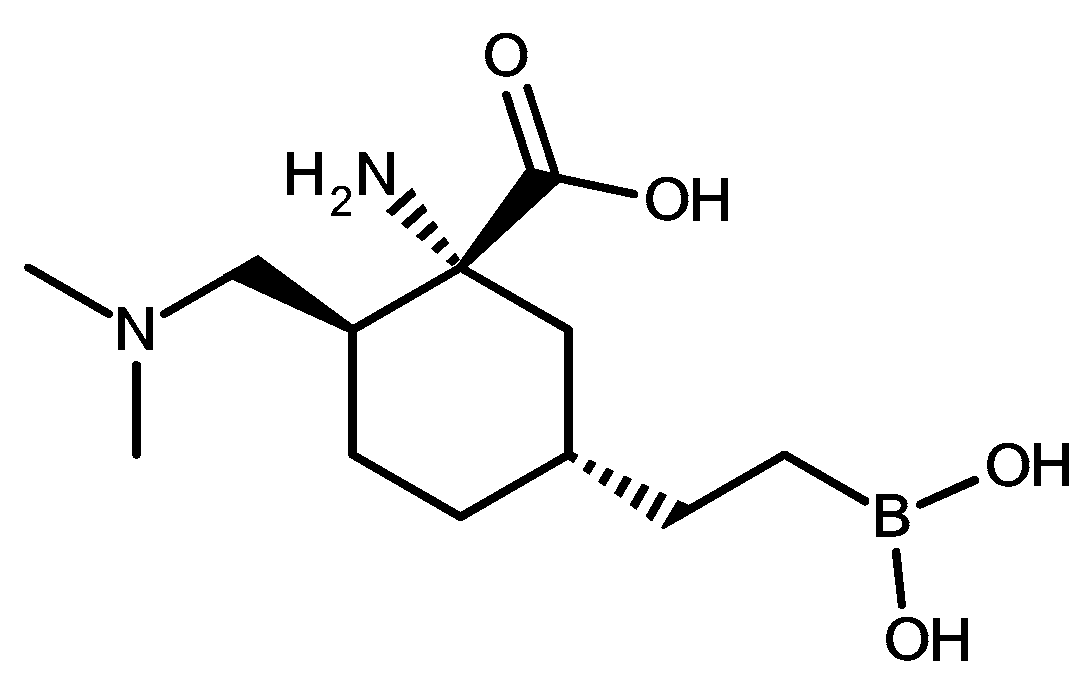
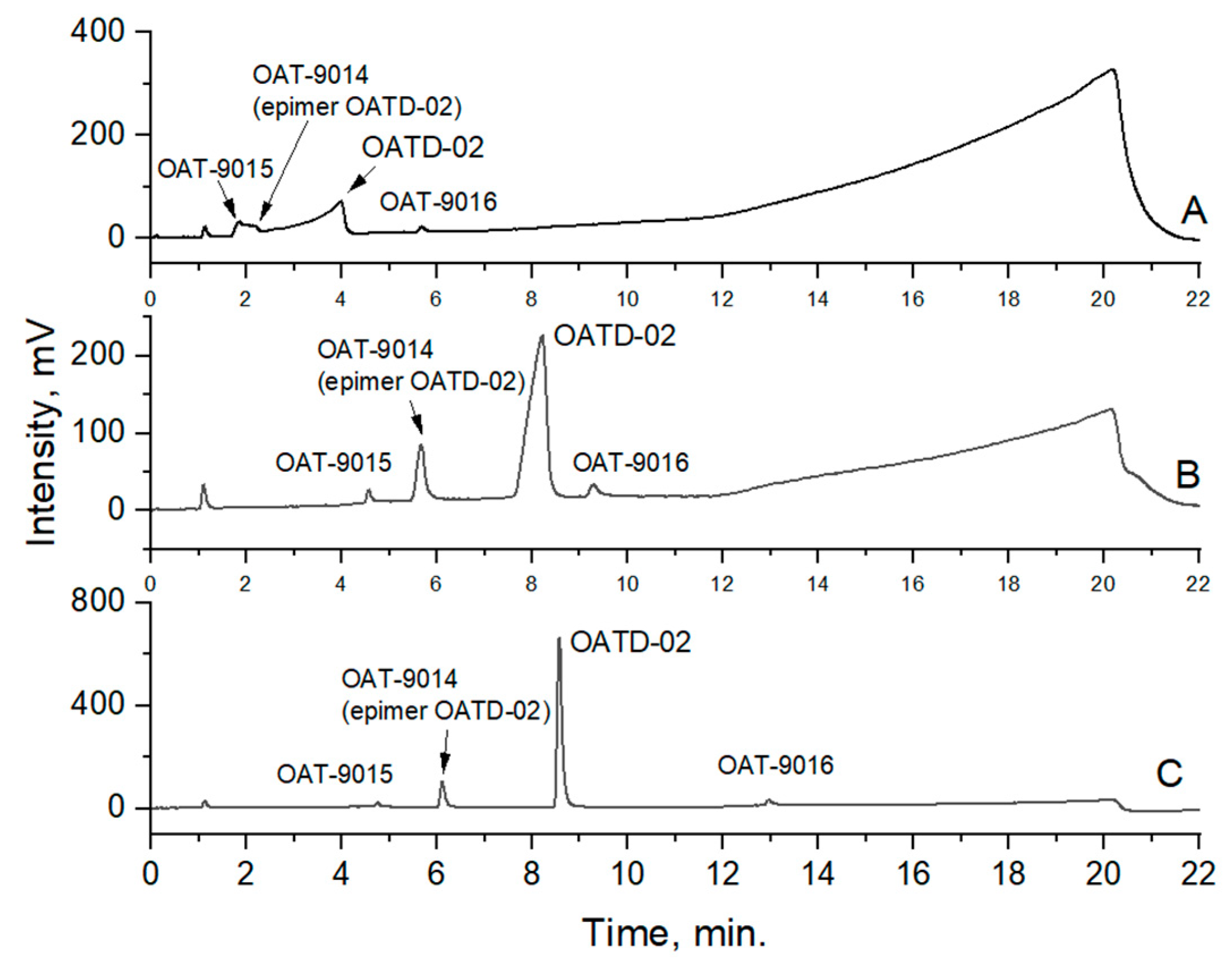

| 10 mM HCOONH4 | |||||||||
| #Peak | Name | Retention Time, Min | Area, µV × Min | Height, µV | Tailing Factor | Efficiency | Rs | Width (1/2)), Min | S/N |
| 1 | OAT-9015 | 1.86 | 351,598 | 29,603 | - | 321 | - | - | 6 |
| 2 | OAT-9014 epi-OATD-02 | 2.01 | 355,836 | 20,317 | - | 12 | 0.1 | - | 4 |
| 3 | OATD-02 | 3.99 | 2,824,963 | 61,963 | - | 196 | 1.1 | 0.58 | 13 |
| 4 | OAT-9016 | 5.67 | 84,399 | 10,901 | 1.47 | 10,689 | 2.5 | 0.12 | 2 |
| 10 mM CH3COONa | |||||||||
| #Peak | Name | Retention Time, Min | Area, µV × Min | Height, µV | Tailing Factor | Efficiency | Rs | Width (1/2)), Min | S/N |
| 1 | OAT-9015 | 4.57 | 111,014 | 17,364 | 1.62 | 11,161 | - | 0.09 | 6 |
| 2 | OAT-9014 epi-OATD-02 | 5.67 | 904,701 | 73,438 | 1.20 | 5206 | 4.5 | 0.18 | 26 |
| 3 | OATD-02 | 8.21 | 5,217,590 | 208,729 | 0.73 | 1965 | 4.8 | 0.41 | 73 |
| 4 | OAT-9016 | 9.28 | 186,428 | 15,203 | 1.40 | 13,737 | 2.0 | 0.19 | 5 |
| 10 mM NaHCO3 | |||||||||
| #Peak | Name | Retention Time, Min | Area, µV × Min | Height, µV | Tailing Factor | Efficiency | Rs | Width (1/2)), Min | S/N |
| 1 | OAT-9015 | 4.75 | 96,289 | 18,010 | 1.38 | 15,272 | - | 0.08 | 9 |
| 2 | OAT-9014 epi-OATD-02 | 6.12 | 735,667 | 101,383 | 1.45 | 15,615 | 7.8 | 0.11 | 49 |
| 3 | OATD-02 | 8.57 | 4,894,797 | 662,409 | 1.49 | 30,321 | 12.5 | 0.11 | 318 |
| 4 | OAT-9016 | 12.96 | 167,195 | 21,002 | 1.54 | 55,442 | 21.0 | 0.12 | 10 |
| OATD-02 | |||||||
|---|---|---|---|---|---|---|---|
| Type of Column | kR | Area, μV × Min | Height, μV | Tf | N/m | Wh1/2, Min | S/N |
| Luna C18 | 10.0 | 5,114,031 | 666,204 | 1.52 | 223,820 | 0.11 | 278 |
| Kinetex Phe-hex | 3.0 | 1,138,000 | 87,869 | 4.01 | 89,284 | 0.15 | 34 |
| Syncronis C18 | 10.4 | 3,997,187 | 548,064 | 1.29 | 260,230 | 0.10 | 344 |
| Kinetex XB-C18 | 6.9 | 4,661,412 | 670,962 | 1.73 | 321,730 | 0.10 | 328 |
| XSelect HSS T3 | 10.1 | 4,941,138 | 779,688 | 1.57 | 303,920 | 0.09 | 440 |
| Zorbax Extend-C18 | 10.4 | 4,555,981 | 644,835 | 1.53 | 175,340 | 0.10 | 239 |
| XSelect CSH-C18 | 6.6 | 4,894,797 | 662,409 | 1.49 | 303,210 | 0.11 | 318 |
| Parameters | Results | |
|---|---|---|
| Column | Waters XSelect CSH C18 2.5 µm, 100 mm × 3 mm | |
| Mobile phase A | 10 mM NaHCO3 (840 mg/L) in H2O | |
| Mobile phase B | 10 mM NaHCO3 (840 mg/L) in H2O:ACN (20:80) | |
| Flow rate | 0.5 mL/min | |
| Mobile phase C | 350 µM alizarin in MeOH | |
| Post-column reagent flow rate C | 0.5 mL/min | |
| Column temperature | 30 °C | |
| Detection | Fluorescence, λex= 470 nm, λem= 580 nm | |
| Injection volume | 5 µL | |
| Sample solvent | water | |
| Sample concentration | 1 mg/mL | |
| Run time | 23 min | |
| Gradient | Time | Conc. %B |
| 0 min | 0 %B | |
| 2–4 min | 6 %B | |
| 5–9 min | 10 %B | |
| 18 min | 100 %B | |
| 18.1–23 min | 0 %B | |
| Sample # | Recovery | ||
|---|---|---|---|
| Accuracy III (80%), % | Accuracy II (100%), % | Accuracy I (120%), % | |
| 1 | 99.6 | 99.4 | 99.0 |
| 2 | 98.5 | 99.5 | 99.2 |
| 3 | 99.6 | 98.8 | 99.2 |
| RSD, % | 0.66 | 0.36 | 0.12 |
| Mean Accuracy, % | 99.2 | ||
| SD | 0.38 | ||
| RSD, % | 0.39 | ||
| Injection # | Peak Area, μV × Min |
|---|---|
| 1 | 4,718,036 |
| 2 | 4,734,661 |
| 3 | 4,767,180 |
| 4 | 4,781,615 |
| 5 | 4,775,214 |
| Mean | 4,755,341 |
| SD | 27,590 |
| RSD, % | 0.58 |
| Temperature | Retention Time, Min | Efficiency | Tailing Factor |
|---|---|---|---|
| 30°C | 8.61 | 29,238 | 1.47 |
| 35°C | 8.38 | 30,358 | 1.50 |
| Flow, mL/min | Retention time, min | Efficiency | Tailing factor |
| 0.4 | 9.80 | 28,664 | 1.50 |
| 0.5 | 8.61 | 29,238 | 1.47 |
| 0.6 | 7.83 | 30,013 | 1.46 |
| Eluent composition | Retention time, min | Efficiency | Tailing factor |
| 0% B | 8.61 | 29,238 | 1.47 |
| 5% B | 8.18 | 27,328 | 1.49 |
| Column S/N# | Retention time, min | Efficiency | Tailing factor |
| 1573018118510 | 8.61 | 29,238 | 1.47 |
| 1733216418402 | 8.55 | 35,545 | 1.50 |
| Flow rate of alizarin, mL/min | Retention time, min | Efficiency | Tailing factor |
| 0.4 | 8.60 | 29,183 | 1.48 |
| 0.5 | 8.61 | 29,238 | 1.47 |
| 0.6 | 8.62 | 29,706 | 1.48 |
| Wavelength λex/λem | % change in peak area | Efficiency | Tailing factor |
| 468/580 | 1.6 | 29,305 | 1.48 |
| 470/580 | - | 29,238 | 1.47 |
| 472/580 | 1.7 | 29,299 | 1.48 |
| 470/578 | 1.9 | 29,353 | 1.47 |
| 470/582 | 1.9 | 29,412 | 1.48 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sobolewska, E.; Tyszkiewicz, M.; Blaszczyk, R.; Biesaga, M. HPLC with Post-Column Derivatization with Alizarin for Determination of OATD-02, an Anticancer Arginase Inhibitor in Clinical Development. Appl. Sci. 2023, 13, 9201. https://doi.org/10.3390/app13169201
Sobolewska E, Tyszkiewicz M, Blaszczyk R, Biesaga M. HPLC with Post-Column Derivatization with Alizarin for Determination of OATD-02, an Anticancer Arginase Inhibitor in Clinical Development. Applied Sciences. 2023; 13(16):9201. https://doi.org/10.3390/app13169201
Chicago/Turabian StyleSobolewska, Elzbieta, Magdalena Tyszkiewicz, Roman Blaszczyk, and Magdalena Biesaga. 2023. "HPLC with Post-Column Derivatization with Alizarin for Determination of OATD-02, an Anticancer Arginase Inhibitor in Clinical Development" Applied Sciences 13, no. 16: 9201. https://doi.org/10.3390/app13169201
APA StyleSobolewska, E., Tyszkiewicz, M., Blaszczyk, R., & Biesaga, M. (2023). HPLC with Post-Column Derivatization with Alizarin for Determination of OATD-02, an Anticancer Arginase Inhibitor in Clinical Development. Applied Sciences, 13(16), 9201. https://doi.org/10.3390/app13169201






