1. Introduction
Atypical ageing (ATA), also called untypical ageing (UTA), is an off-flavor appearing in young white wines, and it is associated with a rapid loss of a wine’s fruity aroma and to the development of unpleasant odors such as acacia blossom, mothball, dirty dish or furniture varnish [
1,
2]. While ATA has been well-documented in the literature, its chemical characterization is still incomplete [
2]. To date, 2-aminoacetophenone (2-AAP) has been identified as the main compound responsible for its occurrence in wines [
1]. Its sensory threshold was reported to be from 0.5 µg/L to 10.5 µg/L according to the wine matrix [
3]. Christoph et al. [
4] described 2-AAP formation by radical co-oxidation of sulfite to sulfate, which leads to the oxidative degradation of 3-indoleacetic acid (3-IAA), a compound derived from tryptophan metabolism. Hoenicke et al. [
5] proposed that the pyrrole ring cleavage of 3-IAA by superoxide radicals, produced by the aerobic oxidation of sulfite during storage of sulfurized wines, form 3-(2-formylaminophenyl)-3-oxopropinoic acid (FAPOP). The decarboxylation of FAPOP leads afterwards to the formation of N-formyl-2-aminoacetophenone (FAP) and finally to 2-AAP, or alternatively, oxidized indole–acetic acid. The possibility that other metabolites such as tryptophan, indole-3-lactic acid (ILA) and kynurenine could be co-oxidized to 2-AAP after sulfite addition was also investigated, but these reactions only had a minor contribution to the formation of 2-AAP in wine [
4]. The photooxidation of free and protein-bound tryptophan in combination with storage conditions could also potentially yield 2-AAP [
6]. On the other hand, 2-AAP could be formed by yeasts during alcoholic fermentation, but again in concentrations below the sensory threshold [
7,
8].
While the chemical reactions yielding 2-AAP concentrations in white wines have been well described, the factors driving its formation in wines are still poorly understood [
2]. The high levels of free 3-IAA at sulfiting increases the risk of obtaining higher levels of 2-AAP later in the wine, but no close correlation was established between the concentrations of 3-IAA and 2-AAP in wines [
2]. The formation of 2-AAP requires only some residual traces of dissolved oxygen, which are incapable of leading to classical oxidative ageing. However, the presence of antioxidants before sulfiting was reported to prevent the formation of 2-AAP [
4,
9]. It has been shown that the addition of ascorbic acid could strongly inhibit the formation of 2-AAP [
10]. However, the use of ascorbic acid in winemaking can promote the formation of strong oxidants if at the same time the level of sulfur dioxide is too low [
11]. Consequently, research has been carried out to find an alternative to ascorbic acid in order to prevent the formation of 2-AAP in wine. Among other antioxidants used in oenology, the effect of tannin and glutathione supplementations in wine did not consistently limit the 2-AAP formation [
2]. In contrast, Nardin et al. [
3] recently showed that the addition of tannins in grape must prior to fermentation was more efficient than glutathione in preventing the formation of 2-AAP in wine, with gallotannin showing an effect almost comparable to ascorbic acid.
Another important factor to consider in wine oxidation chemistry is the role of transition metal ions. This has been well documented for some important chemical reactions like the oxidation of ethanol into acetaldehyde [
12]. On the other hand, the impact of transition metal ions on ATA development has been only suggested by sensory study without any quantification of 2-AAP [
13]. Finally, a preliminary study performed in our laboratory (data not published) suggested that some varieties might be more prone to the formation of 2-AAP than others. This was intriguing, as while historically, Riesling wines have been known to be particularly sensitive to ATA, this off-flavor was observed in different types of white wines all around the world [
2].
This study aimed to improve the understanding of 2-AAP formation in white wines by investigating three main questions. Firstly, the potential varietal effect on 2-AAP concentration in white wines was assessed by analyzing two set of commercial white wines, and included the quantification of 2-AAP after artificial ageing and 2-AAP precursors. Secondly, the effect of different types of antioxidants on 2-AAP formation was investigated, as previously published results were unclear on the topic. Finally, the influence of transition metal ions present in wine (Fe, Cu and Mn) on 2-AAP concentration was performed for the first time.
2. Materials and Methods
L-tryptophan (≤98%), 3-indoleacetic acid (98%), DL-indole-3-lactic acid (99%), DL-kynurenine (≤95%), anthranilic acid (≤98%), 2-aminoacetophenone (98%), glutathione (98%), iron (II) sulfate heptahydrate (99.5%), iron (III) chloride hexahydrate (97%), manganese (II) sulfate monohydrate (98%) and gallic acid (98%) were supplied by Sigma-Aldrich (Steinheim, Germany). LC-MS methanol was from Supelco (Bellefonte, PA, USA), and LC-MS formic acid was from Honeywell-Fluka (Charlotte, NC, USA). Copper (II) sulfate pentahydrate (98%) and high purity grade ethanol (>99.8%) was supplied by Honeywell-Riedel-de Haën (Charlotte, NC, USA). 2-aminoacetophenone-D5 was supplied by EPTES (Vevey, Switzerland). Commercial ellagitannin (TA1), a commercial mixture of ellagitannin and condensed tannins (TA2), and inactivated yeast derivatives (IYD) were kindly provided by the Laffort company (Floirac, France). Chemicals were prepared with mili-Q water obtained from an ELGA PURELAB Option water system supplied by Elga-Veolia (Buckinghamshire, UK).
2.1. Surveys of 2-Aminoacetophenone in Commercial Varietal Wines
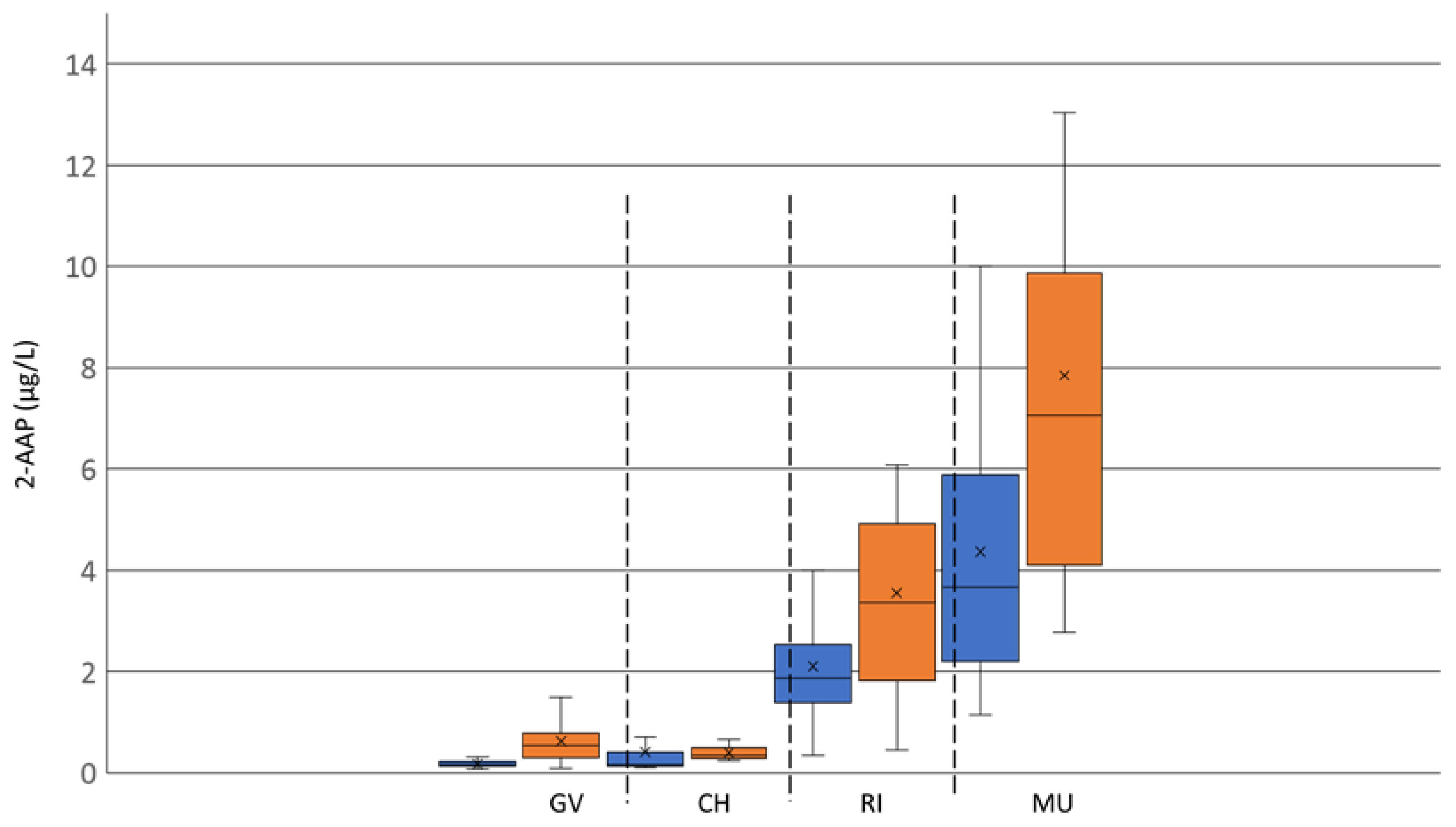
Two surveys of 2-AAP content in commercial varietal wines were carried out. The first survey consisted of analyzing 2-AAP concentration in 86 commercial Austrian white wines from the 2021 vintage prior to and after ageing at 40 °C for four days. This set of wines was sourced from producers and was exclusively composed of four varieties: Grüner Veltliner (20), Chardonnay (16), Riesling (25) and Muscat Ottonel (25) (
Supplementary Data S1). The choice of the varieties was based on their importance for the Austrian wine market and by considering a preliminary study highlighting several trends of higher 2-AAP in some of the selected varieties. Ageing parameters were selected based on a recent study from Nardin et al. [
3] showing that ageing different white varieties for 3 and 6 days at 40 °C was enough to substantially increase the level of 2-AAP. After 4 days of artificial ageing, samples were stored at 4 °C for a few days prior to analysis by gas chromatography coupled to a triple-quad mass detector (GC-MS/MS).
A second commercial white wine screening consisted of 45 wine samples from Slovenia (40) and Croatia (5) purchased from shops and producers. Samples from vintages 2018, 2019, 2020 and 2021 were analyzed in 2022 as in the first survey. Selected varieties were the same as for the Austrian survey, however, Grüner Veltliner was excluded (rarely found in Slovenia and Croatia) and replaced by the locally more important varieties of Zelen (Slovenia) and Welschriesling (Croatia) (
Supplementary Data S1). The 2-AAP concentration was measured in these wines without artificial ageing.
In the same wines, the level of 2-AAP precursors tryptophan, anthranilic acid, indolelactic acid (ILA), 3-indoleacetic acid (3-IAA) and kynurenine were measured by liquid chromatograph coupled to a triple-quad mass detector (LC-MS/MS).
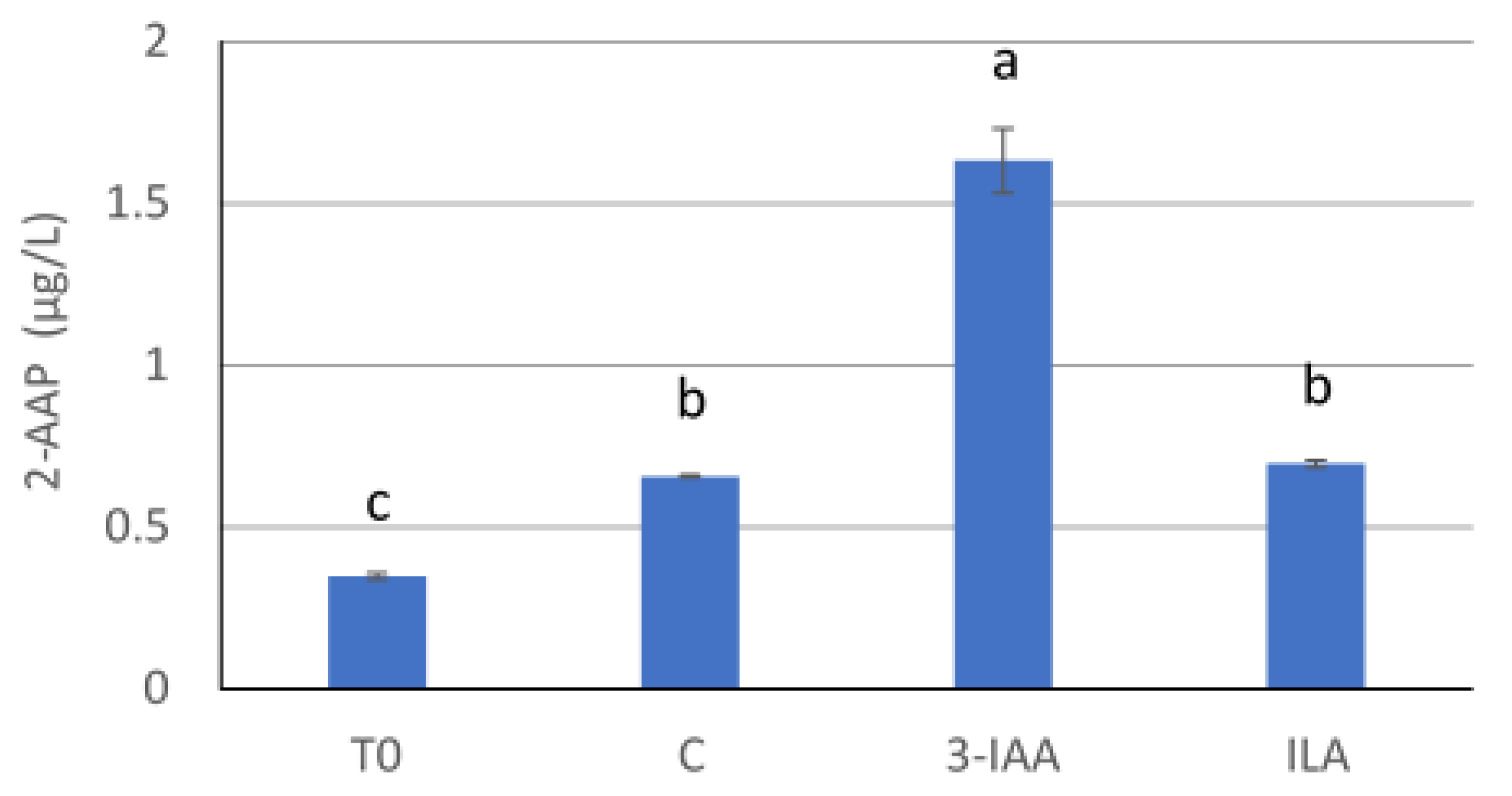
In order to investigate the influence of ILA on 2-AAP formation, 2-AAP was analyzed in a 2021 white blend from Slovenia (wine A) before and after artificial ageing for 4 days at 40 °C, and in the same wine spiked with 3-IAA and ILA at 100 µg/L after artificial ageing.
2.2. Experiments Using Antioxidants and Transition Metal Ions Supplementation
2.2.1. Antioxidants Addition
Two studies investigating the effect of antioxidants on 2-AAP formation were carried out in two white wines (A and B). Both wines were initially spiked with 100 µg/L of 3-IAA in order to ensure 2-AAP production. Chemical parameters of wines A and B are displayed in
Supplementary Data S2.
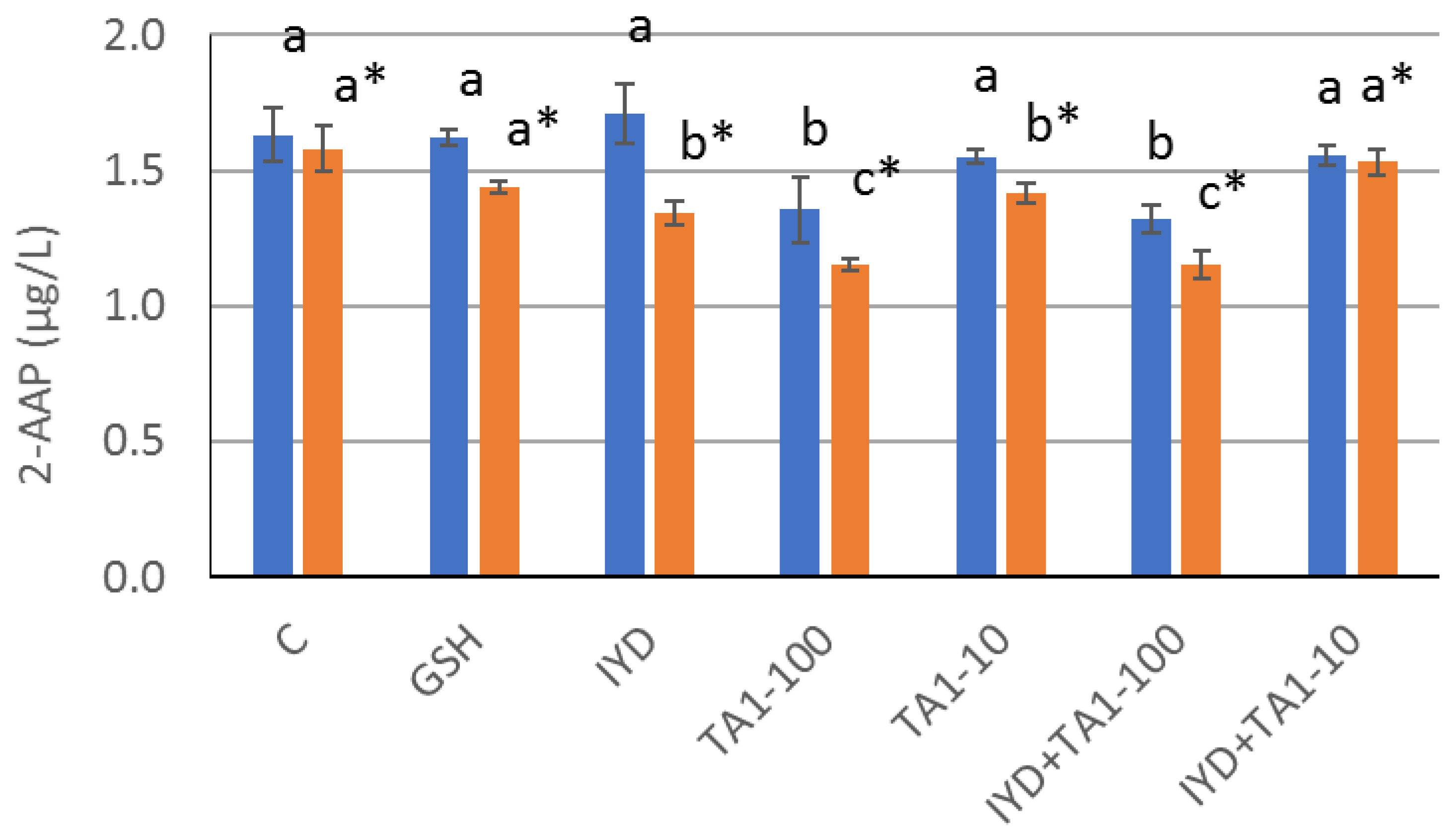
Experiment 1 was performed in 2021 on a 2020 white blend from Slovenia (wine B). Initial free sulfite and 2-AAP concentrations were 25 mg/L and 0.75 µg/L, respectively. A series of different antioxidants was tested as follows: glutathione (GSH) at 12 mg/L, inactivated yeast derivatives (IYD) at 400 mg/L, gallic acid (GA) at 70 mg/L, commercial ellagitannins (TA1) at 100 mg/L, commercial mixture of ellagitannins and condensed tannins (TA2) at 100 mg/L and inactivated yeast derivatives (400 mg/L) in combination with commercial ellagitannins (100 mg/L). The targeted values of antioxidant additions were set according to supplier recommendations.
Experiment 2 was performed in 2022 on wine A again. Initial concentrations of free sulfite, 2-AAP, Cu, Mn and Fe were 22 mg/L, 0.35 µg/L, 75 µg/L, 796 µg/L and 1047 µg/L, respectively. A series of different antioxidants were tested as follows: GSH at 12 mg/L, inactivated yeast derivatives (IYD) at 400 mg/L, commercial ellagitannin (TA1) at 10 mg/L and 100 mg/L and inactivated yeast derivatives (400 mg/L) in combination with commercial ellagitannin (100 mg/L). Each antioxidant treatment was tested alone and in combination with a mixture of metal ions to reach the final concentration as follows: Cu (II) at 1 mg/L using copper (II) sulfate pentahydrate solution; Fe(II)/Fe(III) (1:1) at 5 mg/L using iron (II) sulfate heptahydrate and iron (III) chloride hexahydrate solutions; and Mn (II) at 4 mg/L using manganese (II) sulfate monohydrate solution.
Each treatment was prepared in triplicate adding the antioxidant and metal ion solutions in 20 mL vials filled with 18 mL of wine and tightly sealed with PTFE-lined caps. Vials were then exposed to artificial ageing in the oven for 4 days at 40 °C. Thereafter, vials were stored in the fridge for a few days prior to analysis.
2.2.2. Metal Ions Addition
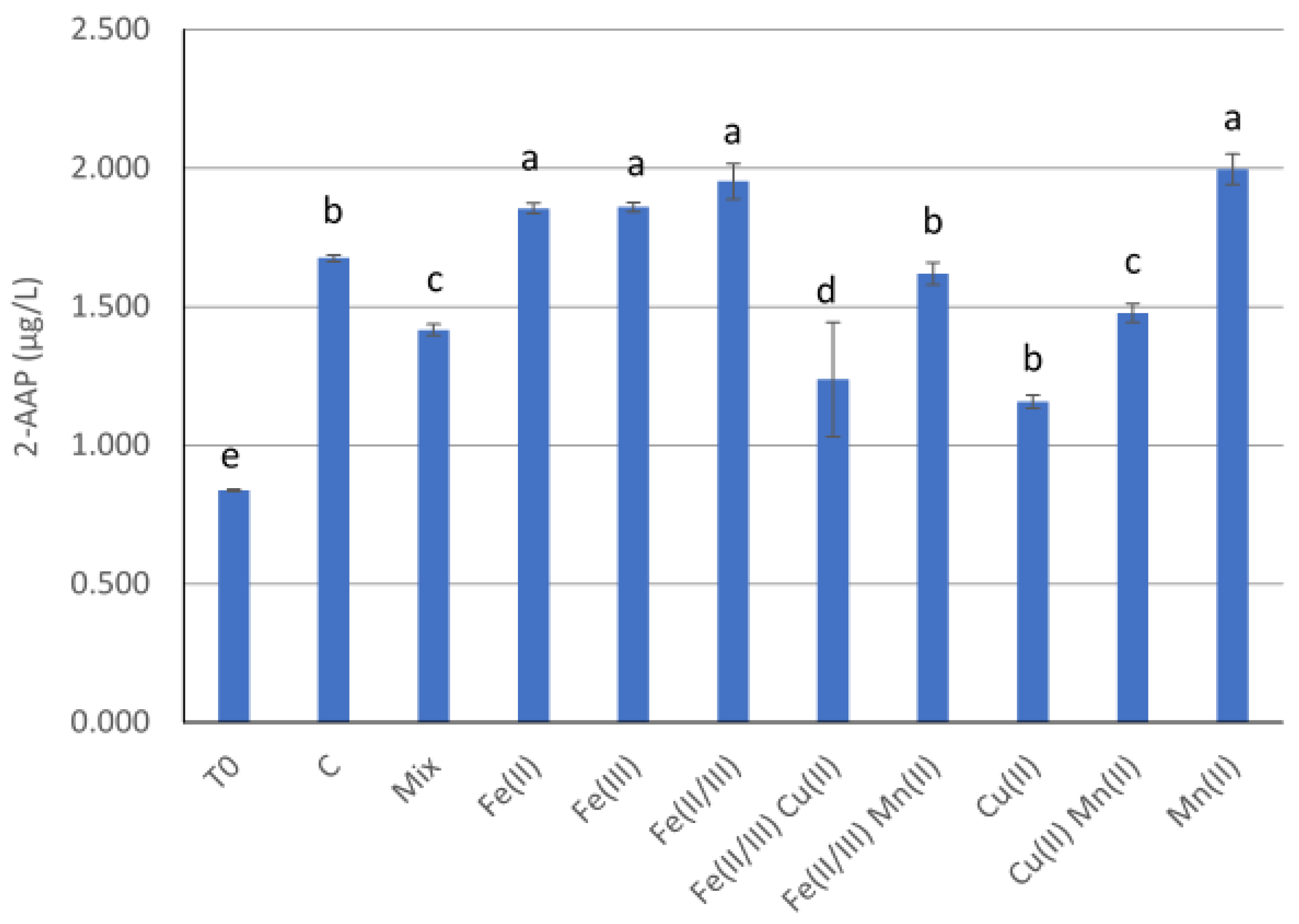
A different wine, a Slovenian white blend from 2021 (wine C), was used in 2022 to test the effect of transition metal ions alone and in mixture on the formation of 2-AAP in white wine. Initial concentrations for free sulfite, 2-AAP, Cu, Mn and Fe were 20 mg/L, 0.83 µg/L, 58 µg/L, 872 µg/L and 564 µg/L, respectively. Transition metals were tested using the same solution freshly prepared as in experiment 2. The following treatments were tested: Fe(II) at 5 mg/L; Fe(III) at 5 mg/L; Fe(II) and Fe(III) (1:1) at 5 mg/L; Fe (II/III) and Mn (II) at 5 mg/L and 4 mg/L, respectively; Cu (II) at 1 mg/L; Mn (II) at 4 mg/L; Cu (II) and Mn (II) at 1 mg/L and 4 mg/L, respectively; Fe(II/III) (1:1), Mn(II) and Cu(II) at 5 mg/L, 4 mg/L and 1 mg/L, respectively.
Each treatment was prepared in triplicate by adding metal ion solutions in 20 mL vials filled with 18 mL of wine and tightly sealed with PTFE-lined caps. Vials were further exposed to artificial ageing in the oven for 4 days at 40 °C. Thereafter, vials were stored in the fridge for a few days prior to analysis.
2.3. GC-MS/MS Measurement of 2-AAP
All wines were analyzed for the content of 2-AAP by HS-SPME-GC-MS/MS. For this purpose, 5 mL of mQ water, 5 mL of sample, 50 µL of 2-AAP-d5 solution prepared at 500 µg/L in high purity grade ethanol and 3 g of NaCl were added to a 20 mL SPME vial. Analysis was performed using a gas chromatograph (type 7890 B A, Agilent Technologies, Santa Clara, CA, USA) with an injector, a controller, a CTC Analytics autosampler (Zwingen, Switzerland) and a triple-quad mass spectrometer (MS/MS) detector (type 7010B GC/MS Triple Quad) (Agilent Technologies, Santa Clara, CA, USA). Separation was performed using a ZB-5MS column (30 m × 0.25 mm I.D. × 0.25 µm df; Agilent Technologies, Santa Clara, CA, USA) at a column flow rate of 1.02 mL/min. Helium was used as the carrier gas. The blue fiber coated with polydimethylsiloxane–divinylbenzene 65 µm (PDMS-DVB) (Supelco, Bellefonte, PA, USA) was used with the following settings: pre-incubation time: 60 s, incubation temp.: 70 °C, pre-incubation agitator speed: 500 rpm, agitator-on time: 5 s, agitator-off time: 2 s, fiber exposure: 12 µL, vial penetration: 15 mm, extraction time: 2400 s, injection penetration: 32 mm, desorption time: 120 s, fib. cond. temp.: 250 °C, and post-fib. cond. time: 600 s. The inlet was set in splitless mode at 250 °C. The oven temperature was programmed at 50 °C for 2 min before a rise of 15 °C/min to reach 160 °C, which was held for 1 min. Then, the temperature was increased by 20 °C/min to reach 230 °C and held for 5 min. The postrun temperature was set at 60 °C. The total run time was 19 min. The transfer line temperature was set at 250 °C. The mass spectrometer was operated in electron ionization (EI) mode at 70 eV with multiple reaction monitoring (MRM). All of the monitored transitions are listed in
Table 1. Data were acquired and analyzed through Agilent MassHunter Workstation software, version B.07.00. The calibration was performed in 8 calibration steps in the range from 0 to 9.6 µg/L in the synthetic wine (R
2 = 0.9993). Limit of detection and limit of quantification were calculated at 0.1 and 0.3 µg/L by considering signal to noise S/N = 3 and S/N = 10, respectively. Recovery was assessed at 99% by spiking one-year old white wine at 0.6 µg/L.
2.4. ICP/MS Measurement of Metal Ions
Transition metal ions were analyzed by ICP-MS. All reagents used were of analytical grade or better. For sample dilution and preparation of standards, ultrapure water (MilliQ, Millipore) and ultrapure acids (HNO3 and HCl, Merck-Suprapure) were used. Standards were prepared in-house by dilution of certified, traceable, inductively coupled plasma (ICP)-grade single-element standards (Merck CertiPUR). The standards and blanks were prepared in model wine consisting of 12% ethanol, 4 g/L glucose and 4 g/L tartaric acid, with the pH adjusted to 3.4. An Agilent Technologies 7900 ICP-MS instrument equipped with a MicroMist glass concentric nebulizer and a Peltier-cooled, Scott type spray chamber was used.
2.5. UHPLC-MS/MS Measurement of 2-AAP Precursors
Analysis was carried out by a 1290 infinity UHPLC system coupled to a 6460 triple-quadrupole mass spectrometer (Agilent Technologies). Wines were diluted in methanol (1:1,
v/
v), vortexed, filtered through a 0.22 μm PVDF filter (Millipore, Billerica, MA, USA) and directly injected. Samples were kept at 4 °C during analysis, and the injection volume was 5 µL. Separation was performed on a 100 mm × 2.1 mm, 1.8 µm column (Acquity HSS T3, Waters) maintained at 40 °C. Flow was 0.4 mL/min; mobile phase A was 0.1% formic acid and B was 0.1% formic acid in methanol. The linear gradient started at 2% B, to 55% B in 45 min, to 100% B in 1 min, at 100% B for 3 min, to 2% B in 1 min and then 5 min at 2% B. Analysis was performed in positive mode using electrospray ionization (ESI, Jet-Stream) and selected reaction monitoring (SRM) acquisition. Nitrogen was the collision gas, and the source parameters were gas temperature 250 °C, gas flow 6 L min
−1, nebulizer 241 Kpa, sheath gas heater 375 °C, sheath gas flow 10 L min
−1, capillary 4000 V. Three mass transitions were selected for each compound: one for the quantifier and two for qualifiers (
Table 2). The linearity of the detector response for 2-AAP precursors was verified in synthetic wine diluted in methanol (1:1,
v/
v). Seven concentration levels were injected in six repetitions for each level. Linearity was determined, and the limit of detection and limit of quantification were calculated by considering signal to noise S/N = 3 and S/N = 10, respectively (
Table 2).
2.6. Statistical Analysis
Comparisons between treatments were performed by one-way ANOVA (analysis of variance) using Microsoft Excel 2016. The threshold for statistical significance was set at 95% (p-value < 0.05). Box plots were also made using Microsoft Excel 2016 software.
4. Discussion
Since the first empirical evidence of the development of ATA in white wines in the late 1980s, most of the published studies investigating the formation of 2-AAP in wine were carried out on Riesling [
2]. This anecdotal observation suggests that the Riesling variety has been known for a long time to be sensitive to the formation of 2-AAP. On the other hand, very few studies have investigated a potential varietal effect on 2-AAP concentration in wine. Košmerl and Zlatić [
15] analyzed 21 Slovenian wines, not including Riesling, and more recently, Alpeza et al. [
16] measured 2-AAP in 20 Croatian white wines from different varieties, including Riesling. While the latter study found that the highest concentrations of 2-AAP were measured in Riesling wines, it is difficult to highlight a consistent trend with such a low number of samples. With two distinctive surveys representing a total of 139 wines analyzed, the present study highlighted that Riesling wines significantly showed a higher concentration of 2-AAP in comparison to Chardonnay and some other local varieties from Central Europe such as Grüner Veltliner, Welschriesling and Zelen. The average 2-AAP concentration in Riesling wines was between 2 and 4 µg/L. This is above the perception threshold reported for white wines (0.5–1.5 µg/L) [
2]. However, the variety that displayed the highest average concentration of 2-AAP was Muscat blanc, and that observation was somewhat unexpected since ATA has been rarely reported in Muscat wines. This is probably due to the presence of a high concentration of monoterpenols imparting an intense floral aroma, which strongly mitigates the impact of 2-AAP on Muscat wine aroma [
12].
The wider distribution of 2-AAP concentrations after ageing for most of the varieties indicated an important matrix effect in the formation of this compound in white wines. In order to clarify the matrix effect and varietal differences on 2-AAP formation, the second survey, performed on Slovenian and Croatian wines, targeted free tryptophan metabolites known as potential precursors of 2-AAP. No relationship between the level of 2-AAP and its precursors could be observed in the wines analyzed in this survey, including the main known precursor 3-indoleacetic acid (3-IAA). The lack of correlation between free 3-IAA and 2-AAP in wines has already been reported by several authors [
8,
9,
17]. Levels of 2-AAP potential precursors were in accordance with previously published concentrations [
18]. The most abundant tryptophan derivate was not 3-IAA but indolelactic acid (ILA). Interestingly, Riesling wines showed the highest ILA concentrations in comparison to other varieties. However, when ILA was added to a white blend, the concentration of 2-AAP after artificial ageing was similar to the same wine without ILA addition. Only the wine with an addition of 3-IAA showed a significantly higher level of 2-AAP (
Figure 2). Therefore, the varietal differences observed in 2-AAP concentration in white wines were not directly linked to the levels of its precursors in wine. Further investigation on varietal factors that could be directly or indirectly involved in 2-AAP formation are needed.
Moreover, Lavigne-Cruege et al. [
19] suggested that glutathione and other yeast biomass derivates could limit 2-AAP formation, but this observation was not consistent with some other studies [
20,
21]. In the present work, phenolic compounds such as gallic acid and commercial tannins were more efficient at limiting the formation of 2-AAP than GSH and IYDs. While commercial tannins reduced the concentration of 2-AAP in both trials, GSH and IYDs had an impact only on one wine. Nardin et al. [
3] recently published similar results using GSH, ellagic tannins, galla and grape tannins. They found that galla and grape tannins were the most efficient at limiting the 2-AAP concentration in the wine and that ellagic tannins had a stronger impact than GSH. In the present study, the mixture of grape and ellagic tannins (TA2) was also slightly more efficient than pure ellagic tannins (TA1). However, Nardin et al. [
3] added antioxidants in must at higher levels in comparison to the present study, where the doses reflect additions suggested by manufacturers to be used in white wine. The addition of tannins in white wine at the upper limit of the recommended range often strongly impacts wine sensory profile. For that reason, our second trial targeted two levels of tannins addition corresponding to 10 g/hL (100 mg/L) and 1 g/hL (10 mg/L), respectively. The higher dosage was the most efficient to reduce 2-AAP concentration.
All these results suggest that when wines are aged in a barrel on gross lees, the tannins extracted from wood might be more efficient to limit the formation of 2-AAP than the antioxidant activity from the lees. Some synergistic effect between both classes of antioxidants cannot be excluded in some wines, as observed during the first trial where the addition of IYD with TA1 was slightly more efficient at reducing 2-AAP than TA1 alone (
Figure 3). However, such an effect was not observed in the second wine, irrespective of tannin dosage (
Figure 4).
Transition metal ions are known to catalyze many chemical reactions, but in a winelike medium, mainly Fe, Cu and, to a lesser extent, Mn have been studied for their role in wine oxidation and reduction [
12,
22]. Hoenicke et al. [
5] speculated that transition metals could potentially influence the formation of 2-AAP in wine without studying this hypothesis. Morozova et al. [
13] reported higher ATA perception in Riesling wines due to the presence of Fe and Cu but 2-AAP was not quantified. The present study is therefore the first to our knowledge to explore the impact of metal ions on 2-AAP production in wine. In the same preliminary study as mentioned earlier, a 2-fold increase in 2-AAP concentration (0.22 to 0.45 µg/L) was observed in the Chardonnay wine aged in an inox tank after the addition of a mixture of Fe, Cu and Mn in order to reach a final concentration at 5, 1 and 4 mg/L, respectively. In contrast, the 2-AAP level did not change when the same addition was made in the wine aged on gross lees in a new oak barrel. This observation suggests that antioxidants from oak or lees could also limit the influence of metal ions in the formation of 2-AAP.
When Fe, Cu and Mn were added together at the same level in two white blends, 2-AAP concentration after artificial ageing was not affected in the first wine (
Figure 4) and was decreased in the second wine (
Figure 5). Interestingly, the presence of higher concentrations of Fe, Cu and Mn in a mixture significantly improved the efficiency of antioxidants to reduce the 2-AAP concentration, while no metal ion effect was observed in the wines without antioxidant addition. Commercial tannins remained the most efficient antioxidant to limit 2-AAP concentration in the presence of metal ions. These results highlight some complex interaction between metal ions and wine matrix involved in the formation of 2-AAP. Metal ions’ reactivity is closely dependent on their redox potential, which is significantly influenced by the presence of ligands such as tartaric acid, malic acid, polyphenols or other antioxidants [
23]. Fe(III/II) redox potential is also influenced by the presence of Cu and Mn [
22,
24]. Copper accelerates Fe(II) oxidation, and that in turn can promote sulfite oxidation [
23,
25], which is suspected to form the superoxide radical responsible for 2-AAP formation in white wines [
5]. In contrast, in our experiment, the addition of Cu(II) tended to decrease the 2-AAP concentration. On the other hand, Mn was proven to be a strong promoter of sulfite oxidation in the presence of traces of Fe and Cu [
22]. In our conditions, the presence of a higher level of Mn was actually rather associated with a higher content of 2-AAP. However, this trend was not observed when the wine had a higher initial Fe(II/III) concentration. This shows the complexity of chemical reactions yielding 2-AAP in a wine medium and can explain why the main limiting factors of 2-AAP formation in wine are not well identified yet. Further research will have to investigate more in detail the interactions between transitional metal ions and wine matrix.
This study opened new perspectives in the understanding of 2-AAP synthesis in white wines. Both varietal differences in 2-AAP formation and interactions between antioxidants and transition metals require further investigations.