Effect of Carbon Dioxide Injection on Limestone Permeability Damage Induced by Alumina Nanoparticles for Enhanced Oil Recovery Applications
Abstract
1. Introduction
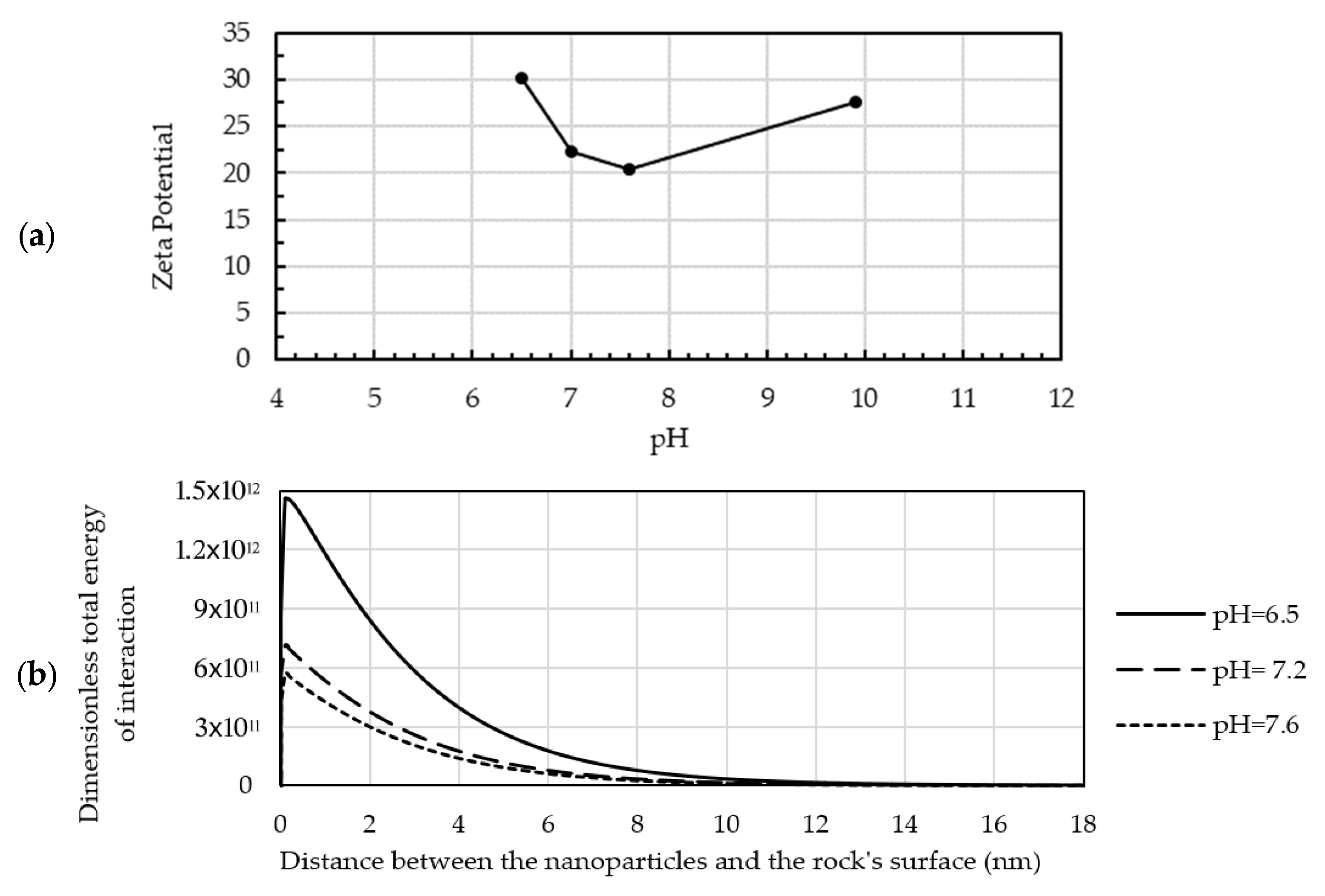
- The pH value is far from the point of zero charge, so the corresponding zeta potentials of nanoparticles and rock surface are high. When the pH value is at or near the point of zero charge, the nanofluid loses its stability because of the weak repulsive force [8].
- The pH value is higher or lower than the point of zero charge so that the signs of zeta potentials of nanoparticles and rock surface match each other.
2. Materials and Methods
2.1. Materials
2.2. Nanofluid Preparation and Properties
2.3. Contact Angle Measurement
2.4. Core Flooding Experiments
2.5. Permeability Damage Evaluation
3. Results and Discussion
3.1. Zeta Potential and Stability
3.2. Limestone Permeability Damage
3.3. Permeability Damage Mechanisms
- Although the nanofluid was stable in the low-pH case, some sedimentation of nanoparticles occurred, but not as much as in the other experiments in which sedimentation was high. In all experiments, sedimentation contributed to the retention of nanoparticles inside the cores.
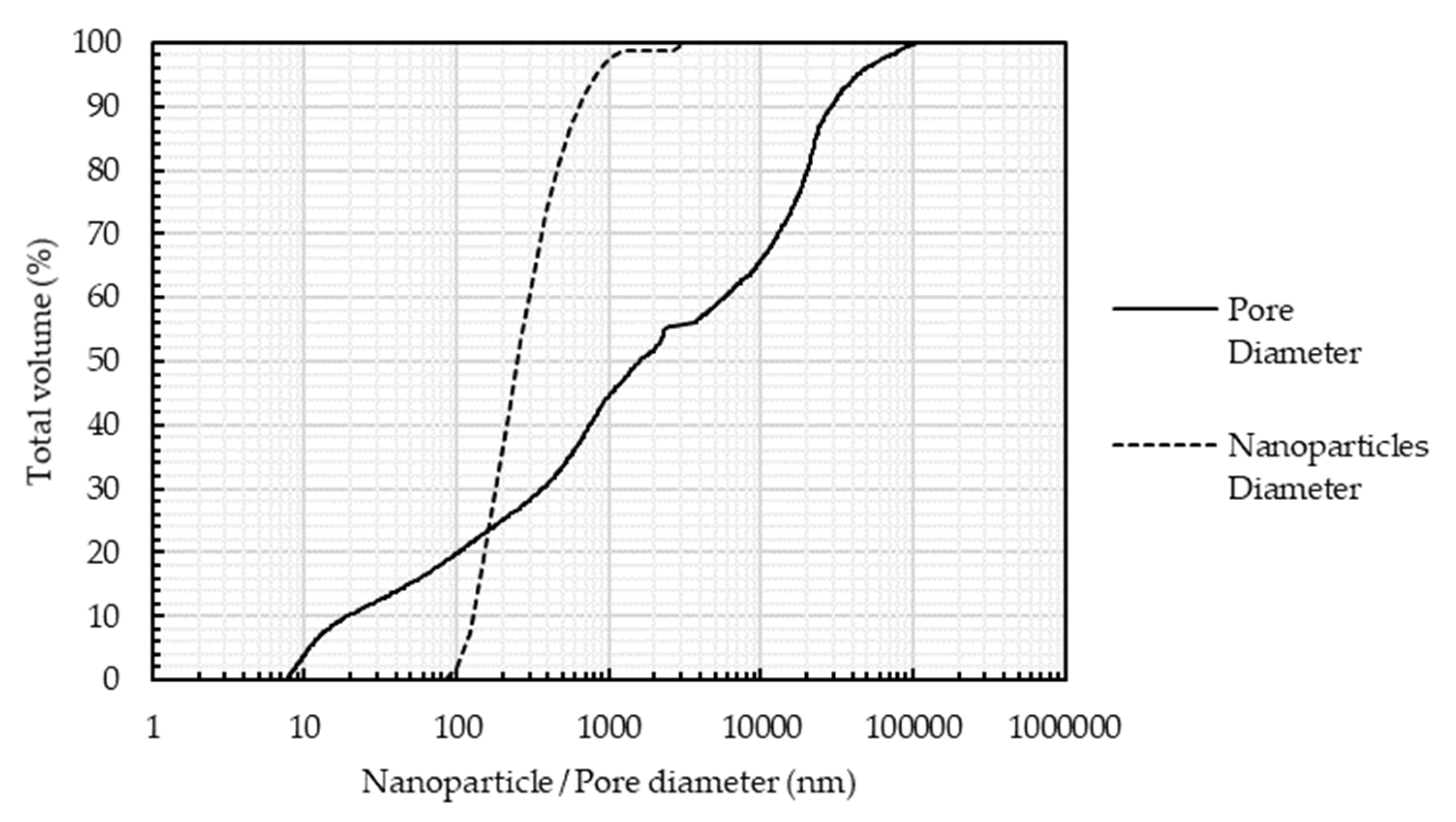
- While the nanoparticles had an average diameter of 50 nm in powder form, they tended to form larger clusters after adding them to the nanofluid. The cluster size ranged between 100 and 1000 nm. The average cluster size of the nanoparticles at a pH of 4.5 was approximately 250 nm, and at a pH of 2.8, the average cluster size was 290 nm. The pH of the nanofluid during ball milling and beads to nanofluid weight ratio are two factors that greatly affected the average cluster size. For example, a pH of 6.5 or a beads to nanofluid weight ratio of 1 resulted in an average cluster size of around 500 nm. The smallest size was 250 ± 10 nm and was consistently achieved using the ball milling method. Adding 30 min of an ultrasonic bath after ball milling did not lower the cluster size. The 250 nm size was not sufficiently small to not cause permeability damage. The pore size distribution of a limestone core similar to the cores used in the experiments and a size distribution of the nanoparticles were used to obtain an approximate difference between the pore size and the nanoparticles’ size. As depicted in Figure 8, approximately 20% of the pores were too small for the smallest nanoparticles, and another 5% of the pores were too small for 25% of the nanoparticles. This case excluded the agglomeration occurring inside the core during the injection process, which rendered the retention through straining more impactful on permeability.
- The roughness of the pore surface considerably influences the retention of nanoparticles inside the pores. The nanoparticles tend to accumulate in the dents of the rough surface because of the Brownian motion or repulsion between them. However, the repulsion between the nanoparticles and pore surface is only effective at a short distance, as displayed in Figure 6b, and that distance is shorter than the depth of the rough surface’s dents. The surface roughness was investigated using SEM images, and the resulting images of the pores’ surfaces are displayed in Figure 9. The images revealed that the surface of the pores was rough in general, but it had some smooth surfaces. Some pore channels were rough on one side and smooth on the other side.
3.4. Oil Recovery
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- World Energy Outlook 2022. Available online: https://www.iea.org/reports/world-energy-outlook-2022/outlook-for-liquid-fuels (accessed on 14 June 2023).
- Chakraborty, S.; Panigrahi, P.K. Stability of nanofluid: A review. Appl. Therm. Eng. 2020, 174, 115259. [Google Scholar] [CrossRef]
- Ajulibe, D.; Ogolo, N.; Ikiensikimama, S. Viability of SiO2 Nanoparticles for enhanced oil recovery in the Niger Delta: A comparative analysis. In Proceedings of the SPE Nigeria Annual International Conference and Exhibition, Lagos, Nigeria, 6–8 August 2018. [Google Scholar]
- Alsaba, M.T.; Al Dushaishi, M.F.; Abbas, A.K. A comprehensive review of nanoparticles applications in the oil and gas industry. J. Pet. Explor. Prod. Technol. 2020, 10, 1389–1399. [Google Scholar] [CrossRef]
- Hogeweg, A.S.; Hincapie, R.E.; Foedisch, H.; Ganzer, L. Evaluation of aluminium oxide and titanium dioxide nanoparticles for EOR applications. In Proceedings of the SPE Europec Featured at 80th EAGE Conference and Exhibition, Copenhagen, Denmark, 11–14 June 2018. [Google Scholar]
- Ogolo, N.A.; Olafuyi, O.A.; Onyekonwu, M.O. Enhanced Oil Recovery Using Nanoparticles. In Proceedings of the SPE Saudi Arabia Section Technical Symposium and Exhibition, Khobar, Saudi Arabia, 8–11 April 2012. [Google Scholar]
- Al-Anssari, S.; Barifcani, A.; Wang, S.; Maxim, L.; Iglauer, S. Wettability alteration of oil-wet carbonate by silica nanofluid. J. Colloid. Interface Sci. 2016, 461, 435–442. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Torsæter, O. The impact of nanoparticles adsorption and transport on wettability alteration of water wet berea sandstone. In Proceedings of the SPE/IATMI Asia Pacific Oil & Gas Conference and Exhibition, Nusa Dua, Bali, Indonesia, 20–22 October 2015. [Google Scholar]
- Neubauer, E.; Hincapie, R.E.; Borovina, A.; Biernat, M.; Clemens, T.; Ahmad, Y.K. Influence of nanofluids on wettability changes and interfacial tension reduction. In Proceedings of the SPE Europec, Virtual, 1–3 December 2020. [Google Scholar]
- Esfandyari Bayat, A.; Junin, R.; Samsuri, A.; Piroozian, A.; Hokmabadi, M. Impact of metal oxide nanoparticles on enhanced oil recovery from limestone media at several temperatures. Energy Fuels 2014, 28, 6255–6266. [Google Scholar] [CrossRef]
- Hendraningrat, L.; Li, S.; Torsaeter, O. A coreflood investigation of nanofluid enhanced oil recovery in low-medium permeability Berea sandstone. In Proceedings of the SPE International Symposium on Oilfield Chemistry, The Woodlands, TX, USA, 8–10 April 2013. [Google Scholar]
- Wasan, D.; Nikolov, A.; Kondiparty, K. The wetting and spreading of nanofluids on solids: Role of the structural disjoining pressure. Curr. Opin. Colloid. Interface Sci. 2011, 16, 344–349. [Google Scholar] [CrossRef]
- Kondiparty, K.; Nikolov, A.; Wu, S.; Wasan, D. Wetting and spreading of nanofluids on solid surfaces driven by the structural disjoining pressure: Statics analysis and experiments. Langmuir 2011, 27, 3324–3335. [Google Scholar] [CrossRef]
- Ilyas, S.U.; Pendyala, R.; Marneni, N. Stability of nanofluids. In Engineering Applications of Nanotechnology: From Energy to Drug Delivery; Springer: Cham, Switzerland, 2017; pp. 1–31. [Google Scholar]
- Udoh, T.H. Improved insight on the application of nanoparticles in enhanced oil recovery process. Sci. Afr. 2021, 13, e00873. [Google Scholar] [CrossRef]
- Iravani, M.; Khalilnezhad, Z.; Khalilnezhad, A. A review on application of nanoparticles for EOR purposes: History and current challenges. J. Pet. Explor. Prod. Technol. 2023, 13, 959–994. [Google Scholar] [CrossRef]
- Esfandyari Bayat, A.; Junin, R.; Shamshirband, S.; Tong Chong, W. Transport and retention of engineered Al2O3, TiO2 and SiO2 nanoparticles through various sedimentary rocks. Sci. Rep. 2015, 5, 1–12. [Google Scholar] [CrossRef]
- Keykhosravi, A.; Bedrikovetsky, P.; Simjoo, M. Experimental insight into the silica nanoparticle transport in dolomite rocks: Spotlight on DLVO theory and permeability impairment. J. Pet. Sci. Eng. 2022, 209, 109830. [Google Scholar] [CrossRef]
- Emeka, O.J.; Ifeanyi, O.; Chikwe, A.; Nwachukwu, A.; Ndubuisi, O.; Ifeanyi, O.; Chidiebere, O.A. Effect of Application of Aluminium Oxide on Recovery in Enhanced Oil Recovery. Eng. Appl. Sci. 2021, 6, 41–48. [Google Scholar]
- Khilar, K.C.; Fogler, H.S. Migrations of Fines in Porous Media; Springer Science & Business Media: Berlin/Heidelberg, Germany, 1998; Volume 12. [Google Scholar]
- Derkani, M.H.; Fletcher, A.J.; Fedorov, M.; Abdallah, W.; Sauerer, B.; Anderson, J.; Zhang, Z.J. Mechanisms of surface charge modification of carbonates in aqueous electrolyte solutions. Colloids Interfaces 2019, 3, 62. [Google Scholar] [CrossRef]
- Jia, B.; Tsau, J.-S.; Barati, R. A review of the current progress of CO2 injection EOR and carbon storage in shale oil reservoirs. Fuel 2019, 236, 404–427. [Google Scholar] [CrossRef]
- Al-Shargabi, M.; Davoodi, S.; Wood, D.A.; Rukavishnikov, V.S.; Minaev, K.M. Carbon dioxide applications for enhanced oil recovery assisted by nanoparticles: Recent developments. ACS Omega 2022, 7, 9984–9994. [Google Scholar] [CrossRef] [PubMed]
- Peng, C.; Crawshaw, J.P.; Maitland, G.C.; Trusler, J.M.; Vega-Maza, D. The pH of CO2-saturated water at temperatures between 308 K and 423 K at pressures up to 15 MPa. J. Supercrit. Fluids 2013, 82, 129–137. [Google Scholar] [CrossRef]
- Kasprzyk-Hordern, B. Chemistry of alumina, reactions in aqueous solution and its application in water treatment. Adv. Colloid. Interface Sci. 2004, 110, 19–48. [Google Scholar] [CrossRef] [PubMed]
- Derkani, M.H.; Fletcher, A.J.; Abdallah, W.; Sauerer, B.; Anderson, J.; Zhang, Z.J. Low salinity waterflooding in carbonate reservoirs: Review of interfacial mechanisms. Colloids Interfaces 2018, 2, 20. [Google Scholar] [CrossRef]
- Bayat, A.E.; Junin, R.; Ghadikolaei, F.D.; Piroozian, A. Transport and aggregation of Al2O3 nanoparticles through saturated limestone under high ionic strength conditions: Measurements and mechanisms. J. Nanoparticle Res. 2014, 16, 1–12. [Google Scholar] [CrossRef]
- Cacua, K.; Ordoñez, F.; Zapata, C.; Herrera, B.; Pabón, E.; Buitrago-Sierra, R. Surfactant concentration and pH effects on the zeta potential values of alumina nanofluids to inspect stability. Colloids Surf. A Physicochem. Eng. Asp. 2019, 583, 123960. [Google Scholar] [CrossRef]
- Nguyen, A.V.; Evans, G.M.; Jameson, G.J. Electrical Double-Layer Interaction between Spherical Particles. In Encyclopedia of Surface and Colloid Science; CRC Press: Boca Raton, FL, USA, 2015; pp. 2017–2027. [Google Scholar]
- Sjoeberg, E.L.; Rickard, D.T. Calcite dissolution kinetics: Surface speciation and the origin of the variable pH dependence. Chem. Geol. 1984, 42, 119–136. [Google Scholar] [CrossRef]










| Core ID | Diameter (mm) | Length (mm) | Gas Permeability (mD) | Pore Volume (cc) | Porosity (%) |
|---|---|---|---|---|---|
| IDLS_1 | 25.52 | 50.78 | 229.444 | 4.515 | 17.38 |
| IDLS_2 | 25.33 | 50.78 | 212.025 | 4.405 | 17.21 |
| IDLS_3 | 25.27 | 50.82 | 208.178 | 4.315 | 16.93 |
| Material | Concentration (wt%) |
|---|---|
| α-Al2O3 nanoparticles | 0.1 |
| NaCl salt | 0.1 |
| Concentrated HCl acid | 0.007 |
| CTAB surfactant | 0.03 |
| Experiment’s Description | Effluent’s Average pH | Original Gas Permeability (mD) | Post Flooding Gas Permeability (MD) | Permeability Reduction Percentage |
|---|---|---|---|---|
| Nanofluid alternating CO2 injection | 6.5 | 229.444 | 176.14 | 23.23% |
| Nanofluid injection | 7.6 | 212.025 | 111.23 | 47.54% |
| Low-pH nanofluid injection | 7.2 | 208.178 | 143.61 | 31.02% |
| Experiment’s Description | Oil Recovery Due to Low-Salinity Water Injection (%) | Oil Recovery Due to Nanofluid and Sweeping Fluid Injections (%) | Overall Oil Recovery |
|---|---|---|---|
| Nanofluid alternating CO2 injection | 40.03 | 10.93 | 50.93 |
| Nanofluid injection | 38.79 | 14.13 | 52.92 |
| Low-pH nanofluid injection | 37.77 | 15.11 | 52.88 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alali, R.; Abe, K.; Seddiqi, K.N.; Fujii, H. Effect of Carbon Dioxide Injection on Limestone Permeability Damage Induced by Alumina Nanoparticles for Enhanced Oil Recovery Applications. Appl. Sci. 2023, 13, 7446. https://doi.org/10.3390/app13137446
Alali R, Abe K, Seddiqi KN, Fujii H. Effect of Carbon Dioxide Injection on Limestone Permeability Damage Induced by Alumina Nanoparticles for Enhanced Oil Recovery Applications. Applied Sciences. 2023; 13(13):7446. https://doi.org/10.3390/app13137446
Chicago/Turabian StyleAlali, Ragheed, Kazunori Abe, Khawaja Naweed Seddiqi, and Hikari Fujii. 2023. "Effect of Carbon Dioxide Injection on Limestone Permeability Damage Induced by Alumina Nanoparticles for Enhanced Oil Recovery Applications" Applied Sciences 13, no. 13: 7446. https://doi.org/10.3390/app13137446
APA StyleAlali, R., Abe, K., Seddiqi, K. N., & Fujii, H. (2023). Effect of Carbon Dioxide Injection on Limestone Permeability Damage Induced by Alumina Nanoparticles for Enhanced Oil Recovery Applications. Applied Sciences, 13(13), 7446. https://doi.org/10.3390/app13137446







