Abstract
Lipids are a diverse group of organic compounds that serve essential roles due to their biological functions for all prokaryotic and eukaryotic organisms. Despite the fundamental role of lipid class in plants, there is still a relatively low level of knowledge regarding the composition of grape lipids. This research represents the first investigation into the lipids of the Croatian grape variety ‘Maraština’ (Vitis vinifera L.), employing a targeted approach and the UHPLC-MS/MS method to conduct a comprehensive analysis of the lipid profile of grape skins. The study investigated the profile of free fatty acids, glycerolipids, glycerophospholipids, triterpenoids, sphingolipids, and free fatty acid esters in eleven vineyards located along the cultivation area of ‘Maraština’ variety (Dalmatia, Croatia). Grape skins from vineyards that were exposed to more sunlight and higher daily temperature during the vegetation period mainly resulted in a richer profile of free fatty acids. Among lipids, ‘Maraština’ grape skins were characterized mainly by a triterpenoid compound; oleanolic acid (249.49 µg/g of FW), and free fatty acids, especially palmitic acid C16:0 (26.09 µg/g of FW), and lignoceric acid C24:0 (12.40 µg/g of FW). Hierarchical clustering analysis represented valuable insights into the diversity of lipids in ‘Maraština’ grape skin.
1. Introduction
Lipids encompass a broad category of organic compounds known for their diversity. According to their chemical building blocks they are classified into eight categories: fatty acyls, glycerolipids, glycerophospholipids, sphingolipids, saccharolipids, polyketides, sterols, and prenol lipids [1]. Lipids possess diverse functions, including serving as a source of energy, contributing to structural elements, regulating biological processes, participating in signaling, and facilitating cellular communication [2,3]. Considering structural diversity, each of these categories encompasses different classes and sub-classes, thereby contributing to the overall complexity of lipids. Among plant cells, the most prevalent lipids are derived from the biosynthetic pathways of fatty acids and glycerolipids. Other groups of lipids are derived from the isoprenoid pathway, such as sterols [4].
The lipid content in grapes ranges from 0.15% to 0.24% of the fresh weight [5]. A study by Higgins and Peng identified the presence of glucolipids, phospholipids, and neutral lipids in both the skins and pulp of Condour grapes back in 1976 [6]. Recently, Garcia-Aloy et al. [7] published work about the comprehensive characterization of the lipidome in Ribolla Gialla grape berries and discovered previously unreported lipid compounds in grapes. The lipidome of the grape berry was found to be complex, with a diverse range of lipid compounds belonging to various lipid classes. These lipids were observed in different compartments of the grape berry and at different stages of maturation. Notably, the presence of methylated phosphatidic acid in grape samples was reported for the first time, suggesting a novel finding. An atypical lipid class known as acylated monogalactosyldiacylglycerols was also identified. These observations highlight the diverse and unique lipid composition of grapes. Lipid concentrations in grapes are affected by climatic conditions such as light exposure [8]. Of the geographical parameters in a wine-growing region, the climate is an important determinant of grape and wine composition [9]. Another study showed that grape berries from the Alvarinho white variety are more photosynthetically active when exposed to a higher number of sunlight hours, especially at the green phase of ripeness [10]. Irrespective of skin color, the most abundant unsaturated fatty acids (UFA) found in Vitis vinifera berries are the C18 acids, specifically linoleic acid (C18:2), oleic acid (C18:1), and linolenic acid (C18:3) [11,12]. The presence and levels of free fatty acids, both unsaturated and saturated, play a crucial role in influencing the production of volatile compounds by wine yeast [13]. During alcoholic fermentation, yeasts utilize only fatty acids in free form but not complex lipids with bounded fatty acids [14]. Furthermore, the lipid composition of grapes is essential in yeast cells because it stimulates the growth of yeasts and affects their metabolism in alcoholic fermentation [15]. The membrane structure of yeast significantly influences stress tolerance and resistance to substances such as acetic acid, ethanol, or acetaldehyde. In addition to encountering stressful environments, yeast cells have developed various molecular pathways to enable them to respond and adapt to changes in their surroundings [16].
Although many analytical techniques for the determination of grape lipidomics have been developed [17,18], they often involve time-consuming preparation procedures. Mass spectrometry has been widely utilized for the analysis of specific lipids. Gas chromatography with mass spectrometry (GC-MS) platform is commonly applied for lipid analysis, particularly for compounds like free fatty acids and steroids, which often require the derivatization of compounds in the sample matrix [19]. The development of high-resolution MS soft ionization methods has greatly facilitated the detection of compounds belonging to the cellular lipidome [20]. In recent years, due to the chemical complexity of grape-wine matrices and the lipid structural diversity and complexity, mass spectrometry (MS) in tandem with ultra-high-performance liquid chromatography (UHPLC) has become the innovative platform for detailed lipidomic analysis [2,21]. UHPLC-MS/MS method has good sensitivity, specificity, and dynamic range [22].
This study focused on the lipid composition of V. vinifera L. cv. ‘Maraština’ berry skin from 11 vineyards located along a cultivation area considering the ‘Maraština’ as one of the significant indigenous white grape cultivars in Dalmatia. According to our knowledge, there are no reports on grape lipidomic in this variety. The objective was to investigate the comprehensive profile of lipids in ‘Maraština’ grape skins using a targeted approach by UHPLC-MS/MS. These results will be valuable for further research related to non-Saccharomyces yeasts isolated from cv. ‘Maraština’ [23].
2. Materials and Methods
2.1. Chemicals
The following chemicals purchased by Sigma-Aldrich (Milan, Italy) were utilized for UHPLC analysis: methanol (CH3OH, LC-MS grade), acetonitrile (CH3CN, LS-MS grade), chloroform (CHCl3), and 2-propanol (CH3CH(OH)CH3). Fluka-Sigma-Aldrich (Milan, Italy) provided LC-MS grade formic acid (HCOOH) and ammonium formate (NH4COOH) for the study. Aldrich-Fluka-Sigma S.r.l. (Milan, Italy) supplied the internal standards stearic acid d3 and cholesterol, along with the antioxidant butylated hydroxytoluene.
2.2. Vineyard Site and Grape Samples
The grapes were selected from the germplasm collection of indigenous cultivars planted at the Institute for Adriatic Crops and Karst Reclamation and 10 vineyards in the 2021 vintage. Vineyards represented the whole cultivation area of the V. vinifera cultivar, ‘Maraština’ in Dalmatia (Croatia, Figure 1). Five vineyards are positioned in the northern part of Dalmatia: M1, M2, M3, M4, and M5. The vineyards M6 and M7 are located in the central part of Dalmatia; and the M8, M9, M10, and M11 are situated on the island of Korčula, southern Dalmatia. Table S1 provides detailed data for the characteristics of the vineyard site and climate data specific to the vineyards during the 2021 vintage are reported in Table S2. Healthy grapes were harvested in three biological replicates at technological maturity, glucose to fructose ratio equal to one. Detailed data about grapes are represented in Table S3. Three randomized rows of vines were chosen for grape picking in each vineyard. The nine representative bunches, approximately 3 kg, were collected from three distinct vines, representing the top, middle, and end of the row. Each set of three vines constituted one sample, and the samples were promptly transported to the laboratory.
Figure 1.
The geographical location of eleven ‘Maraština’ vineyards in Dalmatia, Croatia.
2.3. Samples Preparation for Lipid Extraction
In the laboratory, 100 grape berries were selected from various sections of grape in each batch and stored at −80 °C. The frozen grape skins were separated from berries using a pincette and prepared for homogenization into a powder. The powder was prepared with an IKA A11 basic homogenizer (Staufen, BW, Germany) in a bath under liquid nitrogen. Folch’s method [22,24] with the modifications described by Chitarrini [25] was used for lipids extraction. A precise amount of 0.100 (±0.005) g of the powder was weighed and transferred to the Eppendorf tube. Subsequently, 0.3 mL of CH3OH was added to the tube. Samples were vortexed for 30 s. Afterwards, 0.6 mL of CHCl3 containing butylated hydroxytoluene (BHT) at a concentration of 500 mg/L was added to the mixture and followed by the addition of 10 µL of internal standard (stearic acid d3 at 100 µL/mL). In the next step, the samples were subjected to orbital shaking for a duration of 60 min. Furthermore, 0.25 mL of Mili-Q water was added to the samples. After a 10 min interval, the samples were centrifuged at 3600 rpm for 10 min at 4 °C using a 3–30k Refrigerated Centrifuge (Sigma, Darmstadt, Germany). The lipid-rich layer was carefully collected. The extraction was repeated by adding 0.4 mL of CHCl3 (with BHT)/ CH3OH/H2O in the ratio of 86:14:1 v/v/v. Following the repeated centrifugation at 3600 rpm for 10 min at 4 °C, the total lipid-rich layer (lower phase) was collected and combined with the previously collected fraction. United lipid fractions were evaporated to dryness under N2. Before UHPLC-MS/MS analysis samples were reconstituted in a solution of 300 µL CH3CN/CH3CH(OH)CH3/H2O; 65:30:5 v/v/v which contained the 1 µg/mL of internal standard cholesterol [2].
2.4. UHPLC-MS/MS Analysis of Lipids in Grape Skins
The analysis of lipids was performed using a UHPLC-MS/MS system consisting of a Dionex 3000 UHPLC (Thermo Fisher Scientific, Dreieich, Germany) coupled with an API 5500 triple-quadrupole mass spectrometer (Applied Biosystems/MDS Sciex, Toronto, ON, Canada). The system was equipped with an electrospray source and an autosampler (Dionex Thermo Fisher Scientific, Dreieich, Germany). The chromatographic separation was carried out on a reversed-phase column, Ascentis Express C18 (150 mm × 2.1 mm, 2.7 µm; Sigma, Milan, Italy) maintained at 55 °C [22]. The flow rate was set at 0.26 mL/min, and a sample injection volume of 5 µL was used at 10 °C. The mobile phase used in the analysis consisted of two solvents. Solvent A was prepared by mixing 40% CH3CN in water, along with NH4COOH at a concentration of 10 mM, and HCOOH at a concentration of 0.1%. Solvent B was prepared by mixing 90% CH3CH(OH)CH3 with 10% CH3CN, and also containing NH4COOH at a concentration of 10 mM and HCOOH at a concentration of 0.1%. The instrument control and data acquisition processing was carried out using Analyst™ software version 1.6.1, developed by Applera Corporation, located in Norwalk, CT, USA. Calibration curves were constructed for quantification and based on selected chemical standards. Data were expressed as µg/g of FW after normalization based on the internal standard stearic acid d3. The lipids were detected using the multiple-reaction monitoring (MRM) mode, which offers high selectivity and sensitivity of the ionization method. Compounds were identified based on their reference standards, retention times, and qualifier and quantifier ions. The specific details of compound identification can be found in Table S4 of the referenced study [26].
2.5. Data Analysis
The statistical data analysis was conducted using IBM®SPSS® Statistics for Windows, version 23.0, developed by SPSS Inc. based in Chicago, IL, USA. The lipid compound data obtained from grape skins were subjected to the analysis of variance (ANOVA). Additionally, post hoc multiple comparisons were performed using Tukey’s range test to identify specific pairwise differences between the vineyards. The hierarchical clustering analyses for the metabolomics data were conducted using MetaboAnalyst v.5.0 (University of Alberta, Edmonton, AB, Canada). The specific analysis involved the application of the Ward algorithm and Euclidean distance analysis to generate a heatmap. The heatmap was created to visualize the clustering patterns and relationships among the data points (accessed on 18 March 2023).
3. Results and Discussion
The concentrations of lipid compounds in ‘Maraština’ grape skins determined by a targeted approach with the UHPLC-MS/MS method are shown in Table 1. The detected compounds were categorized into different chemical-lipid classes. In total, 19 compounds were identified, which including free fatty acids (10), triterpenoid (1), glycerolipid (1), glycerophospholipids (2), sphingolipid (1), and free fatty acid esters (4).

Table 1.
Concentration (µg/g of FW) of fatty acids, triterpenoid, glycerolipid, glycerophospholipids, sphingolipid, and free fatty acid esters in ‘Maraština’ grape skin for eleven vineyards.
3.1. Free Fatty Acid Composition
Regarding saturation degree, ‘Maraština’ grape skins showed a higher abundance of saturated fatty acids (SFAs) than unsaturated fatty acids (UFAs). A total of six were saturated fatty acids: myristic acid, palmitic acid, stearic acid, arachidic acid, behenic acid, and lignoceric acid. Saturated fatty acids were in higher concentration than unsaturated fatty acids, especially palmitic acid (13.46–24.01 µg/g of FW) followed by lignoceric acid (6.41–12.40 µg/g of FW), and stearic acid (6.57–9.91 µg/g of FW). In comparison to the results reported by Santos et al. [12] for the V. vinifera cultivars (Benitaka and Brazil), our study found that the ‘Maraština’ grape skin had higher concentrations of myristic acid and lower levels of stearic acid, palmitic acid, behenic acid, and arachidic acid. When we look through the prism of vineyards, the statistically different SFAs were behenic acid (vineyard M11), lignoceric acid (vineyard M4), arachidic acid (vineyard M8), myristic acid (vineyard M6), and palmitic acid (vineyard M8). From this trend, it is obvious that vineyards from the Dalmatian southern part which had higher daily temperatures during vintage 2021 showed higher concentrations of SFAs. This observation is opposite to a previous study on the white variety where Garrido and coworkers [8] showed that a higher level of sunlight during grape maturation had an impact on decreasing fatty acid in grapes. The selected sampling area has a Mediterranean climate. Although the vineyards are located in the same climate zone, their microclimate differences are obvious. Besides sunshine hours, vineyards from the central and southern parts of Dalmatia had higher daily temperatures, especially vineyards located on the island of Korčula (M8, M9, M10, and M11). Indeed, the influence of vineyard factors such as geographical position, agricultural practices, and soil type on the synthesis of fatty acids in grape skins is known. Although these parameters were not specifically correlated in our research, it is important to highlight the impact on the lipid composition of grapes.
On the other side, the UFA grape skin profile was mainly characterized by C18 fatty acids; oleic acid + cis vaccenic acid (0.85–1.95 µg/g of FW), linoleic acid (0.60–0.81 µg/g of FW), linolenic acid (0.32–0.89 µg/of FW), and one C16 fatty acid; palmitoleic acid (0.44–1.54 µg/g of FW). Linolenic acid was the only compound with a statistical difference, the highest concentration was quantified in vineyard M6. Grape skins from vineyard M7 had a lower concentration of free fatty acids (38.34 µg/g of FW) even though the vineyard M7 is located in a central part of Dalmatia with more sunlight and high average daily temperatures. The only difference and possible cause of this is the higher altitude (94 m above sea) in comparison with another vineyard from the central part of Dalmatia (M6–14 m above sea). The analysis of ‘Maraština’ grape skins revealed that free fatty acids are prominent components among the total lipids, which is consistent with findings from other studies [2,11]. In comparison with some other results, ‘Maraština’ grape skin had a similar concentration of total free fatty acids (38.34–52.63 µg/g of FW) like Albillo Dorado (48.52 µg/g of FW) and Bobal Blanca (45.21 µg/g of FW) white variety, but more than Airén white variety (27.78 µg/g of FW) [2]. Also, our results agree with the most recent study [7] on untargeted grape lipidomic profiling confirmed that free fatty acids with 12 and 16 carbon atoms are most common in grape lipidome. Furthermore, Perez-Navarro and coworkers did not reveal the dependence of lipid concentration on grape color. Duan and coworkers [27] suggested that the rational addition of mixtures of unsaturated fatty acids to grape juice can enhance yeast growth and result in a more diverse volatile profile in wine. This can include higher alcohols, acetate esters (such as isoamyl acetate and 2-phenylethyl acetate), and ethyl esters. Linoleic and linolenic acids participate in the lipoxygenase-hydroperoxide lyase pathway, where they serve as precursors for the synthesis of aldehydes and alcohols with six carbons [28]. In addition, alcohols, esters, aldehydes, as well as linoleic and linolenic acids, are involved in the production of volatile thiols. These volatile thiols are responsible for carrying varietal aromas and contribute to the tropical notes in the aroma of wine [29]. The importance of unsaturated fatty acids from grape skin is also manifested in anaerobic conditions in fermentation because fermentation suppresses the yeast’s fatty acid desaturation. The alternative option is the direct uptake of unsaturated fatty acids from grape juice. The addition of oxygen at the end of the growth phase can regulate the process of alcoholic fermentation and promote the synthesis of sterols and UFAs [30]. UFAs are crucial for yeast adaption to fermentation stress and fluidity of membranes, especially one from grapes because in the beginning the biosynthesis of fatty acids can be inhibited by a high content of sugar [31]. Increasing the incorporation of UFAs into yeast cell walls can enhance yeast biomass and provide valuable support during fermentation stresses, such as high sugar levels and ethanol toxicity [32,33]. However, further research is necessary to investigate how different yeast utilize lipids from the ‘Maraština’ grape and their effect on the volatile profile of wine for a better understanding of potential aroma improvement.
3.2. Triterpenoid, Glycerolipid, Glycerophospholipids, Sphingolipid, and Free Fatty Acid Esters Composition
Plants are known for their abundant production of specialized metabolites that fulfil specific roles in adaption to environmental conditions. Indeed, triterpenoids are a prominent subclass of specialized metabolites that play crucial roles in plant defence mechanisms [34]. They are formed from squalene molecules. Oleanolic acid is a pentacyclic triterpenoid and is the most abundant compound in the lipid profile of ‘Maraština’ grape skin (170.15–257.82 µg/g of FW) (Table 1) without statistical differences among observed vineyards. The concentration in the grape skin of white and red varieties was much lower (38.63–57.58 µg/g of FW) than in the ‘Maraština’ variety [2]. Oleanolic acid is a survival factor for yeast cells in conditions with a lack of oxygen and increases the biomass of yeast cells [35]. This presence of this component, unaffected by vineyard, is a notable characteristic of the ‘Maraština’ variety. It is possible that oleanolic acid plays a crucial role in facilitating the survival of the natural microbiota and contributing to the successful completion of the spontaneous fermentation process, as reported in a previous study [36].
Glycerolipids are mono-, di-, and trisubstituted glycerols and represent a large group of biological molecules in plant cells acting as structural and signaling molecules in intracellular processes. The only glycerolipid found in grape skin is 1-linoleoyl-rac-glycerol in a concentration between 1.24 to 3.28 µg/g of FW. ‘Maraština’ grape skin is characterized by a higher concentration of 1-linoleoyl-rac-glycerol than other white varieties such as Airen and Bobal Blanc [2]. Glycerolipids have been acknowledged as a compound group that exhibits elevated levels in grapes cultivated under higher sunlight exposure, which is related to the vineyards M7, M8, and M9 because they had the highest concentration. Garrido and coworkers reported the impact of low and high light, where high light led to the increase in ceramides, triacylglycerols, and glycerophospholipids at a mature stage [8]. In a similar situation as with free fatty acids, the M6 vineyard, situated at a higher altitude, is characterized by the lowest concentration of glycerolipids (1.24 µg/g of FW) and glycerophospholipids (0.49 µg/g of FW).
One of the primary structural constituents of yeast membranes is glycerophospholipids. They are involved in membrane fluidity and signal transduction [37]. The content of two identified and quantified glycerophospholipids in ‘Maraština’ grape skin reach similar concentrations: 0.17–0.54 µg/g of FW for 1,2-dioleoyl-sn-glycerol-3-phosphocholine and 0.27–0.50 µg/g of FW for 1,2-dioleoyl-sn-glycero-3-phospho-rac-(1-glycerol) sodium salt without statistical differences between the vineyards. Furthermore, these two glycerophospholipids can serve as precursors for the synthesis of the sphingolipids and sterols, which are essential for yeast growth and viability, impacting directly on alcoholic fermentation.
Ceramides are a family of waxy lipid molecules that are bioactive sphingolipids. In this study, they are identified and quantified in concentrations of 0.01–0.03 µg/g of FW. They are the least abundant group in lipid content and ‘Maraština’ grape skins from vineyard M7 had the highest concentration.
Fatty acid ethyl esters (FAEEs) are more hydrophobic than triglycerides [38]. The most abundant FAEE quantified and identified in ‘Maraština’ grape skins is ethyl palmitate (0.15–0.39 µg/g of FW), followed by ethyl stearate (0.20–0.43 µg/g of FW), ethyl oleate (0.08–0.31 µg/g of FW), and ethyl linoleate (0.06 µg/g of FW). The only FAEE that showed statistical differences is ethyl palmitate with the highest concentration in grape skin from vineyard M10.
3.3. Multivariate Statistical Analysis
Hierarchical clustering analyses were performed using nine lipid compounds in grape skins with a statistical difference among investigated ‘Maraština’ vineyards. The heatmap generated from the data offered a visually informative representation of the varied composition of grape skin lipids in ‘Maraština’ grapes sourced from different vineyards. It is obvious that the main differences among vineyards are manifested in the content of free fatty acids, which are mainly most abundant in vineyards from the central and southern parts of Dalmatia: M10, M8, M7, M11, and M9. The exception is vineyard M6 (Figure 2; orange color), situated at a higher altitude in central Dalmatia, with smaller contents of free fatty acids, glycerolipids, and glycerophospholipids in the ‘Maraština’ grape skin.
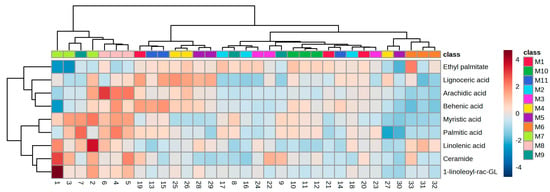
Figure 2.
Hierarchical clustering analysis of the ‘Maraština’ grape skin purchased from the 11 different vineyards obtained by UHPLC-MS/MS analysis. Lipids are represented in the rows, and the columns show samples. The chromatic scale represents the abundance of lipids in samples from dark blue (minimum) to dark red (maximum). 1-linoleoyl-rac-GL—1-linoleoyl-rac-glycerol.
4. Conclusions
This study demonstrates the first results of lipid analysis in the ‘Maraština’ grape. The UHPLC-MS/MS method was applied to identify and quantify various classes of lipids, including free fatty acids, triterpenoids, glycerophospholipids, glycerolipids, sphingolipids, and free fatty acid esters. This method was chosen for its high sensitivity, specificity, and dynamic range, ensuring the reliable analysis of the detailed lipid profiles. Grape skins of the ‘Maraština’ variety were mainly characterized by oleanolic acid belonging to the triterpenoid class, and free fatty acids with a higher concentration of saturated fatty acids than unsaturated fatty acids, especially palmitic acid, and lignoceric acid. Grape skins from vineyards that were exposed to high sunlight and higher daily temperatures throughout the vegetation year mainly resulted in a richer profile of free fatty acids. The obtained results contributed to the characterization of the ‘Maraština’ grape composition. Additionally, the identified lipids will be used for further research on the interactions between lipids and fermentation metabolites produced by isolated non-Saccharomyces yeasts from ‘Maraština’ grapes.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/app13116446/s1, Table S1: General vineyard parameters; Table S2: Climate data for eleven vineyards during vintage 2021 (January–September); Table S3: Physicochemical characteristics of fresh musts obtained from Maraština samples collected from 11 different vineyards; Table S4: Mass transitions (MRM) and instrumental parameters of LC-ESI-MS/MS method.
Author Contributions
Conceptualization, A.B. and I.B.-L.; methodology, A.B., D.M., U.V. and I.B.-L.; formal analysis, A.B. and D.M.; investigation, A.B. and I.B.-L.; resources, U.V. and I.B.-L.; writing—original draft preparation, A.B.; writing—review and editing, D.M., U.V. and I.B.-L.; visualization, U.V. and I.B.-L.; supervision, U.V. and I.B.-L.; project administration, I.B.-L.; funding acquisition, I.B.-L. All authors have read and agreed to the published version of the manuscript.
Funding
This study was supported by the Croatian Science Foundation under the project IP-2020-02-1872: “Impact of native non-Saccharomyces wine yeast on wine aromas” (WINE AROMAS), “Young Researchers’ Career Development Project—Training New Doctoral Students”, and with an STSM grant obtained from COST Action CA 17111 INTEGRAPE (2018–2022).
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Acknowledgments
Authors would like to thank the wine producers from Dalmatia for donating the ‘Maraština’ grape.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Fahy, E.; Cotter, D.; Sud, M.; Subramaniam, S. Lipid classification, structures and tools. BBA-Mol. Cell Biol. Lipids 2011, 1811, 637–647. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Navarro, J.; Da Ros, A.; Masuero, D.; Izquierdo-Cañas, P.M.; Hermosín-Gutiérrez, I.; Gómez-Alonso, S.; Mattivi, F. LC-MS/MS analysis of free fatty acid composition and other lipids in skins and seeds of Vitis vinifera grape cultivars. Int. Food Res. J. 2019, 125, 108556. [Google Scholar] [CrossRef] [PubMed]
- Subramaniam, S.; Fahy, E.; Gupta, S.; Sud, M.; Byrnes, R.W.; Cotter, D.; Maurya, M.R. Bioinformatics and systems biology of the lipidome. Chem. Rev. 2011, 111, 6452–6490. [Google Scholar] [CrossRef]
- Ohlrogge, J.; Browse, J. Lipid biosynthesis. Plant Cell 1995, 7, 957–970. [Google Scholar] [PubMed]
- Gallander, J.F.; Peng, A.C. Lipid and fatty acid compositions of different grape types. Am. J. Enol. Vitic. 1980, 31, 24–27. [Google Scholar] [CrossRef]
- Higgins, P.A.; Peng, A.C. Lipid composition of ‘Concord’ grapes. Am. J. Enol. Vitic. 1976, 27, 32–35. [Google Scholar] [CrossRef]
- Garcia-Aloy, M.; Masuero, D.; Chitarrini, G.; Škrab, D.; Sivilotti, P.; Guella, G.; Vrhovsek, U.; Franceschi, P. Untargeted lipidomic profiling of grapes highlights the importance of modified lipid species beyond the traditional compound classes. Food Chem. 2023, 410, 135360. [Google Scholar] [CrossRef]
- Garrido, A.; Conde, A.; De Vos Ric, C.H.; Cunha, A. The influence of light microclimate on the lipid profile and associated transcripts of photosynthetically active grape berry seeds. Front. Plant Sci. 2023, 13, 1022379. [Google Scholar] [CrossRef]
- van Leeuwen, C.; Barbe, J.C.; Darriet, P.; Geffroy, O.; Gomès, E.; Guillaumie, S.; Helwi, P.; Laboyrie, J.; Lytra, G.; Le Menn, N. Recent advancements in understanding the terroir effect on aromas in grapes and wines. Oeno One 2020, 54, 985–1006. [Google Scholar] [CrossRef]
- Garrido, A.; Serôdio, J.; De Vos, R.; Conde, A.; Cunha, A. Influence of foliar kaolin application and irrigation on photosynthetic activity of grape berries. Agronomy 2019, 9, 685. [Google Scholar] [CrossRef]
- Ancin, C.; Ayestaran, B.; Garcia, A.; Garrido, J. Evolution of fatty acid contents in Garnacha and Viura musts during fermentation and the aging of wine. Eur. Food Res. Technol. 1998, 206, 143–147. [Google Scholar]
- Santos, L.P.; Morais, D.R.; Souza, N.E.; Cottica, S.M.; Boroski, M.; Visentainer, J.V. Phenolic compounds and fatty acids in different parts of Vitis labrusca and V. vinifera grapes. Int. Food Res. J. 2011, 44, 1414–1418. [Google Scholar] [CrossRef]
- Varela, C.; Torrea, D.; Schmidt, S.A.; Ancin-Azpilicueta, C.; Henschke, P.A. Effect of oxygen and lipid supplementation on the volatile composition of chemically defined medium and Chardonnay wine fermented with Saccharomyces cerevisiae. Food Chem. 2012, 135, 2863–2871. [Google Scholar] [CrossRef] [PubMed]
- Dyer, J.M.; Chapital, D.C.; Kuan, J.W.; Mullen, R.T.; Pepperman, A.B. Metabolic engineering of Saccharomyces cerevisiae for production of novel lipid compounds. Appl. Microbiol. Biotechnol. 2002, 59, 224–230. [Google Scholar]
- Tesnière, C. Importance and role of lipids in wine yeast fermentation. Appl. Microbiol. Biotechnol. 2019, 103, 8293–8300. [Google Scholar] [CrossRef] [PubMed]
- Grillitsch, K.; Connerth, M.; Köfeler, H.; Arrey, T.N.; Rietschel, B.; Wagner, B.; Karas, M.; Daum, G. Lipid particles/droplets of the yeast Saccharomyces cerevisiae revisited: Lipidome meets Proteome. BBA-Mol. Cell Biol. Lipids 2011, 1811, 1165–1176. [Google Scholar] [CrossRef]
- Gallart, M.; Francioli, S.; Viu-Marco, A.; López-Tamames, E.; Buxaderas, S. Determination of free fatty acids and their ethyl esters in musts and wines. J. Chromatogr. A 1997, 776, 283–291. [Google Scholar] [CrossRef]
- Abdulkadir, S.; Tsuchiya, M. One-step method for quantitative and qualitative analysis of fatty acids in marine animal samples. J. Exp. Mar. Biol. 2008, 354, 1–8. [Google Scholar] [CrossRef]
- Sjovall, P.; Lausmaa, J.; Johansson, B. Mass spectrometric imaging of lipids in brain tissue. J. Anal. Chem. 2004, 76, 4271–4278. [Google Scholar] [CrossRef]
- Shevchenko, A.; Simons, K. Lipidomics: Coming to grips with lipid diversity. Nat. Rev. 2010, 11, 593–598. [Google Scholar] [CrossRef]
- Wu, B.; Wei, F.; Xu, S.; Xie, Y.; Lv, X.; Chen, H.; Huang, F. Mass spectrometry-based lipidomics as a powerful platform in foodomics research. Trends Food Sci. Technol. 2021, 107, 358–376. [Google Scholar] [CrossRef]
- Della Corte, A.; Chitarrini, G.; Di Gangi, I.M.; Masuero, D.; Soini, E.; Mattivi, F.; Vrhovek, U. A rapid LC-MS/MS method for quantitative profiling of fatty acids, sterols, glycerolipids, glycerophospholipids and sphingolipids in grapes. Talanta 2015, 140, 53–61. [Google Scholar] [CrossRef]
- Milanović, V.; Cardinali, F.; Ferrocino, I.; Boban, A.; Franciosa, I.; Gajdoš Kljusurić, J.; Mucalo, A.; Osimani, A.; Aquilanti, L.; Garofalo, C.; et al. Croatian white grape variety Maraština: First taste of its indigenous mycobiota. Food Res. Int. 2022, 162, 111917. [Google Scholar] [CrossRef]
- Folch, J.; Lees, M.; Sloane, G.H. A simple method for isolation and purification of total lipids from animal tissues. J. Biol. Chem. 1953, 226, 497–509. [Google Scholar] [CrossRef]
- Chitarrini, G.; Zulini, L.; Masuero, D.; Vrhovšek, U. Lipid, phenol and carotenoid changes in ‘Bianca’ grapevine leaves after mechanical wounding: A case study. Protoplasma 2017, 254, 2095–2106. [Google Scholar] [CrossRef] [PubMed]
- Lukic, I.; DaRos, A.; Guella, G.; Camin, F.; Masuero, D.; Mulinacci, N.; Vrhovsek, U.; Mattivi, F. Lipid profiling and stable isotopic data analysis for differentiation of extra virgin olive oils based on their origin. Molecules 2020, 25, 5. [Google Scholar]
- Duan, L.L.; Shi, Y.; Jiang, R.; Yang, Q.; Wang, Y.Q.; Liu, P.T. Effects of adding unsaturated fatty acids on fatty acid composition of Saccharomyces cerevisiae and major volatile compounds in wine. S. Afr. J. Enol. Vitic. 2015, 36, 285–295. [Google Scholar] [CrossRef]
- Shahidi, F.; Hossain, A. Role of Lipids in Food Flavor Generation. Molecules 2022, 27, 5014. [Google Scholar] [CrossRef] [PubMed]
- Tumanov, S.; Pinu, F.; Greenwood, D.; Villas-Bôass, S. Effect of free fatty acids and lipolysis on Sauvignon Blanc fermentation. Aust. J. Grape Wine Res. 2018, 24, 398–405. [Google Scholar] [CrossRef]
- Ochando, T.; Mouret, J.R.; Humbert-Goffard, A.; Sablayrolles, J.M.; Farines, V. Impact of initial lipid content and oxygen supply on alcoholic fermentation in champagne-like musts. Food Res. Int. 2017, 98, 87–94. [Google Scholar] [CrossRef]
- Salmon, J.; Barre, P. Improvement of nitrogen assimilation and fermentation kinetics under enological conditions by derepression of alternative nitrogen-assimilatory pathways in an industrial Saccharomyces cerevisiae strain. Appl. Environ. Microbiol. 1998, 64, 3831–3837. [Google Scholar] [CrossRef] [PubMed]
- You, K.M.; Rosenfield, C.L.; Knipple, D.C. Ethanol tolerance in the yeast Saccharomyces cerevisiae is dependent on cellular oleic acid content. AEM 2003, 69, 1499–1503. [Google Scholar] [CrossRef] [PubMed]
- Yan, G.L.; Duan, L.L.; Liu, P.T.; Duan, C.Q. Transcriptional Comparison Investigating the Influence of the Addition of Unsaturated Fatty Acids on Aroma Compounds During Alcoholic Fermentation. Front Microbiol. 2019, 10, 1115. [Google Scholar] [CrossRef]
- Cárdenas, P.D.; Almeida, A.; Bak, S. Evolution of Structural Diversity of Triterpenoids. Front. Plant Sci. 2019, 10, 1523. [Google Scholar] [CrossRef] [PubMed]
- Ribéreau-Gayon, P.; Dubourdieu, D.; Donèche, B.; Lonvaud, A. Handbook of Enology, the Microbiology of Wine and Vinifications, 2nd ed.; John Wiley & Sons: Chichester, UK, 2006; pp. 91–93. [Google Scholar]
- Boban, A.; Vrhovsek, U.; Carlin, S.; Mucalo, A.; Budić-Leto, I. A Targeted and an Untargeted Metabolomics Approach to the Volatile Aroma Profile of Young ‘Maraština’ Wines. Metabolites 2022, 12, 1295. [Google Scholar] [CrossRef]
- van Meer, G.; Voelker, D.R.; Feigenson, G.W. Membrane lipids: Where they are and how they behave. Nat. Rev. Mol. Cell Biol. 2008, 9, 112–124. [Google Scholar] [CrossRef]
- Musshoff, F.; Wilson, I.D. Encyclopedia of Separation Science, 1st ed.; Academic Press: Oxford, UK, 2000; pp. 1921–1931. [Google Scholar]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).