An Insight into the Chemistry of Cement—A Review
Abstract
1. Introduction
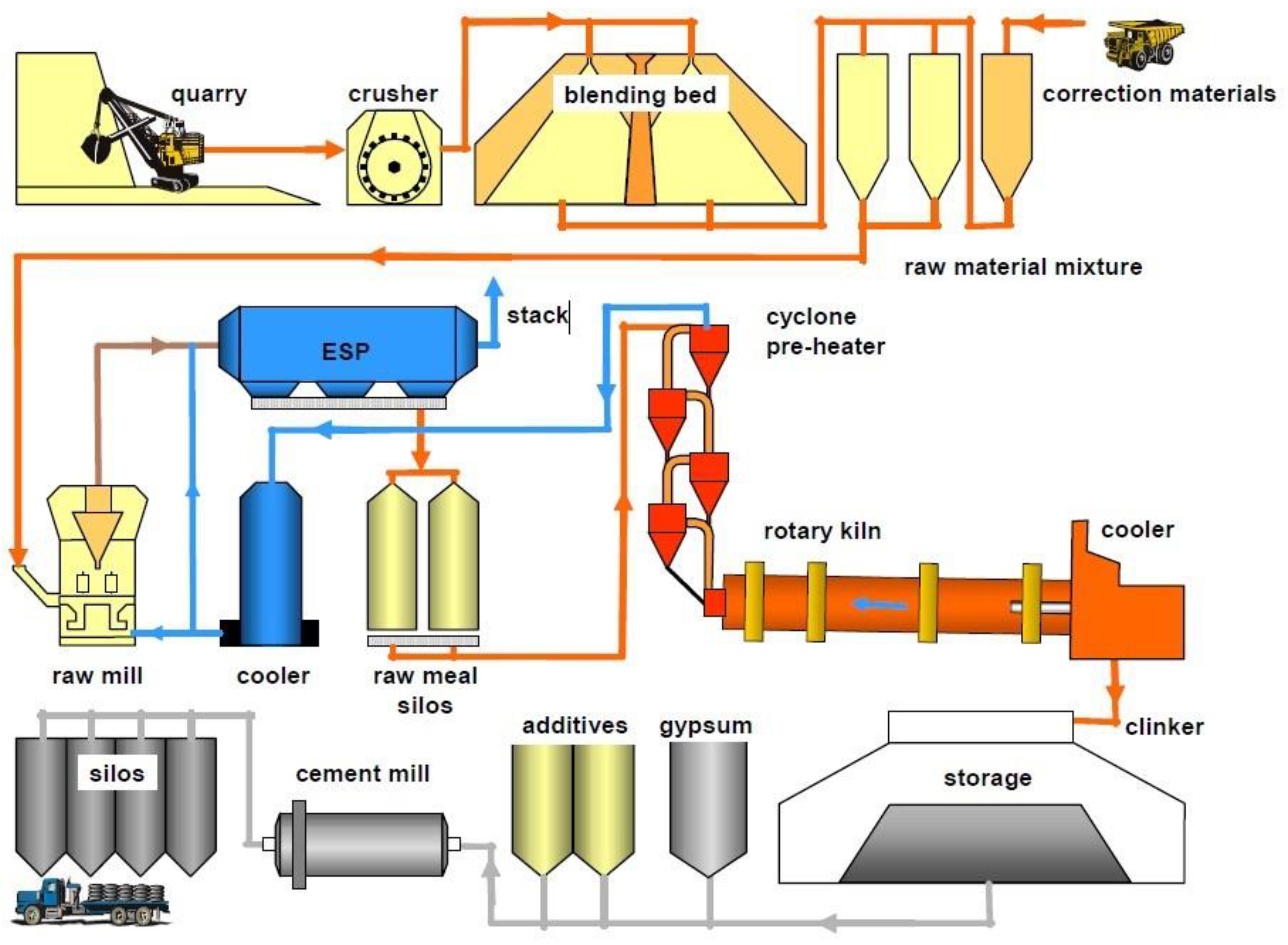
2. The Manufacture of OPC
- Evaporation of the physically sorbed water molecules from the raw mix (20–100 °C).
- Dehydration (100–430 °C) with the production of oxides, such as silica, alumina, and hematite.
- Calcination (800–1100 °C) with the development of calcium oxide, according to the carbonate decomposition reaction:CaCO3 → CaO + CO2
- Exothermic reactions (1100–1300 °C) with the formation of secondary silicate phases:2CaO + SiO2 → 2CaO · SiO2
- Sintering and reactions occurring inside the melt (1300–1450 °C) with conversion of secondary silicate phases into both ternary silicates and tetracalcium aluminoferrites:2CaO · SiO2 + CaO → 3CaO · SiO23CaO · Al2O3 + CaO + Fe2O3 → 4CaO · Al2O3 · Fe2O3
- Cooling of the system, and the crystallization of the other mineral phases.
3. Hydration Step Involving OPC
3.1. Typical Starting Composition of Cement and Mineral Phases
3.2. Chemical Reactions Occurring during the Hydration Process
3.2.1. Reaction Involving C3S
3.2.2. Reaction Involving C2S
3.2.3. Reaction Involving C3A
3.2.4. Reaction Involving C4AF
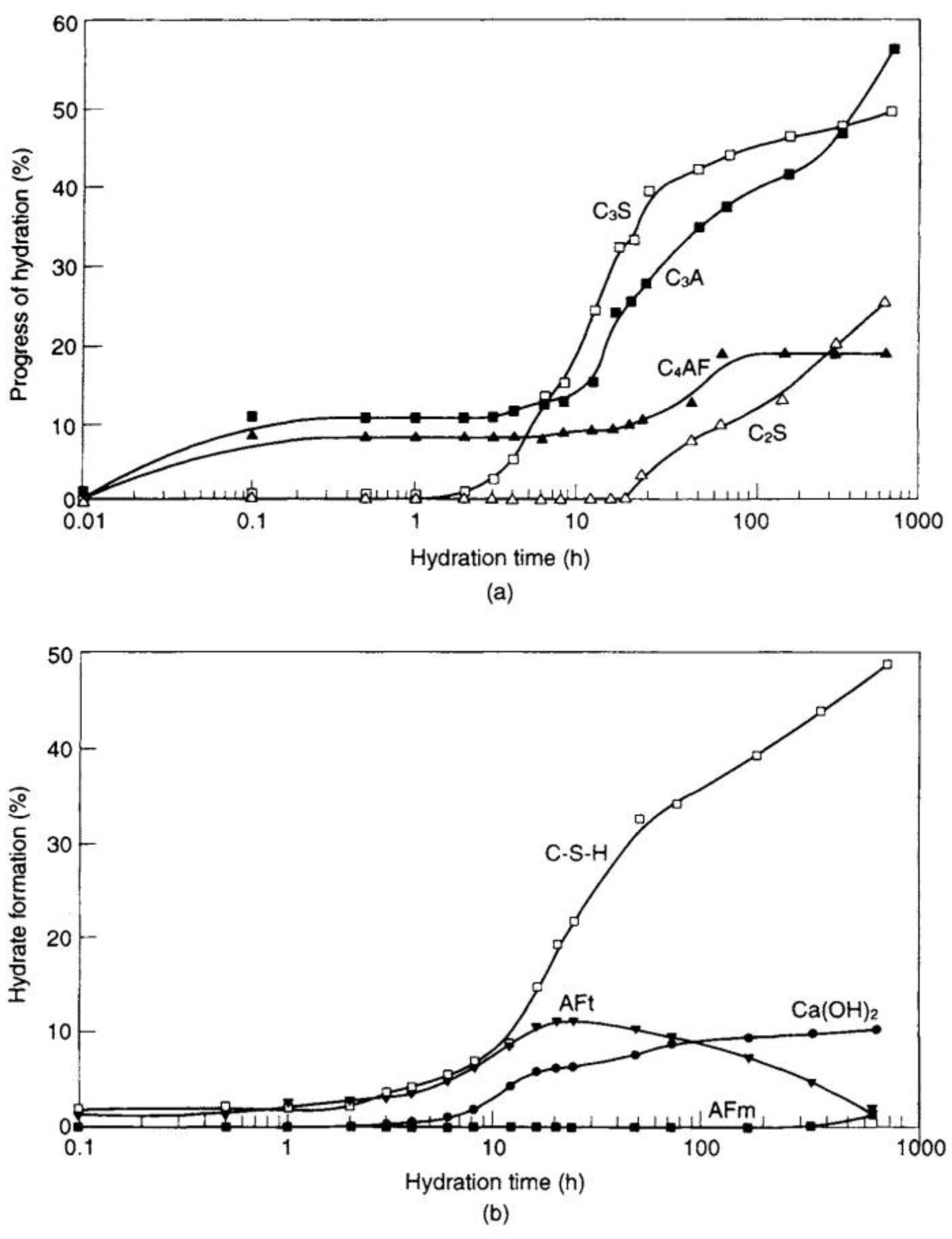
3.3. Mechanism and Heat of Hydration of OPC
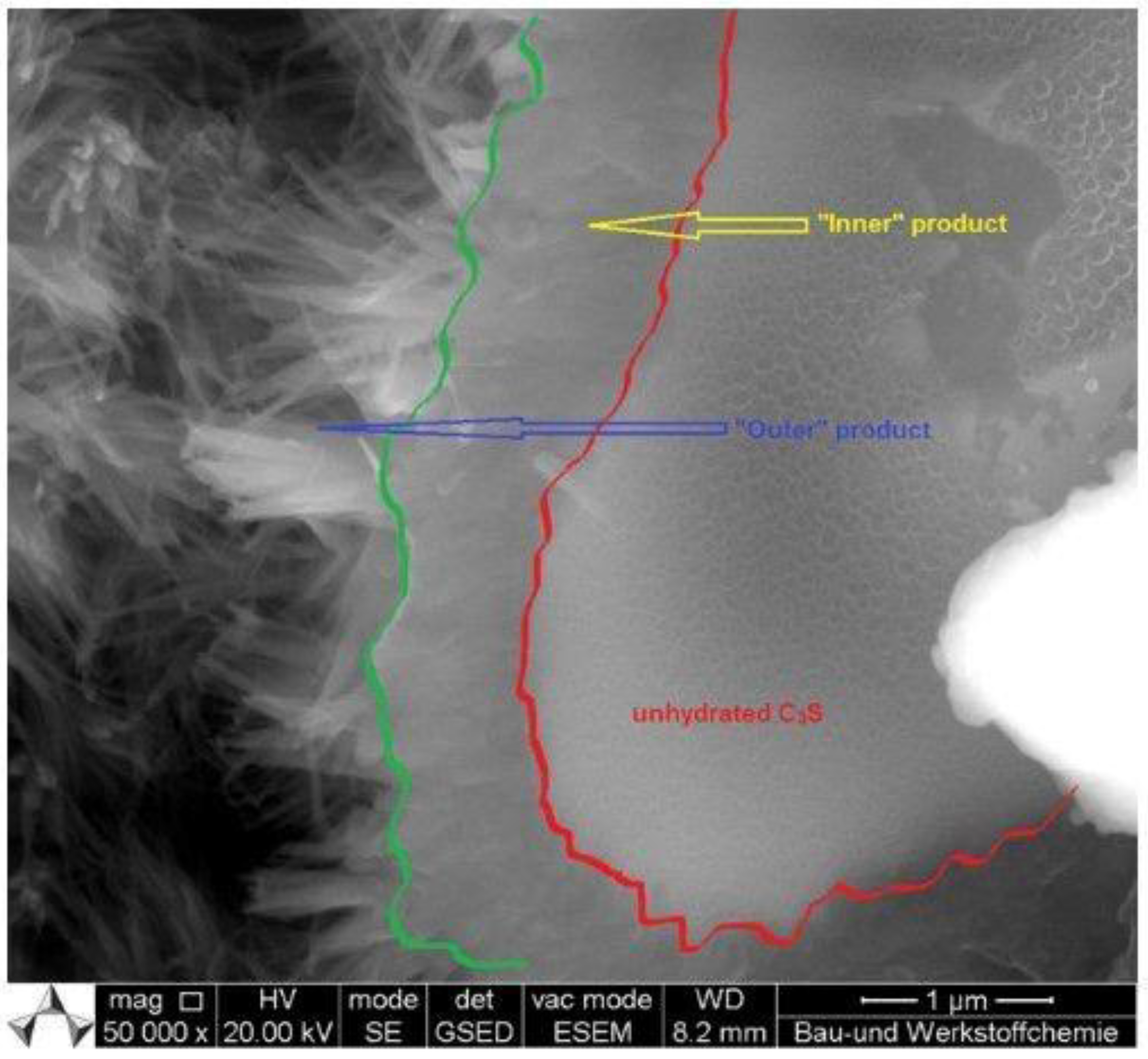
3.3.1. Pre-Induction Period
3.3.2. Induction (Dormant) Period
3.3.3. Acceleration Period
3.3.4. Post-Acceleration Period
3.3.5. Consideration over the Heat of Hydration
4. Setting of OPC
4.1. Mechanism of Setting
4.2. Flash Setting vs. False Setting
5. Pore Structure in OPC
5.1. Type of Porosities
5.2. Type of Water
5.3. Bleeding Phenomenon Involcing the Action of Water in Cementitious Paste
6. Future Perspective in the Use of Recycled (Waste) Materials in Cementitious Matrices
7. Conclusions
- (1)
- Water plays a fundamental role in the evolution of the cement matrix. Since the hydration process consists of a series of different chemical reactions involving the main components of the cement powder with water, the reaction products deriving from the hydration mechanism strongly affect the final chemical composition of the cement.
- (2)
- During the setting time, there is a conversion from a plastic paste towards a set material, whereas the mechanical properties of the formulation are defined after the hardening step.
- (3)
- It is possible to distinguish between three different types of water: (i) chemically bound water, (ii) physically bound (gel) water, and (iii) free (evaporable) water.
- (4)
- Every type of water influences the porosity of the cement, and consequently the mechanical performance.
- (5)
- It is possible to use recycled (waste) materials and reuse them in cementitious matrices to form sustainable (advanced) cementitious composites.
- (6)
- The recent interest in smart inorganic materials for advanced technological applications opens the possibility of using cement as a matrix for novel nanoscopic composites with improved properties (e.g., self-healing and health-monitoring materials).
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Odler, I. 6-Hydration, Setting and Hardening of Portland Cement. In Lea’s Chemistry of Cement and Concrete, 4th ed.; Hewlett, P.C., Ed.; Butterworth-Heinemann: Oxford, UK, 1998; pp. 241–297. ISBN 978-0-7506-6256-7. [Google Scholar]
- Cadix, A.; James, S. Chapter 5-Cementing Additives. In Fluid Chemistry, Drilling and Completion; Wang, Q., Ed.; Gulf Professional Publishing: Dhahram, Saudi Arabia, 2022; pp. 187–254. ISBN 978-0-12-822721-3. [Google Scholar]
- Ludwig, H.-M.; Zhang, W. Research Review of Cement Clinker Chemistry. Cem. Concr. Res. 2015, 78, 24–37. [Google Scholar] [CrossRef]
- Krishnya, S.; Elakneswaran, Y.; Yoda, Y. Proposing a Three-Phase Model for Predicting the Mechanical Properties of Mortar and Concrete. Mater. Today Commun. 2021, 29, 102858. [Google Scholar] [CrossRef]
- Gagg, C.R. Cement and Concrete as an Engineering Material: An Historic Appraisal and Case Study Analysis. Eng. Fail. Anal. 2014, 40, 114–140. [Google Scholar] [CrossRef]
- Liu, M.; Xia, Y.; Zhao, Y.; Cao, Z. Immobilization of Cu (Ⅱ), Ni (Ⅱ) and Zn (Ⅱ) in Silica Fume Blended Portland Cement: Role of Silica Fume. Constr. Build. Mater. 2022, 341, 127772. [Google Scholar] [CrossRef]
- Xue, C. Performance and Mechanisms of Stimulated Self-Healing in Cement-Based Composites Exposed to Saline Environments. Cem. Concr. Compos. 2022, 129, 104470. [Google Scholar] [CrossRef]
- Papaioannou, S.; Amenta, M.; Kilikoglou, V.; Gournis, D.; Karatasios, I. Synthesis and Integration of Cement-Based Capsules Modified with Sodium Silicate for Developing Self-Healing Cements. Constr. Build. Mater. 2022, 316, 125803. [Google Scholar] [CrossRef]
- Sun, M.; Liew, R.J.Y.; Zhang, M.-H.; Li, W. Development of Cement-Based Strain Sensor for Health Monitoring of Ultra High Strength Concrete. Constr. Build. Mater. 2014, 65, 630–637. [Google Scholar] [CrossRef]
- Cassese, P.; Rainieri, C.; Occhiuzzi, A. Applications of Cement-Based Smart Composites to Civil Structural Health Monitoring: A Review. Appl. Sci. 2021, 11, 8530. [Google Scholar] [CrossRef]
- Lavagna, L.; Bartoli, M.; Suarez-Riera, D.; Cagliero, D.; Musso, S.; Pavese, M. Oxidation of Carbon Nanotubes for Improving the Mechanical and Electrical Properties of Oil-Well Cement-Based Composites. ACS Appl. Nano Mater. 2022, 5, 6671–6678. [Google Scholar] [CrossRef]
- Jia, L.; Shi, C.; Pan, X.; Zhang, J.; Wu, L. Effects of Inorganic Surface Treatment on Water Permeability of Cement-Based Materials. Cem. Concr. Compos. 2016, 67, 85–92. [Google Scholar] [CrossRef]
- Lavagna, L.; Burlon, D.; Nisticò, R.; Brancato, V.; Frazzica, A.; Pavese, M.; Chiavazzo, E. Cementitious Composite Materials for Thermal Energy Storage Applications: A Preliminary Characterization and Theoretical Analysis. Sci. Rep. 2020, 10, 12833. [Google Scholar] [CrossRef] [PubMed]
- Nisticò, R.; Lavagna, L.; Boot, E.A.; Ivanchenko, P.; Lorusso, M.; Bosia, F.; Pugno, N.M.; D’Angelo, D.; Pavese, M. Improving Rubber Concrete Strength and Toughness by Plasma-induced End-of-life Tire Rubber Surface Modification. Plasma Process Polym. 2021, 18, 2100081. [Google Scholar] [CrossRef]
- Siddika, A.; Mamun, M.A.A.; Alyousef, R.; Amran, Y.H.M.; Aslani, F.; Alabduljabbar, H. Properties and Utilizations of Waste Tire Rubber in Concrete: A Review. Constr. Build. Mater. 2019, 224, 711–731. [Google Scholar] [CrossRef]
- Lavagna, L.; Nisticò, R.; Sarasso, M.; Pavese, M. An Analytical Mini-Review on the Compression Strength of Rubberized Concrete as a Function of the Amount of Recycled Tires Crumb Rubber. Materials 2020, 13, 1234. [Google Scholar] [CrossRef] [PubMed]
- Roychand, R.; Gravina, R.J.; Zhuge, Y.; Ma, X.; Youssf, O.; Mills, J.E. A Comprehensive Review on the Mechanical Properties of Waste Tire Rubber Concrete. Constr. Build. Mater. 2020, 237, 117651. [Google Scholar] [CrossRef]
- Chen, Y.K.; Sun, Y.; Wang, K.Q.; Kuang, W.Y.; Yan, S.R.; Wang, Z.H.; Lee, H.S. Utilization of Bio-Waste Eggshell Powder as a Potential Filler Material for Cement: Analyses of Zeta Potential, Hydration and Sustainability. Constr. Build. Mater. 2022, 325, 126220. [Google Scholar] [CrossRef]
- Nisticò, R.; Lavagna, L.; Versaci, D.; Ivanchenko, P.; Benzi, P. Chitosan and Its Char as Fillers in Cement-Base Composites: A Case Study. Boletín Soc. Española Cerámica Vidr. 2020, 59, 186–192. [Google Scholar] [CrossRef]
- Suarez-Riera, D.; Merlo, A.; Lavagna, L.; Nisticò, R.; Pavese, M. Mechanical Properties of Mortar Containing Recycled Acanthocardia Tuberculata Seashells as Aggregate Partial Replacement. Boletín Soc. Española Cerámica Vidr. 2021, 60, 206–210. [Google Scholar] [CrossRef]
- Cement|OEC. Available online: https://oec.world/en/profile/hs/cement (accessed on 12 December 2022).
- Xu, Z.; Guo, Z.; Zhao, Y.; Li, S.; Luo, X.; Chen, G.; Liu, C.; Gao, J. Hydration of Blended Cement with High-Volume Slag and Nano-Silica. J. Build. Eng. 2023, 64, 105657. [Google Scholar] [CrossRef]
- Balachandran, C.; Muñoz, J.F.; Peethamparan, S.; Arnold, T.S. Alkali -Silica Reaction and Its Dynamic Relationship with Cement Pore Solution in Highly Reactive Systems. Constr. Build. Mater. 2023, 362, 129702. [Google Scholar] [CrossRef]
- Xia, Y.; Liu, M.; Zhao, Y.; Chi, X.; Guo, J.; Du, D.; Du, J. Hydration Mechanism and Phase Assemblage of Blended Cement with Iron-Rich Sewage Sludge Ash. J. Build. Eng. 2023, 63, 105579. [Google Scholar] [CrossRef]
- Zheng, X.; Liu, K.; Gao, S.; Wang, F.; Wu, Z. Effect of Pozzolanic Reaction of Zeolite on Its Internal Curing Performance in Cement-Based Materials. J. Build. Eng. 2023, 63, 105503. [Google Scholar] [CrossRef]
- Deng, X.; Guo, H.; Tan, H.; Zhang, J.; Zheng, Z.; Li, M.; Chen, P.; He, X.; Yang, J.; Wang, J. Comparison on Early Hydration of Portland Cement and Sulphoaluminate Cement in the Presence of Nano Ettringite. Constr. Build. Mater. 2022, 360, 129516. [Google Scholar] [CrossRef]
- Fang, Z.; Wang, C.; Hu, H.; Zhou, S.; Luo, Y. Effect of Electrical Field on the Stability of Hydration Products of Cement Paste in Different Liquid Media. Constr. Build. Mater. 2022, 359, 129489. [Google Scholar] [CrossRef]
- Hou, J.; He, X.; Ni, X. Hydration Mechanism and Thermodynamic Simulation of Ecological Ternary Cements Containing Phosphogypsum. Mater. Today Commun. 2022, 33, 104621. [Google Scholar] [CrossRef]
- Zhang, Y.; Yang, W.; Wang, Y.; Gong, Z.; Zhang, K. Molecular Dynamics Simulation of C-S-H Corrosion in Chloride Environment. Mater. Today Commun. 2022, 33, 104568. [Google Scholar] [CrossRef]
- Aretxabaleta, X.M.; López-Zorrilla, J.; Labbez, C.; Etxebarria, I.; Manzano, H. A Potential C-S-H Nucleation Mechanism: Atomistic Simulations of the Portlandite to C-S-H Transformation. Cem. Concr. Res. 2022, 162, 106965. [Google Scholar] [CrossRef]
- Yi, Y.; Ma, W.; Sidike, A.; Ma, Z.; Fang, M.; Lin, Y.; Bai, S.; Chen, Y. Synergistic Effect of Hydration and Carbonation of Ladle Furnace Aslag on Cementitious Substances. Sci. Rep. 2022, 12, 14526. [Google Scholar] [CrossRef]
- Zunino, F.; Dhandapani, Y.; Ben Haha, M.; Skibsted, J.; Joseph, S.; Krishnan, S.; Parashar, A.; Juenger, M.C.G.; Hanein, T.; Bernal, S.A.; et al. Hydration and Mixture Design of Calcined Clay Blended Cements: Review by the RILEM TC 282-CCL. Mater. Struct. 2022, 55, 234. [Google Scholar] [CrossRef]
- Maruyama, I.; Sugimoto, H.; Umeki, S.; Kurihara, R. Effect of Fineness of Cement on Drying Shrinkage. Cem. Concr. Res. 2022, 161, 106961. [Google Scholar] [CrossRef]
- Li, H.-W.; Wang, R.; Wei, M.-W.; Lei, N.-Z.; Sun, H.-X.; Fan, J.-J. Mechanical Properties and Hydration Mechanism of High-Volume Ultra-Fine Iron Ore Tailings Cementitious Materials. Constr. Build. Mater. 2022, 353, 129100. [Google Scholar] [CrossRef]
- Ma, Y.; Li, W.; Jin, M.; Liu, J.; Zhang, J.; Huang, J.; Lu, C.; Zeng, H.; Wang, J.; Zhao, H.; et al. Influences of Leaching on the Composition, Structure and Morphology of Calcium Silicate Hydrate (C–S–H) with Different Ca/Si Ratios. J. Build. Eng. 2022, 58, 105017. [Google Scholar] [CrossRef]
- Liu, R.; Fang, B.; Zhang, G.; Guo, J.; Yang, Y. Investigation of Sodium Alginate as a Candidate Retarder of Magnesium Phosphate Cement: Hydration Properties and Its Retarding Mechanism. Ceram. Int. 2022, 48, 30846–30852. [Google Scholar] [CrossRef]
- Wang, X.; Zhao, Q.; Zhang, S.; Lu, N. Pre-Curing Time Effect on Reactive Powder Concrete Impact Resistance. AIP Adv. 2022, 12, 105025. [Google Scholar] [CrossRef]
- Li, B.; Tang, Z.; Huo, B.; Liu, Z.; Cheng, Y.; Ding, B.; Zhang, P. The Early Age Hydration Products and Mechanical Properties of Cement Paste Containing GBFS under Steam Curing Condition. Buildings 2022, 12, 1746. [Google Scholar] [CrossRef]
- Yan, Z.; Zhang, H.; Zhu, Y. Hydration Kinetics of Sulfoaluminate Cement with Different Water/Cement Ratios as Grouting Material Used for Coal Mines. Mag. Concr. Res. 2022, 74, 1056–1064. [Google Scholar] [CrossRef]
- Dong, P.; Allahverdi, A.; Andrei, C.M.; Bassim, N.D. The Effects of Nano-Silica on Early-Age Hydration Reactions of Nano Portland Cement. Cem. Concr. Compos. 2022, 133, 104698. [Google Scholar] [CrossRef]
- Ma, J.; Liu, X.; Yu, Z.; Shi, H.; Wu, Q.; Shen, X. Effects of Limestone Powder on the Early Hydration of Tricalcium Aluminate. J. Therm. Anal. Calorim. 2022, 147, 10351–10362. [Google Scholar] [CrossRef]
- Achternbosch, M.; Bräutigam, K.R.; Hartlieb, N.; Kupsch, C.; Richers, U.; Stemmermann, P.; Gleis, M. Heavy Metals in Cement and Concrete Resulting from the Co-Incineration of Wastes in Cement Kilns with Regard to the Legitimacy of Waste Utilisation; Forschungszentrum Karlsruhe GmbH: Karlsruhe, Germany, 2003. [Google Scholar]
- de Queiroz Lamas, W.; Palau, J.C.F.; de Camargo, J.R. Waste Materials Co-Processing in Cement Industry: Ecological Efficiency of Waste Reuse. Renew. Sustain. Energy Rev. 2013, 19, 200–207. [Google Scholar] [CrossRef]
- Hwidi, R.S.; Tengku Izhar, T.N.; Mohd Saad, F.N. Characterization of Limestone as Raw Material to Hydrated Lime. E3S Web Conf. 2018, 34, 02042. [Google Scholar] [CrossRef]
- Schumacher, G.; Juniper, L. 15-Coal Utilisation in the Cement and Concrete Industries. In The Coal Handbook: Towards Cleaner Production; Osborne, D., Ed.; Woodhead Publishing: Sawston, UK, 2013; Volume 2, pp. 387–426. ISBN 978-1-78242-116-0. [Google Scholar]
- Lea, F.M.; Mason, T.O. Cement-Extraction and Processing; Encyclopædia Britannica, Inc.: Chicago, IL, USA, 2019. [Google Scholar]
- Whittaker, M.; Dubina, E.; Plank, J.; Black, L. The Effects of Prehydration on Cement Performance. In Proceedings of the 30th Cement and Concrete Science Conference, Birmingham, UK, 13–15 September 2010. [Google Scholar]
- Nisticò, R. A Comprehensive Study on the Applications of Clays into Advanced Technologies, with a Particular Attention on Biomedicine and Environmental Remediation. Inorganics 2022, 10, 40. [Google Scholar] [CrossRef]
- Nisticò, R. The Importance of Surfaces and Interfaces in Clays for Water Remediation Processes. Surf. Topogr. Metrol. Prop. 2018, 6, 043001. [Google Scholar] [CrossRef]
- Powers, T.C.; Brownyard, T.L. Studies of the Physical Properties of Hardened Portland Cement Paste. ACI J. Proc. 1946, 43, 249–336. [Google Scholar] [CrossRef]
- Alesiani, M.; Pirazzoli, I.; Maraviglia, B. Factors Affecting Early-Age Hydration of Ordinary Portland Cement Studied by NMR: Fineness, Water-to-Cement Ratio and Curing Temperature. Appl. Magn. Reson. 2007, 32, 385–394. [Google Scholar] [CrossRef]
- Soler, J.M. Thermodynamic Description of the Solubility of C-S-H Gels in Hydrated Portland Cement Literature Review; Posiva Oy: Helsinki, Finland, 2007; Available online: http://www.posiva.fi/publications/WR2007-88web.pdf (accessed on 12 December 2022).
- Wang, L.; Yang, H.Q.; Zhou, S.H.; Chen, E.; Tang, S.W. Hydration, Mechanical Property and C-S-H Structure of Early-Strength Low-Heat Cement-Based Materials. Mater. Lett. 2018, 217, 151–154. [Google Scholar] [CrossRef]
- de Jong, J.G.M.; Stein, H.N.; Stevels, J.M. Hydration of Tricalcium Silicate. J. Appl. Chem. 2007, 17, 246–250. [Google Scholar] [CrossRef]
- Cuesta, A.; Zea-Garcia, J.D.; Londono-Zuluaga, D.; De La Torre, A.G.; Santacruz, I.; Vallcorba, O.; Dapiaggi, M.; Sanfélix, S.G.; Aranda, M.A.G. Multiscale Understanding of Tricalcium Silicate Hydration Reactions. Sci. Rep. 2018, 8, 8544. [Google Scholar] [CrossRef]
- Thomas, J.J.; Ghazizadeh, S.; Masoero, E. Kinetic Mechanisms and Activation Energies for Hydration of Standard and Highly Reactive Forms of β-Dicalcium Silicate (C2S). Cem. Concr. Res. 2017, 100, 322–328. [Google Scholar] [CrossRef]
- Park, S.M.; Jang, J.G.; Son, H.M.; Lee, H.K. Stable Conversion of Metastable Hydrates in Calcium Aluminate Cement by Early Carbonation Curing. J. CO2 Util. 2017, 21, 224–226. [Google Scholar] [CrossRef]
- Lea, F.M.; Hewlett, P.C.; Liška, M. (Eds.) Lea’s Chemistry of Cement and Concrete, 5th ed.; Butterworth-Heinemann: Oxford, UK, 2019; ISBN 978-0-08-100773-0. [Google Scholar]
- Taylor, H.F.W.; Famy, C.; Scrivener, K.L. Delayed Ettringite Formation. Cem. Concr. Res. 2001, 31, 683–693. [Google Scholar] [CrossRef]
- Marchon, D.; Flatt, R.J. 8-Mechanisms of Cement Hydration. In Science and Technology of Concrete Admixtures; Aïtcin, P.-C., Flatt, R.J., Eds.; Woodhead Publishing: Sawston, UK, 2016; pp. 129–145. ISBN 978-0-08-100693-1. [Google Scholar]
- Meller, N.; Hall, C.; Crawshaw, J. ESEM Evidence for Through-Solution Transport during Brownmillerite Hydration. J. Mater. Sci. 2004, 39, 6611–6614. [Google Scholar] [CrossRef]
- Rose, J.; Bénard, A.; El Mrabet, S.; Masion, A.; Moulin, I.; Briois, V.; Olivi, L.; Bottero, J.Y. Evolution of Iron Speciation during Hydration of C4AF. Waste Manag. 2006, 26, 720–724. [Google Scholar] [CrossRef]
- Flick, E.W. Handbook of Adhesive Raw Materials; William Andrew: Norwich, NY, USA, 1989; ISBN 0-8155-1185-X. [Google Scholar]
- Nicoleau, L.; Schreiner, E.; Nonat, A. Ion-Specific Effects Influencing the Dissolution of Tricalcium Silicate. Cem. Concr. Res. 2014, 59, 118–138. [Google Scholar] [CrossRef]
- Scherer, G.W.; Zhang, J.; Thomas, J.J. Nucleation and Growth Models for Hydration of Cement. Cem. Concr. Res. 2012, 42, 982–993. [Google Scholar] [CrossRef]
- Scrivener, K.L.; Juilland, P.; Monteiro, P.J.M. Advances in Understanding Hydration of Portland Cement. Cem. Concr. Res. 2015, 78, 38–56. [Google Scholar] [CrossRef]
- Double, D.D.; Hellawell, A.; Perry, S.J.; Hirsch, P.B. The Hydration of Portland Cement. Proc. R. Soc. Lond. A Math. Phys. Sci. 1978, 359, 435–451. [Google Scholar] [CrossRef]
- Meredith, P.; Donald, A.M.; Luke, K. Pre-Induction and Induction Hydration of Tricalcium Silicate: An Environmental Scanning Electron Microscopy Study. J. Mater. Sci. 1995, 30, 1921–1930. [Google Scholar] [CrossRef]
- Thomas, N.L.; Double, D.D. Calcium and Silicon Concentrations in Solution during the Early Hydration of Portland Cement and Tricalcium Silicate. Cem. Concr. Res. 1981, 11, 675–687. [Google Scholar] [CrossRef]
- Jiahui, P.; Jianxin, Z.; Jindong, Q. The Mechanism of the Formation and Transformation of Ettringite. J. Wuhan Univ. Technol. -Mater. Sci. Ed. 2006, 21, 158–161. [Google Scholar] [CrossRef]
- El-Didamony, H.; Sharara, A.M.; Helmy, I.M.; Abd El-Aleem, S. Hydration Characteristics of β-C2S in the Presence of Some Accelerators. Cem. Concr. Res. 1996, 26, 1179–1187. [Google Scholar] [CrossRef]
- Jennings, H.M.; Pratt, P.L. An Experimental Argument for the Existence of a Protective Membrane Surrounding Portland Cement during the Induction Period. Cem. Concr. Res. 1979, 9, 501–506. [Google Scholar] [CrossRef]
- Jia, F.; Yao, Y.; Wang, J. Influence and Mechanism Research of Hydration Heat Inhibitor on Low-Heat Portland Cement. Front. Mater. 2021, 8, 697380. [Google Scholar] [CrossRef]
- Ouzia, A.R.C.W.C. Modeling the Kinetics of the Main Peak and Later Age of Alite Hydration; EPFL: Lausanne, Switzerland, 2019; p. 288. [Google Scholar] [CrossRef]
- Juilland, P.; Nicoleau, L.; Arvidson, R.S.; Gallucci, E. Advances in Dissolution Understanding and Their Implications for Cement Hydration. RILEM Tech. Lett. 2017, 2, 90–98. [Google Scholar] [CrossRef]
- Tennis, P.D.; Jennings, H.M. Model for Two Types of Calcium Silicate Hydrate in the Microstructure of Portland Cement Pastes. Cem. Concr. Res. 2000, 30, 855–863. [Google Scholar] [CrossRef]
- Sakalli, Y.; Trettin, R. Investigation of C3S Hydration by Environmental Scanning Electron Microscope. J. Microsc. 2015, 259, 53–58. [Google Scholar] [CrossRef]
- Zhutovsky, S.; Kovler, K. Chemical Shrinkage of High-Strength/High-Performance Cementitious Materials. Int. Rev. Chem. Eng. 2018, 10, 110–118. [Google Scholar] [CrossRef]
- Bazzoni, A.; Ma, S.; Wang, Q.; Shen, X.; Cantoni, M.; Scrivener, K.L. The Effect of Magnesium and Zinc Ions on the Hydration Kinetics of C3S. J. Am. Ceram. Soc. 2014, 97, 3684–3693. [Google Scholar] [CrossRef]
- Barnes, P.; Bensted, J. Structure and Performance of Cements; CRC Press: Boca Raton, FL, USA, 2002; ISBN 0-429-17842-5. [Google Scholar]
- Ben Haha, M.; Winnefeld, F.; Pisch, A. Advances in Understanding Ye’elimite-Rich Cements. Cem. Concr. Res. 2019, 123, 105778. [Google Scholar] [CrossRef]
- C01 Committee. Test Methods for Time of Setting of Hydraulic Cement by Vicat Needle; ASTM International: West Conshohocken, PA, USA, 2006. [Google Scholar]
- Yuan, Q.; Liu, Z.; Zheng, K.; Ma, C. Chapter 2-Inorganic Cementing Materials. In Civil Engineering Materials; Yuan, Q., Liu, Z., Zheng, K., Ma, C., Eds.; Elsevier: Amsterdam, The Netherlands, 2021; pp. 17–57. ISBN 978-0-12-822865-4. [Google Scholar]
- Taylor, H.F.W. 4 Properties of Portland Clinker and Cement. In Cement Chemistry; Thomas Telford Publishing: London, UK, 1997; pp. 89–112. [Google Scholar]
- Wang, L.; Jin, M.; Wu, Y.; Zhou, Y.; Tang, S. Hydration, Shrinkage, Pore Structure and Fractal Dimension of Silica Fume Modified Low Heat Portland Cement-Based Materials. Constr. Build. Mater. 2021, 272, 121952. [Google Scholar] [CrossRef]
- Aïtcin, P.-C.; Lessard, J.-M. 8-The Composition and Design of High-Strength Concrete and Ultrahigh-Strength Concrete. In Developments in the Formulation and Reinforcement of Concrete, 2nd ed.; Mindess, S., Ed.; Woodhead Publishing: Sawston, UK, 2019; pp. 171–192. ISBN 978-0-08-102616-8. [Google Scholar]
- Ley-Hernandez, A.M.; Lapeyre, J.; Cook, R.; Kumar, A.; Feys, D. Elucidating the Effect of Water-To-Cement Ratio on the Hydration Mechanisms of Cement. ACS Omega 2018, 3, 5092–5105. [Google Scholar] [CrossRef]
- Mesboua, N.; Benyounes, K.; Benmounah, A. Study of the Impact of Bentonite on the Physico-Mechanical and Flow Properties of Cement Grout. Cogent Eng. 2018, 5, 1446252. [Google Scholar] [CrossRef]
- Nguyen, D.D.; Devlin, L.P.; Koshy, P.; Sorrell, C.C. Impact of Water-Soluble Cellulose Ethers on Polymer-Modified Mortars. J. Mater. Sci. 2014, 49, 923–951. [Google Scholar] [CrossRef]
- Xie, N.; Dang, Y.; Shi, X. New Insights into How MgCl2 Deteriorates Portland Cement Concrete. Cem. Concr. Res. 2019, 120, 244–255. [Google Scholar] [CrossRef]
- Villoria Sáez, P.; Osmani, M. A Diagnosis of Construction and Demolition Waste Generation and Recovery Practice in the European Union. J. Clean. Prod. 2019, 241, 118400. [Google Scholar] [CrossRef]
- Liikanen, M.; Grönman, K.; Deviatkin, I.; Havukainen, J.; Hyvärinen, M.; Kärki, T.; Varis, J.; Soukka, R.; Horttanainen, M. Construction and Demolition Waste as a Raw Material for Wood Polymer Composites—Assessment of Environmental Impacts. J. Clean. Prod. 2019, 225, 716–727. [Google Scholar] [CrossRef]
- Coelho, A.; de Brito, J. Economic Analysis of Conventional versus Selective Demolition—A Case Study. Resour. Conserv. Recycl. 2011, 55, 382–392. [Google Scholar] [CrossRef]
- Borghi, G.; Pantini, S.; Rigamonti, L. Life Cycle Assessment of Non-Hazardous Construction and Demolition Waste (CDW) Management in Lombardy Region (Italy). J. Clean. Prod. 2018, 184, 815–825. [Google Scholar] [CrossRef]
- Contreras, M.; Teixeira, S.R.; Lucas, M.C.; Lima, L.C.N.; Cardoso, D.S.L.; da Silva, G.A.C.; Gregório, G.C.; de Souza, A.E.; dos Santos, A. Recycling of Construction and Demolition Waste for Producing New Construction Material (Brazil Case-Study). Constr. Build. Mater. 2016, 123, 594–600. [Google Scholar] [CrossRef]
- Ossa, A.; García, J.L.; Botero, E. Use of Recycled Construction and Demolition Waste (CDW) Aggregates: A Sustainable Alternative for the Pavement Construction Industry. J. Clean. Prod. 2016, 135, 379–386. [Google Scholar] [CrossRef]
- Coelho, A.; De Brito, J. 9-Preparation of Concrete Aggregates from Construction and Demolition Waste (CDW). In Handbook of Recycled Concrete and Demolition Waste; Pacheco-Torgal, F., Tam, V.W.Y., Labrincha, J.A., Ding, Y., de Brito, J., Eds.; Woodhead Publishing: Sawston, UK, 2013; pp. 210–245. ISBN 978-0-85709-682-1. [Google Scholar]
- Marzouk, M.; Azab, S. Environmental and Economic Impact Assessment of Construction and Demolition Waste Disposal Using System Dynamics. Resour. Conserv. Recycl. 2014, 82, 41–49. [Google Scholar] [CrossRef]
- Gálvez-Martos, J.-L.; Styles, D.; Schoenberger, H.; Zeschmar-Lahl, B. Construction and Demolition Waste Best Management Practice in Europe. Resour. Conserv. Recycl. 2018, 136, 166–178. [Google Scholar] [CrossRef]
- Jesus, S.; Maia, C.; Brazão Farinha, C.; de Brito, J.; Veiga, R. Rendering Mortars with Incorporation of Very Fine Aggregates from Construction and Demolition Waste. Constr. Build. Mater. 2019, 229, 116844. [Google Scholar] [CrossRef]
- Ma, Z.; Shen, J.; Wang, C.; Wu, H. Characterization of Sustainable Mortar Containing High-Quality Recycled Manufactured Sand Crushed from Recycled Coarse Aggregate. Cem. Concr. Compos. 2022, 132, 104629. [Google Scholar] [CrossRef]
- Saikia, N.; de Brito, J. Use of Plastic Waste as Aggregate in Cement Mortar and Concrete Preparation: A Review. Constr. Build. Mater. 2012, 34, 385–401. [Google Scholar] [CrossRef]
- El Bitouri, Y.; Perrin, D. Compressive and Flexural Strengths of Mortars Containing ABS and WEEE Based Plastic Aggregates. Polymers 2022, 14, 3914. [Google Scholar] [CrossRef]
- Albano, C.; Camacho, N.; Hernández, M.; Matheus, A.; Gutiérrez, A. Influence of Content and Particle Size of Waste Pet Bottles on Concrete Behavior at Different w/c Ratios. Waste Manag. 2009, 29, 2707–2716. [Google Scholar] [CrossRef]
- Choi, Y.W.; Moon, D.J.; Kim, Y.J.; Lachemi, M. Characteristics of Mortar and Concrete Containing Fine Aggregate Manufactured from Recycled Waste Polyethylene Terephthalate Bottles. Constr. Build. Mater. 2009, 23, 2829–2835. [Google Scholar] [CrossRef]
- Choi, Y.-W.; Moon, D.-J.; Chung, J.-S.; Cho, S.-K. Effects of Waste PET Bottles Aggregate on the Properties of Concrete. Cem. Concr. Res. 2005, 35, 776–781. [Google Scholar] [CrossRef]
- Kim, S.B.; Yi, N.H.; Kim, H.Y.; Kim, J.-H.J.; Song, Y.-C. Material and Structural Performance Evaluation of Recycled PET Fiber Reinforced Concrete. Cem. Concr. Compos. 2010, 32, 232–240. [Google Scholar] [CrossRef]
- Marzouk, O.Y.; Dheilly, R.M.; Queneudec, M. Valorization of Post-Consumer Waste Plastic in Cementitious Concrete Composites. Waste Manag. 2007, 27, 310–318. [Google Scholar] [CrossRef]
- Silva, D.A.; Betioli, A.M.; Gleize, P.J.P.; Roman, H.R.; Gómez, L.A.; Ribeiro, J.L.D. Degradation of Recycled PET Fibers in Portland Cement-Based Materials. Cem. Concr. Res. 2005, 35, 1741–1746. [Google Scholar] [CrossRef]
- Yesilata, B.; Isıker, Y.; Turgut, P. Thermal Insulation Enhancement in Concretes by Adding Waste PET and Rubber Pieces. Constr. Build. Mater. 2009, 23, 1878–1882. [Google Scholar] [CrossRef]
- Akçaözoğlu, S.; Atiş, C.D.; Akçaözoğlu, K. An Investigation on the Use of Shredded Waste PET Bottles as Aggregate in Lightweight Concrete. Waste Manag. 2010, 30, 285–290. [Google Scholar] [CrossRef] [PubMed]
- Nisticò, R. Polyethylene Terephthalate (PET) in the Packaging Industry. Polym. Test. 2020, 90, 106707. [Google Scholar] [CrossRef]
- Kou, S.C.; Lee, G.; Poon, C.S.; Lai, W.L. Properties of Lightweight Aggregate Concrete Prepared with PVC Granules Derived from Scraped PVC Pipes. Waste Manag. 2009, 29, 621–628. [Google Scholar] [CrossRef]
- Merlo, A.; Lavagna, L.; Suarez-Riera, D.; Pavese, M. Mechanical Properties of Mortar Containing Waste Plastic (PVC) as Aggregate Partial Replacement. Case Stud. Constr. Mater. 2020, 13, e00467. [Google Scholar] [CrossRef]
- Naik, T.R.; Singh, S.S.; Huber, C.O.; Brodersen, B.S. Use of Post-Consumer Waste Plastics in Cement-Based Composites. Cem. Concr. Res. 1996, 26, 1489–1492. [Google Scholar] [CrossRef]
- Ismail, Z.Z.; AL-Hashmi, E.A. Use of Waste Plastic in Concrete Mixture as Aggregate Replacement. Waste Manag. 2008, 28, 2041–2047. [Google Scholar] [CrossRef]
- Merlo, A.; Lavagna, L.; Suarez-Riera, D.; Pavese, M. Recycling of WEEE Plastics Waste in Mortar: The Effects on Mechanical Properties. Recycling 2021, 6, 70. [Google Scholar] [CrossRef]
- Kan, A.; Demirboğa, R. A Novel Material for Lightweight Concrete Production. Cem. Concr. Compos. 2009, 31, 489–495. [Google Scholar] [CrossRef]
- Kan, A.; Demirboğa, R. A New Technique of Processing for Waste-Expanded Polystyrene Foams as Aggregates. J. Mater. Process. Technol. 2009, 209, 2994–3000. [Google Scholar] [CrossRef]
- Hannawi, K.; Kamali-Bernard, S.; Prince, W. Physical and Mechanical Properties of Mortars Containing PET and PC Waste Aggregates. Waste Manag. 2010, 30, 2312–2320. [Google Scholar] [CrossRef] [PubMed]
- Mounanga, P.; Gbongbon, W.; Poullain, P.; Turcry, P. Proportioning and Characterization of Lightweight Concrete Mixtures Made with Rigid Polyurethane Foam Wastes. Cem. Concr. Compos. 2008, 30, 806–814. [Google Scholar] [CrossRef]
- Ben Fraj, A.; Kismi, M.; Mounanga, P. Valorization of Coarse Rigid Polyurethane Foam Waste in Lightweight Aggregate Concrete. Constr. Build. Mater. 2010, 24, 1069–1077. [Google Scholar] [CrossRef]
- Sgobba, S.; Borsa, M.; Molfetta, M.; Marano, G.C. Mechanical Performance and Medium-Term Degradation of Rubberised Concrete. Constr. Build. Mater. 2015, 98, 820–831. [Google Scholar] [CrossRef]
- Nacif, G.L.; Panzera, T.H.; Strecker, K.; Christoforo, A.L.; Paine, K. Investigations on Cementitious Composites Based on Rubber Particle Waste Additions. Mater. Res. 2012, 16, 259–268. [Google Scholar] [CrossRef][Green Version]
- Asdrubali, F.; D’Alessandro, F.; Schiavoni, S.; Baldinelli, G. Lightweight Screeds Made of Concrete and Recycled Polymers: Acoustic, Thermal, Mechanical and Chemical Characterization; Forum Acusticum: Aalborg, Denmark, 2011; pp. 821–826. [Google Scholar]
- Abaza, O.A.; Shtayeh, S.M. Crumbed Rubber for Non-Structural Portland Cement Concrete Applications. Eng. Sci. 2010, 37, 214–225. [Google Scholar]



| Compounds | Chemical Formula | Bogue Formula | Amount (wt.%) |
|---|---|---|---|
| Alite, or Tricalcium silicate | Ca3SiO5 [3CaO · SiO2] | C3S | 30–50 |
| Belite, or Dicalcium silicate | Ca2SiO4 [2CaO · SiO2] | C2S | 20–45 |
| Celite, or Tricalcium aluminate | Ca3Al2O6 [3CaO · Al2O3] | C3A | 8–12 |
| Brownmillerite, or Tetracalcium aluminoferrite | Ca4Al2Fe2O10 [4CaO · Al2O3 · Fe2O3] | C4AF | 6–10 |
| Gypsum, or Calcium sulphate dihydrated | CaSO4 · 2H2O | - | 4–8 |
| Potassium oxide | K2O | K | <2 |
| Sodium oxide | Na2O | N | <2 |
| Component | Amount (wt.%) |
|---|---|
| CaO | 58.0–68.0 |
| SiO2 | 16.0–26.0 |
| Al2O3 | 4.0–8.0 |
| Fe2O3 | 2.0–5.0 |
| MgO | 1.0–4.0 |
| SO3 | 0.1–2.5 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lavagna, L.; Nisticò, R. An Insight into the Chemistry of Cement—A Review. Appl. Sci. 2023, 13, 203. https://doi.org/10.3390/app13010203
Lavagna L, Nisticò R. An Insight into the Chemistry of Cement—A Review. Applied Sciences. 2023; 13(1):203. https://doi.org/10.3390/app13010203
Chicago/Turabian StyleLavagna, Luca, and Roberto Nisticò. 2023. "An Insight into the Chemistry of Cement—A Review" Applied Sciences 13, no. 1: 203. https://doi.org/10.3390/app13010203
APA StyleLavagna, L., & Nisticò, R. (2023). An Insight into the Chemistry of Cement—A Review. Applied Sciences, 13(1), 203. https://doi.org/10.3390/app13010203







