Silver Thread-Based Microfluidic Platform for Detection of Essential Oils Using Impedance Spectroscopy
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials and Instruments
2.2. Design and Fabrication of Textile-Based Microfluidic Sensing Platform
2.3. Preparation of Essential Oil Solutions
2.4. Experimental Setup
3. Results and Discussion
3.1. GCMS Measurements
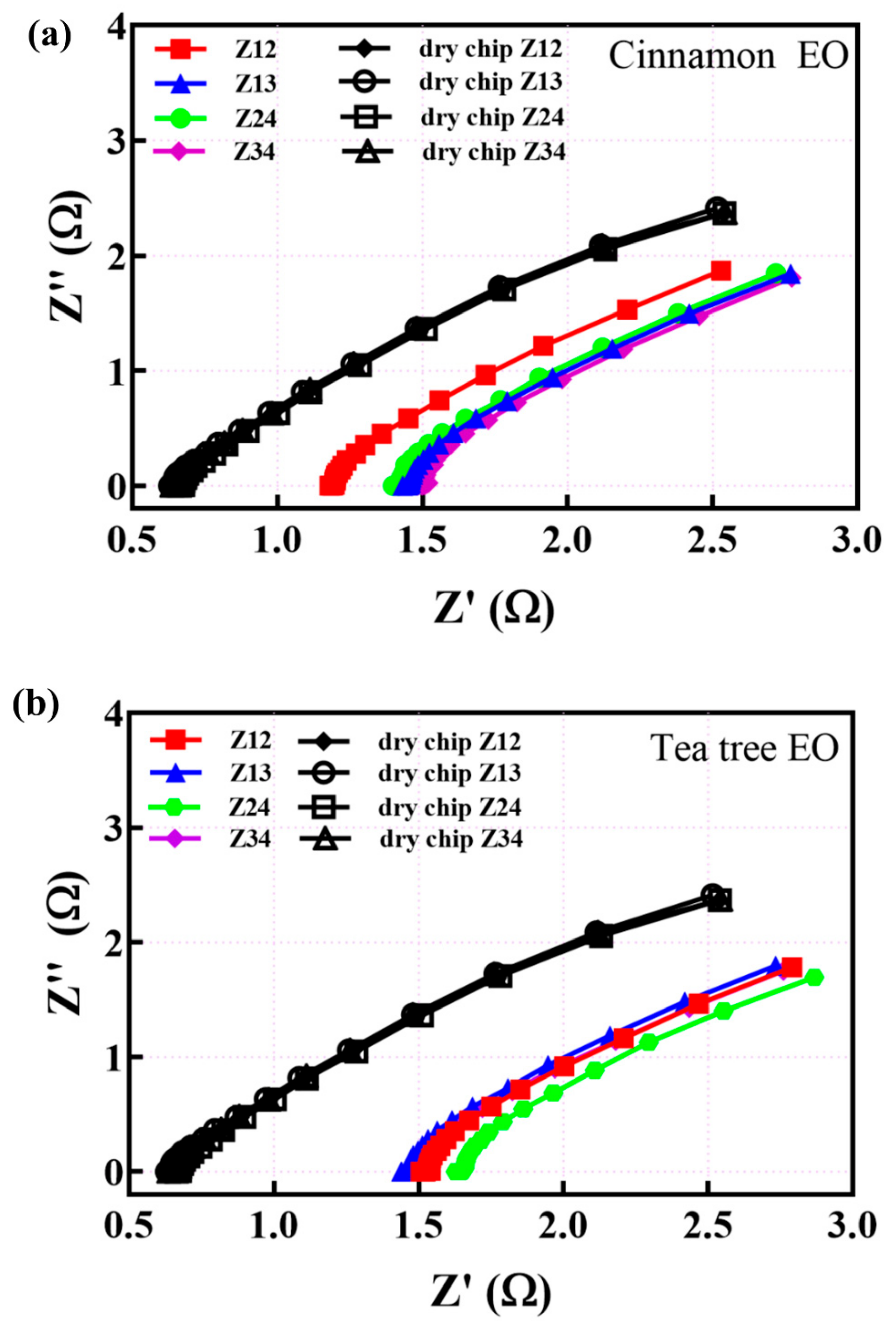
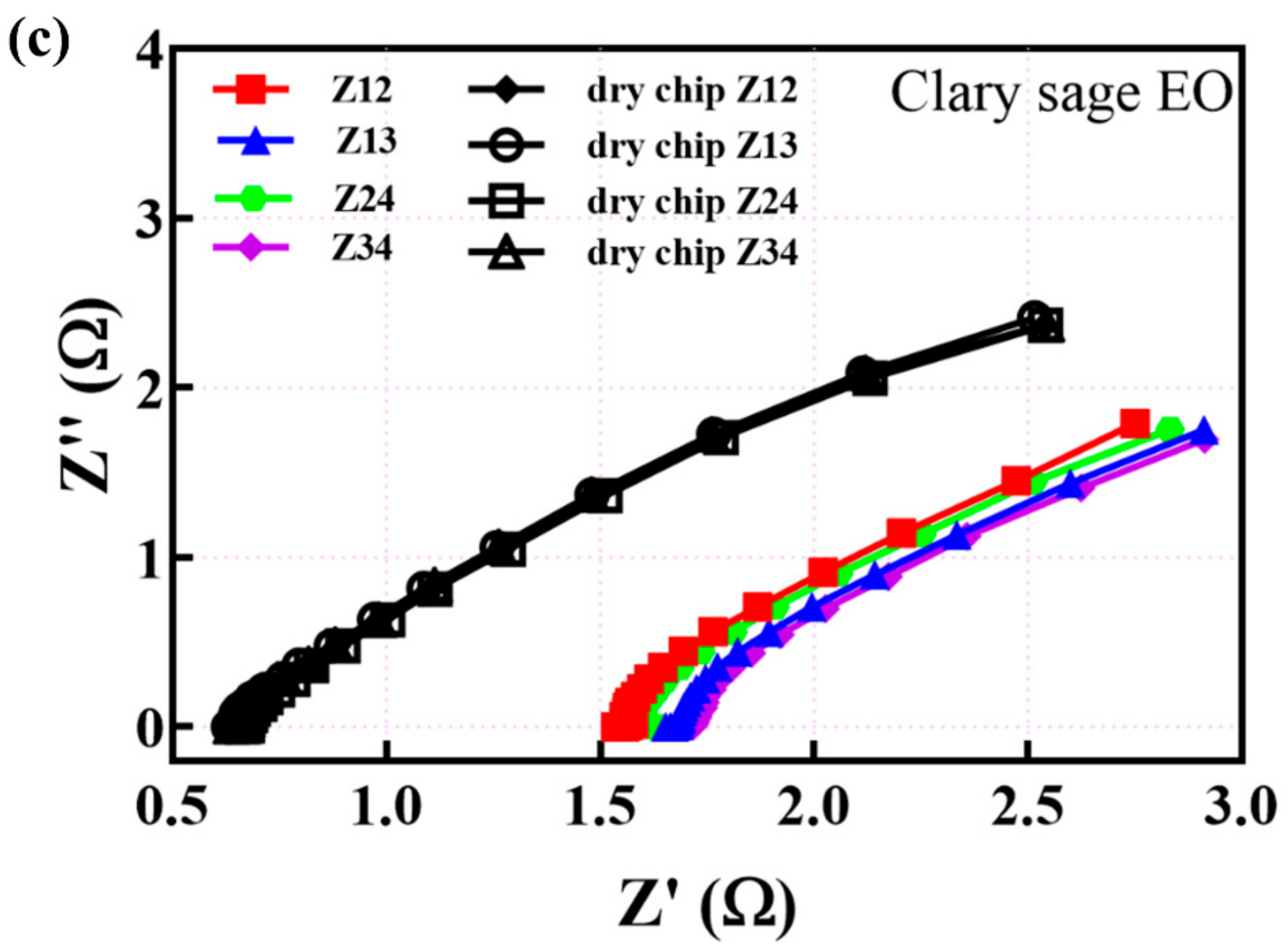
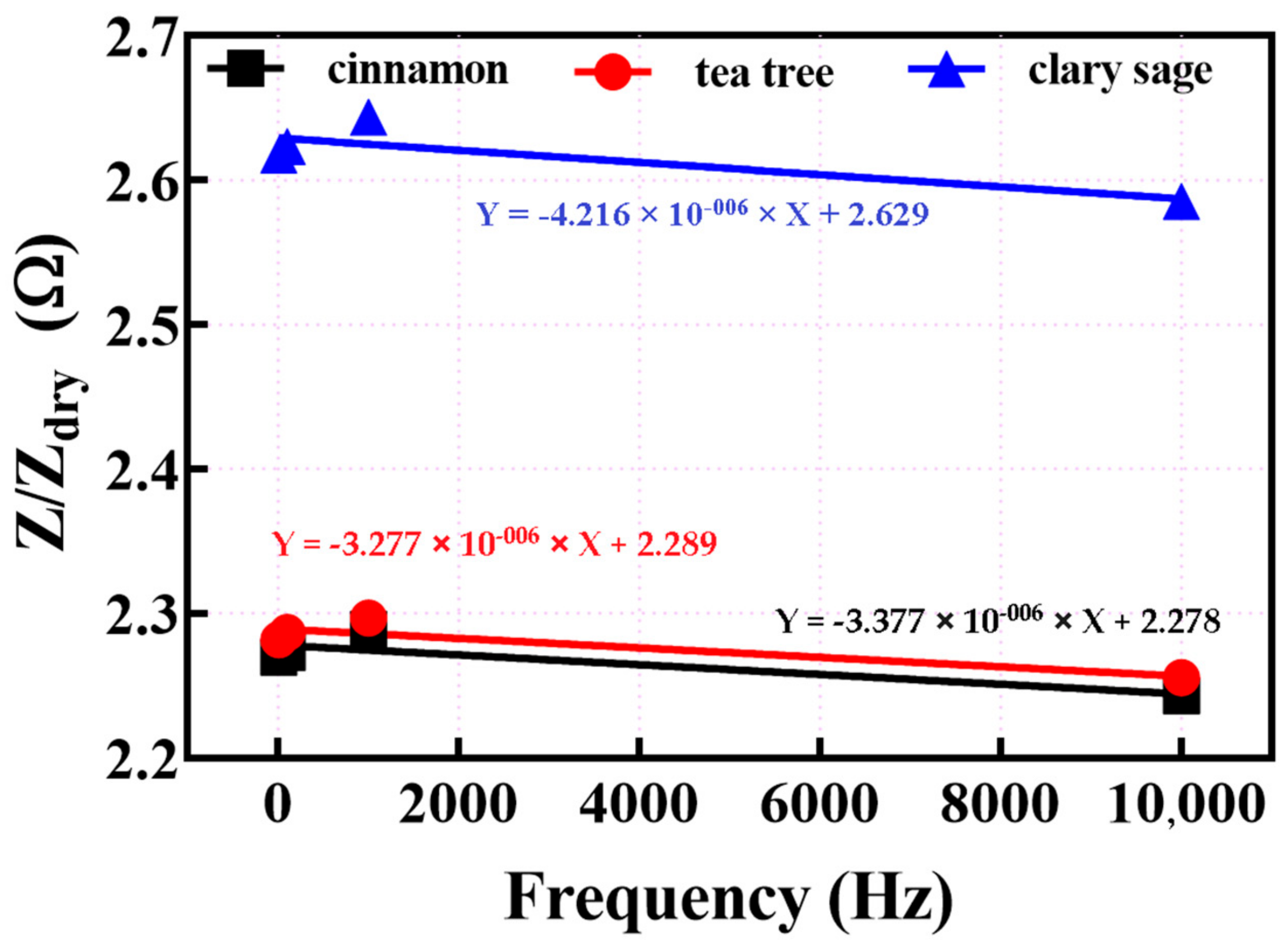
3.2. EIS Measurements
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Shi, J.; Liu, S.; Zhang, L.; Yang, B.; Shu, L.; Yang, Y.; Ren, M.; Wang, Y.; Chen, J.; Chen, W.; et al. Smart Textile-Integrated Microelectronic Systems for Wearable Applications. Adv. Mater. 2020, 32, 1901958. [Google Scholar] [CrossRef] [PubMed]
- Seyedin, S.; Zhang, P.; Naebe, M.; Qin, S.; Chen, J.; Wang, X.; Razal, J.M. Textile strain sensors: A review of the fabrication technologies, performance evaluation and applications. Mater. Horiz. 2019, 6, 219–249. [Google Scholar] [CrossRef]
- Cerempei, A.; Muresan, E.; Sandu, I.; Chirila, L. Textile Materials with Controlled Release of Rosemary Essential Oil. Rev. Chim. 2014, 65, 1154–1157. [Google Scholar]
- Chirila, L.; Constantinescu, G.; Danila, A.; Popescu, A.; Constantinescu, R.; Sandulache, I. Functionalization of textile materials with bioactive polymeric systems based on propolis and cinnamon essential oil. Ind. Text. 2020, 71, 186–192. [Google Scholar] [CrossRef]
- Ghayempour, S.; Montazer, M. Micro/nanoencapsulation of essential oils and fragrances: Focus on perfumed, antimicrobial, mosquito-repellent and medical textiles. J. Microencapsul. 2016, 33, 497–510. [Google Scholar] [CrossRef]
- Stan, M.S.; Chirila, L.; Popescu, A.; Radulescu, D.M.; Radulescu, D.E.; Dinischiotu, A. Essential Oil Microcapsules Immobilized on Textiles and Certain Induced Effects. Materials 2019, 12, 2029. [Google Scholar] [CrossRef] [Green Version]
- Štular, D.; Jerman, I.; Mihelčič, M.; Simončič, B.; Tomšič, B. Antimicrobial activity of essential oils and their controlled release from the smart PLA fabric. IOP Conf. Ser. Mater. Sci. Eng. 2018, 460, 012011. [Google Scholar] [CrossRef]
- Matusiak, K.; Machnowski, W.; Wrzosek, H.; Polak, J.; Rajkowska, K.; Smigielski, K.; Kunicka-Styczynska, A.; Gutarowska, B. Application of Cinnamomum zeylanicum essential oil in vapour phase for heritage textiles disinfection. Int. Biodeterior. Biodegrad. 2017, 131, 88–96. [Google Scholar] [CrossRef]
- Julaeha, E.; Puspita, S.; Rakhmawaty, D.; Tatang Wahyudi, E.; Nurzaman, M.; Nugraha, J.; Herlina, T.; Al Anshori, J. Microencapsulation of lime (Citrus aurantifolia) oil for antibacterial finishing of cotton fabric. RSC Adv. 2021, 11, 1743. [Google Scholar] [CrossRef]
- Bakkali, F.; Averbeck, S.; Averbeck, D.; Idaomar, M. Biological effects of essential oils–A review. Food Chem. Toxicol. 2008, 46, 446–475. [Google Scholar] [CrossRef]
- Walentowska, J.; Foksowicz-Flaczyk, J. Thyme essential oil for antimicrobial protection of natural textiles. Int. Biodeterior. Biodegrad. 2013, 84, 407–411. [Google Scholar] [CrossRef]
- Kamrudi, N.; Akbari, S.; Haghighat Kish, M. The odour assessment of thyme essential oils in electrospun fibre mat with a virtual sensor array data and its relation to antibacterial activity. J. Microencapsul. 2020, 37, 144–159. [Google Scholar] [CrossRef] [PubMed]
- Dhouibi, I.; Masmoudi, F.; Bouaziz, M.; Masmoud, M. A study of the anti-corrosive effects of essential oils of rosemary and myrtle for copper corrosion in chloride media. Arab. J. Chem. 2021, 14, 102961. [Google Scholar] [CrossRef]
- Mzioud, K.; Habsaoui, A.; Ouakki, M.; Galai, M.; Souad, E.; Touhami, M. Inhibition of copper corrosion by the essential oil of Allium sativum in 0.5M H2SO4 solutions. SN Appl. Sci. 2020, 2, 1611. [Google Scholar] [CrossRef]
- Elvina, R.; Mojic, E.-R. Orange Peel Essential Oil as Component of a Metal Sensor for Lead (II) Ion Determination in Aqueous Solutions. J. Appl. Sci. Environ. Manag. 2005, 9, 23–25. [Google Scholar]
- de Azevedo Sá, C.S.; Ladchumananandasivam, R.; Rossi, C.G.F.T.; da Silva, R.K.; da Silva Camboim, W.; Zille, A.; Padrão, J.; de Oliveira Souto Silva, K.K. Characterization of a natural surfactant from an essential oil from neem (Azadirachta indica A. Juss) for textile industry applications. Text. Res. J. 2021, 55, 1444–1454. [Google Scholar] [CrossRef]
- Do, T.; Hadji-Minaglou, F.; Antoniotti, S.; Fernandez, X. Authenticity of essential oils. TrAC Trends Anal. Chem. 2014, 66, 146–157. [Google Scholar] [CrossRef]
- Jjunju, F.P.M.; Giannoukos, S.; Marshall, A.; Taylor, S. In-Situ Analysis of Essential Fragrant Oils Using a Portable Mass Spectrometer. Int. J. Anal. Chem. 2019, 2019, 1780190. [Google Scholar] [CrossRef] [Green Version]
- Graboski, A.; Zakrzevski, C.; Shimizu, F.; Paschoalin, R.; Soares, A.; Steffens, J.; Paroul, N.; Steffens, C. Electronic Nose Based on Carbon Nanocomposite Sensors for Clove Essential Oil Detection. ACS Sens. 2020, 5, 1814–1821. [Google Scholar] [CrossRef]
- Sudarmaji, A.; Margiwiyatno, A.; Ediati, R.; Mustofa, A. Vapor Measurement System of Essential Oil Based on MOS Gas Sensors Driven with Advanced Temperature Modulation Technique. IOP Conf. Ser. Earth Environ. Sci. 2018, 147, 012046. [Google Scholar] [CrossRef]
- Wu, T.; Wu, H.; Du, Y.; Peng, Z. Progress and trend of sensor technology for on-line oil monitoring. Sci. China Technol. Sci. 2013, 56, 2914–2926. [Google Scholar] [CrossRef]
- Liu, H.; Tang, X.; Lu, H.; Xie, W.; Hu, Y.; Xue, Q. An interdigitated impedance microsensor for detection of moisture content in engine oil. Nanotechnol. Precis. Eng. 2020, 3, 75–80. [Google Scholar] [CrossRef]
- Amann Group, Textile Threads. Available online: https://www.amann.com/ (accessed on 20 February 2022).
- Bell, E.W.; Zhang, Y. DockRMSD: An open-source tool for atom mapping and RMSD calculation of symmetric molecules through graph isomorphism. J. Cheminform. 2019, 11, 40. [Google Scholar] [CrossRef] [Green Version]
- Ayna, A.; Moody, P.C. Crystal structures of a dual coenzyme specific glyceraldehyde-3-phosphate dehydrogenase from the enteric pathogen Campylobacter jejuni. J. Mol. Struct. 2021, 1242, 130820. [Google Scholar] [CrossRef]
- Alaoui Ismaili, S.; Belgharza, M.; Marmouzi, I.; Saaid, H.; Kitane, S.; Alaoui El Belghiti, M. Study of the electrical resistivity of vegetable and essential oils. Der Pharma Chemica 2015, 7, 294–296. [Google Scholar]
- Banerjee, S.; Bonde, C. Total phenolic content and antioxidant activity of extracts of Bridelia Retusa Spreng Bark: Impact of dielectric constant and geographical location. J. Med. Plants Res. 2011, 5, 817–822. [Google Scholar]
- Pratap Singh, S.; Singh Chandel, V.; Manohar, R. Dielectric study of Clove oil. J. Ayurveda Integr. Med. 2018, 9, 53–56. [Google Scholar] [CrossRef]




| EOs | Linearity (R2) | |LOD| | |LOQ| |
|---|---|---|---|
| Tea Tree | 0.8045 | 5780.43 | 17,516.40 |
| Clary Sage | 0.7088 | 7515.51 | 22,774.30 |
| Cinnamon | 0.7307 | 7118.50 | 21,571.20 |
| EO Type | Concentration | Time (Minutes) | RMSD |
|---|---|---|---|
| Cinnamon | 0.2% | 3 | 34.03 |
| Clary Sage | 0.2% | 3 | 55.37 |
| Clary Sage | 0.2% | 30 | 60.64 |
| Clary Sage | 0.2% | 60 | 67.23 |
| Tea Tree | 0.2% | 3 | 34.65 |
| Tea Tree | 0.2% | 30 | 36.10 |
| Tea Tree | 0.2% | 60 | 38.15 |
| Tea Tree | 4% | 3 | 40.86 |
| Tea Tree | 10% | 3 | 51.36 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kojic, T.; Kovacevic, B.; Sinha, A.; Simić, M.; Stojanović, G.M. Silver Thread-Based Microfluidic Platform for Detection of Essential Oils Using Impedance Spectroscopy. Appl. Sci. 2022, 12, 3596. https://doi.org/10.3390/app12073596
Kojic T, Kovacevic B, Sinha A, Simić M, Stojanović GM. Silver Thread-Based Microfluidic Platform for Detection of Essential Oils Using Impedance Spectroscopy. Applied Sciences. 2022; 12(7):3596. https://doi.org/10.3390/app12073596
Chicago/Turabian StyleKojic, Tijana, Bozica Kovacevic, Ankita Sinha, Mitar Simić, and Goran M. Stojanović. 2022. "Silver Thread-Based Microfluidic Platform for Detection of Essential Oils Using Impedance Spectroscopy" Applied Sciences 12, no. 7: 3596. https://doi.org/10.3390/app12073596
APA StyleKojic, T., Kovacevic, B., Sinha, A., Simić, M., & Stojanović, G. M. (2022). Silver Thread-Based Microfluidic Platform for Detection of Essential Oils Using Impedance Spectroscopy. Applied Sciences, 12(7), 3596. https://doi.org/10.3390/app12073596








