In Vitro Antibacterial Activity of Marine Microalgae Extract against Vibrio harveyi
Abstract
:Featured Application
Abstract
1. Introduction
2. Materials and Methods
2.1. Cultures and Preparation of Microalgae Extract
2.2. Preparation of Vibrio harveyi Inoculums
2.3. Disc Diffusion Antibacterial Assay
2.4. Co-Culture Antibacterial Assay
2.5. Statistical Analysis
3. Results
3.1. Disc Diffusion Antibacterial Assay
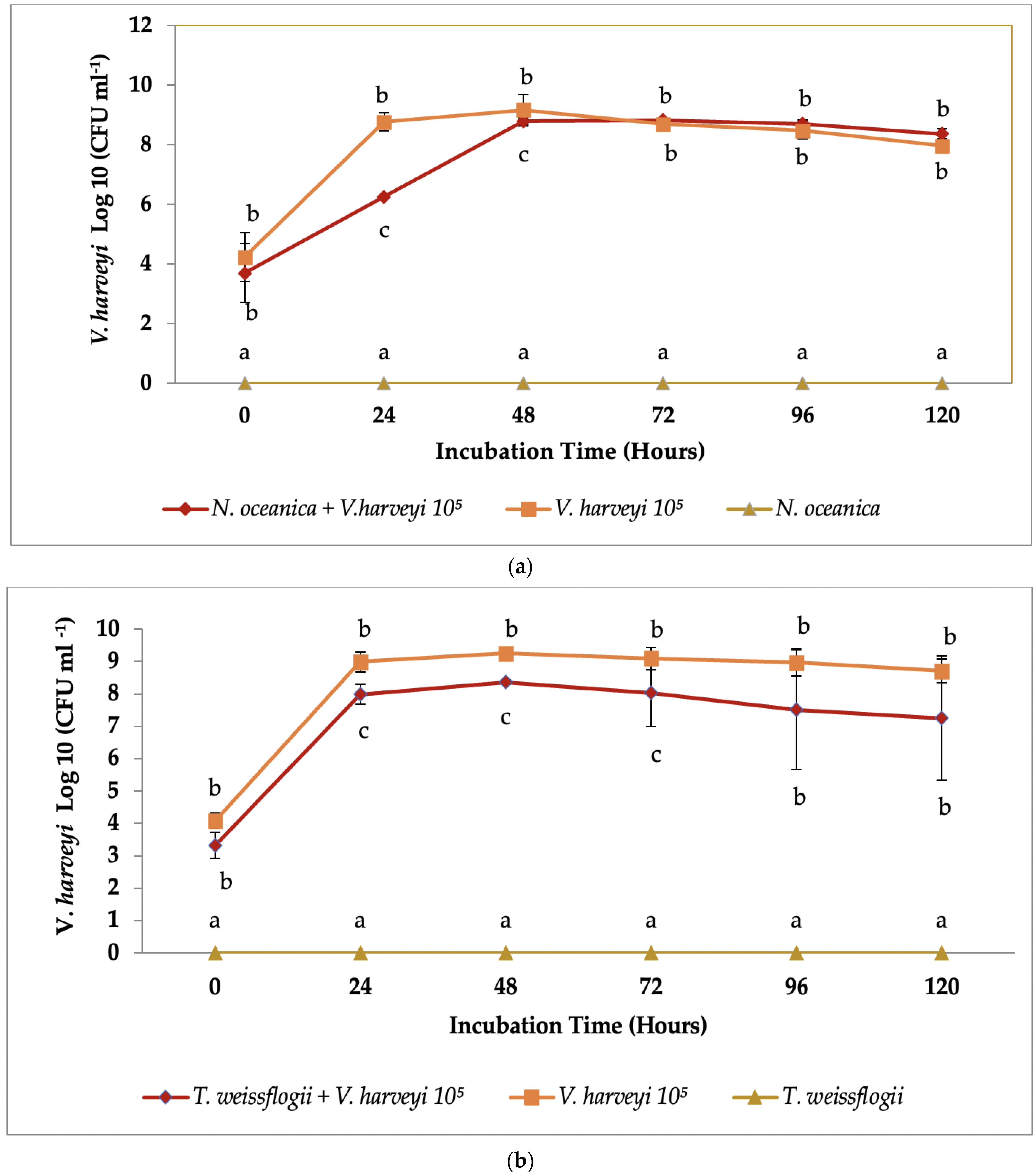
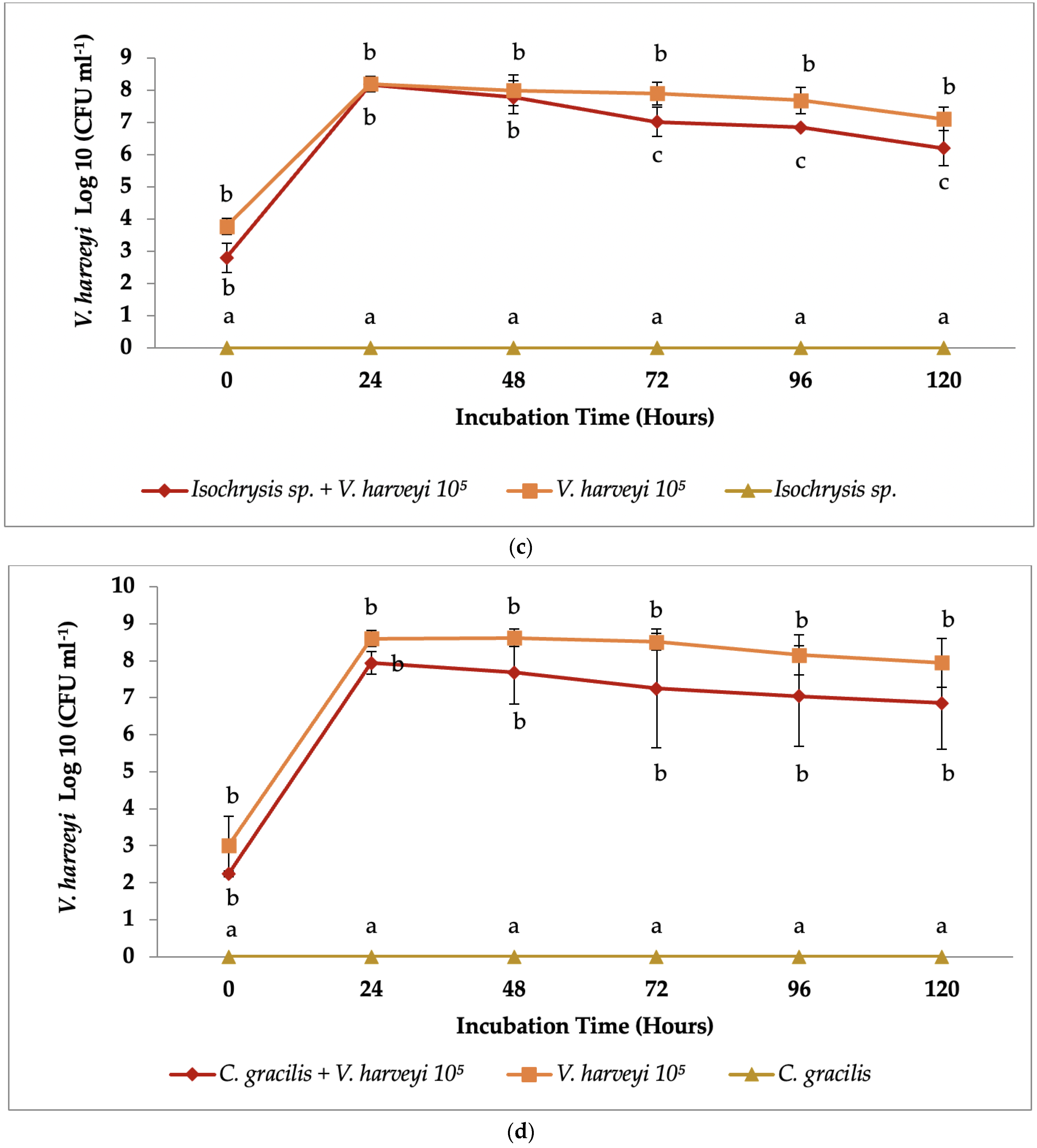
3.2. Co-Culture Antibacterial Assay
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Pauly, D.; Zeller, D. Comments on FAOs State of World Fisheries and Aquaculture (SOFIA 2016). Mar. Policy 2017, 77, 176–181. [Google Scholar] [CrossRef]
- Ma, K.; Bao, Q.; Wu, Y.; Chen, S.; Zhao, S.; Wu, H.; Fan, J. Evaluation of Microalgae as Immunostimulants and Recombinant Vaccines for Diseases Prevention and Control in Aquaculture. Front. Bioeng. Biotechnol. 2020, 8, 1331. [Google Scholar] [CrossRef] [PubMed]
- Wei, L.S.; Wee, W. Diseases in aquaculture. Res. J. Ani. Vet. Sci. 2014, 7, 1–6. [Google Scholar]
- Mohd Yazid, S.H.; Mohd Daud, H.; Azmai, M.N.A.; Mohamad, N.; Mohd Nor, N. Estimating the Economic Loss Due to Vibriosis in Net-Cage Cultured Asian Seabass (Lates calcarifer): Evidence from the East Coast of Peninsular Malaysia. Front. Vet. Sci. 2021, 8, 1111. [Google Scholar] [CrossRef]
- Ina-Salwany, M.Y.; Al-Saari, N.; Mohamad, A.; Mursidi, F.A.; Mohd-Aris, A.; Amal, M.N.A.; Kasai, H.; Mino, S.; Sawabe, T.; Zamri-Saad, M. Vibriosis in Fish: A Review on Disease Development and Prevention. J. Aquat. Anim. Health 2019, 31, 3–22. [Google Scholar] [CrossRef]
- Farisa, M.Y.; Namaskara, K.E.; Yusuf, M.B.; Desrina. Antibacterial Potention of Extract of Rotifers Fed with Different Microalgae to Control Vibrio harveyi. IOP Conf. Ser. Earth Environ. Sci. 2019, 246, 012058. [Google Scholar] [CrossRef]
- Aftabuddin, S.; Siddique, M.A.M.; Habib, A.; Akter, S.; Hossen, S.; Tanchangya, P.; Abdullah, M.A.l. Effects of seaweeds extract on growth, survival, antibacterial activities, and immune responses of Penaeus monodon against Vibrio parahaemolyticus. Ital. J. Anim. Sci. 2021, 20, 243–255. [Google Scholar] [CrossRef]
- Manyi-Loh, C.; Mamphweli, S.; Meyer, E.; Okoh, A. Antibiotic Use in Agriculture and Its Consequential Resistance in Environmental Sources: Potential Public Health Implications. Molecules 2018, 23, 795. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Molina-Cárdenas, C.A.; Sánchez-Saavedra, M.P.; Lizárraga-Partida, M.L. Inhibition of pathogenic Vibrio by the microalgae Isochrysis galbana. J. Appl. Phycol. 2014, 26, 2347–2355. [Google Scholar] [CrossRef]
- Reverter, M.; Bontemps, N.T.; Sasal, P.; Saulnier, D. Use of medicinal plants in aquaculture. In Diagnosis and Control of Diseases of Fish and Shellfish; Austin, B., Newaj-Fyzul, A., Eds.; Wiley: Hoboken, NJ, USA, 2017; pp. 223–261. [Google Scholar]
- Khavari, F.; Saidijam, M.; Taheri, M.; Nouri, F. Microalgae: Therapeutic potentials and applications. Mol. Biol. Rep. 2021, 48, 4757–4765. [Google Scholar] [CrossRef]
- Abdullah, A.; Ramli, R.; Ridzuan, M.S.M.; Murni, M.; Hashim, S.; Sudirwan, F.; Abdullah, S.Z.; Mansor, N.N.; Amira, S.; Zamri-Saad, M.; et al. The presence of Vibrionaceae, Betanodavirus and Iridovirus in marine cage-cultured fish: Role of fish size, water physicochemical parameters and relationships among the pathogens. Aquac. Rep. 2017, 7, 57–65. [Google Scholar] [CrossRef]
- Mohamad, N.; Mustafa, M.; Amal, M.N.A.; Saad, M.Z.; Md-Yasin, I.S.; Al-saari, N. Environmental factors associated with the presence of Vibrionaceae in tropical cage-cultured marine fishes. J. Aquat. Anim. Health 2019, 31, 154–167. [Google Scholar] [CrossRef]
- Salvesen, I.; Reitan, K.I.; Skjermo, J.; Oie, G. Microbial environments in marine larviculture: Impacts of algal growth rates on the bacterial load in six microalgae. Aquac. Int. 2000, 8, 275–287. [Google Scholar] [CrossRef]
- Guillard, R.R.; Ryther, J.H. Studies on marine planktonic diatoms. I. Cyclotella nana Hustedt and Detonula confervacaea (Cleve) Gran. Can. J. Microbiol. 1962, 8, 229–239. [Google Scholar] [CrossRef]
- Sasireka, G.; Muthuvelayudham, R. Effect of Salinity and Iron Stressed on Growth and Lipid Accumulation in Skeletonema costatum for Biodiesel Production. Res. J. Chem. Sci. 2015, 5, 69–72. [Google Scholar]
- Gonzalez, D.V.A.; Platas, G.; Basilio, A. Screening of antimicrobial activities in red, green and brown macroalgae from Gran Canaria (Canary Islands, Spain). Int. Microbiol. 2001, 4, 35–40. [Google Scholar]
- Bauer, A.N.; Kirby, W.M.M.; Sherries, J.C.; Truck, M. Antibiotic susceptibility testing by a standardized single disk method. Am. J. Clin. Pathol. 1966, 45, 493–496. [Google Scholar] [CrossRef] [PubMed]
- Corona, E.; Fernandez-Acero, J.; Bartual, A. Screening study for antibacterial activity from marine and freshwater microalgae. Int. J. Pharm. Bio Sci. 2017, 8, 189–194. [Google Scholar]
- Prasetiya, F.S.; Sunarto, S.; Bachtiar, E.; Mochamad, U.K.A.; Nathanael, B.; Pambudi, A.C.; Lestari, A.D.; Astuty, S.; Mouget, J.-L. Effect of the blue pigment produced by the tropical diatom Haslea nusantara on marine organisms from different trophic levels and its bioactivity. Aquac. Rep. 2020, 17, 100389. [Google Scholar] [CrossRef]
- Soto-Rodriguez, S.A.; Lopez-Vela, M.; Soto, M.N. 2021 Inhibitory effect of marine microalgae used in shrimp hatcheries on Vibrio parahaemolyticus responsible for acute hepatopancreatic necrosis disease. Aquac. Res. 2021, 1–11. [Google Scholar]
- Barone, M.E.; Murphy, E.; Parkes, R.; Fleming, G.T.A.; Campanile, F.; Thomas, O.P.; Touzet, N. Antibacterial Activity and Amphidinol Profiling of the Marine Dinoflagellate Amphidinium carterae (Subclade III). Int. J. Mol. Sci. 2021, 22, 12196. [Google Scholar] [CrossRef]
- Desbois, A.P.; Mearns-Spragg, A.; Smith, V.J. A fatty acid from the diatom Phaeodactylum tricornutum is antibacterial against diverse bacteria including multi-resistant Staphylococcus aureus (MRSA). Mar. Biotechnol. 2009, 11, 45–52. [Google Scholar] [CrossRef]
- Duff, D.C.B.; Bruce, D.L. The antibacterial activity of marine planktonic algae. Can. J. Microbiol. 1966, 12, 877–884. [Google Scholar] [CrossRef] [PubMed]
- Borowitzka, M.A. Microalgae as sources of pharmaceuticals and other biologically active compounds. J. Appl. Phycol. 1995, 7, 3–15. [Google Scholar] [CrossRef]
- Navarro, F.; Forján, E.; Vázquez, M.; Toimil, A.; Montero, Z.; Ruiz-Domínguez, M.D.C.; Garbayo, I.; Castaño, M.A.; Vilchez, C.; Vega, J.M. Antimicrobial activity of the acidophilic eukaryotic microalga Coccomyxa onubensis. Phycol Res. 2017, 65, 38–43. [Google Scholar] [CrossRef]
- Falaise, C.; François, C.; Travers, M.A.; Morga, B.; Haure, J.; Tremblay, R.; Turcotte, F.; Pasetto, P.; Gastineau, R.; Hardivillier, Y.; et al. Antimicrobial Compounds from Eukaryotic Microalgae against Human Pathogens and Diseases in Aquaculture. Mar. Drugs 2016, 14, 159. [Google Scholar] [CrossRef] [Green Version]
- Pawlik-Skowrońska, B. Resistance, accumulation and allocation of zinc in two ecotypes of the green alga Stigeoclonium tenue Kütz. coming from habitats of different heavy metal concentrations. Aquat. Bot. 2003, 75, 189–198. [Google Scholar] [CrossRef]
- Lustigman, B. Comparison of antibiotic production from four ecotypes of the marine alga, Dunaliella Bull. Environ. Contam. Toxicol. 1988, 40, 18–22. [Google Scholar] [CrossRef] [PubMed]
- Zanella, L.; Vianello, F. Microalgae of the genus Nannochloropsis: Chemical composition and functional implications for human nutrition. J. Funct. Foods 2020, 68, 103919. [Google Scholar] [CrossRef]
- Sun, Z.; Wang, X.; Liu, J. Screening of Isochrysis strains for simultaneous production of docosahexaenoic acid and fucoxanthin. Algal Res. 2019, 41, 101545. [Google Scholar] [CrossRef]
- Marella, T.K.; Tiwari, A. Marine diatom Thalassiosira weissflogii based biorefinery for co-production of eicosapentaenoic acid and fucoxanthin. Bioresour. Technol. 2020, 307, 123245. [Google Scholar] [CrossRef]
- Merchie, G.; Lavens, P.; Sorgeloos, P. Optimization of dietary vitamin C in fish and crustacean larvae: A review. Aquaculture 1997, 155, 165–181. [Google Scholar] [CrossRef]
- Kanjana, K.; Radtanatip, T.; Asuvapongpatana, S.; Withyachumnarnkul, B.; Wongprasert, K. Solvent extracts of the red seaweed Gracilaria fisheri prevent Vibrio harveyi infections in the black tiger shrimp Penaeus monodon, Fish. Shellfish Immunol. 2011, 30, 389–396. [Google Scholar] [CrossRef]
- Padmakumar, K.; Ayyakkannu, K. Seasonal variation of antibacterial and antifungal activities of marine algae from southern coast of India. Bot. Mar. 1997, 40, 507–515. [Google Scholar] [CrossRef]
- Pradhan, J.; Das, B.K.; Sahu, S.; Marhual, N.P.; Swain, A.K.; Mishra, B.K.; Eknath, A.E. Traditional antibacterial activity of fresh water microalga Spirulina platensis to aquatic pathogens. Aquac. Res. 2012, 43, 1287–1295. [Google Scholar] [CrossRef]
- Patil, L.; Kaliwal, B.B. Microalga Scenedesmus bajacalifornicus BBKLP-07, a new source of bioactive compounds with in vitro pharmacological applications. Bioprocess Biosyst. Eng. 2019, 42, 979–994. [Google Scholar] [CrossRef] [PubMed]
- Zea-Obando, C.; Tunin Ley, A.; Turquet, J.; Culioli, G.; Briand, J.F.; Bazire, A.; Réhel, K.; Faÿ, F.; Linossier, I. Anti-bacterial adhesion activity of tropical microalgae extracts. Molecules 2018, 23, 2180. [Google Scholar] [CrossRef] [Green Version]
- Kilic, N.K.; Erdem, K.; Donmez, G. Bioactive compounds produced by Dunaliella species, antimicrobial effects and optimization of the efficiency. Turk. J. Fish. Aquat. Sci. 2018, 19, 923–933. [Google Scholar]
- Prakash, J.W.; Antonisamy, J.M.; Jeeve, S. Antimicrobial activity of certain fresh water microalgae from Thamirabarani River, Tamil Nadu, South India. Asian Pac. J. Tro. Biomed. 2011, 1, S170–S173. [Google Scholar] [CrossRef]
- Pradhan, B.; Patra, S.; Dash, S.R.; Nayak, R.; Behera, C.; Jena, M. Evaluation of the anti-bacterial activity of methanolic extract of Chlorella vulgaris Beyerinck [Beijerinck] with special reference to antioxidant modulation. Futur. J. Pharm. Sci. 2021, 7, 17. [Google Scholar] [CrossRef]
- Dantas, D.M.D.M.; Oliveira, C.Y.B.D.; Costa, R.M.P.B.; Carneiro-da-Cunha, M.D.G.; Gálvez, A.O.; Bezerra, R.D.S. Evaluation of antioxidant and antibacterial capacity of green microalgae Scenedesmus subspicatus. Food Sci. Technol. Int. 2019, 25, 318–326. [Google Scholar] [CrossRef] [PubMed]
- Herrero, M.; Jaime, L.; Martin-Alvarez, J.M.; Cifuentes, A.; Ibanez, E. Optimization of the Extraction of Antioxidants from Dunaliella salina Microalga by Pressurized Liquids. J. Agric. Food Chem. 2006, 54, 5597–5603. [Google Scholar] [CrossRef] [PubMed]
- Kokou, F.; Makridis, P.; Kentouri, M.; Divanach, P. Antibacterial activity in microalgae cultures. Aquac. Res. 2012, 43, 1520–1527. [Google Scholar] [CrossRef]
- Roncarati, A.; Meluzzi, A.; Acciari, S.; Tallarico, N.; Melotti, P. Fatty acid composition of different microalgae strains (Nannochloropsis sp., Nannochloropsis oculata (Droop) Hibberd, Nannochloropsis atomus Butcher and Isochrysis sp. according to the culture phase and the carbon dioxide concentration. J. World Aquac. Soc. 2004, 35, 401–411. [Google Scholar] [CrossRef]
- Defoirdt, T.; Boon, N.; Sorgeloos, P.; Verstraete, W.; Bossier, P. Alternatives to antibiotics to control bacterial infections: Luminescent vibriosis in aquaculture as an example. Trends Biotechol. 2007, 25, 472–479. [Google Scholar] [CrossRef] [Green Version]
- Elkomy, R.; Ibraheem, I.B.M.; Shreadah, M.; Mohammed, R. Optimal conditions for antimicrobial activity production from two microalgae Chlorella marina and Navicula f. delicatula. J. Pure Appl. Microbiol. 2015, 9, 2725–2732. [Google Scholar]
- Dineshkumar, R.; Narendran, R.; Jayasingam, P.; Sampath-kumar, P. Cultivation and chemical composition of microalgae Chlorella vulgaris and its antibacterial activity against human pathogens. J. Aquac. Mar. Biol. 2017, 5, 00119. [Google Scholar]
- Hamouda, R.A.E.; Abou-El-Souod, G.W. Influence of various concentrations of phosphorus on the antibacterial, antioxidant and bioactive components of green microalgae Scenedesmus obliquus. Int. J. Pharmacol. 2018, 14, 99–107. [Google Scholar]
- Ruffell, S.E.; Müller, K.M.; McConkey, B.J. Comparative assessment of microalgal fatty acids as topical antibiotics. J. Appl. Phycol. 2016, 28, 1695–1704. [Google Scholar] [CrossRef]
- Schuelter, A.R.; Kroumov, A.D.; Hinterholz, C.L.; Fiorini, A.; Trigueros, D.E.G.; Vendruscolo, E.G.; Zaharieva, M.M.; Módenes, A.N. Isolation and identification of new microalgae strains with antibacterial activity on food-borne pathogens: Engineering approach to optimize synthesis of desired metabolites. Biochem. Eng. J. 2019, 144, 28–39. [Google Scholar] [CrossRef]
- Santhakumaran, P.; Kookal, S.K.; Mathew, L.; Ray, J.G. Experimental evaluation of the culture parameters for optimum yield of lipids and other nutraceutically valuable compounds in Chloroidium saccharophillum (Kruger) comb. Nov. Renew. Energy 2020, 147, 1082–1097. [Google Scholar] [CrossRef]
- Noaman, N.H.; Fattah, A.; Khaleafa, M.; Zaky, S.H. Factors affecting antimicrobial activity of Synechococcus leopoliensis. Microbiol. Res. 2004, 159, 395–402. [Google Scholar] [CrossRef] [PubMed]
| Concentration of V. harvey | Extractant | Diameter of the Inhibition Zone (D) in mm by Marine Microalgae Extract (106 Cell/mL) and Control (Extractant) after 24 h Incubation with V. harveyi | ||||
|---|---|---|---|---|---|---|
| N. oceanica | T. weissflogii | C. gracilis | Isochrysis sp. | Control (Extractant) | ||
| 105 CFU/mL | Ethanol | +++ | +++ | +++ | +++ | − |
| Methanol | ++ | ++ | ++ | ++ | − | |
| Saline water | ++ | + | ++ | ++ | − | |
| DMSO | ++ | + | + | + | − | |
| 106 CFU/mL | Ethanol | +++ | ++ | ++ | +++ | − |
| Methanol | ++ | ++ | + | ++ | − | |
| Saline water | ++ | + | + | ++ | − | |
| DMSO | + | + | + | + | − | |
| 107 CFU/mL | Ethanol | +++ | ++ | + | ++ | − |
| Methanol | + | + | + | + | − | |
| Saline water | + | + | + | + | − | |
| DMSO | + | + | + | + | − | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jusidin, M.R.; Othman, R.; Shaleh, S.R.M.; Ching, F.F.; Senoo, S.; Oslan, S.N.H. In Vitro Antibacterial Activity of Marine Microalgae Extract against Vibrio harveyi. Appl. Sci. 2022, 12, 1148. https://doi.org/10.3390/app12031148
Jusidin MR, Othman R, Shaleh SRM, Ching FF, Senoo S, Oslan SNH. In Vitro Antibacterial Activity of Marine Microalgae Extract against Vibrio harveyi. Applied Sciences. 2022; 12(3):1148. https://doi.org/10.3390/app12031148
Chicago/Turabian StyleJusidin, Mohd Ridzwan, Rafidah Othman, Sitti Raehanah Muhamad Shaleh, Fui Fui Ching, Shigeharu Senoo, and Siti Nur Hazwani Oslan. 2022. "In Vitro Antibacterial Activity of Marine Microalgae Extract against Vibrio harveyi" Applied Sciences 12, no. 3: 1148. https://doi.org/10.3390/app12031148
APA StyleJusidin, M. R., Othman, R., Shaleh, S. R. M., Ching, F. F., Senoo, S., & Oslan, S. N. H. (2022). In Vitro Antibacterial Activity of Marine Microalgae Extract against Vibrio harveyi. Applied Sciences, 12(3), 1148. https://doi.org/10.3390/app12031148







