Toxic Metals in Meat Contributed by Helicopter and Rifle Thoracic Killing of Game Meat Animals
Abstract
1. Introduction
1.1. Occurrences and Accumulation of Toxic Metals in Farmed Animals
1.2. Composition of Shotgun and Rifle Ammunition
2. Materials and Methods
2.1. Sampling and Sample Handling
2.2. Sample Analysis
2.3. Data Analysis
3. Results
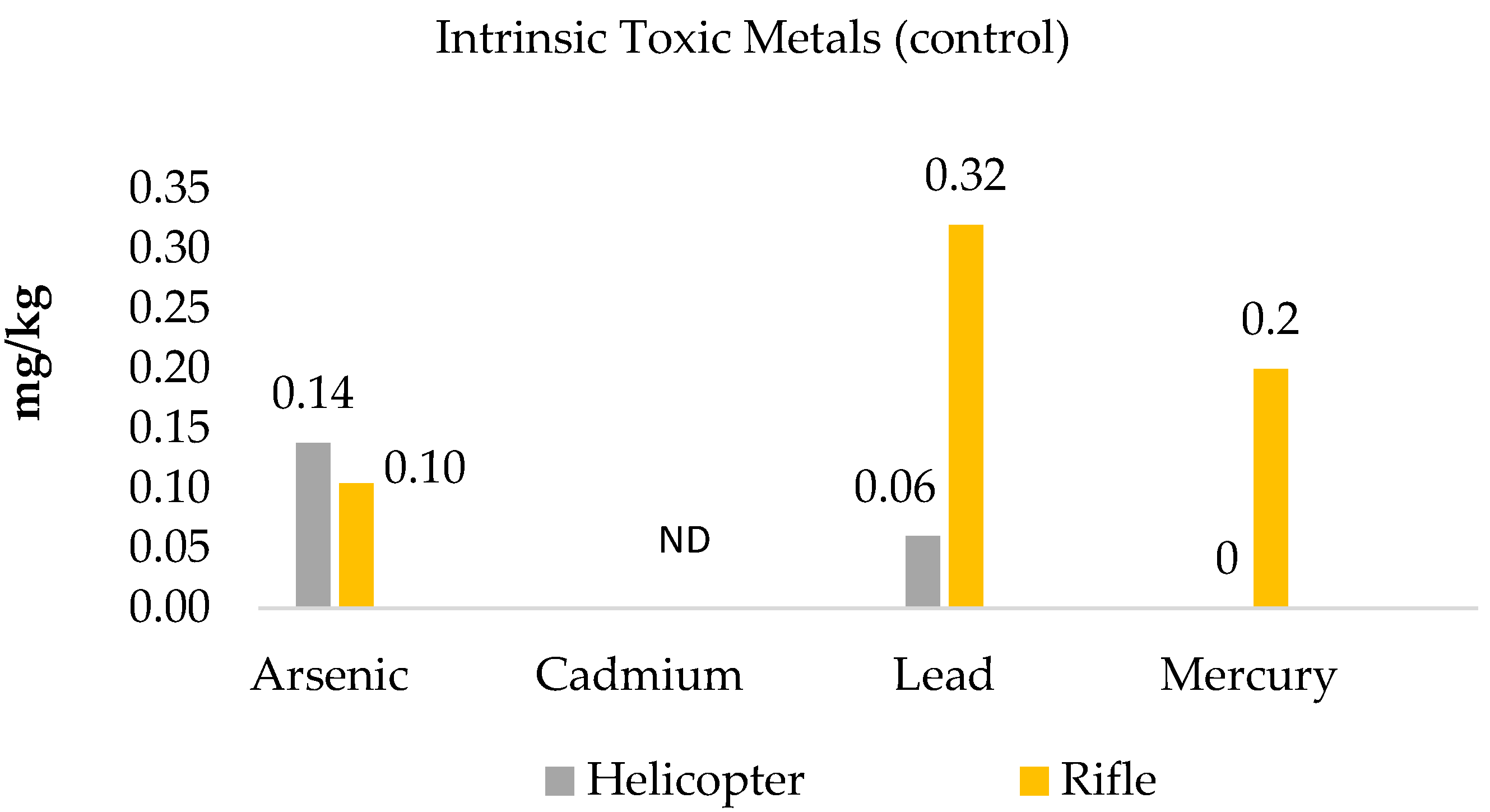
3.1. Intrinsic Toxic Metals
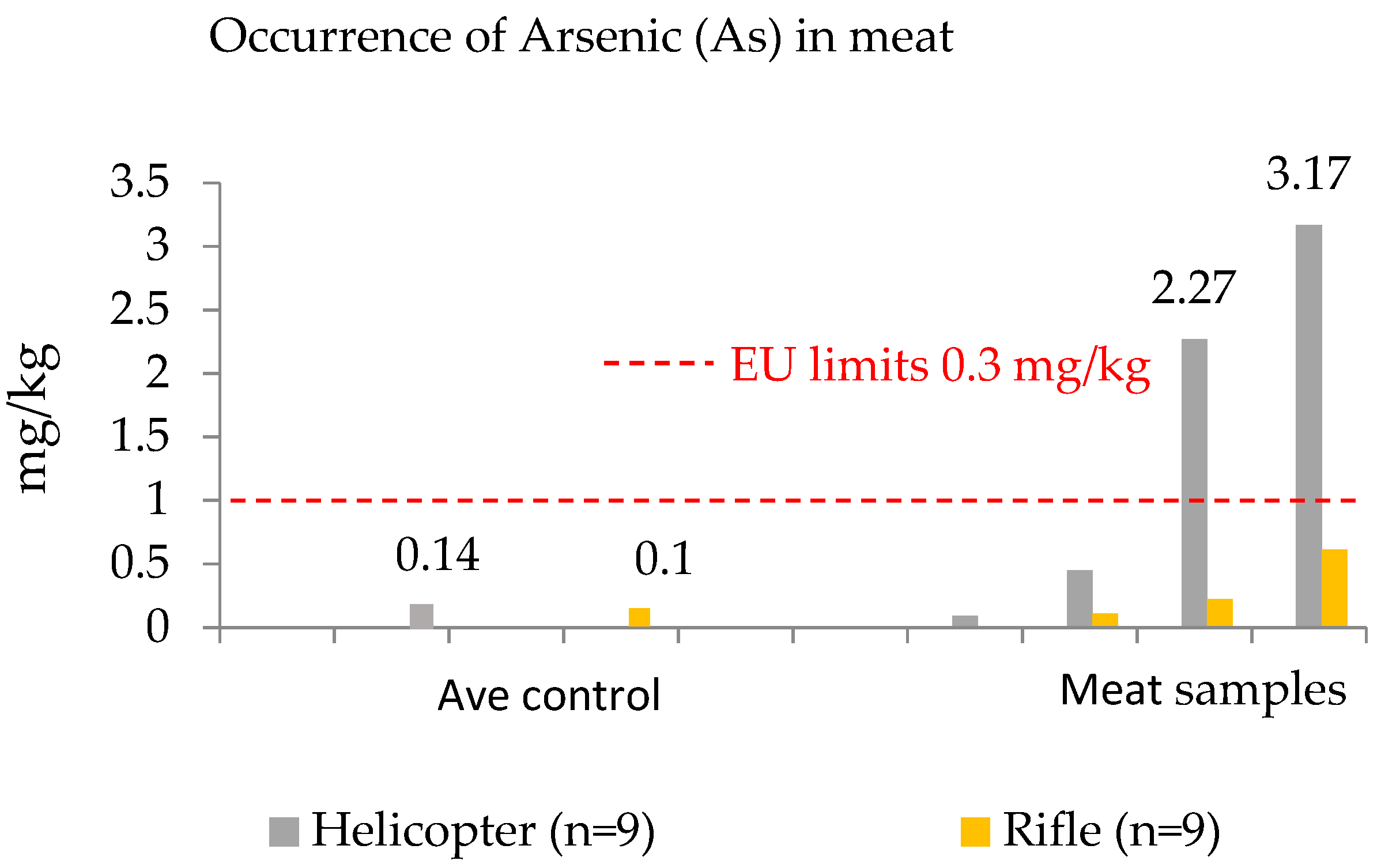
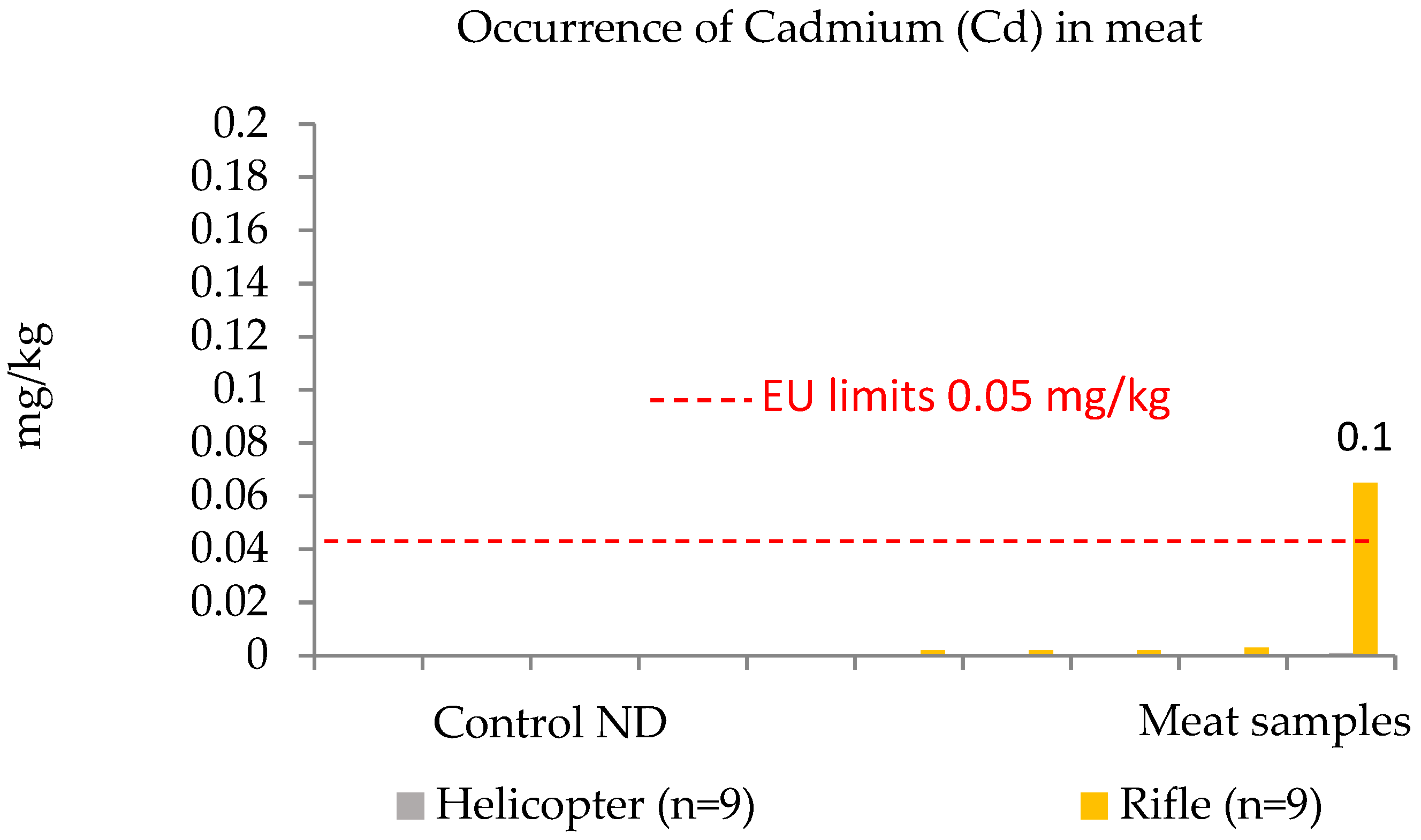
3.2. Toxic Metals Contributed by Killing Methods
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Hoffman, L.C.; Wiklund, E. Game and venison–meat for the modern consumer. Meat Sci. 2006, 74, 197–208. [Google Scholar] [CrossRef] [PubMed]
- White, P.A.; Belant, J.L. Provisioning of game meat to rural communities as a benefit of sport hunting in Zambia. PLoS ONE 2015, 10, e0117237. [Google Scholar] [CrossRef] [PubMed]
- Flora, S.J. Toxic metals: Health effects, and therapeutic measures. J. Biomed. Sci. 2014, 1, 48–64. [Google Scholar]
- Prashanth, L.; Kattapagari, K.K.; Chitturi, R.T.; Baddam, V.R.R.; Prasad, L.K. A review on role of essential trace elements in health and disease. J. Dr. NTR Univ. Health Sci. 2015, 4, 75. [Google Scholar]
- Kamunda, C.; Mathuthu, M.; Madhuku, M. Health risk assessment of heavy metals in soils from Witwatersrand gold mining basin, South Africa. Int. J. Environ. Res. Public Health 2016, 13, 663. [Google Scholar] [CrossRef]
- Nkosi, D.V.; Bekker, J.L.; Hoffman, L.C. Toxic Metals in Wild Ungulates and Domestic Meat Animals Slaughtered for Food Purposes: A Systemic Review. Foods 2021, 10, 2853. [Google Scholar] [CrossRef]
- Falandysz, J.; Szymczyk–Kobrzyńska, K.; Brzostowski, A.; Zalewski, K.; Zasadowski, A. Concentrations of heavy metals in the tissues of red deer (Cervus elaphus) from the region of Warmia and Mazury, Poland. Food Addit. Contam. 2005, 22, 141–149. [Google Scholar] [CrossRef]
- Mudgal, V.; Madaan, N.; Mudgal, A.; Singh, R.; Mishra, S. Effect of toxic metals on human health. Open Nutraceuticals J. 2010, 3, 94–99. [Google Scholar] [CrossRef]
- Kortei, N.K.; Heymann, M.E.; Essuman, E.K.; Kpodo, F.M.; Akonor, P.T.; Lokpo, S.Y.; Boadi, N.O.; Ayim-Akonor, M.; Tettey, C. Health risk assessment and levels of toxic metals in fishes (Oreochromis noliticus and Clarias anguillaris) from Ankobrah and Pra basins: Impact of illegal mining activities on food safety. Toxicol. Rep. 2020, 7, 360–369. [Google Scholar] [CrossRef]
- Yakupa, N.Y.; Sabowa, A.B.; Saleha, S.J.; Mohammed, G.R. Assessment of heavy metal in imported red meat available in the markets of Erbil city. J. Univ. Babylon Pure Appl. Sci. 2018, 26, 177–183. [Google Scholar]
- Di Bella, C.; Traina, A.; Giosuè, C.; Carpintieri, D.; Lo Dico, G.M.; Bellante, A.; Core, M.D.; Falco, F.; Gherardi, S.; Uccello, M.M.; et al. Heavy metals and PAHs in meat, milk, and seafood from Augusta area (Southern Italy): Contamination levels, dietary intake, and human exposure assessment. Front. Public Health 2020, 8, 273. [Google Scholar] [CrossRef] [PubMed]
- Fowler, B.A.; Alexander, J.; Oskarsson, A. Toxic metals in food. In Handbook on the Toxicology of Metals; Elsevier: Amsterdam, The Netherlands, 2015; pp. 123–140. [Google Scholar]
- Sudaric, T.; Zmaic, K.; Petrac, M.; Boskovic, I.; Vuksic, N.; Kranjac, D.; Florijancic, T. Multifunctional Aspect of Hunting Tourism—The Case of Croatia. In Proceedings of the 79th International Scientific Conference on Economic and Social Development, Rabat, Morocco, 25–26 March 2022; pp. 170–176. [Google Scholar]
- Moswete, N.N.; Saarinen, J.; Thapa, B. Socio-economic Impacts of Community-Based Ecotourism on Rural Livelihoods: A Case Study of Khawa Village in the Kalahari Region, Botswana. In Southern African Perspectives on Sustainable Tourism Management; Springer: Berlin/Heidelberg, Germany, 2022; pp. 109–124. [Google Scholar]
- Khan, A.; Khan, S.; Khan, M.A.; Qamar, Z.; Waqas, M. The uptake and bioaccumulation of heavy metals by food plants, their effects on plants nutrients, and associated health risk: A review. Environ. Sci. Pollut. Res. 2015, 22, 13772–13799. [Google Scholar] [CrossRef] [PubMed]
- Guynup, S.; Safina, B. Mercury: Sources in the environment, health effects, and politics. Blue Ocean. Inst. 2012, 1, 7–54. [Google Scholar]
- Levin, R.; Vieira, C.L.Z.; Mordarski, D.C.; Rosenbaum, M.H. Lead seasonality in humans, animals, and the natural environment. Environ. Res. 2020, 180, 108797. [Google Scholar] [CrossRef]
- Lazarus, M.; Prevendar Crnić, A.; Bilandžić, N.; Kusak, J.; Reljić, S. Cadmium, lead, and mercury exposure assessment among Croatian consumers of free-living game. Arh. Hig. Rada Toksikol. 2014, 65, 281–291. [Google Scholar] [CrossRef]
- Juric, A.K.; Batal, M.; David, W.; Sharp, D.; Schwartz, H.; Ing, A.; Fediuk, K.; Black, A.; Tikhonov, A.; Chan, H.M. A total diet study and probabilistic assessment risk assessment of dietary mercury exposure among First Nations living on-reserve in Ontario, Canada. Environ. Res. 2017, 158, 409–420. [Google Scholar] [CrossRef]
- McAuley, C.; Ng, C.; McFarland, C.; Dersch, A.; Koppe, B.; Sowan, D. Lead exposure through consumption of small game harvested using lead-based ammunition and the corresponding health risks to First Nations in Alberta, Canada. Cogent Environ. Sci. 2018, 4, 1557316. [Google Scholar] [CrossRef]
- Department of Health. Food Stuff, Cosmetics and Disinfactant Act (Act 54 of 1972): Regulations Relating to Maximum Levels of Metals in Food. In Pretoria 2018. Available online: https://www.dpsa.gov.za (accessed on 2 April 2022).
- Europian Commission. Commision Regulation (EC) No: 78/2005 of 16 January 2005 amending Regulation EC No: 466/2001 as regards heavy metals. Off. J. Eur. Union Legis. Ser. 2005, 16, 43–45. [Google Scholar]
- GB2762-2012; National Food Safety Standard: Maximum Levels of Contaminants in Food. Ministry of Health, People’s Republic of China: Beijing, China, 2013. Available online: https://www.dgav.pt/ (accessed on 2 April 2022).
- Food Standard Australia New Zealand. Contaminants and Natural Toxicants Federal Register of Legislative Instruments 2015. Available online: https://www.foodstandards.gov.au (accessed on 2 March 2022).
- FDA. Potential Hazards Associated with the Manufacturing, Processing, Packing, and Holding of Human Food. Food Safety in the Center for Food Safety and Applied Nutrition at the U.S. Food and Drug Administration; 2018. Available online: https://www.fda.gov (accessed on 1 April 2022).
- Codex Alimentarius Commission. Codex General Standard for Contaminants and Toxins in Food and Feed. 2010. Available online: https://www.fao.org (accessed on 2 March 2022).
- Lü, J.; Jiao, W.-B.; Qiu, H.-Y.; Chen, B.; Huang, X.-X.; Kang, B. Origin and spatial distribution of heavy metals and carcinogenic risk assessment in mining areas at You’xi County southeast China. Geoderma 2018, 310, 99–106. [Google Scholar] [CrossRef]
- Marrugo-Negrete, J.; Pinedo-Hernández, J.; Díez, S. Assessment of heavy metal pollution, spatial distribution and origin in agricultural soils along the Sinú River Basin, Colombia. Environ. Res. 2017, 154, 380–388. [Google Scholar] [CrossRef]
- Bosch, A.C.; O’Neill, B.; Sigge, G.O.; Kerwath, S.E.; Hoffman, L.C. Heavy metals in marine fish meat and consumer health: A review. J. Sci. Food Agric. 2016, 96, 32–48. [Google Scholar] [CrossRef] [PubMed]
- Hampton, J.O.; Laidlaw, M.; Buenz, E.; Arnemo, J.M. Heads in the sand: Public health and ecological risks of lead-based bullets for wildlife shooting in Australia. Wildl. Res. 2018, 45, 287–306. [Google Scholar] [CrossRef]
- Lehel, J.; Laczay, P.; Gyurcsó, A.; Jánoska, F.; Majoros, S.; Lányi, K.; Marosán, M. Toxic heavy metals in the muscle of roe deer (Capreolus capreolus)—Food toxicological significance. Environ. Sci. Pollut. Res. 2016, 23, 4465–4472. [Google Scholar] [CrossRef]
- Oropesa, A.-L.; Ramos, A.; Gómez, L.-J. Toxic and essential metal levels in the hair of red deer (Cervus elaphus) and wild boar (Sus scrofa) for monitoring the contamination in protected areas of South-Western Spain. Environ. Sci. Pollut. Res. 2022, 29, 27430–27442. [Google Scholar] [CrossRef] [PubMed]
- Mateo, R.; Kanstrup, N. Regulations on lead ammunition adopted in Europe and evidence of compliance. Ambio 2019, 48, 989–998. [Google Scholar] [CrossRef] [PubMed]
- Kanstrup, N.; Thomas, V.G. Transitioning to lead-free ammunition use in hunting: Socio-economic and regulatory considerations for the European Union and other jurisdictions. Environ. Sci. Eur. 2020, 32, 91. [Google Scholar] [CrossRef]
- Durkalec, M.; Szkoda, J.; Kolacz, R.; Opalinski, S.; Nawrocka, A.; Zmudzki, J. Bioaccumulation of lead, cadmium and mercury in roe deer and wild boars from areas with different levels of toxic metal pollution. Int. J. Environ. Res. 2015, 9, 205–212. [Google Scholar]
- Irschik, I.; Bauer, F.; Sager, M.; Paulsen, P. Copper residues in meat from wild artiodactyls hunted with two types of rifle bullets manufactured from copper. Eur. J. Wildl. Res. 2013, 59, 129–136. [Google Scholar] [CrossRef]
- Irschik, I.; Wanek, C.; Bauer, F.; Sager, M.; Paulsen, P. Composition of bullets used for hunting and food safety considerations. In Trends in Game Meat Hygiene: From Forest to Fork; Wageningen Academic Publishers: Wageningen, The Netherlands, 2014; pp. 283–289. [Google Scholar]
- Sanchez, D.M.; Epps, C.W.; Taylor, D.S. Estimating lead fragmentation from ammunition for muzzleloading and black powder cartridge rifles. J. Fish Wildl. Manag. 2016, 7, 467–479. [Google Scholar] [CrossRef]
- Dobrowolska, A.; Melosik, M. Bullet-derived lead in tissues of the wild boar (Sus scrofa) and red deer (Cervus elaphus). Eur. J. Wildl. Res. 2008, 54, 231–235. [Google Scholar] [CrossRef]
- Gerofke, A.; Martin, A.; Schlichting, D.; Gremse, C.; Müller-Graf, C. Heavy metals in game meat. In Chemical Hazards in Foods of Animal Origin; Wageningen Academic Publishers: Wageningen, The Netherlands, 2019; pp. 435–439. [Google Scholar]
- Bekker, J.L.; Hoffman, L.C.; Jooste, P.J. Essential food safety management points in the supply chain of game meat in South Africa. In Game Meat Hygiene in Focus; Springer: Berlin/Heidelberg, Germany, 2011; pp. 39–65. [Google Scholar]
- Cooper, S.M.; Van der Merwe, M. Game ranching for meat production in marginal African agricultural lands. J. Arid. Land Stud. 2014, 24, 249–252. [Google Scholar]
- Nkosi, D.V.; Bekker, J.L.; Gower, L.A.; Van der Watt, M.; Hoffman, L.C. Physical Hazards in Aepyceros melampus Carcasses Killed for Meat Purposes by Aerial and Thoracic Shots. Appl. Sci. 2022, 12, 6861. [Google Scholar] [CrossRef]
- Needham, T.; Hoffman, L. Chapter 8: Species destined for non-traditional meat production: 1. African game species, cervids, ostriches, crocodiles and kangaroos. In Preslaughter Handling and Slaughter of Meat Animals; Wageningen Academic Publishers: Wageningen, The Netherlands, 2022; pp. 1–11. [Google Scholar]
- South Africa.; Standard for the Registration of Game Harvesteres for Harvesting Wild Game Intended for Export of Game Meat. Pretoria Government Gazzett. VPN/08/2010-01. 2010. Available online: https://www.dalrrd.gov.za/ (accessed on 17 April 2022).
- Neethling, J.; Hoffman, L.; Muller, M. Factors influencing the flavour of game meat: A review. Meat Sci. 2016, 113, 139–153. [Google Scholar] [CrossRef]
- Hampton, J.O.; Eccles, G.; Hunt, R.; Bengsen, A.J.; Perry, A.L.; Parker, S.; Miller, C.J.; Joslyn, S.K.; Stokke, S.; Arnemo, J.M.; et al. A comparison of fragmenting lead-based and lead-free bullets for aerial shooting of wild pigs. PLoS ONE 2021, 16, e0247785. [Google Scholar] [CrossRef] [PubMed]
- Fountain, A.J.; Corey, A.; Malko, J.A.; Strozier, D.; Allen, J.W. Imaging appearance of ballistic wounds predicts bullet composition: Implications for MRI safety. Am. J. Roentgenol. 2021, 216, 542–551. [Google Scholar] [CrossRef] [PubMed]
- Hampton, J.O.; Bengsen, A.J.; Pople, A.; Brennan, M.; Leeson, M.; Forsyth, D.M. Animal welfare outcomes of helicopter-based shooting of deer in Australia. Wildl. Res. 2021, 49, 264–273. [Google Scholar] [CrossRef]
- Hösli, R.; König, S.; Mühlebach, S.F. Development and validation of an LC-MS/MS method and comparison with a GC-MS method to measure phenytoin in human brain dialysate, blood, and saliva. J. Anal. Methods Chem. 2018, 2018, 8274131. [Google Scholar] [CrossRef]
- Chahrour, O.; Malone, J.; Collins, M.; Salmon, V.; Greenan, C.; Bombardier, A.; Ma, Z.; Dunwoody, N. Development and validation of an ICP-MS method for the determination of elemental impurities in TP-6076 active pharmaceutical ingredient (API) according to USP<232>/<233>. J. Pharm. Biomed. Anal. 2017, 145, 84–90. [Google Scholar] [CrossRef]
- Abernethy, D.R.; DeStefano, A.J.; Cecil, T.L.; Zaidi, K.; Williams, R.L. Metal impurities in food and drugs. Pharm. Res. 2010, 27, 750–755. [Google Scholar] [CrossRef]
- Mitranescu, E.; Butaru, A.; Tudor, L.; Lataretu, A.; Furnaris, F.; Ghimpeteanu, O.M.; Silviu, M. Researches concerning the occurrence of cadmium and lead residues in game meat. Bull. Univ. Agric. Sci. Vet. Med. Cluj-Napoca Vet. Med. 2011, 68, 222–224. [Google Scholar]
- Szkoda, J.; Durkalec, M.; Kołacz, R.; Opaliński, S.; Żmudzki, J. Content of cadmium, lead and mercury in the tissues of game animals. Med. Weter. 2012, 68, 689–692. [Google Scholar]
- Kim, J.; Lee, Y.; Yang, M. Environmental exposure to lead (Pb) and variations in its susceptibility. J. Environ. Sci. Health Part C 2014, 32, 159–185. [Google Scholar] [CrossRef] [PubMed]
- O’Neill, B.; Bosch, A.C.; Kerwath, S.E.; Sigge, G.O.; Hoffman, L.C. Presence, concentration and trends of metals: A baseline study of blacktail (Diplodus sargus capensis) and hottentot (Pachymetopon blochii) along the South African coastline. Mar. Pollut. Bull. 2017, 121, 352. [Google Scholar] [CrossRef]
- Mehouel, F.; Bouayad, L.; Hammoudi, A.; Ayadi, O.; Regad, F. Evaluation of the heavy metals (mercury, lead, and cadmium) contamination of sardine (Sardina pilchardus) and swordfish (Xiphias gladius) fished in three Algerian coasts. Vet. World 2019, 12, 7–11. [Google Scholar] [CrossRef] [PubMed]
- Trinogga, A.L.; Courtiol, A.; Krone, O. Fragmentation of lead-free and lead-based hunting rifle bullets under real life hunting conditions in Germany. Ambio 2019, 48, 1056–1064. [Google Scholar] [CrossRef]
- European Union (EU). Maximum levels of cadmium in foodstuffs. J. Eur. Union 2014. No 32014R0488. Available online: http://data.europa.eu/eli/reg/2014/488/oj (accessed on 21 April 2022).
- Bukovjan, K.; Wittlingerová, Z.; Kutlvašr, K. Arsenic deposition in tissues of the European hare (Lepus europaeus). Acta Vet. Brno 2016, 85, 215–221. [Google Scholar] [CrossRef][Green Version]
- Benini, J.C. Frangible Metal Bullets, Ammunition and Method of Making such Articles. U.S. Patent No. 6,090,178, 18 July 2000. [Google Scholar]
- Menozzi, A.; Menotta, S.; Fedrizzi, G.; Lenti, A.; Cantoni, A.M.; Di Lecce, R.; Gnudi, G.; Pérez-López, M.; Bertini, S. Lead and copper in hunted wild boars and radiographic evaluation of bullet fragmentation between ammunitions. Food Addit. Contam. Part B 2019, 12, 182–190. [Google Scholar] [CrossRef]
- de Oliveira, D.F.; de Castro, B.S.; do Nascimento Recktenvald, M.C.N.; da Costa Júnior, W.A.; da Silva, F.X.; de Menezes Alves, C.L.; Froehlich, J.D.; Bastos, W.R.; Ott, A.M.T. Mercury in wild animals and fish and health risk for indigenous Amazonians. Food Addit. Contam. Part B 2021, 14, 161–169. [Google Scholar] [CrossRef]
- Pietrzkiewicz, K.; Maliszewski, G.; Bombik, E. Mercury accumulation level in meat and organs of farm and game animals. FPUTS Ser. Agric. Aliment. Piscaria Zootech. 2018, 343, 55–62. [Google Scholar] [CrossRef]
- Jędruch, A.; Falkowska, L.; Saniewska, D.; Durkalec, M.; Nawrocka, A.; Kalisińska, E.; Kowalski, A.; Pacyna, J.M. Status and trends of mercury pollution of the atmosphere and terrestrial ecosystems in Poland. Ambio 2021, 50, 1698–1717. [Google Scholar] [CrossRef]






| Toxic Elements | ||||||
|---|---|---|---|---|---|---|
| Country | Type | As (Semi-Metal) | Pb | Hg | Cd | Source(s) |
| South Africa | Regulation | 1.0 | 0.10 | - | 0.05 | [21] |
| EU countries | Regulation | 0.3 | 0.10 | 1.0 | 0.05 | [22] |
| Republic of China | Regulation | 0.5 | 0.5 | 0.05 | 0.1 | [23] |
| Australia and New Zealand | Regulation | 0.01 | 0.10 | 0.002 | 0.05 | [24] |
| United States of America | Regulation | 0.01 | 0.01 | 0.01 | 0.01 | [25] |
| Countries without specified limits | Codex Alimentarius Standards | 2.1 | 0.10 | - | 0.05 | [26] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nkosi, D.V.; Bekker, J.L.; Hoffman, L.C. Toxic Metals in Meat Contributed by Helicopter and Rifle Thoracic Killing of Game Meat Animals. Appl. Sci. 2022, 12, 8095. https://doi.org/10.3390/app12168095
Nkosi DV, Bekker JL, Hoffman LC. Toxic Metals in Meat Contributed by Helicopter and Rifle Thoracic Killing of Game Meat Animals. Applied Sciences. 2022; 12(16):8095. https://doi.org/10.3390/app12168095
Chicago/Turabian StyleNkosi, Davies Veli, Johan Leon Bekker, and Louwrens Christiaan Hoffman. 2022. "Toxic Metals in Meat Contributed by Helicopter and Rifle Thoracic Killing of Game Meat Animals" Applied Sciences 12, no. 16: 8095. https://doi.org/10.3390/app12168095
APA StyleNkosi, D. V., Bekker, J. L., & Hoffman, L. C. (2022). Toxic Metals in Meat Contributed by Helicopter and Rifle Thoracic Killing of Game Meat Animals. Applied Sciences, 12(16), 8095. https://doi.org/10.3390/app12168095





