Nanometer-Resolution Imaging of Living Cells Using Soft X-ray Contact Microscopy
Abstract
Featured Application
Abstract
1. Introduction
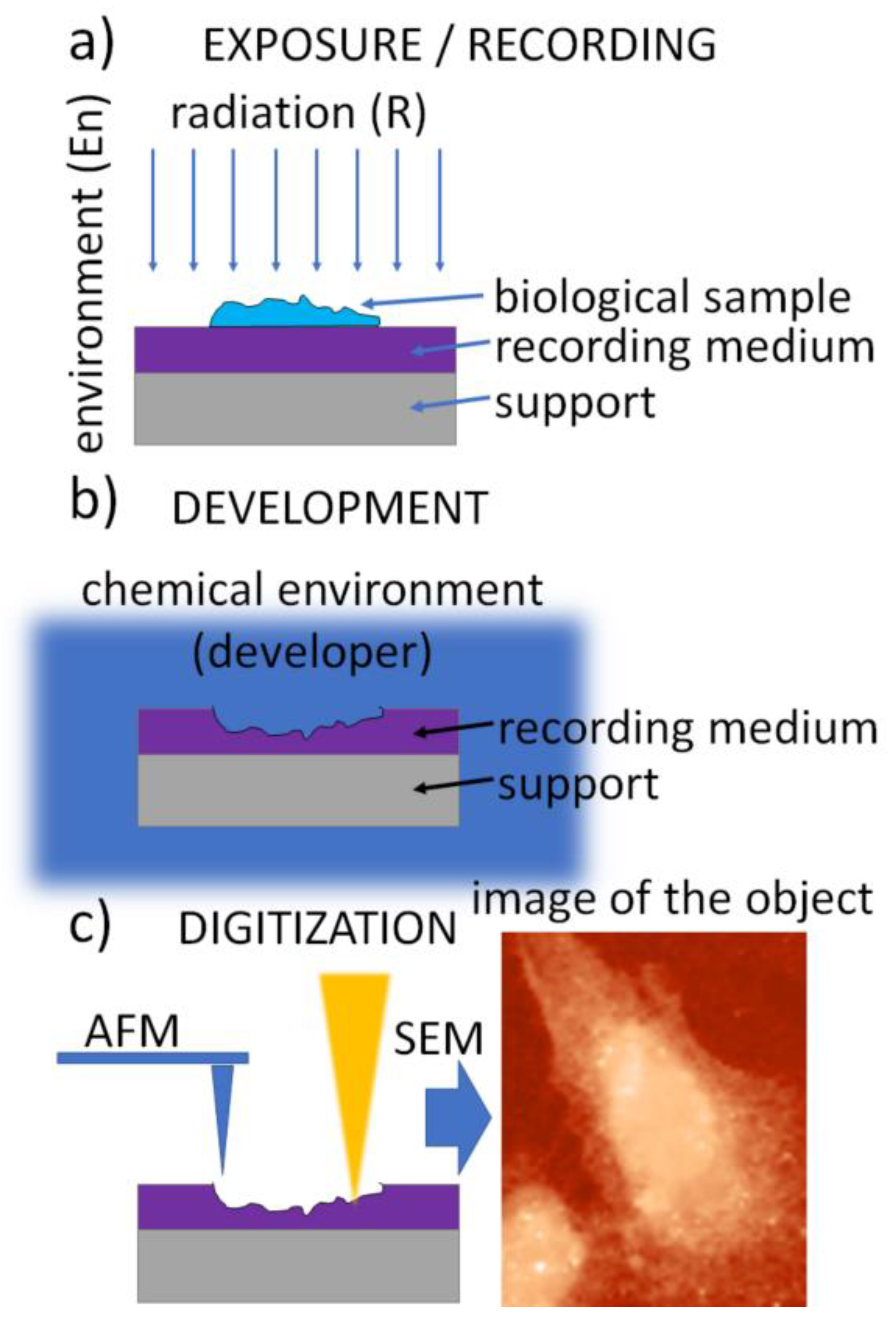
2. Soft X-ray Contact Microscopy
2.1. Radiation Sources
2.2. Recording Media
2.3. Exposure Environment
2.4. Development Procedure
2.5. Digitization
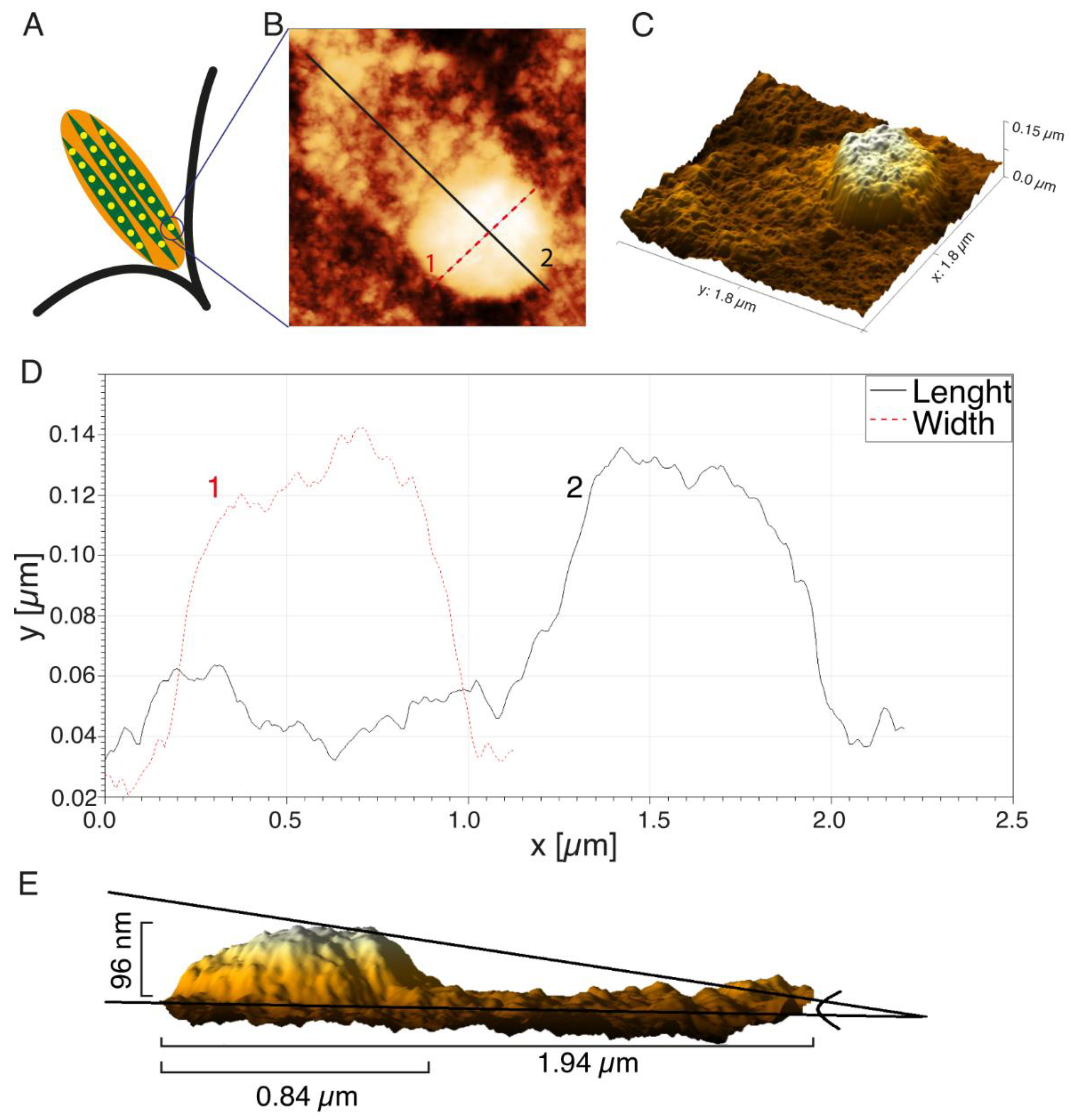
2.6. A Note on Spatial Resolution
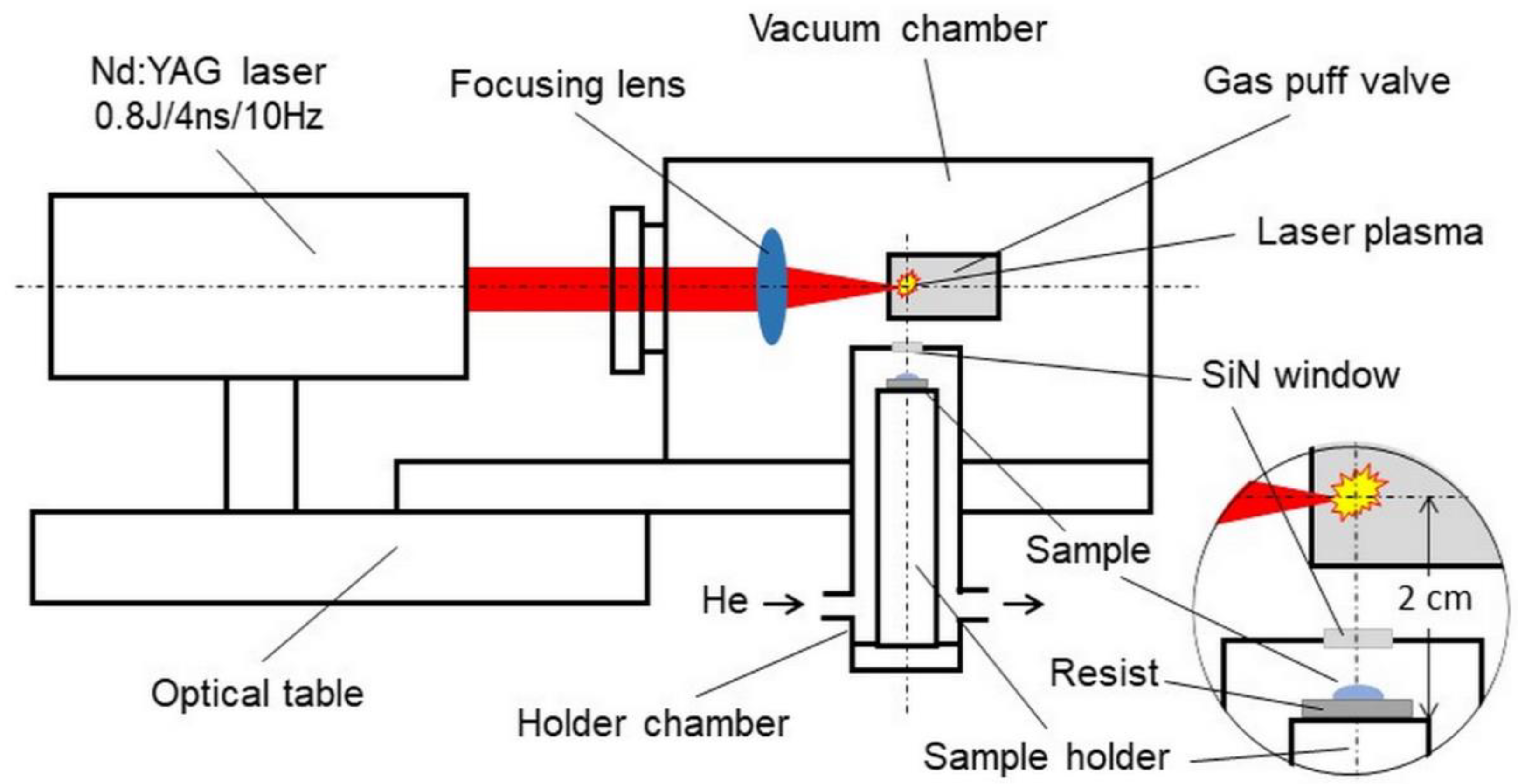
2.7. Desk-Top Laboratory SXCM System
3. Cell Imaging with SXCM
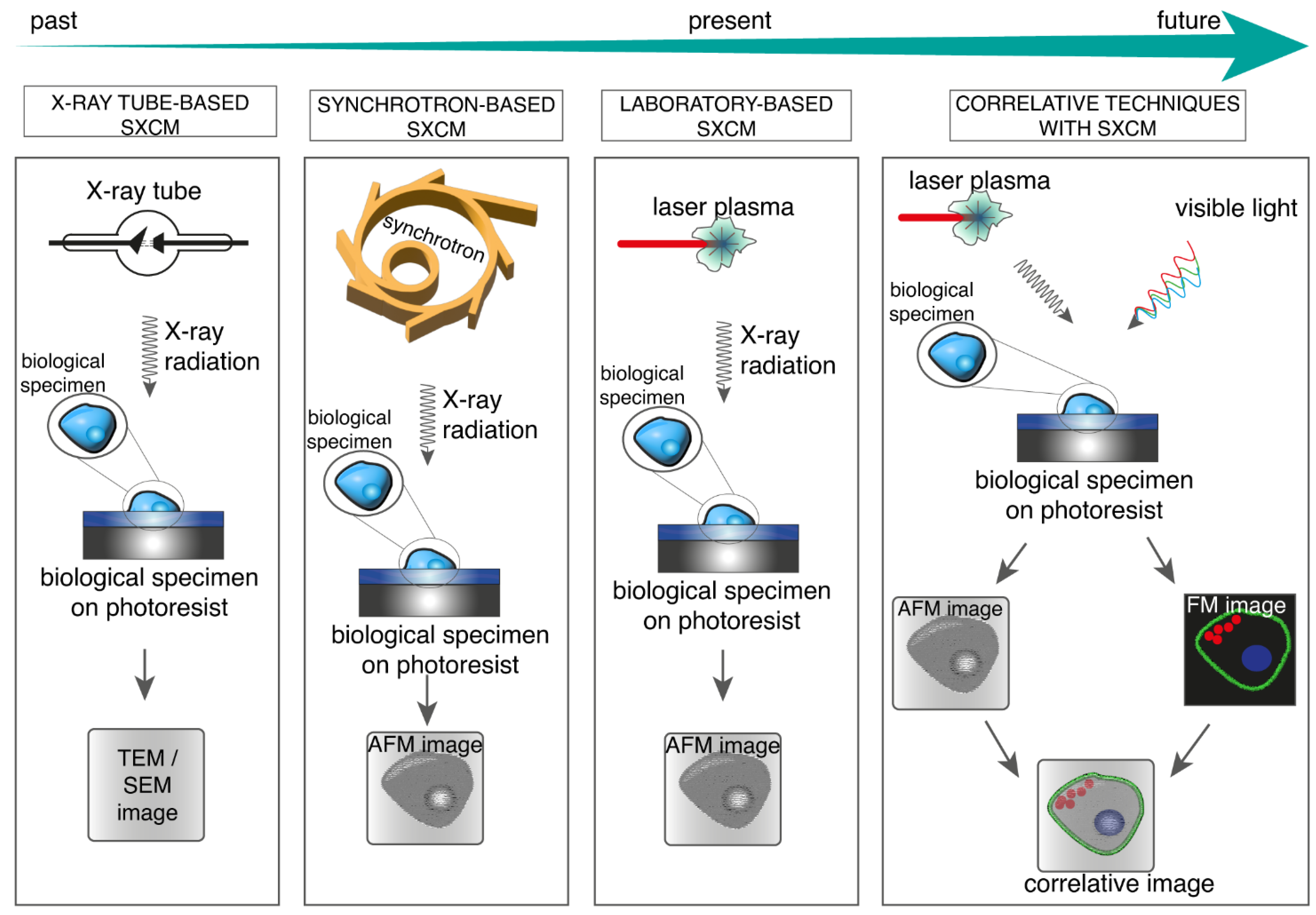
4. SXCM and Correlative Microscopy
5. Conclusions and Future Directions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Lidke, D.S.; Lidke, K.A. Advances in high-resolution imaging–techniques for three-dimensional imaging of cellular structures. J. Cell Sci. 2012, 125, 2571–2580. [Google Scholar] [CrossRef] [PubMed]
- Sousa, A.A.; Kruhlak, M.J. Introduction: Nanoimaging Techniques in Biology. In Nanoimaging: Methods and Protocols; Sousa, A.A., Kruhlak, M.J., Eds.; Humana Press: Totowa, NJ, USA, 2013; pp. 1–10. [Google Scholar]
- Hylton, R.K.; Swulius, M.T. Challenges and triumphs in cryo-electron tomography. iScience 2021, 24, 102959. [Google Scholar] [CrossRef] [PubMed]
- Leis, A.; Rockel, B.; Andrees, L.; Baumeister, W. Visualizing cells at the nanoscale. Trends Biochem. Sci. 2009, 34, 60–70. [Google Scholar] [CrossRef]
- Pascolo, L.; Sena, G.; Gianoncelli, A.; Cernogoraz, A.; Kourousias, G.; Metscher, B.D.; Romano, F.; Zito, G.; Pacile, S.; Barroso, R.; et al. Hard and soft X-ray imaging to resolve human ovarian cortical structures. J. Synchrotron Radiat. 2019, 26, 1322–1329. [Google Scholar] [CrossRef] [PubMed]
- Pedrosa, P.; Baptista, P.V.; Fernandes, A.R.; Guerra, M. Benchtop X-ray fluorescence imaging as a tool to study gold nanoparticle penetration in 3D cancer spheroids. RSC Adv. 2021, 11, 26344–26353. [Google Scholar] [CrossRef]
- Penedo, M.; Miyazawa, K.; Okano, N.; Furusho, H.; Ichikawa, T.; Alam, M.S.; Miyata, K.; Nakamura, C.; Fukuma, T. Visualizing intracellular nanostructures of living cells by nanoendoscopy-AFM. Sci. Adv. 2021, 7, eabj4990. [Google Scholar] [CrossRef] [PubMed]
- Jacobsen, C. Soft X-ray microscopy. Trends Cell Biol. 1999, 9, 44–47. [Google Scholar] [CrossRef]
- Osuchowska, P.N.; Wachulak, P.; Kasprzycka, W.; Nowak-Stępniowska, A.; Wakuła, M.; Bartnik, A.; Fiedorowicz, H.; Trafny, E.A. Adhesion of Triple-Negative Breast Cancer Cells under Fluorescent and Soft X-ray Contact Microscopy. Int. J. Mol. Sci. 2021, 22, 7279. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, Y.; Shinohara, K. Application of X-ray microscopy in analysis of living hydrated cells. Anat. Rec. 2002, 269, 217–223. [Google Scholar] [CrossRef] [PubMed]
- Kirz, J.; Jacobsen, C.; Howells, M. Soft X-ray microscopes and their biological applications. Q. Rev. Biophys. 1995, 28, 33–130. [Google Scholar] [CrossRef]
- Jacobsen, C.; Howells, M.; Warwick, T. Zone-Plate X-ray Microscopy. In Springer Handbook of Microscopy; Hawkes, P.W., Spence, J.C.H., Eds.; Springer International Publishing: Cham, Switzerland, 2019; pp. 1145–1204. [Google Scholar]
- Weiß, D.; Schneider, G.; Niemann, B.; Guttmann, P.; Rudolph, D.; Schmahl, G. Computed tomography of cryogenic biological specimens based on X-ray microscopic images. Ultramicroscopy 2000, 84, 185–197. [Google Scholar] [CrossRef]
- Chao, W.; Fischer, P.; Tyliszczak, T.; Rekawa, S.; Anderson, E.; Naulleau, P. Real space soft X-ray imaging at 10 nm spatial resolution. Opt. Express 2012, 20, 9777–9783. [Google Scholar] [CrossRef] [PubMed]
- Chao, W.; Harteneck, B.D.; Liddle, J.A.; Anderson, E.H.; Attwood, D.T. Soft X-ray microscopy at a spatial resolution better than 15 nm. Nature 2005, 435, 1210–1213. [Google Scholar] [CrossRef]
- Kaulich, B.; Thibault, P.; Gianoncelli, A.; Kiskinova, M. Transmission and emission X-ray microscopy: Operation modes, contrast mechanisms and applications. J. Phys. Condens. Matter 2011, 23, 083002. [Google Scholar] [CrossRef]
- Harkiolaki, M.; Darrow, M.C.; Spink, M.C.; Kosior, E.; Dent, K.; Duke, E. Cryo-soft X-ray tomography: Using soft X-rays to explore the ultrastructure of whole cells. Emerg. Top. Life Sci. 2018, 2, 81–92. [Google Scholar] [CrossRef]
- Larabell, C.A.; Nugent, K.A. Imaging cellular architecture with X-rays. Curr. Opin. Struct. Biol. 2010, 20, 623–631. [Google Scholar] [CrossRef]
- Weinhardt, V.; Chen, J.-H.; Ekman, A.; McDermott, G.; Le Gros, M.A.; Larabell, C. Imaging cell morphology and physiology using X-rays. Biochem. Soc. Trans. 2019, 47, 489–508. [Google Scholar] [CrossRef] [PubMed]
- White, K.L.; Singla, J.; Loconte, V.; Chen, J.-H.; Ekman, A.; Sun, L.; Zhang, X.; Francis, J.P.; Li, A.; Lin, W.; et al. Visualizing subcellular rearrangements in intact β cells using soft X-ray tomography. Sci. Adv. 2020, 6, eabc8262. [Google Scholar] [CrossRef]
- Cosslett, V.E. Microscopy with X-rays. Nature 1959, 183, 1423–1427. [Google Scholar] [CrossRef]
- Wachulak, P.W. Nanometer-Scale and Low-Density Imaging with Extreme Ultraviolet and Soft X-ray Radiation. In 21st Century Nanoscience—A Handbook; CRC Press: Boca Raton, FL, USA, 2020. [Google Scholar]
- Greulich, R.C.; Engström, A. A new approach to high resolution microradiography using extremely soft X-rays. Exp. Cell Res. 1956, 10, 251–254. [Google Scholar] [CrossRef]
- Rommel, M.; Nilsson, B.; Jedrasik, P.; Bonanni, V.; Dmitriev, A.; Weis, J. Sub-10nm resolution after lift-off using HSQ/PMMA double layer resist. Microelectron. Eng. 2013, 110, 123–125. [Google Scholar] [CrossRef]
- Feder, R.; Sayre, D.; Spiller, E.; Topalian, J.; Kirz, J. Specimen replication for electron microscopy using X rays and X-ray resist. J. Appl. Phys. 1976, 47, 1192–1193. [Google Scholar] [CrossRef]
- Stevens, R.M. New carbon nanotube AFM probe technology. Mater. Today 2009, 12, 42–45. [Google Scholar] [CrossRef]
- Spiller, E.; Feder, R.; Topalian, J.; Eastman, D.; Gudat, W.; Sayre, D. X-ray Microscopy of Biological Objects with Carbon Kα and with Synchrotron Radiation. Science 1976, 191, 1172–1174. [Google Scholar] [CrossRef]
- Kado, M.; Kishimoto, M.; Tamotsu, S.; Yasuda, K.; Shinohara, K. In situ observation of cellular organelles with a contact X-ray microscope. J. Phys. Conf. Ser. 2013, 463, 012056. [Google Scholar] [CrossRef]
- Feder, R.; Spiller, E.; Topalian, J.; Broers, A.N.; Gudat, W.; Panessa, B.J.; Zadunaisky, Z.A.; Sedat, J. High-Resolution Soft X-ray Microscopy. Science 1977, 197, 259–260. [Google Scholar] [CrossRef]
- Ford, T.W.; Stead, A.D.; Cotton, R.A. Soft X-ray contact microscopy of biological materials. Electron Microsc. Rev. 1991, 4, 269–292. [Google Scholar] [CrossRef]
- Rosser, R.J.; Feder, R.; Ng, A.; Adams, F.; Celliers, P.; Speer, R.J. Nondestructive single-shot soft X-ray lithography and contact microscopy using a laser-produced plasma source. Appl. Opt. 1987, 26, 4313–4318. [Google Scholar] [CrossRef]
- Kado, M.; Richardson, M.; Gaebel, K.; Torres, D.; Rajyaguru, J.; Muszynski, M. Ultrastructural Imaging and Molecular Modeling of Live Bacteria Using Soft X-ray Contact Microscopy with Nanoseconds Laser-Plasma Radiation; SPIE: Bellingham, WA, USA, 1995; Volume 2523. [Google Scholar]
- Michette, A.G.; Cheng, P.C.; Easons, R.W.; Feder, R.; O’Neill, F.; Owadano, Y.; Rosser, R.J.; Rumsby, P.; Shaw, M.J. Soft X-ray contact microscopy using laser plasma sources. J. Phys. D Appl. Phys. 1986, 19, 363–372. [Google Scholar] [CrossRef]
- Albertano, P.; Reale, L.; Palladino, L.; Reale, A.; Cotton, R.; Bollanti, S.; Di Lazzaro, P.; Flora, F.; Lisi, N.; Nottola, A.; et al. X-ray contact microscopy using an excimer laser plasma source with different target materials and laser pulse durations. J. Microsc. 1997, 187, 96–103. [Google Scholar] [CrossRef]
- Rosser, R.J.; Feder, R.; Ng, A.; Adams, F.; Caldarolo, M.; Celliers, P.; Cheng, P.C.; Da Silva, L.; Parfeniuk, D.; Speer, R.J. Biological specimens imaged by soft X-ray contact microscopy using a plasma source produced with a laboratory sized laser. J. Microsc. 1986, 144, RP5–RP6. [Google Scholar] [CrossRef]
- Kinjo, Y.; Watanabe, M.; Fiedorowicz, H.; Daido, H.; Yanase, E.; Fujii, S.; Sato, E.; Shinohara, K. Fine structures of human chromosomes observed by X-ray contact microscopy coupled with atomic force microscopy. J. Phys. IV Fr. 2003, 104, 313–316. [Google Scholar] [CrossRef]
- Yasin, S.; Hasko, D.G.; Ahmed, H. Comparison of MIBK/IPA and water/IPA as PMMA developers for electron beam nanolithography. Microelectron. Eng. 2002, 61–62, 745–753. [Google Scholar] [CrossRef]
- Cefalas, A.C.; Sarantopoulou, E.; Kollia, Z.; Argitis, P.; Tegou, E.; Ford, T.W.; Stead, A.D.; Danson, C.N.; Neely, D.; Kobe, S. Nanostructured imaging of biological specimens in vivo with laser plasma X-ray contact microscopy. Mater. Sci. Eng. C 2003, 23, 105–108. [Google Scholar] [CrossRef]
- Bonfigli, F.; Faenov, A.Y.; Flora, F.; Marolo, T.; Montereali, R.M.; Nichelatti, E.; Pikuz, T.A.; Reale, L.; Baldacchini, G. Point defects in lithium fluoride films for micro-radiography, X-ray microscopy and photonic applications. Phys. Status Solidi (a) 2005, 202, 250–255. [Google Scholar] [CrossRef]
- Batani, D.; Botto, C.; Moret, M.; Milani, M.; Lucchini, G.; Eidmann, K.; Cotelli, F.; Donin, C.L.L.; Poletti, G.; Ford, T.; et al. The use of high energy laser-plasma sources in soft X-ray contact microscopy of living biological samples. Eur. Phys. J. D 2002, 21, 167–179. [Google Scholar] [CrossRef]
- Majima, T. Soft X-ray imaging of living cells in water: Flash contact soft X-ray microscope. TrAC Trends Anal. Chem. 2004, 23, 520–526. [Google Scholar] [CrossRef]
- Ayele, M.G.; Wachulak, P.W.; Czwartos, J.; Adjei, D.; Bartnik, A.; Wegrzynski, Ł.; Szczurek, M.; Pina, L.; Fiedorowicz, H. Development and characterization of a laser-plasma soft X-ray source for contact microscopy. Nucl. Instrum. Methods Phys. Res. Sect. B: Beam Interact. Mater. At. 2017, 411, 35–43. [Google Scholar] [CrossRef]
- Wang, Y.; Jacobsen. A numerical study of resolution and contrast in soft X-ray contact microscopy. J. Microsc. 1998, 191, 159–169. [Google Scholar] [CrossRef]
- Osuchowska, P.N.; Wachulak, P.; Nowak-Stępniowska, A.; Bartnik, A.; Gnanachandran, K.; Lekka, M.; Czwartos, J.; Fiedorowicz, H.; Trafny, E.A. Imaging of Cell Structures Using Optimized Soft X-ray Contact Microscopy. Appl. Sci. 2020, 10, 6895. [Google Scholar] [CrossRef]
- Kado, M.; Ishino, M.; Tamotsu, S.; Yasuda, K.; Kishimoto, M.; Nishikino, M.; Kinjo, Y.; Shinohara, K. Observation of Organelles in Leydig Cells by Contact Soft X-ray Microscopy with a Laser Plasma X-ray Source. AIP Conf. Proc. 2011, 1365, 391–394. [Google Scholar] [CrossRef]
- Panessa-Warren, B.J. Contact microscopy with synchrotron radiation. Biol Trace Elem Res 1987, 12, 167–183. [Google Scholar] [CrossRef] [PubMed]
- Spiller, E.; Feder, R.; Topallan, J. Soft X-rays for biological and industrial pattern replications. J. Phys. Colloq. 1978, 39, C4–C205. [Google Scholar] [CrossRef]
- Cheng, P.C.; Shinozaki, D.M.; Tan, K.H. Recent Advances in Contact Imaging of Biological Materials. In X-ray Microscopy: Instrumentation and Biological Applications; Cheng, P.-C., Jan, G.-J., Eds.; Springer: Berlin/Heidelberg, Germany, 1987; pp. 65–104. [Google Scholar]
- Feder, R.; Costa Jonathan, L.; Chaudhari, P.; Sayre, D. Improved Detail in Biological Soft X-ray Microscopy: Study of Blood Platelets. Science 1981, 212, 1398–1400. [Google Scholar] [CrossRef] [PubMed]
- Kinjo, Y.; Watanabe, M.; Ito, A.; Shinohara, K. X-ray Microscopy and Chromosome Research. In 8th International Conference on X-ray Microscopy; IPAP Conference Series 7; Institute of Pure and Applied Physics: Tokyo, Japan, 2005; pp. 227–229. [Google Scholar]
- Reale, L.; Bonfigli, F.; Lai, A.; Flora, F.; Albertano, P.; Di Giorgio, M.L.; Mezi, L.; Montereali, R.M.; Faenov, A.; Pikuz, T.; et al. Contact X-ray microscopy of living cells by using LiF crystal as imaging detector. J. Microsc. 2015, 258, 127–139. [Google Scholar] [CrossRef]
- Baldacchini, G.; Bollanti, S.; Bonfigli, F.; Flora, F.; Di Lazzaro, P.; Lai, A.; Marolo, T.; Montereali, R.M.; Murra, D.; Faenov, A.; et al. Soft X-ray submicron imaging detector based on point defects in LiF. Rev. Sci. Instrum. 2005, 76, 113104. [Google Scholar] [CrossRef]
- Hu, W.; Sarveswaran, K.; Lieberman, M.; Bernstein, G.H. Sub-10 nm electron beam lithography using cold development of poly(methylmethacrylate). J. Vac. Sci. Technol. B Microelectron. Nanometer Struct. Processing Meas. Phenom. 2004, 22, 1711–1716. [Google Scholar] [CrossRef]
- Ayele, M.G.; Czwartos, J.; Adjei, D.; Wachulak, P.; Ahad, I.U.; Bartnik, A.; Wegrzynski, Ł.; Szczurek, M.; Jarocki, R.; Fiedorowicz, H.; et al. Contact Microscopy using a Compact Laser Produced Plasma Soft X-ray Source. Acta Phys. Pol. A 2016, 129, 237–240. [Google Scholar] [CrossRef]
- Beese, L.; Feder, R.; Sayre, D. Contact X-ray microscopy. A new technique for imaging cellular fine structure. Biophys. J. 1986, 49, 259–268. [Google Scholar] [CrossRef][Green Version]
- Richardson, M.C.; Shinohara, K.; Tanaka, K.A.; Kinjo, Y.; Ikeda, N.; Kado, M. Pulsed X-ray microscopy of biological specimens with laser plasma sources. In Soft X-ray Microscopy; SPIE: Bellingham, WA, USA, 1993. [Google Scholar]
- Feder, R.; Banton, V.; Sayre, D.; Costa, J.; Baldini, M.; Kim, B. Direct Imaging of Live Human Platelets by Flash X-ray Microscopy. Science 1985, 227, 63–64. [Google Scholar] [CrossRef] [PubMed]
- Kinjo, Y.; Shinohara, K.; Ito, A.; Nakano, H.; Watanabe, M.; Horiike, Y.; Kikuchi, Y.; Richardson, M.C.; Tanaka, K.A. Direct imaging in a water layer of human chromosome fibres composed of nucleosomes and their higher-order structures by laser-plasma X-ray contact microscopy. J. Microsc. 1994, 176, 63–74. [Google Scholar] [CrossRef] [PubMed]
- Kunio, S.; Yasuhito, K.; Martin, C.R.; Atsushi, I.; Noboru, M.; Yasuhiro, H.; Makoto, W.; Keiji, Y.; Kazuo, A.T. Observation of human chromosome fibers in a water layer by laser-plasma X-ray contact microscopy. In Soft X-ray Microscopy; SPIE: Bellingham, WA, USA, 1993. [Google Scholar]
- Kado, M.; Daido, H.; Yamamoto, Y.; Shinohara, K.; Richardson, M.C. Development of a laser plasma X-ray microscope to observe live hydrated biological specimens. Laser Phys. Lett. 2006, 3, 205–207. [Google Scholar] [CrossRef]
- Kado, M.; Richardson, M.; Rajyaguru, J.M.; Muszynski, M.J.; Friedman, H.; Yamamoto, Y. Direct Ultrastructural Imaging of Macrophages Using a Novel X-ray Contact Microscopy. Proc. Soc. Exp. Biol. Med. 1999, 220, 27–30. [Google Scholar] [CrossRef] [PubMed]
- Kishimoto, M.; Kado, M.; Ishino, M.; Tamotsu, S.; Yasuda, K.; Shinohara, K. Development of single shot soft X-ray contact microscopy system for nano-scale dynamics measurement of living biological specimen. AIP Conf. Proc. 2012, 1465, 43–47. [Google Scholar] [CrossRef]
- Rajyaguru, J.M.; Kado, M.; Nekula, K.; Richardson, M.C.; Muszynski, M.J. High resolution X-ray micrography of live Candida albicans using laser plasma pulsed point X-ray sources. Microbiology 1997, 143, 733–738. [Google Scholar] [CrossRef]
- Rajyaguru, J.M.; Torres, D.S.; Abel, E.; Richardson, M.C.; Muszynski, M.J. Application of X-ray micrography and imaging to study the effect of gentamicin on Pseudomonas aeruginosa. J. Antimicrob. Chemother. 1998, 41, 557–561. [Google Scholar] [CrossRef][Green Version]
- Tomie, T.; Shimizu, H.; Majima, T.; Yamada, M.; Kanayama, T.; Kondo, H.; Yano, M.; Ono, M. Three-Dimensional Readout of Flash X-ray Images of Living Sperm in Water by Atomic-Force Microscopy. Science 1991, 252, 691–693. [Google Scholar] [CrossRef]
- Shinohara, K.; Nakano, H.; Kinjo, Y.; Watanabe, M. Fine structure of unstained human chromosome fibres dried with no fixative as observed by X-ray contact microscopy. J. Microsc. 1990, 158, 335–342. [Google Scholar] [CrossRef]
- Wachulak, P.; Torrisi, A.; Ayele, M.; Czwartos, J.; Bartnik, A.; Węgrzyński, Ł.; Fok, T.; Parkman, T.; Salačová, Š.; Turňová, J.; et al. Bioimaging Using Full Field and Contact EUV and SXR Microscopes with Nanometer Spatial Resolution. Appl. Sci. 2017, 7. [Google Scholar] [CrossRef]
- Lo, C.-A.; Greben, A.W.; Chen, B.E. Generating stable cell lines with quantifiable protein production using CRISPR/Cas9-mediated knock-in. BioTechniques 2017, 62, 165–174. [Google Scholar] [CrossRef] [PubMed]
- Sokolovski, M.; Bhattacherjee, A.; Kessler, N.; Levy, Y.; Horovitz, A. Thermodynamic Protein Destabilization by GFP Tagging: A Case of Interdomain Allostery. Biophys. J. 2015, 109, 1157–1162. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Trastoy, M.O.; Defais, M.; Larminat, F. Resistance to the antibiotic Zeocin by stable expression of the Sh ble gene does not fully suppress Zeocin-induced DNA cleavage in human cells. Mutagenesis 2005, 20, 111–114. [Google Scholar] [CrossRef] [PubMed]
- Miranda, A.; Gómez-Varela, A.I.; Stylianou, A.; Hirvonen, L.M.; Sánchez, H.; De Beule, P.A.A. How did correlative atomic force microscopy and super-resolution microscopy evolve in the quest for unravelling enigmas in biology? Nanoscale 2021, 13, 2082–2099. [Google Scholar] [CrossRef]
- Navikas, V.; Leitao, S.M.; Grussmayer, K.S.; Descloux, A.; Drake, B.; Yserentant, K.; Werther, P.; Herten, D.-P.; Wombacher, R.; Radenovic, A.; et al. Correlative 3D microscopy of single cells using super-resolution and scanning ion-conductance microscopy. Nat. Commun. 2021, 12, 4565. [Google Scholar] [CrossRef]
- Duke, E.M.H.; Razi, M.; Weston, A.; Guttmann, P.; Werner, S.; Henzler, K.; Schneider, G.; Tooze, S.A.; Collinson, L.M. Imaging endosomes and autophagosomes in whole mammalian cells using correlative cryo-fluorescence and cryo-soft X-ray microscopy (cryo-CLXM). Ultramicroscopy 2014, 143, 77–87. [Google Scholar] [CrossRef]
- Hagen, C.; Guttmann, P.; Klupp, B.; Werner, S.; Rehbein, S.; Mettenleiter, T.C.; Schneider, G.; Grünewald, K. Correlative VIS-fluorescence and soft X-ray cryo-microscopy/tomography of adherent cells. J. Struct. Biol. 2012, 177, 193–201. [Google Scholar] [CrossRef]
- Kado, M.; Kishimoto, M.; Tamotsu, S.; Yasuda, K.; Aoyama, M.; Tone, S.; Shinohara, K. Correlative imaging of live biological cells with a soft X-ray microscope and a fluorescence microscope. AIP Conf. Proc. 2016, 1696, 020019. [Google Scholar] [CrossRef]
- Kounatidis, I.; Stanifer, M.L.; Phillips, M.A.; Paul-Gilloteaux, P.; Heiligenstein, X.; Wang, H.; Okolo, C.A.; Fish, T.M.; Spink, M.C.; Stuart, D.I.; et al. 3D Correlative Cryo-Structured Illumination Fluorescence and Soft X-ray Microscopy Elucidates Reovirus Intracellular Release Pathway. Cell 2020, 182, 515–530.e17. [Google Scholar] [CrossRef]
- Okolo, C.A.; Kounatidis, I.; Groen, J.; Nahas, K.L.; Balint, S.; Fish, T.M.; Koronfel, M.A.; Cortajarena, A.L.; Dobbie, I.M.; Pereiro, E.; et al. Sample preparation strategies for efficient correlation of 3D SIM and soft X-ray tomography data at cryogenic temperatures. Nat. Protoc. 2021, 16, 2851–2885. [Google Scholar] [CrossRef]
- Deng, J.; Vine, D.J.; Chen, S.; Jin, Q.; Nashed, Y.S.G.; Peterka, T.; Vogt, S.; Jacobsen, C. X-ray ptychographic and fluorescence microscopy of frozen-hydrated cells using continuous scanning. Sci. Rep. 2017, 7, 445. [Google Scholar] [CrossRef] [PubMed]
- Bernhardt, M.; Nicolas, J.D.; Osterhoff, M.; Mittelstädt, H.; Reuss, M.; Harke, B.; Wittmeier, A.; Sprung, M.; Köster, S.; Salditt, T. Correlative microscopy approach for biology using X-ray holography, X-ray scanning diffraction and STED microscopy. Nat. Commun. 2018, 9, 3641. [Google Scholar] [CrossRef] [PubMed]
- Kourousias, G.; Billè, F.; Borghes, R.; Pascolo, L.; Gianoncelli, A. Megapixel scanning transmission soft X-ray microscopy imaging coupled with compressive sensing X-ray fluorescence for fast investigation of large biological tissues. Analyst 2021, 146, 5836–5842. [Google Scholar] [CrossRef] [PubMed]
- Nicolas, J.-D.; Bernhardt, M.; Krenkel, M.; Richter, C.; Luther, S.; Salditt, T. Combined scanning X-ray diffraction and holographic imaging of cardiomyocytes. J. Appl. Crystallogr. 2017, 50, 612–620. [Google Scholar] [CrossRef]

| Year | X-ray Source | Sample | Photoresist | Imaging | Object of Interest | Spatial Resolution (nm) | References |
|---|---|---|---|---|---|---|---|
| 1981 | X-ray tube | Dry/fixed | PMMA | TEM | Human blood platelets | 5 | (Feder et al., 1981) [49] |
| 1985 | Discharge plasma | Hydrated | PMMA | TEM | Human blood platelets | NS * | |
| 1990 | Synchrotron | Dry/fixed, isolated | PMMA | TEM | Chromosomes from human lymphocytes | 10 | (Feder et al., 1985) [57] |
| 1991 | Laser plasma | Hydrated | PMMA | AFM | Sea urchin sperm | 100 | |
| 1992 | Laser plasma | Hydrated, isolated | PMMA | TEM | Chromosomes from human lymphocytes | 10 | (Shinohara et al., 1990) [66] |
| 1994 | Laser plasma | Hydrated, isolated | PMMA | TEM | Chromosomes from human lymphocytes | 10–100 | |
| 1997 | Synchrotron | Hydrated | PMMA | AFM | Candida albicans | 20–90 | (Tomie et al., 1991) [65] |
| 1998 | Laser plasma | Hydrated | PMMA | AFM | Pseudomonas aeruginosa | 90 | |
| 1999 | Laser plasma | Hydrated | PMMA | AFM | Macrophages | - | (Shinohara et al., 1993) [59] |
| 2003 | Laser plasma | Hydrated | EPR | SEM, AFM | Flagella from green algae | 100 | |
| 2003 | Laser plasma | Hydrated, isolated | PMMA | AFM | Chromosomes from Vicia faba L. | 10 | (Kinjo et al., 1994) [58] |
| 2005 | Synchrotron | Hydrated | PMMA | AFM | Macrophages | 10 | |
| 2011 | Laser plasma | Hydrated | PMMA | AFM | Leydig cells | 90 | (Rajyaguru et al., 1997) [63] |
| 2012 | Laser plasma | Hydrated | PMMA | AFM | Leydig cells | 100 | |
| 2015 | Laser plasma | Hydrated | PMMA | AFM | Macrophages, Leptolyngbya sp., Chlamydomonas reinhardtii, Chlamydomonas dysosmos | 100 | (Rajyaguru et al., 1998) [64] |
| 2016 | Laser plasma | Dry/fixed | PMMA | AFM | Non-malignant human bladder cells HCV29 | 80 | |
| 2017 | Laser plasma | Dry/fixed | PMMA | AFM | Human epidermal keratinocytes HEK | 80 | (Kado et al., 1999) [61] |
| 2017 | Laser plasma | Dry/fixed | PMMA | AFM | Murine colorectal carcinoma CT26, human urinary bladder carcinoma T24 | 80 | |
| 2020 | Laser plasma | Dry/fixed | PMMA | AFM | Human Poietics™ mesenchymal stem cells, breast cancer cells HCC38, prostate cancer cells DU145 | 80 | (Cefalas et al., 2003) [38] |
| 2021 | Laser plasma | Hydrated | PMMA | AFM | Human breast cancer cells HCC38 | 80 | |
| 2015 | Laser plasma | Hydrated | PMMA | AFM | Macrophages, Leptolyngbya sp., Chlamydomonas reinhardtii, Chlamydomonas dysosmos | 100 | (Kinjo et al., 2003) [36] |
| 2016 | Laser plasma | Dry/fixed | PMMA | AFM | Non-malignant human bladder cells HCV29 | 80 | |
| 2017 | Laser plasma | Dry/fixed | PMMA | AFM | Human epidermal keratinocytes HEK | 80 | (Kado et al., 2006) [60] |
| 2020 | Laser plasma | Dry/fixed | PMMA | AFM | Human Poietics™ mesenchymal stem cells, breast cancer cells HCC38, prostate cancer cells DU145 | 80 | |
| 2021 | Laser plasma | Hydrated | PMMA | AFM | Human breast cancer cells HCC38 | 80 | (Kado et al., 2011) [45] |
| Techniques | X-ray Source | Sample | Object of Interest | Spatial Resolution (nm) | References | |
|---|---|---|---|---|---|---|
| X-ray Technique | Correlated Method | |||||
| Cryo-SXM/SXT | Cryo-FL microscopy | Synchrotron | Cryo-preserved; fluorescently tagged | Porcine epithelial-like embryonic EFN-R kidney cells stably co-expressing pseudorabies viruses pUL31 and pUL34 | NS * | (Hagen et al., 2012) [74] |
| Cryo-SXM/SXT | Cryo-FL microscopy | Synchrotron | Cryo-preserved; fluorescently tagged | Human epidermal keratinocytes HEK293 | 40 | (Duke et al., 2014) [73] |
| LPSXM | FL microscopy | Laser plasma | Hydrated; fluorescently tagged | Leydig cells | 90 | (Kado et al., 2016) [75] |
| X-ray ptychography | XRF | Synchrotron | Cryo-preserved | Chlamydomonas reinhardtii | 85 | (Deng et al., 2017) [78] |
| X-ray holography, X-ray scanning diffraction | STED | Synchrotron | Dry/fixed; cryo-preserved; fluorescently tagged | Neonatal rat cardiomyocytes | nm | (Bernhardt et al., 2018) [79] |
| Cryo-SXT | Cryo-SIM | Synchrotron | Cryo-preserved; fluorescently tagged | Human bone osteosarcoma cells U2OS | 60–200 | (Kounatidis et al., 2020) [76] |
| STXM | XRF | Synchrotron | Paraffin-embedded | Human lung and ovarian tissues | 1000 | (Kourousias et al., 2021) [80] |
| Cryo-SXT | Cryo-SIM | Synchrotron | Cryo-preserved; fluorescently tagged | Mouse NIH3T3 embryo fibroblast cells, human bone osteosarcoma cells U2OS, lysosomes in Drosophila melanogaster plasmatocytes | 25–50 | (Okolo et al., 2021) [77] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nowak-Stępniowska, A.; Kasprzycka, W.; Osuchowska, P.N.; Trafny, E.A.; Bartnik, A.; Fiedorowicz, H.; Wachulak, P. Nanometer-Resolution Imaging of Living Cells Using Soft X-ray Contact Microscopy. Appl. Sci. 2022, 12, 7030. https://doi.org/10.3390/app12147030
Nowak-Stępniowska A, Kasprzycka W, Osuchowska PN, Trafny EA, Bartnik A, Fiedorowicz H, Wachulak P. Nanometer-Resolution Imaging of Living Cells Using Soft X-ray Contact Microscopy. Applied Sciences. 2022; 12(14):7030. https://doi.org/10.3390/app12147030
Chicago/Turabian StyleNowak-Stępniowska, Agata, Wiktoria Kasprzycka, Paulina Natalia Osuchowska, Elżbieta Anna Trafny, Andrzej Bartnik, Henryk Fiedorowicz, and Przemysław Wachulak. 2022. "Nanometer-Resolution Imaging of Living Cells Using Soft X-ray Contact Microscopy" Applied Sciences 12, no. 14: 7030. https://doi.org/10.3390/app12147030
APA StyleNowak-Stępniowska, A., Kasprzycka, W., Osuchowska, P. N., Trafny, E. A., Bartnik, A., Fiedorowicz, H., & Wachulak, P. (2022). Nanometer-Resolution Imaging of Living Cells Using Soft X-ray Contact Microscopy. Applied Sciences, 12(14), 7030. https://doi.org/10.3390/app12147030







