Flavour Generation during Lactic Acid Fermentation of Brassica Vegetables—Literature Review
Abstract
1. Introduction
2. The Importance of Flavour
3. Strategies Used for Aroma Analysis
4. Lactic Acid Fermentation—State-of-Art
5. Flavour of Brassica Vegetables
6. Changes in Flavour during the Fermentation of Brassica Vegetables
6.1. Carbohydrate Metabolism
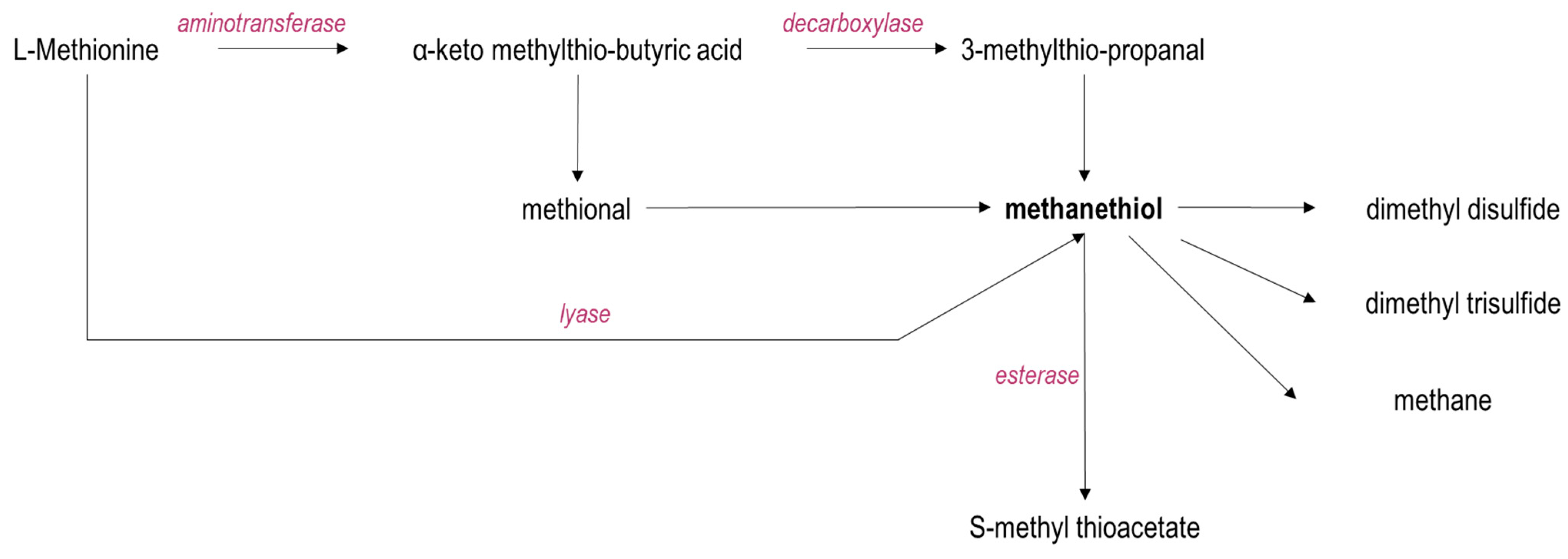
6.2. Amino Acid Metabolism
6.3. Lipid Metabolism
6.4. Glucosinolate Metabolism
7. Brassica Fermented Products
7.1. Sauerkraut
7.2. Kimchi
8. Summary and Future Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Clark, J.E. Taste and Flavour: Their Importance in Food Choice and Acceptance. Proc. Nutr. Soc. 1998, 57, 639–643. [Google Scholar] [CrossRef]
- You Searched for Flavor—Food Insight. Available online: https://foodinsight.org/?s=flavor (accessed on 14 May 2022).
- Reineccius, G. Flavor Chemistry and Technology; CRC Press: Boca Raton, FL, USA, 2005. [Google Scholar] [CrossRef]
- Chandrashekar, J.; Kuhn, C.; Oka, Y.; Yarmolinsky, D.A.; Hummler, E.; Ryba, N.J.P.; Zuker, C.S. The Cells and Peripheral Representation of Sodium Taste in Mice. Nature 2010, 464, 297–301. [Google Scholar] [CrossRef]
- Li, X.; Staszewski, L.; Xu, H.; Durick, K.; Zoller, M.; Adler, E. Human Receptors for Sweet and Umami Taste. Proc. Natl. Acad. Sci. USA 2002, 99, 4692–4696. [Google Scholar] [CrossRef]
- Beckett, E.L.; Martin, C.; Yates, Z.; Veysey, M.; Duesing, K.; Lucock, M. Bitter Taste Genetics-the Relationship to Tasting, Liking, Consumption and Health. Food Funct. 2014, 5, 3040–3054. [Google Scholar] [CrossRef]
- Allen, A.L.; McGeary, J.E.; Knopik, V.S.; Hayes, J.E. Bitterness of the Non-Nutritive Sweetener Acesulfame Potassium Varies with Polymorphisms in TAS2R9 and TAS2R31. Chem. Senses 2013, 38, 379–389. [Google Scholar] [CrossRef]
- Negoias, S.; Visschers, R.; Boelrijk, A.; Hummel, T. New Ways to Understand Aroma Perception. Food Chem. 2008, 108, 1247–1254. [Google Scholar] [CrossRef]
- Hummel, T.; Livermore, A. Intranasal Chemosensory Function of the Trigeminal Nerve and Aspects of Its Relation to Olfaction. Int. Arch. Occup. Environ. Health 2002, 75, 305–313. [Google Scholar] [CrossRef]
- Jelen, H. Food Flavors: Chemical, Sensory and Technological Properties; CRC Press: Boca Raton, FL, USA, 2012; ISBN 9781138034976. [Google Scholar]
- Buttery, R.G. Flavor chemistry and odor thresholds. In Flavor Chemistry; Springer: Boston, MA, USA, 1999; pp. 353–365. [Google Scholar] [CrossRef]
- Frank, O.; Ottinger, H.; Hofmann, T. Characterization of an Intense Bitter-Tasting 1H,4H-Quinolizinium-7-Olate by Application of the Taste Dilution Analysis, a Novel Bioassay for the Screening and Identification of Taste-Active Compounds in Foods. J Agric. Food Chem. 2001, 49, 231–238. [Google Scholar] [CrossRef]
- Yahia, E.M.; Gardea-Béjar, A.; de JesúsOrnelas-Paz, J.; Maya-Meraz, I.O.; Rodríguez-Roque, M.J.; Rios-Velasco, C.; Ornelas-Paz, J.; Salas-Marina, M.A. Chapter 4—Preharvest Factors Affecting Postharvest Quality. In Postharvest Technology of Perishable Horticultural Commodities; Yahia, E.M., Ed.; Woodhead Publishing: Sawston, UK, 2019; pp. 99–128. ISBN 978-0-12-813276-0. [Google Scholar]
- Wieczorek, M.N.; Walczak, M.; Skrzypczak-Zielińska, M.; Jeleń, H.H. Bitter Taste of Brassica Vegetables: The Role of Genetic Factors, Receptors, Isothiocyanates, Glucosinolates, and Flavor Context. Crit. Rev. Food Sci. Nutr. 2018, 58, 3130–3140. [Google Scholar] [CrossRef]
- Granvogl, M.; Schieberle, P. Chapter Two—The Sensomics Approach: A Useful Tool to Unravel the Genuine Aroma Blueprint of Foods and Aroma Changes during Food Processing. In Comprehensive Analytical Chemistry; Cordero, C.E.I., Ed.; Elsevier: Amsterdam, The Netherlands, 2022; Volume 96, pp. 41–68. ISBN 0166-526X. [Google Scholar]
- Engel, W.; Schieberle, P.; Bahr, W.; Bahr, G.; Engel, W.; Bahr, W.; Schieberle, P. Solvent Assisted Flavour Evaporation—A New and Versatile Technique for the Careful and Direct Isolation of Aroma Compounds from Complex Food Matrices; Springer: Berlin/Heidelberg, Germany, 1999; Volume 209. [Google Scholar]
- Zhu, W.; Cadwallader, K.R. Streamlined Approach for Careful and Exhaustive Aroma Characterization of Aged Distilled Liquors. Food Chem. X 2019, 3, 100038. [Google Scholar] [CrossRef]
- Guth, H.; Grosch, W. Evaluation of important odorants in foods by dilution techniques. In Flavor Chemistry; Springer: Boston, MA, USA, 1999; pp. 377–386. [Google Scholar] [CrossRef]
- Schieberle, P. New Developments in Methods for Analysis of Volatile Flavor Compounds and Their Precursors. Charact. Food 1995, 403–431. [Google Scholar] [CrossRef]
- Greger, V.; Schieberle, P. Characterization of the Key Aroma Compounds in Apricots (Prunus Armeniaca) by Application of the Molecular Sensory Science Concept. J. Agric. Food Chem. 2007, 55, 5221–5228. [Google Scholar] [CrossRef]
- Di Cagno, R.; Coda, R.; de Angelis, M.; Gobbetti, M. Exploitation of Vegetables and Fruits through Lactic Acid Fermentation. Food Microbiol. 2013, 33, 1–10. [Google Scholar] [CrossRef]
- Ogrodowczyk, A.M.; Drabińska, N. Crossroad of Tradition and Innovation—The Application of Lactic Acid Fermentation to Increase the Nutritional and Health-Promoting Potential of Plant-Based Food Products—A Review. Pol. J. Food Nutr. Sci. 2021, 71, 107–134. [Google Scholar] [CrossRef]
- Ruiz Rodríguez, L.G.; Zamora Gasga, V.M.; Pescuma, M.; van Nieuwenhove, C.; Mozzi, F.; Sánchez Burgos, J.A. Fruits and Fruit By-Products as Sources of Bioactive Compounds. Benefits and Trends of Lactic Acid Fermentation in the Development of Novel Fruit-Based Functional Beverages. Food Res. Int. 2021, 140, 109854. [Google Scholar] [CrossRef] [PubMed]
- Smid, E.J.; Kleerebezem, M. Production of Aroma Compounds in Lactic Fermentations. Annu. Rev. Food Sci. Technol. 2014, 5, 313–326. [Google Scholar] [CrossRef]
- Mozzi, F.; Raya, R.R.; Vignolo, G.M. (Eds.) Biotechnology of Lactic Acid Bacteria: Novel Applications, 2nd ed.; Edited Production of Flavor Compounds by Lactic Acid Bacteria in Fermented Foods; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2016. [Google Scholar]
- Blajman, J.E.; Vinderola, G.; Páez, R.B.; Signorini, M.L. The Role of Homofermentative and Heterofermentative Lactic Acid Bacteria for Alfalfa Silage: A Meta-Analysis. J. Agric. Sci. 2020, 158, 107–118. [Google Scholar] [CrossRef]
- Moon, S.H.; Kim, C.R.; Chang, H.C. Heterofermentative Lactic Acid Bacteria as a Starter Culture to Control Kimchi Fermentation. LWT 2018, 88, 181–188. [Google Scholar] [CrossRef]
- Zaunmüller, T.; Eichert, M.; Richter, H.; Unden, G. Variations in the Energy Metabolism of Biotechnologically Relevant Heterofermentative Lactic Acid Bacteria during Growth on Sugars and Organic Acids. Appl. Microbiol. Biotechnol. 2006, 72, 421–429. [Google Scholar] [CrossRef]
- Zheng, J.; Wittouck, S.; Salvetti, E.; Franz, C.M.A.P.; Harris, H.M.B.; Mattarelli, P.; O’Toole, P.W.; Pot, B.; Vandamme, P.; Walter, J.; et al. A Taxonomic Note on the Genus Lactobacillus: Description of 23 Novel Genera, Emended Description of the Genus Lactobacillus Beijerinck 1901, and Union of Lactobacillaceae and Leuconostocaceae. Int. J. Syst. Evol. Microbiol. 2020, 70, 2782–2858. [Google Scholar] [CrossRef]
- Hu, W.; Yang, X.; Ji, Y.; Guan, Y. Effect of Starter Cultures Mixed with Different Autochthonous Lactic Acid Bacteria on Microbial, Metabolome and Sensory Properties of Chinese Northeast Sauerkraut. Food Res. Int. 2021, 148, 110605. [Google Scholar] [CrossRef] [PubMed]
- Bell, L.; Oloyede, O.O.; Lignou, S.; Wagstaff, C.; Methven, L. Taste and Flavor Perceptions of Glucosinolates, Isothiocyanates, and Related Compounds. Mol. Nutr. Food Res. 2018, 62, 1700990. [Google Scholar] [CrossRef]
- Ciska, E.; Horbowicz, M.; Rogowska, M.; Kosson, R.; Drabińska, N.; Honke, J. Evaluation of Seasonal Varaitions in the Glucosinolate Content in Leaves and Roots of Four. Pol. J. Food Nutr. Sci. 2017, 67, 301–308. [Google Scholar] [CrossRef][Green Version]
- Wieczorek, M.N.; Dunkel, A.; Szwengiel, A.; Czaczyk, K.; Drożdżyńska, A.; Zawirska-Wojtasiak, R.; Jeleń, H.H. The Relation between Phytochemical Composition and Sensory Traits of Selected Brassica Vegetables. LWT 2022, 156, 113028. [Google Scholar] [CrossRef]
- Bell, L.; Methven, L.; Signore, A.; Oruna-Concha, M.J.; Wagstaff, C. Analysis of Seven Salad Rocket (Eruca Sativa) Accessions: The Relationships between Sensory Attributes and Volatile and Non-Volatile Compounds. Food Chem. 2017, 218, 181–191. [Google Scholar] [CrossRef]
- Fenwick, G.R.; Griffiths, N.M.; Heaney, R.K. Bitterness in Brussels Sprouts (Brassica oleracea L. var. gemmifera): The Role of Glucosinolates and Their Breakdown Products. J. Sci. Food Agric. 1983, 34, 73–80. [Google Scholar] [CrossRef]
- Pasini, F.; Verardo, V.; Cerretani, L.; Caboni, M.F.; D’Antuono, L.F. Rocket salad (Diplotaxis and Eruca spp.) sensory analysis and relation with glucosinolate and phenolic content. J. Sci. Food Agric. 2011, 91, 2858–2864. [Google Scholar] [CrossRef]
- Del Carmen Martinez-Ballesta, C.; Carvajal, M. Myrosinase in Brassicaceae: The Most Important Issue for Glucosinolate Turnover and Food Quality. Phytochem. Rev. 2015, 14, 1045–1051. [Google Scholar] [CrossRef]
- Rask, L.; Andréasson, E.; Ekbom, B.; Eriksson, S.; Pontoppidan, B.; Meijer, J. Myrosinase: Gene Family Evolution and Herbivore Defense in Brassicaceae. Plant Mol. Biol. 2000, 42, 93–113. [Google Scholar] [CrossRef]
- Kissen, R.; Rossiter, J.T.; Bones, A.M. The “Mustard Oil Bomb”: Not so Easy to Assemble?! Localization, Expression and Distribution of the Components of the Myrosinase Enzyme System. Phytochem. Rev. 2009, 8, 69–86. [Google Scholar] [CrossRef]
- Andréasson, E.; Jørgensen, L.B.; Höglund, A.S.; Rask, L.; Meijer, J. Different Myrosinase and Idioblast Distribution in Arabidopsis and Brassica Napus. Plant Physiol. 2001, 127, 1750–1763. [Google Scholar] [CrossRef]
- Marcinkowska, M.; Jeleń, H.H. Determination of the Odor Threshold Concentrations and Partition Coefficients of Isothiocyanates from Brassica Vegetables in Aqueous Solution. LWT 2020, 131, 109793. [Google Scholar] [CrossRef]
- Ortner, E.; Granvogl, M. Thermally Induced Generation of Desirable Aroma-Active Compounds from the Glucosinolate Sinigrin. J. Agric. Food Chem. 2018, 66, 2485–2490. [Google Scholar] [CrossRef] [PubMed]
- Hong, F.; Freeman, M.L.; Liebler, D.C. Identification of Sensor Cysteines in Human Keap1 Modified by the Cancer Chemopreventive Agent Sulforaphane. Chem. Res. Toxicol. 2005, 18, 1917–1926. [Google Scholar] [CrossRef]
- Hinman, A.; Chuang, H.H.; Bautista, D.M.; Julius, D. TRP Channel Activation by Reversible Covalent Modification. Proc. Natl. Acad. Sci. USA 2006, 103, 19564–19568. [Google Scholar] [CrossRef] [PubMed]
- Marcinkowska, M.; Frank, S.; Steinhaus, M.; Jeleń, H.H. Key Odorants of Raw and Cooked Green Kohlrabi (Brassica oleracea var. gongylodes L.). J. Agric. Food Chem. 2021, 69, 12270–12277. [Google Scholar] [CrossRef] [PubMed]
- Wieczorek, M.N.; Majcher, M.A.; Jeleń, H.H. Identification of Aroma Compounds in Raw and Cooked Broccoli. Flavour Fragr. J. 2021, 36, 576–583. [Google Scholar] [CrossRef]
- Cartea, M.E.; Francisco, M.; Soengas, P.; Velasco, P. Molecules Phenolic Compounds in Brassica Vegetables. Molecules 2011, 16, 251–280. [Google Scholar] [CrossRef]
- Zabaras, D.; Roohani, M.; Krishnamurthy, R.; Cochet, M.; Delahunty, C.M. Characterisation of Taste-Active Extracts from Raw Brassica Oleracea Vegetables. Food Funct 2013, 4, 592–601. [Google Scholar] [CrossRef]
- Park, S.-Y.; Jang, H.-L.; Lee, J.-H.; Choi, Y.; Kim, H.; Hwang, J.; Seo, D.; Kim, S.; Nam, J.-S. Changes in the Phenolic Compounds and Antioxidant Activities of Mustard Leaf (Brassica juncea) Kimchi Extracts during Different Fermentation Periods. Food Sci. Biotechnol. 2017, 26, 105–112. [Google Scholar] [CrossRef]
- Liu, M.; Bienfait, B.; Sacher, O.; Gasteiger, J.; Siezen, R.J.; Nauta, A.; Geurts, J.M.W. Combining Chemoinformatics with Bioinformatics: In Silico Prediction of Bacterial Flavor-Forming Pathways by a Chemical Systems Biology Approach “Reverse Pathway Engineering”. PLoS ONE 2014, 9, e84769. [Google Scholar] [CrossRef] [PubMed]
- De Palencia, P.F.; Plaza, M.; Amárita, F.; Requena, T.; Peláez, C. Diversity of Amino Acid Converting Enzymes in Wild Lactic Acid Bacteria. Enzym. Microb. Technol. 2006, 38, 88–93. [Google Scholar] [CrossRef]
- Liu, S.-Q. Practical Implications of Lactate and Pyruvate Metabolism by Lactic Acid Bacteria in Food and Beverage Fermentations. Int. J. Food Microbiol. 2003, 83, 115–131. [Google Scholar] [CrossRef]
- Choi, Y.J.; Yong, S.; Lee, M.J.; Park, S.J.; Yun, Y.R.; Park, S.H.; Lee, M.A. Changes in Volatile and Non-Volatile Compounds of Model Kimchi through Fermentation by Lactic Acid Bacteria. LWT 2019, 105, 118–126. [Google Scholar] [CrossRef]
- Yang, X.; Hu, W.; Xiu, Z.; Jiang, A.; Yang, X.; Sarengaowa; Ji, Y.; Guan, Y.; Feng, K. Comparison of Northeast Sauerkraut Fermentation between Single Lactic Acid Bacteria Strains and Traditional Fermentation. Food Res. Int. 2020, 137, 109553. [Google Scholar] [CrossRef]
- Wang, Y.; Wu, J.; Lv, M.; Shao, Z.; Hungwe, M.; Wang, J.; Bai, X.; Xie, J.; Wang, Y.; Geng, W. Metabolism Characteristics of Lactic Acid Bacteria and the Expanding Applications in Food Industry. Front. Bioeng. Biotechnol. 2021, 9, 612285. [Google Scholar] [CrossRef]
- Bekal, S.; van Beeumen, J.; Samyn, B.; Garmyn, D.; Henini, S.; Diviès, C.; Prévost, H. Purification of Leuconostoc Mesenteroides Citrate Lyase and Cloning and Characterization of the CitCDEFG Gene Cluster. J. Bacteriol. 1998, 180, 647–654. [Google Scholar] [CrossRef]
- Pripis-Nicolau, L.; de Revel, G.; Bertrand, A.; Maujean, A. Formation of Flavor Components by the Reaction of Amino Acid and Carbonyl Compounds in Mild Conditions. J. Agric. Food Chem. 2000, 48, 3761–3766. [Google Scholar] [CrossRef]
- Zhao, C.J.; Schieber, A.; Gänzle, M.G. Formation of Taste-Active Amino Acids, Amino Acid Derivatives and Peptides in Food Fermentations—A Review. Food Res. Int. 2016, 89, 39–47. [Google Scholar] [CrossRef]
- Drabińska, N.; Jeż, M.; Nogueira, M. Variation in the Accumulation of Phytochemicals and Their Bioactive Properties among the Aerial Parts of Cauliflower. Antioxidants 2021, 10, 1597. [Google Scholar] [CrossRef]
- Drabińska, N. The Evaluation of Amino Acid Profiles in Gluten-Free Mini Sponge Cakes Fortified with Broccoli By-Product. Separations 2022, 9, 81. [Google Scholar] [CrossRef]
- Oliveira, A.P.; Pereira, D.M.; Andrade, P.B.; Valentão, P.; Sousa, C.; Pereira, J.A.; Bento, A.; Rodrigues, M.Â.; Seabra, R.M.; Silva, B.M. Free Amino Acids of Tronchuda Cabbage (Brassica oleracea L. Var. Costata DC): Influence of Leaf Position (Internal or External) and Collection Time. J. Agric. Food Chem. 2008, 56, 5216–5221. [Google Scholar] [CrossRef] [PubMed]
- Zhao, N.; Zhang, C.; Yang, Q.; Guo, Z.; Yang, B.; Lu, W.; Li, D.; Tian, F.; Liu, X.; Zhang, H.; et al. Selection of Taste Markers Related to Lactic Acid Bacteria Microflora Metabolism for Chinese Traditional Paocai: A Gas Chromatography–Mass Spectrometry-Based Metabolomics Approach. J. Agric. Food Chem. 2016, 64, 2415–2422. [Google Scholar] [CrossRef] [PubMed]
- Bel-Rhlid, R.; Berger, R.G.; Blank, I. Bio-Mediated Generation of Food Flavors—Towards Sustainable Flavor Production Inspired by Nature. Trends Food Sci. Technol. 2018, 78, 134–143. [Google Scholar] [CrossRef]
- Smit, G.; Smit, B.A.; Engels, W.J.M. Flavour Formation by Lactic Acid Bacteria and Biochemical Flavour Profiling of Cheese Products. FEMS Microbiol. Rev. 2005, 29, 591–610. [Google Scholar] [CrossRef]
- Peyer, L.C.; Zannini, E.; Arendt, E.K. Lactic Acid Bacteria as Sensory Biomodulators for Fermented Cereal-Based Beverages. Trends Food Sci. Technol. 2016, 54, 17–25. [Google Scholar] [CrossRef]
- Van Gemert, L.J. Compilations of Odour Threshold Values in Air, Water and Other Media, 2nd ed.; Oliemans, Punter & Partners BV: Utrecht, The Netherlands, 2011; ISBN 9789081089401. [Google Scholar]
- Ortner, E.; Granvogl, M.; Schieberle, P. Elucidation of Thermally Induced Changes in Key Odorants of White Mustard Seeds (Sinapis alba L.) and Rapeseeds (Brassica napus L.) Using Molecular Sensory Science. J. Agric. Food Chem. 2016, 64, 8179–8190. [Google Scholar] [CrossRef]
- Holland, R.; Liu, S.-Q.; Crow, V.L.; Delabre, M.-L.; Lubbers, M.; Bennett, M.; Norris, G. Esterases of Lactic Acid Bacteria and Cheese Flavour: Milk Fat Hydrolysis, Alcoholysis and Esterification. Int. Dairy J. 2005, 15, 711–718. [Google Scholar] [CrossRef]
- Major, N.; Bažon, I.; Išić, N.; Kovačević, T.K.; Ban, D.; Radeka, S.; Goreta Ban, S. Bioactive Properties, Volatile Compounds, and Sensory Profile of Sauerkraut Are Dependent on Cultivar Choice and Storage Conditions. Foods 2022, 11, 1218. [Google Scholar] [CrossRef]
- Zhang, J.; Zhang, C.; Xin, X.; Liu, D.; Zhang, W. Comparative Analysis of Traditional and Modern Fermentation for Xuecai and Correlations Between Volatile Flavor Compounds and Bacterial Community. Front. Microbiol. 2021, 12, 631054. [Google Scholar] [CrossRef]
- Vincenti, S.; Mariani, M.; Alberti, J.C.; Jacopini, S.; de Caraffa, V.B.B.; Berti, L.; Maury, J. Biocatalytic Synthesis of Natural Green Leaf Volatiles Using the Lipoxygenase Metabolic Pathway. Catalysts 2019, 9, 873. [Google Scholar] [CrossRef]
- Ciska, E.; Honke, J.; Drabińska, N. Changes in Glucosinolates and Their Breakdown Products during the Fermentation of Cabbage and Prolonged Storage of Sauerkraut: Focus on Sauerkraut Juice. Food Chem. 2021, 365, 130498. [Google Scholar] [CrossRef] [PubMed]
- Daxenbichler, M.E.; VanEtten, C.H.; Williams, P.H. Glucosinolate Products in Commercial Sauerkraut. J. Agric. Food Chem. 1980, 28, 809–811. [Google Scholar] [CrossRef] [PubMed]
- Sikorska-Zimny, K.; Beneduce, L. The Glucosinolates and Their Bioactive Derivatives in Brassica: A Review on Classification, Biosynthesis and Content in Plant Tissues, Fate during and after Processing, Effect on the Human Organism and Interaction with the Gut Microbiota. Crit. Rev. Food Sci. Nutr. 2020, 61, 2544–2571. [Google Scholar] [CrossRef] [PubMed]
- Witzel, K.; Abu Risha, M.; Albers, P.; Börnke, F.; Hanschen, F.S. Identification and Characterization of Three Epithiospecifier Protein Isoforms in Brassica Oleracea. Front. Plant Sci. 2019, 10, 1552. [Google Scholar] [CrossRef]
- Verkerk, R.; Schreiner, M.; Krumbein, A.; Ciska, E.; Holst, B.; Rowland, I.; de Schrijver, R.; Hansen, M.; Gerhäuser, C.; Mithen, R.; et al. Glucosinolates in Brassica Vegetables: The Influence of the Food Supply Chain on Intake, Bioavailability and Human Health. Mol. Nutr. Food Res. 2009, 53, S219. [Google Scholar] [CrossRef] [PubMed]
- Mullaney, J.A.; Kelly, W.J.; McGhie, T.K.; Ansell, J.; Heyes, J.A. Lactic Acid Bacteria Convert Glucosinolates to Nitriles Efficiently Yet Differently from Enterobacteriaceae. J. Agric. Food Chem. 2013, 61, 3039–3046. [Google Scholar] [CrossRef]
- Tolonen, M.; Taipale, M.; Viander, B.; Pihlava, J.-M.; Korhonen, H.; Ryhänen, E.-L. Plant-Derived Biomolecules in Fermented Cabbage. J. Agric. Food Chem. 2002, 50, 6798–6803. [Google Scholar] [CrossRef]
- Ciska, E.; Pathak, D.R. Glucosinolate Derivatives in Stored Fermented Cabbage. J. Agric. Food Chem. 2004, 52, 7938–7943. [Google Scholar] [CrossRef]
- Palliyaguru, D.L.; Yuan, J.-M.; Kensler, T.W.; Fahey, J.W. Isothiocyanates: Translating the Power of Plants to People. Mol. Nutr. Food Res. 2018, 62, e1700965. [Google Scholar] [CrossRef]
- Paramithiotis, S.; Hondrodimou, O.L.; Drosinos, E.H. Development of the Microbial Community during Spontaneous Cauliflower Fermentation. Food Res. Int. 2010, 43, 1098–1103. [Google Scholar] [CrossRef]
- Ye, J.-H.; Huang, L.-Y.; Terefe, N.S.; Augustin, M.A. Fermentation-Based Biotransformation of Glucosinolates, Phenolics and Sugars in Retorted Broccoli Puree by Lactic Acid Bacteria. Food Chem. 2019, 286, 616–623. [Google Scholar] [CrossRef] [PubMed]
- Maifreni, M.; Marino, M.; Conte, L. Lactic Acid Fermentation of Brassica Rapa: Chemical and Microbial Evaluation of a Typical Italian Product (Brovada). Eur. Food Res. Technol. 2004, 218, 469–473. [Google Scholar] [CrossRef]
- Liu, M.C.; Li, Z.G.; Deng, W.; Wang, G.M.; Yang, Y.W. Changes in Volatile Compounds of Pickled Mustard Tuber (Brassica Juncea Var. Tsatsai) during the Pickling Process. Int. J. Food Sci. Technol. 2009, 44, 2278–2286. [Google Scholar] [CrossRef]
- Zhang, C.; Zhang, J.; Liu, D. Biochemical Changes and Microbial Community Dynamics during Spontaneous Fermentation of Zhacai, a Traditional Pickled Mustard Tuber from China. Int. J. Food Microbiol. 2021, 347, 109199. [Google Scholar] [CrossRef]
- Zhao, D.; Tang, J.; Ding, X. Analysis of Volatile Components during Potherb Mustard (Brassica juncea, Coss.) Pickle Fermentation Using SPME-GC-MS. LWT Food Sci. Technol. 2007, 40, 439–447. [Google Scholar] [CrossRef]
- Liu, D.; Zhang, C.; Zhang, J.; Xin, X.; Liao, X. Metagenomics Reveals the Formation Mechanism of Flavor Metabolites during the Spontaneous Fermentation of Potherb Mustard (Brassica juncea Var. Multiceps). Food Res. Int. 2021, 148, 110622. [Google Scholar] [CrossRef]
- Tomita, S.; Watanabe, J.; Kuribayashi, T.; Tanaka, S.; Kawahara, T. Metabolomic Evaluation of Different Starter Culture Effects on Water-Soluble and Volatile Compound Profiles in Nozawana Pickle Fermentation. Food Chem. Mol. Sci. 2021, 2, 100019. [Google Scholar] [CrossRef]
- Suzuki, C.; Ohnishi-Kameyama, M.; Sasaki, K.; Murata, T.; Yoshida, M. Behavior of Glucosinolates in Pickling Cruciferous Vegetables. J. Agric. Food Chem. 2006, 54, 9430–9436. [Google Scholar] [CrossRef]
- Peñas, E.; Martinez-Villaluenga, C.; Frias, J. Chapter 24—Sauerkraut: Production, Composition, and Health Benefits. In Fermented Foods in Health and Disease Prevention; Frias, J., Martinez-Villaluenga, C., Peñas, E., Eds.; Academic Press: Boston, MA, USA, 2017; pp. 557–576. ISBN 978-0-12-802309-9. [Google Scholar]
- Wiander, B.; Ryhänen, E.-L. Laboratory and Large-Scale Fermentation of White Cabbage into Sauerkraut and Sauerkraut Juice by Using Starters in Combination with Mineral Salt with a Low NaCl Content. Eur. Food Res. Technol. 2005, 220, 191–195. [Google Scholar] [CrossRef]
- Wolkers-Rooijackers, J.C.M.; Thomas, S.M.; Nout, M.J.R. Effects of Sodium Reduction Scenarios on Fermentation and Quality of Sauerkraut. LWT Food Sci. Technol. 2013, 54, 383–388. [Google Scholar] [CrossRef]
- Kapusta-Duch, J.; Kusznierewicz, B.; Leszczyńska, T.; Borczak, B. The Effect of Package Type on Selected Parameters of Nutritional Quality of the Chilled Stored Red Sauerkraut. J. Food Process. Preserv. 2017, 41, 1–12. [Google Scholar] [CrossRef]
- Kapusta-Duch, J.; Kusznierewicz, B.; Leszczyńska, T.; Borczak, B. Effect of Package Type on Selected Parameters of Nutritional Quality of Chill-Stored White Sauerkraut. Pol. J. Food Nutr. Sci. 2017, 67, 137–144. [Google Scholar] [CrossRef]
- Mcfeeters, R.F. Fermentation Microorganisms and Flavor Changes in Fermented Foods. J. Food Sci. 2004, 69, FMS35–FMS37. [Google Scholar] [CrossRef]
- Lonergan, D.; Lindsay, R.C. Evaluation of Sauerkraut-Like Products From Direct-Acidification of Cabbage1. J. Food Prot. 1979, 42, 38–42. [Google Scholar] [CrossRef] [PubMed]
- Peñas, E.; Frias, J.; Sidro, B.; Vidal-Valverde, C. Chemical Evaluation and Sensory Quality of Sauerkrauts Obtained by Natural and Induced Fermentations at Different NaCl Levels from Brassica Oleracea Var. Capitata Cv. Bronco Grown in Eastern Spain. Effect of Storage. J. Agric. Food Chem. 2010, 58, 3549–3557. [Google Scholar] [CrossRef]
- Peñas, E.; Pihlava, J.M.; Vidal-Valverde, C.; Frias, J. Influence of Fermentation Conditions of (Brassica Oleracea L. Var. Capitata) on the Volatile Glucosinolate Hydrolysis Compounds of Sauerkrauts. LWT Food Sci. Technol. 2012, 48, 16–23. [Google Scholar] [CrossRef]
- Martinez-Villaluenga, C.; Peñas, E.; Frias, J.; Ciska, E.; Honke, J.; Piskula, M.K.; Kozlowska, H.; Vidal-Valverde, C. Influence of Fermentation Conditions on Glucosinolates, Ascorbigen, and Ascorbic Acid Content in White Cabbage (Brassica oleracea Var. Capitata Cv. Taler) Cultivated in Different Seasons. J. Food Sci. 2009, 74, C62–C67. [Google Scholar] [CrossRef]
- Sarvan, I.; Valerio, F.; Lonigro, S.L.; de Candia, S.; Verkerk, R.; Dekker, M.; Lavermicocca, P. Glucosinolate Content of Blanched Cabbage (Brassica oleracea Var. Capitata) Fermented by the Probiotic Strain Lactobacillus Paracasei LMG-P22043. Food Res. Int. 2013, 54, 706–710. [Google Scholar] [CrossRef]
- Śmiechowska Anna The Assessment of the Sensory Quality of the Sauerkraut from Organic and Conven-Tional Farming. J. Res. Appl. Agric. Eng. 2017, 62, 168–172.
- Satora, P.; Skotniczny, M.; Strnad, S.; Piechowicz, W. Chemical Composition and Sensory Quality of Sauerkraut Produced from Different Cabbage Varieties. LWT 2021, 136, 110325. [Google Scholar] [CrossRef]
- Yang, X.; Hu, W.; Xiu, Z.; Jiang, A.; Yang, X.; Saren, G.; Ji, Y.; Guan, Y.; Feng, K. Microbial Community Dynamics and Metabolome Changes during Spontaneous Fermentation of Northeast Sauerkraut From Different Households. Front. Microbiol. 2020, 11, 1878. [Google Scholar] [CrossRef] [PubMed]
- Jung, J.Y.; Lee, S.H.; Jeon, C.O. Kimchi Microflora: History, Current Status, and Perspectives for Industrial Kimchi Production. Appl. Microbiol. Biotechnol. 2014, 98, 2385–2393. [Google Scholar] [CrossRef]
- Cheigh, H.-S.; Park, K.-Y.; Lee, C.Y. Biochemical, Microbiological, and Nutritional Aspects of Kimchi (Korean Fermented Vegetable Products). Crit. Rev. Food Sci. Nutr. 1994, 34, 175–203. [Google Scholar] [CrossRef] [PubMed]
- Chang, J.-H.; Shim, Y.Y.; Cha, S.-K.; Chee, K.M. Probiotic Characteristics of Lactic Acid Bacteria Isolated from Kimchi. J. Appl. Microbiol. 2010, 109, 220–230. [Google Scholar] [CrossRef]
- You, S.Y.; Yang, J.S.; Kim, S.H.; Hwang, I.M. Changes in the Physicochemical Quality Characteristics of Cabbage Kimchi with Respect to Storage Conditions. J. Food Qual. 2017, 2017, 9562981. [Google Scholar] [CrossRef]
- Rhee, S.J.; Lee, J.E.; Lee, C.H. Importance of Lactic Acid Bacteria in Asian Fermented Foods. Microb. Cell Fact. 2011, 10, S5. [Google Scholar] [CrossRef]
- Chang, J.Y.; Chang, H.C. Improvements in the Quality and Shelf Life of Kimchi by Fermentation with the Induced Bacteriocin-Producing Strain, Leuconostoc Citreum GJ7 as a Starter. J Food Sci 2010, 75, M103–M110. [Google Scholar] [CrossRef]
- Lee, J.J.; Choi, Y.J.; Lee, M.J.; Park, S.J.; Oh, S.J.; Yun, Y.R.; Min, S.G.; Seo, H.Y.; Park, S.H.; Lee, M.A. Effects of Combining Two Lactic Acid Bacteria as a Starter Culture on Model Kimchi Fermentation. Food Res. Int. 2020, 136, 109591. [Google Scholar] [CrossRef]
- Seo, S.H.; Park, S.E.; Kim, E.J.; Lee, K.I.; Na, C.S.; Son, H.S. A GC-MS Based Metabolomics Approach to Determine the Effect of Salinity on Kimchi. Food Res. Int. 2018, 105, 492–498. [Google Scholar] [CrossRef]
- Yun, J.W.; Kang, S.C.; Song, S.K. Mannitol Accumulation during Fermentation of Kimchi. J. Ferment. Bioeng. 1996, 81, 279–280. [Google Scholar] [CrossRef]
- Chambers, E.; Lee, J.; Chun, S.; Miller, A.E. Development of a Lexicon for Commercially Available Cabbage (Baechu) Kimchi. J. Sens. Stud. 2012, 27, 511–518. [Google Scholar] [CrossRef]
- Cha, Y.J.; Kim, H.; Cadwallader, K.R. Aroma-Active Compounds in Kimchi during Fermentation. J. Agric. Food Chem. 1998, 46, 1944–1953. [Google Scholar] [CrossRef]
- Hong, S.P.; Lee, E.J.; Kim, Y.H.; Ahn, D.U. Effect of Fermentation Temperature on the Volatile Composition of Kimchi. J. Food Sci. 2016, 81, C2623–C2629. [Google Scholar] [CrossRef] [PubMed]
- Jang, S.H.; Kim, M.J.; Lim, J.; Hong, J.H. Cross-Cultural Comparison of Consumer Acceptability of Kimchi with Different Degree of Fermentation. J. Sens. Stud. 2016, 31, 124–134. [Google Scholar] [CrossRef]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wieczorek, M.N.; Drabińska, N. Flavour Generation during Lactic Acid Fermentation of Brassica Vegetables—Literature Review. Appl. Sci. 2022, 12, 5598. https://doi.org/10.3390/app12115598
Wieczorek MN, Drabińska N. Flavour Generation during Lactic Acid Fermentation of Brassica Vegetables—Literature Review. Applied Sciences. 2022; 12(11):5598. https://doi.org/10.3390/app12115598
Chicago/Turabian StyleWieczorek, Martyna Natalia, and Natalia Drabińska. 2022. "Flavour Generation during Lactic Acid Fermentation of Brassica Vegetables—Literature Review" Applied Sciences 12, no. 11: 5598. https://doi.org/10.3390/app12115598
APA StyleWieczorek, M. N., & Drabińska, N. (2022). Flavour Generation during Lactic Acid Fermentation of Brassica Vegetables—Literature Review. Applied Sciences, 12(11), 5598. https://doi.org/10.3390/app12115598






