The Role of Brownmillerite in Preparation of High-Belite Sulfoaluminate Cement Clinker
Abstract
:1. Introduction
2. Experimental Section
2.1. Materials
2.2. Preparation
2.2.1. Mineral Design of Clinker
2.2.2. Clinker Preparation
2.2.3. Cement Preparation
2.3. Characterization
3. Results and Discussion
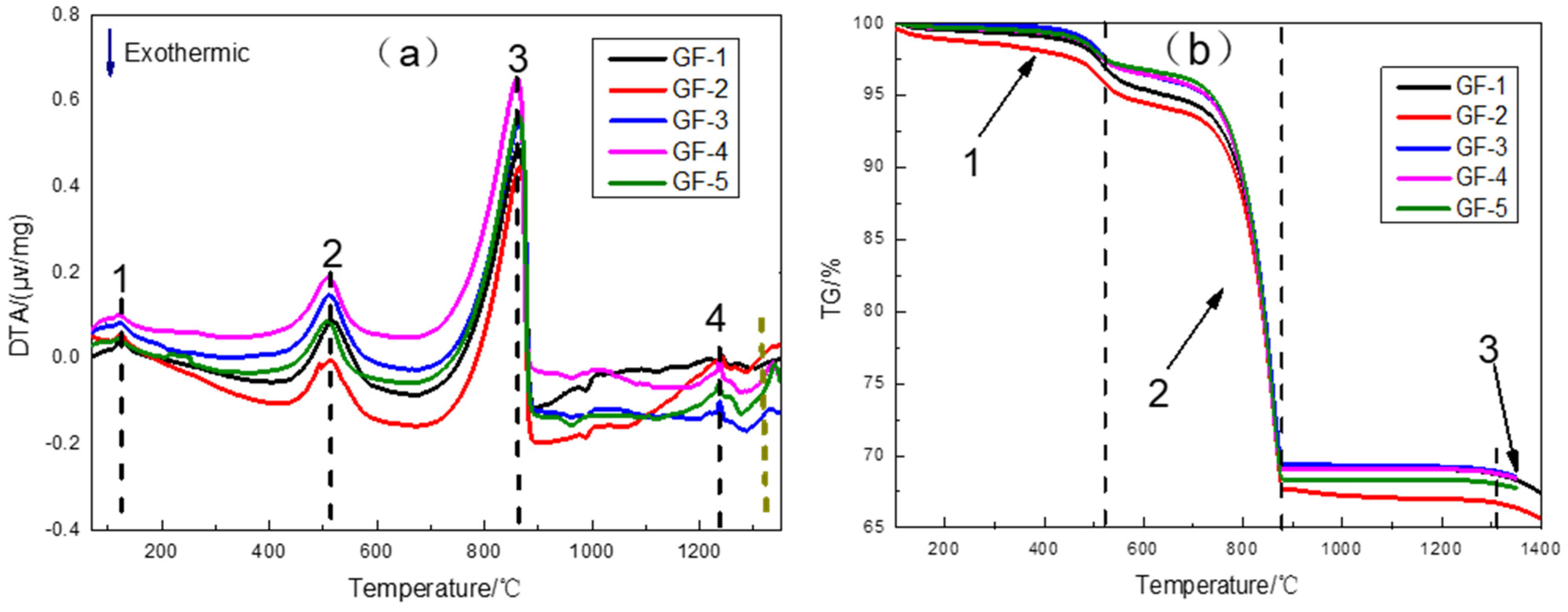
3.1. Thermal Evolution of HBSC Raw Materials
3.2. Sintering of HBSC Clinker Containing Brownmillerite
3.2.1. Burnability
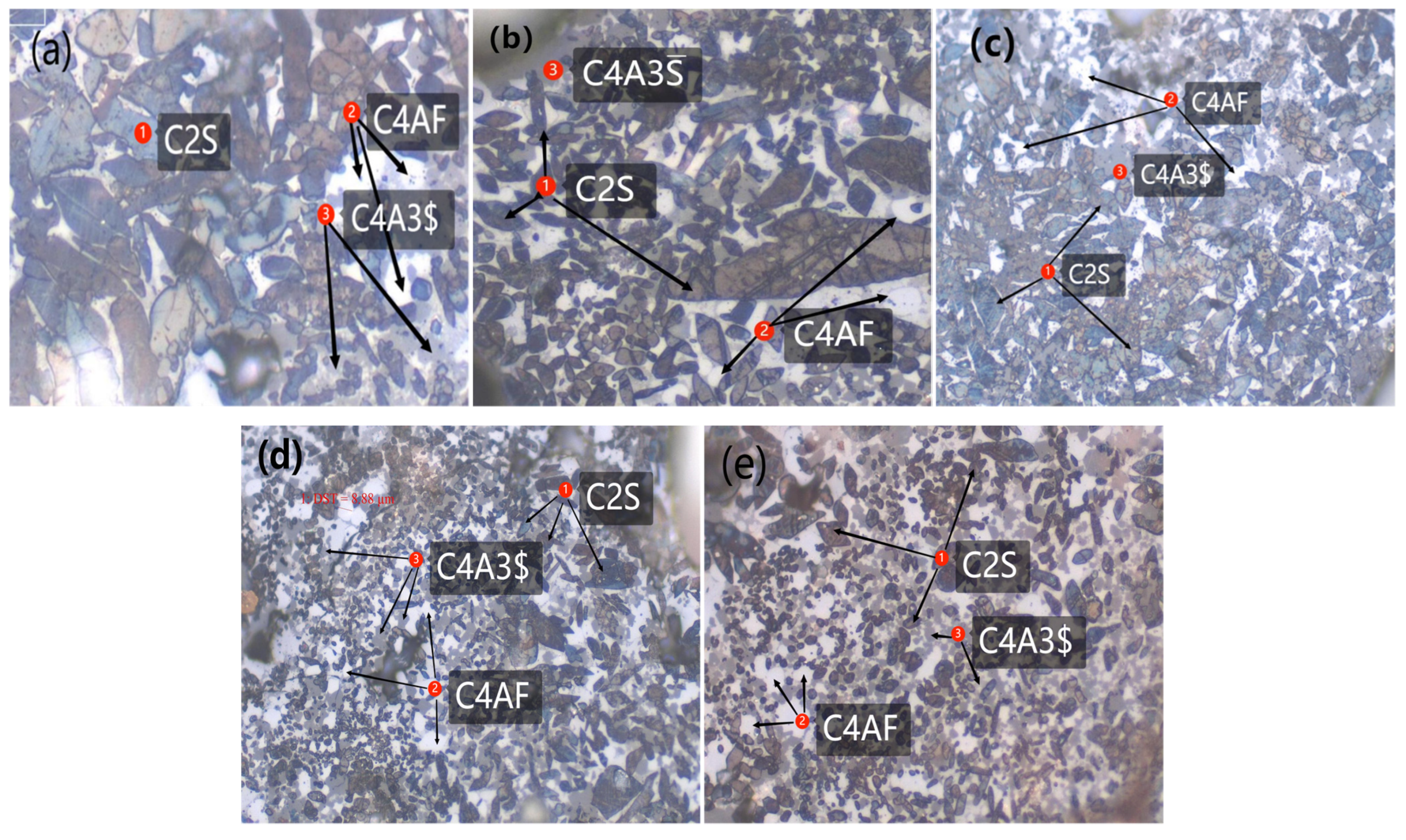
3.2.2. Mineral Morphology
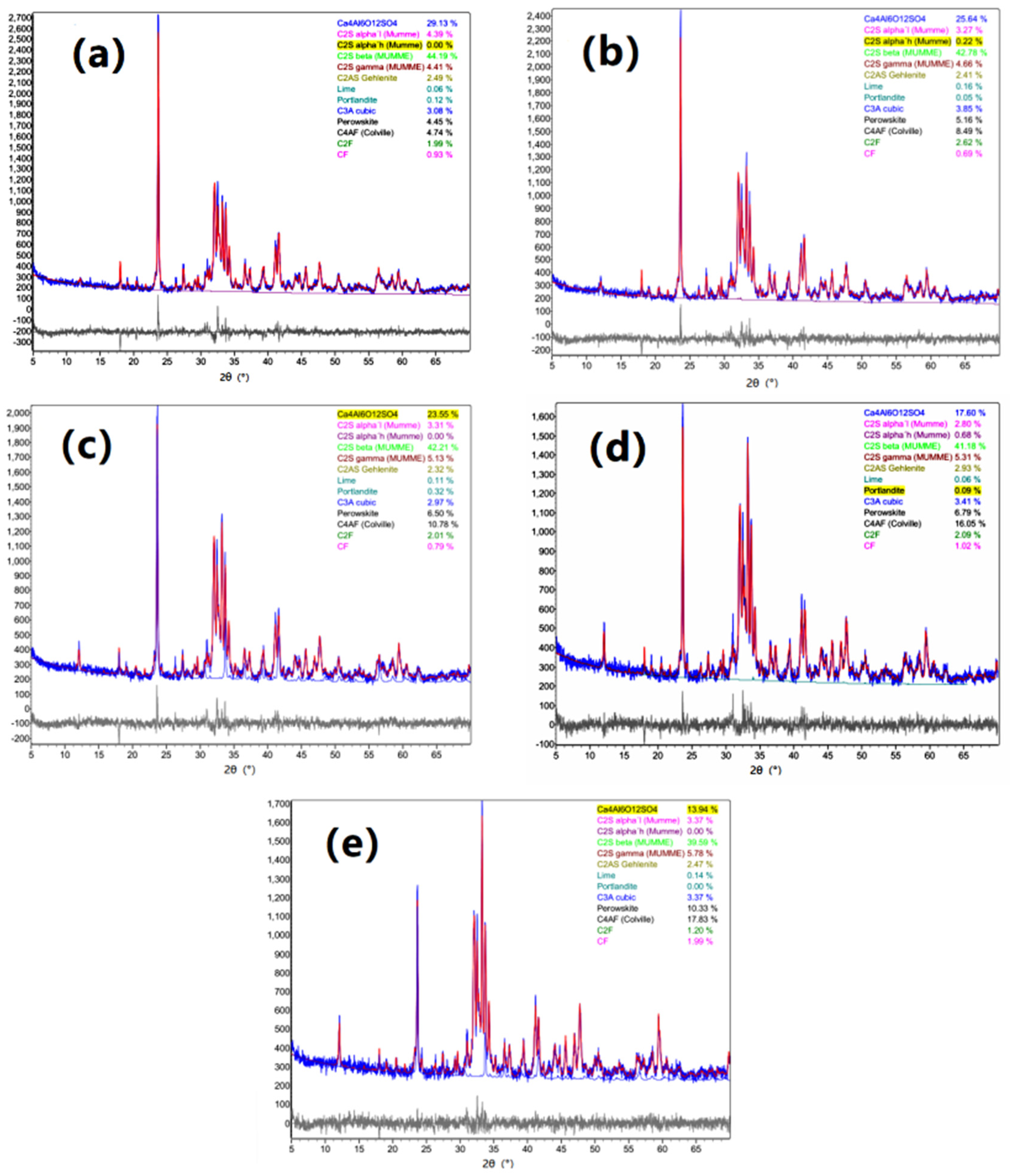
3.2.3. Mineral Composition
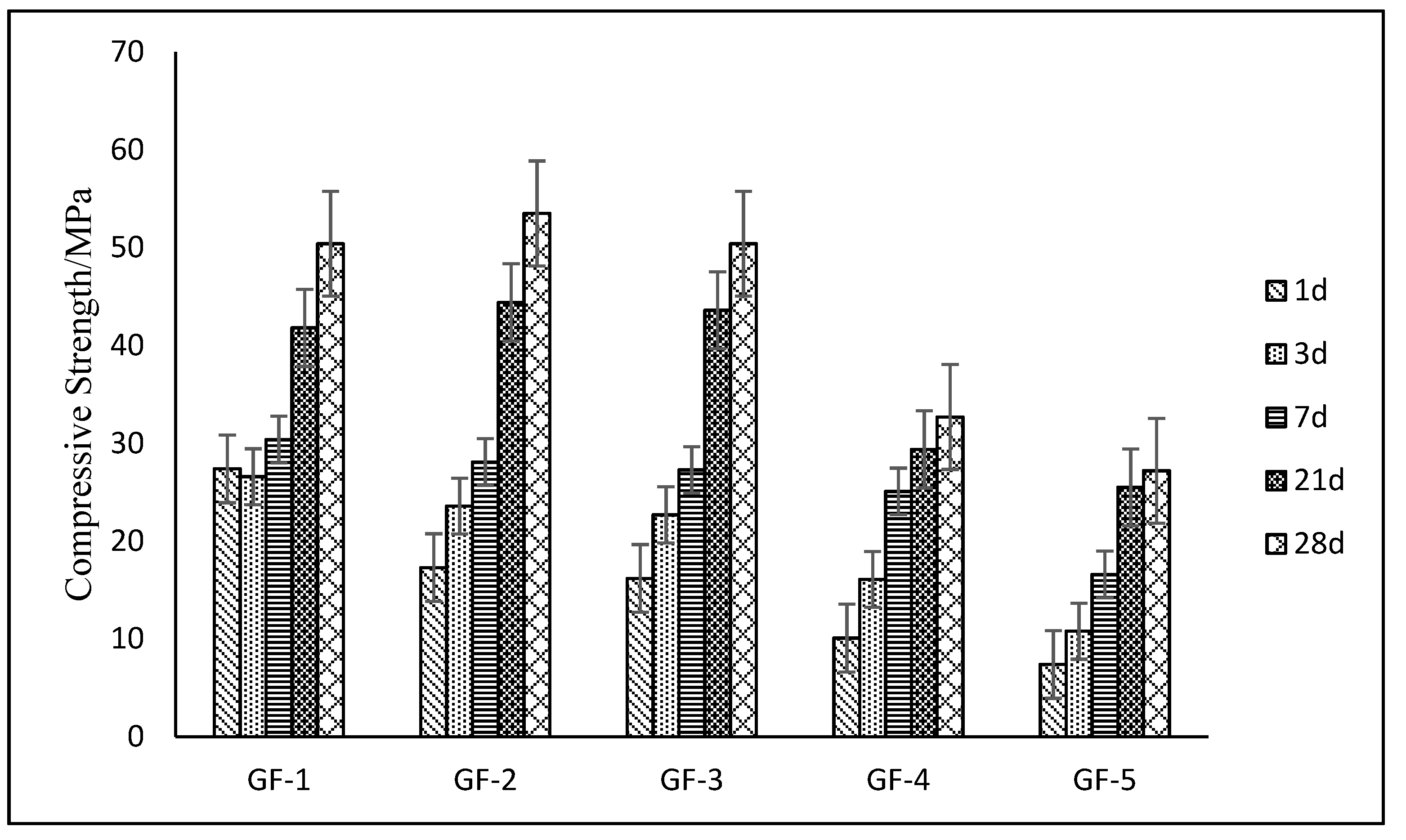
3.3. Mechanical Performance of HBSC Containing Brownmillerite
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Gartner, E.; Hirao, H. A review of alternative approaches to the reduction of CO2 emissions associated with the manufacture of the binder phase in concrete. Cem. Concr. Res. 2015, 78, 126–142. [Google Scholar] [CrossRef] [Green Version]
- Cuberos, A.J.M.; de la Torre, Á.G.; Álvarez-Pinazo, G.; Martín-Sedeño, M.C.; Schollbach, K.; Pöllmann, H.; Aranda, M.A.G. Active iron-rich belite sulfoaluminate cements: Clinkering and hydration. Environ. Sci. Technol. 2010, 44, 6855–6862. [Google Scholar] [CrossRef] [PubMed]
- Baltakys, K.; Dambrauskas, T.; Rubinaite, D.; Siauciunas, R.; Grineviciene, A. Formation and hydration of eco-friendly cement using industrial wastes as raw materials. Sci. Rep. 2021, 11, 14742. [Google Scholar] [CrossRef] [PubMed]
- Han, S.; Zhong, J.; Yu, Q.; Yan, L.; Ou, J. Sulfate resistance of eco-friendly and sulfate-resistant concrete using seawater sea-sand and high-ferrite Portland cement. Constr. Build. Mater. 2021, 305, 753–760. [Google Scholar] [CrossRef]
- Popescu, C.; Muntean, M.; Sharp, J. Industrial trial production of low energy belite cement. Cem. Concr. Compos. 2003, 25, 689–693. [Google Scholar] [CrossRef]
- Gartner, E.; Sui, T. Alternative cement clinkers. Cem. Concr. Res. 2018, 114, 27–39. [Google Scholar] [CrossRef]
- El Khessaimi, Y.; El Hafiane, Y.; Smith, A.; Trauchessec, R.; Diliberto, C.; Lecomte, A. Solid-state synthesis of pure ye’elimite. J. Eur. Ceram. Soc. 2018, 38, 3401–3411. [Google Scholar] [CrossRef]
- Gao, Y.; Li, Z.; Zhang, J.; Zhang, Q.; Wang, Y. Synergistic use of industrial solid wastes to prepare belite-rich sulphoaluminate cement and its feasibility use in repairing materials. Constr. Build. Mater. 2020, 264, 201–213. [Google Scholar] [CrossRef]
- Isteri, V.; Ohenoja, K.; Hanein, T.; Kinoshita, H.; Tanskanen, P.; Illikainen, M.; Fabritius, T. Production and properties of ferrite-rich CSAB cement from metallurgical industry residues. Sci. Total Environ. 2020, 712, 208–219. [Google Scholar] [CrossRef]
- Chang, J.; Zhang, X.; Gu, Y.Y.; Zhang, Y.Y. Effects of pozzolanic reaction on carbonation degree and strength of steel slag compacts containing zeolite. Constr. Build. Mater. 2021, 277, 334–343. [Google Scholar] [CrossRef]
- Kiventerä, J.; Piekkari, K.; Isteri, V.; Ohenoja, K.; Tanskanen, P.; Illikainen, M. Solidifification/stabilization of gold mine tailings using calcium sulfoaluminate-belite cement. J. Clean. Prod. 2019, 239, 008–015. [Google Scholar] [CrossRef]
- Zhang, G.; Ren, Q.; He, J.; Jiang, S.; Cheng, X.; Yu, Y.; Huang, S.; Zhang, S.; Zhou, M. New understanding of early hydration of C4AF under surface vitrifification. Powder Technol. 2021, 377, 372–378. [Google Scholar] [CrossRef]
- Hertel, T.; Van den Bulck, A.; Onisei, S.; Sivakumar, P.P.; Pontikes, Y. Boosting the use of bauxite residue (red mud) in cement—Production of an Fe-rich calciumsulfoaluminate-ferrite clinker and characterisation of the hydration. Cem. Con.Res. 2021, 145, 463–480. [Google Scholar]
- Tao, Y.; Wan, D.; Zhang, W.; Wang, F.; Hu, S. Intrinsic reactivity and dissolution characteristics of tetracalcium aluminoferrite. Cem. Con.Res. 2021, 146, 485–493. [Google Scholar] [CrossRef]
- Yao, X.; Yang, S.; Huang, Y.; Wu, S.; Yao, Y.; Wang, W. Effect of CaSO4 batching in raw material on the iron-bearing mineral transition of ferric-rich sulfoaluminate cement. Constr. Build. Mater. 2020, 250, 783–792. [Google Scholar] [CrossRef]
- Hertel, T.; Van den Bulck, A.; Onisei, S.; Sivakumar, P.P.; Pontikes, Y. Synthesis Mechanism and Material Characterization of C4AF Obtained by the Self-propagating Combustion Reaction Method. J. Wuhan Univ. Technol. Mater. 2018, 3, 1099–1107. [Google Scholar]
- Martín-Sedeño, M.C.; Cuberos, A.J.; Ángeles, G.; Álvarez-Pinazo, G.; Ordónez, L.M.; Gateshki, M.; Aranda, M.A. Aluminum-rich belite sulfoaluminate cements: Clinkering and early age hydration. Cem. Concr. Rese 2019, 40, 359–369. [Google Scholar] [CrossRef] [Green Version]
- Benarchid, M.Y.; Rogez, J. The effect of Cr2O3 and P2O5 additions on the phase transformations during the formation of calcium sulfoaluminate C4A3S. Cem. Concr. Res. 2005, 35, 2074–2080. [Google Scholar] [CrossRef]
- Touzo, B.; Scrivener, K.L.; Glasser, F.P. Phase compositions and equilibria in the CaO-Al2O3-Fe2O3-SO3 system, for assemblages containing ye’elimite and ferrite Ca2(Al, Fe)O5. Cem. Concr. Res. 2013, 54, 77–86. [Google Scholar] [CrossRef]
- Idrissi, M.; Diouri, A.; Damidot, D.; Greneche, J.M.; Talbi, M.A.; Taibi, M. Characterisation of brownmillerite inclusion during the formation of calcium sulfoaluminate phase. Cem. Concr. Res. 2010, 40, 1314–1319. [Google Scholar] [CrossRef]
- Bullerjahn, F.; Schmitt, D.; Haha, M.B. Effect of raw mix design and of clinkering process on the formation and mineralogical composition of (ternesite) belite calcium sulphoaluminate ferrite clinker. Cem. Concr. Res. 2014, 59, 87–95. [Google Scholar] [CrossRef]
- Jadhav, R.; Debnath, N.C. Computation of X-ray powder diffractograms of cement components. Bull. Mater. Sci. 2021, 34, 1137–1150. [Google Scholar] [CrossRef] [Green Version]
- Álvarez-Pinazo, G.; Cuesta, A.; García-Maté, M.; Santacruz, I.; Losilla, E.R.; De la Torre, A.G.; León-Reinab, L.; Arandaa, M.A.G. Rietveld quantitative phase analysis of Yeelimite-containing cements. Cem. Concr. Res. 2012, 42, 960–971. [Google Scholar] [CrossRef] [Green Version]
- Irico, S.; Gastaldi, D.; Canonico, F.; Magnacca, G. Investigation of the microstructural evolution of calcium sulfoaluminate cements by thermoporometry. Cem. Concr. Res. 2013, 53, 239–247. [Google Scholar] [CrossRef]
- Morsli, K.; Angeles, G.; Zahir, M.; Aranda, M.A. Mineralogical phase analysis of alkali and sulfate bearing belite rich laboratory clinkers. Cem. Concr. Res. 2007, 37, 639–646. [Google Scholar] [CrossRef]
- Abir, F.; Mesnaoui, M.; Abouliatim, Y.; Nibou, L.; Labbilta, T.; El Hafiane, Y.; Smith, A. Effect of the addition of iron oxide on the microstructure of ye’elimite. Cem. Conr. Res. 2022, 151, 625–641. [Google Scholar] [CrossRef]
- Ndzila, J.S.; Liu, S.; Jing, G.; Wang, S.; Ye, Z. The effect of Fe3+ ion substitution on the crystal structure of ye’elimite. Ceram. Silik. 2020, 64, 18–28. [Google Scholar] [CrossRef] [Green Version]
- Cuesta, A.; De la Torre, G.; Losilla, E.R.; Santacruz, I.; Aranda, M.A.G. Pseudocubic crystal structure and phase transition in doped ye’elimite. Cryst. Growth Des. 2014, 14, 5158–5163. [Google Scholar] [CrossRef] [Green Version]
- Rungchet, A.; Poon, C.S.; Chindaprasirt, P.; Pimraksa, K. Synthesis of low temperature calcium sulfoaluminate-belite cements from industrial wastes and their hydration: Comparative studies between lignite fly ash and bottom ash. Cem. Concr. Compos. 2017, 83, 10–19. [Google Scholar] [CrossRef]
- De la Torre, A.G.; Cuberos, A.J.M.; Álvarez-Pinazo, G.; Cuesta, A.; Aranda, M.A.G. In situ powder diffraction study of belite sulfoaluminate clinkering. J. Synchrotron Radiat. 2011, 18, 506–514. [Google Scholar] [CrossRef]
- da Costa, E.B.; Rodríguez, E.D.; A Bernal, S.; Provis, J.; Gobbo, L.A.; Kirchheim, A.P. Production and hydration of calcium sulfoaluminate-belite cements derived from aluminium anodising sludge. Constr. Build. Mater. 2016, 122, 373–383. [Google Scholar] [CrossRef]
- Le Saoût, G.; Lothenbach, B.; Taquet, P.; Fryda, H.; Winnefeld, F. Hydration of calcium aluminate cement blended with anhydrite. Adv. Cem. Res. Thomas Telford 2018, 30, 24–36. [Google Scholar] [CrossRef] [Green Version]
- Huang, X.; Wang, F.; Hu, S.; Lu, Y.; Rao, M.; Mu, Y. Brownmillerite hydration in the presence of gypsum: The effect of Al/Fe ratio and sulfate ions. J. Am. Ceram. Soc. 2019, 102, 5545–5554. [Google Scholar] [CrossRef]

| Materials | Loss | SiO2 | Fe2O3 | Al2O3 | CaO | MgO | TiO2 | SO3 | ∑ |
|---|---|---|---|---|---|---|---|---|---|
| LS | 43.37 | 1.03 | 0.16 | 0.26 | 54.13 | 0.44 | - | - | 99.39 |
| AT | 14.06 | 37.20 | 1.88 | 43.15 | 0.57 | 0.41 | - | 0.05 | 97.32 |
| AM | 9.70 | 39.18 | 24.46 | 19.74 | 1.69 | 0.60 | 3.87 | - | 99.24 |
| AH | 8.47 | 4.05 | 1.02 | 0.41 | 33.85 | 5.12 | - | 46.60 | 99.52 |
| Materials | Cl | Cr2O3 | MnO | NiO | CuO | ZnO | SrO | ZrO2 | BaO |
|---|---|---|---|---|---|---|---|---|---|
| LS | 0.004 | 0.010 | 0.082 | 0.015 | 0.028 | 0.022 | 0.019 | 0.143 | 0.065 |
| AT | 0.002 | 0.014 | 0.068 | 0.020 | 0.007 | 0.020 | 0.028 | 0.065 | 0.062 |
| AM | 0.010 | 0.009 | 0.059 | 0.010 | 0.021 | 0.015 | 0.036 | 0.237 | 0.057 |
| AH | 0.006 | 0.003 | 0.000 | 0.004 | 0.005 | 0.003 | 0.408 | 0.000 | 0.020 |
| Sample | Mix Design of Raw Materials (wt%) | Minerals Composition (wt%) | ||||||
|---|---|---|---|---|---|---|---|---|
| LS | AM | AH | AT | C4A3$ | C2S | C4AF | f-CS | |
| GF-1 | 60.6 | 12.4 | 7.0 | 20.0 | 28.79 | 51.50 | 15.18 | 1.70 |
| GF-2 | 61.2 | 17.8 | 6.0 | 15.0 | 24.26 | 51.00 | 20.24 | 1.56 |
| GF-3 | 62.0 | 23.0 | 5.0 | 10.0 | 19.69 | 50.29 | 25.14 | 1.44 |
| GF-4 | 62.8 | 28.2 | 4.0 | 5.0 | 15.09 | 49.57 | 30.07 | 1.31 |
| GF-5 | 63.1 | 33.9 | 3.0 | 0.0 | 10.57 | 49.39 | 35.45 | 1.16 |
| Mineralogical Phase | ICSD |
|---|---|
| α-C2S | 81097 |
| β-C2S | 81096 |
| γ-C2S | 81095 |
| C4A3$ | 9560 |
| C4AF | 9197 |
| Perovskite | 62149 |
| C2AS | 87144 |
| Anhydrite | 16382 |
| C2F | 27808 |
| CF | 16695 |
| Sample | Peak 1 | Peak 2 | Peak 3 | Peak 4 |
|---|---|---|---|---|
| GF-1 | 125.4 | 516.5 | 863.1 | 1241.9 |
| GF-2 | 121.9 | 512.2 | 864.5 | 1239.3 |
| GF-3 | 121.8 | 512.3 | 864.2 | 1238.1 |
| GF-4 | 120.7 | 510.5 | 860.4 | 1237.3 |
| GF-5 | 118.0 | 507.5 | 863.6 | 1235.8 |
| Sample | Stage I | Stage II | Stage III |
|---|---|---|---|
| GF-1 | 4.65 | 25.63 | 0.89 |
| GF-2 | 4.51 | 26.63 | 1.08 |
| GF-3 | 3.70 | 26.84 | 0.89 |
| GF-4 | 3.26 | 27.30 | 0.65 |
| GF-5 | 2.87 | 28.48 | 0.58 |
| Sample | 1350 °C | 1330 °C | 1320 °C | 1300 °C |
|---|---|---|---|---|
| GF-1 | Reddish brown, highly dense | Reddish brown, dense | Reddish brown, loose | Reddish brown, loose |
| GF-2 | Brown, sticky | Brown, dense | Brown, dense | Brown, loose |
| GF-3 | Gray, highly sticky | Gray, dense | Gray, dense | Gray, dense |
| GF-4 | Dark gray, highly sticky | Dark gray, slightly sticky | Dark gray, dense | Dark gray, dense |
| GF-5 | Light black, highly sticky | Light black, sticky | Light black, dense | Light black, very dense |
| Sample | 1300 °C | 1320 °C | 1330 °C | 1350 °C |
|---|---|---|---|---|
| GF-1 | 0.92 | 0.41 | 0.29 | 0.32 |
| GF-2 | 0.67 | 0.57 | 0.00 | 0.29 |
| GF-3 | 0.93 | 0.53 | 0.23 | 0.15 |
| GF-4 | 0.93 | 0.73 | 0.26 | 0.22 |
| GF-5 | 0.64 | 0.70 | 0.26 | 0.13 |
| No. | C4A3$ | C2S | C2AS | C3A | Brownmillerite | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| α | β | γ | ∑ | C4AF | C2F | CF | ∑ | ||||
| GF-1 | 29.13 | 4.39 | 44.19 | 4.41 | 52.99 | 2.49 | 3.08 | 4.74 | 1.99 | 0.93 | 7.66 |
| GF-2 | 25.64 | 3.49 | 42.78 | 4.66 | 50.93 | 2.41 | 3.85 | 8.49 | 2.62 | 0.69 | 11.80 |
| GF-3 | 23.55 | 3.31 | 42.21 | 5.13 | 50.65 | 2.32 | 2.97 | 10.78 | 2.01 | 0.79 | 13.58 |
| GF-4 | 17.60 | 3.48 | 41.18 | 5.31 | 49.97 | 2.93 | 3.41 | 16.05 | 2.09 | 1.02 | 19.16 |
| GF-5 | 13.94 | 3.37 | 39.59 | 5.78 | 48.74 | 2.47 | 3.37 | 17.83 | 1.20 | 1.99 | 21.02 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, X.; Li, J.; Lu, Z.; Ng, S.; Niu, Y.; Jiang, J.; Xu, Y.; Lai, Z.; Liu, H. The Role of Brownmillerite in Preparation of High-Belite Sulfoaluminate Cement Clinker. Appl. Sci. 2022, 12, 4980. https://doi.org/10.3390/app12104980
Chen X, Li J, Lu Z, Ng S, Niu Y, Jiang J, Xu Y, Lai Z, Liu H. The Role of Brownmillerite in Preparation of High-Belite Sulfoaluminate Cement Clinker. Applied Sciences. 2022; 12(10):4980. https://doi.org/10.3390/app12104980
Chicago/Turabian StyleChen, Xuemei, Jun Li, Zhongyuan Lu, Serina Ng, Yunhui Niu, Jun Jiang, Yigang Xu, Zhenyu Lai, and Huan Liu. 2022. "The Role of Brownmillerite in Preparation of High-Belite Sulfoaluminate Cement Clinker" Applied Sciences 12, no. 10: 4980. https://doi.org/10.3390/app12104980
APA StyleChen, X., Li, J., Lu, Z., Ng, S., Niu, Y., Jiang, J., Xu, Y., Lai, Z., & Liu, H. (2022). The Role of Brownmillerite in Preparation of High-Belite Sulfoaluminate Cement Clinker. Applied Sciences, 12(10), 4980. https://doi.org/10.3390/app12104980





