Beneficial Health Potential of Algerian Polysaccharides Extracted from Plantago ciliata Desf. (Septentrional Sahara) Leaves and Seeds
Abstract
Featured Application
Abstract
1. Introduction
2. Materials and Methods
2.1. Raw Material and Chemicals
2.2. Polysaccharides Extractions
2.3. Determining Biochemical Compositions
2.4. FTIR Footprints
2.5. Monosaccharide Compositions by HPAEC-PAD en GC/MS-EI
2.5.1. GC/MS-EI Experiments
2.5.2. HPAEC-PAD Experiments
2.6. Biological Activities
2.6.1. Phagocytotic Activity
2.6.2. Anti-inflammatory Activity
2.6.3. Anti-complement Activity
2.6.4. Anti-diabetic Activity
2.7. Statistical Analysis
3. Results and Discussions
3.1. Structural Characterization of L-PSPC and S-PSPC
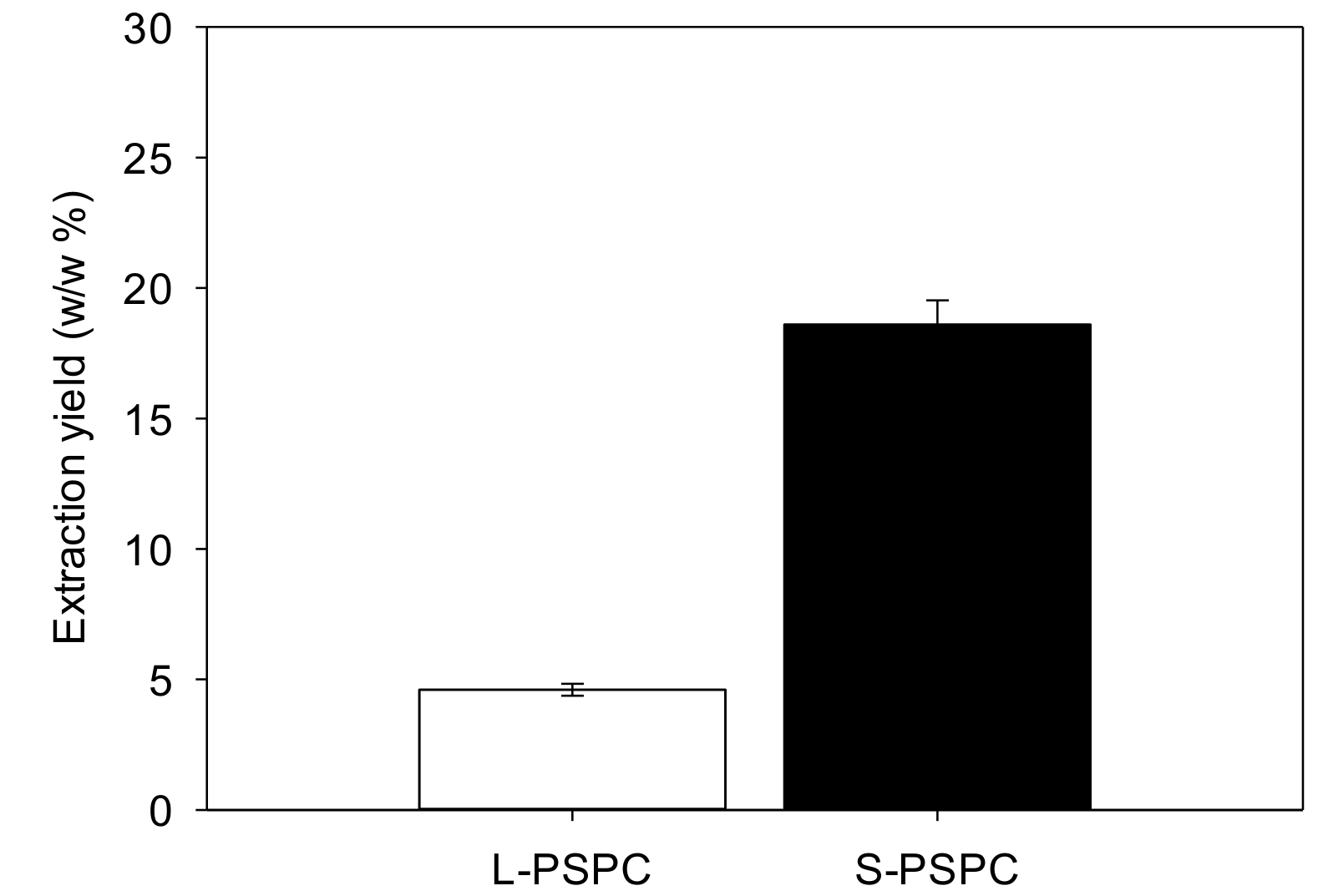
3.1.1. Main Biochemical Compositions
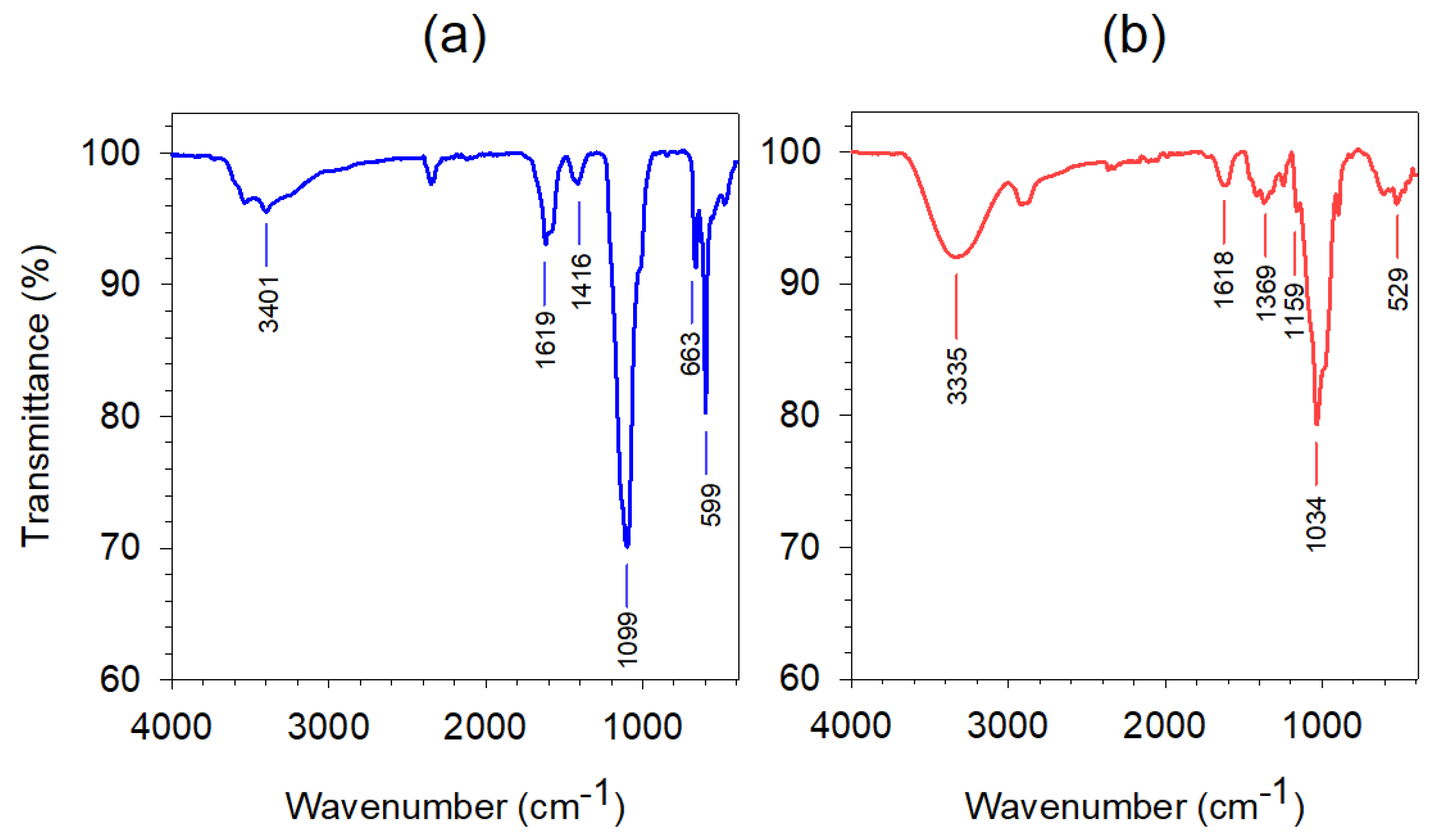
3.1.2. FTIR Spectroscopy
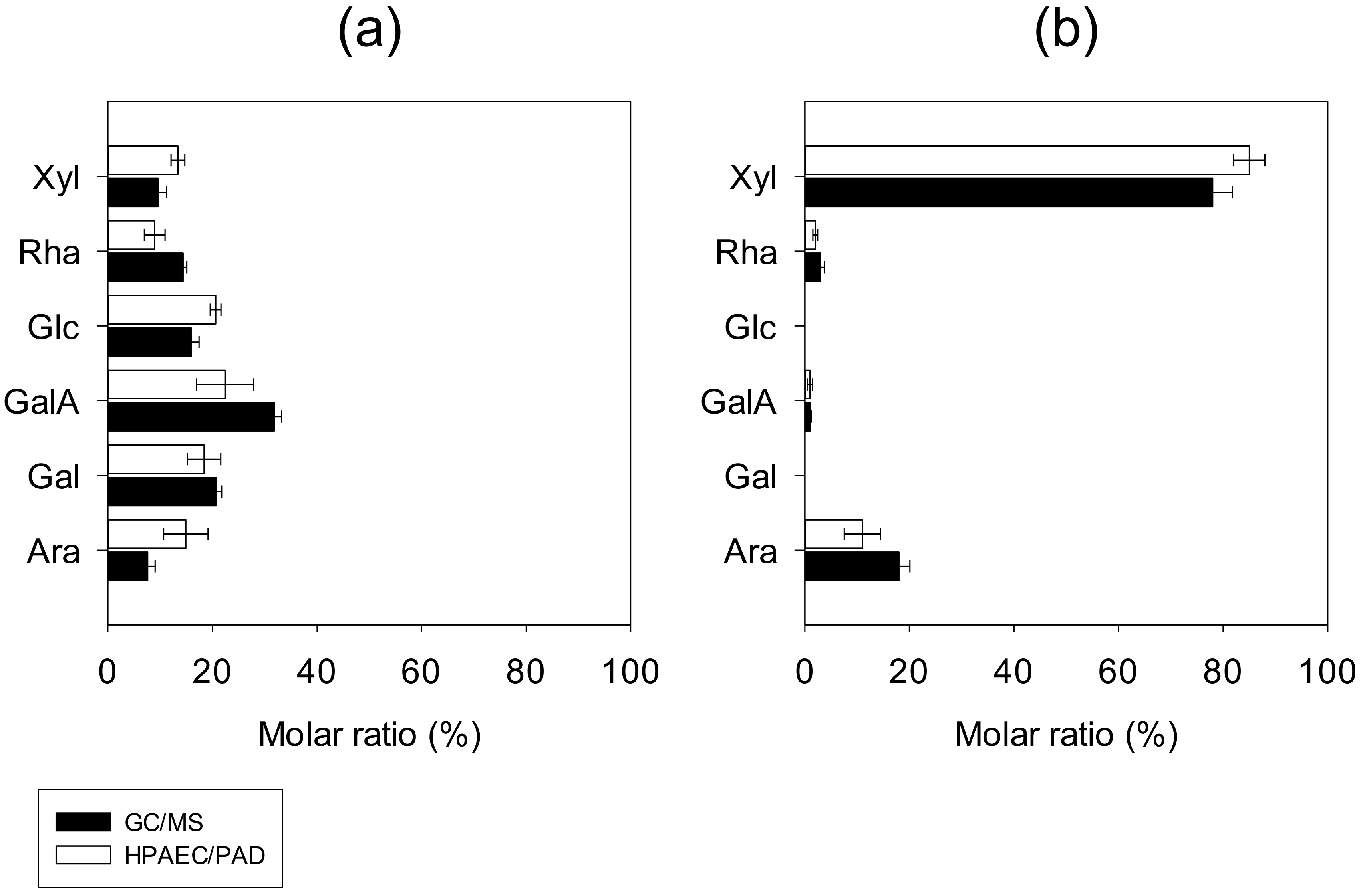
3.1.3. Monosaccharide Compositions and Main Structural Features
- Composition of L-PCPC
- Composition of S-PCPC
3.2. Potential Health Benefits of L-PSPC and S-PSPC
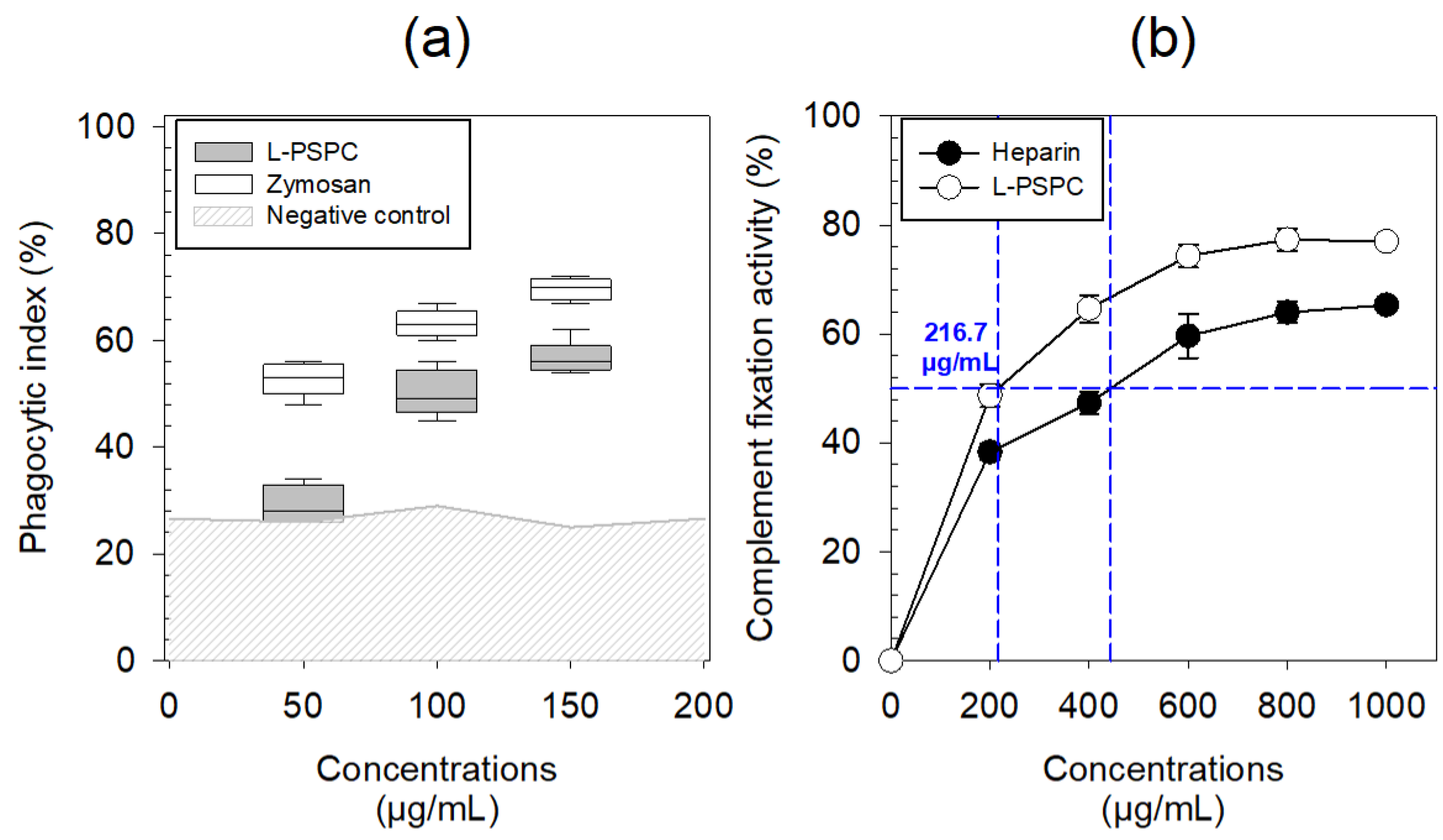
3.2.1. Biological Activities of L-PSPC
3.2.2. Biological Activities of S-PSPC
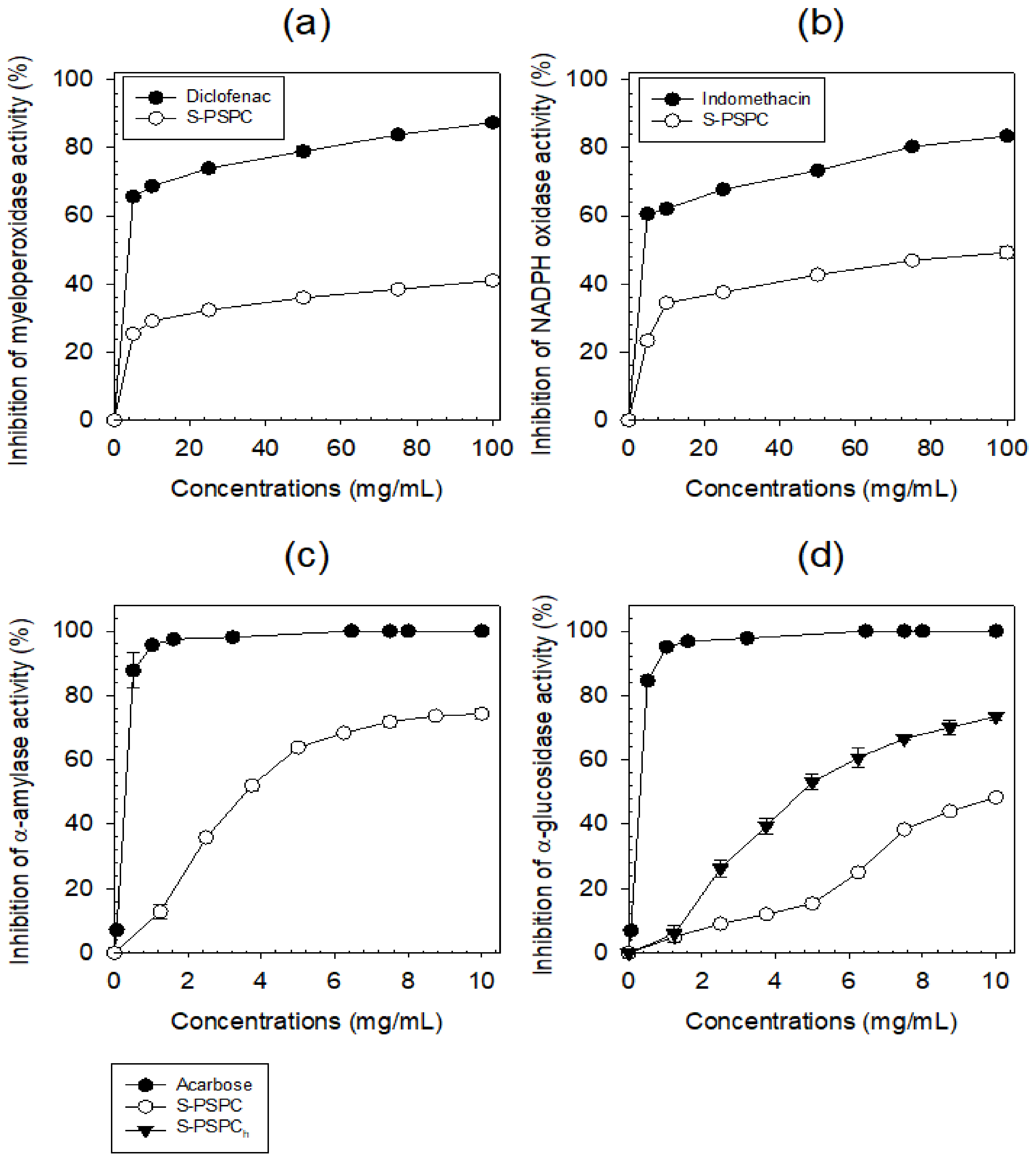
- Anti-inflammation potential of S-PCPC
- Antihyperglymic potential of S-PCPC
Author Contributions
Funding
Institutional Review Board Statement
Conflicts of Interest
References
- Sung, B.; Prasad, S.; Gupta, S.C.; Patchva, S.; Aggarwal, B.B. Regulation of Inflammation-Mediated Chronic Diseases by Botanicals. Adv. Bot. Res. 2012, 62, 58–94. [Google Scholar]
- Yang, X.; Guo, D.; Zhang, J.; Wu, M. Characterization and anti-tumor activity of pollen polysaccharide. Int. Immunopharmacol. 2007, 7, 401–408. [Google Scholar] [CrossRef]
- Ooi, V.E.C.; Liu, F. Immunomodulation and anti-cancer activity of polysaccharide-protein complexes. Curr. Med. Chem. 2000, 7, 715–729. [Google Scholar] [CrossRef] [PubMed]
- Shao, B.M.; Xu, W.; Dai, H.; Tu, P.; Li, Z.; Gao, X.M. A study on the immune receptors for polysaccharides from the roots of Astragalus membranaceus, a Chinese medicinal herb. Biochem. Biophys. Res. Commun. 2004, 320, 1103–1111. [Google Scholar] [CrossRef] [PubMed]
- Huang, D.F.; Tang, Y.F.; Nie, S.P.; Wan, Y.; Xie, M.Y.; Xie, X.M. Effect of phenylethanoid glycosides and polysaccharides from the seed of Plantago asiatica L. on the maturation of murine bone marrow-derived dendritic cells. Eur. J. Pharmacol. 2009, 620, 105–111. [Google Scholar] [CrossRef] [PubMed]
- Samuelsen, A.B.; Paulsen, B.S.; Weld, J.K.; Knutsen, S.H.; Yamada, H. Characterization of a biologically active arabinogalactan from the leaves of Plantago major L. Carbohydr. Polym. 1998, 35, 145–153. [Google Scholar] [CrossRef]
- Hu, J.L.; Nie, S.P.; Li, C.; Xie, M.Y. In vitro fermentation of polysaccharide from the seeds of Plantago asiatica L. by human fecal microbiota. Food Hydrocoll. 2013, 33, 384–392. [Google Scholar] [CrossRef]
- Galisteo, M.; Sanchez, M.; Vera, R.; Gonzalez, M.; Anguera, A.; Duarte, J.; Zarzuelo, A. A diet supplemented with husks of Plantago ovata deduces the development of endothelial dysfunction, hypertension, and obesity by affecting adiponectin and TNF-α in obese zucker rats. J. Nutr. 2005, 135, 2399–2404. [Google Scholar] [CrossRef]
- Biringanine, G.; Vray, B.; Vercruysse, V.; Vanhaelen-Fastre, R.; Vanhaelena, M.; Duez, P. Polysaccharides extracted from the leaves of Plantago palmata Hook.f. induce nitric oxide and tumor necrosis factor-α production by interferon-γ-activated macrophages. Nitric Oxide 2005, 12, 1–8. [Google Scholar] [CrossRef]
- Kardosova, A.; Machova, E. Antioxidant activity of medicinal plant polysaccharides. Fitoterapia 2006, 77, 367–373. [Google Scholar] [CrossRef]
- Djeridane, A. Evaluation du pouvoir antioxydant et de l’inhibition d’enzymes (la Carboxylestérase et l’Acylase) par des extraits phénoliques de dix-neuf plantes médicinales locales. Thèse de Doctorat, Ecole Normale Supérieure De Kouba-Alger, Kouba-Alger, Algeria, 2008. [Google Scholar]
- Dubois, M.; Gilles, K.A.; Hamilton, J.K.; Pebers, P.A.; Smith, F. Colorimetric method for determination of sugar and relayed substances. Anal. Chem. 1956, 28, 350–356. [Google Scholar] [CrossRef]
- Monsigny, M.; Petit, C.; Roche, A.C. Colorimetric determination of neutral sugars by a resorcinol sulfuric acids micromethod. Anal. Biochem. 1988, 175, 525–530. [Google Scholar] [CrossRef]
- Blumenkrantz, N.; Asboe-Hansen, G. New method for quantitative determination of uronic acids. Anal. Biochem. 1973, 54, 484–489. [Google Scholar] [CrossRef]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Singleton, V.L.; Rossi, J.A. Colorimetry of total phenolics with phosphomolybdic–phosphotungstic acid reagents. Am. J. Enol. Vitic. 1965, 16, 144–158. [Google Scholar]
- Pierre, G.; Graber, M.; Rafiliposon, B.A.; Dupuy, C.; Orvain, F.; De Crignis, M.; Maugard, T. Biochemical composition and changes of extracellular polysaccharides (ECPS) produced during microphytobenthic biofilm development (Marennes-Oléron, France). Microb. Ecol. 2011, 63, 157–169. [Google Scholar] [CrossRef] [PubMed]
- Ghoneum, M.; Gollapudi, S. Phagocytosis of Candida albicans by metastatic and non-metastatic human breast cancer cell lines in vitro. Cancer Detect. Prev. 2004, 28, 17–26. [Google Scholar] [CrossRef]
- Tavanti, A.; Campa, D.; Bertozzi, A.; Pardini, G.; Naglik, J.R.; Barale, R.; Senesi, S. Candida albicans isolates with different genomic backgrounds display a differential response to macrophage infection. Microb. Infect. 2006, 8, 791–800. [Google Scholar] [CrossRef]
- Wanikiat, P.; Panthong, A.; Sujayanon, P.; Yoosook, C.; Rossi, A.G.; Reutrakul, V. The anti-inflammatory effects and the inhibition of neutrophil responsiveness by Barleria lupulina and Clinacanthus nutans extracts. J. Ethnopharmacol. 2008, 116, 234–244. [Google Scholar] [CrossRef]
- Meziti, A. Régulation de L’inflammation par les Extraits de Rubus fruticosus et Zizyphus vulgaris. Ph.D. Thesis, Université Ferhat Abbas-Sétif, Sétif, Algeria, 2018. [Google Scholar]
- Boudoukha, C. Etude de Certaines Fonctions du Neutrophile Humain et Leur Modulation par les Extraits de Santolina chamaecyparissus. Ph.D. Thesis, Université Ferhat Abbas-Sétif, Sétif, Algeria, 2018. [Google Scholar]
- Kumar, A.; Lakshman, K.; Jayaveera, K.; Shekar, S.; Swamy, N.; Khan, S.; Velumurga, C. In vitro α-amylase inhibition and antioxidant activities of methanolic extract of Amaranthus caudatus Linn. Oman Med. J. 2011, 26, 166–170. [Google Scholar] [CrossRef] [PubMed]
- Kajaria, D.; Tripathi, J.; Tripathi, Y.B.; Tiwari, S. In-vitro α-amylase and glycosidase inhibitory effect of ethanolic extract of antiasthmatic drug-Shirishadi. J. Adv. Pharm. Technol. Res. 2013, 4, 206–209. [Google Scholar] [CrossRef] [PubMed]
- Bisht, S.; Kant, R.; Kumar, V. α-d-glucosidase inhibitory activity of polysaccharide isolated from Acacia tortilis gum exudate. Int. J. Biol. Macromol. 2013, 59, 214–220. [Google Scholar] [CrossRef]
- Qian, J.Y.; Bai, Y.Y.; Tang, J.; Chen, W. Antioxidation and α-glucosidase inhibitory activities of barley polysaccharides modified with sulfation. LWT-Food Sci. Technol. 2015, 64, 104–111. [Google Scholar] [CrossRef]
- Telagari, M.; Hulltti, K. In-vitro α-amylase and α-glucosidase inhibitory activity of Adiantum caudatum Linn. and Celosia argentea Linn. extracts and fractions. Ind. J. Pharmacol. 2015, 47, 425–429. [Google Scholar]
- Gong, L.; Zhang, H.; Niu, Y.; Chen, L.; Liu, J.; Alaxi, S.; Shang, P.; Yu, W.; Yu, L. A novel alkali extractable polysaccharide from Plantago asiatica L. seeds and its radical-scavenging and bile acid-binding activities. J. Agric. Food Chem. 2015, 63, 569–577. [Google Scholar] [CrossRef] [PubMed]
- Benaoun, F.; Delattre, C.; Boual, Z.; Ursu, A.V.; Vial, C.; Gardarin, C.; Wadouachi, A.; Le Cerf, D.; Varacavoudin, T.; Ould El-Hadj, M.D.; et al. Structural characterization and rheological behavior of a heteroxylan extracted from Plantago notata Lagasca (Plantaginaceae) seeds. Carbohydr. Polym. 2017, 175, 96–104. [Google Scholar] [CrossRef]
- Han, N.; Wang, L.; Song, Z.; Lin, J.; Ye, C.; Liu, Z.; Yin, J. Optimization and antioxidant activity of polysaccharides from Plantago depressa. Int. J. Biol. Macromol. 2016, 93, 644–654. [Google Scholar] [CrossRef]
- Guo, Q.; Cui, S.W.; Wang, Q.; Young, J.C. Fractionation and physicochemical characterization of psyllium gum. Carbohydr. Polym. 2008, 73, 35–43. [Google Scholar] [CrossRef]
- Yin, J.; Lin, H.; Li, J.; Wang, Y.; Cui, S.W.; Nie, S.; Xie, M. Structural characterization of a highly branched polysaccharide from the seeds of Plantago asiatica L. Carbohydr. Polym. 2012, 87, 2416–2424. [Google Scholar] [CrossRef]
- Craeyveld, V.V.; Delcour, J.A.; Christophe, M.C. Extractability and chemical and enzymic degradation of psyllium (Plantago ovata Forsk) seed husk arabinoxylans. Food Chem. 2009, 112, 812–819. [Google Scholar] [CrossRef]
- Lukova, P.; Nikolova, M.; Petit, E.; Elboutachfaiti, R.; Vasileva, T.; Katsarov, P.; Manev, H.; Gardarin, C.; Pierre, G.; Michaud, P.; et al. Prebiotic activity of poly- and oligosaccharides obtained from Plantago major L. leaves. Appl. Sci. 2020, 10, 2648. [Google Scholar] [CrossRef]
- Zha, H.; Wang, Q.; Sun, Y.; Yang, B.; Wang, Z.; Chai, G.; Guan, Y.; Shu, Z.; Lei, X.; Kuang, H. Purification, characterization and immunomodulatory effects of Plantago depressa polysaccharides. Carbohydr. Polym. 2014, 112, 63–72. [Google Scholar] [CrossRef]
- Olennikov, D.N.; Tankhaeva, L.M.; Samuelsen, A.B. Quantitative analysis of polysaccharides from Plantago major leaves using the dreywood method. Chem. Nat. Compd. 2006, 42, 265–268. [Google Scholar] [CrossRef]
- Bichara, L.C.; Alvarez, P.E.; Bimbi, M.V.F.; Vaca, H.; Gervasi, C.; Brandán, S.A. Structural and spectroscopic study of a pectin isolated from citrus peel by using FTIR and FT-Raman spectra and DFT calculations. Infrared Phys. Technol. 2016, 76, 315–327. [Google Scholar] [CrossRef]
- Patel, M.K.; Tanna, B.; Mishra, A.; Jha, B. Physicochemical characterization, antioxidant and anti-proliferative activities of a polysaccharide extracted from psyllium (P. ovata) leaves. Int. J. Biol. Macromol. 2018, 118, 976–987. [Google Scholar] [CrossRef]
- Gorin, A.G. Polysaccharides from Plantago major leaves. I. Analysis of monosaccharide composition of polysaccharide complex. Chem. Abstracts 1966, 64, 8277. [Google Scholar]
- Boual, Z.; Kemassi, A.; Daddi Bouhoun, M.; Michaud, P.; Ould El Hadj, M.D. Isolation and partial characterization of water-soluble polysaccharides from one saharian medicinal plant: Plantago Notata Lagasca. Int. Conf. Environ. Chem. Biol. 2012, 49, 61–65. [Google Scholar]
- Zhong, X.K.; Jin, X.; Lai, F.Y.; Lin, Q.S.; Jiang, J.G. Chemical analysis and antioxidant activities in vitro of polysaccharide extracted from Opuntia ficus indica Mill. cultivated in China. Carbohydr. Polym. 2010, 82, 722–727. [Google Scholar] [CrossRef]
- Fischer, M.H.; Yu, N.; Gray, G.R.; Ralph, J.; Anderson, L.; Marlett, J.A. The gel-forming polysaccharide of psyllium husk (Plantago ovata Forsk). Carbohydr. Res. 2004, 339, 2009–2017. [Google Scholar] [CrossRef] [PubMed]
- Saghir, S.; Iqbal, M.S.; Hussain, M.A.; Koschella, A.; Heinze, T. Structure characterization and carboxymethylation of arabinoxylan isolated from Ispaghula (Plantago ovata) seed husk. Carbohydr. Polym. 2008, 74, 309–317. [Google Scholar] [CrossRef]
- Samuelsen, A.B.; Ingrid, L.; Djahromi, J.M.; Paulsen, B.S.; Wold, J.K.; Knutsen, S.H. Structural features and anti-complementary activity of some heteroxylan polysaccharide fractions from the seeds of Plantago major L. Carbohydr. Polym. 1999, 38, 133–143. [Google Scholar] [CrossRef]
- Addoun, N.; Boual, Z.; Delattre, C.; Ursu, A.V.; Desbrières, J.; Le Cerf, D.; Gardarin, C.; Hentati, F.; Ould El-Hadj, M.D.; Michaud, P.; et al. Structural features and rheological behavior of a water-soluble polysaccharide extracted from the seeds of Plantago ciliata Desf. Int. J. Biol. Macromol. 2020, 155, 1333–1341. [Google Scholar] [CrossRef]
- Blondin, C.; Chaubet, F.; Nardella, A.; Sinquin, C.; Jozefonvicz, J. Relationships between chemical characterstics and anticomplementary activity of fucans. Biomaterials 1996, 17, 597–603. [Google Scholar] [CrossRef]
- Xi, Z.; Chen, W.; Wu, Z.; Wang, Y.; Zeng, P.; Zhao, G.; Li, X.; Sun, L. Anti-complementary activity of flavonoids from Gnaphalium affine D. Don. Food Chem. 2012, 130, 165–170. [Google Scholar] [CrossRef]
- Xie, G.; Schepetkin, I.A.; Quinn, M.T. Immunomodulatory activity of acidic polysaccharides isolated from Tanacetum vulgare L. Int. Immunopharmacol. 2007, 7, 1639–1650. [Google Scholar] [CrossRef]
- Lojek, A.; Denev, P.; Ciz, M.; Vasicek, O.; Kratchanova, M. The effects of biologically active substances in medicinal plants on the metabolic activity of neutrophils. Phytochem. Rev. 2014, 13, 499–510. [Google Scholar] [CrossRef]
- Song, X.; Shen, Q.; Liu, M.; Zhang, C.; Zhang, L.; Ren, Z.; Wang, W.; Dong, Y.; Wang, X.; Zhang, J.; et al. Antioxidant and hepatoprotective effects of intracellular mycelium polysaccharides from Pleurotus geesteranus against alcoholic liver diseases. Int. J. Biol. Macromol. 2018, 114, 979–988. [Google Scholar] [CrossRef]
- Iwalewa, E.O.; McGaw, L.J.; Naidoo, V.; Eloff, J.N. Inflammation: The foundation of diseases and disorders. A review of phytomedicines of South African origin used to treat pain and inflammatory conditions. Afr. J. Biotechnol. 2007, 6, 2868–2885. [Google Scholar]
- Xie, G.; Schepetkin, I.A.; Siemsen, D.W.; Kirpotina, L.N.; Wiley, J.A.; Quinn, M.T. Fractionation and characterization of biologically active polysaccharides from Artemisia tripartita. Phytochem. 2008, 69, 1359–1371. [Google Scholar] [CrossRef]
- Wang, Y.; Liu, D.; Chen, S.; Wang, Y.; Jiang, H.; Yin, H. A new glucomannan from Bletilla striata: Structural and anti-fibrosis effects. Fitoterapia 2014, 92, 72–78. [Google Scholar] [CrossRef] [PubMed]
- Huang, D.; Xia, Q.; Li, F.; Yang, W.; Nie, S.; Xie, M. Attenuation of intestinal inflammation of polysaccharides from the seeds of Plantago asiatica L. as affected by ultrasonication. J. Food Biochem. 2018, 42, e12656–e12663. [Google Scholar] [CrossRef]
- Stipp, M.C.; de Lacerda Bezerra, I.; Corso, C.R.; dos Reis Livero, F.A.; Lomba, L.A.; Caillot, A.R.C.; Zampronio, A.R.; Queiroz-Telles, J.E.; Klassen, G.; Ramos, E.A.S.; et al. Necroptosis mediates the antineoplastic effects of the soluble fraction of polysaccharide from red wine in Walker-256 tumor-bearing rats. Carbohydr. Polym. 2017, 160, 123–133. [Google Scholar] [CrossRef]
- Wasser, S. Medicinal mushrooms as a source of antitumor and immunomodulating polysaccharides. Appl. Microbiol. Biotechnol. 2003, 60, 258–274. [Google Scholar]
- Dey, L.; Attele, A.S.; Yuan, C.S. Alternative therapies for type 2 diabetes. Altern. Med. Rev. 2002, 7, 45–58. [Google Scholar]
- Yu, L.; Xie, Z.; Liu, W. Nutraceutical and health properties of psyllium. In Cereals and Pulses: Nutraceutical Properties and Health Benefits, 1st ed.; Yu, L., Tsao, R., Shahidi, F., Eds.; Wiley Blackwell: Hoboken, NJ, USA, 2012; pp. 1–139. [Google Scholar]
- Poovitha, S.; Parani, M. In vitro and in vivo α-amylase and α-glucosidase inhibiting activities of the protein extracts from two varieties of bitter gourd (Momordica charantia L.). BMC Compl. Alternative Med. 2016, 16, 185–200. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Zhang, B.; Huang, Q.; Fu, X.; Liu, R.H. Microwave-assisted extraction of polysaccharides from Moringa oleifera Lam. leaves: Characterization and hypoglycemic activity. Ind. Crop. Prod. 2017, 100, 1–11. [Google Scholar] [CrossRef]
- Jia, X.; Hu, J.; He, M.; Zhang, Q.; Li, P.; Wan, J.; He, C. α-Glucosidase inhibitory activity and structural characterization of polysaccharide fraction from Rhynchosia minima root. J. Funct. Foods 2017, 28, 76–82. [Google Scholar] [CrossRef]
- Malunga, L.N.; Izydorczyk, M.; Beta, T. Antiglycemic Effect of Water Extractable Arabinoxylan from wheat aleurone and bran. J. Nutr. Metab. 2017, 2017, 5784759. [Google Scholar] [CrossRef]
- Nie, Q.; Hu, J.; Gao, H.; Fan, L.; Chen, H.; Nie, S. Polysaccharide from Plantago asiatica L. attenuates hyperglycemia, hyperlipidemia and affects colon microbiota in type 2 diabetic rats. Food Hydrocoll. 2019, 86, 34–42. [Google Scholar] [CrossRef]
- Gourgue, C.M.P.; Champ, M.M.J.; Lozano, Y.; Delort-Laval, J. Dietary fiber from mango by products: Characterization and hypoglycemic effect determined by in vitro methods. J. Agric. Food Chem. 1992, 40, 1864–1868. [Google Scholar] [CrossRef]
- Annison, G.; Topping, D.L. Nutritional role of resistant starch: Chemical structure vs Physiological function. Annu. Rev. Nutr. 1994, 14, 297–320. [Google Scholar] [CrossRef] [PubMed]
- Madgulkar, A.R.; Rao, M.R.P.; Warrier, D. Characterization of Psyllium (Plantago ovata) polysaccharide and its uses. In Polysaccharides; Ramawat, K.G., Mérillon, J.M., Eds.; Springer International Publishing: Switzerland, 2014; pp. 871–890. [Google Scholar]
- Hussain, M.A.; Muhammad, G.; Jantan, I.; Bukhari, S.N.A. Psyllium arabinoxylan: A versatile biomaterial for potential medicinal and pharmaceutical applications. Polym. Rev. 2015, 56, 1–30. [Google Scholar] [CrossRef]
- Zhang, Z.; Kong, F.; Ni, H.; Mo, Z.; Wan, J.B.; Hua, D.; Yan, C. Structural characterization, α-glucosidase inhibitory and DPPH• scavenging activities of polysaccharides from guava. Carbohydr. Polym. 2016, 144, 106–114. [Google Scholar] [CrossRef] [PubMed]
| Fractions | Carbohydrate (w/w %) | Proteins (w/w %) | Phenols (w/w %) | Ash (w/w %) | ||
|---|---|---|---|---|---|---|
| Total | Neutral | Uronic Acids | ||||
| L-PSPC | 66.6 ± 1.42 | 42.6 ± 1.37 | 24.0 ± 1.12 | 2.30 ± 0.09 | 0.6 ± 0.03 | 3.65 ± 0.08 |
| S-PSPC | 86.5 ± 4.32 | 63.3 ± 3.17 | 7.96 ± 0.39 | 0.35 ± 0.02 | 0 | 5.12 ± 0.24 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Addoun, N.; Boual, Z.; Delattre, C.; Chouana, T.; Gardarin, C.; Dubessay, P.; Benaoun, F.; Addaoud, S.; Ould El Hadj, M.D.; Michaud, P.; et al. Beneficial Health Potential of Algerian Polysaccharides Extracted from Plantago ciliata Desf. (Septentrional Sahara) Leaves and Seeds. Appl. Sci. 2021, 11, 4299. https://doi.org/10.3390/app11094299
Addoun N, Boual Z, Delattre C, Chouana T, Gardarin C, Dubessay P, Benaoun F, Addaoud S, Ould El Hadj MD, Michaud P, et al. Beneficial Health Potential of Algerian Polysaccharides Extracted from Plantago ciliata Desf. (Septentrional Sahara) Leaves and Seeds. Applied Sciences. 2021; 11(9):4299. https://doi.org/10.3390/app11094299
Chicago/Turabian StyleAddoun, Noura, Zakaria Boual, Cédric Delattre, Toufik Chouana, Christine Gardarin, Pascal Dubessay, Fatima Benaoun, Seddik Addaoud, Mohamed Didi Ould El Hadj, Philippe Michaud, and et al. 2021. "Beneficial Health Potential of Algerian Polysaccharides Extracted from Plantago ciliata Desf. (Septentrional Sahara) Leaves and Seeds" Applied Sciences 11, no. 9: 4299. https://doi.org/10.3390/app11094299
APA StyleAddoun, N., Boual, Z., Delattre, C., Chouana, T., Gardarin, C., Dubessay, P., Benaoun, F., Addaoud, S., Ould El Hadj, M. D., Michaud, P., & Pierre, G. (2021). Beneficial Health Potential of Algerian Polysaccharides Extracted from Plantago ciliata Desf. (Septentrional Sahara) Leaves and Seeds. Applied Sciences, 11(9), 4299. https://doi.org/10.3390/app11094299









