Mushroom Nutrition as Preventative Healthcare in Sub-Saharan Africa
Abstract
1. Introduction
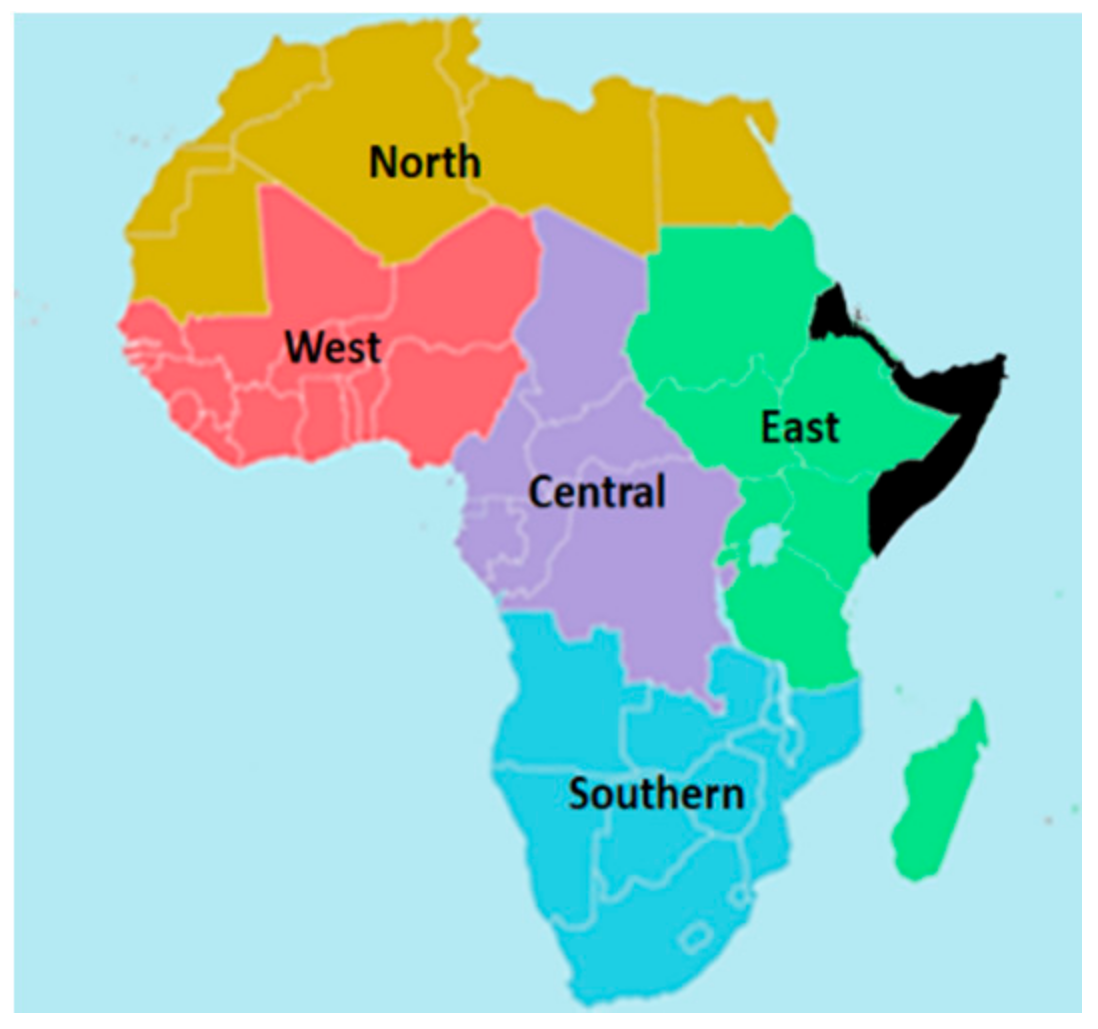
2. The African Diets
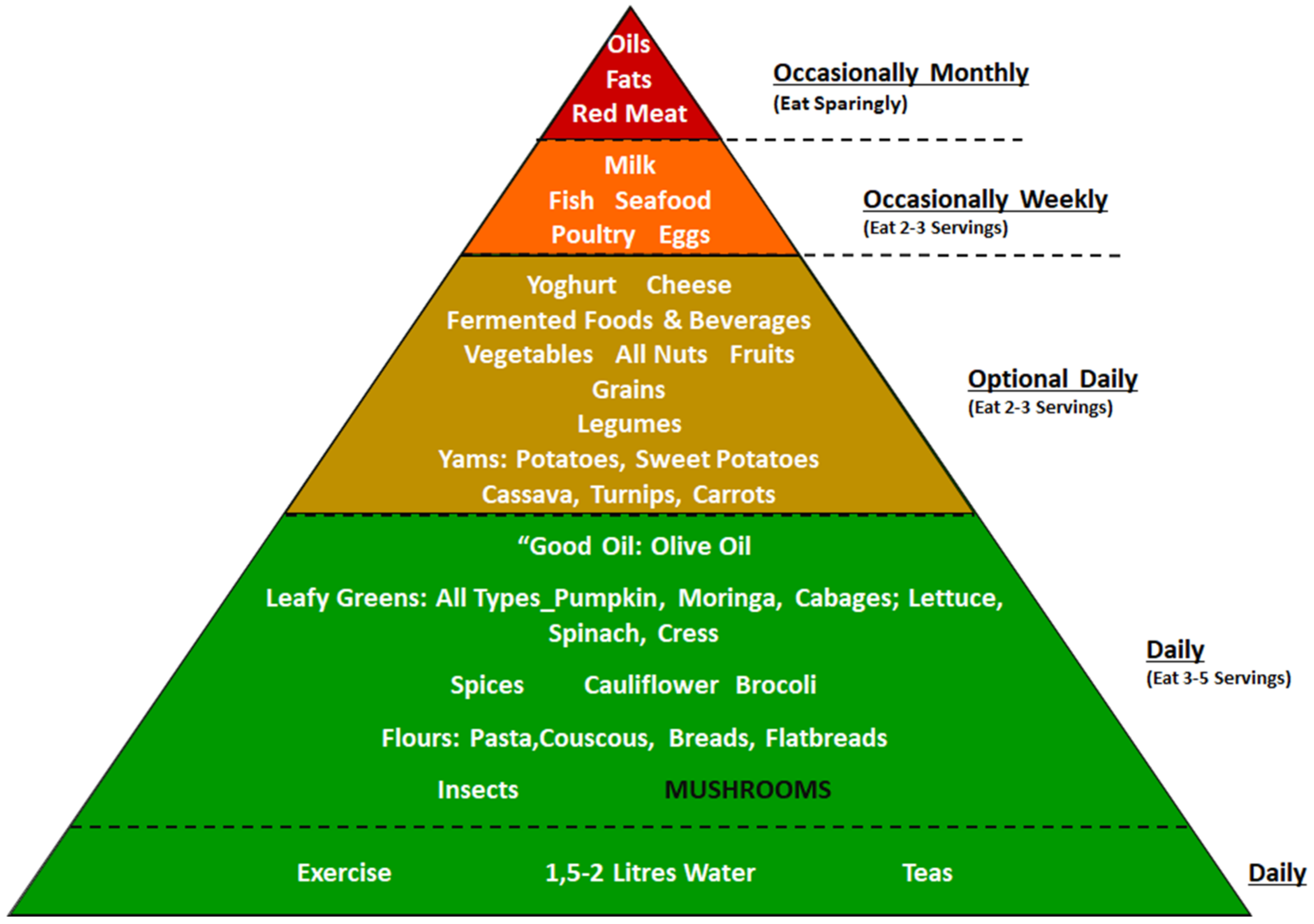
3. African Dietary Guidelines
Role of Mushrooms in the Dietary Guidelines
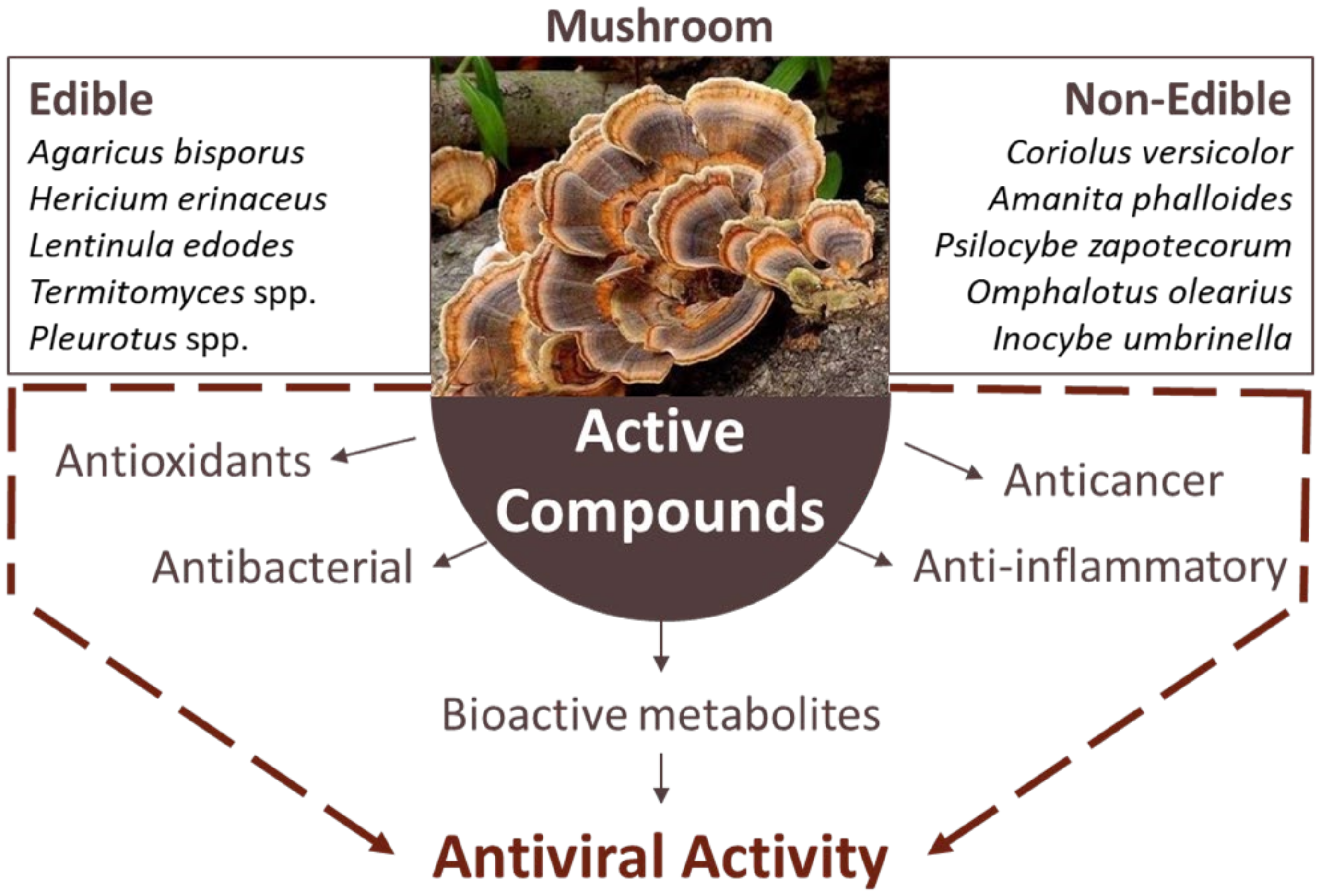
4. African Mushrooms
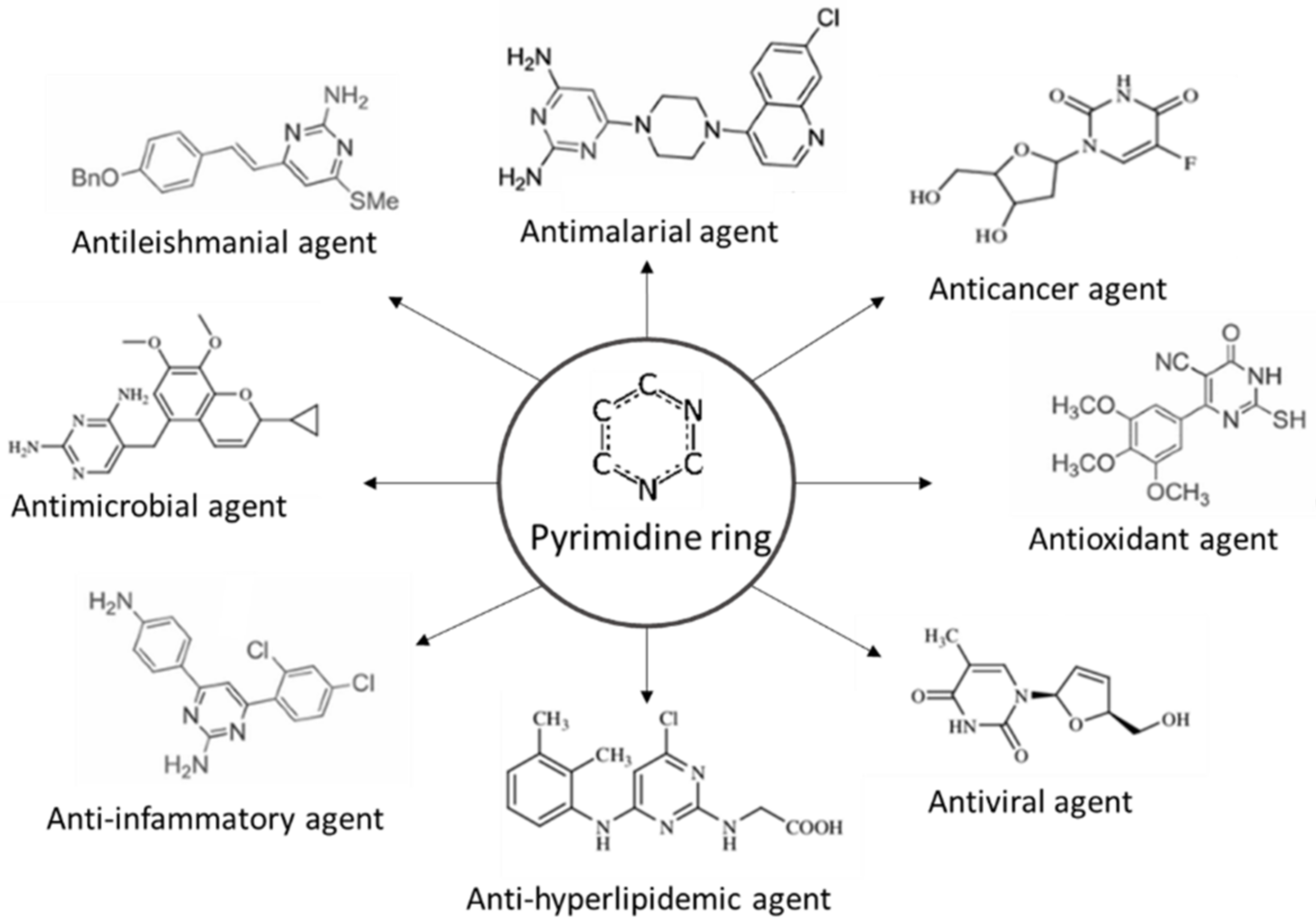
Bioactive Compounds of Mushrooms
5. Anti-Inflammatory Role of Mushrooms
6. The Antiviral Role of Mushrooms
6.1. HIV/AIDS
6.2. Herpes Virus
6.3. Influenza Virus
6.4. Human Papillomaviruses (HPVs)
6.5. The Novel Coronavirus (SARS-CoV-2)
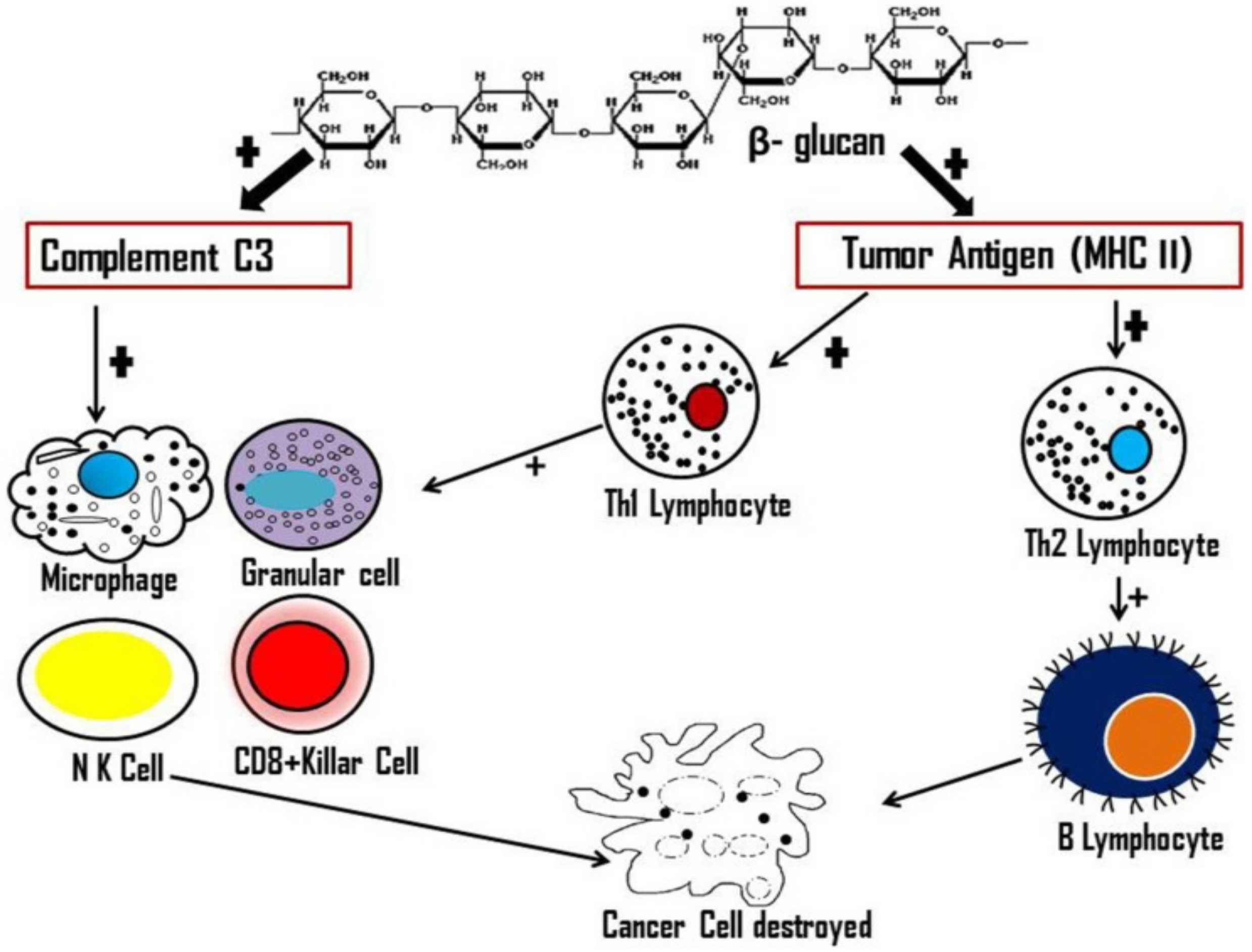
7. Antitumour Activity of Mushrooms
8. Prebiotic Activity of Mushrooms
9. Mushrooms and Neurological Disorders
Role of Mushrooms in Autism
10. Concluding Remarks
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Dodo, K.M. Understanding Africa’s Food Security Challenges. In Food Security in Africa; Mahmoud, B., Ed.; IntechOpen: London, UK, 8 April 2020. [Google Scholar] [CrossRef]
- Raheem, D.; Dayoub, M.; Birech, R.; Nakiyemba, A. The Contribution of Cereal Grains to Food Security and Sustainability in Africa: Potential Application of UAV in Ghana, Nigeria, Uganda, and Namibia. Urban Sci. 2021, 5, 8. [Google Scholar] [CrossRef]
- Gassner, A.; Harris, D.; Mausch, K.; Terheggen, A.; Lopes, C.; Finlayson, R.; Dobie, P. Poverty eradication and food security through agriculture in Africa: Rethinking objectives and entry points. Outlook Agric. 2019, 48, 309–315. [Google Scholar] [CrossRef]
- Pawlak, K.; Kołodziejczak, M. The Role of Agriculture in Ensuring Food Security in Developing Countries: Considerations in the Context of the Problem of Sustainable Food Production. Sustainability 2020, 12, 5488. [Google Scholar] [CrossRef]
- Asongu, S.A.; Odhiambo, N.M. Environmental degradation and inclusive human development in sub-Saharan Africa. Sustain. Dev. 2019, 27, 25–34. [Google Scholar] [CrossRef]
- Forouzanfar, M.H.; Afshin, A.; Alexander, L.T.; Anderson, H.R.; Bhutta, Z.A.; Biryukov, S.; Brauer, M.; Burnett, R.; Cercy, K.; Charlson, F.J.; et al. Global, regional, and national comparative risk assessment of 79 behavioural, environmental and occupational, and metabolic risks or clusters of risks, 1990–2015: A systematic analysis for the Global Burden of Disease Study 2015. Lancet 2016, 388, 1659–1724. [Google Scholar] [CrossRef]
- U.S. Department of Agriculture; U.S. Department of Health and Human Services. Dietary Guidelines for Americans, 2020–2025, 9th ed.; U.S. Department of Agriculture: Washington, DC, USA, December 2020. Available online: https://www.dietaryguidelines.gov/sites/default/files/2020-12/Dietary_Guidelines_for_Americans_2020-2025.pdf (accessed on 14 January 2021).
- Hansen, M.E.B.; Rubel, M.A.; Bailey, A.G.; Ranciaro, A.; Thompson, S.R.; Campbell, M.C.; Beggs, W.; Dave, J.R.; Mokone, G.G.; Mpoloka, S.W.; et al. Population structure of human gut bacteria in a diverse cohort from rural Tanzania and Botswana. Genome Biol. 2019, 20, 16. [Google Scholar] [CrossRef] [PubMed]
- Kearney, J. Food consumption trends and drivers. Philos. Trans. R. Soc. B Biol. Sci. 2010, 365, 2793–2807. [Google Scholar] [CrossRef]
- O’Keefe, S.J.D.; Li, J.V.; Lahti, L.; Ou, J.; Carbonero, F.; Mohammed, K.; Posma, J.M.; Kinross, J.; Wahl, E.; Ruder, E.; et al. Fat, fibre and cancer risk in African Americans and rural Africans. Nat. Commun. 2015. [Google Scholar] [CrossRef] [PubMed]
- Oniang’o, R.K.; Mutuku, J.M.; Malaba, S.J. Contemporary African food habits and their nutritional and health implications. Asia Pac. J. Clin. Nutr. 2003, 12, 331–336. [Google Scholar] [PubMed]
- Nyadanu, D.; Lowor, S.T. Promoting competitiveness of neglected and underutilized crop species: Comparative analysis of nutritional composition of indigenous and exotic leafy and fruit vegetables in Ghana. Genet. Resour. Crop Evol. 2014. [Google Scholar] [CrossRef]
- Imamura, F.; Micha, R.; Khatibzadeh, S.; Fahimi, S.; Shi, P.; Powles, J.; Mozaffarian, D. Dietary quality among men and women in 187 countries in 1990 and 2010: A systematic assessment. Lancet Glob. Health 2015, 3, e132–e142. [Google Scholar] [CrossRef]
- Bell, V.; Barros, A.B.; Fernandes, T.H. Food Fortification in Sub Saharan Africa: Science or Business. In Food and Nutrition Security in Africa; Fernandes, T.H., Ferrão, J., Facknath, S., Eds.; Alcance Publishers: Maputo, Mozambique, 2020; ISBN 978-989-8934-05-5. [Google Scholar]
- EFSA Panel on Dietetic Products Nutrition and Allergies (NDA) Scientific Opinion on establishing Food-Based Dietary Guidelines. EFSA J. 2010, 8, 1460.
- Vapnek, J.; Spreij, M. Perspectives and Guidelines on Food Legislation, with a New Model Food Law. FAO Legis. Study; Chief, Publishing Management Service, Information Division, FAO: Rome, Italy, 2005; Available online: http://www.fao.org/3/a0274e/a0274e.pdf (accessed on 3 January 2021).
- Bell, V.; Ferrão, J.; Fernandes, T. Nutritional Guidelines and Fermented Food Frameworks. Foods 2017, 6, 65. [Google Scholar] [CrossRef]
- Fischer, C.G.; Garnett, T. Plates, Pyramids, Planet: Developments in National Healthy and Sustainable Dietary Guidelines: A State of Play Assessment; FAO: Rome, Italy, 2016; ISBN 9789251092224. Available online: http://www.fao.org/documents/card/en/c/d8dfeaf1-f859-4191-954f-e8e1388cd0b7/ (accessed on 11 December 2020).
- WHO; FAO. Understanding Codex, 5th ed.; FAO: Rome, Italy, 2018; ISBN 978-92-5-130928-5. Available online: http://www.fao.org/3/CA1176EN/ca1176en.pdf (accessed on 13 January 2021).
- Bell, V.; Ferrão, J.; Fernandes, T. Nutrition, Food Safety and Quality in Sub-Saharan Africa. EC Nutr. 2017, 9, 243–255. Available online: https://www.ecronicon.com/ecnu/pdf/ECNU-09-00322.pdf (accessed on 17 December 2020).
- Mingle, C.L.; Darko, G.; Asare-Donkor, N.K.; Borquaye, L.S.; Woode, E. Patterns in protein consumption in Ghanaian cities. Sci. Afr. 2021, 11, e00684. [Google Scholar] [CrossRef]
- Borelli, T.; Hunter, D.; Padulosi, S.; Amaya, N.; Meldrum, G.; de Oliveira Beltrame, D.M.; Samarasinghe, G.; Wasike, V.W.; Güner, B.; Tan, A.; et al. Local Solutions for Sustainable Food Systems: The Contribution of Orphan Crops and Wild Edible Species. Agronomy 2020, 10, 231. [Google Scholar] [CrossRef]
- Ritchie, H.; Roser, M. Environmental Impacts of Food Production. Our World in DataPublished Online at OurWorldInData.org. 2020. Available online: https://ourworldindata.org/environmental-impacts-of-food (accessed on 13 November 2020).
- Vorster, H. Make starchy foods part of most meals: A food-based dietary guideline for South Africa. S. Afr. J. Clin. Nutr. 2013, 26, S28–S35. [Google Scholar]
- Cisse, F. African Starchy Foods, Gastric Emptying, and Starch Digestion in Malian Stunted Children. Ph.D. Thesis, Purdue University, West Lafayette, IN, USA, 2014. Available online: https://docs.lib.purdue.edu/open_access_dissertations/247 (accessed on 15 January 2021).
- Chastanet, M.; Chouin, G.; de Lima, D.; Guindeuil, T. Towards a history of foodways in Africa before the 20th century. Afriques 2014. [Google Scholar] [CrossRef]
- Tanumihardjo, S.A.; Anderson, C.; Kaufer-Horwitz, M.; Bode, L.; Emenaker, N.J.; Haqq, A.M.; Satia, J.A.; Silver, H.J.; Stadler, D.D. Poverty, Obesity, and Malnutrition: An International Perspective Recognizing the Paradox. J. Am. Diet. Assoc. 2007, 107, 1966–1972. [Google Scholar] [CrossRef] [PubMed]
- Ferrão, J.; Bell, V.; Fernandes, T. Food and Beverage Fortification in Africa? EC Nutr. 2017, SI.01, 17–26. [Google Scholar]
- Derbyshire, E.J. Is There Scope for a Novel Mycelium Category of Proteins alongside Animals and Plants? Foods 2020, 9, 1151. [Google Scholar] [CrossRef]
- Agarwal, S.; Fulgoni, V. Nutritional impact of adding a serving of mushrooms to USDA Food Patterns—A dietary modeling analysis. Food Nutr. Res. 2021, 65, 5618. [Google Scholar] [CrossRef] [PubMed]
- Love, P.V. Developing and Assessing the Appropriateness of the Preliminary Food-Based Dietary Guidelines for South Africans. Ph.D. Thesis, University of KwaZulu-Natal, Durban, South Africa, 2002. Available online: https://citeseerx.ist.psu.edu/viewdoc/download?doi=10.1.1.915.8160&rep=rep1&type=pdf (accessed on 14 January 2021).
- Maunder, E.; Steyn, N. A spoonful of sugar helps the medicine go down! S. Afr. J. Clin. Nutr. 2012, 25, 90–93. [Google Scholar] [CrossRef]
- Samorin, G. The oldest representations of hallucinogenic mushrooms in the world (sahara desert, 9000–7000 B.P.). Integration 1992, 2, 69–78. [Google Scholar]
- Boa, E. Wild Edible Fungi A Global Overview of Their Use and Importance to People; FAO: Rome, Italy, 2004. [Google Scholar]
- Goldman, G.B.; Gryzenhout, M. Field Guide To Mushrooms & Other Fungi of South Africa; EBook; Penguin Random House South Africa: Cape Town, South Africa, 2019; ISBN 9781775846604. [Google Scholar]
- Kalač, P. A review of chemical composition and nutritional value of wild-growing and cultivated mushrooms. J. Sci. Food Agric. 2013, 93, 209–218. [Google Scholar] [CrossRef]
- Kinge, T.R.; Goldman, G.; Jacobs, A.; Ndiritu, G.G.; Gryzenhout, M. A first checklist of macrofungi for South Africa. MycoKeys 2020, 63. [Google Scholar] [CrossRef]
- Kasolo, W.; Chemining’wa, G.; Temu, A. Neglected and Underutilized Species (NUS) for Improved Food Security and Resilience to Climate Change: A Contextualized Learning Manual for African Colleges and Universities; ANAFE: Nairobi, Kenya, 2018; ISBN 9789966108104. [Google Scholar]
- Barroetaveña, C.; Toledo, C.V. The Nutritional Benefits of Mushrooms. In Wild Plants, Mushrooms and Nuts; John Wiley & Sons, Ltd.: Chichester, UK, 2016; pp. 65–81. [Google Scholar]
- Wedaralalage, T.C.K.; Jayawardena, R.S.; Hyde, K.D. Hurdles in fungal taxonomy: Effectiveness of recent methods in discriminating taxa. Megataxa 2020, 1, 114–122. [Google Scholar] [CrossRef]
- Aryal, K.P.; Poudel, S.; Chaudhary, R.P.; Chettri, N.; Chaudhary, P.; Ning, W.; Kotru, R. Diversity and use of wild and non-cultivated edible plants in the Western Himalaya. J. Ethnobiol. Ethnomed. 2018, 14, 10. [Google Scholar] [CrossRef] [PubMed]
- Tibuhwa, D. Folk taxonomy and use of mushrooms in communities around Ngorongoro and Serengeti National Park, Tanzania. J. Ethnobiol. Ethnomed. 2012, 8, 36. [Google Scholar] [CrossRef]
- Sitotaw, R.; Lulekal, E.; Abate, D. Ethnomycological study of edible and medicinal mushrooms in Menge District, Asossa Zone, Benshangul Gumuz Region, Ethiopia. J. Ethnobiol. Ethnomed. 2020, 16, 11. [Google Scholar] [CrossRef]
- Jumbam, B.; Haelewaters, D.; Koch, R.; Al, E. A new and unusual species of Hericium (Basidiomycota: Russulales, Hericiaceae) from the Dja Biosphere Reserve, Cameroon. Mycol. Prog. 2019, 18, 1253–1262. [Google Scholar] [CrossRef]
- Kumla, J.; Suwannarach, N.; Sujarit, K.; Penkhrue, W.; Kakumyan, P.; Jatuwong, K.; Vadthanarat, S.; Lumyong, S. Cultivation of Mushrooms and Their Lignocellulolytic Enzyme Production through the Utilization of Agro-Industrial Waste. Molecules 2020, 25, 2811. [Google Scholar] [CrossRef] [PubMed]
- Trappe, J.M.; Claridge, A.W.; Arora, D.; Smit, W.A. Desert Truffles of the African Kalahari: Ecology, Ethnomycology, and Taxonomy. Econ. Bot. 2008, 62, 521–529. [Google Scholar] [CrossRef]
- El Enshasy, H.; Elsayed, E.A.; Aziz, R.; Wadaan, M.A. Mushrooms and Truffles: Historical Biofactories for Complementary Medicine in Africa and in the Middle East. Evid. Based Complement. Altern. Med. 2013, 2013, 1–10. [Google Scholar] [CrossRef]
- Wang, S.; Marcone, M.F. The biochemistry and biological properties of the world’s most expensive underground edible mushroom: Truffles. Food Res. Int. 2011, 44, 2567–2581. [Google Scholar] [CrossRef]
- Morte, A.; Honrubia, M.; Gutiérrez, A. Biotechnology and cultivation of desert truffles. In Mycorrhiza: State of the Art, Genetics and Molecular Biology, Eco-Function, Biotechnology, Eco-Physiology, Structure and Systematics, 3rd ed.; Springer: Cham, Switzerland; New York, NY, USA, 2008; ISBN 9783540788263. [Google Scholar]
- Osarenkhoe, O.O.; John, O.A.; Theophilus, D.A. Ethnomycological Conspectus of West African Mushrooms: An Awareness Document. Adv. Microbiol. 2014, 4, 39–54. [Google Scholar] [CrossRef]
- Oyetayo, O.V. Medicinal uses of mushrooms in Nigeria: Towards full and sustainable exploitation. Afr. J. Tradit. Complement. Altern. Med. 2011, 8, 267–274. [Google Scholar] [CrossRef]
- van Huis, A. Cultural significance of termites in sub-Saharan Africa. J. Ethnobiol. Ethnomed. 2017, 13, 8. [Google Scholar] [CrossRef]
- Mukiibi, J. The nutritional value of some Uganda mushrooms. Acta Hortic. 1973, 171–176. [Google Scholar] [CrossRef]
- Okhuoya, J.A.; Akpaja, E.O.; Osemwegie, O.O.; Oghenekaro, A.O.; Ihayaere, C.A. Nigerian Mushrooms: Underutilized Non-Wood Forest Resources. J. Appl. Sci. Environ. Manag. 2010, 14, 43–54. [Google Scholar] [CrossRef]
- Gumbo, D.J.; Dumas-Johansen, M.; Muir, G.; Boerstler, F.; Xia, Z. Sustainable Management of Miombo Woodlands-Food Security, Nutrition and Wood Energy; FAO: Rome, Italy, 2018; ISBN 978-92-5-130423-5. [Google Scholar]
- Del Kesel, A.; Malaisse, F. Edible Wild Food: Fungi. In How to Live and Survive in Zambezian Open Forest (Miombo Ecoregion); Les Presses Agronomiques de Gembloux: Gembloux, Belguim, 2010; pp. 41–56. Available online: https://www.researchgate.net/publication/313791952_Edible_Wild_Food_Fungi (accessed on 12 February 2021).
- Rammeloo, J.; Walleyn, R. The Edible Fungi of Africa South of the Sahara: A Literature Survey; Ministry of Agriculture, Administration of Agricultural Research, National Botanic Gardens of Belgium: Meise, Belgium, 1993; ISBN 9789072619129.
- Tesfay, T.; Godifey, T.; Mesfin, R.; Kalayu, G. Evaluation of waste paper for cultivation of oyster mushroom (Pleurotus ostreatus) with some added supplementary materials. AMB Express 2020, 10, 15. [Google Scholar] [CrossRef]
- Degreef, J.; Demuynck, L.; Mukandera, A.; Nyirandayambaje, G.; Nzigidahera, B.; De Kesel, A. Wild edible mushrooms, a valuable resource for food security and rural development in Burundi and Rwanda. Biotechnol. Agron. Soc. Environ. 2016, 20. [Google Scholar] [CrossRef]
- Camassola, M. Mushrooms-The Incredible Factory for Enzymes and Metabolites Productions. Ferment. Technol. 2013, 2. [Google Scholar] [CrossRef]
- Gupta, A.; Gupta, R.; Singh, R.L. Microbes and Environment. In Principles and Applications of Environmental Biotechnology for a Sustainable Future; Springer Nature Singapore Private Limited: Singapore, 2017; pp. 43–84. Available online: https://www.springer.com/gp/book/9789811018657 (accessed on 12 December 2020).
- Barros, L.; Cruz, T.; Baptista, P.; Estevinho, L.M.; Ferreira, I.C.F.R. Wild and commercial mushrooms as source of nutrients and nutraceuticals. Food Chem. Toxicol. 2008, 46, 2742–2747. [Google Scholar] [CrossRef] [PubMed]
- Tung, Y.-T.; Pan, C.-H.; Chien, Y.-W.; Huang, H.-Y. Edible Mushrooms: Novel Medicinal Agents to Combat Metabolic Syndrome and Associated Diseases. Curr. Pharm. Des. 2020, 26, 4970–4981. [Google Scholar] [CrossRef] [PubMed]
- Chaturvedi, V.K.; Agarwal, S.; Gupta, K.K.; Ramteke, P.W.; Singh, M.P. Medicinal mushroom: Boon for therapeutic applications. 3 Biotech 2018, 8, 334. [Google Scholar] [CrossRef]
- Dimitrijević, M.; Stankov Jovanović, V.; Cvetković, J.; Mitić, M.; Petrović, G.; Đorđević, A.; Mitić, V. Phenolics, Antioxidant Potentials, and Antimicrobial Activities of Six Wild Boletaceae Mushrooms. Anal. Lett. 2017, 50, 1691–1709. [Google Scholar] [CrossRef]
- Rahi, D.K.; Malik, D. Diversity of Mushrooms and Their Metabolites of Nutraceutical and Therapeutic Significance. J. Mycol. 2016, 2016, 7654123. [Google Scholar] [CrossRef]
- Al-Laith, A.A.A. Antioxidant components and antioxidant/antiradical activities of desert truffle (Tirmania nivea) from various Middle Eastern origins. J. Food Compos. Anal. 2010, 23, 15–22. [Google Scholar] [CrossRef]
- El Enshasy, H. Immunomodulators. In The Mycota: A Comprehensive Treatise on Fungi as Experimental Systems for Basic and Applied Research. Industrial Applications; Hofrichter., M., Ed.; Springer: Berlin/Heidelberg, Germany, 2010; Volume X, pp. 165–194. [Google Scholar]
- Mocan, A.; Fernandes, Â.; Barros, L.; Crişan, G.; Smiljković, M.; Soković, M.; Ferreira, I.C.F.R. Chemical composition and bioactive properties of the wild mushroom: Polyporus squamosus (Huds.) Fr: A study with samples from Romania. In Proceedings of the Food and Function. Food Funct. 2018, 9, 160–170. [Google Scholar] [CrossRef]
- Ruiz-Dueñas, F.J.; Martínez, Á.T. Microbial degradation of lignin: How a bulky recalcitrant polymer is efficiently recycled in nature and how we can take advantage of this. Microb. Biotechnol. 2009, 2, 164–167. [Google Scholar] [CrossRef]
- Nghi, D.H.; Bittner, B.; Kellner, H.; Jehmlich, N.; Ullrich, R.; Pecyna, M.J.; Nousiainen, P.; Sipilä, J.; Huong, L.M.; Hofrichter, M.; et al. The Wood Rot Ascomycete Xylaria polymorpha Produces a Novel GH78 Glycoside Hydrolase That Exhibits α-l-Rhamnosidase and Feruloyl Esterase Activities and Releases Hydroxycinnamic Acids from Lignocelluloses. Appl. Environ. Microbiol. 2012, 78, 4893–4901. [Google Scholar] [CrossRef] [PubMed]
- Hashimoto, K.; Kaneko, S.; Yoshida, M. Extracellular Carbohydrate Esterase from the Basidiomycete Coprinopsis cinerea Released Ferulic and Acetic Acids from Xylan. Biosci. Biotechnol. Biochem. 2010, 74, 1722–1724. [Google Scholar] [CrossRef]
- Andlar, M.; Rezić, T.; Marđetko, N.; Kracher, D.; Ludwig, R.; Šantek, B. Lignocellulose degradation: An overview of fungi and fungal enzymes involved in lignocellulose degradation. Eng. Life Sci. 2018, 18, 768–778. [Google Scholar] [CrossRef] [PubMed]
- Kim, S. Antioxidant Compounds for the Inhibition of Enzymatic Browning by Polyphenol Oxidases in the Fruiting Body Extract of the Edible Mushroom Hericium erinaceus. Foods 2020, 9, 951. [Google Scholar] [CrossRef]
- Qin, M.; Geng, Y.; Lu, Z.; Xu, H.; Shi, J.-S.; Xu, X.; Xu, Z.-H. Anti-Inflammatory Effects of Ethanol Extract of Lion’s Mane Medicinal Mushroom, Hericium erinaceus (Agaricomycetes), in Mice with Ulcerative Colitis. Int. J. Med. Mushrooms 2016, 18, 227–234. [Google Scholar] [CrossRef] [PubMed]
- Abdulla, M.A.; Noor, S.M.; Sabaratnam, V.; Abdullah, N.; Wong, K.H.; Ali, H.M. Effect of culinary-medicinal lion’s mane mushroom, Hericium erinaceus (Bull.: Fr.) Pers. (Aphyllophoromycetideae), on ethanol-induced gastric ulcers in rats. Int. J. Med. Mushrooms 2008, 10, 325–330. [Google Scholar] [CrossRef]
- Imtiaj, A.; Jayasinghe, C.; Lee, G.; Al, E. Vegetative Growth of Four Strains of Hericium erinaceus Collected from Different Habitats. Mycobiology 2008, 36, 88–92. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.; Chen, Q.-J.; Cao, Q.-Q.; Wu, Y.-Y.; Xu, L.-J.; Zhu, M.-J.; Ng, T.-B.; Wang, H.-X.; Zhang, G.-Q. A Laccase with Antiproliferative and HIV-I Reverse Transcriptase Inhibitory Activities from the Mycorrhizal Fungus Agaricus placomyces. J. Biomed. Biotechnol. 2012, 2012, 736472. [Google Scholar] [CrossRef]
- Xu, L.; Wang, H.; Ng, T. A laccase with HIV-1 reverse transcriptase inhibitory activity from the broth of mycelial culture of the mushroom Lentinus tigrinus. J. Biomed. Biotechnol. 2012, 2012, 536725. [Google Scholar] [CrossRef]
- Angelova, M.; Dolashka-Angelova, P.; Ivanova, E.; Serkedjieva, J.; Slokoska, L.; Pashova, S.; Toshkova, R.; Vassilev, S.; Simeonov, I.; Hartmann, H.J.; et al. A novel glycosylated Cu/Zn-containing superoxide dismutase: Production and potential therapeutic effect. Microbiology 2001, 147, 1641–1650. [Google Scholar] [CrossRef]
- Sabotič, J.; Kos, J. Microbial and fungal protease inhibitors—current and potential applications. Appl. Microbiol. Biotechnol. 2012, 93, 1351–1375. [Google Scholar] [CrossRef] [PubMed]
- Perduca, M.; Destefanis, L.; Bovi, M.; Galliano, M.; Munari, F.; Assfalg, M.; Ferrari, F.; Monaco, H.L.; Capaldi, S. Structure and properties of the oyster mushroom (Pleurotus ostreatus) lectin. Glycobiology 2020, 30, 550–562. [Google Scholar] [CrossRef] [PubMed]
- Olombrada, M.; Lázaro-Gorines, R.; López-Rodríguez, J.C.; Martínez-Del-Pozo, Á.; Oñaderra, M.; Maestro-López, M.; Lacadena, J.; Gavilanes, J.G.; García-Ortega, L. Fungal ribotoxins: A review of potential biotechnological applications. Toxins 2017, 9, 71. [Google Scholar] [CrossRef]
- Cruz, A.; Pimentel, L.; Rodríguez-Alcalá, L.M.; Fernandes, T.; Pintado, M. Health Benefits of Edible Mushrooms Focused on Coriolus versicolor: A Review. J. Food Nutr. Res. 2016, 4, 773–781. [Google Scholar]
- Kardideh, B.; Samimi, Z.; Norooznezhad, F.; Kiani, S.; Mansouri, K. Autophagy, cancer and angiogenesis: Where is the link? Cell Biosci. 2019, 9, 65. [Google Scholar] [CrossRef]
- Keller, N.P. Fungal secondary metabolism: Regulation, function and drug discovery. Nat. Rev. Microbiol. 2019, 17, 167–180. [Google Scholar] [CrossRef] [PubMed]
- Vultaggio-Poma, V.; Sarti, A.; Di Virgilio, F. Extracellular ATP: A Feasible Target for Cancer Therapy. Cells 2020, 9, 2496. [Google Scholar] [CrossRef]
- Phan, C.-W.; Wang, J.-K.; Cheah, S.-C.; Naidu, M.; David, P.; Sabaratnam, V. A review on the nucleic acid constituents in mushrooms: Nucleobases, nucleosides and nucleotides. Crit. Rev. Biotechnol. 2018, 38, 762–777. [Google Scholar] [CrossRef]
- Kumar, S.; Narasimhan, B. Therapeutic potential of heterocyclic pyrimidine scaffolds. Chem. Cent. J. 2018, 12, 38. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.; Hwang, I.; Kim, S.; Hong, E.-J.; Jeung, E.-B. Lentinus edodes promotes fat removal in hypercholesterolemic mice. Exp. Ther. Med. 2013, 6, 1409–1413. [Google Scholar] [CrossRef] [PubMed]
- Jonas, D.A.; Elmadfa, I.; Engel, K.-H.; Heller, K.J.; Kozianowski, G.; König, A.; Müller, D.; Narbonne, J.F.; Wackernagel, W.; Kleiner, J. Safety Considerations of DNA in Food. Ann. Nutr. Metab. 2001, 45, 235–254. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.-S.; Yao, G.-S.; Shi, X.-H.; Rehman, S.U.; Xu, Y.; Fu, X.-M.; Zhang, X.-L.; Liu, Y.; Wang, C.-Y. Epigenetic Agents Trigger the Production of Bioactive Nucleoside Derivatives and Bisabolane Sesquiterpenes From the Marine-Derived Fungus Aspergillus versicolor. Front. Microbiol. 2020, 11. [Google Scholar] [CrossRef] [PubMed]
- Obodai, M.; Cleland-Okine, J.; Vowotor, K. Comparative study on the growth and yield of Pleurotus ostreatus mushroom on different lignocellulosic by-products. J. Ind. Microbiol. Biotechnol. 2003, 30, 146–149. [Google Scholar] [CrossRef]
- Chen, L.; Deng, H.; Cui, H.; Fang, J.; Zuo, Z.; Deng, J.; Li, Y.; Wang, X.; Zhao, L. Inflammatory responses and inflammation-associated diseases in organs. Oncotarget 2018, 9, 7204–7218. [Google Scholar] [CrossRef]
- Punchard, N.A.; Whelan, C.J.; Adcock, I. The Journal of Inflammation. J. Inflamm. 2004, 1. [Google Scholar] [CrossRef]
- Minihane, A.M.; Vinoy, S.; Russell, W.R.; Baka, A.; Roche, H.M.; Tuohy, K.M.; Teeling, J.L.; Blaak, E.E.; Fenech, M.; Vauzour, D.; et al. Low-grade inflammation, diet composition and health: Current research evidence and its translation. Br. J. Nutr. 2015, 114, 999–1012. [Google Scholar] [CrossRef]
- Yu, S.; Weaver, V.; Martin, K.; Cantorna, M.T. The effects of whole mushrooms during inflammation. BMC Immunol. 2009, 10, 12. [Google Scholar] [CrossRef]
- Zhang, P.; Sutheerawattananonda, M. Kinetic Models for Glucosamine Production by Acid Hydrolysis of Chitin in Five Mushrooms. Int. J. Chem. Eng. 2020, 2020, 1–8. [Google Scholar] [CrossRef]
- Salazar, J.; Bello, L.; Chávez, M.; Añez, R.; Rojas, J.; Bermúdez, V. Glucosamine for Osteoarthritis: Biological Effects, Clinical Efficacy, and Safety on Glucose Metabolism. Arthritis 2014, 2014, 1–13. [Google Scholar] [CrossRef]
- Steen, E.H.; Wang, X.; Balaji, S.; Butte, M.J.; Bollyky, P.L.; Keswani, S.G. The Role of the Anti-Inflammatory Cytokine Interleukin-10 in Tissue Fibrosis. Adv. Wound Care 2019, 9, 184–198. [Google Scholar] [CrossRef]
- Elkhateeb, W.; Daba, G.; Thomas, P.; Wen, T.-C. Medicinal mushrooms as a new source of natural therapeutic bioactive compounds. Egypt. Pharm. J. 2019, 18, 88–101. [Google Scholar]
- Nie, A.; Chao, Y.; Zhang, X.; Al, E. Phytochemistry and Pharmacological Activities of Wolfiporia cocos (F.A. Wolf) Ryvarden & Gilb. Front. Pharmacol. 2020, 11, 505249. [Google Scholar] [CrossRef]
- Staniszewska, J.; Szymański, M.; Ignatowicz, E. Antitumor and immunomodulatory activity of Inonotus obliquus. Herba Pol. 2017, 63, 48–58. [Google Scholar] [CrossRef]
- Venturella, G.; Ferraro, V.; Cirlincione, F.; Al, E. Medicinal Mushrooms: Bioactive Compounds, Use, and Clinical Trials. Int. J. Mol. Sci. 2021, 22, 634. [Google Scholar] [CrossRef]
- Dicks, L.; Ellinger, S. Effect of the Intake of Oyster Mushrooms (Pleurotus ostreatus) on Cardiometabolic Parameters—A Systematic Review of Clinical Trials. Nutrients 2020, 12, 1134. [Google Scholar] [CrossRef] [PubMed]
- Baeva, E.; Bleha, R.; Lavrova, E.; Al, E. Polysaccharides from Basidiocarps of Cultivating Mushroom Pleurotus ostreatus: Isolation and Structural Characterization. Molecules 2019, 24, 2740. [Google Scholar] [CrossRef] [PubMed]
- Xiao, Z.; Zhou, W.; Zhang, Y. Fungal polysaccharides. Adv. Pharmacol. 2020, 87, 277–299. [Google Scholar] [CrossRef] [PubMed]
- Vetvicka, V.; Vannucci, L.; Sima, P.; Al, E. Beta Glucan: Supplement or Drug? From Laboratory to Clinical Trials. Molecules 2019, 24, 1251. [Google Scholar] [CrossRef]
- Huang, Q.; Li, L.; Chen, H.; Liu, Q.; Wang, Z. GPP (Composition of Ganoderma Lucidum Poly-saccharides and Polyporus Umbellatus Poly-saccharides) Enhances Innate Immune Function in Mice. Nutrients 2019, 11, 1480. [Google Scholar] [CrossRef]
- Habtemariam, S. Trametes versicolor (Synn. Coriolus versicolor) Polysaccharides in Cancer Therapy: Targets and Efficacy. Biomedicines 2020, 8, 135. [Google Scholar] [CrossRef] [PubMed]
- Ferreiro, E.; Pita, I.R.; Mota, S.I.; Valero, J.; Ferreira, N.R.; Fernandes, T.; Calabrese, V.; Fontes-Ribeiro, C.A.; Pereira, F.C.; Rego, A.C. Coriolus versicolor biomass increases dendritic arborization of newly-generated neurons in mouse hippocampal dentate gyrus. Oncotarget 2018, 9, 32929–32942. [Google Scholar] [CrossRef] [PubMed]
- Stephenson, J.; Nutma, E.; van der Valk, P.; Amor, S. Inflammation in CNS neurodegenerative diseases. Immunology 2018, 154, 204–219. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.-W.; Zhang, X.; Huang, W.-J. Role of neuroinflammation in neurodegenerative diseases (Review). Mol. Med. Rep. 2016, 13, 3391–3396. [Google Scholar] [CrossRef]
- Cheah, I.K.; Halliwell, B. Could Ergothioneine Aid in the Treatment of Coronavirus Patients? Antioxidants 2020, 9, 595. [Google Scholar] [CrossRef] [PubMed]
- Borodina, I.; Kenny, L.; McCarthy, C.; Al, E. The biology of ergothioneine, an antioxidant nutraceutical. Nutr. Res. Rev. 2020, 33, 190–217. [Google Scholar] [CrossRef] [PubMed]
- Seo, D.; Choi, C. Antiviral Bioactive Compounds of Mushrooms and Their Antiviral Mechanisms: A Review. Viruses 2021, 13, 350. [Google Scholar] [CrossRef]
- Suwannarach, N.; Kumla, J.; Sujarit, K.; Pattananandecha, T.; Saenjum, C.; Lumyong, S. Natural Bioactive Compounds from Fungi as Potential Candidates for Protease Inhibitors and Immunomodulators to Apply for Coronaviruses. Molecules 2020, 25, 1800. [Google Scholar] [CrossRef] [PubMed]
- McCarty, M.F.; DiNicolantonio, J.J. Nutraceuticals have potential for boosting the type 1 interferon response to RNA viruses including influenza and coronavirus. Prog. Cardiovasc. Dis. 2020, 63, 383–385. [Google Scholar] [CrossRef]
- Zhang, W.; Zhao, Y.; Zhang, F.; Wang, Q.; Li, T.; Liu, Z.; Wang, J.; Qin, Y.; Zhang, X.; Yan, X.; et al. The use of anti-inflammatory drugs in the treatment of people with severe coronavirus disease 2019 (COVID-19): The experience of clinical immunologists from China. Clin. Immunol. 2020, 214, 108393. [Google Scholar] [CrossRef]
- Roy, B.G. Potential of small-molecule fungal metabolites in antiviral chemotherapy. Antivir. Chem. Chemother. 2017, 25, 20–52. [Google Scholar] [CrossRef]
- Geller, A.; Yan, J. Could the Induction of Trained Immunity by β-Glucan Serve as a Defense Against COVID-19? Front. Immunol. 2020, 11. [Google Scholar] [CrossRef]
- Lin, B.; Li, S. Cordyceps as an Herbal Drug. In Herbal Medicine: Biomolecular and Clinical Aspects; Benzie, I., Wachtel-Galor, S., Eds.; CRC Press/Taylor & Francis: Boca Raton, FL, USA, 2011. [Google Scholar]
- Guidry, C.A.; Mansfield, S.A.; Sawyer, R.G.; Cook, C.H. Resistant Pathogens, Fungi, and Viruses. Surg. Clin. N. Am. 2014, 94, 1195–1218. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Wang, S.; Welte, T.; Fang, H.; Chang, G.-J.J.; Born, W.K.; O’Brien, R.L.; Sun, B.; Fujii, H.; Kosuna, K.-I.; Wang, T. Oral Administration of Active Hexose Correlated Compound Enhances Host Resistance to West Nile Encephalitis in Mice. J. Nutr. 2009, 139, 598–602. [Google Scholar] [CrossRef]
- Teplyakova, T.V.; Kosogova, T.A. Antiviral Effect of Agaricomycetes Mushrooms (Review). Int. J. Med. Mushrooms 2016, 18, 375–386. [Google Scholar] [CrossRef] [PubMed]
- Chunchao, H.; Guo, J. A Hypothesis: Supplementation with Mushroom-Derived Active Compound Modulates Immunity and Increases Survival in Response to Influenza Virus (H1N1) Infection. Evid. Based Complement. Altern. Med. 2011, 2011, 1–3. [Google Scholar] [CrossRef] [PubMed]
- He, M.; Su, D.; Liu, Q.; Gao, W.; Kang, Y. Mushroom lectin overcomes hepatitis B virus tolerance via TLR6 signaling. Sci. Rep. 2017, 7, 5814. [Google Scholar] [CrossRef]
- Rodríguez-Valentín, M.; López, S.; Rivera, M.; Ríos-Olivares, E.; Cubano, L.; Boukli, N.M. Naturally Derived Anti-HIV Polysaccharide Peptide (PSP) Triggers a Toll-Like Receptor 4-Dependent Antiviral Immune Response. J. Immunol. Res. 2018, 2018, 1–14. [Google Scholar] [CrossRef]
- Teplyakova, T.V.; Psurtseva, N.V.; Kosogova, T.A.; Mazurkova, N.A.; Khanin, V.A.; Vlasenko, V.A. Antiviral Activity of Polyporoid Mushrooms (Higher Basidiomycetes) from Altai Mountains (Russia). Int. J. Med. Mushrooms 2012, 14, 37–45. [Google Scholar] [CrossRef]
- Foster, M.; Samman, S. Zinc and Regulation of Inflammatory Cytokines: Implications for Cardiometabolic Disease. Nutrients 2012, 4, 676–694. [Google Scholar] [CrossRef]
- Jang, I.-S.; Ko, Y.-H.; Moon, Y.-S.; Sohn, S.-H. Effects of Vitamin C or E on the Pro-inflammatory Cytokines, Heat Shock Protein 70 and Antioxidant Status in Broiler Chicks under Summer Conditions. Asian Australas. J. Anim. Sci. 2014, 27, 749–756. [Google Scholar] [CrossRef]
- Catanzaro, M.; Corsini, E.; Rosini, M.; Racchi, M.; Lanni, C. Immunomodulators Inspired by Nature: A Review on Curcumin and Echinacea. Molecules 2018, 23, 2778. [Google Scholar] [CrossRef]
- Guggenheim, A.G.; Wright, K.M.; Zwickey, H.L. Immune modulation from five major mushrooms: Application to integrative oncology. Integr. Med. 2014, 13, 32–44. [Google Scholar]
- Fernandes, T.; Chaquisse, E.; Ferrão, J. HIV and the Antiviral Role of Mushroom Nutraceuticals. Adv. Image Video Process. 2020, 8, 64–100. [Google Scholar] [CrossRef]
- World Health Organization. Essential Prevention and Care Interventions for Adults and Adolescents Living with HIV in Resource-Limited Settings/Coordinated by Kevin O’Reilly; World Health Organization: Geneva, Switzerland, 2008; ISBN 978-92-4-159670-1. Available online: https://www.who.int/hiv/pub/guidelines/EP/en/ (accessed on 17 January 2021).
- Rose, A.M.; Hall, C.S.; Martinez-Alier, N. Aetiology and management of malnutrition in HIV-positive children. Arch. Dis. Child. 2014, 99, 546–551. [Google Scholar] [CrossRef] [PubMed]
- Maldonado, S.; Fitzgerald-Bocarsly, P. Antifungal Activity of Plasmacytoid Dendritic Cells and the Impact of Chronic HIV Infection. Front. Immunol. 2017, 8. [Google Scholar] [CrossRef] [PubMed]
- Lindequist, U.; Niedermeyer, T.H.J.; Jülich, W.-D. The Pharmacological Potential of Mushrooms. Evid. Based Complement. Altern. Med. 2005, 2, 285–299. [Google Scholar] [CrossRef]
- Guo, W.-L.; Guo, J.-B.; Liu, B.-Y.; Lu, J.-Q.; Chen, M.; Liu, B.; Bai, W.-D.; Rao, P.-F.; Ni, L.; Lv, X.-C. Ganoderic acid A from Ganoderma lucidum ameliorates lipid metabolism and alters gut microbiota composition in hyperlipidemic mice fed a high-fat diet. Food Funct. 2020, 11, 6818–6833. [Google Scholar] [CrossRef]
- Cao, F.-R.; Feng, L.; Ye, L.-H.; Wang, L.-S.; Xiao, B.-X.; Tao, X.; Chang, Q. Ganoderic Acid A Metabolites and Their Metabolic Kinetics. Front. Pharmacol. 2017, 8. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Gallo, R.L. Antimicrobial peptides. Curr. Biol. 2016, 26, R14–R19. [Google Scholar] [CrossRef] [PubMed]
- Florindo, H.F.; Kleiner, R.; Vaskovich-Koubi, D.; Acúrcio, R.C.; Carreira, B.; Yeini, E.; Tiram, G.; Liubomirski, Y.; Satchi-Fainaro, R. Immune-mediated approaches against COVID-19. Nat. Nanotechnol. 2020, 15, 630–645. [Google Scholar] [CrossRef]
- Rouse, B.T.; Sehrawat, S. Immunity and immunopathology to viruses: What decides the outcome? Nat. Rev. Immunol. 2010, 10, 514–526. [Google Scholar] [CrossRef]
- Magden, J.; Kääriäinen, L.; Ahola, T. Inhibitors of virus replication: Recent developments and prospects. Appl. Microbiol. Biotechnol. 2005, 66, 612–621. [Google Scholar] [CrossRef] [PubMed]
- Weber, F. Antiviral Innate Immunity: Introduction. Encycl. Virol. 2021, 577–583. [Google Scholar] [CrossRef]
- Ellan, K.; Thayan, R.; Raman, J.; Hidari, K.I.P.J.; Ismail, N.; Sabaratnam, V. Anti-viral activity of culinary and medicinal mushroom extracts against dengue virus serotype 2: An in-vitro study. BMC Complement. Altern. Med. 2019, 19, 260. [Google Scholar] [CrossRef] [PubMed]
- Lederman, M.M.; Margolis, L. The lymph node in HIV pathogenesis. Semin. Immunol. 2008, 20, 187–195. [Google Scholar] [CrossRef] [PubMed]
- Jeong, Y.-U.; Park, Y.-J. Ergosterol Peroxide from the Medicinal Mushroom Ganoderma lucidum Inhibits Differentiation and Lipid Accumulation of 3T3-L1 Adipocytes. Int. J. Mol. Sci. 2020, 21, 460. [Google Scholar] [CrossRef]
- Cagno, V.; Tintori, C.; Civra, A.; Cavalli, R.; Tiberi, M.; Botta, L.; Brai, A.; Poli, G.; Tapparel, C.; Lembo, D.; et al. Novel broad spectrum virucidal molecules against enveloped viruses. PLoS ONE 2018, 13, e0208333. [Google Scholar] [CrossRef] [PubMed]
- Raut, J.K. Mushroom: A potent source of natural antiviral drugs. Appl. Sci. Technol. Ann. 2020, 1, 81–91. [Google Scholar] [CrossRef]
- James, C.; Harfouche, M.; Welton, N.J.; Turner, K.M.E.; Abu-Raddad, L.J.; Gottlieb, S.L.; Looker, K.J. Herpes simplex virus: Global infection prevalence and incidence estimates, 2016. Bull. World Health Organ. 2020, 98, 315–329. [Google Scholar] [CrossRef]
- Looker, K.J.; Welton, N.J.; Sabin, K.M.; Dalal, S.; Vickerman, P.; Turner, K.M.E.; Boily, M.-C.; Gottlieb, S.L. Global and regional estimates of the contribution of herpes simplex virus type 2 infection to HIV incidence: A population attributable fraction analysis using published epidemiological data. Lancet Infect. Dis. 2020, 20, 240–249. [Google Scholar] [CrossRef]
- Mangold, C.A.; Szpara, M.L. Persistent Infection with Herpes Simplex Virus 1 and Alzheimer’s Disease—A Call to Study How Variability in Both Virus and Host may Impact Disease. Viruses 2019, 11, 966. [Google Scholar] [CrossRef]
- Duarte, L.F.; Farías, M.A.; Álvarez, D.M.; Bueno, S.M.; Riedel, C.A.; González, P.A. Herpes Simplex Virus Type 1 Infection of the Central Nervous System: Insights Into Proposed Interrelationships With Neurodegenerative Disorders. Front. Cell. Neurosci. 2019, 13. [Google Scholar] [CrossRef] [PubMed]
- Thaker, S.K.; Ch’ng, J.; Christofk, H.R. Viral hijacking of cellular metabolism. BMC Biol. 2019, 17, 59. [Google Scholar] [CrossRef]
- Vlasenko, V.; Vlasenko, A. Antiviral activity of fungi of the Novosibirsk Region: Pleurotus ostreatus and P. pulmonarius (Review). BIO Web Conf. 2018, 11, 00044. [Google Scholar] [CrossRef][Green Version]
- Elkhateeb, W.; Elnahas, M.; Thomas, P. Fomes fomentarius and Polyporus squamosus Models of Marvel Medicinal Mushrooms. Biomed. Res. Rev. 2020, 3, 119. [Google Scholar] [CrossRef]
- Eguchi, N.; Fujino, K.; Thanasut, K.; Taharaguchi, M.; Motoi, M.; Motoi, A.; Taharaguchi, S. In vitro Anti-Influenza Virus Activity of Agaricus brasiliensis KA21. Biocontrol Sci. 2017, 22, 171–174. [Google Scholar] [CrossRef]
- Couto, J.S.; Silva, D.P. Coriolus versicolor supplementation in HPV patients. In Proceedings of the 20th European Congress of Obstetrics and Gynaecology, Lisbon, Portugal, 4–8 March 2008; Available online: https://www.mycologyresearch.com/articles/coriolus-versicolor-supplementation-in-hpv-patients (accessed on 15 February 2021).
- Smith, J.A.; Mathew, L.; Gaikwad, A.; Rech, B.; Burney, M.N.; Faro, J.P.; Lucci, J.A.; Bai, Y.; Olsen, R.J.; Byrd, T.T. From Bench to Bedside: Evaluation of AHCC Supplementation to Modulate the Host Immunity to Clear High-Risk Human Papillomavirus Infections. Front. Oncol. 2019, 9. [Google Scholar] [CrossRef] [PubMed]
- Gincheva, D.; Gincheva, V.; Konova, E. Effect of combined therapy polyhexamethylene biguanide and Coriolus-MRL on human papilloma virus (HPV) cervical and vulvar-related lesions. IJMDAT 2020, 3, e220. [Google Scholar] [CrossRef]
- Shahzad, F.; Anderson, D.; Najafzadeh, M. The Antiviral, Anti-Inflammatory Effects of Natural Medicinal Herbs and Mushrooms and SARS-CoV-2 Infection. Nutrients 2020, 12, 2573. [Google Scholar] [CrossRef] [PubMed]
- Kaymakci, M.; Guler, E. Promising Potential Pharmaceuticals from the Genus Cordyceps for COVID-19 Treatment: A Review Study. Bezmialem Sci. 2020, 8, 140–144. [Google Scholar] [CrossRef]
- Hetland, G.; Johnson, E.; Bernardshaw, S.; Grinde, B. Can medicinal mushrooms have prophylactic or therapeutic effect against COVID–19 and its pneumonic superinfection and complicating inflammation? Scand J. Immunol. 2021, 93, e12937. [Google Scholar] [CrossRef]
- Al-jumaili, M.; Al-dulaimi, F.; Ajeel, M. The Role of Ganoderma lucidum Uptake on Some Hematological and Immunological Response in Patients with Coronavirus (COVID-19). Sys. Rev. Pharm. 2020, 11, 537–541. [Google Scholar]
- USA Unsubstantiated Claims for Coronavirus Prevention and Treatment—Carlin Creative Concepts LLC. Federal Trade Commission. Published 2020. Available online: https://www.ftc.gov/system/files/warning-letters/covid-19-letter_to_swro_carlin_creative_concepts_llc_0.pdf (accessed on 23 November 2020).
- Lam, C.; Cheng, L.; Zhou, L.; Al, E. Herb-drug interactions between the medicinal mushrooms Lingzhi and Yunzhi and cytotoxic anticancer drugs: A systematic review. Chin. Med. 2020, 15, 75. [Google Scholar] [CrossRef] [PubMed]
- Ngai, P.H.K.; Ng, T.B. A hemolysin from the mushroom Pleurotus eryngii. Appl. Microbiol. Biotechnol. 2006, 72, 1185–1191. [Google Scholar] [CrossRef]
- Matuszewska, A.; Stefaniuk, D.; Jaszek, M.; Pięt, M.; Zając, A.; Matuszewski, Ł.; Cios, I.; Grąz, M.; Paduch, R.; Bancerz, R. Antitumor potential of new low molecular weight antioxidative preparations from the white rot fungus Cerrena unicolor against human colon cancer cells. Sci. Rep. 2019, 9, 1975. [Google Scholar] [CrossRef] [PubMed]
- Chan, W.Y.; Ng, T.B.; Lam, J.S.Y.; Wong, J.H.; Chu, K.T.; Ngai, P.H.K.; Lam, S.K.; Wang, H.X. The mushroom ribosome-inactivating protein lyophyllin exerts deleterious effects on mouse embryonic development in vitro. Appl. Microbiol. Biotechnol. 2010, 85, 985–993. [Google Scholar] [CrossRef]
- Ivanova, T.S.; Krupodorova, T.A.; Barshteyn, V.Y.; Artamonova, A.B.; Shlyakhovenko, V.A. Anticancer substances of mushroom origin. Exp. Oncol. 2014, 36, 58–66. [Google Scholar] [PubMed]
- Cappadocia, L.; Lima, C.D. Ubiquitin-like Protein Conjugation: Structures, Chemistry, and Mechanism. Chem. Rev. 2018, 118, 889–918. [Google Scholar] [CrossRef]
- Roca-Lema, D.; Martinez-Iglesias, O.; Portela, C.F.d.A.; Rodríguez-Blanco, A.; Valladares-Ayerbes, M.; Díaz-Díaz, A.; Casas-Pais, A.; Prego, C.; Figueroa, A. In Vitro Anti-proliferative and Anti-invasive Effect of Polysaccharide-rich Extracts from Trametes Versicolor and Grifola Frondosa in Colon Cancer Cells. Int. J. Med. Sci. 2019, 16, 231–240. [Google Scholar] [CrossRef]
- Jakopovic, B.; Oršoli’c, N.; Paveli´c, S. Antitumor, Immunomodulatory and Antiangiogenic Efficacy of Medicinal Mushroom Extract Mixtures in Advanced Colorectal Cancer Animal Model. Molecules 2020, 25, 5005. [Google Scholar] [CrossRef]
- Kany, S.; Vollrath, J.T.; Relja, B. Cytokines in Inflammatory Disease. Int. J. Mol. Sci. 2019, 20, 6008. [Google Scholar] [CrossRef] [PubMed]
- Yamanaka, D.; Takatsu, K.; Kimura, M.; Swamydas, M.; Ohnishi, H.; Umeyama, T.; Oyama, F.; Lionakis, M.S.; Ohno, N. Development of a novel β-1,6-glucan–specific detection system using functionally-modified recombinant endo-β-1,6-glucanase. J. Biol. Chem. 2020, 295, 5362–5376. [Google Scholar] [CrossRef] [PubMed]
- Han, B.; Baruah, K.; Cox, E.; Al, E. Structure-Functional Activity Relationship of β-Glucans From the Perspective of Immunomodulation: A Mini-Review. Front. Immunol. 2020, 11, 658. [Google Scholar] [CrossRef]
- Jameel, G.H.; AL-Ezzy, A.I.A.; Mohammed, I.H. Immunomodulatory, Apoptosis Induction and Antitumor Activities of Aqueous and Methanolic Extract of Calvatia Craniiformis in Mice Transfected with Murine Hepatocellular Carcinoma Cells. Open Access Maced. J. Med. Sci. 2018, 6, 1206–1214. [Google Scholar] [CrossRef] [PubMed]
- Bryant, J.M.; Bouchard, M.; Haque, A. Anticancer Activity of Ganoderic Acid DM: Current Status and Future Perspective. J. Clin. Cell. Immunol. 2017, 8. [Google Scholar] [CrossRef]
- Wang, X.-Y.; Zhang, D.; Yin, J.-Y.; Nie, S.-P.; Xie, M.-Y. Recent developments in Hericium erinaceus polysaccharides: Extraction, purification, structural characteristics and biological activities. Crit. Rev. Food Sci. Nutr. 2019, 59, S96–S115. [Google Scholar] [CrossRef]
- Blagodatski, A.; Yatsunskaya, M.; Mikhailova, V.; Tiasto, V.; Kagansky, A.; Katanaev, V.L. Medicinal mushrooms as an attractive new source of natural compounds for future cancer therapy. Oncotarget 2018, 9, 29259–29274. [Google Scholar] [CrossRef]
- Wong, J.H.; Ng, T.B.; Chan, H.H.L.; Liu, Q.; Man, G.C.W.; Zhang, C.Z.; Guan, S.; Ng, C.C.W.; Fang, E.F.; Wang, H.; et al. Mushroom extracts and compounds with suppressive action on breast cancer: Evidence from studies using cultured cancer cells, tumor-bearing animals, and clinical trials. Appl. Microbiol. Biotechnol. 2020, 104, 4675–4703. [Google Scholar] [CrossRef]
- Del Cornò, M.; Gessani, S.; Conti, L. Shaping the Innate Immune Response by Dietary Glucans: Any Role in the Control of Cancer? Cancers 2020, 12, 155. [Google Scholar] [CrossRef]
- Guarino, M.; Altomare, A.; Emerenziani, S.; Di Rosa, C.; Ribolsi, M.; Balestrieri, P.; Iovino, P.; Rocchi, G.; Cicala, M. Mechanisms of Action of Prebiotics and Their Effects on Gastro-Intestinal Disorders in Adults. Nutrients 2020, 12, 1037. [Google Scholar] [CrossRef]
- Bell, V.; Garrine, C.; Ferrão, J.; Calabrese, V.; Fernandes, T. Effects of Nutrition on Neurology. Open Access J. Agric. Res. 2018, 3, 1–6. [Google Scholar] [CrossRef]
- Lordan, C.; Thapa, D.; Ross, R.P.; Cotter, P.D. Potential for enriching next-generation health-promoting gut bacteria through prebiotics and other dietary components. Gut Microbes 2020, 11, 1–20. [Google Scholar] [CrossRef] [PubMed]
- Nowak, R.; Nowacka-Jechalke, N.; Juda, M.; Malm, A. The preliminary study of prebiotic potential of Polish wild mushroom polysaccharides: The stimulation effect on Lactobacillus strains growth. Eur. J. Nutr. 2018, 57, 1511–1521. [Google Scholar] [CrossRef]
- Mitsou, E.; Saxami, G.; Stamoulou, E.; Al, E. Effects of Rich in B-Glucans Edible Mushrooms on Aging Gut Microbiota Characteristics: An In Vitro Study. Molecules 2020, 25, 2806. [Google Scholar] [CrossRef]
- Costa, C.M.d.S.F. Evaluation of the Prebiotic Potential of Coriolus Versicolor-Effect upon the Human Gut Microbiota. Master’s Thesis, Universidade Católica Portuguesa, Porto, Portugal, 2018. Available online: https://repositorio.ucp.pt/bitstream/10400.14/30621/1/TeseMSc_CeliaCosta.pdf (accessed on 17 January 2021).
- Winkler, A.; Mosser, P.; Schmutzhard, E. Neurological disorders in rural Africa: A systematic approach. Trop Doct. 2009, 39, 102–104. [Google Scholar] [CrossRef] [PubMed]
- Dewhurst, F.; Dewhurst, M.; Gray, W. The prevalence of neurological disorders in older people in Tanzania. Acta Neurol. Scand. 2013, 127, 198–207. [Google Scholar] [CrossRef]
- Kausar, S.; Wang, F.; Cui, H. The Role of Mitochondria in Reactive Oxygen Species Generation and Its Implications for Neurodegenerative Diseases. Cells 2018, 7, 274. [Google Scholar] [CrossRef] [PubMed]
- Lourenço, S.C.; Moldão-Martins, M.; Alves, V.D. Antioxidants of Natural Plant Origins: From Sources to Food Industry Applications. Molecules 2019, 24, 4132. [Google Scholar] [CrossRef]
- Ma, Q.; Xing, C.; Long, W.; Wang, H.Y.; Liu, Q.; Wang, R.-F. Impact of microbiota on central nervous system and neurological diseases: The gut-brain axis. J. Neuroinflamm. 2019, 16, 53. [Google Scholar] [CrossRef] [PubMed]
- Suganya, K.; Koo, B. Gut-Brain Axis: Role of Gut Microbiota on Neurological Disorders and How Probiotics/Prebiotics Beneficially Modulate Microbial and Immune Pathways to Improve Brain Functions. Int. J. Mol. Sci. 2020, 21, 7551. [Google Scholar] [CrossRef]
- Feng, L.; Cheah, I.K.-M.; Ng, M.M.-X.; Li, J.; Chan, S.M.; Lim, S.L.; Mahendran, R.; Kua, E.-H.; Halliwell, B. The Association between Mushroom Consumption and Mild Cognitive Impairment: A Community-Based Cross-Sectional Study in Singapore. J. Alzheimer’s Dis. 2019, 68, 197–203. [Google Scholar] [CrossRef] [PubMed]
- Habtemariam, S. The Chemistry, Pharmacology and Therapeutic Potential of the Edible Mushroom Dictyophora indusiata (Vent ex. Pers.) Fischer (Synn. Phallus indusiatus). Biomedicines 2019, 7, 98. [Google Scholar] [CrossRef]
- Li, I.-C.; Lee, L.-Y.; Tzeng, T.-T.; Chen, W.-P.; Chen, Y.-P.; Shiao, Y.-J.; Chen, C.-C. Neurohealth Properties of Hericium erinaceus Mycelia Enriched with Erinacines. Behav. Neurol. 2018, 2018, 1–10. [Google Scholar] [CrossRef]
- Trovato, A.; Pennisi, M.; Crupi, R.; Al, E. Neuroinflammation and Mitochondrial Dysfunction in the Pathogenesis of Alzheimer’s Disease: Modulation by Coriolus Versicolor (Yun-Zhi) Nutritional Mushroom. J. Neurol. Neuromed. 2017, 2, 19–28. Available online: http://www.jneurology.com/articles/pneuroinflammation-and-mitochondrial-dysfunction-in-the-pathogenesis-of-alzheimerrsquos-disease-modulation-by-coriolus-versicolor.pdf (accessed on 17 December 2020).
- Khan, A.; Ikram, M.; Hahm, J.R.; Kim, M.O. Antioxidant and Anti-Inflammatory Effects of Citrus Flavonoid Hesperetin: Special Focus on Neurological Disorders. Antioxidants 2020, 9, 609. [Google Scholar] [CrossRef]
- Novellino, F.; Saccà, V.; Donato, A.; Zaffino, P.; Spadea, M.F.; Vismara, M.; Arcidiacono, B.; Malara, N.; Presta, I.; Donato, G. Innate Immunity: A Common Denominator between Neurodegenerative and Neuropsychiatric Diseases. Int. J. Mol. Sci. 2020, 21, 1115. [Google Scholar] [CrossRef] [PubMed]
- Salinaro, A.T.; Pennisi, M.; Di Paola, R.; Scuto, M.; Crupi, R.; Cambria, M.T.; Ontario, M.L.; Tomasello, M.; Uva, M.; Maiolino, L.; et al. Neuroinflammation and neurohormesis in the pathogenesis of Alzheimer’s disease and Alzheimer-linked pathologies: Modulation by nutritional mushrooms. Immun Ageing. 2018, 15. [Google Scholar] [CrossRef]
- Scuto, M.; Di Mauro, P.; Ontario, M.L.; Amato, C.; Modafferi, S.; Ciavardelli, D.; Trovato Salinaro, A.; Maiolino, L.; Calabrese, V. Nutritional Mushroom Treatment in Meniere’s Disease with Coriolus versicolor: A Rationale for Therapeutic Intervention in Neuroinflammation and Antineurodegeneration. Int. J. Mol. Sci. 2019, 21, 284. [Google Scholar] [CrossRef]
- Kushairi, N.; Phan, C.W.; Sabaratnam, V.; David, P.; Naidu, M. Lion’s Mane Mushroom, Hericium erinaceus (Bull.: Fr.) Pers. Suppresses H2O2-Induced Oxidative Damage and LPS-Induced Inflammation in HT22 Hippocampal Neurons and BV2 Microglia. Antioxidants 2019, 8, 261. [Google Scholar] [CrossRef] [PubMed]
- Williams, J.; McKune, A.J.; Georgousopoulou, E.N.; Kellett, J.; D’Cunha, N.M.; Sergi, D.; Mellor, D.; Naumovski, N. The Effect of L-Theanine Incorporated in a Functional Food Product (Mango Sorbet) on Physiological Responses in Healthy Males: A Pilot Randomised Controlled Trial. Foods 2020, 9, 371. [Google Scholar] [CrossRef] [PubMed]
- Franz, L.; Chambers, N.; von Isenburg, M.; de Vries, P.J. Autism spectrum disorder in sub-saharan africa: A comprehensive scoping review. Autism Res. 2017, 10, 723–749. [Google Scholar] [CrossRef] [PubMed]
- Abdullahi, A.; Farouk, Z.; Imam, A. Common mental disorders in mothers of children attending out-patient malnutrition clinics in rural North-western Nigeria: A cross-sectional study. BMC Public Health 2021, 21, 185. [Google Scholar] [CrossRef] [PubMed]
- Hodges, H.; Fealko, C.; Soares, N. Autism spectrum disorder: Definition, epidemiology, causes, and clinical evaluation. Transl. Pediatr. 2020, 9, S55–S65. [Google Scholar] [CrossRef] [PubMed]
- Ruparelia, K.; Abubakar, A.; Badoe, E.; Bakare, M.; Visser, K.; Chugani, D.C.; Chugani, H.T.; Donald, K.A.; Wilmshurst, J.M.; Shih, A.; et al. Autism Spectrum Disorders in Africa: Current Challenges in Identification, Assessment, and Treatment: A Report on the International Child Neurology Association Meeting on ASD in Africa, Ghana, April 3-5, 2014. J. Child Neurol. 2016, 31, 1018–1026. [Google Scholar] [CrossRef] [PubMed]
- Adaku Chilaka, C.; Mally, A. Mycotoxin Occurrence, Exposure and Health Implications in Infants and Young Children in Sub-Saharan Africa: A Review. Foods 2020, 9, 1585. [Google Scholar] [CrossRef]
- Bell, V.; Ferrão, J.; Chaquisse, E.; Manuel, B. Role of Mushrooms in Autism. Austin J. Nutr. Food Sci. 2019, 6, 1–8. [Google Scholar]
- Chong, P.; Fung, M.-L.; Kah, K.; Al, E. Therapeutic Potential of Hericium erinaceus for Depressive Disorder. Int. J. Mol. Sci. 2020, 21, 163. [Google Scholar] [CrossRef]
- Li, I.-C.; Chang, H.-H.; Lin, C.-H.; Chen, W.-P.; Lu, T.-H.; Lee, L.-Y.; Chen, Y.-W.; Chen, Y.-P.; Chen, C.-C.; Lin, D.P.-C. Prevention of Early Alzheimer’s Disease by Erinacine A-Enriched Hericium erinaceus Mycelia Pilot Double-Blind Placebo-Controlled Study. Front. Aging Neurosci. 2020, 12. [Google Scholar] [CrossRef]




Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fernandes, T.; Garrine, C.; Ferrão, J.; Bell, V.; Varzakas, T. Mushroom Nutrition as Preventative Healthcare in Sub-Saharan Africa. Appl. Sci. 2021, 11, 4221. https://doi.org/10.3390/app11094221
Fernandes T, Garrine C, Ferrão J, Bell V, Varzakas T. Mushroom Nutrition as Preventative Healthcare in Sub-Saharan Africa. Applied Sciences. 2021; 11(9):4221. https://doi.org/10.3390/app11094221
Chicago/Turabian StyleFernandes, Tito, Carmen Garrine, Jorge Ferrão, Victoria Bell, and Theodoros Varzakas. 2021. "Mushroom Nutrition as Preventative Healthcare in Sub-Saharan Africa" Applied Sciences 11, no. 9: 4221. https://doi.org/10.3390/app11094221
APA StyleFernandes, T., Garrine, C., Ferrão, J., Bell, V., & Varzakas, T. (2021). Mushroom Nutrition as Preventative Healthcare in Sub-Saharan Africa. Applied Sciences, 11(9), 4221. https://doi.org/10.3390/app11094221






