Submerged Cultivation of Inonotus obliquus Mycelium Using Statistical Design of Experiments and Mathematical Modeling to Increase Biomass Yield
Abstract
1. Introduction
2. Materials and Methods
2.1. Inoculum Preparation
2.2. Optimization Design of the Nutrient Broth Components
2.3. Broth Variants Cultivation
2.4. Bioreactor Cultivation
2.5. Mycelial Biomass Harvesting
3. Results and Discussion
3.1. Analysis of the Dry Mycelium Response and Model Building
3.2. Predicted Optimum Nutrient Broth for I. obliquus Biomass Production
3.3. Bioreactor Biomass Yield with Predicted Optimum and PDB
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Kidd, P.M. The use of mushroom glucans and proteoglycans in cancer treatment. Altern. Med. Rev. 2000, 5, 4–27. [Google Scholar] [PubMed]
- Jong, S.C.; Donovick, R. Antitumour and antiviral substances from fungi. Adv. Appl. Microbiol. 1989, 34, 183–262. [Google Scholar] [CrossRef] [PubMed]
- Hou, X.; Chen, W. Optimization of extraction process of crude polysaccharides from wild edible BaChu mushroom by response surface methodology. Carbohydr. Polym. 2008, 72, 67–74. [Google Scholar] [CrossRef]
- Hsieh, C.; Tsai, M.J.; Hsu, T.H.; Chang, D.M.; Lo, C.T. Medium optimization for polysaccharide production of Cordyceps sinensis. Appl. Biochem. Biotechnol. 2005, 120, 145–157. [Google Scholar] [CrossRef]
- Jonathan, G.S.; Ayodele, S.; Damilola, A. Optimization of sub-merged culture conditions for biomass production in Pleurotus florida (Mont.) Singer, a Nigerian edible fungus. Afr. J. Biotechnol. 2006, 5, 1464–1469. [Google Scholar]
- Wasser, S.P.; Elisashvili, V.; Tan, K.K. Effects of carbon and nitrogen sources in the medium on Tremella mesenterica Retz:Fr. (Heterobasidiomycetes) growth and polysaccharide production. Int. J. Med. Mushrooms 2003, 5, 49–56. [Google Scholar] [CrossRef]
- Handa, N.; Yamada, T.; Tanaka, R. An unusual lanostane-type triterpenoid, spiroinonotsuoxodiol, and other triterpenoids from Inonotus obliquus. Phytochemistry 2010, 71, 1774–1779. [Google Scholar] [CrossRef] [PubMed]
- Zheng, W.; Zhang, M.; Zhao, Y.; Wang, Y. Accumulation of antioxidant phenolic constituents in submerged cultures of Inonotus obliquus. Bioresour. Technol. 2009, 100, 1327–1335. [Google Scholar] [CrossRef] [PubMed]
- Lee, I.K.; Yun, B.S. Highly oxygenated and unsaturated metabolites providing a diversity of hispidin class antioxidants in the medicinal mushrooms Inonotus and Phellinus. Bioorg. Med. Chem. 2007, 15, 3309–3314. [Google Scholar] [CrossRef] [PubMed]
- Zheng, W.F.; Gu, Q.; Chen, C.F.; Yang, S.Z.; Wei, J.C.; Chu, C.C. Aminophenols and mold-water-extracts affect the accumulation of flavonoids and their antioxidant activity in cultured mycelia of Inonotus obliquus. Mycosystema 2007, 26, 414–426. [Google Scholar]
- Babitskaya, V.G.; Shcherba, V.V.; Lkonnikova, N.V. Melanin complex of the fungus Inonotus obliquus. Appl. Biochem. Microbiol. 2000, 36, 377. [Google Scholar] [CrossRef]
- Yang, S.Z.; Zheng, W.F. Factors affecting the accumulation of hydrolysable tannins in cultured mycelia of Inonotus obliquus. Chin. Tradit. Herb. Drugs 2007, 38, 798–803. [Google Scholar]
- Kang, J.H.; Jang, J.E.; Mishra, S.K.; Lee, H.J.; Nho, C.W.; Shin, D.; Jin, M.; Kim, M.K.; Choi, C.; Oh, S.H. Ergosterol peroxide from Chaga mushroom (Inonotus obliquus) exhibits anti-cancer activity by down-regulation of the β-catenin pathway in colorectal cancer. J. Ethnopharmacol. 2015, 173, 303–312. [Google Scholar] [CrossRef] [PubMed]
- Bai, Y.H.; Feng, Y.Q.; Mao, D.B.; Xu, C. Optimization for betulin production from mycelial culture of Inonotus obliquus by orthogonal design and evaluation of its antioxidant activity. J. Taiwan Inst. Chem. Eng. 2012, 43, 663–669. [Google Scholar] [CrossRef]
- Chen, H.; Xu, X.; Zhu, Y. Optimization of hydroxyl radical scavenging activity of exo-polysaccharides from Inonotus obliquus in submerged fermentation using response surface methodology. J. Microbiol. Biotechnol. 2010, 20, 835–843. [Google Scholar] [PubMed]
- Xiang, C.; Xiang, Q.X.; Ling, H.Z. Submerged culture optimisation for triterpenoids production by Inonotus obliquus using response surface methodology. Adv. Mat. Res. 2012, 560–561, 784–790. [Google Scholar] [CrossRef]
- Kūka, M.; Čakste, I.; Geršebeka, E. Determination of bioactive compounds and mineral substances in Latvian birch and maple saps. Proc. Latv. Acad. Sci. Sect. B Nat. Exact Appl. Sci. 2013, 67, 437–441. [Google Scholar] [CrossRef]
- Cho, N.S.; Shin, Y.S. Optimization of in vitro cultivation of Inonotus obliquus. J. Korean Wood Sci. Technol. 2005, 33, 92–98. [Google Scholar]
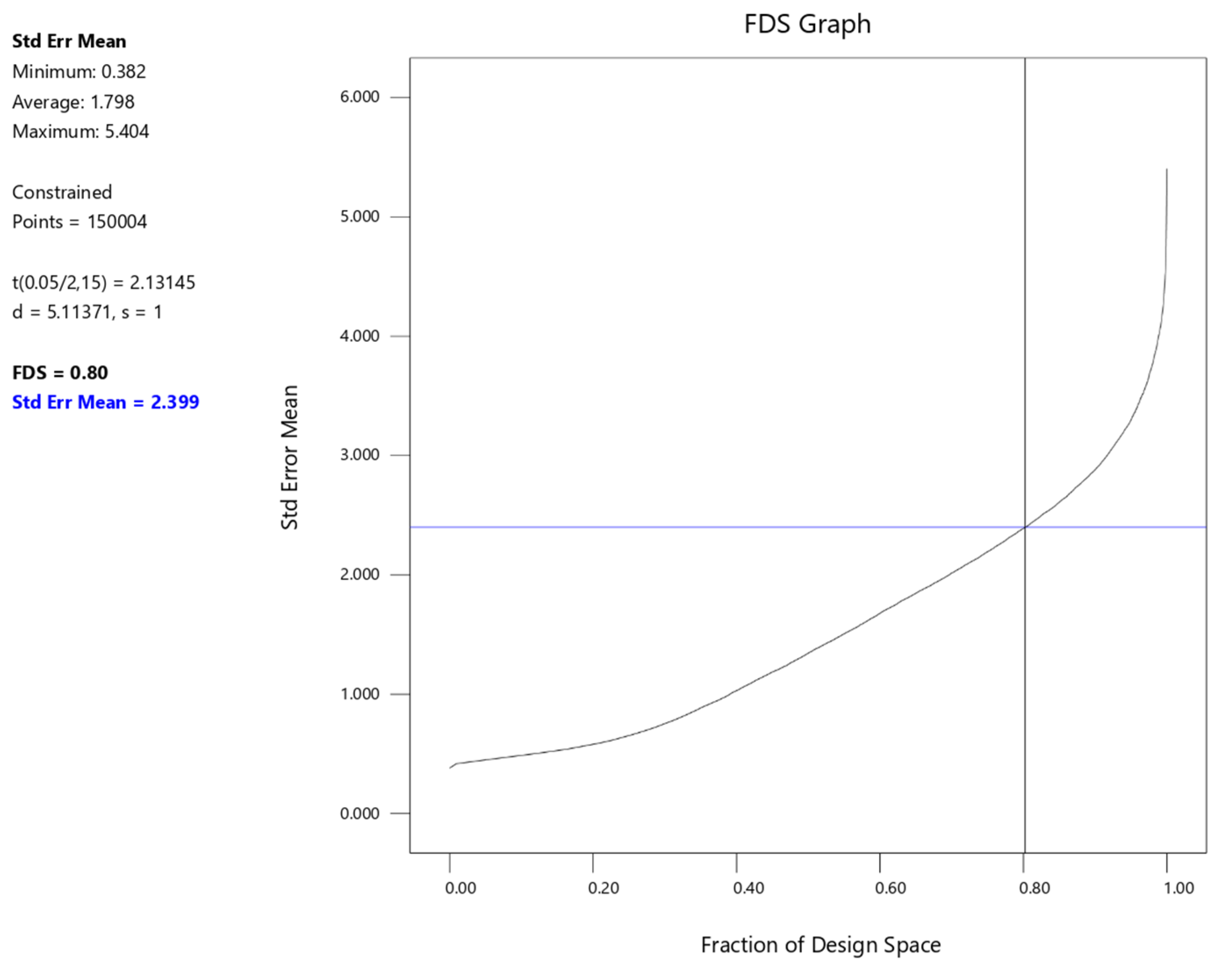
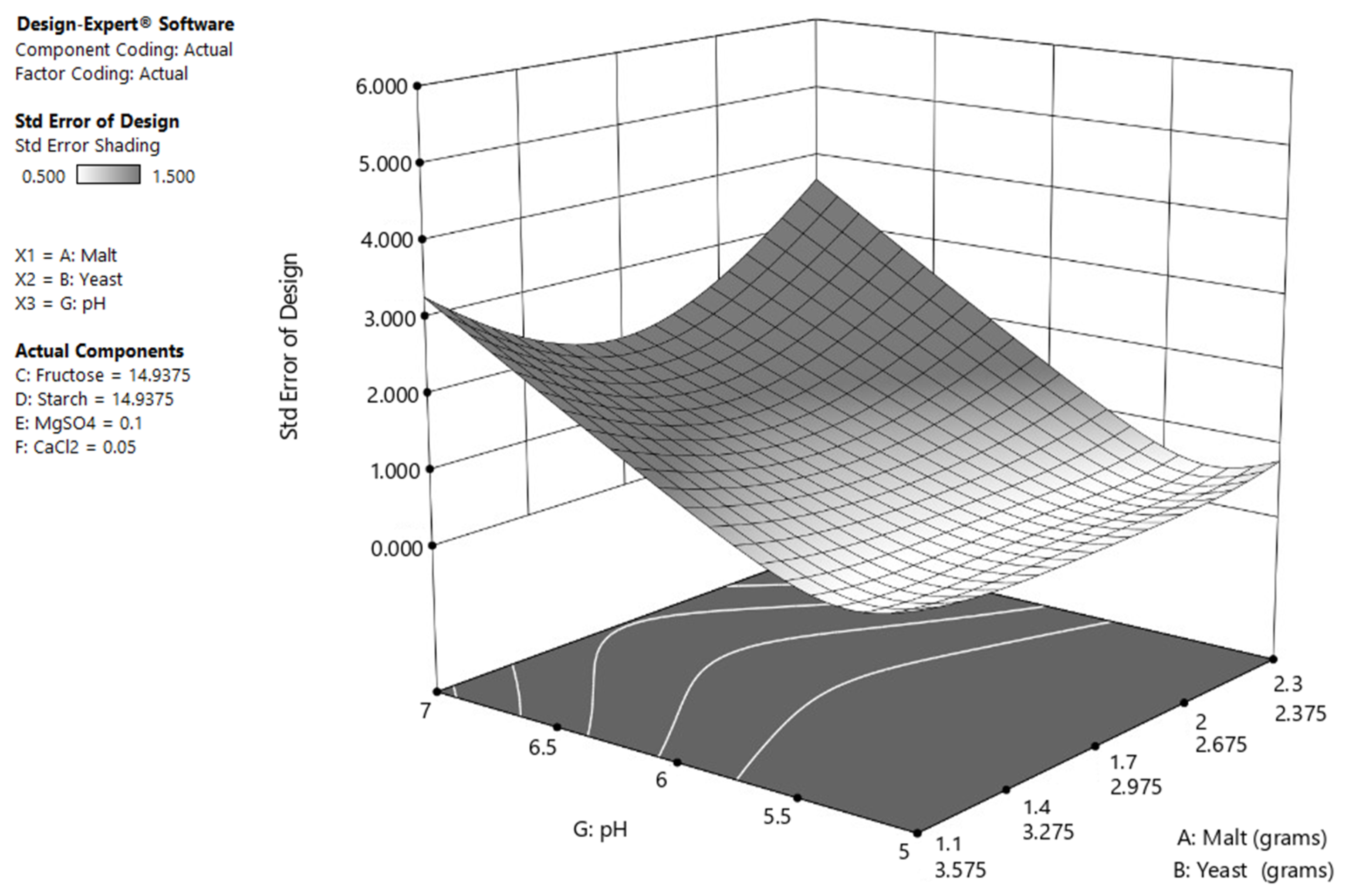
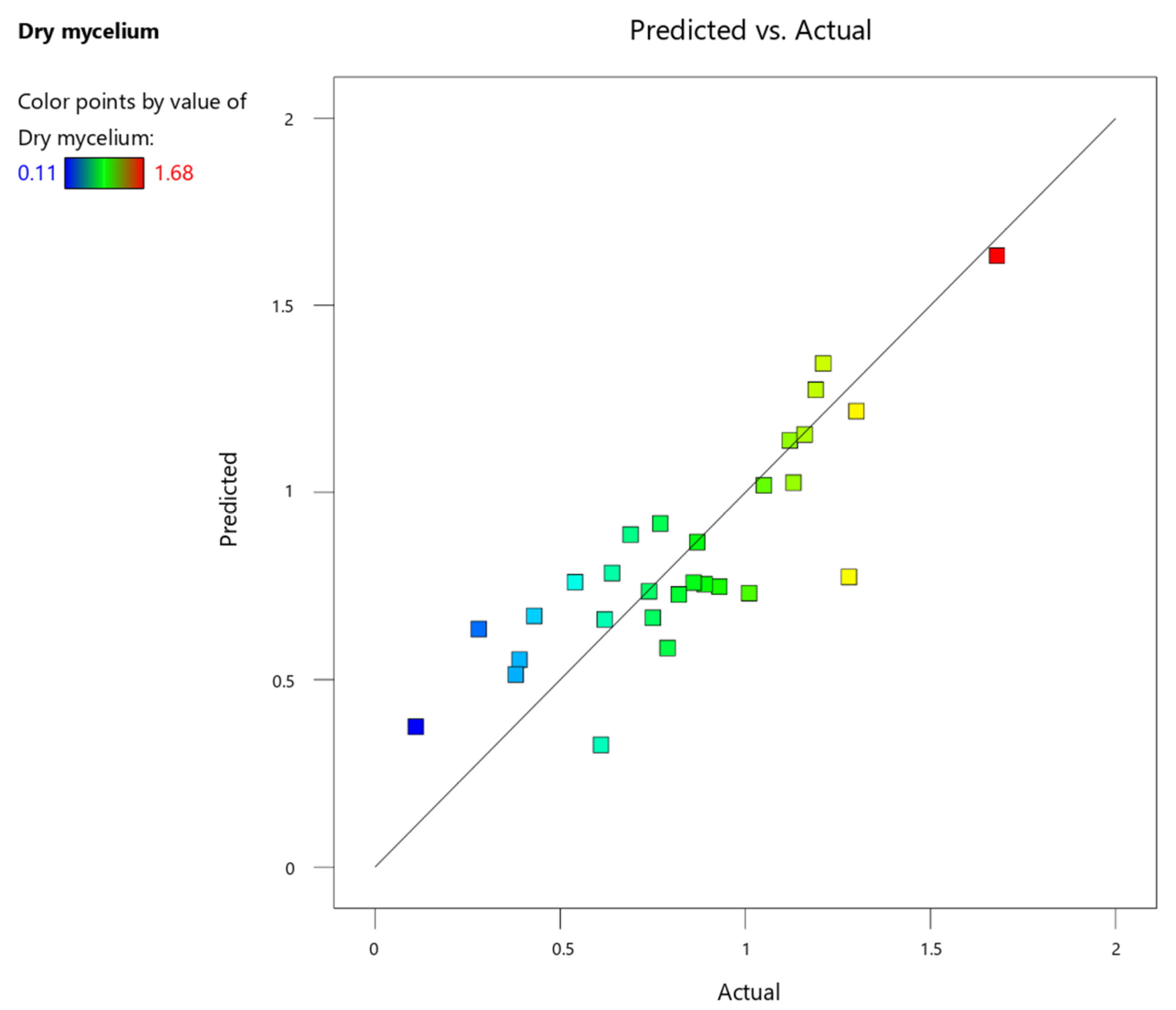


| Factor | Symbol | Low Value (g/L) | High Value (g/L) |
|---|---|---|---|
| Malt extract | A | 1.10 | 2.30 |
| Yeast extract | B | 2.00 | 4.00 |
| Fructose | C | 11.00 | 19.00 |
| Soluble starch | D | 11.00 | 19.00 |
| MgSO4 | E | 0.10 | 0.10 |
| CaCl2 | F | 0.05 | 0.05 |
| pH | G | 4.97 | 5.67 |
| Run | Malt Extract (g/L) | Yeast Extract (g/L) | Fructose (g/L) | Soluble Starch (g/L) | MgSO4 (g/L) | CaCl2 (g/L) | pH |
|---|---|---|---|---|---|---|---|
| 1 | 1.14725 | 2 | 14.12359301 | 17.27915699 | 0.1 | 0.05 | - |
| 2 | 1.1 | 2 | 12.45 | 19 | 0.1 | 0.05 | 5.55 |
| 3 | 1.1 | 2.70875 | 19 | 11.74125 | 0.1 | 0.05 | 5.33 |
| 4 | 2.3 | 2.780591346 | 18.46940865 | 11 | 0.1 | 0.05 | 4.97 |
| 5 | 1.1 | 4 | 13.81857624 | 15.63142376 | 0.1 | 0.05 | 5.55 |
| 6 | 2.3 | 4 | 13.35966064 | 14.89033936 | 0.1 | 0.05 | 5.13 |
| 7 | 1.551558201 | 4 | 17.9984418 | 11 | 0.1 | 0.05 | 5.01 |
| 8 | 1.759389627 | 3.01120613 | 11.48678705 | 18.29261719 | 0.1 | 0.05 | 5.14 |
| 9 | 1.115150333 | 3.119596696 | 15.5597747 | 14.75547827 | 0.1 | 0.05 | 5.1 |
| 10 | 1.93376577 | 4 | 14.60469819 | 14.01153604 | 0.1 | 0.05 | 5.33 |
| 11 | 2.3 | 2 | 19 | 11.25 | 0.1 | 0.05 | 5.21 |
| 12 | 1.1 | 2 | 12.45 | 19 | 0.1 | 0.05 | 5.15 |
| 13 | 1.781034944 | 2 | 15.79942516 | 14.96953989 | 0.1 | 0.05 | 5.05 |
| 14 | 1.1 | 4 | 11 | 18.45 | 0.1 | 0.05 | 5.18 |
| 15 | 1.104378427 | 3.445621573 | 11 | 19 | 0.1 | 0.05 | 5.44 |
| 16 | 1.803890875 | 3.730662742 | 18.01544638 | 11 | 0.1 | 0.05 | 5.39 |
| 17 | 1.654403265 | 3.132022743 | 11.23077409 | 18.5327999 | 0.1 | 0.05 | 5.62 |
| 18 | 2.3 | 4 | 11 | 17.25 | 0.1 | 0.05 | 5.67 |
| 19 | 2.3 | 2 | 11.25 | 19 | 0.1 | 0.05 | 5.54 |
| 20 | 1.1 | 2 | 19 | 12.45 | 0.1 | 0.05 | 4.99 |
| 21 | 2.3 | 2.936180941 | 14.98784285 | 14.32597621 | 0.1 | 0.05 | 5.41 |
| 22 | 2.3 | 2.936180941 | 14.98784285 | 14.32597621 | 0.1 | 0.05 | 5.4 |
| 23 | 2.3 | 2 | 11.25 | 19 | 0.1 | 0.05 | 5.14 |
| 24 | 2.078933462 | 2 | 18.77101112 | 11.70005541 | 0.1 | 0.05 | 5.12 |
| 25 | 1.115150333 | 3.119596696 | 15.5597747 | 14.75547827 | 0.1 | 0.05 | 5.06 |
| 26 | 1.683913211 | 2.065251896 | 15.77432099 | 15.0265139 | 0.1 | 0.05 | 5.32 |
| 27 | 1.654403265 | 3.132022743 | 11.23077409 | 18.5327999 | 0.1 | 0.05 | 5.52 |
| 28 | 1.1 | 3.45 | 19 | 11 | 0.1 | 0.05 | 5.22 |
| 29 | 1.759389627 | 3.01120613 | 11.48678705 | 18.29261719 | 0.1 | 0.05 | 5.15 |
| 30 | 1.683913211 | 2.065251896 | 15.77432099 | 15.0265139 | 0.1 | 0.05 | 5.29 |
| Run Order | Actual Value (g/500 mL) | Predicted Value (g/500 mL) | Residual | Leverage | Internally Studentized Residuals | Externally Studentized Residuals | Cook’s Distance |
|---|---|---|---|---|---|---|---|
| 2 | 0.8200 | 0.7275 | 0.0925 | 0.696 | 0.647 | 0.634 | 0.068 |
| 3 | 1.16 | 1.15 | 0.0052 | 0.577 | 0.031 | 0.030 | 0.000 |
| 4 | 0.7900 | 0.5846 | 0.2054 | 0.665 | 1.367 | 1.412 | 0.265 |
| 5 | 1.05 | 1.02 | 0.0309 | 0.736 | 0.232 | 0.225 | 0.011 |
| 6 | 0.1100 | 0.3745 | −0.2645 | 0.711 | −1.894 | −2.098 | 0.630 |
| 7 | 0.7500 | 0.6654 | 0.0846 | 0.664 | 0.562 | 0.549 | 0.045 |
| 8 | 0.9300 | 0.7486 | 0.1814 | 0.309 | 0.841 | 0.832 | 0.023 |
| 9 | 0.3900 | 0.5534 | −0.1634 | 0.299 | −0.752 | −0.741 | 0.017 |
| 10 | 0.8700 | 0.8672 | 0.0028 | 0.260 | 0.013 | 0.012 | 0.000 |
| 11 | 0.2800 | 0.6352 | −0.3552 | 0.504 | −1.944 | −2.171 | 0.275 |
| 12 | 0.6200 | 0.6609 | −0.0409 | 0.703 | −0.289 | −0.280 | 0.014 |
| 13 | 0.4300 | 0.6702 | −0.2402 | 0.349 | −1.147 | −1.160 | 0.050 |
| 14 | 0.6100 | 0.3260 | 0.2840 | 0.706 | 2.017 | 2.282 | 0.696 |
| 15 | 0.6900 | 0.8875 | −0.1975 | 0.329 | −0.929 | −0.924 | 0.030 |
| 16 | 1.19 | 1.27 | −0.0845 | 0.595 | −0.511 | −0.498 | 0.027 |
| 17 | 1.21 | 1.34 | −0.1346 | 0.354 | −0.645 | −0.632 | 0.016 |
| 18 | 1.68 | 1.63 | 0.0469 | 0.728 | 0.346 | 0.336 | 0.023 |
| 19 | 0.7700 | 0.9171 | −0.1471 | 0.691 | −1.020 | −1.022 | 0.167 |
| 20 | 1.13 | 1.03 | 0.1040 | 0.605 | 0.637 | 0.624 | 0.044 |
| 21 | 0.6400 | 0.7846 | −0.1446 | 0.331 | −0.682 | −0.669 | 0.016 |
| 22 | 1.28 | 0.7742 | 0.5058 | 0.323 | 2.368 | 2.892 | 0.191 |
| 23 | 0.8900 | 0.7551 | 0.1349 | 0.718 | 0.978 | 0.977 | 0.174 |
| 24 | 0.8600 | 0.7589 | 0.1011 | 0.311 | 0.469 | 0.457 | 0.007 |
| 25 | 0.3800 | 0.5139 | −0.1339 | 0.338 | −0.634 | −0.621 | 0.015 |
| 26 | 0.7400 | 0.7358 | 0.0042 | 0.276 | 0.019 | 0.018 | 0.000 |
| 27 | 1.30 | 1.22 | 0.0826 | 0.269 | 0.372 | 0.361 | 0.004 |
| 28 | 1.12 | 1.14 | −0.0187 | 0.400 | −0.093 | −0.090 | 0.000 |
| 29 | 0.5400 | 0.7604 | −0.2204 | 0.300 | −1.015 | −1.016 | 0.032 |
| 30 | 1.01 | 0.7308 | 0.2792 | 0.254 | 1.246 | 1.271 | 0.038 |
| Source | Sum of Squares | df | Mean Square | F-Value | p-Value |
|---|---|---|---|---|---|
| Model | 2.42 | 13 | 0.1860 | 2.76 | 0.0316 |
| (1) Linear Mixture | 0.0752 | 3 | 0.0251 | 0.3721 | 0.7743 |
| AB | 0.1874 | 1 | 0.1874 | 2.78 | 0.1161 |
| AC | 0.0859 | 1 | 0.0859 | 1.28 | 0.2764 |
| AD | 0.1174 | 1 | 0.1174 | 1.74 | 0.2066 |
| AG | 0.0070 | 1 | 0.0070 | 0.1038 | 0.7518 |
| BC | 0.0356 | 1 | 0.0356 | 0.5291 | 0.4782 |
| BD | 0.0244 | 1 | 0.0244 | 0.3629 | 0.5559 |
| BG | 0.4574 | 1 | 0.4574 | 6.79 | 0.0199 |
| CD | 0.2314 | 1 | 0.2314 | 3.44 | 0.0836 |
| CG | 0.0031 | 1 | 0.0031 | 0.0456 | 0.8338 |
| DG | 0.0075 | 1 | 0.0075 | 0.1108 | 0.7439 |
| Residual | 1.01 | 15 | 0.0674 | ||
| Cor Total | 3.43 | 28 |
| Factor | Optimum Value (g/L) |
|---|---|
| Malt extract | 2.15299 |
| Yeast extract | 3.99296 |
| Fructose | 11.0041 |
| Soluble starch | 17.40 |
| MgSO4 | 0.10 |
| CaCl2 | 0.05 |
| Analysis | Predicted Mean | Predicted Median | SD | n | SE Prediction | 95% PI Low | Data Mean | 95% PI High |
|---|---|---|---|---|---|---|---|---|
| Dry mycelium | 2.34028 | 2.34028 | 0.259524 | 1 | 0.432599 | 1.41822 | 2.185 | 3.26234 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Petre, A.; Ene, M.; Vamanu, E. Submerged Cultivation of Inonotus obliquus Mycelium Using Statistical Design of Experiments and Mathematical Modeling to Increase Biomass Yield. Appl. Sci. 2021, 11, 4104. https://doi.org/10.3390/app11094104
Petre A, Ene M, Vamanu E. Submerged Cultivation of Inonotus obliquus Mycelium Using Statistical Design of Experiments and Mathematical Modeling to Increase Biomass Yield. Applied Sciences. 2021; 11(9):4104. https://doi.org/10.3390/app11094104
Chicago/Turabian StylePetre, Alexandru, Mihaela Ene, and Emanuel Vamanu. 2021. "Submerged Cultivation of Inonotus obliquus Mycelium Using Statistical Design of Experiments and Mathematical Modeling to Increase Biomass Yield" Applied Sciences 11, no. 9: 4104. https://doi.org/10.3390/app11094104
APA StylePetre, A., Ene, M., & Vamanu, E. (2021). Submerged Cultivation of Inonotus obliquus Mycelium Using Statistical Design of Experiments and Mathematical Modeling to Increase Biomass Yield. Applied Sciences, 11(9), 4104. https://doi.org/10.3390/app11094104






