Evaluation of Multivariate Biomarker Indexes Application in Ecotoxicity Tests with Marine Diatoms Exposed to Emerging Contaminants
Abstract
Featured Application
Abstract
1. Introduction
2. Materials and Methods
2.1. Diatom Exposure Trials and Biomarker Analysis
2.2. Multivariate Index Calculation
2.2.1. Integrated Biomarker Response (IBR) Index
2.2.2. Principal Component Analysis (PCA)-Based Index
2.2.3. Bioeffect Assessment Index (BAI)
- Values > 3rd quartile: 40
- Values between 3rd and 2nd quartiles: 30
- Values between 2nd and 1st quartiles: 20
- Values < 1st quartile: 10
2.3. Statistical Analysis
3. Results
3.1. Diatom Growth and Ecotoxicological Parameters (IC50)
3.2. Oxidative Stress Biomarkers
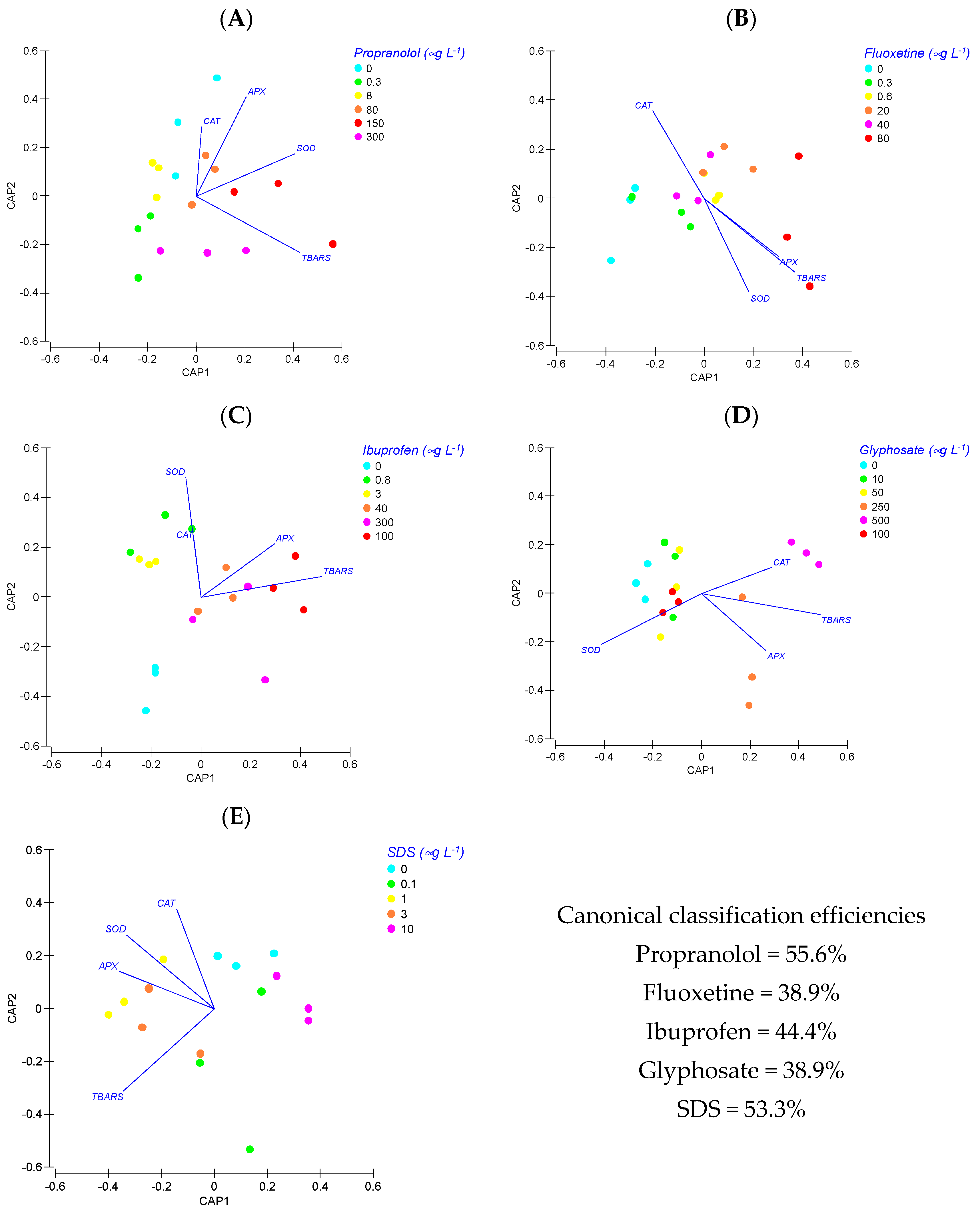
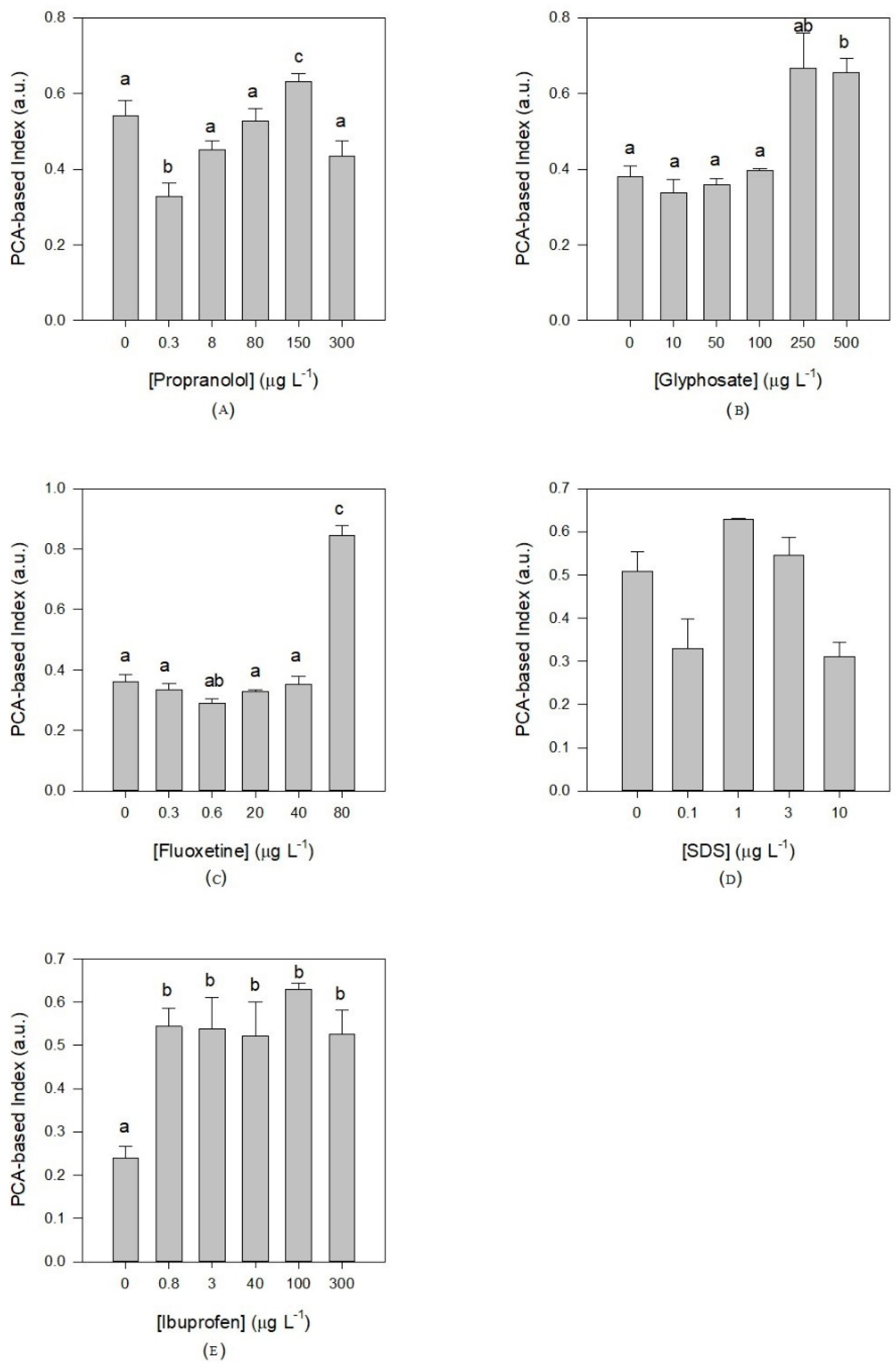
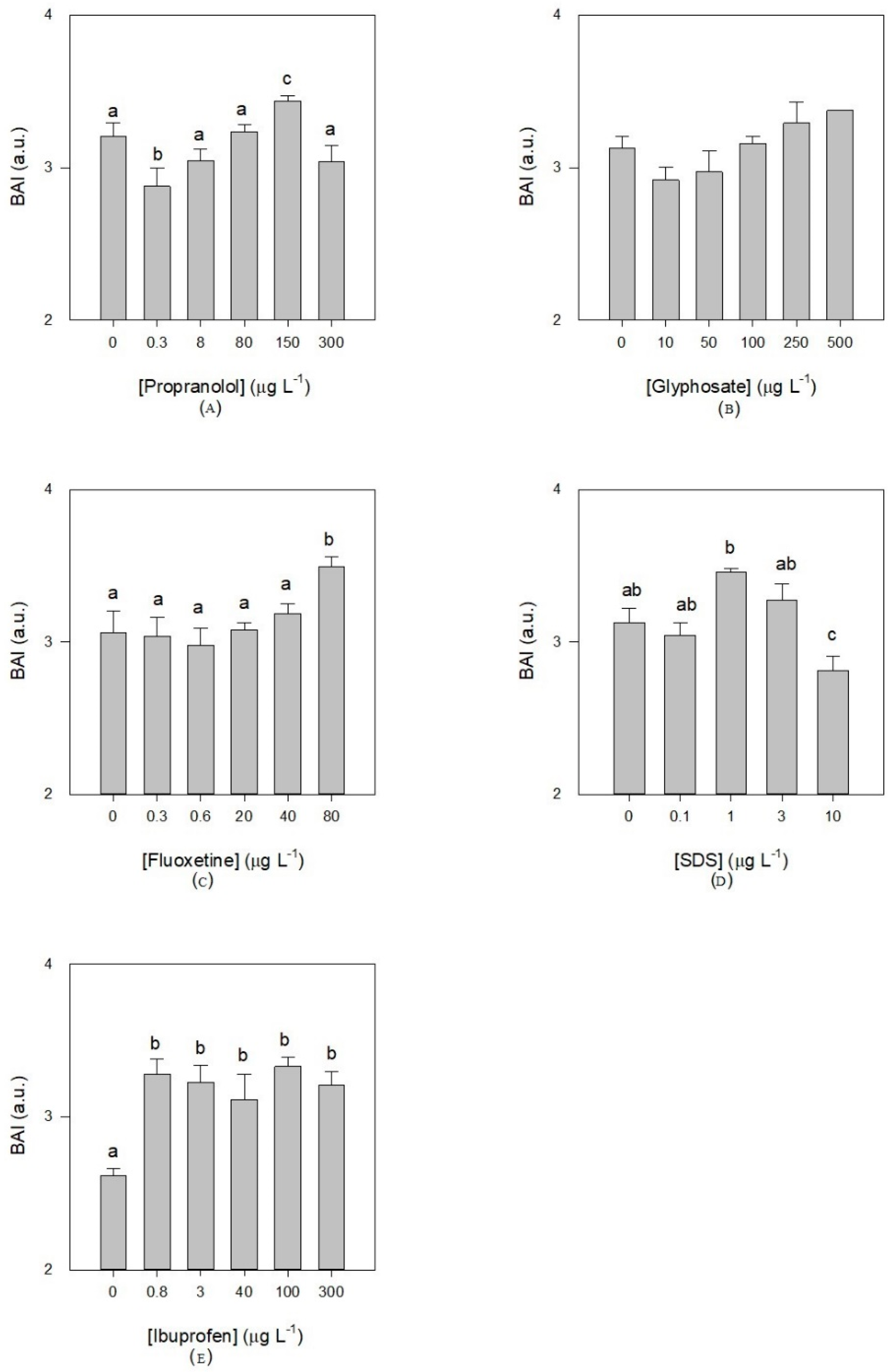
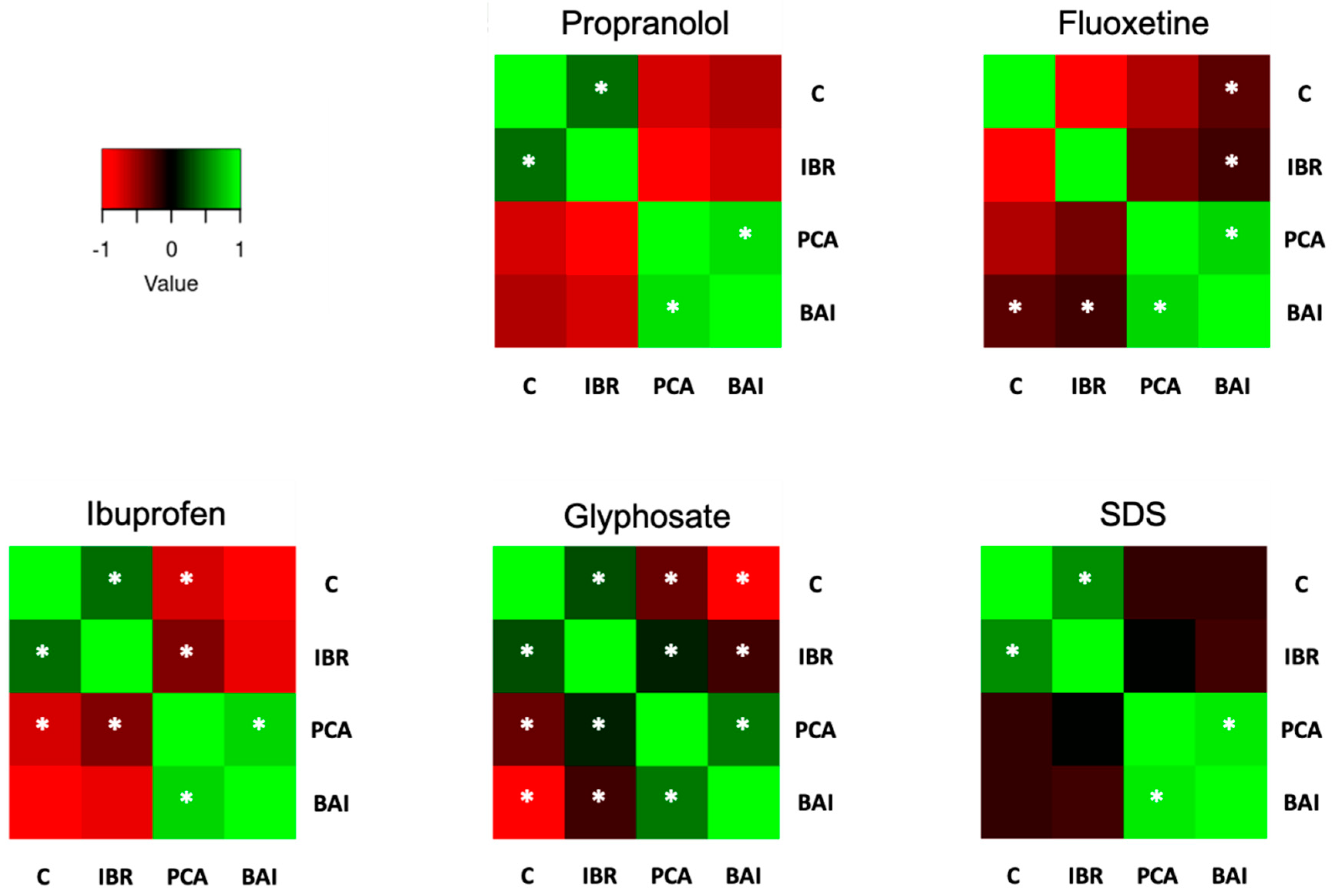
3.3. Oxidative Stress Biomarkers Indexes
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Asimakopoulos, A.; Bletsou, A.; Thomaidis, N. Emerging contaminants: A tutorial mini-review. Glob. Nest J. 2012, 14, 72–79. [Google Scholar]
- EUROSTAT. EUROSTAT. Available online: https://ec.europa.eu/eurostat/statisticsexplained/index.php?title=Archive:Chemicals_production_statistics&oldid=199190 (accessed on 11 February 2021).
- Duarte, B.; Caçador, I. Ecotoxicology of Marine Organisms, 1st ed.; Duarte, B., Caçador, I., Eds.; CRC Press: Boca Raton, FL, USA; Taylor & Francis Group: Abingdon-on-Thames, UK, 2019; ISBN 1138035491. [Google Scholar]
- Lopes, D.G.; Duarte, I.A.; Antunes, M.; Fonseca, V.F. Effects of antidepressants in the reproduction of aquatic organisms: A meta-analysis. Aquat. Toxicol. 2020, 227, 105569. [Google Scholar] [CrossRef]
- Beliaeff, B.; Burgeot, T. Integrated biomarker response: A useful tool for ecological risk assessment. In Proceedings of the Environmental Toxicology and Chemistry; SETAC Press: Pensacola, FL, USA, 2002; Volume 21, pp. 1316–1322. [Google Scholar]
- Schettino, T.; Caricato, R.; Calisi, A.; Giordano, M.E.; Lionetto, M.G. Biomarker Approach in Marine Monitoring and Assessment: New Insights and Perspectives. Open Environ. Sci. 2012, 12. [Google Scholar] [CrossRef]
- Duarte, I.A.; Reis-Santos, P.; França, S.; Cabral, H.; Fonseca, V.F. Biomarker responses to environmental contamination in estuaries: A comparative multi-taxa approach. Aquat. Toxicol. 2017, 189, 31–41. [Google Scholar] [CrossRef]
- European Comission. European Comission Marine Strategy Framework Directive (MSFD); Europen Comision: Luxembourg, 2017. [Google Scholar]
- WHO. Biomarkers and Risk Assessment: Concepts and Principles, Environmental Health Criteria; WHO: Geneva, Switzerland, 1993. [Google Scholar]
- Valavanidis, A.; Vlahogianni, T.; Dassenakis, M.; Scoullos, M. Molecular biomarkers of oxidative stress in aquatic organisms in relation to toxic environmental pollutants. Ecotoxicol. Environ. Saf. 2006, 64, 178–189. [Google Scholar] [CrossRef]
- Livingstone, D.R. Contaminant-stimulated reactive oxygen species production and oxidative damage in aquatic organisms. Mar. Pollut. Bull. 2001, 42, 656–666. [Google Scholar]
- Draper, H.H.; Squires, E.J.; Mahmoodi, H.; Wu, J.; Agarwal, S.; Hadley, M. A comparative evaluation of thiobarbituric acid methods for the determination of malondialdehyde in biological materials. Free Radic. Biol. Med. 1993, 15, 353–363. [Google Scholar] [CrossRef]
- Anjum, N.A.; Duarte, B.; Caçador, I.; Sleimi, N.; Duarte, A.C.; Pereira, E. Biophysical and biochemical markers of metal/metalloid-impacts in salt marsh halophytes and their implications. Front. Environ. Sci. 2016, 4, 24. [Google Scholar]
- Borja, A.; Bricker, S.B.; Dauer, D.M.; Demetriades, N.T.; Ferreira, J.G.; Forbes, A.T.; Hutchings, P.; Jia, X.; Kenchington, R.; Marques, J.C.; et al. Overview of integrative tools and methods in assessing ecological integrity in estuarine and coastal systems worldwide. Mar. Pollut. Bull. 2008, 56, 1519–1537. [Google Scholar] [CrossRef]
- De Carvalho, R.C.; Feijão, E.; Matos, A.R.; Cabrita, M.T.; Novais, S.C.; Lemos, M.F.L.; Caçador, I.; Marques, J.C.; Reis-Santos, P.; Fonseca, V.F.; et al. Glyphosate-based herbicide toxicophenomics in marine diatoms: Impacts on primary production and physiological fitness. Appl. Sci. 2020, 10, 7391. [Google Scholar] [CrossRef]
- Borja, A.; Dauer, D.M. Assessing the environmental quality status in estuarine and coastal systems: Comparing methodologies and indices. Ecol. Indic. 2008, 8, 331–337. [Google Scholar] [CrossRef]
- Beck, M.W.; O’Hara, C.; Lowndes, J.S.S.; Ma-Zor, R.D.; Theroux, S.; Gillett, D.J.; Lane, B.; Gearheart, G. The importance of open science for biological assessment of aquatic environments. PeerJ 2020, 8, 1–27. [Google Scholar] [CrossRef]
- Cabral, H.N.; Fonseca, V.F.; Gamito, R.; Gonalves, C.I.; Costa, J.L.; Erzini, K.; Gonalves, J.; Martins, J.; Leite, L.; Andrade, J.P.; et al. Ecological quality assessment of transitional waters based on fish assemblages in Portuguese estuaries: The Estuarine Fish Assessment Index (EFAI). Ecol. Indic. 2012, 19, 144–153. [Google Scholar] [CrossRef]
- Broeg, K.; Westernhagen, H.V.; Zander, S.; Körting, W.; Koehler, A. The “bioeffect assessment index” (BAI): A concept for the quantification of effects of marine pollution by an integrated biomarker approach. Mar. Pollut. Bull. 2005, 50, 495–503. [Google Scholar] [CrossRef]
- Duarte, B.; Freitas, J.; Caçador, I. Sediment microbial activities and physic-chemistry as progress indicators of salt marsh restoration processes. Ecol. Indic. 2012, 19, 231–239. [Google Scholar] [CrossRef]
- Bastida, F.; Luis Moreno, J.; Teresa, H.; García, C. Microbiological degradation index of soils in a semiarid climate. Soil Biol. Biochem. 2006, 38, 3463–3473. [Google Scholar] [CrossRef]
- Duarte, B.; Feijão, E.; de Carvalho, R.C.; Duarte, I.A.; Silva, M.; Matos, A.R.; Cabrita, M.T.; Novais, S.C.; Lemos, M.F.L.; Marques, J.C.; et al. Effects of propranolol on growth, lipids and energy metabolism and oxidative stress response of Phaeodactylum tricornutum. Biology 2020, 9, 478. [Google Scholar] [CrossRef] [PubMed]
- Feijão, E.; Cruz de Carvalho, R.; Duarte, I.A.; Matos, A.R.; Cabrita, M.T.; Novais, S.C.; Lemos, M.F.L.; Caçador, I.; Marques, J.C.; Reis-Santos, P.; et al. Fluoxetine Arrests Growth of the Model Diatom Phaeodactylum tricornutum by Increasing Oxidative Stress and Altering Energetic and Lipid Metabolism. Front. Microbiol. 2020, 11, 1803. [Google Scholar] [CrossRef]
- Silva, M.; Feijão, E.; da Cruz de Carvalho, R.; Duarte, I.A.; Matos, A.R.; Cabrita, M.T.; Barreiro, A.; Lemos, M.F.L.; Novais, S.C.; Marques, J.C.; et al. Comfortably numb: Ecotoxicity of the non-steroidal anti-inflammatory drug ibuprofen on Phaeodactylum tricornutum. Mar. Environ. Res. 2020, 161. [Google Scholar] [CrossRef]
- Silva, M.; Feijão, E.; Carvalho, R.C.; Matos, A.R.; Cabrita, M.T.; Lemos, M.F.L.; Novais, S.C.; Marques, J.C.; Caçador, I.; Reis-Santos, P.; et al. Ecotoxicity of the detergent SDS on Phaeodactylum tricornutum primary productivity, fatty acid metabolism and oxidative stress biomarkers. 2021; preprint. [Google Scholar]
- Guillard, R.R.; Ryther, J.H. Studies of marine planktonic diatoms. I. Cyclotella nana Hustedt, and Detonula confervacea (cleve) Gran. Can. J. Microbiol. 1962, 8, 229–239. [Google Scholar] [CrossRef] [PubMed]
- Feijão, E.; Gameiro, C.; Franzitta, M.; Duarte, B.; Caçador, I.; Cabrita, M.T.; Matos, A.R. Heat wave impacts on the model diatom Phaeodactylum tricornutum: Searching for photochemical and fatty acid biomarkers of thermal stress. Ecol. Indic. 2018, 95, 1026–1037. [Google Scholar] [CrossRef]
- OECD. OECD guidelines for the testing of chemicals. Freshwater alga and cyanobacteria, growth inhibition test. Organ. Econ. Coop. Dev. 2011, 1–25. [Google Scholar] [CrossRef]
- Franzellitti, S.; Buratti, S.; Du, B.; Haddad, S.P.; Chambliss, C.K.; Brooks, B.W.; Fabbri, E. A multibiomarker approach to explore interactive effects of propranolol and fluoxetine in marine mussels. Environ. Pollut. 2015, 205, 60–69. [Google Scholar] [CrossRef] [PubMed]
- Claessens, M.; Vanhaecke, L.; Wille, K.; Janssen, C.R. Emerging contaminants in Belgian marine waters: Single toxicant and mixture risks of pharmaceuticals. Mar. Pollut. Bull. 2013, 71, 41–50. [Google Scholar] [CrossRef] [PubMed]
- Teranishi, Y.; Tanaka, A.; Osumi, M.; Fukui, S. Catalase activities of hydrocarbon-utilizing candida yeasts. Agric. Biol. Chem. 1974, 38, 1213–1220. [Google Scholar] [CrossRef]
- Tiryakioglu, M.; Eker, S.; Ozkutlu, F.; Husted, S.; Cakmak, I. Antioxidant defense system and cadmium uptake in barley genotypes differing in cadmium tolerance. J. Trace Elem. Med. Biol. 2006, 20, 181–189. [Google Scholar] [CrossRef]
- Marklund, S.; Marklund, G. Involvement of the Superoxide Anion Radical in the Autoxidation of Pyrogallol and a Convenient Assay for Superoxide Dismutase. Eur. J. Biochem. 1974, 47, 469–474. [Google Scholar] [CrossRef]
- Heath, R.L.; Packer, L. Photoperoxidation in isolated chloroplasts. I. Kinetics and stoichiometry of fatty acid peroxidation. Arch. Biochem. Biophys. 1968, 125, 189–198. [Google Scholar] [CrossRef]
- Broeg, K.; Lehtonen, K.K. Indices for the assessment of environmental pollution of the Baltic Sea coasts: Integrated assessment of a multi-biomarker approach. Mar. Pollut. Bull. 2006, 53, 508–522. [Google Scholar] [CrossRef]
- Sinha, S.; Masto, R.E.; Ram, L.C.; Selvi, V.A.; Srivastava, N.K.; Tripathi, R.C.; George, J. Rhizosphere soil microbial index of tree species in a coal mining ecosystem. Soil Biol. Biochem. 2009, 41, 1824–1832. [Google Scholar] [CrossRef]
- Clarke, K.; RN, G. Primer v6: User Manual/Tutorial. 2006. Available online: https://www.scienceopen.com/document?vid=87ec9d6d-cae1-4f49-9204-0fbf723dcdbc (accessed on 3 March 2021).
- Masto, R.E.; Chhonkar, P.K.; Singh, D.; Patra, A.K. Alternative soil quality indices for evaluating the effect of intensive cropping, fertilisation and manuring for 31 years in the semi-arid soils of India. Environ. Monit. Assess. 2008, 136, 419–435. [Google Scholar] [CrossRef] [PubMed]
- Cabrita, M.T.; Duarte, B.; Gameiro, C.; Godinho, R.M.; Caçador, I. Photochemical features and trace element substituted chlorophylls as early detection biomarkers of metal exposure in the model diatom Phaeodactylum tricornutum. Ecol. Indic. 2018, 95, 1038–1052. [Google Scholar] [CrossRef]
- Duarte, B.; Cabrita, M.T.; Vidal, T.; Pereira, J.L.; Pacheco, M.; Pereira, P.; Canário, J.; Gonçalves, F.J.M.; Matos, A.R.; Rosa, R.; et al. Phytoplankton community-level bio-optical assessment in a naturally mercury contaminated Antarctic ecosystem (Deception Island). Mar. Environ. Res. 2018, 140, 412–421. [Google Scholar] [CrossRef] [PubMed]
- Duarte, B.; Prata, D.; Matos, A.R.; Cabrita, M.T.; Caçador, I.; Marques, J.C.; Cabral, H.N.; Reis-Santos, P.; Fonseca, V.F. Ecotoxicity of the lipid-lowering drug bezafibrate on the bioenergetics and lipid metabolism of the diatom Phaeodactylum tricornutum. Sci. Total Environ. 2019, 650, 2085–2094. [Google Scholar] [CrossRef] [PubMed]
- Duarte, B.; Pedro, S.; Marques, J.C.; Adão, H.; Caçador, I. Zostera noltii development probing using chlorophyll a transient analysis (JIP-test) under field conditions: Integrating physiological insights into a photochemical stress index. Ecol. Indic. 2017, 76, 219–229. [Google Scholar] [CrossRef]
- Kaviraj, A.; Unlu, E.; Gupta, A.; El Nemr, A. Biomarkers of environmental pollutants. Biomed Res. Int. 2014, 2014, 806598. [Google Scholar] [CrossRef]
- Devin, S.; Burgeot, T.; Giambérini, L.; Minguez, L.; Pain-Devin, S. The integrated biomarker response revisited: Optimization to avoid misuse. Environ. Sci. Pollut. Res. 2014, 21, 2448–2454. [Google Scholar] [CrossRef]
- Meng, F.-P.; Yang, F.-F.; Cheng, F.-L. Marine environmental assessment approaches based on biomarker index: A review. J. Appl. Ecol. 2012, 23, 1128–1136. [Google Scholar]
- Marigómez, I.; Garmendia, L.; Soto, M.; Orbea, A.; Izagirre, U.; Cajaraville, M.P. Marine ecosystem health status assessment through integrative biomarker indices: A comparative study after the Prestige oil spill “mussel Watch.”. Ecotoxicology 2013, 22, 486–505. [Google Scholar] [CrossRef]
- Novais, S.C.; Gomes, N.C.; Soares, A.M.V.M.; Amorim, M.J.B. Antioxidant and neurotoxicity markers in the model organism Enchytraeus albidus (Oligochaeta): Mechanisms of response to atrazine, dimethoate and carbendazim. Ecotoxicology 2014, 23, 1220–1233. [Google Scholar] [CrossRef]
- Brooks, S.J.; Escudero-Oñate, C.; Gomes, T.; Ferrando-Climent, L. An integrative biological effects assessment of a mine discharge into a Norwegian fjord using field transplanted mussels. Sci. Total Environ. 2018, 644, 1056–1069. [Google Scholar] [CrossRef] [PubMed]
- Serafim, A.; Company, R.; Lopes, B.; Fonseca, V.F.; Frana, S.; Vasconcelos, R.P.; Bebianno, M.J.; Cabral, H.N. Application of an integrated biomarker response index (IBR) to assess temporal variation of environmental quality in two Portuguese aquatic systems. Ecol. Indic. 2012, 19, 215–225. [Google Scholar] [CrossRef]
- Raftopoulou, E.K.; Dimitriadis, V.K. Assessment of the health status of mussels Mytilus galloprovincialis along Thermaikos Gulf (Northern Greece): An integrative biomarker approach using ecosystem health indices. Ecotoxicol. Environ. Saf. 2010, 73, 1580–1587. [Google Scholar] [CrossRef]
- Felício, A.A.; Freitas, J.S.; Scarin, J.B.; de Souza Ondei, L.; Teresa, F.B.; Schlenk, D.; de Almeida, E.A. Isolated and mixed effects of diuron and its metabolites on biotransformation enzymes and oxidative stress response of Nile tilapia (Oreochromis niloticus). Ecotoxicol. Environ. Saf. 2018, 149, 248–256. [Google Scholar] [CrossRef]
- Rodrigues, A.P.; OlivaeTeles, T.; Mesquita, S.R.; DelerueeMatos, C.; Guimarães, L. Integrated biomarker responses of an estuarine invertebrate to high abiotic stress and decreased metal contamination. Mar. Environ. Res. 2014, 101, 101–114. [Google Scholar] [CrossRef] [PubMed]
- Silva, C.S.E.; Novais, S.C.; Simões, T.; Caramalho, M.; Gravato, C.; Rodrigues, M.J.; Maranhão, P.; Lemos, M.F.L. Using biomarkers to address the impacts of pollution on limpets (Patella depressa) and their mechanisms to cope with stress. Ecol. Indic. 2018, 95, 1077–1086. [Google Scholar] [CrossRef]
- Duarte, B.; Carreiras, J.; Pérez-Romero, J.A.; Mateos-Naranjo, E.; Redondo-Gómez, S.; Matos, A.R.; Marques, J.C.; Caçador, I. Halophyte fatty acids as biomarkers of anthropogenic-driven contamination in Mediterranean marshes: Sentinel species survey and development of an integrated biomarker response (IBR) index. Ecol. Indic. 2018, 87, 86–96. [Google Scholar] [CrossRef]
- Duarte, B.; Durante, L.; Marques, J.C.; Reis-Santos, P.; Fonseca, V.F.; Caçador, I. Development of a toxicophenomic index for trace element ecotoxicity tests using the halophyte Juncus acutus: Juncus-TOX. Ecol. Indic. 2021, 121, 107097. [Google Scholar] [CrossRef]
- Adams, S.M.; Brown, A.M.; Goede, R.W. A Quantitative Health Assessment Index for Rapid Evaluation of Fish Condition in the Field. Trans. Am. Fish. Soc. 1993, 122. [Google Scholar] [CrossRef]
- Clarke, K.R. Nonmetric multivariate analysis in community-level ecotoxicology. Environ. Toxicol. Chem. 1999, 18, 118–127. [Google Scholar] [CrossRef]
- Jollands, N.; Lermit, J.; Patterson, M. Aggregate eco-efficiency indices for New Zealand—A principal components analysis. J. Environ. Manag. 2004, 73, 293–305. [Google Scholar] [CrossRef]
- Isibor, P.O.; Imoobe, T.O.T.; Enuneku, A.A.; Akinduti, P.A.; Dedeke, G.A.; Adagunodo, T.A.; Obafemi, D.Y. Principal Components and Hierarchical Cluster Analyses of Trace Metals and Total Hydrocarbons in Gills, Intestines and Muscles of Clarias gariepinus (Burchell, 1822). Sci. Rep. 2020, 10, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Shin, P.K.S.; Lam, W.K.C. Development of a Marine Sediment Pollution Index. Environ. Pollut. 2001, 113, 281–291. [Google Scholar] [CrossRef]
- Reid, M.K.; Spencer, K.L. Use of principal components analysis (PCA) on estuarine sediment datasets: The effect of data pre-treatment. Environ. Pollut. 2009, 157, 2275–2281. [Google Scholar] [CrossRef] [PubMed]
- Caçador, I.; Neto, J.M.; Duarte, B.; Barroso, D.V.; Pinto, M.; Marques, J.C. Development of an Angiosperm Quality Assessment Index (AQuA-Index) for ecological quality evaluation of Portuguese water bodies—A multi-metric approach. Ecol. Indic. 2013, 25, 141–148. [Google Scholar] [CrossRef]
- Lever, J.; Krzywinski, M.; Altman, N. Principal component analysis. Nat. Methods 2017, 14, 641–642. [Google Scholar] [CrossRef]

| Compound | Concentrations Tested (μg L−1) | Inhibition (%) | IC50 (μg L−1) | Reference |
|---|---|---|---|---|
| Propranolol | 0.3 | 0.15 | 194.6 | [22] |
| 8 | 4.11 | |||
| 80 | 41.11 | |||
| 150 | 77.08 | |||
| 300 | 154.16 | |||
| Fluoxetine | 0.3 | 0.63 | 47.3 | [23] |
| 0.6 | 1.27 | |||
| 20 | 42.26 | |||
| 40 | 84.52 | |||
| 80 | 169.04 | |||
| Ibuprofen | 0.8 | 0.23 | 350.6 | |
| 3 | 0.86 | [24] | ||
| 40 | 11.41 | |||
| 100 | 28.52 | |||
| 300 | 85.57 | |||
| Glyphosate | 10 | 4.43 | 225.9 | |
| 50 | 22.13 | [15] | ||
| 100 | 44.27 | |||
| 250 | 110.67 | |||
| 500 | 221.34 | |||
| SDS | 0.1 | 15.5 | 11.4 | |
| 1 | 21.1 | [25] | ||
| 3 | 24.6 | |||
| 10 | 43.8 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pires, V.L.; Novais, S.C.; Lemos, M.F.L.; Fonseca, V.F.; Duarte, B. Evaluation of Multivariate Biomarker Indexes Application in Ecotoxicity Tests with Marine Diatoms Exposed to Emerging Contaminants. Appl. Sci. 2021, 11, 3878. https://doi.org/10.3390/app11093878
Pires VL, Novais SC, Lemos MFL, Fonseca VF, Duarte B. Evaluation of Multivariate Biomarker Indexes Application in Ecotoxicity Tests with Marine Diatoms Exposed to Emerging Contaminants. Applied Sciences. 2021; 11(9):3878. https://doi.org/10.3390/app11093878
Chicago/Turabian StylePires, Vanessa Leal, Sara C. Novais, Marco F. L. Lemos, Vanessa F. Fonseca, and Bernardo Duarte. 2021. "Evaluation of Multivariate Biomarker Indexes Application in Ecotoxicity Tests with Marine Diatoms Exposed to Emerging Contaminants" Applied Sciences 11, no. 9: 3878. https://doi.org/10.3390/app11093878
APA StylePires, V. L., Novais, S. C., Lemos, M. F. L., Fonseca, V. F., & Duarte, B. (2021). Evaluation of Multivariate Biomarker Indexes Application in Ecotoxicity Tests with Marine Diatoms Exposed to Emerging Contaminants. Applied Sciences, 11(9), 3878. https://doi.org/10.3390/app11093878










