Spectroscopic and Spectrometric Applications for the Identification of Bioactive Compounds from Vegetal Extracts
Abstract
Featured Application
Abstract
1. Introduction
2. Discussion
- Glycosides
- Flavonoids and proanthocyanidins
- Tannins
- Terpenes and terpenoids
- Resins
- Lignans
- Alkaloids
- Furanocoumarins and naphthodianthrones
- Proteins and peptides
- Extraction of bioactive compounds from plants
3. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Wadood, S.A.; Boli, G.; Zhang, X.W.; Hussain, I.; Wei, Y.M. Recent development in the application of analytical techniques for the traceability and authenticity of food of plant origin. Microchem. J. 2020, 152, 104295. [Google Scholar] [CrossRef]
- Lindinger, W.; Hansel, A.; Jordan, A. On-line monitoring of volatile organic compounds at pptv levels by means of proton-transfer reaction mass spectrometry (PTR-MS) medical applications, food control and environmental research. Int. J. Mass Spectrom. 1998, 173, 191–241. [Google Scholar] [CrossRef]
- Yang, X.; Neta, P.; Stein, S.E. Extending a tandem mass spectral library to include MS2 spectra of fragment ions produced in-source and MSn spectra. J. Am. Soc. Mass Spectrom. 2017, 28, 2280–2287. [Google Scholar] [CrossRef] [PubMed]
- Conceição, R.S.; Perez, C.J.; Branco, A.; Botura, M.B.; Ifa, D.R. Identification of Sassafras albidum alkaloids by high-performance thin-layer chromatography tandem mass spectrometry and mapping by desorption electrospray ionization mass spectrometry imaging. J. Mass Spectrom. 2020, e4674. [Google Scholar] [CrossRef]
- Govindaraj, N.; Gangadoo, S.; Truong, V.K.; Chapman, J.; Gill, H.; Cozzolino, D. The use of derivatives and chemometrics to interrogate the UV-Visible spectra of gin samples to monitor changes related to storage. Spectrochim. Acta A Mol. Biomol. Spectrosc. 2020, 227, 117548. [Google Scholar] [CrossRef]
- Emwas, A.H.; Roy, R.; McKay, R.T.; Tenori, L.; Saccenti, E.; Gowda, G.A.; Raftery, D.; Alahmari, F.; Jaremko, L.; Jaremko, M.; et al. NMR spectroscopy for metabolomics research. Metabolites 2019, 9, 123. [Google Scholar] [CrossRef]
- Wallace, R.J. Antimicrobial properties of plant secondary metabolites. Proc. Nutr. Soc. 2004, 63, 621–629. [Google Scholar] [CrossRef]
- Seca, A.M.; Pinto, D.C. Plant secondary metabolites as anticancer agents: Successes in clinical trials and therapeutic application. Int. J. Mol. Sci. 2018, 19, 263. [Google Scholar] [CrossRef]
- Kong, J.M.; Chia, L.S.; Goh, N.K.; Chia, T.F.; Brouillard, R. Analysis and biological activities of anthocyanins. Phytochemistry 2003, 64, 923–933. [Google Scholar] [CrossRef]
- Bernhoft, A. A brief review on bioactive compounds in plants. In Bioactive Compounds in Plants—Benefits and Risks for Man and Animals; Bernhoft, A., Ed.; The Norwegian Academy of Science and Letters: Oslo, Norway, 2010. [Google Scholar]
- Zhao, M.; Bai, L.; Wang, L.; Toki, A.; Hasegawa, T.; Kikuchi, M.; Abe, M.; Sakai, J.; Hasegawa, R.; Bai, Y.; et al. Bioactive cardenolides from the stems and twigs of Nerium oleander. J. Nat. Prod. 2007, 70, 1098–1103. [Google Scholar] [CrossRef]
- Binienda, A.; Ziolkowska, S.; Pluciennik, E. The anticancer properties of silibinin: Its molecular mechanism and therapeutic effect in breast cancer. Anticancer Agents Med. Chem. 2020, 20, 1787–1796. [Google Scholar] [CrossRef] [PubMed]
- Gillessen, A.; Schmidt, H.H.J. Silymarin as Supportive Treatment in Liver Diseases: A Narrative Review. Adv. Ther. 2020, 37, 1279–1301. [Google Scholar] [CrossRef] [PubMed]
- Elateeq, A.A.; Sun, Y.; Nxumalo, W.; Gabr, A.M. Biotechnological production of silymarin in Silybum marianum L.: A review. Biocatal. Agric. Biotechnol. 2020, 29, 101775. [Google Scholar] [CrossRef]
- Petrásková, L.; Káňová, K.; Biedermann, D.; Křen, V.; Valentová, K. Simple and Rapid HPLC Separation and Quantification of Flavonoid, Flavonolignans, and 2,3-Dehydroflavonolignans in Silymarin. Foods 2020, 9, 116. [Google Scholar] [CrossRef]
- Rauf, A.; Imran, M.; Abu-Izneid, T.; Patel, S.; Pan, X.; Naz, S.; Silva, A.S.; Saeed, F.; Suleria, H.A.R. Proanthocyanidins: A comprehensive review. Biomed. Pharmacother. 2019, 116, 108999. [Google Scholar] [CrossRef]
- Reeves, S.G.; Somogyi, A.; Zeller, W.E.; Ramelot, T.A.; Wrighton, K.C.; Hagerman, A.E. Proanthocyanidin Structural Details Revealed by Ultrahigh Resolution FT-ICR MALDI-Mass Spectrometry, 1H–13C HSQC NMR, and Thiolysis-HPLC–DAD. J. Agric. Food Chem. 2020, 68, 14038–14048. [Google Scholar] [CrossRef]
- Mattio, L.M.; Catinella, G.; Dallavalle, S.; Pinto, A. Stilbenoids: A Natural Arsenal against Bacterial Pathogens. Antibiotics 2020, 9, 336. [Google Scholar] [CrossRef]
- Kim, H.W.; Kim, S.S.; Kang, K.B.; Ryu, B.; Park, E.; Huh, J.; Jeon, W.K.; Chae, H.-S.; Oh, W.K.; Kim, J.; et al. Combined MS/MS-NMR Annotation Guided Discovery of Iris lactea var. chinensis Seed as a Source of Viral Neuraminidase Inhibitory Polyphenols. Molecules 2020, 25, 3383. [Google Scholar] [CrossRef]
- Watrelot, A.A.; Norton, E.L. Chemistry and Reactivity of Tannins in Vitis spp.: A Review. Molecules 2020, 25, 2110. [Google Scholar] [CrossRef]
- Cardullo, N.; Muccilli, V.; Cunsolo, V.; Tringali, C. Mass Spectrometry and 1H-NMR Study of Schinopsis lorentzii (Quebracho) Tannins as a Source of Hypoglycemic and Antioxidant Principles. Molecules 2020, 25, 3257. [Google Scholar] [CrossRef]
- Yu, L.; Yan, J.; Sun, Z. D-limonene exhibits anti-inflammatory and antioxidant properties in an ulcerative colitis rat model via regulation of iNOS, COX-2, PGE2 and ERK signaling pathways. Mol. Med. Rep. 2017, 15, 2339–2346. [Google Scholar] [CrossRef]
- John, I.; Muthukumar, K.; Arunagiri, A. A review on the potential of citrus waste for D-Limonene, pectin, and bioethanol production. Int. J. Green Energy 2017, 14, 599–612. [Google Scholar] [CrossRef]
- Bsaibes, S.; Piel, F.; Gros, V.; Truong, F.; Lafouge, F.; Ciuraru, R.; Buysse, P.; Kammer, J.; Loubet, B.; Staudt, M. Monoterpene Chemical Speciation with High Time Resolution Using a FastGC/PTR-MS: Results from the COV3ER Experiment on Quercus ilex. Atmosphere 2020, 11, 690. [Google Scholar] [CrossRef]
- Rana, V.S.; Ahluwalia, V.; Shakil, N.A.; Prasad, L. Essential oil composition, antifungal, and seedling growth inhibitory effects of zerumbone from Zingiber zerumbet Smith. J. Essent. Oil Res. 2017, 29, 320–329. [Google Scholar] [CrossRef]
- Girisa, S.; Shabnam, B.; Monisha, J.; Fan, L.; Halim, C.E.; Arfuso, F.; Ahn, K.S.; Sethi, G.; Kunnumakkara, A.B. Potential of Zerumbone as an Anti-Cancer Agent. Molecules 2019, 24, 734. [Google Scholar] [CrossRef] [PubMed]
- Srikrishna, D.; Godugu, C.; Dubey, P.K. A review on pharmacological properties of coumarins. Mini Rev. Med. Chem. 2018, 18, 113–141. [Google Scholar] [CrossRef]
- Gonçalves, G.A.; Spillere, A.R.; Machado das Neves, G.; Kagami, L.P.; Lino von Poser, G.; Santos Canto, R.F.; Eifler-Lima, V.L. Natural and synthetic coumarins as antileishmanial agents: A review. Eur. J. Med. Chem. 2020, 203, 112514. [Google Scholar] [CrossRef] [PubMed]
- Majhi, S. Diterpenoids: Natural Distribution, Semisynthesis at Room Temperature and Pharmacological Aspects-A Decade Update. Chem. Sel. 2020, 15, 1–16. [Google Scholar] [CrossRef]
- Yilmaz, A.; Crowley, R.S.; Sherwood, A.M.; Prisinzano, T.E. Semisynthesis and kappa-opioid receptor activity of derivatives of columbin, a furanolactone diterpene. J. Nat. Prod. 2017, 80, 2094–2100. [Google Scholar] [CrossRef]
- Islam, A.K.M.M.; Hong, S.M.; Lee, H.S.; Moon, B.C.; Kim, D.; Kwon, H. Identification and characterization of matrix components in spinach during QuEChERS sample preparation for pesticide residue analysis by LC–ESI–MS/MS, GC–MS and UPLC-DAD. J. Food Sci. Technol. 2018, 55, 3930–3938. [Google Scholar] [CrossRef]
- Dai, Y.; Row, K.H. Isolation and determination of beta-carotene in carrots by magnetic chitosan beta-cyclodextrin extraction and high-performance liquid chromatography (HPLC). Anal. Lett. 2019, 52, 1828–1843. [Google Scholar] [CrossRef]
- Horst, D.J.; Tebcherani, S.M.; Kubaski, E.T.; de Almeida Vieira, R. Bioactive Potential of 3D-Printed Oleo-Gum-Resin Disks: B. papyrifera, C. myrrha, and S. benzoin Loading Nanooxides—TiO2, P25, Cu2O, and MoO3. Bioinorg. Chem. Appl. 2017, 2017, 6398167. [Google Scholar] [CrossRef] [PubMed]
- Wang, F.; Zhang, L.; Zhang, Q.; Chen, A.; Wang, S.; Fang, Z. Two new phenylpropanoids from the resin of Styrax tonkinensis (Pierre) Craib ex Hartw. J. Nat. Med. 2020, 74, 819–824. [Google Scholar] [CrossRef]
- Cui, Q.; Du, R.; Liu, M.; Rong, L. Lignans and Their Derivatives from Plants as Antivirals. Molecules 2020, 25, 183. [Google Scholar] [CrossRef]
- Liu, B.; Yang, Y.; Liu, H.; Xie, Z.; Li, Q.; Deng, M.; Li, F.; Peng, J.; Wu, H. Screening for cytotoxic chemical constituents from Justicia procumbens by HPLC–DAD–ESI–MS and NMR. Chem. Cent. J. 2018, 12, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Debnath, B.; Singh, W.S.; Das, M.; Goswami, S.; Singh, M.K.; Maiti, D.; Manna, K. Role of plant alkaloids on human health: A review of biological activities. Mater. Today Chem. 2018, 9, 56–72. [Google Scholar] [CrossRef]
- Kopp, T.; Abdel-Tawab, M.; Mizaikoff, B. Extracting and Analyzing Pyrrolizidine Alkaloids in Medicinal Plants: A Review. Toxins 2020, 12, 320. [Google Scholar] [CrossRef] [PubMed]
- Melough, M.M.; Cho, E.; Chun, O.K. Furocoumarins: A review of biochemical activities, dietary sources and intake, and potential health risks. Food Chem Toxicol. 2018, 113, 99–107. [Google Scholar] [CrossRef]
- Bruni, R.; Barreca, D.; Protti, M.; Brighenti, V.; Righetti, L.; Anceschi, L.; Mercolini, L.; Benvenuti, S.; Gattuso, G.; Pellati, F. Botanical Sources, Chemistry, Analysis, and Biological Activity of Furanocoumarins of Pharmaceutical Interest. Molecules 2019, 24, 2163. [Google Scholar] [CrossRef]
- Mir, M.Y.; Hamid, S.; Kamili, A.N.; Hassan, Q.P. Sneak peek of Hypericum perforatum L.: Phytochemistry, phytochemical efficacy and biotechnological interventions. J. Plant Biochem. Biotechnol. 2019, 28, 357–373. [Google Scholar] [CrossRef]
- Kimáková, K.; Kimáková, A.; Idkowiak, J.; Stobiecki, M.; Rodziewicz, P.; Marczak, Ł.; Čellárová, E. Phenotyping the genus Hypericum by secondary metabolite profiling: Emodin vs. skyrin, two possible key intermediates in hypericin biosynthesis. Anal. Bioanal. Chem. 2018, 410, 7689–7699. [Google Scholar] [CrossRef]
- Hayes, M.; Bleakley, S. 21 Peptides from plants and their applications. In Peptide Applications in Biomedicine, Biotechnology and Bioengineering; Koutsopoulos, S., Ed.; Woodhead Publishing: Sawston, UK, 2018; pp. 603–622. [Google Scholar] [CrossRef]
- Fideler, J.; Johanningsmeier, S.D.; Ekelöf, M.; Muddiman, D.C. Discovery and quantification of bioactive peptides in fermented cucumber by direct analysis IR-MALDESI mass spectrometry and LC-QQQ-MS. Food Chem. 2019, 271, 715–723. [Google Scholar] [CrossRef]
- Kumar, K.K.; Chandra, K.L.P.; Sumanthi, J.; Reddy, G.S.; Shekar, P.C.; Reddy, B.V.R. Biological role of lectins: A review. J. Orofac. Sci. 2012, 4, 20. [Google Scholar] [CrossRef]
- Gnanesh Kumar, B.S.; Surolia, A. Identification of Banana Lectin Isoforms and Differential Acetylation through Mass Spectrometry Approaches. Prot. J. 2017, 37, 38–46. [Google Scholar] [CrossRef] [PubMed]
- Hong, V.; Wrolstad, R.E. Use of HPLC separation/photodiode array detection for characterization of anthocyanins. J. Agric. Food Chem. 1990, 38, 708–715. [Google Scholar] [CrossRef]
- Chen, L.; Xin, X.; Feng, H.; Li, S.; Cao, Q.; Wang, X.; Vriesekoop, F. Isolation and Identification of Anthocyanin Component in the Fruits of Acanthopanax sessiliflorus (Rupr. & Maxim.) Seem. by Means of High-Speed Counter Current Chromatography and Evaluation of Its Antioxidant Activity. Molecules 2020, 25, 1781. [Google Scholar] [CrossRef]
- Wang, L.S.; Stoner, G.D. Anthocyanins and their role in cancer prevention. Cancer Lett. 2008, 269, 281–290. [Google Scholar] [CrossRef] [PubMed]
- Mourtzinos, I.; Makris, D.P.; Yannakopoulou, K.; Kalogeropoulos, N.; Michali, I.; Karathanos, V.T. Thermal Stability of Anthocyanin Extract of Hibiscus sabdariffa L. in the Presence of β-Cyclodextrin. J. Agric. Food Chem. 2008, 56, 10303–10310. [Google Scholar] [CrossRef] [PubMed]
- Bonvehí, J.S.; Coll, F.V. Phenolic composition of propolis from China and from South America. Z. Naturforsch. C 1994, 49, 712–718. [Google Scholar] [CrossRef]
- Jiménez-Martínez, M.D.; Bautista-Ortín, A.B.; Gil-Muñoz, R.; Gómez-Plaza, E. Fining with purified grape pomace. Effect of dose, contact time and varietal origin on the final wine phenolic composition. Food Chem. 2019, 271, 570–576. [Google Scholar] [CrossRef]
- Folin, O.; Ciocalteu, V. Tyrosine and tryptophan determinations proteins. J. Biol. Chem. 1927, 73, 627. [Google Scholar] [CrossRef]
- Singleton, V.L.; Orthofer, R.; Lamuela-Raventos, R.M. Analysis of total phenols and other oxidation substrates and antioxidants by means of Folin-Ciocalteu reagent. Meth. Enzymol. 1999, 299, 152–178. [Google Scholar] [CrossRef]
- Jiménez-Aguilar, D.M.; Ortega-Regules, A.E.; Lozada-Ramírez, J.D.; Pérez-Pérez, M.C.I.; Vernon-Carter, E.J.; Welti-Chanes, J. Color and chemical stability of spray-dried blueberry extract using mesquite gum as wall material. J. Food Compost. Anal. 2011, 24, 889–894. [Google Scholar] [CrossRef]
- Lee, J.; Durst, R.W.; Wrolstad, R.E. Determination of total monomeric anthocyanin pigment content of fruit juices, beverages, natural colorants, and wines by the pH differential method: Collaborative study. J. AOAC Int. 2005, 88, 1269–1278. [Google Scholar] [CrossRef] [PubMed]
- Sarni-Manchado, P.; Fulcrand, H.; Souquet, J.M.; Cheynier, V.; Moutounet, M. Stability and color of unreported wine anthocyanin-derived pigments. J. Food Sci. 1996, 61, 938–941. [Google Scholar] [CrossRef]
- Boulton, R. The copigmentation of anthocyanins and its role in the color of red wine: A critical review. Am. J. Enol. Vitic. 2001, 52, 67–87. [Google Scholar]
- Osete-Alcaraz, A.; Bautista-Ortín, A.B.; Ortega-Regules, A.; Gómez-Plaza, E. Elimination of suspended cell wall material in musts improves the phenolic content and color of red wines. Am. J. Enol. Vitic. 2019, 70, 201–204. [Google Scholar] [CrossRef]
- Ortega-Regules, A.; Romero-Cascales, I.; García, J.M.R.; Bautista-Ortín, A.B.; López-Roca, J.M.; Fernández-Fernández, J.I.; Gómez-Plaza, E. Anthocyanins and tannins in four grape varieties (Vitis vinifera L.). Evolution of their content and extractability. OENO One 2008, 42, 147–156. [Google Scholar] [CrossRef]
- Bautista-Ortín, A.B.; Martínez-Hernández, A.; Ruiz-García, Y.; Gil-Muñoz, R.; Gómez-Plaza, E. Anthocyanins influence tannin-cell wall interactions. Food Chem. 2016, 206, 239–248. [Google Scholar] [CrossRef]
- Gómez-Plaza, E.; Osete-Alcaraz, A.; Jurado, R.; Iniesta, J.A.; Bautista-Ortín, A.B. The application of high power ultrasounds for improving the phenolic extraction and color from grape-derived products. Acta Hortic. 2020, 1274, 1–8. [Google Scholar] [CrossRef]
- Reiter, R.J. Oxidative processes and antioxidative defense mechanisms in the aging brain 1. FASEB J. 1995, 9, 526–533. [Google Scholar] [CrossRef] [PubMed]
- Sherer, T.B.; Betarbet, R.; Stout, A.K.; Lund, S.; Baptista, M.; Panov, A.V.; Cookson, M.R.; Greenamyre, J.T. An in vitro model of Parkinson’s disease: Linking mitochondrial impairment to altered α-synuclein metabolism and oxidative damage. J. Neurosci. 2002, 22, 7006–7015. [Google Scholar] [CrossRef]
- Apel, K.; Hirt, H. Reactive oxygen species: Metabolism, oxidative stress, and signal transduction. Annu. Rev. Plant. Biol. 2004, 55, 373–399. [Google Scholar] [CrossRef]
- Rius-Pérez, S.; Torres-Cuevas, I.; Millán, I.; Ortega, Á.L.; Pérez, S. PGC-1α, Inflammation, and Oxidative Stress: An Integrative View in Metabolism. Oxidative Med. Cell. Longev. 2020, 2020, 1452696. [Google Scholar] [CrossRef] [PubMed]
- Flora, S.J. Structural, chemical and biological aspects of antioxidants for strategies against metal and metalloid exposure. Oxidative Med. Cell. Longev. 2009, 2, 191–206. [Google Scholar] [CrossRef] [PubMed]
- Brand-Williams, W.; Cuvelier, M.E.; Berset, C. Use of free radical method to evaluate antioxidant activity. Lebensm. Wiss. Technol. 1995, 28, 25–30. [Google Scholar] [CrossRef]
- Re, R.; Pellegrini, N.; Proteggente, A.; Pannala, A.; Yang, M.; Rice-Evans, C. Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radic. Biol. Med. 1999, 26, 1231–1237. [Google Scholar] [CrossRef]
- Schlesier, K.; Harwat, M.; Böhm, V.; Bitsch, R. Assessment of antioxidant activity by using different in vitro methods. Free Radic. Res. 2002, 36, 177–187. [Google Scholar] [CrossRef]
- Huang, D.; Ou, B.; Prior, R.L. The chemistry behind antioxidant capacity assays. J. Agric. Food Chem. 2005, 53, 1841–1856. [Google Scholar] [CrossRef]
- Benzie, I.F.F.; Strain, J.J. The ferric reducing ability of plasma as a measure of “antioxidant power”: The FRAP assay. Anal. Biochem. 1996, 239, 70–76. [Google Scholar] [CrossRef]
- Cao, G.; Alessio, H.M.; Cutler, R.G. Oxygen-radical absorbance capacity assay for antioxidants. Free Radic. Biol. Med. 1993, 14, 303–311. [Google Scholar] [CrossRef]
- Miller, N.J.; Rice-Evans, C.; Davies, M.J.; Gopinathan, V.; Milner, A. A novel method for measuring antioxidant capacity and its application to monitoring the antioxidant status in premature neonates. Clin. Sci. 1993, 84, 407–412. [Google Scholar] [CrossRef] [PubMed]
- Fogliano, V.; Verde, V.; Randazzo, G.; Ritieni, A. Method for measuring antioxidant activity and its application to monitoring the antioxidant capacity of wines. J. Agric. Food Chem. 1999, 47, 1035–1040. [Google Scholar] [CrossRef] [PubMed]
- Apak, R.; Güçlü, K.; Ozyürek, M.; Oğlu, B.B.; Bener, M. Cupric ion reducing antioxidant capacity assay for food antioxidants: Vitamins, polyphenolics, and flavonoids in food extracts. Methods Mol. Biol. 2008, 477, 163–193. [Google Scholar] [CrossRef]
- Hidalgo, G.I.; Almajano, M.P. Red Fruits: Extraction of Antioxidants, Phenolic Content, and Radical Scavenging Determination: A Review. Antioxidants 2017, 6, 7. [Google Scholar] [CrossRef]
- Metzner, C.R.; Lupitu, A.I.; Moisa, C.; Rivis, A.; Copolovici, L.O.; Poiana, M.A. Investigation on High-Value Bioactive Compounds and Antioxidant Properties of Blackberries and Their Fractions Obtained by Home-Scale Juice Processing. Sustainability 2020, 12, 5681. [Google Scholar] [CrossRef]
- Magrone, T.; Magrone, M.; Russo, M.A.; Jirillo, E. Recent Advances on the Anti-Inflammatory and Antioxidant Properties of Red Grape Polyphenols: In Vitro and In Vivo Studies. Antioxidants 2020, 9, 35. [Google Scholar] [CrossRef]
- Ljevar, A.; Ćurko, N.; Tomašević, M.; Radošević, K.; Srček, V.G.; Ganić, K.K. Phenolic Composition, Antioxidant Capacity and in vitro Cytotoxicity Assessment of Fruit Wines. Food Technol. Biotechnol. 2016, 54, 145–155. [Google Scholar] [CrossRef]
- Nie, X.R.; Li, H.Y.; Wei, S.Y.; Han, Q.H.; Zhao, L.; Zhang, Q.; Li, S.Q.; Qin, W.; Wu, D.T. Changes of phenolic compounds, antioxidant capacities, and inhibitory effects on digestive enzymes of kiwifruits (Actinidia chinensis) during maturation. J. Food Meas. Charact. 2020, 14, 1765–1774. [Google Scholar] [CrossRef]
- Tong, T.; Liu, Y.J.; Kang, J.; Zhang, C.M.; Kang, S.G. Antioxidant Activity and Main Chemical Components of a Novel Fermented Tea. Molecules 2019, 24, 2917. [Google Scholar] [CrossRef]
- Kim, H.J.; Kim, B.; Mun, E.G.; Jeong, S.Y.; Cha, Y.S. The antioxidant activity of steamed ginger and its protective effects on obesity induced by high-fat diet in C57BL/6J mice. Nutr. Res. Pract. 2018, 12, 503–511. [Google Scholar] [CrossRef]
- Ahmad, T.; Cawood, M.; Iqbal, Q.; Ariño, A.; Batool, A.; Tariq, R.; Azam, M.; Akhtar, S. Phytochemicals in Daucus carota and Their Health Benefits-Review Article. Foods 2019, 8, 424. [Google Scholar] [CrossRef]
- Park, J.H.; Lee, Y.J.; Kim, Y.H.; Yoon, K.S. Antioxidant and Antimicrobial Activities of Quinoa (Chenopodium quinoa Willd.) Seeds Cultivated in Korea. Prev. Nutr. Food Sci. 2017, 22, 195–202. [Google Scholar] [CrossRef] [PubMed]
- Žilić, S.; Serpen, A.; Akıllıoǧlu, G.; Gökmen, V.; Vančetović, J. Phenolic compounds, carotenoids, anthocyanins, and antioxidant capacity of colored maize (Zea mays L.) kernels. J. Agric. Food Chem. 2012, 60, 1224–1231. [Google Scholar] [CrossRef]
- Francavilla, A.; Joye, I.J. Anthocyanins in Whole Grain Cereals and Their Potential Effect on Health. Nutrients 2020, 12, 2922. [Google Scholar] [CrossRef]
- Goh, L.M.; Barlow, P.J. Antioxidant capacity in Ginkgo biloba. Food Res. Int. 2002, 35, 815–820. [Google Scholar] [CrossRef]
- Estupiñán, D.C.; Schwartz, S.J.; Garzón, G.A. Antioxidant activity, total phenolics content, anthocyanin, and color stability of isotonic model beverages colored with andes berry (Rubus glaucus Benth) anthocyanin powder. J. Food Sci. 2011, 76, S26–S34. [Google Scholar] [CrossRef] [PubMed]
- Tsai, P.J.; McIntosh, J.; Pearce, P.; Camden, B.; Jordan, B.R. Anthocyanin and antioxidant capacity in Roselle (Hibiscus Sabdariffa, L.) extract. Food Res. Int. 2002, 35, 351–356. [Google Scholar] [CrossRef]
- Fernández-Arroyo, S.; Rodríguez-Medina, I.C.; Beltrán-Debón, R.; Pasini, F.; Joven, J.; Micol, V.; Segura-Carretero, A.; Fernández-Gutiérrez, A. Quantification of the polyphenolic fraction and in vitro antioxidant and in vivo anti-hyperlipemic activities of Hibiscus sabdariffa aqueous extract. Food Res. Int. 2011, 44, 1490–1495. [Google Scholar] [CrossRef]
- Owoade, A.O.; Lowe, G.M.; Khalid, R. The in vitro antioxidant properties of Hibiscus anthocyanins rich extract (HAE). Nat. Sci. 2015, 13, 22–29. [Google Scholar]
- Cissé, M.; Bohuon, P.; Sambe, F.; Kane, C.; Sakho, M.; Dornier, M. Aqueous extraction of anthocyanins from Hibiscus sabdariffa: Experimental kinetics and modeling. J. Food Eng. 2012, 109, 16–21. [Google Scholar] [CrossRef]
- Lao, F.; Giusti, M.M. The effect of pigment matrix, temperature and amount of carrier on the yield and final color properties of spray dried purple corn (Zea mays L.) cob anthocyanin powders. Food Chem. 2017, 227, 376–382. [Google Scholar] [CrossRef] [PubMed]
- Araujo, N.M.P.; Arruda, H.S.; Dos Santos, F.N.; de Morais, D.R.; Pereira, G.A.; Pastore, G.M. LC-MS/MS screening and identification of bioactive compounds in leaves, pulp and seed from Eugenia calycina Cambess. Food Res. Int. 2020, 137, 109556. [Google Scholar] [CrossRef]
- Li, H.W.; Liu, P.; Zhang, H.Q.; Feng, W.M.; Yan, H.; Guo, S.; Qian, D.; Duan, J.A. Determination of bioactive compounds in the nonmedicinal parts of Scrophularia ningpoensis using ultra-high-performance liquid chromatography coupled with tandem mass spectrometry and chemometric analysis. J. Sep. Sci. 2020, 43, 4191–4201. [Google Scholar] [CrossRef] [PubMed]
- Różańska, A.; Russo, M.; Cacciola, F.; Salafia, F.; Polkowska, Ż.; Dugo, P.; Mondello, L. Concentration of potentially bioactive compounds in italian extra virgin olive oils from various sources by using LC-MS and multivariate data analysis. Foods 2020, 9, 1120. [Google Scholar] [CrossRef]
- Russo, M.; Cacciola, F.; Arena, K.; Mangraviti, D.; de Gara, L.; Dugo, P.; Mondello, L. Characterization of the polyphenolic fraction of pomegranate samples by comprehensive two-dimensional liquid chromatography coupled to mass spectrometry detection. Nat. Prod. Res. 2020, 34, 39–45. [Google Scholar] [CrossRef] [PubMed]
- Zhishen, J.; Mengcheng, T.; Jianming, W. The determination of flavanoid contents in mulberry and their scavenging effects on superoxid radicals. Food Chem. 1999, 64, 555–559. [Google Scholar] [CrossRef]
- Urias-Lugo, D.A.; Heredia, J.B.; Muy-Rangel, M.D.; Valdez-Torres, J.B.; Serna-Saldivar, S.O.; Gutiérrez-Uribe, J.A. Anthocyanins and Phenolic Acids of Hybrid and Native Blue Maize (Zea mays L.) Extracts and Their Antiproliferative Activity in Mammary (MCF7), Liver (HepG2), Colon (Caco2 and HT29) and Prostate (PC3) Cancer Cells. Plant. Food Hum. Nutr. 2015, 70, 193–199. [Google Scholar] [CrossRef]
- Yang, Z.; Chen, Z.; Yuan, S.; Zhai, W.; Piao, X. Extraction and identification of anthocyanin from purple corn (Zea mays L.). Int. J. Sci. Technol. 2009, 44, 2485–2492. [Google Scholar] [CrossRef]
- Dewanto, V.; Wu, X.; Liu, R.H. Processed sweet corn has higher antioxidant activity. J. Agric. Food Chem. 2002, 50, 4959–4964. [Google Scholar] [CrossRef]
- De Pascual-Teresa, S.; Santos-Buelga, C.; Rivas-Gonzalo, J.C. LC–MS analysis of anthocyanins from purple corn cob. J. Sci. Food Agric. 2002, 82, 1003–1006. [Google Scholar] [CrossRef]
- Fossen, T.; Slimestad, R.; Andersen, Ø.M. Anthocyanins from Maize (Zea mays) and Reed Canarygrass (Phalaris arundinacea). J. Agric. Food Chem. 2001, 49, 2318–2321. [Google Scholar] [CrossRef] [PubMed]
- Gođevac, D.; Tešević, V.; Veličković, M.; Vujisić, L.; Vajs, V.; Milosavljević, S. Polyphenolic compounds in seeds from some grape cultivars grown in Serbia. J. Serb. Chem. Soc. 2010, 75, 1641–1652. [Google Scholar] [CrossRef]
- Yonekura-Sakakibara, K.; Fukushima, A.; Nakabayashi, R.; Hanada, K.; Matsuda, F.; Sugawara, S.; Inoue, E.; Kuromori, T.; Ito, T.; Shinozaki, K.; et al. Two glycosyltransferases involved in anthocyanin modification delineated by transcriptome independent component analysis in Arabidopsis thaliana. Plant J. 2012, 69, 154–167. [Google Scholar] [CrossRef] [PubMed]
- Du, H.; Wu, J.; Ji, K.X.; Zeng, Q.Y.; Bhuiya, M.W.; Su, S.; Shu, Q.Y.; Ren, H.X.; Liu, Z.A.; Wang, L.S. Methylation mediated by an anthocyanin, O-methyltransferase, is involved in purple flower coloration in Paeonia. J. Exp. Bot. 2015, 66, 6563–6577. [Google Scholar] [CrossRef]
- Guzmán-Figueroa, M.D.P.; Ortega-Regules, A.E.; Bautista-Ortín, A.B.; Gómez-Plaza, E.; Anaya-Berríos, C. New pyranoanthocyanins synthesized from roselle (Hibiscus sabdariffa L.) anthocyanins. J. Mex. Chem. Soc. 2016, 60, 13–18. [Google Scholar] [CrossRef]
- Santillán-Urquiza, E.; Méndez-Rojas, M.Á.; Vélez-Ruiz, J.F. Fortification of yogurt with nano and micro sized calcium, iron and zinc, effect on the physicochemical and rheological properties. LWT 2017, 80, 462–469. [Google Scholar] [CrossRef]
- Martínez-Pacheco, M.; Lozada-Ramírez, J.D.; Martínez-Huitle, C.A.; Cerro-López, M. Hibiscus sabdariffa L. Anthocyanins Immobilization on TiO2 Nanotubes and Its Electrochemical Characterization as a Hydrogen Peroxide Sensing Electrode. J. Electrochem. Soc. 2019, 166, B1506. [Google Scholar] [CrossRef]
- Hid-Cadena, R.H.; Bautista-Ortín, A.B.; Ortega-Regules, A.E.; Welti-Chanes, J.; Lozada-Ramírez, J.D.; Anaya de Parrodi, C. Cambios en contenido de compuestos fenólicos y color de extractos de Jamaica (Hibiscus sabdariffa) sometidos a calentamiento con energía de microondas. In Proceedings of the IX Congreso Nacional del Color, Alicante, Spain, 29 June–2 July 2010; Publicaciones de la Universidad de Alicante: Madrid, Spain, 2010; pp. 300–303. (In Spanish). [Google Scholar]
- Maciel, L.G.; do Carmo, M.A.V.; Azevedo, L.; Daguer, H.; Molognoni, L.; de Almeida, M.M.; Granato, D.; Rosso, N.D. Hibiscus sabdariffa anthocyanins-rich extract: Chemical stability, in vitro antioxidant and antiproliferative activities. Food Chem. Toxicol. 2018, 113, 187–197. [Google Scholar] [CrossRef]
- Deshpande, S.S.; Sathe, S.K.; Salunkhe, D.K. Chemistry and safety of plant phenols. Adv. Exp. Med. Biol. 1984, 177, 457–495. [Google Scholar] [CrossRef]
- Thompson, R.J.; Jacques, D.; Haslam, E.; Tanner, R.J.N. Plant proanthocyanidins. I. Introduction: The isolation, structure and distribution in nature of plant procyanidins. J. Chem. Soc. Perkins Trans. 1972, 1387–1399. [Google Scholar] [CrossRef]
- Chung, K.T.; Wong, T.Y.; Wei, C.I.; Huang, Y.W.; Lin, Y. Tannins and Human Health: A Review. Crit. Rev. Food Sci. Nutr. 1998, 38, 421–464. [Google Scholar] [CrossRef]
- Guoruoluo, Y.; Zhou, H.; Zhou, J.; Zhao, H.; Aisa, H.A.; Yao, G. Isolation and characterization of sesquiterpenoids from Cassia buds and their antimicrobial activities. J. Agric. Food Chem. 2017, 65, 5614–5619. [Google Scholar] [CrossRef] [PubMed]
- Kabir, T.; Uddin, S.; Zaman, S.; Rahman, S.; Behl, T.; Ahmad, A.; Hafeez, A.; Perveen, A.; Ashraf, G. Exploring the Anti-Neuroinflammatory Potential of Steroid and Terpenoid-Derived Phytochemicals to Combat Alzheimer’s Disease. Curr. Pharm. Des. 2020, 26, 1–15. [Google Scholar] [CrossRef]
- Zhou, H.; Guoruoluo, Y.; Tuo, Y.; Zhou, J.; Zhang, H.; Wang, W.; Xiang, M.; Aisa, H.A.; Yao, G. Cassiabudanols A and B, immunostimulative diterpenoids with a Cassiabudane carbon skeleton featuring a 3-oxatetracyclo [6.6.1.02,6.010,14] pentadecane scaffold from Cassia buds. Org. Lett. 2019, 21, 549–553. [Google Scholar] [CrossRef]
- Chopra, B.; Dhingra, A.K.; Dhar, K.L.; Nepali, K. Emerging role of terpenoids for the treatment of cancer: A review. Mini Rev. Med. Chem. 2021. [Google Scholar] [CrossRef]
- Hentschel, V.; Kranl, K.; Hollmann, J.; Lindhauer, M.G.; Bohmand, V.; Bitsch, R. Spectrophotometric determination of yellow pigment content and evaluation of carotenoids by highperformance liquid chromatography in durum wheat grain. J. Agric. Food Chem. 2002, 50, 6663–6668. [Google Scholar] [CrossRef]
- Sommer, A.; Davidson, F.R. Assessment and control of vitamin a deficiency: The annecy accords. J. Nutr. 2002, 132, 2845S–2850S. [Google Scholar] [CrossRef]
- Orta-Amaro, M. Copal: Microestructura, Composición y Algunas Propiedades Relevantes. Ph.D. Thesis, National Polytechnic Institute of Mexico, Mexico City, Mexico, 2007. [Google Scholar]
- Langenheim, J.H. Plant Resins: Chemistry, Evolution, Ecology and Ethnobotany; Timber Press: Portland, OR, USA, 2003; ISBN 978-0881925746. [Google Scholar]
- Perveen, K.; Bokhari, N.A.; Siddique, I.; Al-Rashid, S.A.I. Antifungal Activity of Essential Oil of Commiphora molmol Oleo Gum Resin. J. Essent. Oil Bear. Plants 2018, 21, 667–673. [Google Scholar] [CrossRef]
- Khaled, M.; Jiang, Z.Z.; Zhang, L.Y. Deoxypodophyllotoxin: A promising therapeutic agent from herbal medicine. J. Ethnopharmacol. 2013, 149, 24–34. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.Y.; Zhang, Y.F.; Liu, L.J.; Wang, Y.; Shang, M.Y.; Xu, F.; Liu, G.X.; Cai, S.Q. Lignans and diterpenes isolated from Tirpitzia ovoidea and their biological activities. Chin. J. Nat. Med. 2017, 15, 938–943. [Google Scholar] [CrossRef]
- Tsuchiya, H. Anesthetic Agents of Plant Origin: A Review of Phytochemicals with Anesthetic Activity. Molecules 2017, 22, 1369. [Google Scholar] [CrossRef] [PubMed]
- Yana, Y.X.; Suna, Y.; Lia, Z.R.; Zhoua, L.; Qiu, M.H. Chemistry and Biological Activities of Buxus Alkaloids. Curr. Bioact. Compd. 2011, 7, 47–64. [Google Scholar] [CrossRef]
- Yan, Y.X.; Chen, J.C.; Sun, Y.; Wang, Y.Y.; Su, J.; Li, Y.; Qiu, M.H. Triterpenoid alkaloids from Buxus microphylla. Chem. Biodivers. 2010, 7, 1822–1827. [Google Scholar] [CrossRef] [PubMed]
- Afek, U.; Aharoni, N.; Carmeli, S. Increasing celery resistance to pathogens during storage and reducing high-risk psoralen concentration by treatment with GA3. J. Am. Soc. Hort. Sci. 1995, 120, 562–565. [Google Scholar] [CrossRef]
- Yasaman, M.; Abbaspour, H.; Peyvandi, M.; Naghavi, M.R. Investigation of gene expression diversity in Hypericum spp. before and after flowering under different nitrogen fertilization levels. Acta Agric. Slov. 2020, 116, 83–92. [Google Scholar] [CrossRef]
- Gallego, R.; Bueno, M.; Herrero, M. Sub- and supercritical fluid extraction of bioactive compounds from plants, food-by-products, seaweeds and microalgae—An update. Trends Anal. Chem. 2019, 116, 198–213. [Google Scholar] [CrossRef]
- Wang, M.; Wang, J.; Zhou, Y.; Zhang, M.; Xia, Q.; Bi, W.; Chen, D.D.Y. Ecofriendly mechanochemical extraction of bioactive compounds from plants with deep eutectic solvents. ACS Sustain. Chem. Eng. 2017, 5, 6297–6303. [Google Scholar] [CrossRef]
- Sati, P.; Dhyani, P.; Bhatt, I.D.; Pandey, A. Ginkgo biloba flavonoid glycosides in antimicrobial perspective with reference to extraction method. J. Trad. Complement. Med. 2017, 9, 15–23. [Google Scholar] [CrossRef]
- Lisjak, K.; Lelova, Z.; Žigon, U.; Bolta, Š.V.; Teissedre, P.L.; Vanzo, A. Effect of extraction time on content, composition and sensory perception of proanthocyanidins in wine-like medium and during industrial fermentation of Cabernet Sauvignon. J. Sci. Food Agric. 2020, 100, 1887–1896. [Google Scholar] [CrossRef]
- Suh, S.; Kim, Y.E.; Yang, H.J.; Ko, S.; Hong, G.P. Influence of autoclave treatment and enzymatic hydrolysis on the antioxidant activity of Opuntia ficus-indica fruit extract. Food Sci. Biotechnol. 2017, 26, 581–590. [Google Scholar] [CrossRef]
- Novaes, F.J.M.; Lima, F.A.; Calado, V.; Marriott, P.J.; de Aquino Neto, F.R.; Rezende, C.M. Isolating valuable coffee diterpenes by using an inexpensive procedure. Ind. Crops Prod. 2020, 152, 112494. [Google Scholar] [CrossRef]
- Zhou, L.; Jiang, B.; Zhang, T.; Li, S. Ultrasound-assisted aqueous two-phase extraction of resveratrol from the enzymatic hydrolysates of Polygonum cuspidatum. Food Biosci. 2019, 31, 100442. [Google Scholar] [CrossRef]
- Kovacevic, D.B.; Maras, M.; Barba, F.J.; Granato, D.; Roohinejad, S.; Mallikarjunan, K.; Montesano, D.; Lorenzo, J.M.; Putnik, P. Innovative technologies for the recovery of phytochemicals from Stevia rebaudiana Bertoni leaves: A review. Food Chem. 2018, 268, 513–521. [Google Scholar] [CrossRef] [PubMed]
- Kovačević, D.B.; Barba, F.J.; Granato, D.; Galanakis, C.M.; Herceg, Z.; Dragović-Uzelac, V.; Putnik, P. Pressurized hot water extraction (PHWE) for the green recovery of bioactive compounds and steviol glycosides from Stevia rebaudiana Bertoni leaves. Food Chem. 2018, 254, 150–157. [Google Scholar] [CrossRef] [PubMed]
- Hatami, T.; Johner, J.C.F.; Zabot, G.L.; Meireles, M.A.A. Supercritical fluid extraction assisted by cold pressing from clove buds: Extraction performance, volatile oil composition, and economic evaluation. J. Supercrit. Fluids 2018, 144, 39–47. [Google Scholar] [CrossRef]
- Nastić, N.; Švarc-Gajić, J.; Delerue-Matos, C.; Barroso, M.F.; Soares, C.; Moreira, M.M.; Morais, S.; Maskovic, P.; Srcek, V.G.; Slivac, I.; et al. Subcritical water extraction as an environmentally-friendly technique to recover bioactive compounds from traditional Serbian medicinal plants. Ind. Crops Prod. 2018, 111, 579–589. [Google Scholar] [CrossRef]
- El Kantar, S.; Boussetta, N.; Lebovka, N.; Foucart, F.; Rajha, H.N.; Maroun, R.G.; Louka, N.; Vorobiev, E. Pulsed electric field treatment of citrus fruits: Improvement of juice and polyphenols extraction. Innov. Food Sci. Emerg. Technol. 2018, 46, 153–161. [Google Scholar] [CrossRef]
- Barba, F.J.; Brianceau, S.; Turk, M.; Boussetta, N.; Vorobiev, N. Effect of Alternative Physical Treatments (Ultrasounds, Pulsed Electric Fields, and High-Voltage Electrical Discharges) on Selective Recovery of Bio-compounds from Fermented Grape Pomace. Food Bioprocess. Technol. 2015, 8, 1139–1148. [Google Scholar] [CrossRef]
- Zhou, Y.H.; Vidyarthi, S.K.; Zhong, C.S.; Zheng, Z.A.; An, Y.; Wang, J.; Wei, Q.; Xiao, H.W. Cold plasma enhances drying and color, rehydration ratio and polyphenols of wolfberry via microstructure and ultrastructure alteration. LWT 2020, 134, 110173. [Google Scholar] [CrossRef]
- Naviglio, D. Naviglio’s principle and presentation of an innovative solid–liquid extraction technology: Extractor Naviglio®. Anal. Lett. 2003, 36, 1647–1659. [Google Scholar] [CrossRef]
- Rocchetti, G.; Blasi, F.; Montesano, D.; Ghisoni, S.; Marcotullio, M.C.; Sabatini, S.; Cossignani, L.; Lucini, L. Impact of conventional/non-conventional extraction methods on the untargeted phenolic profile of Moringa oleifera leaves. Food Res. Int. 2019, 115, 319–327. [Google Scholar] [CrossRef] [PubMed]
- Duan, L.; Zhang, W.H.; Zhang, Z.H.; Liu, E.H.; Guo, L. Evaluation of natural deep eutectic solvents for the extraction of bioactive flavone C-glycosides from Flos Trollii. Microchem. J. 2019, 145, 180–186. [Google Scholar] [CrossRef]
- Shikov, A.N.; Kosman, V.M.; Flissyuk, E.V.; Smekhova, I.E.; Elameen, A.; Pozharitskaya, O.N. Natural Deep Eutectic Solvents for the Extraction of Phenyletanes and Phenylpropanoids of Rhodiola rosea L. Molecules 2020, 25, 1826. [Google Scholar] [CrossRef] [PubMed]
- Fernández, K.; Vega, M.; Aspé, E. An enzymatic extraction of proanthocyanidins from País grape seeds and skins. Food Chem. 2015, 168, 7–13. [Google Scholar] [CrossRef]
- Nadar, S.S.; Rao, P.; Rathod, V.K. Enzyme assisted extraction of biomolecules as an approach to novel extraction technology: A review. Food Res. Int. 2017, 108, 308–330. [Google Scholar] [CrossRef]
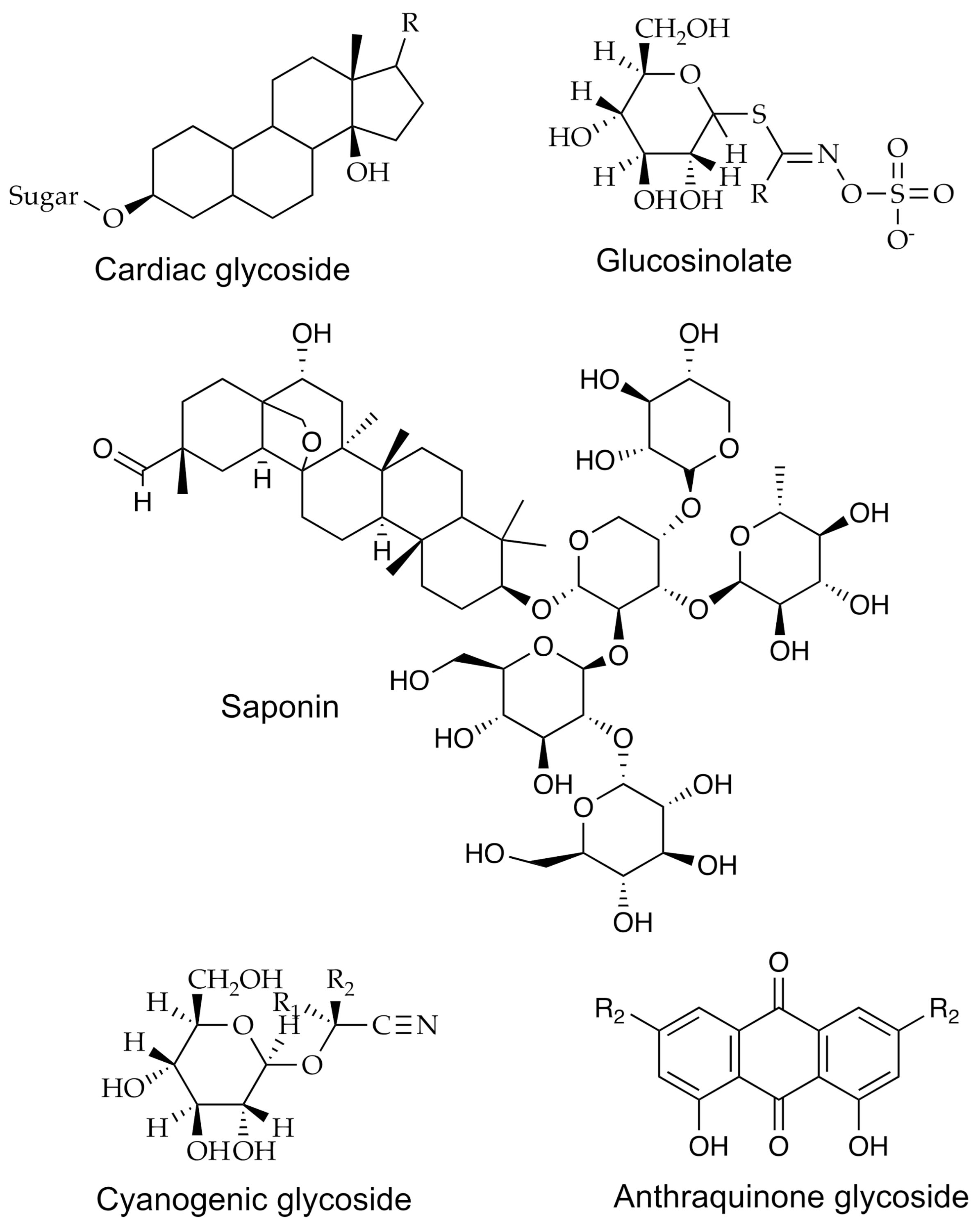
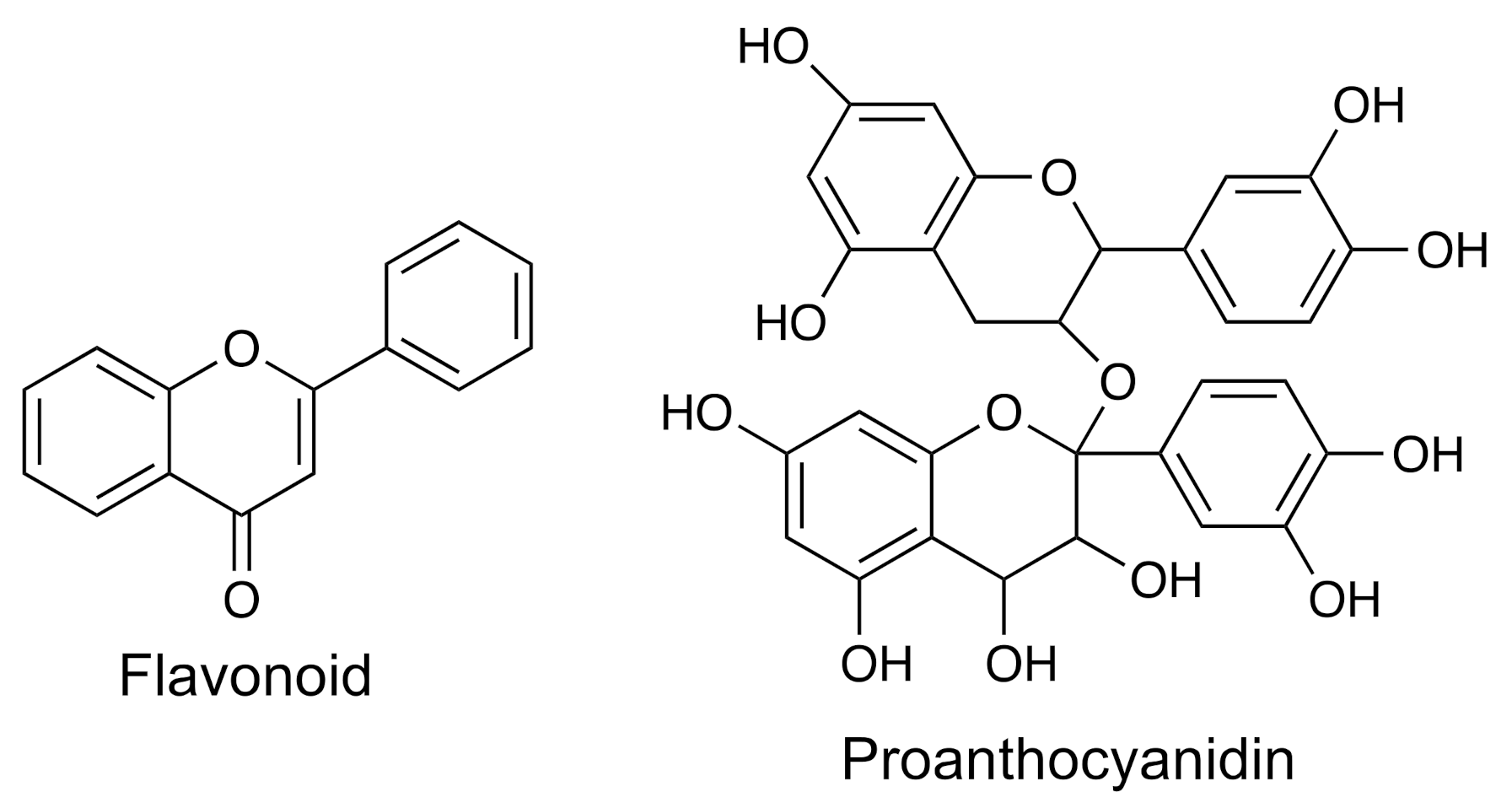


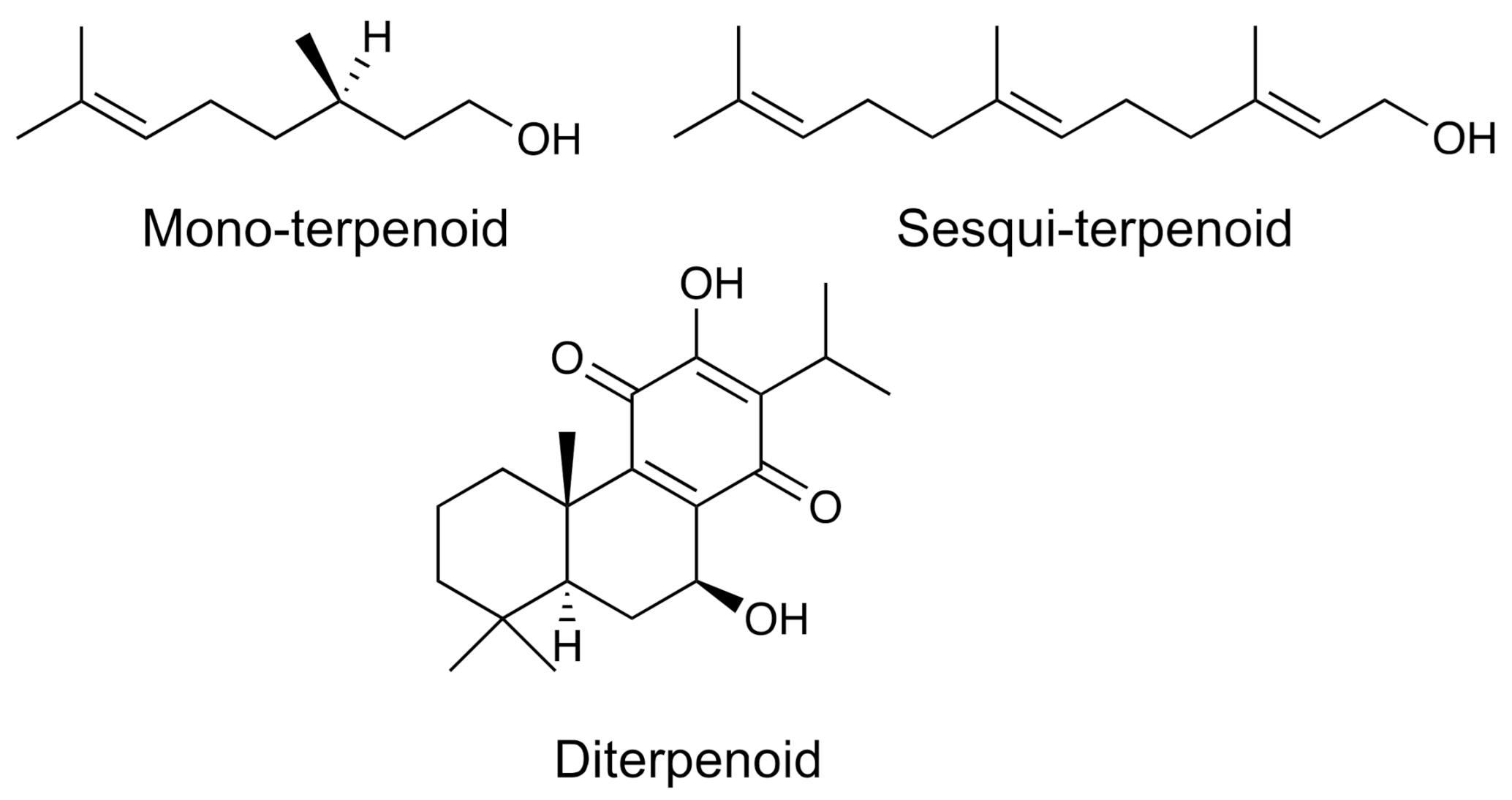

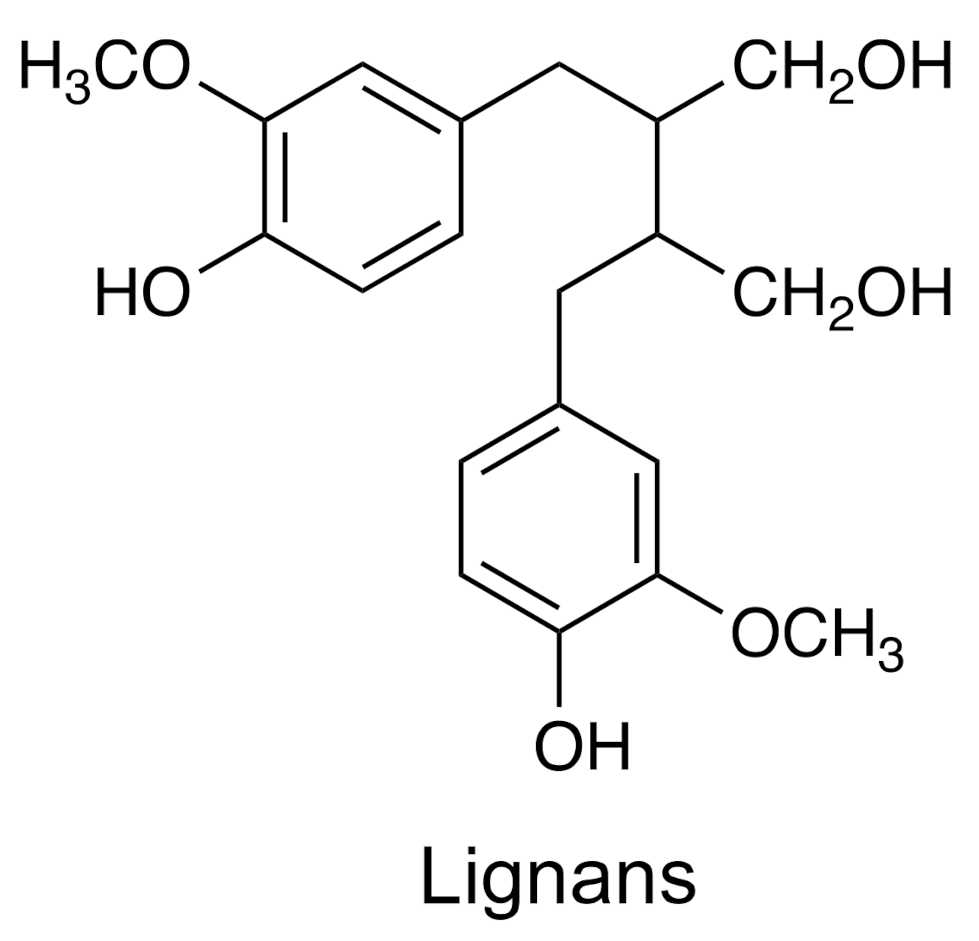
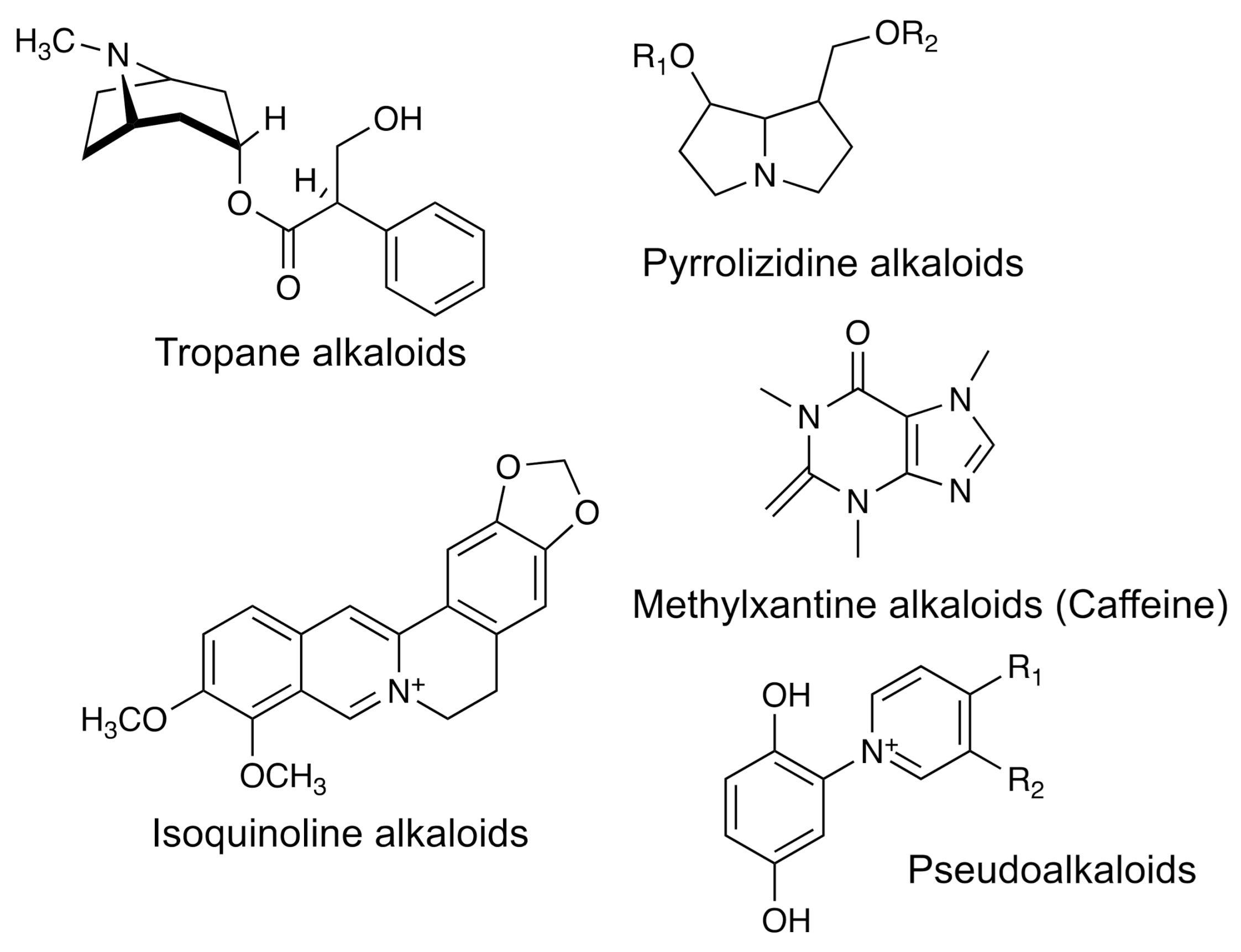
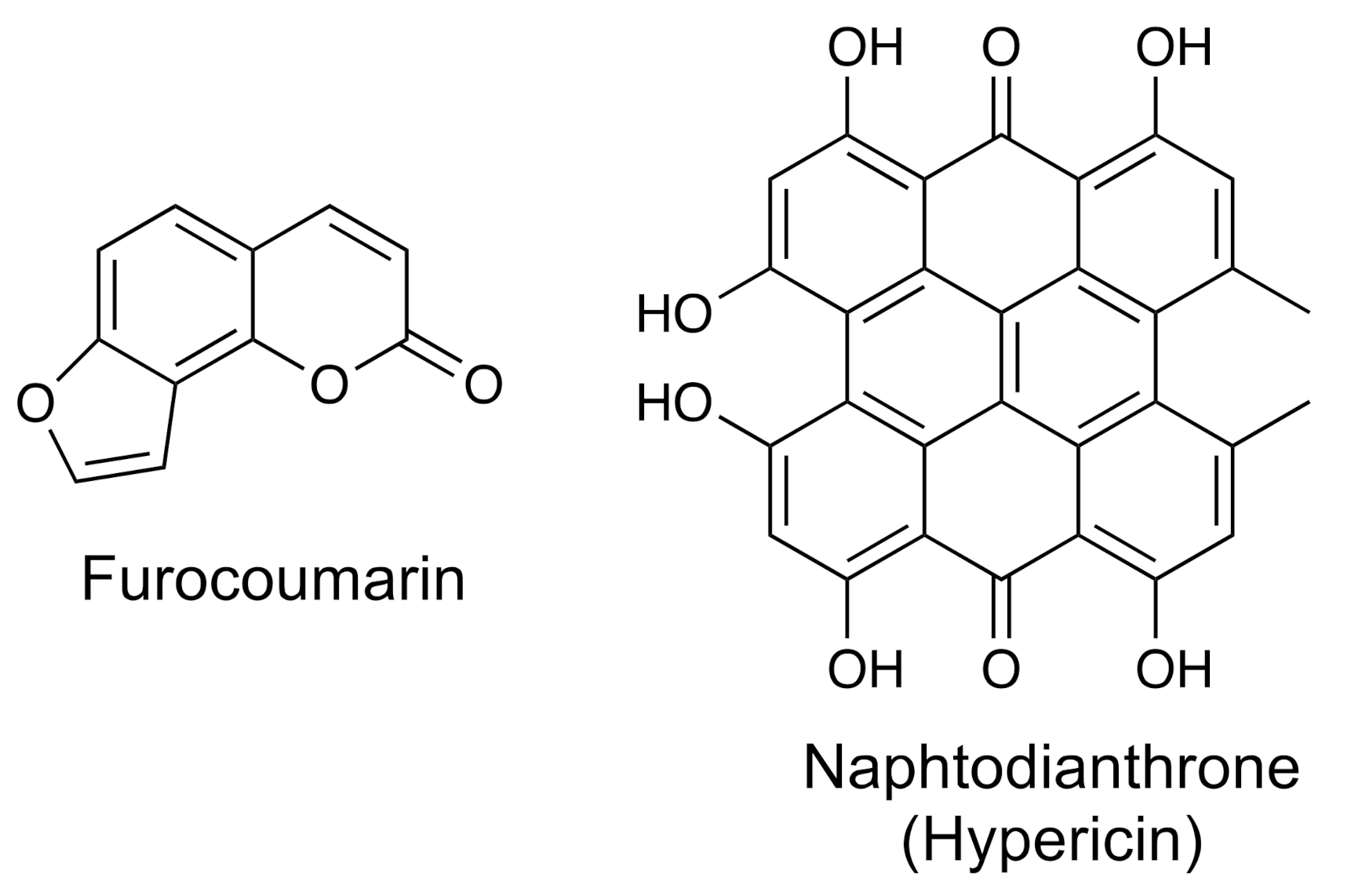
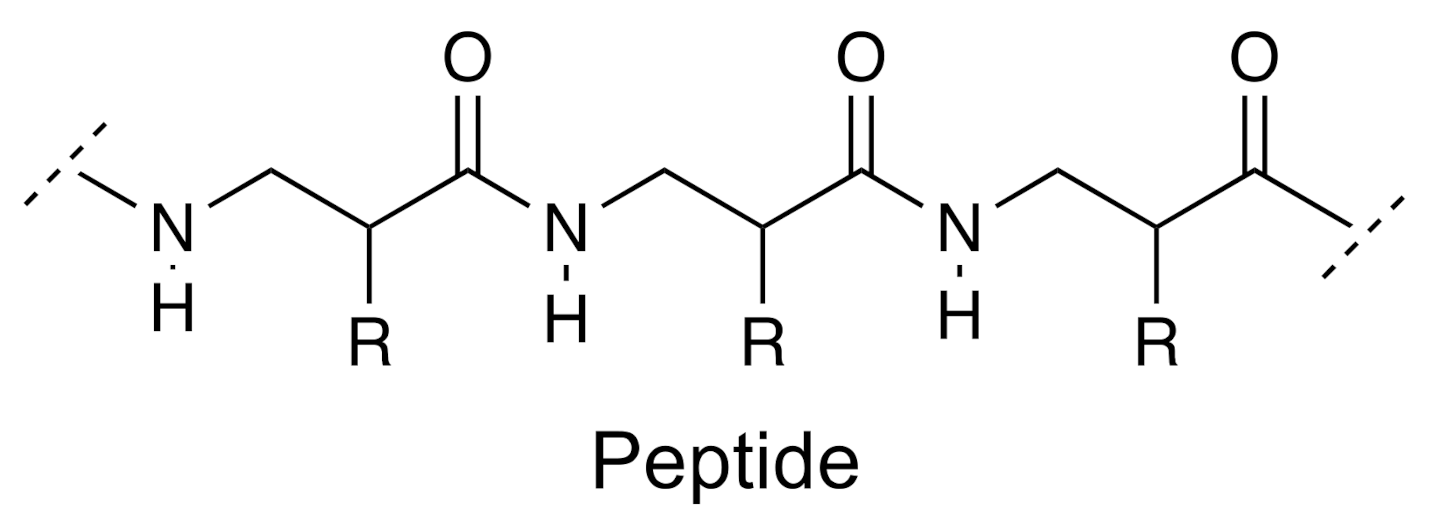
| Group | Example | Source | Activity | References | Methods |
|---|---|---|---|---|---|
| Glycosides | Cardenolide | Nerium oleander | Anti-inflammatory, anticancer | [11] | IR, UV–vis; HPLC-UV–vis NMR |
| Flavonoids | Silibinin | Silybum marianum | Anti-inflammatory, anticancer | [12,13,14,15] | IR; HPLC-UV–vis; LC-MS |
| Proanthocyanidins | A- and B-type | Cranberry, bilberry, curry, cinnamon, tea | Anti-inflammatory, anticancer, anti-microbial, hypolipidemic | [16,17] | IR; NMR; FT-ICR-ESI-MS-MS; UPLC-IM-HR-MS |
| Stilbenoids | Resveratrol | Grape, apple, berries, pistachios, peanuts | Anti-inflammatory, anticancer; anti-microbial, anti-diabetic | [18,19] | HPLC-UV–vis; UPLC-MS-MS; NMR |
| Tannins | Ellagitannins | Sorghum, apple, grape | Anti-inflammatory, anticancer, antimicrobial, antiviral | [20,21] | HPLC-UV–vis; NMR; HPLC ESI-MS-MS |
| Monoterpenoids | Limonene | Quercus ilex | Anti-inflammatory, anticancer | [22,23,24] | FGC-PTR-MS; NMR |
| Sesquiterpenoids | Zerumbone | Zingiber zerumbet | Anti-inflammatory, anticancer, antimicrobial, antipyretic | [25,26] | GC-MS; IR, NMR; ESI-MS |
| Phenylpropanoids | Coumarins | Artemisa annua | Anti-inflammatory, anticancer, antimicrobial, antiviral, antiparasitic | [27,28] | HPLC-UV–vis; LC-ESI-QTOF-MS-MS |
| Diterpenoids | Columbin | Jateorhiza columba | Analgesic, antipyretic | [29,30] | NMR; LC-MS; UPLC-HR-MS |
| Tetraterpenoids (Carotenoids) | α- and β-carotene | Carrot, cantaloupe, tomato | Antioxidant, chemopreventive | [31,32] | HPLC-UV–vis; HPLC-ESI-MS-MS; GC-MS |
| Resins | Stytonkinol A and B | Styrax benzoin | Antimicrobial, antifungal | [33,34] | IR; UV–vis; NMR |
| Lignans | Dibenzylbutane | Phyllanthus niruri | Antiviral, antioxidant | [35,36] | HPLC–DAD–ESI–QTOF–MS; NMR |
| Alkaloids | Triangularine | Asteraceae family | Anti-inflammatory, antioxidant, antimicrobial, anesthetic | [37,38] | HPLC-UV–vis: LC-MS-MS; NMR |
| Furocoumarines | Bergamottin | Grapefruit, orange | Anticancer | [39,40] | UV–vis; UPLC-MS-MS |
| Naphthodianthrones | Hypericin | Hypericum triquetrifolium | Anti-inflammatory, anticancer, antimicrobial, antiviral, antidepressant | [41,42] | UPLC-ESI-MS-MS |
| Peptides | Sesquin | Vigna sesquipedalis | Anticancer | [43,44] | IR; MALDESI-MS-MS |
| Proteins | Lectins | Ricinus communis | Antimicrobial | [45,46] | LC-QTOF-MS-MS |
| Method of Extraction and Fundamentals | Bioactive Compound | Plant Source | Reference |
|---|---|---|---|
| Maceration extraction (ME): | Flavonoid glycoside | Ginkgo biloba | [132,134,135] |
| The plant material is soaked with solvent in a container and left to stand for a minimum of three days with agitation. The cell wall softens, releasing the desired material. | |||
| Soxhlet extraction (SE): | Cafestol and kahweol (diterpenes) | Coffea arabica | [136,137] |
| The sample is put on a porous filter inside a Soxhlet apparatus chamber. The solvent is heated reaching its boiling point and passes through the matrix extracting the desired compound. The solvent is condensed reaching a volume allowing a siphon effect, thus continuously using the solvent for the extraction. | |||
| Ultrasound-assisted extraction (UAE): | Resveratrol (flavonoid) | Polygonum cuspidatum | [138,139] |
| Waves of high frequencies and longer wavelengths propagating through the medium, causing compression and rarefaction. Bubbles generated are unstable with contracts, collapsing, thus causing cavitation effect that cause cell disruption and improved solvent penetration to the matrix. | |||
| Microwave-assisted extraction (MAE): | Berberine and palmatine (alkaloids) | Berberis jaeschkeana | [134,139] |
| Uses microwave energies that interact with dipoles of polar solvents and samples causing heat and allowing the solvent penetration to the matrix. For nonpolar solvents, heating is poor and there is a transfer of the energy by dielectric absorbing. | |||
| High-pressure-assisted extraction (HPAE): | Condensed tannin | Stevia rebaudiana | [136,140] |
| High-applied pressure led to increased solubility and produces an increase in permeability. Solvent penetration into the cell is promoted and more target compounds can permeate the cell membrane, facilitating the extraction process. | |||
| Supercritical fluid extraction (SFE): | Euglenol, beta-caryophyllene, alpha-humulene (terpenoids) | Clove | [132,140,141,142] |
| Uses mainly, CO2 as the solvent, which is fluid at 31 °C and 74 bar, dissolving CO2 soluble compounds from plants. A variant is the use of water (SWE) at 100–374 °C and high pressures to maintain water liquid, allowing to extract polar constituents. | |||
| Pulsed electric field extraction (PEFE) and high voltage electrical discharge extraction (HVED): | Carotenoids and flavonoids | Citrus fruits | [139,143,144] |
| Pulses of electricity under high-intensity electric fields are applied to the samples. The cell membranes generate pores that allow the release of intracellular components to the media. | |||
| Cold plasma extraction (CPE): | Polyphenols | Wolfberry | [139,144,145] |
| Cold plasma causes cell wall damage, an effect that results in the release of active molecules from the cell. | |||
| Rapid solid–liquid dynamic extraction (RSLDE): | Phenolic acids | Moringa oleifera | [139,146,147] |
| Uses a solid–liquid extractor working at 8–9 bar. Requires water as a solvent and is based on the principle of pressure/depression. | |||
| Eutectic solvent extraction (ESE): | Rosin (phenylpropanoid) | Rhodiola rosea L. | [133,148,149] |
| Uses environmentally friendly solvents for the extraction of bioactive compounds from plants. This kind of solvents is prepared when two solid compounds are mixed under certain conditions to diminish their melting points to a point low to form a eutectic mixture with specific physicochemical characteristics. | |||
| Enzyme-assisted extraction (EAE): | |||
| Pretreatment for the extraction that uses enzymes with specific activities, promoting disruption of the cell wall of the plant material to enhance or facilitate the solvent extraction of the biologically active compound. | Proanthocyanidins | Grapes | [150,151] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lozada-Ramírez, J.D.; Ortega-Regules, A.E.; Hernández, L.R.; Anaya de Parrodi, C. Spectroscopic and Spectrometric Applications for the Identification of Bioactive Compounds from Vegetal Extracts. Appl. Sci. 2021, 11, 3039. https://doi.org/10.3390/app11073039
Lozada-Ramírez JD, Ortega-Regules AE, Hernández LR, Anaya de Parrodi C. Spectroscopic and Spectrometric Applications for the Identification of Bioactive Compounds from Vegetal Extracts. Applied Sciences. 2021; 11(7):3039. https://doi.org/10.3390/app11073039
Chicago/Turabian StyleLozada-Ramírez, José Daniel, Ana E. Ortega-Regules, Luis Ricardo Hernández, and Cecilia Anaya de Parrodi. 2021. "Spectroscopic and Spectrometric Applications for the Identification of Bioactive Compounds from Vegetal Extracts" Applied Sciences 11, no. 7: 3039. https://doi.org/10.3390/app11073039
APA StyleLozada-Ramírez, J. D., Ortega-Regules, A. E., Hernández, L. R., & Anaya de Parrodi, C. (2021). Spectroscopic and Spectrometric Applications for the Identification of Bioactive Compounds from Vegetal Extracts. Applied Sciences, 11(7), 3039. https://doi.org/10.3390/app11073039







